Parametric Finite Element Evaluation of Load Redistribution Under Progressive Lumbar Disc Degeneration
Abstract
1. Introduction
2. Materials and Methods
2.1. Initial Data and Numerical Model Development
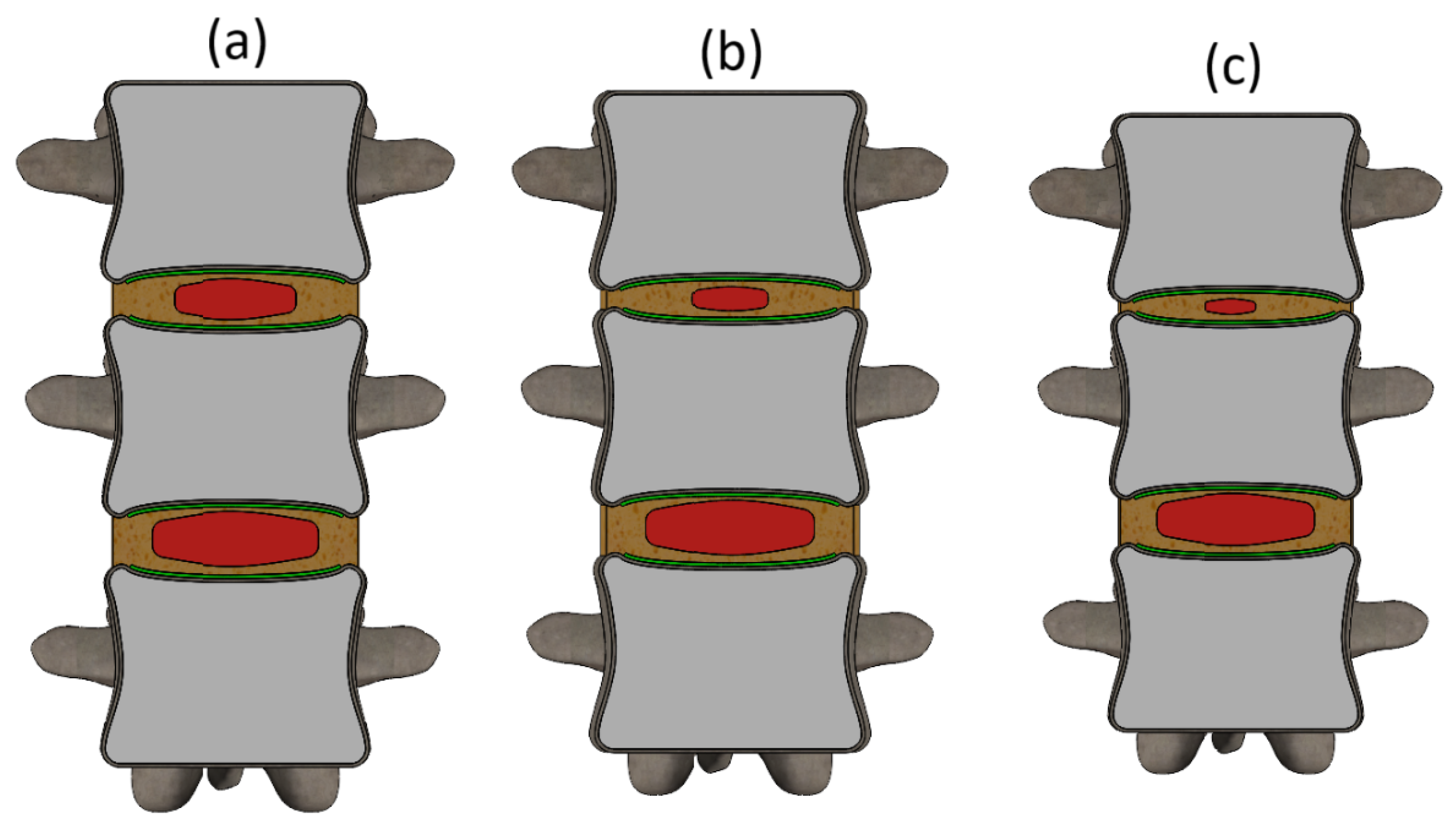
2.2. Modelling the Geometrical Impact of Degenerative Changes on IVD
2.3. Mechanical Properties of Model Components
2.4. Problem Formulation
2.5. Boundary Conditions and Finite Element Mesh
2.6. Modeling Assumptions and Their Implications for Interpretation
3. Results and Discussion
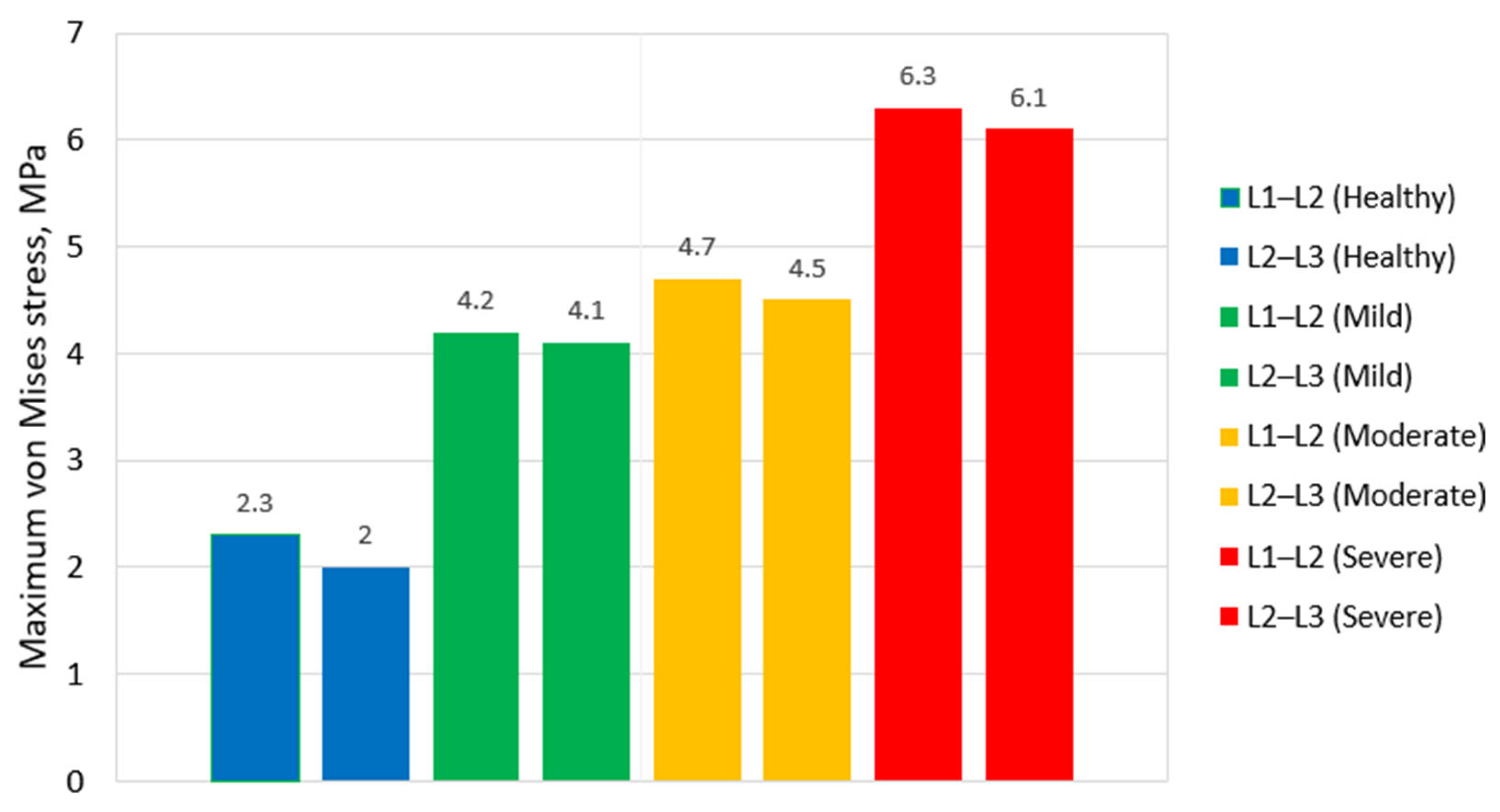
3.1. Stress Analysis Across Degeneration Stages in the L1–L3 Segment
3.2. Analysis of Reaction Forces Under Different Degrees of Disc Degeneration
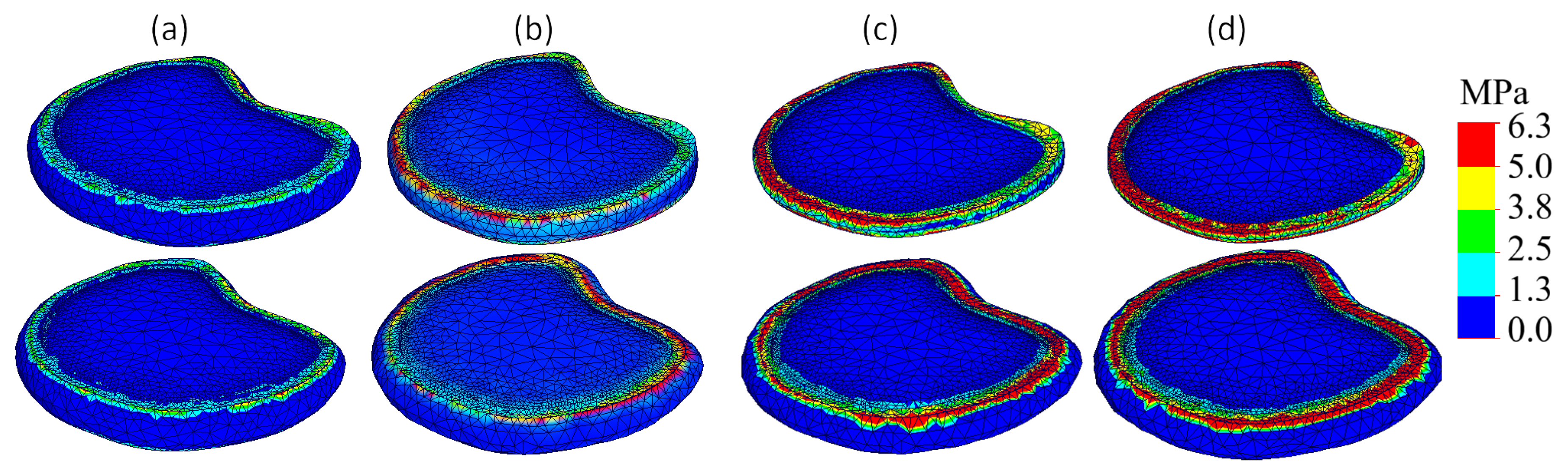
3.3. Stress Patterns in the Intervertebral Disc Across Degeneration Stages
3.4. Results Verification and Discussion
3.5. Sensitivity Analysis
- Disc height: ±10% from baseline (healthy: 10 mm → 9 mm/11 mm; severe: 4 mm → 3.6 mm/4.4 mm).
- Nucleus pulposus volume (cross-sectional area): ±15% from baseline.
- Annulus fibrosus stiffness (Mooney–Rivlin constants C1 and C2): ±20% from baseline values.
- The model is most sensitive to disc height variations: a 10% reduction increases peak AF stress by 13–18% and total reaction force by 15–23%, while a 10% increase reduces these values accordingly. This confirms disc height as one of the dominant geometric factors influencing segmental stiffness and annulus loading.
- Nucleus pulposus volume has a moderate effect, primarily on NP stress (changes up to ±13–15%), with minimal impact on AF stress (±3–4%) and reaction force (±8–11%).
- Annulus fibrosus stiffness (C1 and C2) exerts the strongest influence on peak AF stress (up to ±26%) and reaction force (±13–19%), but has negligible effect on NP stress (±2–6%).
- Importantly, all qualitative trends remain consistent across the tested ranges: progressive degeneration still leads to increased AF stress, decreased NP stress, and a shift of load toward posterior elements. No reversal of trends was observed.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Oktay, A.B.; Albayrak, N.B.; Akgul, Y.S. Computer Aided Diagnosis of Degenerative Intervertebral Disc Diseases from Lumbar MR Images. Comput. Med. Imaging Graph. 2014, 38, 613–619. [Google Scholar] [CrossRef]
- Galbusera, F.; Van Rijsbergen, M.; Ito, K.; Huyghe, J.M.; Brayda-Bruno, M.; Wilke, H.J. Ageing and Degenerative Changes of the Intervertebral Disc and Their Impact on Spinal Flexibility Ageing and Degenerative Changes of the Intervertebral Disc and Their Impact on Spinal Flexibility. Eur. Spine J. 2014, 23, 324–332. [Google Scholar] [CrossRef]
- Liu, Y.; Dou, Y.; Sun, X.; Yang, Q. Mechanisms and Therapeutic Strategies for Senescence-Associated Secretory Phenotype in the Intervertebral Disc Degeneration Microenvironment. J. Orthop. Transl. 2024, 45, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Y.; Han, Y. Comparison of the Biomechanical Effects of Lumbar Disc Degeneration on Normal Patients and Osteoporotic Patients: A Finite Element Analysis. Med. Eng. Phys. 2023, 112, 103952. [Google Scholar] [CrossRef] [PubMed]
- Griffith, J.F.; Wang, Y.-X.J.; Antonio, G.E.; Choi, K.C.; Yu, A.; Ahuja, A.T.; Leung, P.C. Modified Pfirrmann Grading System for Lumbar Intervertebral Disc Degeneration. Spine 2007, 32, E708–E712. [Google Scholar] [CrossRef] [PubMed]
- Salamat, S.; Hutchings, J.; Kwong, C.; Magnussen, J.; Hancock, M.J. The Relationship between Quantitative Measures of Disc Height and Disc Signal Intensity with Pfirrmann Score of Disc Degeneration. Springerplus 2016, 5, 829. [Google Scholar] [CrossRef]
- Teichtahl, A.J.; Urquhart, D.M.; Wang, Y.; Wluka, A.E.; Heritier, S.; Cicuttini, F.M. A Dose-Response Relationship between Severity of Disc Degeneration and Intervertebral Disc Height in the Lumbosacral Spine. Arthritis Res. Ther. 2015, 17, 297. [Google Scholar] [CrossRef]
- Jo, M.; Chae, S.W. Stress Analysis of Intervertebral Disc during Occupational Activities. Comput. Methods Programs Biomed. 2021, 208, 106298. [Google Scholar] [CrossRef]
- Ruiz, C.; Noailly, J.; Lacroix, D. Material Property Discontinuities in Intervertebral Disc Porohyperelastic Finite Element Models Generate Numerical Instabilities Due to Volumetric Strain Variations. J. Mech. Behav. Biomed. Mater. 2013, 26, 1–10. [Google Scholar] [CrossRef]
- Zhu, Q.; Gao, X.; Chen, S.; Gu, W.; Brown, M.D. Effect of Intervertebral Disc Degeneration on Mechanical and Electric Signals at the Interface between Disc and Vertebra. J. Biomech. 2020, 104, 109756. [Google Scholar] [CrossRef]
- Holzapfel, G.A.; Ogden, R.W. Biomechanical Stresses in a Residually Stressed Idealized Intervertebral Disc. Int. J. Non-Linear Mech. 2024, 161, 104687. [Google Scholar] [CrossRef]
- Ghezelbash, F.; Eskandari, A.H.; Shirazi-Adl, A.; Kazempour, M.; Tavakoli, J.; Baghani, M.; Costi, J.J. Modeling of Human Intervertebral Disc Annulus Fibrosus with Complex Multi-Fiber Networks. Acta Biomater. 2021, 123, 208–221. [Google Scholar] [CrossRef] [PubMed]
- Kandil, K.; Zaïri, F.; Messager, T.; Zaïri, F. A Microstructure-Based Model for a Full Lamellar-Interlamellar Displacement and Shear Strain Mapping inside Human Intervertebral Disc Core. Comput. Biol. Med. 2021, 135, 104629. [Google Scholar] [CrossRef]
- Xi, Z.; Xie, Y.; Sun, S.; Wang, N.; Chen, S.; Wang, G.; Li, J. IVD Fibrosis and Disc Collapse Comprehensively Aggravate Vertebral Body Disuse Osteoporosis and Zygapophyseal Joint Osteoarthritis by Posteriorly Shifting the Load Transmission Pattern. Comput. Biol. Med. 2024, 170, 108019. [Google Scholar] [CrossRef] [PubMed]
- 3D Slicer Image Computing Platform. Available online: https://www.slicer.org/ (accessed on 23 March 2023).
- MESHLAB. Available online: https://www.meshlab.net/ (accessed on 23 March 2023).
- SOLIDWORKS Systemes. Available online: https://help.solidworks.com/ (accessed on 23 March 2023).
- Kim, Y.H.; Wu, M.; Kim, K. Stress Analysis of Osteoporotic Lumbar Vertebra Using Finite Element Model with Microscaled Beam-Shell Trabecular-Cortical Structure. J. Appl. Math. 2013, 2013, 285165. [Google Scholar] [CrossRef]
- Cai, X.; Sun, M.; Huang, Y.; Liu, Z.; Liu, C.; Du, C.; Yang, Q. Biomechanical Effect of L4–L5 Intervertebral Disc Degeneration on the Lower Lumbar Spine: A Finite Element Study. Orthop. Surg. 2020, 12, 917–930. [Google Scholar] [CrossRef]
- McDonald, K.; Little, J.; Pearcy, M.; Adam, C. Development of a Multi-Scale Finite Element Model of the Osteoporotic Lumbar Vertebral Body for the Investigation of Apparent Level Vertebra Mechanics and Micro-Level Trabecular Mechanics. Med. Eng. Phys. 2010, 32, 653–661. [Google Scholar] [CrossRef]
- Jaramillo, H.E. Evaluation of the Use of the Yeoh and Mooney-Rivlin Functions as Strain Energy Density Functions for the Ground Substance Material of the Annulus Fibrosus. Math. Probl. Eng. 2018, 2018, 1570142. [Google Scholar] [CrossRef]
- Gruber, G.; Nicolini, L.F.; Ribeiro, M.; Lerchl, T.; Wilke, H.; Jaramillo, H.E.; Senner, V.; Kirschke, J.S.; Nispel, K. Comparative FEM Study on Intervertebral Disc Modeling: Holzapfel-Gasser-Ogden vs. Structural Rebars. Front. Bioeng. Biotechnol. 2024, 12, 1391957. [Google Scholar] [CrossRef]
- Xie, F.; Zhou, H.; Zhao, W.; Huang, L. A Comparative Study on the Mechanical Behavior of Intervertebral Disc Using Hyperelastic Finite Element Model. Technol. Health Care 2017, 25, 177–187. [Google Scholar] [CrossRef]
- Mimura, M.; Panjabi, M.; Oxland, T.; Crisco, J.; Yamamoto, I.; Vasavada, A. Disc Degeneration Affects the Multidirectional Flexibility of the Lumbar Spine. Spine 1994, 19, 1371–1380. [Google Scholar] [CrossRef] [PubMed]
- Zirbel, S.; Stolworthy, D.; Howell, L.; Bowden, A. Intervertebral Disc Degeneration Alters Lumbar Spine Segmental Stiffness in All Modes of Loading under a Compressive Follower Load. Spine J. 2013, 13, 1134–1147. [Google Scholar] [CrossRef] [PubMed]
- Ruberté, L.M.; Natarajan, R.N.; Andersson, G.B. Influence of Single-Level Lumbar Degenerative Disc Disease on the Behavior of the Adjacent Segments—A Finite Element Model Study. J. Biomech. 2009, 42, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Galbusera, F.; Schmidt, H.; Neidlinger-Wilke, C.; Gottschalk, A.; Wilke, H.J. The Mechanical Response of the Lumbar Spine to Different Combinations of Disc Degenerative Changes Investigated Using Randomized Poroelastic Finite Element Models. Eur. Spine J. 2011, 20, 563–571. [Google Scholar] [CrossRef]
- Schmidt, H.; Kettler, A.; Rohlmann, A.; Claes, L.; Wilke, H.J. The Risk of Disc Prolapses with Complex Loading in Different Degrees of Disc Degeneration—A Finite Element Analysis. Clin. Biomech. 2007, 22, 988–998. [Google Scholar] [CrossRef]
- Poojara, V.; Thaker, M.; Trivedi, R.; Modi, B.; Patel, R. Three Dimensional Analysis of Mechanical Behavior of Lumbar Vertebra under Impact Load and Its Biomechanical Importance. Mater. Today Proc. 2020, 38, 402–406. [Google Scholar] [CrossRef]
- Kurutz, M. Finite Element Modelling of Human Lumbar Spine. In Finite Element Analysis; IntechOpen: London, UK, 2010; ISBN 978-953-307-123-7. [Google Scholar]
- Ma, J.; Ding, Y.; Rozi, R.; Han, J.; Jiang, Q. From Single Ligament to Multi-Ligament Injury: A Finite Element Study on the Contribution of the Posterior Ligamentous Complex to Segmental Stability and Intervertebral Disc Stress Distribution. BMC Musculoskelet. Disord. 2025, 26, 820. [Google Scholar] [CrossRef]
- Zhang, Q.; Jin, Y.; He, L.; Zhang, K.; Chen, L.; Chen, W.; Feng, H. Biomechanical Effect of Cage Size in Single—Level Anterior Cervical Discectomy and Fusion: A Finite Element Analysis. BMC Musculoskelet. Disord. 2025, 26, 594. [Google Scholar] [CrossRef]
- Wang, B.; Liu, Y.; Wang, Z.; Niu, C.; Tang, J.; Sun, L. Osteoporosis in Adjacent Cervical Segments Exacerbates Disc Herniation. Sci. Rep. 2025, 15, 22901. [Google Scholar] [CrossRef]
- Wan, C.; Shen, X.; Wu, X.; Yu, C.; Shao, Y.; Zhang, R.; Shang, J.; Li, J.; Zhang, Y.; Li, Y. Assessing the Biomechanics of Scheuermann’ s Kyphosis Affected Thoracolumbar Spine in Forward Flexion at the Tissue-Level Using a Finite Element Model. Sci. Rep. 2025, 15, 27408. [Google Scholar] [CrossRef]
- Flor, O.; Rubio, J.E.; Tong, J.; Sundaramurthy, A.; Pant, A.; Nagaraja, S.; Owen, M.K.; Samaan, M.A.; Noehren, B.; Reifman, J. Effect of Walking with an Active Ankle Exoskeleton on the Biomechanical Responses of the Lumbar Spine. Front. Bioeng. Biotechnol. 2025, 13, 1654585. [Google Scholar] [CrossRef] [PubMed]
- Mu, X.; Wei, X.; Nong, J.; Ye, H.; Li, Z.; Wei, M.; Wei, J. Clinical Evaluation and Fi Nite Element Analysis of Bone Cement-Augmented Anterolateral Screw Fi Xation versus Percutaneous Bilateral Pedicle Screw Fi Xation Co-Applied with Oblique Lumbar Interbody Fusion for Single-Level Lumbar Degenerative Diseases with Osteoporosis. Front. Bioeng. Biotechnol. 2025, 13, 1571849. [Google Scholar] [CrossRef]
- Park, W.M.; Kim, Y.H.; Lee, S. Effect of Intervertebral Disc Degeneration on Biomechanical Behaviors of a Lumbar Motion Segment under Physiological Loading Conditions. J. Mech. Sci. Technol. 2013, 27, 483–489. [Google Scholar] [CrossRef]
- Inoue, N.; Orías, A.A.E.; Segami, K. Biomechanics of the Lumbar Facet Joint. Spine Surg. Relat. Res. 2020, 4, 1–7. [Google Scholar] [CrossRef]
- Park, J.S.; Goh, T.S.; Lee, J.S.; Lee, C. Analyzing Isolated Degeneration of Lumbar Facet Joints: Implications for Degenerative Instability and Lumbar Biomechanics Using Fi Nite Element Analysis. Front. Bioeng. Biotechnol. 2024, 12, 1294658. [Google Scholar] [CrossRef]
- Chabarova, O.; Selivonec, J. Virtual Modelling the Impact of Torsional Loading on Osteoporotic Vertebrae Buckling. Acta Bioeng. Biomech. 2024, 26, 13–22. [Google Scholar] [CrossRef]
- Wilke, H.J.; Neef, P.; Caimi, M.; Hoogland, T.; Claes, L.E. New in Vivo Measurements of Pressures in the Intervertebral Disc in Daily Life. Spine 1999, 24, 755–762. [Google Scholar] [CrossRef]
- Sato, K.; Kikuchi, S.; Yonezawa, T. In Vivo Intradiscal Pressure Measurement in Healthy Individuals and in Patients with Ongoing Back Problems. Spine 1999, 24, 2468–2474. [Google Scholar] [CrossRef]
- Schmidt, H.; Heuer, F.; Drumm, J.; Klezl, Z.; Claes, L.; Wilke, H.-J. Application of a Calibration Method Provides More Realistic Results for a Finite Element Model of a Lumbar Spinal Segment. Clin. Biomech. 2007, 22, 377–384. [Google Scholar] [CrossRef]
- Newell, N.; Little, J.P.; Christou, A.; Adams, M.A.; Adam, C.J.; Masouros, S.D. Biomechanics of the Human Intervertebral Disc: A Review of Testing Techniques and Results. J. Mech. Behav. Biomed. Mater. 2017, 69, 420–434. [Google Scholar] [CrossRef]
- Fujiwara, A.; Tamai, K.; An, H.S.; Kurihashi, T.; Lim, T.H.; Yoshida, H.; Saotome, K. The Relationship between Disc Degeneration, Facet Joint Osteoarthritis, and Stability of the Degenerative Lumbar Spine. J. Spinal Disord. 2000, 13, 444–450. [Google Scholar] [CrossRef]
- Dreischarf, M.; Shirazi-Adl, A.; Arjmand, N.; Rohlmann, A.; Schmidt, H. Estimation of Loads on Human Lumbar Spine: A Review of in Vivo and Computational Model Studies. J. Biomech. 2016, 49, 833–845. [Google Scholar] [CrossRef]
- Heuer, F.; Schmidt, H.; Klezl, Z.; Claes, L.; Wilke, H.-J. Stepwise Reduction of Functional Spinal Structures Increase Range of Motion and Change Lordosis Angle. J. Biomech. 2007, 40, 271–280. [Google Scholar] [CrossRef]






| Model Component | Young’s Modulus, MPa | Poisson’s Ratio | Yield Strength, MPa |
|---|---|---|---|
| Cortical bone | 8000 | 0.3 | 64 |
| Cancellous Bone | 100 | 0.3 | |
| Posterior elements | 3500 | 0.3 | |
| Endplates | 24 | 0.4 |
| Degeneration Stage | C1, MPa (AF) | C2, MPa (AF) | C1, MPa (NP) | C2, MPa (NP) |
|---|---|---|---|---|
| Healthy | 0.18 | 0.045 | 0.12 | 0.030 |
| Mild | 0.40 | 0.100 | 0.14 | 0.035 |
| Moderate | 0.60 | 0.150 | 0.17 | 0.041 |
| Severe | 0.90 | 0.230 | 0.19 | 0.045 |
| Model | Number of Elements | Number of Nodes | Number of Degrees of Freedom | Element Size, mm | Skewness; Aspect Ratio | Number of Jacobian Points |
|---|---|---|---|---|---|---|
| Healthy | 576,345 | 704,576 | 2,111,946 | 0.0029–3 | <0.8; <3 | 16 |
| Mild | 559,054 | 683,457 | 2,009,363 | |||
| Moderate | 536,692 | 656,108 | 1,922,396 | |||
| Severe | 509,857 | 623,296 | 1,832,490 |
| Simplification | Justification | Implications for Interpretation |
|---|---|---|
| Absence of follower load and muscle forces | Avoids additional assumptions related to curvature-dependent load paths and subject-specific muscle activation, preserving a controlled parametric framework. | Load transmission occurs through simplified axial compression; deformation modes and reaction forces may differ from physiological loading scenarios, while comparative degeneration-driven trends remain interpretable. |
| No ligament structures | Avoids adding nonlinear, tension-only ligaments with subject-specific properties and attachment uncertainty, ensuring a controlled, reproducible comparison across degeneration stages under compression load. | The segmental mechanical response under compression primarily reflects disc- and bone-driven behavior. Ligament-mediated stabilization is not included, so results characterize internal load redistribution in the absence of soft-tissue tension contributions. |
| Flat endplates | Controls geometric variability and isolates the mechanical contribution of disc degeneration without additional curvature-related effects. | Flattened boundaries may over constrain the segment, restricting small, coupled motions under compression and thereby increasing apparent stiffness and potentially inflating absolute reaction forces. |
| Homogeneous cancellous bone representation | CT-based heterogeneity was not the focus; uniform cancellous properties improve numerical robustness and reduce computational cost. | Local stress heterogeneity within vertebral bodies is not resolved; the vertebral body response represents an effective, averaged mechanical behavior. |
| Isotropic hyperelastic annulus fibrosus | Prioritizes numerical stability and enables a consistent parametric degeneration framework; explicit fiber-level anisotropy was beyond the intended scope. | The annulus response represents bulk mechanical behavior; direction-dependent fiber effects are not explicitly captured, which may influence absolute stress magnitudes. |
| Fixed inferior constraint and rigid displacement loading | Ensures numerical stability and reproducible kinematic conditions across all degeneration stages, allowing controlled comparison of disc-driven mechanical effects. | The flattened external boundaries may increase apparent stiffness. However, relative mechanical trends across degeneration stages remain valid due to consistent application. |
| Single-sample geometry | Reduces inter-subject anatomical variability and isolates the mechanical effects of progressive disc degeneration, enabling a controlled comparison across degeneration stages without confounding geometric differences. | The results reflect degeneration-driven mechanical trends within a fixed anatomical configuration and may not capture population-level variability associated with subject-specific geometry; therefore, findings should be interpreted as mechanistic rather than statistical generalizations. |
| Parameter Varied | Variation | Configuration | % Change in Peak AF Stress (L1–L2) | % Change in Peak NP Stress (L1–L2) | % Change in Total Reaction Force |
|---|---|---|---|---|---|
| Disc height | +10% | Healthy | −14% | +9% | −16% |
| Disc height | –10% | +18% | −11% | +23% | |
| Disc height | +10% | Severe | −13% | +6% | −12% |
| Disc height | –10% | +16% | −8% | +15% | |
| Nucleus pulposus volume | +15% | Healthy | −3% | +13% | −8% |
| Nucleus pulposus volume | –15% | +4% | −14% | +11% | |
| AF stiffness (C1 and C2) | +20% | Healthy | +21% | −6% | +15% |
| AF stiffness (C1 and C2) | –20% | −17% | +5% | −13% | |
| AF stiffness (C1 and C2) | +20% | Severe | +26% | −4% | +19% |
| AF stiffness (C1 and C2) | –20% | −20% | +2% | −16% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ardatov, O.; Fernandes, S.R.; Kilikevičius, A.; Alekna, V. Parametric Finite Element Evaluation of Load Redistribution Under Progressive Lumbar Disc Degeneration. Bioengineering 2026, 13, 234. https://doi.org/10.3390/bioengineering13020234
Ardatov O, Fernandes SR, Kilikevičius A, Alekna V. Parametric Finite Element Evaluation of Load Redistribution Under Progressive Lumbar Disc Degeneration. Bioengineering. 2026; 13(2):234. https://doi.org/10.3390/bioengineering13020234
Chicago/Turabian StyleArdatov, Oleg, Sofia Rita Fernandes, Artūras Kilikevičius, and Vidmantas Alekna. 2026. "Parametric Finite Element Evaluation of Load Redistribution Under Progressive Lumbar Disc Degeneration" Bioengineering 13, no. 2: 234. https://doi.org/10.3390/bioengineering13020234
APA StyleArdatov, O., Fernandes, S. R., Kilikevičius, A., & Alekna, V. (2026). Parametric Finite Element Evaluation of Load Redistribution Under Progressive Lumbar Disc Degeneration. Bioengineering, 13(2), 234. https://doi.org/10.3390/bioengineering13020234







