Digital and Intelligent Rehabilitation Technologies in Stroke and Neurological Disorders: A Systematic Review of Artificial Intelligence, Virtual Reality, Gamification, and Emerging Therapeutic Platforms in Neurorehabilitation
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Protocol Registration
2.2. Search Strategy
2.3. Eligibility Criteria
2.4. Screening and Study Selection
2.5. Data Extraction and Classification
2.6. Risk of Bias Assessment
2.7. Data Synthesis Strategy
3. Results
| Author, Year | Study Design | Objective and Population | Intervention | Outcome Measures | Results | Conclusion | AI Method/ Gamification Feature |
|---|---|---|---|---|---|---|---|
| Rodríguez-Hernández et al., 2021 [34] | RCT (Clinical) | To assess the effect of VR-based therapy on upper-limb motor function in patients with strokes (N = 43) | Conventional therapy + VR exposure therapy vs. conventional therapy alone for 4 weeks | Fugl-Meyer Assessment-Upper Extremity (FMA-UE), Modified Ashworth Scale, Stroke Impact Scale 3.0 | Significant improvement in FMA-UE (~11-point increase), η2 = 0.633, p < 0.001 | VR enhances traditional therapy and demonstrates a large effect size | Immersive VR environment with real-time performance feedback and adaptation |
| Kang et al., 2023 [54] | Protocol (Planned RCT–Clinical) | To evaluate home-based VR exergame training for post-patients with strokes (N = 120 planned) | 8 weeks of home-based VR exergame vs. daily life activity (control) | Endurance, strength, ADLs, gait, QoL | Pending-protocol stage | Home-based VR is expected to improve community-level rehabilitation outcomes | Home VR exergaming with scoring and progressive difficulty (telerehabilitation) |
| House et al., 2016 [44] | Pilot Study (Clinical Feasibility) | To evaluate team-based gamified VR rehabilitation for chronic stroke in nursing homes (N = 23) | BrightArm Duo VR using robotic-enabled workstation; collaborative competition with remote participants | ROM (18/23 variables), task completion time, engagement, depressive symptoms | 18/23 ROM variables improved significantly (p = 0.01) | Team-based gamified VR is feasible and beneficial in nursing home settings | Robot-assisted VR; multiplayer collaboration/competition; score feedback |
| Zhou et al., 2022 [43] | Protocol (Robot-Assisted Clinical Trial-In Progress) | To assess NeuCir-VR combined with robotic lower-limb rehabilitation (N = 40 planned) | Robot assistance + NeuCir-VR vs. robot + standard VR, 5 sessions/week for 4 weeks | FMA-LE, Berg Balance Scale, fMRI, Modified Ashworth Scale | Pending | NeuCir-VR expected to promote neuroplasticity and balance recovery | Robotic assistance + neural-circuit VR training framework |
| Bai et al., 2022 [42] | RCT (AI-Integrated Clinical Intervention) | To compare an AI-enhanced VR rehabilitation system with medication-only controls in patients with strokes (N = 50) | AI-VR personalized adaptive therapy for 10 weeks | FMA-UE, FMA-LE, FTHUE-HK, Barthel Index, Berg Balance Scale | Significant improvements in all motor outcomes (p < 0.05); Barthel Index ↑ ~25 points | AI-driven personalization improves functional recovery across domains | AI-adaptive VR tasks with continuous monitoring and feedback |
| Morone et al., 2014 [32] | RCT (Gamified Clinical Intervention) | To examine Wii Fit gamified balance therapy in subacute stroke (N = 50) | Wii Fit + PT vs. balance therapy + PT | BBS, Barthel Index, 10 MWT, FAC | BBS ↑ 7.6 vs. 4.2 (p = 0.004); BI ↑ ~22.8 points | Low-cost gamified balance therapy can enhance post-stroke recovery | Commercial games (Wii Fit) with goal-oriented balance tasks |
| Chen et al., 2022 [55] | Meta-analysis (43 RCTs) | To evaluate overall effects of VR-supported UL rehabilitation | VR-based therapy vs. conventional therapy | FMA-UE, ROM, strength, FIM, QoL | SMD values: UE = 0.45; ROM = 1.01; strength = 0.79 (all p < 0.001) | VR-supported therapy is effective for upper-limb recovery across trials | Adaptive VR motor-learning environments |
| Ahmed et al., 2020 [56] | Protocol (Immersive VR RCT-Planned) | To test immersive VR for upper-limb rehabilitation in ischemic stroke (N = 262) | Task-oriented multisensory rehabilitation (TMSR) + immersive VR vs. TMSR | FMA-UE, UK FIM-FAM | Pending | Immersive VR expected to enhance early subacute motor rehabilitation | Fully immersive 3-D VR environment with structured progression |
| Faria et al., 2018 [33] | RCT (Clinical Cognitive–Motor) | To test Reh@Task VR platform for cognitive-motor rehabilitation in chronic stroke (N = 24) | Reh@Task + OT vs. OT alone | MoCA, Bell’s test, Digit Cancellation, FMA-UE, Barthel Index | Greater improvements in FMA-UE and cognitive measures | Combined cognitive-motor VR provides additional benefit beyond OT | Gamified VR dual motor-cognitive tasks with adaptive performance tracking |
| Myung-Mo Lee et al., 2016 [45] | Pilot Study (Clinical Usability) | To evaluate VR canoe game for trunk stability and upper-limb function (N = 10) | VR canoe game 30 min, 3×/week for 4 weeks + PT | Trunk stability, balance, UL coordination, SUS | All outcomes improved; high usability scores | VR canoe-based therapy is feasible and supports motor/postural improvement | Game-based dynamic trunk control; high usability ratings |
| Maggio et al., 2023 [41] | RCT (Cognitive Telerehabilitation–MS) | Evaluate VR cognitive telerehabilitation in multiple sclerosis (N = 36) | VRRS-based cognitive TR (Khymeia Group, Padova, Italy) | MSQoL-54 | Mental QoL ↑ 20.5 points (p < 0.001) | VR cognitive TR improves mental QoL | VRRS cognitive platform with interactive session tasks |
| Lutokhin et al., 2023 [35] | RCT (Exoskeleton + FES + VR) | Evaluate combined robotic, FES, and VR rehabilitation for early ischemic stroke recovery (N = 130) | Exoskeleton + FES + VR vs. comparators | Tinetti scale, muscle strength, stabilometry | Balance ↑ 7.1; gait ↑ 6.4; strength ↑ 13.6% | Multimodal VR-robotic systems yield strong early recovery benefits | Robotic gait + FES with VR-enhanced feedback |
| Ali et al., 2023 [46] | RCT (Parkinson’s-Gamified VR) | Compare VR vs. conventional PT for balance and QoL (N = 46) | VR balance/motor rehabilitation | SF-36, Barthel Index, BBS | BI ↑ 11 points; BBS ↑ 5.8 (p < 0.05) | VR improves balance and QoL in Parkinson’s | Gamified VR tasks with rewards and feedback |
| Paul et al., 2024 [57] | Protocol (VR-cRGS RCT) | To test VR-cRGS for stroke upper-limb recovery (N = 162 planned) | VR-cRGS vs. PT | FMA-UE, WMFT, Barthel Index, SF-36 | Pending | VR-cRGS may improve upper-limb outcomes | Mirror-based VR gaming; feedback-based movement control |
| Lülsdorff et al., 2023 [40] | RCT (Immersive VR) | Compare immersive VR vs. robotic electromechanical training (N = 52) | CUREO (iVR) (CUREosity GmbH, Düsseldorf, Germany) vs. ARMEOSpring + therapy | ARAT, UEQ | ARAT ↑ 9.8 vs. 5.1; 84% vs. 50% achieved MCID | iVR may be equal or superior to robotic training | Immersive VR with real-time arm tracking |
| Held et al., 2017 [37] | Pilot (Home-Based VR TR) | Evaluate home-based VR telerehabilitation (N = 15) | REWIRE VR platform, 12-week balance exergames | Compliance, usability, adverse events | 71% completion; 95% TAM usability; no adverse events | Safe, feasible, effective home VR system | Gamified VR balance tasks; telerehab monitoring |
| de Castro-Cros et al., 2020 [52] | Pilot (BCI-Partially Simulated) | Evaluate gamified vs. non-gamified BCI rehabilitation (N = 16) | BCI + avatar/FES vs. BCI without gamification | Classification accuracy, user satisfaction | Accuracy similar, engagement higher with gamification | Gamification boosts engagement in BCI rehab | Avatar-based BCI with reward structure |
| Alsheikhy et al., 2025 [50] | Simulation (In silico AI Model) | Develop personalized VR stroke therapy using Bi-LSTM + Firefly | AI-driven adaptive VR system | Prediction accuracy, task success | 99.06% accuracy; 98% task success; task duration ~50 s | Strong AI potential for personalized VR | Bi-LSTM + Firefly optimization |
| Pelosi et al., 2024 [51] | Simulation (reinforcement learning) | RL-driven VR reaching-movement adaptation | Q-learning-based bubble-reaching VR system | Spatial adaptation performance | Effective adaptation across sessions; works for 2 participants | RL may support autonomous difficulty progression | Reinforcement learning for spatial cue modification |
| Zhang et al., 2025 [36] | RCT (AI–Gamified Dysphagia Rehab) | Evaluate AI-video game swallowing therapy post-stroke (N = 84) | AI-VG with lip, tongue, CTAR exercises | GUSS, SSA, FOIS, MNA-SF, SWAL-QoL, adherence | GUSS ↑ 4.02; FOIS ↑ 1.07; adherence ↑ (18 vs. 16 days) | AI-based gamified dysphagia rehab is effective | AI adaptive difficulty + gamified swallowing tasks |
| Burdea et al., 2021 [48] | Usability Study | Evaluate AI-adaptive BBG controller + BrightBrainer VR | VR games + AI controller | Error rate, completion, USE scale | Usability 6–7/7; difficulty scaling worked as intended | AI-adaptive controller is feasible and usable | Automatic difficulty adaptation |
| Chen et al., 2024 [15] | Simulation (GAN-Based) | Develop GAN-based difficulty-modulation engine for rehab games | GAN model (“Egg Catcher”) | Pearson r, training loss, variation, convergence | Pearson r = 0.74; 4.5× less variation; faster convergence | GANs promising for auto-tuning difficulty | GAN-based difficulty engine |
| Author, Year | Platform | Description | Examples of Application | Benefits | Key Insights |
|---|---|---|---|---|---|
| Rodríguez-Hernández et al., 2021 [34] | VR exposure therapy | Interactive VR therapy environment combined with conventional rehab | Upper limb function, tone, stroke recovery | Enhanced motor function and recovery | VR augments traditional therapy; high effect size (η2 = 0.633) |
| House et al., 2016 [44] | BrightArm Duo system (Bright Cloud International Corp., North Brunswick, NJ, USA) | Robotic table + VR team-based gaming | Upper-limb ROM, motivation, depression | Improved ROM, enjoyment, and compliance | Gamified teamwork model feasible in nursing homes |
| Bai et al., 2022 [42] | AI-VR rehab system | Game-based rehab guided by AI system for stroke | Motor scores, ADLs, balance | Significant gains in FMA and Barthel Index | AI-driven personalization improves recovery |
| Morone et al., 2014 [32] | Nintendo Wii Fit (Nintendo Co., Ltd., Kyoto, Japan) | Commercial gaming system adapted for stroke rehab | Balance training in subacute stroke | Superior gains in BBS and ADL vs. standard therapy | Low-cost, accessible game-based therapy works |
| Faria et al., 2024 [58] | Reh@Task | VR cognitive–motor dual-task training platform | Cognition, motor, ADLs | Better arm recovery and cognitive gains | Dual-targeted VR intervention is effective |
| Myung-Mo Lee et al., 2016 [45] | Canoe Game-based VR | Trunk postural training using a canoe-themed VR interface | Trunk stability and upper-limb motor control | Usability confirmed; improved stability and function | Novel VR settings like canoe are engaging and effective |
| Lülsdorff et al., 2023 [40] | CUREO (immersive VR) | Immersive virtual reality system for upper-limb rehab | Motor recovery, user experience | Comparable or superior to robotic therapy | iVR is clinically effective and better accepted |
| de Castro-Cros et al., 2020 [52] | Gamified BCI + FES | Brain–computer interface linked to functional electrical stimulation and gamified avatar | User satisfaction, stroke recovery | High engagement, preserved accuracy | Gamification enhances BCI-based rehab usability |
| Burdea et al., 2021 [48] | BrightBrainer BBG system | AI-adaptive game controller for home-based VR rehab | Task adaptation, usability testing | Highly usable, customizable rehab tool | AI improves user-level personalization in telerehab |
| Chen et al., 2024 [15] | GAN-based Adaptive Difficulty Planner | AI model to generate personalized rehab task difficulty levels for stroke therapy games | Adaptive game difficulty in upper-limb rehab simulations | Reduces training loss and difficulty variance; generalizes well across demographics | Automates personalization of task difficulty, enabling scalable game design |
| Zhang et al., 2025 [36] | AI-based Gamified Swallowing System | Tablet-based gamified rehab with AI-driven feedback for lips, tongue, and CTAR training | Post-stroke dysphagia therapy | Improves swallowing function, oral intake, QoL, and adherence | First AI-gamified platform targeting dysphagia with high satisfaction and effectiveness |
| Outcome | Description | Examples of Application | Representative Quantitative Outcomes | Benefits | Key Insights |
|---|---|---|---|---|---|
| Upper-Limb Motor Recovery [34,58,59] | Improvement in arm and hand function using gamified systems | VR therapy, BrightArm Duo, NeuroAlreh@b | FMA-UE + 9–11 pts (p < 0.05); η2 = 0.63; ARAT + 9.8 pts; Adherence ≥ 85% | Enhanced FMA scores, ROM, coordination, and functional independence | Gamification appears to support motor learning and adherence |
| Balance and Gait Improvement [32,35,37] | Recovery of postural control and walking through VR or robotic games | Wii Fit, Exoskeleton+ FES + VR), REWIRE | BBS + 5–8 pts; Tinetti + 6–7 pts (p < 0.05); Adherence > 80% | Improved BBS, Tinetti scores, reduced fall risk | Interactive balance games were generally well tolerated and may support balance improvement at home |
| Cognitive Engagement and Compliance [40,48,52] | Patient motivation and sustained use of VR or AI platforms | BBG System, BCI + FES, iVR | SUS > 85%; TAM 95%; USE 6–7/7 scale | High usability ratings, engagement scores, sustained task repetition | Gamified telerehab is well accepted and may reduce dropout rates |
| Swallowing Function [36] | Gamified AI-based therapy for post-stroke dysphagia rehabilitation | AI-VG exercises for lips, tongue, CTAR | GUSS + 4.0; FOIS + 1.1 (p < 0.001); Adherence ≈ 90% | Improved GUSS, FOIS, and SWAL-QOL scores; higher adherence and satisfaction | Gamified telerehab shows encouraging results for specialized domains such as dysphagia |
| Personalization and Adaptive Training [15,50] | AI-driven systems that adjust rehab tasks in real-time | GAN difficulty design, Bi-LSTM Firefly system | Accuracy 98–99%; r = 0.74 vs. real data | Better matching of task to user ability, faster progress | Generative and predictive AI tools show potential to enhance self-guided telerehab precision |
| Study Design | No. of Studies | Sample Size (Mean ± Range) | Regions Represented (with No. of Studies) |
|---|---|---|---|
| Randomized Controlled Trials (RCTs) | 9 | ≈515 participants (68 ± 35; 24–130) | Europe (4): [34,40,41,58] |
| Asia (3): [32,36,42] | |||
| Middle East (1): [46] | |||
| North America (1): [35] | |||
| Pilot Studies | 4 | ≈64 participants (16 ± 6; 10–23) | Europe (2): [45,52] |
| Asia (1): [37] | |||
| North America (1): [44] | |||
| Protocols (Registered/Ongoing) | 4 | ≈584 planned (181 ± 73; 120–262) | Asia (2): [43,54] |
| Europe (1): [57] | |||
| Multinational (1): [56] | |||
| Meta-Analysis/Systematic Review | 1 | 43 RCTs pooled (N = 1893) | Global/Multinational: [55] |
| Simulation/AI Model Studies | 3 | Not applicable (Simulated datasets) | Asia (2): [15,50] |
| Europe (1): [51] | |||
| Usability Studies | 1 | N = 2 | North America (1): [48] |
| Total | 22 studies | ≈3129 participants (clinical + simulated) | Europe (9), Asia (7), North America (3), Middle East (1), Multinational (2) |
| Outcome/Domain | No. of Studies (Designs) | Risk of Bias (1–4) | Inconsistency (1–4) | Indirectness (1–4) | Imprecision (1–4) | Publication Bias (1–4) | Mean Score | Overall Certainty (GRADE) |
|---|---|---|---|---|---|---|---|---|
| Upper-limb Motor Function | 11 (8 RCTs, 3 pilots) | 3 | 3 | 4 | 3 | 4 | 3.4 | Moderate |
| Balance and Gait | 4 RCTs | 3 | 3 | 4 | 2 | 4 | 3.2 | Moderate |
| Swallowing Function | 1 RCT | 4 | 2 | 4 | 2 | 4 | 3.2 | Low |
| Activities of Daily Living (ADLs) | 2 RCTs | 3 | 2 | 4 | 2 | 4 | 3.0 | Low |
| Cognition/Engagement | 3 (2 RCTs, 1 pilot) | 2 | 2 | 4 | 2 | 4 | 2.8 | Low |
| Usability/Adherence | 5 pilot or feasibility studies | 2 | 2 | 2 | 1 | 4 | 2.2 | Very Low |
| Quality of Life/Psychosocial Well-being | 3 RCTs | 3 | 2 | 4 | 2 | 4 | 3.0 | Low |
| Overall Summary of Evidence | 22 studies (9 RCTs, 6 pilots, 4 simulations, 3 protocols) | - | - | - | - | - | ≈3.0 | Moderate → Low overall certainty |
4. Discussion
5. Conclusions
Registration and Protocol
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADLs | Activities of Daily Living |
| AI | Artificial Intelligence |
| AR | Augmented Reality |
| ARAT | Action Research Arm Test |
| BBT | Box and Block Test |
| BCI | Brain–Computer Interface |
| BI | Barthel Index |
| Bi-LSTM | Bidirectional Long Short-Term Memory |
| BBG | Balance-Based Games |
| DL | Deep Learning |
| FMA | Fugl–Meyer Assessment |
| FMA-UE | Fugl–Meyer Assessment—Upper Extremity |
| FOIS | Functional Oral Intake Scale |
| GAN | Generative Adversarial Network |
| GRADE | Grading of Recommendations Assessment, Development, and Evaluation |
| GUSS | Gugging Swallowing Screen |
| HCI | Human–Computer Interaction |
| IEEE | Institute of Electrical and Electronics Engineers |
| κ | Cohen’s Kappa |
| LMICs | Low- and Middle-Income Countries |
| ML | Machine Learning |
| MS | Multiple Sclerosis |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| PROSPERO | International Prospective Register of Systematic Reviews |
| QoL | Quality of Life |
| RCT | Randomized Controlled Trial |
| RL | Reinforcement Learning |
| RoB 2 | Risk of Bias Tool 2 |
| ROBINS-I | Risk of Bias in Non-randomized Studies of Interventions |
| SCI | Spinal Cord Injury |
| SWAL-QOL | Swallowing Quality of Life Questionnaire |
| TBI | Traumatic Brain Injury |
| VR | Virtual Reality |
| AR/VR | Augmented Reality/Virtual Reality |
| Wii Fit | Nintendo Wii Fit Balance Training System |
References
- Johnson, C.O.; Nguyen, M.; Roth, G.A.; Nichols, E.; Alam, T.; Abate, D.; Abd-Allah, F.; Abdelalim, A.; Abraha, H.N.; Abu-Rmeileh, N.M.; et al. Global, regional, and national burden of stroke, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 439–458. [Google Scholar] [CrossRef]
- Langhorne, P.; Bernhardt, J.; Kwakkel, G. Stroke care 2. Stroke rehabilitation. Lancet 2011, 377, 1693–1702. [Google Scholar] [CrossRef]
- Laver, K.E.; Adey-Wakeling, Z.; Crotty, M.; Lannin, N.A.; George, S.; Sherrington, C.J. Telerehabilitation services for stroke. Cochrane Database Syst. Rev. 2020, 1, CD010255. [Google Scholar] [CrossRef]
- Linder, S.M.; Rosenfeldt, A.B.; Bay, R.C.; Sahu, K.; Wolf, S.L.; Alberts, J.L. Improving quality of life and depression after stroke through telerehabilitation. Am. J. Occup. Ther. 2015, 69, 6902290020p1–6902290020p10. [Google Scholar] [CrossRef]
- Verma, A.; Towfighi, A.; Brown, A.; Abhat, A.; Casillas, A. Moving towards equity with digital health innovations for stroke care. Stroke 2022, 53, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Dhingra, D.; Dabas, A. Global strategy on digital health. Indian Pediatr. 2020, 57, 356–358. [Google Scholar] [CrossRef] [PubMed]
- Maclean, N.; Pound, P.; Wolfe, C.; Rudd, A. Qualitative analysis of stroke patients’ motivation for rehabilitation. BMJ 2000, 321, 1051–1054. [Google Scholar] [CrossRef]
- Nam, H.S.; Park, E.; Heo, J.H. Facilitating stroke management using modern information technology. J. Stroke 2013, 15, 135. [Google Scholar] [CrossRef]
- Sardi, L.; Idri, A.; Fernández-Alemán, J.L. A systematic review of gamification in e-Health. J. Biomed. Inform. 2017, 71, 31–48. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Escamez, F.; Cortés-Pérez, I.; Obrero-Gaitán, E.; Fusco, A. Virtual reality applications in neurorehabilitation: Current panorama and challenges. Brain Sci. 2023, 13, 819. [Google Scholar] [CrossRef]
- Yamato, T.P.; Pompeu, J.E.; Pompeu, S.M.; Hassett, L. Virtual reality for stroke rehabilitation. Phys. Ther. 2026, 96, 1508–1513. [Google Scholar] [CrossRef]
- Kamari, M.; Siqueira, V.; Bakare, J.; Sebastião, E. Virtual reality technology for physical and cognitive function rehabilitation in people with multiple sclerosis. Rehabil. Res. Pract. 2024, 2024, 2020263. [Google Scholar] [CrossRef]
- Gangemi, A.; De Luca, R.; Fabio, R.A.; Lauria, P.; Rifici, C.; Pollicino, P.; Marra, A.; Olivo, A.; Quartarone, A.; Calabrò, R.S. Effects of virtual reality cognitive training on neuroplasticity: A quasi-randomized clinical trial in patients with stroke. Biomedicines 2023, 11, 3225. [Google Scholar] [CrossRef]
- Xu, F.; Dai, Z.; Ye, Y.; Hu, P.; Cheng, H. Bibliometric and visualized analysis of the application of artificial intelligence in stroke. Front. Neurosci. 2024, 18, 1411538. [Google Scholar] [CrossRef]
- Chen, K.; Vinjamuri, R.; Wang, H.; Kadiyala, S.P. Generative AI based difficulty level design of serious games for stroke rehabilitation. IEEE Internet Things J. 2024, 11, 39560–39569. [Google Scholar] [CrossRef]
- El-Banna, M.M.; Tebbenhoff, B.; Whitlow, M.; Wyche, K.F. Motivated strategies for learning in accelerated second-degree nursing students. Nurse Educ. 2017, 42, 308–312. [Google Scholar] [CrossRef]
- Cameirão, M.S.; Bermúdez i Badia, S.; Duarte Oller, E.; Verschure, P.F. The rehabilitation gaming system: A review. In Advanced Technologies in Rehabilitation; IOS Press: Amsterdam, The Netherlands, 2009; pp. 65–83. [Google Scholar]
- Vourvopoulos, A.; Faria, A.L.; Cameirao, M.S.; i Badia, S.B. RehabNet: A distributed architecture for motor and cognitive neuro-rehabilitation. In 2013 IEEE 15th International Conference on e-Health Networking, Applications and Services (Healthcom 2013); IEEE: New York, NY, USA, 2013; pp. 454–459. [Google Scholar]
- Mulpuri, R.P.; Konda, N.; Gadde, S.T.; Amalakanti, S.; Valiveti, S.C. Artificial intelligence and machine learning in neuroregeneration: A systematic review. Cureus 2024, 16, e61400. [Google Scholar] [CrossRef]
- Prats-Bisbe, A.; López-Carballo, J.; García-Molina, A.; Leno-Colorado, D.; García-Rudolph, A.; Opisso, E.; Jané, R. Virtual reality–based neurorehabilitation support tool for people with cognitive impairments resulting from an acquired brain injury: Usability and feasibility study. JMIR Neurotechnol. 2024, 3, e50538. [Google Scholar] [CrossRef] [PubMed]
- Norouzi-Gheidari, N.; Archambault, P.S.; Fung, J. Effects of robot-assisted therapy on stroke rehabilitation in upper limbs: Systematic review and meta-analysis of the literature. J. Rehabil. Res. Dev. 2012, 49, 479–496. [Google Scholar] [CrossRef] [PubMed]
- Lo, A.C.; Guarino, P.; Krebs, H.I.; Volpe, B.T.; Bever, C.T.; Duncan, P.W.; Ringer, R.J.; Wagner, T.H.; Richards, L.G.; Bravata, D.M.; et al. Multicenter randomized trial of robot-assisted rehabilitation for chronic stroke: Methods and entry characteristics for VA ROBOTICS. Neurorehabilit. Neural Repair 2009, 23, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Elaklouk, A.M.S.; Ramli, R.Z.; Alakklouk, S.M.M.; Rahim, N.J. Design Guidelines for a Game-Based Physical Rehabilitation System: Focus Group Study. JMIR Hum. Factors 2025, 12, e67336. [Google Scholar] [CrossRef]
- Bussard, M.E.; Jessee, M.A.; El-Banna, M.M.; Cantrell, M.A.; Alrimawi, I.; Marchi, N.M.; Gonzalez, L.I.; Rischer, K.; Coy, M.L.; Poledna, M.; et al. Current practices for assessing clinical judgment in nursing students and new graduates: A scoping review. Nurse Educ. Today 2024, 134, 106078. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Moher, D. Updating guidance for reporting systematic reviews: Development of the PRISMA 2020 statement. J. Clin. Epidemiol. 2021, 134, 103–112. [Google Scholar] [CrossRef]
- Laver, K.E.; Lange, B.; George, S.; Deutsch, J.E.; Saposnik, G.; Crotty, M. Virtual reality for stroke rehabilitation. Stroke 2018, 49, e160–e161. [Google Scholar] [CrossRef]
- Chen, Y.; Abel, K.T.; Janecek, J.T.; Chen, Y.; Zheng, K.; Cramer, S.C. Home-based technologies for stroke rehabilitation: A systematic review. Int. J. Med. Inform. 2019, 123, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Massetti, T.; Da Silva, T.D.; Crocetta, T.B.; Guarnieri, R.; De Freitas, B.L.; Bianchi Lopes, P.; Watson, S.; Tonks, J.; de Mello Monteiro, C.B. The clinical utility of virtual reality in neurorehabilitation: A systematic review. J. Cent. Nerv. Syst. Dis. 2018, 10, 1179573518813541. [Google Scholar] [CrossRef]
- Szeto, S.G.; Wan, H.; Alavinia, M.; Dukelow, S.; MacNeill, H. Rehabilitation. Effect of mobile application types on stroke rehabilitation: A systematic review. J. Neuroeng. Rehabil. 2023, 20, 12. [Google Scholar] [CrossRef]
- Mickenautsch, S.; Yengopal, V. Selection bias risk in randomized controlled trials rated as low bias using Risk of Bias, Version 2 (RoB2) tool. Cureus 2024, 16, e63581. [Google Scholar] [CrossRef] [PubMed]
- Popay, J.; Roberts, H.; Sowden, A.; Petticrew, M.; Arai, L.; Rodgers, M.; Britten, N.; Roen, K.; Duffy, S. Guidance on the Conduct of Narrative Synthesis in Systematic Reviews: A Product from the ESRC Methods Programme; Lancaster University: Lancaster, UK, 2006; Volume 1, p. b92. [Google Scholar]
- Morone, G.; Tramontano, M.; Iosa, M.; Shofany, J.; Iemma, A.; Musicco, M.; Paolucci, S.; Caltagirone, C. The efficacy of balance training with video game-based therapy in subacute stroke patients: A randomized controlled trial. BioMed Res. Int. 2014, 2014, 580861. [Google Scholar] [CrossRef]
- Faria, A.L.; Cameirão, M.S.; Couras, J.F.; Aguiar, J.R.; Costa, G.M.; Bermúdez i Badia, S. Combined cognitive-motor rehabilitation in virtual reality improves motor outcomes in chronic stroke—A pilot study. Front. Psychol. 2018, 9, 854. [Google Scholar] [CrossRef]
- Rodríguez-Hernández, M.; Polonio-Lopez, B.; Corregidor-Sánchez, A.-I.; Martin-Conty, J.L.; Mohedano-Moriano, A.; Criado-Álvarez, J.-J. Effects of specific virtual reality-based therapy for the rehabilitation of the upper limb motor function post-ictus: Randomized controlled trial. Brain Sci. 2021, 11, 555. [Google Scholar] [CrossRef]
- Lutokhin, G.M.; Kashezhev, A.G.; Pogonchenkova, I.V.; Rassulova, M.A.; Turova, E.A.; Utegenova, Y.V.; Shulkina, A.V.; Samokhvalov, R.I. Effectiveness and Safety of Robotic Mechanotherapy with FES and VR in Restoring Gait and Balance in the Acute and Early Rehabilitation Period of Ischemic Stroke: Prospective Randomized Comparative Study. Bull. Rehabil. Med. 2023, 22, 22–29. [Google Scholar] [CrossRef]
- Zhang, B.; Wong, K.P.; Liu, M.; Hui, V.; Guo, C.; Liu, Z.; Liu, Y.; Xiao, Q.; Qin, J. Effect of artificial intelligence-based video-game system on dysphagia in patients with stroke: A randomized controlled trial. Clin. Nutr. 2025, 45, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Held, J.P.; Ferrer, B.; Mainetti, R.; Steblin, A.; Hertler, B.; Moreno-Conde, A.; Dueñas, A.; Pajaro, M.; Vargiu, E.; Zarco, M.; et al. Autonomous rehabilitation at stroke patients home for balance and gait: Safety, usability and compliance of a virtual reality system. Eur. J. Phys. Rehabil. Med. 2018, 54, 545–555. [Google Scholar] [CrossRef]
- Lloréns, R.; Noé, E.; Colomer, C.; Alcañiz, M. Effectiveness, usability, and cost-benefit of a virtual reality–based telerehabilitation program for balance recovery after stroke: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2015, 96, 418–425.e2. [Google Scholar] [CrossRef] [PubMed]
- Swanson, V.A.; Johnson, C.; Zondervan, D.K.; Bayus, N.; McCoy, P.; Ng, Y.F.J.; Schindele, B.; Schindele, J.; Reinkensmeyer, D.J.; Shaw, S.; et al. Optimized home rehabilitation technology reduces upper extremity impairment compared to a conventional home exercise program: A randomized, controlled, single-blind trial in subacute stroke. Neurorehabilit. Neural Repair 2023, 37, 53–65. [Google Scholar] [CrossRef]
- Lülsdorff, K.; Junker, F.B.; Studer, B.; Wittenberg, H.; Pickenbrock, H.; Schmidt-Wilcke, T. Neurorehabilitation of the upper extremity–immersive virtual reality vs. electromechanically assisted training. A comparative study. Front. Neurol. 2023, 14, 1290637. [Google Scholar] [CrossRef]
- Maggio, M.G.; Cannavò, A.; Quartarone, A.; Manuli, A.; Tonin, P.; Calabrò, R.S. Enhancing the quality of life of patients with multiple sclerosis: Promising results on the role of cognitive tele-rehabilitation plus virtual reality. Brain Sci. 2023, 13, 1636. [Google Scholar] [CrossRef]
- Bai, Y.; Liu, F.; Zhang, H. Artificial Intelligence Limb Rehabilitation System on Account of Virtual Reality Technology on Long-Term Health Management of Stroke Patients in the Context of the Internet. Comput. Math. Methods Med. 2022, 2022, 2688003. [Google Scholar] [CrossRef]
- Zhou, Z.-Q.; Hua, X.-Y.; Wu, J.-J.; Xu, J.-J.; Ren, M.; Shan, C.-L.; Xu, J.-G. Combined robot motor assistance with neural circuit-based virtual reality (NeuCir-VR) lower extremity rehabilitation training in patients after stroke: A study protocol for a single-centre randomised controlled trial. BMJ Open 2022, 12, e064926. [Google Scholar] [CrossRef]
- House, G.; Burdea, G.; Polistico, K.; Grampurohit, N.; Roll, D.; Damiani, F.; Keeler, S.; Hundal, J. A rehabilitation first-tournament between teams of nursing home residents with chronic stroke. Games Health J. 2016, 5, 75–83. [Google Scholar] [CrossRef]
- Lee, M.-M.; Shin, D.-C.; Song, C.-H. Canoe game-based virtual reality training to improve trunk postural stability, balance, and upper limb motor function in subacute stroke patients: A randomized controlled pilot study. J. Phys. Ther. Sci. 2016, 28, 2019–2024. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.; Suleman, R.; Noor, A.; Ahmad, I.; Shakeel, M.; Aqeel, M. Effectiveness of a Virtual Reality-Based Rehabilitation Program versus Conventional Physical Therapy in Improving Motor Function and Balance in Stroke Survivors: A Randomized Controlled Trial. J. Health Rehabil. Res. 2023, 3, 817–821. [Google Scholar] [CrossRef]
- Saywell, N.L.; Vandal, A.C.; Mudge, S.; Hale, L.; Brown, P.; Feigin, V.; Hanger, C.; Taylor, D. Telerehabilitation after stroke using readily available technology: A randomized controlled trial. Neurorehabilit. Neural Repair 2021, 35, 88–97. [Google Scholar] [CrossRef]
- Burdea, G.; Kim, N.; Polistico, K.; Kadaru, A.; Grampurohit, N.; Roll, D.; Damiani, F. Assistive game controller for artificial intelligence-enhanced telerehabilitation post-stroke. Assist. Technol. 2021, 33, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.D.; Zhou, X.H.; Freeman, D.H., Jr.; Freeman, J. A non-parametric method for the comparison of partial areas under ROC curves and its application to large health care data sets. Stat. Med. 2002, 21, 701–715. [Google Scholar] [CrossRef]
- Alsheikhy, A.A.; Shawly, T.; Said, Y.E.; Ahmed, H.E.; Alazzam, M.B. Developing machine learning models for personalized game-based stroke rehabilitation therapy in virtual reality. Alex. Eng. J. 2025, 121, 358–369. [Google Scholar] [CrossRef]
- Pelosi, A.D.; Roth, N.; Yehoshua, T.; Itah, D.; Braun Benyamin, O.; Dahan, A. Personalized rehabilitation approach for reaching movement using reinforcement learning. Sci. Rep. 2024, 14, 17675. [Google Scholar] [CrossRef]
- de Castro-Cros, M.; Sebastian-Romagosa, M.; Rodríguez-Serrano, J.; Opisso, E.; Ochoa, M.; Ortner, R.; Guger, C.; Tost, D. Effects of gamification in BCI functional rehabilitation. Front. Neurosci. 2020, 14, 882. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Or, C.K.; Chen, T. Effectiveness of using virtual reality–supported exercise therapy for upper extremity motor rehabilitation in patients with stroke: Systematic review and meta-analysis of randomized controlled trials. J. Med. Internet Res. 2022, 24, e24111. [Google Scholar] [CrossRef]
- Kang, D.; Park, J.; Eun, S.-D. Home-based virtual reality exergame program after stroke rehabilitation for patients with stroke: A study protocol for a multicenter, randomized controlled trial. Life 2023, 13, 2256. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Shih, H.-M.; Wu, J.-E.; Huang, F.-W.; Chen, W.-K.; Chen, D.-M.; Chung, Y.-T.; Wang, C.C. Machine learning–based triage to identify low-severity patients with a short discharge length of stay in emergency department. BMC Emerg. Med. 2022, 22, 88. [Google Scholar] [CrossRef]
- Ahmed, N.; Mauad, V.A.Q.; Gomez-Rojas, O.; Sushea, A.; Castro-Tejada, G.; Michel, J.; Liñares, J.M.; Pedrosa Salles, L.; Candido Santos, L.; Shan, M. The impact of rehabilitation-oriented virtual reality device in patients with ischemic stroke in the early subacute recovery phase: Study protocol for a phase iii, single-blinded, randomized, controlled clinical trial. J. Central Nerv. Syst. Dis. 2020, 12, 1179573519899471. [Google Scholar] [CrossRef]
- Paul, R.; Elango, S.; Chakravarthy, S.; Sinha, A.; PR, S.; Raju, B.; Sarma, P.S.; Hafsath, S.; Francis, A.J.A.; Darshini, D. Feasibility and efficacy of virtual reality rehabilitation compared with conventional physiotherapy for upper extremity impairment due to ischaemic stroke: Protocol for a randomised controlled trial. BMJ Open 2024, 14, e086556. [Google Scholar] [CrossRef]
- Faria, A.L.; Almeida, Y.; Branco, D.; Câmara, J.; Cameirão, M.; Ferreira, L.; Moreira, A.; Paulino, T.; Rodrigues, P.; Spinola, M. NeuroAIreh@ b: An artificial intelligence-based methodology for personalized and adaptive neurorehabilitation. Front. Neurol. 2024, 14, 1258323. [Google Scholar] [CrossRef]
- House, G.; Burdea, G.; Polistico, K.; Roll, D.; Kim, J.; Damiani, F.; Keeler, S.; Hundal, J.; Pollack, S. BrightArm duo integrative rehabilitation for post-stroke maintenance in skilled nursing facilities. In 2015 International Conference on Virtual Rehabilitation (ICVR); IEEE: New York, NY, USA, 2015; pp. 207–214. [Google Scholar]
- Bishop, C.M.; Bishop, H. Deep Learning: Foundations and Concepts; Springer: Cham, Switzerland, 2023. [Google Scholar] [CrossRef]
- Fayaz, S.A.; Jahangeer Sidiq, S.; Zaman, M.; Butt, M.A. Machine learning: An introduction to reinforcement learning. In Machine Learning and Data Science: Fundamentals and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2022; pp. 1–22. [Google Scholar]
- Goodfellow, I.J.; Pouget-Abadie, J.; Mirza, M.; Xu, B.; Warde-Farley, D.; Ozair, S.; Courville, A.; Bengio, Y. Generative adversarial networks. Commun. ACM 2020, 63, 139–144. [Google Scholar] [CrossRef]
- Attoh-Mensah, E.; Boujut, A.; Desmons, M.; Perrochon, A. Artificial intelligence in personalized rehabilitation: Current applications and a SWOT analysis. Front. Digit. Health 2025, 7, 1606088. [Google Scholar] [CrossRef] [PubMed]
- Winstein, C.J.; Stein, J.; Arena, R.; Bates, B.; Cherney, L.R.; Cramer, S.C.; Deruyter, F.; Eng, J.J.; Fisher, B.; Harvey, R.L.; et al. Guidelines for adult stroke rehabilitation and recovery: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2016, 47, e98–e169. [Google Scholar] [CrossRef] [PubMed]
- Catania, V.; Rundo, F.; Panerai, S.; Ferri, R. Virtual reality for the rehabilitation of acquired cognitive disorders: A narrative review. Bioengineering 2023, 11, 35. [Google Scholar] [CrossRef] [PubMed]
- Park, C.; Lee, B.-C. A systematic review of the effects of interactive telerehabilitation with remote monitoring and guidance on balance and gait performance in older adults and individuals with neurological conditions. Bioengineering 2024, 11, 460. [Google Scholar] [CrossRef]
- Holden, M.K. Virtual environments for motor rehabilitation. Cyberpsychol. Behav. 2005, 8, 187–211. [Google Scholar] [CrossRef]
- Burke, J.W.; McNeill, M.; Charles, D.K.; Morrow, P.J.; Crosbie, J.H.; McDonough, S.M. Optimising engagement for stroke rehabilitation using serious games. Vis. Comput. 2009, 25, 1085–1099. [Google Scholar] [CrossRef]
- Forman, C.R.; Nielsen, J.B.; Lorentzen, J. Neuroplasticity at home: Improving home-based motor learning through technological solutions. A review. Front. Rehabil. Sci. 2021, 2, 789165. [Google Scholar] [CrossRef]
- Stinear, C.M.; Lang, C.E.; Zeiler, S.; Byblow, W.D. Advances and challenges in stroke rehabilitation. Lancet Neurol. 2020, 19, 348–360. [Google Scholar] [CrossRef]
- Silva-Batista, C.; de Almeida, F.O.; Wilhelm, J.L.; Horak, F.B.; Mancini, M.; King, L.A. Telerehabilitation by videoconferencing for balance and gait in people with Parkinson’s disease: A scoping review. Geriatrics 2024, 9, 66. [Google Scholar] [CrossRef]
- Khan, A.; Imam, Y.Z.; Muneer, M.; Al Jerdi, S.; Gill, S.K. Virtual reality in stroke recovery: A meta-review of systematic reviews. Bioelectron. Med. 2024, 10, 23. [Google Scholar] [CrossRef]
- Laver, K.E.; Lange, B.; George, S.; Deutsch, J.E.; Saposnik, G.; Chapman, M.; Crotty, M. Virtual reality for stroke re-habilitation. Cochrane Database Syst. Rev. 2025, 6, CD008349. [Google Scholar] [CrossRef]
- El-Banna, M.M.; Whitlow, M.; McNelis, A.M. Improving pharmacology standardized test and final examination scores through team-based learning. Nurse Educ. 2020, 45, 47–50. [Google Scholar] [CrossRef]
- Sailer, M.; Hense, J.U.; Mayr, S.K.; Mandl, H. How gamification motivates: An experimental study of the effects of specific game design elements on psychological need satisfaction. Comput. Hum. Behav. 2017, 69, 371–380. [Google Scholar] [CrossRef]
- Ding, K.; Ma, Y.; Zhang, L.; Gu, Y.; Pan, H.; Gu, Z.-E.; Zhang, H.J. Patient-centered insights into virtual reality rehabilitation for stroke: A systematic review and qualitative meta-synthesis. J. Neuroeng. Rehabil. 2025, 22, 124. [Google Scholar] [CrossRef]
- Surya, N.; Someshwar, H.P. Low-Cost telerehabilitation in low-and middle-income countries (LMICs): Overcoming barriers to access and improving healthcare delivery. NeuroRehabilitation 2025, 56, 30–36. [Google Scholar] [CrossRef]
- Zhang, J.-R.; Wu, Y.-E.; Huang, Y.-F.; Zhang, S.-Q.; Pan, W.-L.; Huang, J.-X.; Huang, Q.-P. Effectiveness of smart health-based rehabilitation on patients with poststroke dysphagia: A brief research report. Front. Neurol. 2023, 13, 1110067. [Google Scholar] [CrossRef]
- Alshami, A.; Nashwan, A.; AlDardour, A.; Qusini, A. Artificial Intelligence in rehabilitation: A narrative review on advancing patient care. Rehabilitación 2025, 59, 100911. [Google Scholar] [CrossRef] [PubMed]
- Caramia, F.; D’Angelantonio, E.; Lucangeli, L.; Camomilla, V. Validation of low cost wearables sensors in motor telerehabilitation exercises. In 2024 IEEE International Symposium on Medical Measurements and Applications (MeMeA); IEEE: New York, NY, USA, 2024; pp. 1–6. [Google Scholar]
- Topol, E.J. High-performance medicine: The convergence of human and artificial intelligence. Nat. Med. 2019, 25, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Holzinger, A.; Carrington, A.; Müller, H. Measuring the quality of explanations: The system causability scale (SCS) comparing human and machine explanations. KI-Künstliche Intell. 2020, 34, 193–198. [Google Scholar] [CrossRef] [PubMed]


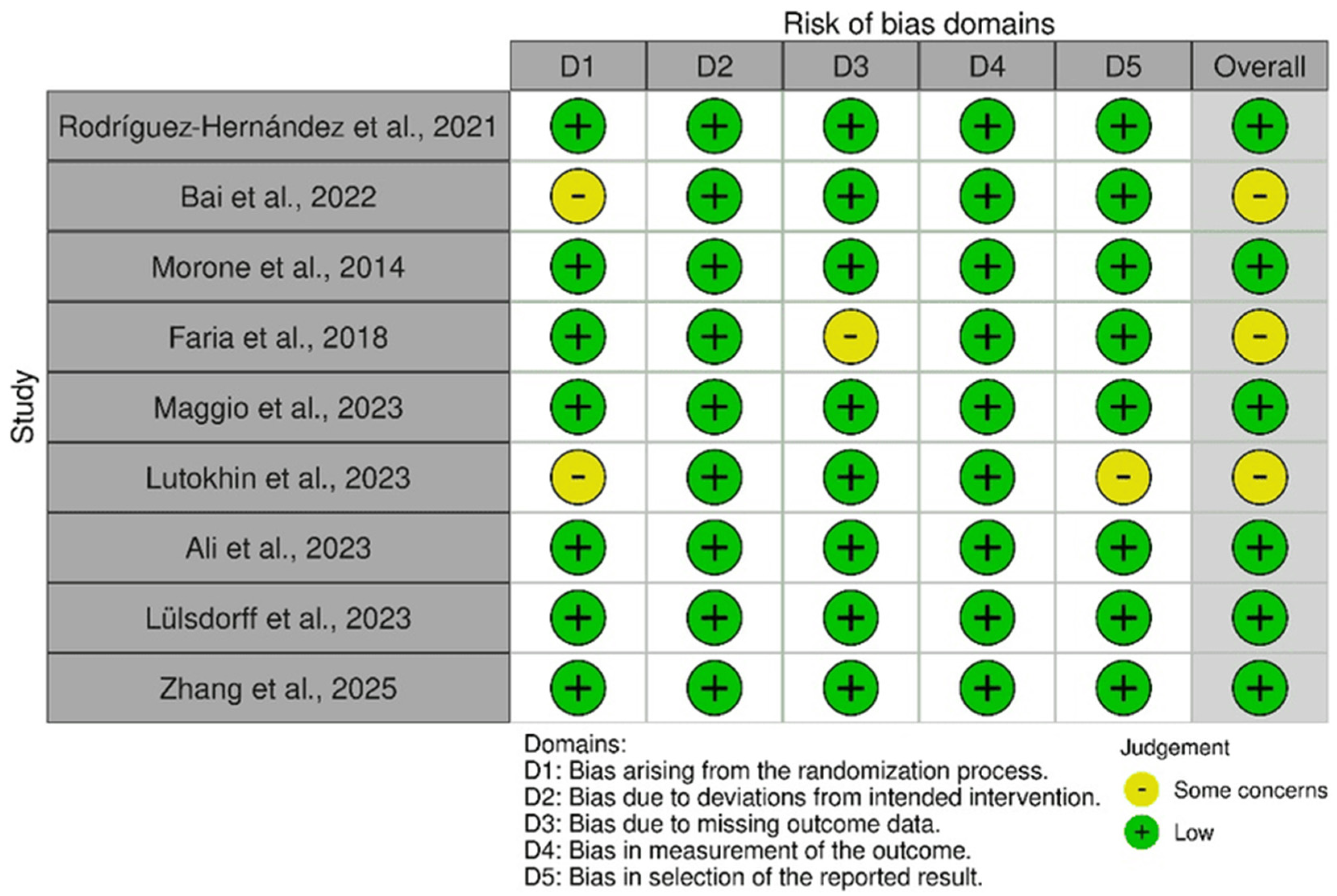
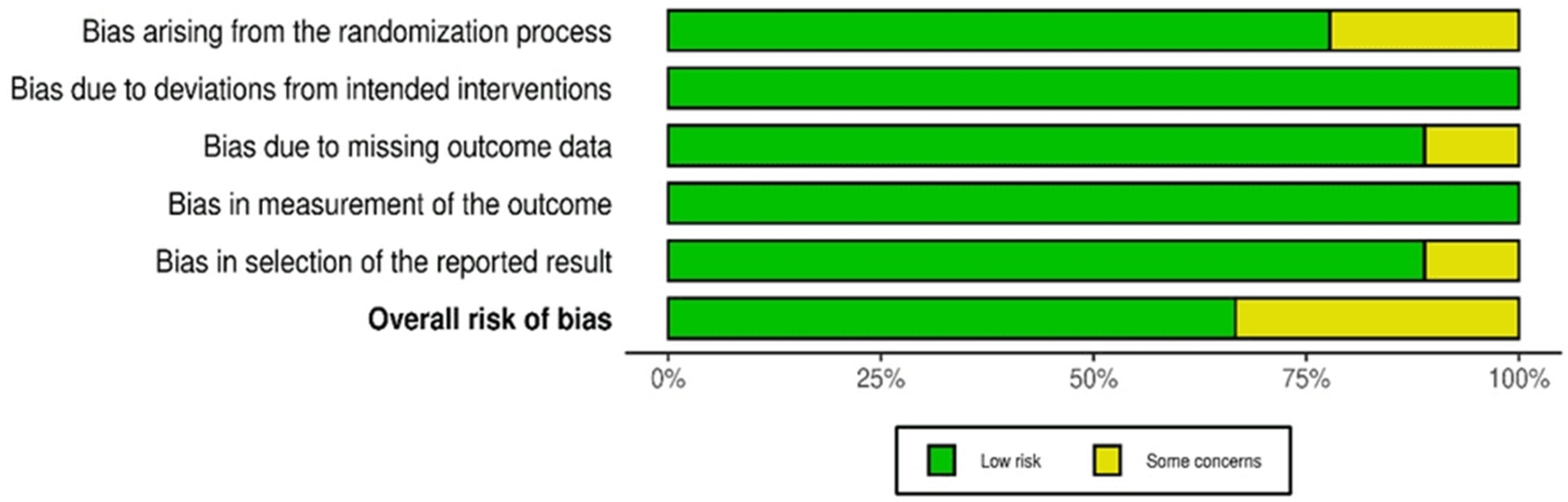


| Study (Author, Year) | AI Methodology | Primary Inputs | Algorithm/Decision Logic Reported | Validation Context |
|---|---|---|---|---|
| Bai (2022) [42] | AI-adaptive VR system | Kinematics, task performance | Supervised learning-based performance monitoring used to adjust task difficulty and progression in real time; adaptation described functionally | Clinical RCT |
| Zhang (2025) [36] | AI-gamified video-game therapy | Task accuracy, session frequency | Rule-based AI system with adaptive difficulty modulation based on user performance trends across sessions | Clinical RCT |
| Burdea (2021) [48] | AI-adaptive controller | Error rate, task completion time | Automatic difficulty scaling triggered by performance thresholds; internal decision rules not explicitly specified | Usability study |
| Lutokhin (2023) [35] | Multimodal AI-assisted rehabilitation | Sensor signals, motor performance metrics | AI-supported personalization combining sensor feedback and performance metrics to modulate training intensity | Clinical RCT |
| Alsheikhy (2025) [50] | Bi-LSTM + Firefly optimization | Synthetic performance data | Bi-LSTM network predicts task performance; Firefly algorithm optimizes difficulty parameters for personalized VR therapy | Simulation-only |
| Chen (2024) [15] | GAN-based difficulty generator | Synthetic game/task data | GAN trained to generate task-difficulty levels matching real-data distributions (reported via correlation analysis) | Simulation-only |
| Pelosi (2024) [51] | Reinforcement learning (Q-learning) | Task success, spatial performance | Q-learning updates task difficulty based on reward signals derived from reaching performance | Simulation/proof-of-concept |
| de Castro-Cros (2020) [52] | Gamified BCI paradigm | BCI classification output | Decision logic compares gamified vs. non-gamified feedback; no change in classifier accuracy but increased engagement | Pilot/partially simulated |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
El-Banna, M.M.; Rizvi, M.R.; Sami, W.; Sharma, A.; Atyeh, R.R. Digital and Intelligent Rehabilitation Technologies in Stroke and Neurological Disorders: A Systematic Review of Artificial Intelligence, Virtual Reality, Gamification, and Emerging Therapeutic Platforms in Neurorehabilitation. Bioengineering 2026, 13, 195. https://doi.org/10.3390/bioengineering13020195
El-Banna MM, Rizvi MR, Sami W, Sharma A, Atyeh RR. Digital and Intelligent Rehabilitation Technologies in Stroke and Neurological Disorders: A Systematic Review of Artificial Intelligence, Virtual Reality, Gamification, and Emerging Therapeutic Platforms in Neurorehabilitation. Bioengineering. 2026; 13(2):195. https://doi.org/10.3390/bioengineering13020195
Chicago/Turabian StyleEl-Banna, Majeda M., Moattar Raza Rizvi, Waqas Sami, Ankita Sharma, and Rushdy R. Atyeh. 2026. "Digital and Intelligent Rehabilitation Technologies in Stroke and Neurological Disorders: A Systematic Review of Artificial Intelligence, Virtual Reality, Gamification, and Emerging Therapeutic Platforms in Neurorehabilitation" Bioengineering 13, no. 2: 195. https://doi.org/10.3390/bioengineering13020195
APA StyleEl-Banna, M. M., Rizvi, M. R., Sami, W., Sharma, A., & Atyeh, R. R. (2026). Digital and Intelligent Rehabilitation Technologies in Stroke and Neurological Disorders: A Systematic Review of Artificial Intelligence, Virtual Reality, Gamification, and Emerging Therapeutic Platforms in Neurorehabilitation. Bioengineering, 13(2), 195. https://doi.org/10.3390/bioengineering13020195








