A Parametric Finite Element Analysis of Chick Embryo Aortic Valve Leaflet Biomechanics
Abstract
1. Introduction
2. Materials and Methods
2.1. Doppler Ultrasound Measurement of Aortic Flow in Chick Embryo
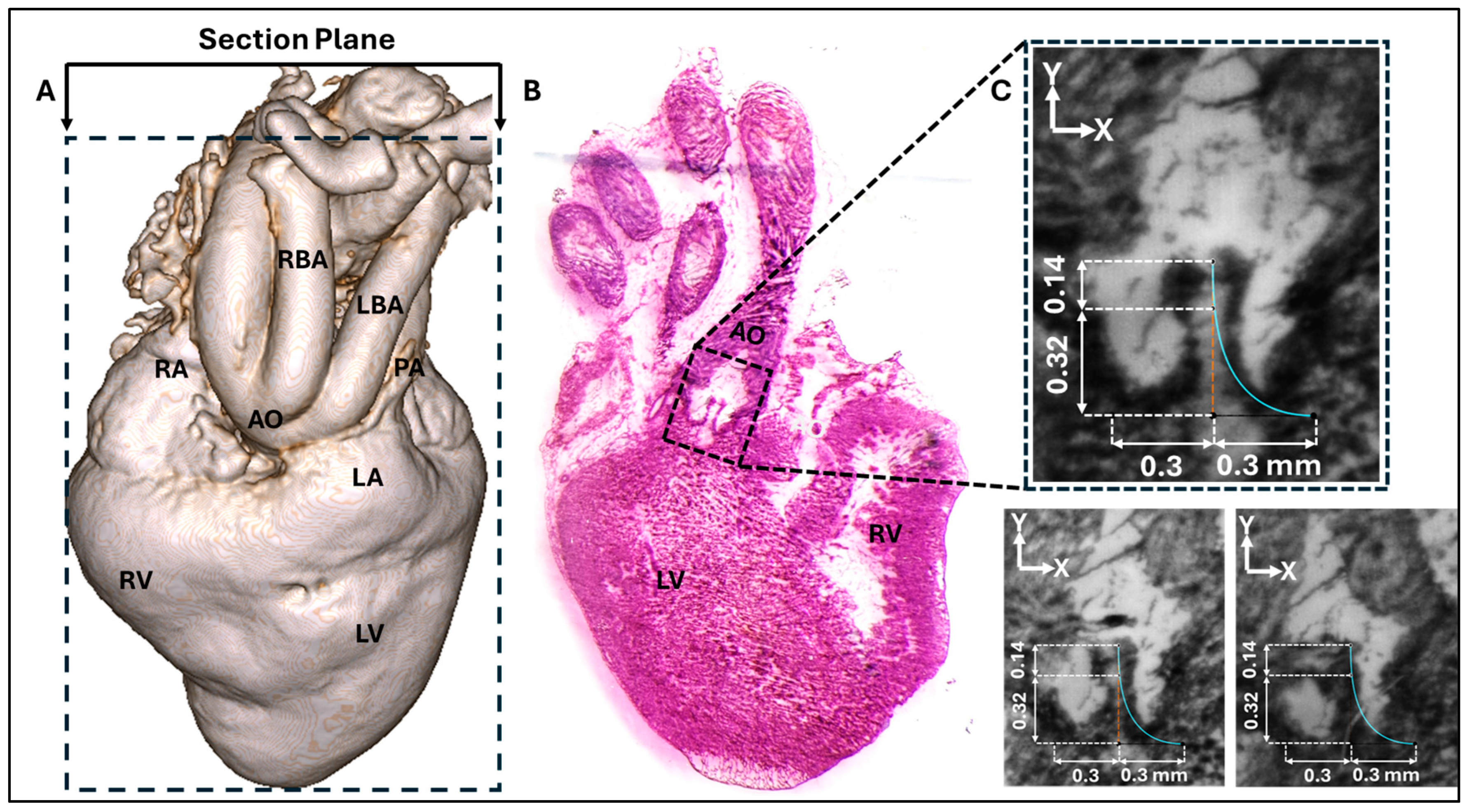
2.2. Cryostat Sectioning
2.3. Aortic Valve Modeling
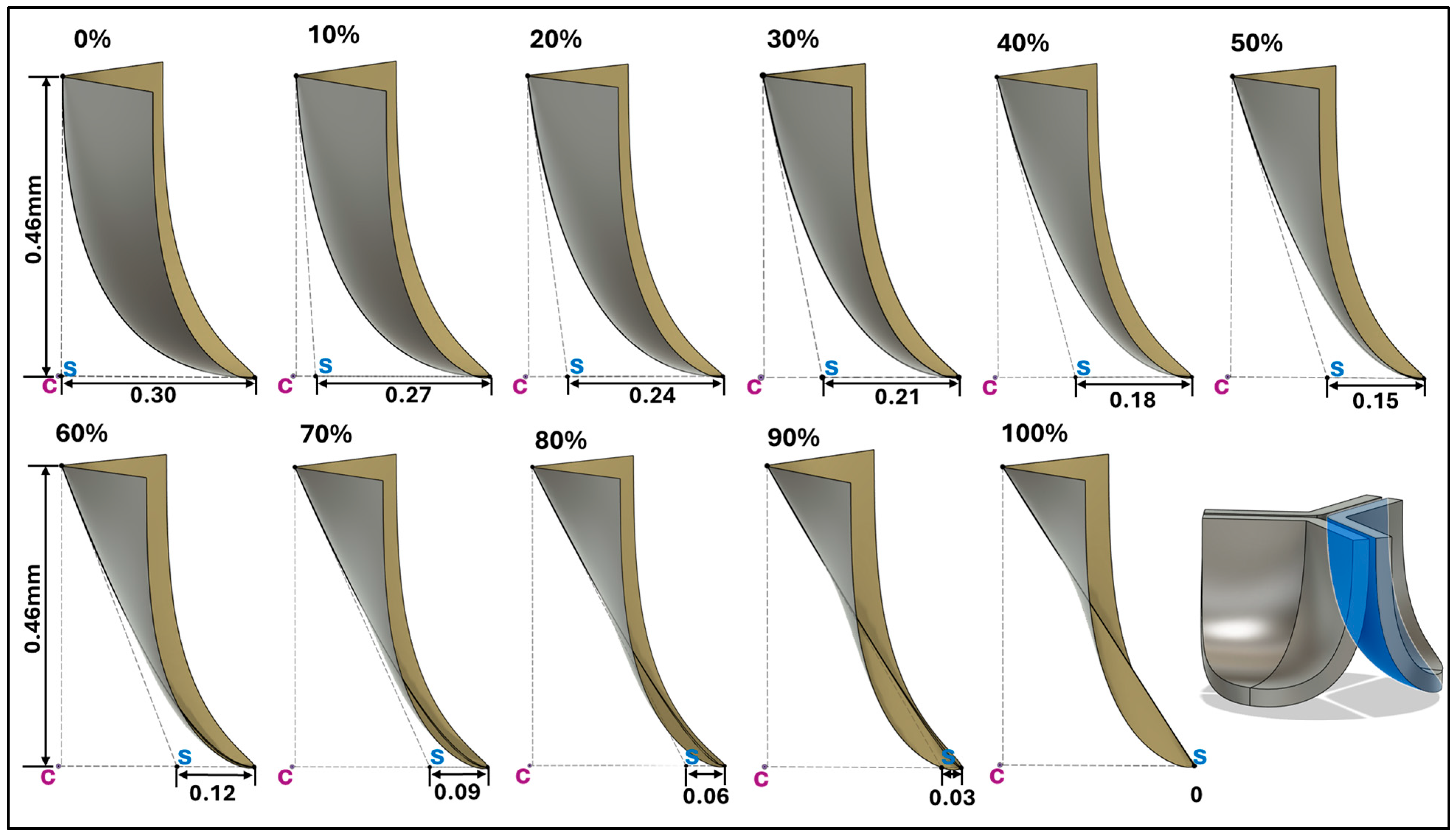
2.4. Middle Curve Shift-Based Parametric Modeling of Aortic Valve Leaflets
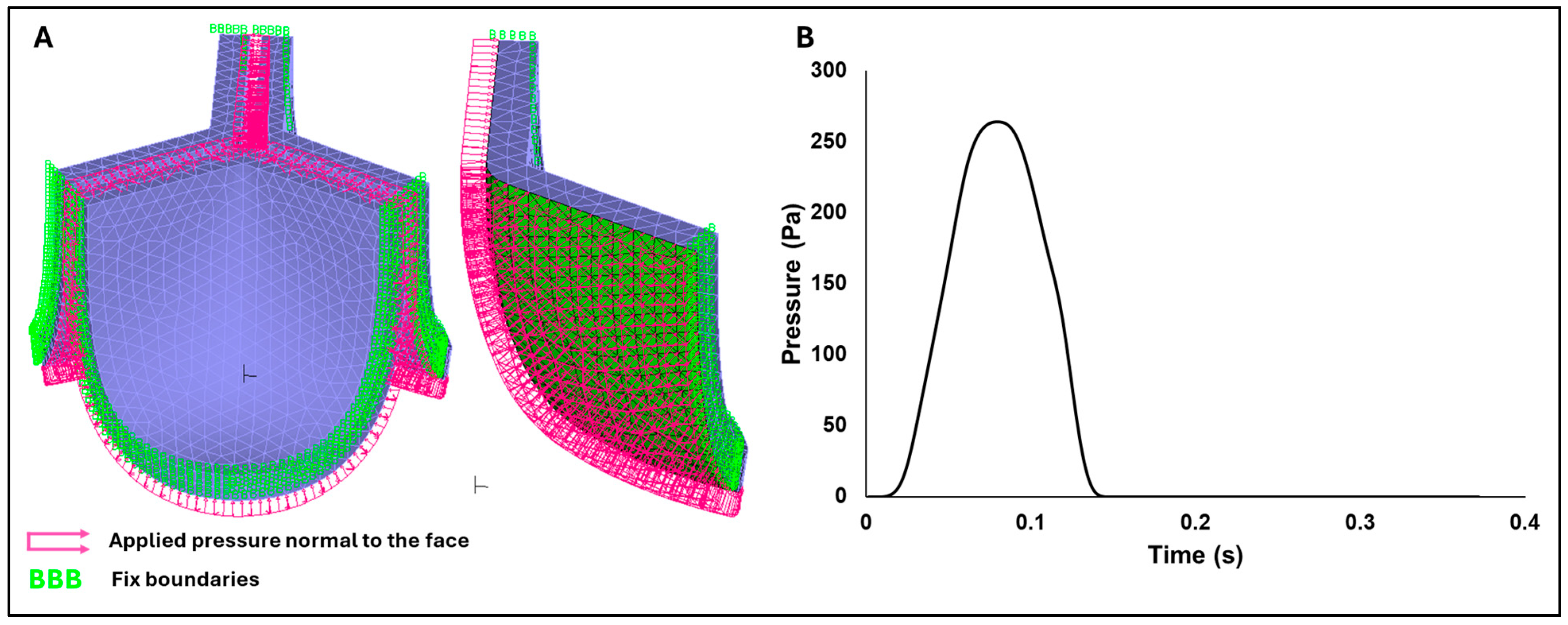
2.5. Finite Element Analysis Setup
2.6. Mesh Generation and Convergence
2.7. Material Sensitivity Analysis
| TEST | μ1 (Pa) | α1 | (Pa) |
|---|---|---|---|
| 0 | 577.60 | 26.260 | 7583.89 |
| 1 | 530.34 | 24.634 | 6532.20 |
| 2 | 483.08 | 23.008 | 5557.35 |
| 3 | 435.82 | 21.382 | 4659.35 |
| 4 | 388.56 | 19.756 | 3838.20 |
| 5 | 341.30 | 18.130 | 3093.88 |
| 6 | 294.04 | 16.504 | 2426.42 |
| 7 | 246.78 | 14.878 | 1835.80 |
| 8 | 199.52 | 13.252 | 1322.02 |
| 9 | 152.26 | 11.626 | 885.09 |
| 10 | 105 | 10 | 525.00 |
2.8. Frequency Analysis and Damping Coefficient Calculation
3. Results
3.1. Mesh Convergence and Material Sensitivity
3.2. Damping Coefficient Sensitivity
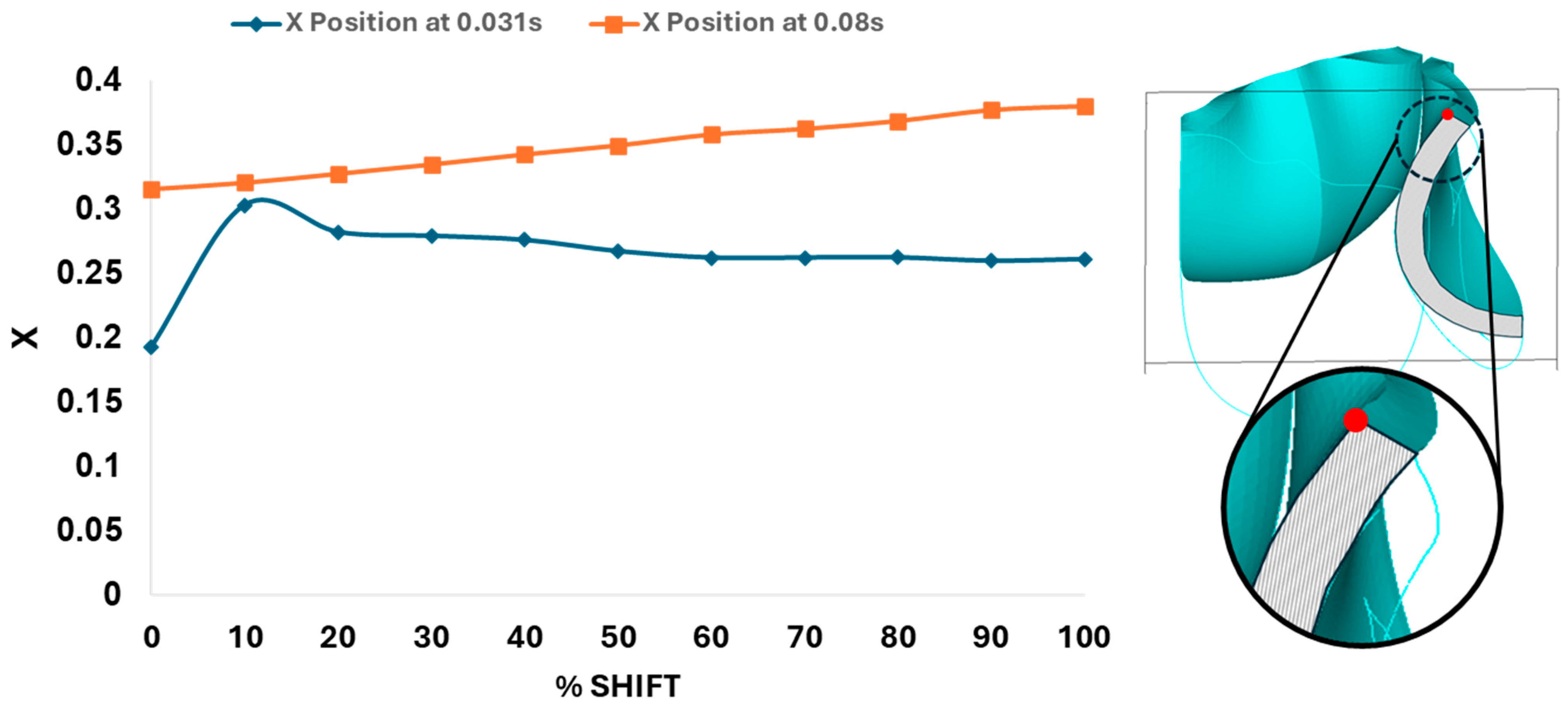
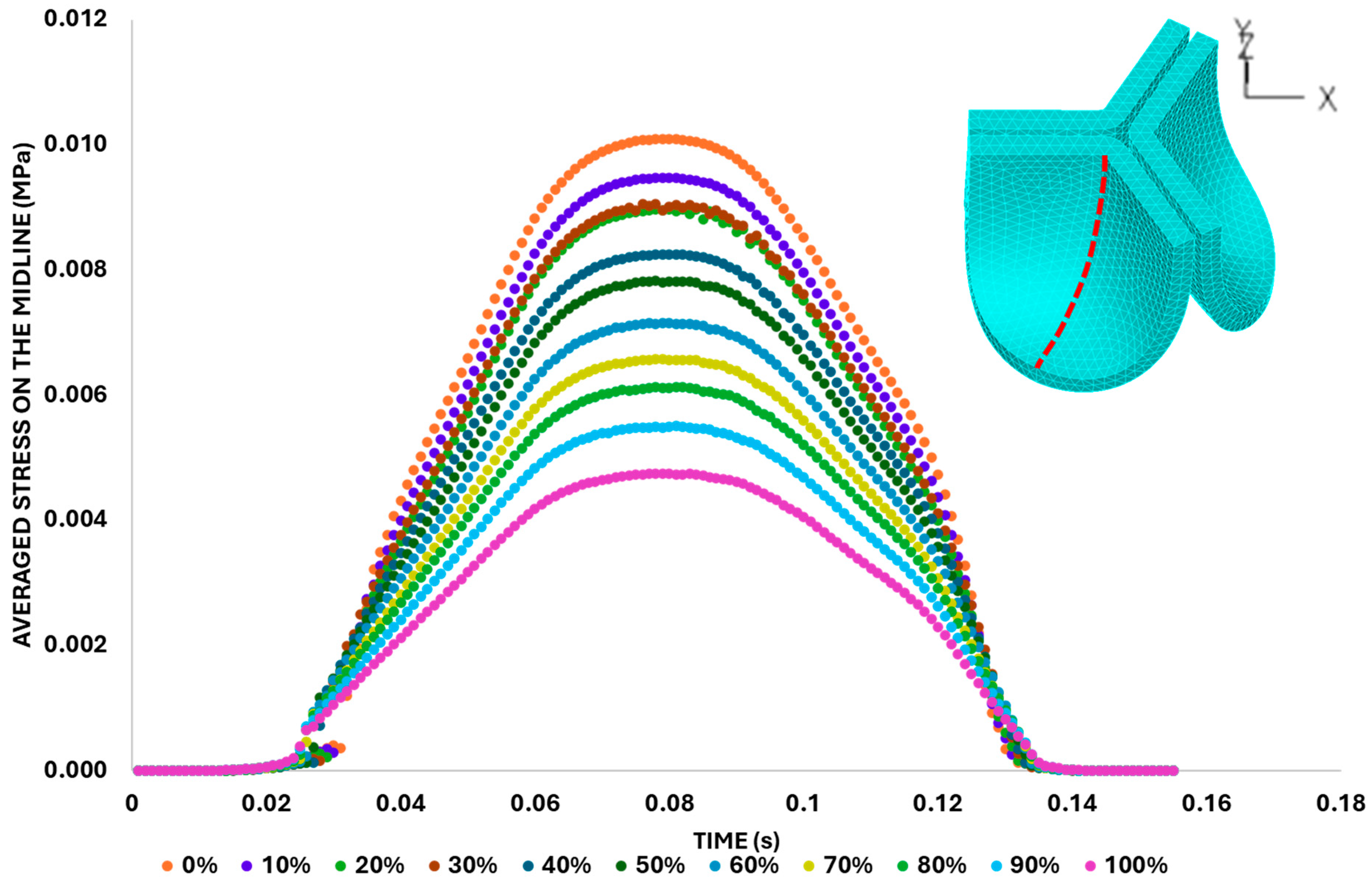
3.3. Valve Leaflet Middle Curve Shift
4. Discussion
4.1. Limitations
4.2. Parametric Valve Modeling
4.3. Leaflet Material Properties
4.4. Biomechanical Analysis
4.5. Effect of Leaflet Midline Curve on Valve Biomechanics
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crawford, P.T.; Arbor, T.C.; Bordoni, B. Anatomy, Thorax, Aortic Valve; StatPearls: Orlando, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK559384/ (accessed on 18 August 2025).
- Hsu, C.-P.D.; Tchir, A.; Mirza, A.; Chaparro, D.; Herrera, R.E.; Hutcheson, J.D.; Ramaswamy, S. Valve Endothelial Cell Exposure to High Levels of Flow Oscillations Exacerbates Valve Interstitial Cell Calcification. Bioengineering 2022, 9, 393. [Google Scholar] [CrossRef]
- Lindman, B.R.; Patel, J.N. Multimorbidity in Older Adults with Aortic Stenosis. Clin. Geriatr. Med. 2016, 32, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Hulin, A.; Hego, A.; Lancellotti, P.; Oury, C. Advances in Pathophysiology of Calcific Aortic Valve Disease Propose Novel Molecular Therapeutic Targets. Front. Cardiovasc. Med. 2018, 5, 21. [Google Scholar] [CrossRef]
- Gomez-Stallons, M.V.; Tretter, J.T.; Hassel, K.; Gonzalez-Ramos, O.; Amofa, D.; Ollberding, N.J.; Mazur, W.; Choo, J.K.; Smith, J.M.; Kereiakes, D.J.; et al. Calcification and extracellular matrix dysregulation in human postmortem and surgical aortic valves. Heart 2019, 105, 1616–1621. [Google Scholar] [CrossRef]
- Kodigepalli, K.M.; Thatcher, K.; West, T.; Howsmon, D.P.; Schoen, F.J.; Sacks, M.S.; Breuer, C.K.; Lincoln, J. Biology and Biomechanics of the Heart Valve Extracellular Matrix. J. Cardiovasc. Dev. Dis. 2020, 7, 57. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, S.; Ferrari, G.; Gorman, R.C.; Gorman, J.H., III; Schoen, F.J.; Sacks, M.S. Heart Valve Biomechanics and Underlying Mechanobiology. Compr. Physiol. 2016, 6, 1743–1780. [Google Scholar] [CrossRef]
- Arjunon, S.; Rathan, S.; Jo, H.; Yoganathan, A.P. Aortic valve: Mechanical environment and mechanobiology. Ann. Biomed. Eng. 2013, 41, 1331–1346. [Google Scholar] [CrossRef]
- Salman, H.E.; Yalcin, H.C. Computational Modeling of Blood Flow Hemodynamics for Biomechanical Investigation of Cardiac Development and Disease. J. Cardiovasc. Dev. Dis. 2021, 8, 14. [Google Scholar] [CrossRef]
- Pang, K.L.; Parnall, M.; Loughna, S. Effect of altered haemodynamics on the developing mitral valve in chick embryonic heart. J. Mol. Cell. Cardiol. 2017, 108, 114–126. [Google Scholar] [CrossRef]
- Goddard, L.M.; Duchemin, A.-L.; Ramalingan, H.; Wu, B.; Chen, M.; Bamezai, S.; Yang, J.; Li, L.; Morley, M.P.; Wang, T.; et al. Hemodynamic Forces Sculpt Developing Heart Valves through a KLF2-WNT9B Paracrine Signaling Axis. Dev. Cell 2017, 43, 274–289.e5. [Google Scholar] [CrossRef] [PubMed]
- Thubrikar, M. The Aortic Valve; Routledge: London, UK, 2018; pp. 8–9. [Google Scholar]
- Butcher, J.T.; Markwald, R.R. Valvulogenesis: The moving target. Philos. Trans. R. Soc. B Biol. Sci. 2007, 362, 1489–1503. [Google Scholar] [CrossRef]
- Taber, L.A. Biomechanics of Cardiovascular Development. Annual Review Biomedical Engineering 2001, 3, 1–25. [Google Scholar] [CrossRef]
- Midgett, M.; López, C.S.; David, L.; Maloyan, A.; Rugonyi, S. Increased hemodynamic load in early embryonic stages alters endocardial to mesenchymal transition. Front. Physiol. 2017, 8, 56. [Google Scholar] [CrossRef]
- England, J.; Loughna, S. Vertebrate Embryo: An Overview of Heart Development and the Utilisation of Multiple Animal Models in Research. Encycl. Life Sci. 2018, 1–11. [Google Scholar] [CrossRef]
- Midgett, M.; Rugonyi, S. Congenital heart malformations induced by hemodynamic altering surgical interventions. Front. Physiol. 2014, 5, 287. [Google Scholar] [CrossRef] [PubMed]
- Baratchi, S.; Alser, M.; Zakaria, Z.Z.; Sharma, A.; Abdelrahman, H.A.; Yalcin, H.C. Adaptation of a Mice Doppler Echocardiography Platform to Measure Cardiac Flow Velocities for Embryonic Chicken and Adult Zebrafish. Front. Bioeng. Biotechnol. 2019, 7, 445257. [Google Scholar] [CrossRef]
- Goenezen, S.; Chivukula, V.K.; Midgett, M.; Phan, L.; Rugonyi, S. 4D subject-specific inverse modeling of the chick embryonic heart outflow tract hemodynamics. Biomech. Model. Mechanobiol. 2016, 15, 723–743. [Google Scholar] [CrossRef] [PubMed]
- Dabiri, Y.; Ronsky, J.; Ali, I.; Basha, A.; Bhanji, A.; Narine, K. Effects of Leaflet Design on Transvalvular Gradients of Bioprosthetic Heart Valves. Cardiovasc. Eng. Technol. 2016, 7, 363–373. [Google Scholar] [CrossRef]
- Zhou, J.; Wu, Y.; Chen, L.; Li, T.; Xiong, Y.; Chen, Y. Numerical Simulation Studies on the Design of the Prosthetic Heart Valves Belly Curves. Fluids 2024, 9, 209. [Google Scholar] [CrossRef]
- Dabiri, Y.; Narine, K. The Roles of the Leaflet Geometry in the Structural Deterioration of Bioprosthetic Aortic Valves. Prosthesis 2025, 7, 86. [Google Scholar] [CrossRef]
- Sebastian, K.; Sylvia, P.; Stefan, S.; Pfensig, S.; Siewert, S.; Schmitz, K.; Stiehm, M.; Stefanie, K.; Niels, G.; Alper, Ö. Influence of leaflet geometry on hydrodynamic performance of transcatheter aortic valve prostheses. Current Dir. Biomed. Eng. 2019, 5, 473–475. [Google Scholar] [CrossRef]
- Salman, H.E.; Saltik, L.; Yalcin, H.C. Computational Analysis of Wall Shear Stress Patterns on Calcified and Bicuspid Aortic Valves: Focus on Radial and Coaptation Patterns. Fluids 2021, 6, 287. [Google Scholar] [CrossRef]
- Bakhaty, A.A.; Govindjee, S.; Mofrad, M.R.K. A Coupled Multiscale Approach to Modeling Aortic Valve Mechanics in Health and Disease. Appl. Sci. 2021, 11, 8332. [Google Scholar] [CrossRef]
- Gilmanov, A.; Stolarski, H.; Sotiropoulos, F. Flow-Structure Interaction Simulations of the Aortic Heart Valve at Physiologic Conditions: The Role of Tissue Constitutive Model. J. Biomech. Eng. 2018, 140, 041003. [Google Scholar] [CrossRef]
- Flamini, V.; DeAnda, A.; Griffith, B.E. Immersed boundary-finite element model of fluid–structure interaction in the aortic root. Theor. Comput. Fluid Dyn. 2016, 30, 139–164. [Google Scholar] [CrossRef]
- Hasan, A.; Kolahdouz, E.M.; Enquobahrie, A.; Caranasos, T.G.; Vavalle, J.P.; Griffith, B.E. Image-based immersed boundary model of the aortic root. Med. Eng. Phys. 2017, 47, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Bialas, O.; Zmudzki, J. Fea of displacements and stresses of aortic heart valve leaflets during the opening phase. J. Achiev. Mater. Manuf. Eng. 2019, 92, 29–35. [Google Scholar] [CrossRef]
- Kuchumov, A.G.; Makashova, A.; Vladimirov, S.; Borodin, V.; Dokuchaeva, A. Fluid–Structure Interaction Aortic Valve Surgery Simulation: A Review. Fluids 2023, 8, 295. [Google Scholar] [CrossRef]
- Chandran, K.B. Role of Computational Simulations in Heart Valve Dynamics and Design of Valvular Prostheses. J. Med Eng. Technol. 2010, 46, 220–230. [Google Scholar] [CrossRef]
- Whiting, R.; Sander, E.; Conway, C.; Vaughan, T.J. In silico modelling of aortic valve implants–predicting in vitro performance using finite element analysis. J. Med. Eng. Technol. 2022, 46, 220–230. [Google Scholar] [CrossRef]
- Mutlu, O.; Mazhar, N.; Saribay, M.; Yavuz, M.M.; Ozturk, D.; Ghareeb, A.N.; Alnabti, A.; Yalcin, H.C. Finite Element Analysis of Evolut Transcatheter Heart Valves: Effects of Aortic Geometries and Valve Sizes on Post-TAVI Wall Stresses and Deformations. J. Clin. Med. 2025, 14, 850. [Google Scholar] [CrossRef] [PubMed]
- Haj-Ali, R.; Marom, G.; Zekry, S.B.; Rosenfeld, M.; Raanani, E. A general three-dimensional parametric geometry of the native aortic valve and root for biomechanical modeling. J. Biomech. 2012, 45, 2392–2397. [Google Scholar] [CrossRef] [PubMed]
- Hamburger, V.; Hamilton, H.L. A Series of Normal Stages in the Development of the Chick Embryo. Dev. Dyn. 1992, 195, 231–272. [Google Scholar] [CrossRef]
- Baumgartner, H.; Hung, J.; Bermejo, J.; Chambers, J.B.; Evangelista, A.; Griffin, B.P.; Iung, B.; Otto, C.M.; Pellikka, P.A.; Quiñones, M. Echocardiographic Assessment of Valve Stenosis: EAE/ASE Recommendations for Clinical Practice. J. Am. Soc. Echocardiogr. 2009, 22, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, A.; Sacks, M.S. An inverse modeling approach for semilunar heart valve leaflet mechanics: Exploitation of tissue structure. Biomech. Model. Mechanobiol. 2016, 15, 909–932. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Armour, C.; Kandail, H.; O’REgan, D.P.; Bahrami, T.; Mirsadraee, S.; Pirola, S.; Xu, X.Y. Fluid–structure interaction analysis of a healthy aortic valve and its surrounding haemodynamics. Int. J. Numer. Method. Biomed. Eng. 2024, 40, e3865. [Google Scholar] [CrossRef]
- Pfensig, S.; Kaule, S.; Sämann, M.; Stiehm, M.; Grabow, N.; Schmitz, K.-P.; Siewert, S. Assessment of heart valve performance by finite-element design studies of polymeric leaflet-structures. Curr. Dir. Biomed. Eng. 2017, 3, 631–634. [Google Scholar] [CrossRef]
- Lohr, M.J.; Sugerman, G.P.; Kakaletsis, S.; Lejeune, E.; Rausch, M.K. An introduction to the Ogden model in biomechanics: Benefits, implementation tools and limitations. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2022, 380, 20210365. [Google Scholar] [CrossRef]
- Tango, A.M.; Salmonsmith, J.; Ducci, A.; Burriesci, G. Validation and Extension of a Fluid–Structure Interaction Model of the Healthy Aortic Valve. Cardiovasc. Eng. Technol. 2018, 9, 739–751. [Google Scholar] [CrossRef]
- Yao, J.; Varner, V.D.; Brilli, L.L.; Young, J.M.; Taber, L.A.; Perucchio, R. Viscoelastic material properties of the myocardium and cardiac jelly in the looping chick heart. J. Biomech. Eng. 2012, 134, 024502. [Google Scholar] [CrossRef]
- Bathe, K.J. Finite Element Procedures; Klaus-Jurgen Bathe: Cambridge, MA, USA, 1996. [Google Scholar]
- Dassault Systèmes. ABAQUS 6.6 Documentation: Effects of Damping on the Stable Time Increment in ABAQUS/Explicit. Section 20.1.1. 2023. Available online: https://classes.engineering.wustl.edu/2009/spring/mase5513/abaqus/docs/v6.6/books/usb/default.htm?startat=pt05ch20s01abm43.html (accessed on 18 August 2025).
- Phoon, C.K.L.; Aristizábal, O.; Turnbull, D.H. Spatial velocity profile in mouse embryonic aorta and Doppler-derived volumetric flow: A preliminary model. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, H908–H916. [Google Scholar] [CrossRef]
- Kowalski, W.J.; Dur, O.; Wang, Y.; Patrick, M.J.; Tinney, J.P.; Keller, B.B.; Pekkan, K. Critical Transitions in Early Embryonic Aortic Arch Patterning and Hemodynamics. PLoS ONE 2013, 8, e60271. [Google Scholar] [CrossRef]
- Buskohl, P.R.; Gould, R.A.; Butcher, J.T. Quantification of embryonic atrioventricular valve biomechanics during morphogenesis. J. Biomech. 2012, 45, 895–902. [Google Scholar] [CrossRef] [PubMed]
- Gharaie, S.H.; Morsi, Y. A novel design of a polymeric aortic valve. Int. J. Artif. Organs 2015, 38, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Morganti, S.; Zakerzadeh, R.; Kamensky, D.; Auricchio, F.; Reali, A.; Hughes, T.J.R.; Sacks, M.S.; Hsu, M. A framework for designing patient-specific bioprosthetic heart valves using immersogeometric fluid–structure interaction analysis. Int. J. Numer. Method. Biomed. Eng. 2018, 34, e2938. [Google Scholar] [CrossRef]
- Saraeian, M.; Corpuz, A.M.; Hsu, M.C.; Krishnamurthy, A. PARAVALVE: An open source framework for parametric design and fluid–structure interaction simulation of bioprosthetic heart valves in patient-specific aortic geometries. Comput. Aided Geom. Des. 2025, 120, 102455. [Google Scholar] [CrossRef]
- Pase, G.; Brinkhuis, E.; De Vries, T.; Kosinka, J.; Willems, T.; Bertoglio, C. A parametric geometry model of the aortic valve for subject-specific blood flow simulations using a resistive approach. Biomech. Model. Mechanobiol. 2023, 22, 987–1002. [Google Scholar] [CrossRef]
- Smuts, A.N.; Blaine, D.C.; Scheffer, C.; Weich, H.; Doubell, A.F.; Dellimore, K.H. Application of finite element analysis to the design of tissue leaflets for a percutaneous aortic valve. J. Mech. Behav. Biomed. Mater. 2011, 4, 85–98. [Google Scholar] [CrossRef]
- Li, K.; Sun, W. Simulated transcatheter aortic valve deformation: A parametric study on the impact of leaflet geometry on valve peak stress. Int. J. Numer. Method. Biomed. Eng. 2017, 33, e02814. [Google Scholar] [CrossRef]
- Travaglino, S.; Murdock, K.; Tran, A.; Martin, C.; Liang, L.; Wang, Y.; Sun, W. Computational optimization study of transcatheter aortic valve leaflet design using porcine and bovine leaflets. J. Biomech. Eng. 2020, 142, 011007. [Google Scholar] [CrossRef]
- Corso, P.; Obrist, D. On the role of aortic valve architecture for physiological hemodynamics and valve replacement, Part I: Flow configuration and vortex dynamics. Comput. Biol. Med. 2024, 176, 108526. [Google Scholar] [CrossRef]
- Mao, W.; Li, K.; Sun, W. Fluid–Structure Interaction Study of Transcatheter Aortic Valve Dynamics Using Smoothed Particle Hydrodynamics. Cardiovasc. Eng. Technol. 2016, 7, 374–388. [Google Scholar] [CrossRef]
- De Hart, J.; Baaijens, F.P.T.; Peters, G.W.M.; Schreurs, P.J.G. A computational fluid-structure interaction analysis of a fiber-reinforced stentless aortic valve. J. Biomech. 2003, 36, 699–712. [Google Scholar] [CrossRef]
- Wertheimer, S.; Habayib, L.; Nudelman, R.; Richter, S.; Rami, H.A. Soft-coral collagen fiber biocomposites for aortic valve leaflet engineering: From material design to computational analysis. Mech. Adv. Mater. Struct. 2025, 1–14. [Google Scholar] [CrossRef]
- Jahren, S.E.; Vennemann, B.; Bornemann, K.M.; Rösgen, T.; Obrist, D. Modes of Leaflet Fluttering: Quantitative Characterization of a Bovine Bioprosthetic Heart Valve. Ann. Biomed. Eng. 2025, 54, 410–421. [Google Scholar] [CrossRef]
- Fringand, T.; Mace, L.; Cheylan, I.; Lenoir, M.; Favier, J. Analysis of Fluid–Structure Interaction Mechanisms for a Native Aortic Valve, Patient-Specific Ozaki Procedure, and a Bioprosthetic Valve. Ann. Biomed. Eng. 2024, 52, 3021–3036. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Jin, M.; Li, W.; Zhang, H.; Li, H.; Li, S.; Yang, Y.; Zhu, M.; Shi, Y.; Zhang, X.; et al. In Silico Trials of Prosthetic Valves Replicate Methodologies for Evaluating the Fatigue Life of Artificial Leaflets to Expand Beyond In Vitro Tests and Conventional Clinical Trials. Biomedicines 2025, 13, 1135. [Google Scholar] [CrossRef] [PubMed]
- Sarrafzadeh-Ghadimi, N.; Ghalichi, F.; Niroomand-Oscuii, H.; Fatouraee, N. Design of an aortic polymeric valve with asymmetric leaflets and evaluation of its performance by finite element method. Comput. Biol. Med. 2022, 145, 105440. [Google Scholar] [CrossRef]
- Marro, M.; Kossar, A.P.; Xue, Y.; Frasca, A.; Levy, R.J.; Ferrari, G. Noncalcific mechanisms of bioprosthetic structural valve degeneration. J. Am. Heart Assoc. 2021, 10, e018921. [Google Scholar] [CrossRef] [PubMed]









| = 1 | (s−1) |
|---|---|
| 0.1 | 9.5589 |
| 0.5 | 47.7947 |
| 1 | 95.5894 |
| 2.5 | 238.9735 |
| 10 | 955.8940 |
| 20 | 1911.7880 |
| Displacement Mag. (mm) | Effective Stress (MPa) | Ascending Transition | Peak Pressure | |
|---|---|---|---|---|
 |  | 0.031 s | 0.08 s | |
| 0% SHIFT | 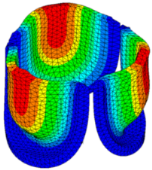 |  |  |  |
| 10% SHIFT |  |  |  |  |
| 20% SHIFT | 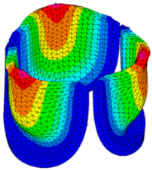 |  |  |  |
| 30% SHIFT |  |  |  |  |
| 40% SHIFT |  |  |  |  |
| 50% SHIFT |  |  |  |  |
| 60% SHIFT |  |  |  |  |
| 70% SHIFT |  |  |  |  |
| 80% SHIFT |  | 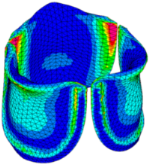 |  |  |
| 90% SHIFT | 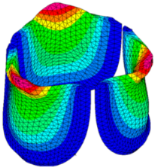 |  |  |  |
| 100% SHIFT |  |  |  |  |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mutlu, O.; Rugonyi, S. A Parametric Finite Element Analysis of Chick Embryo Aortic Valve Leaflet Biomechanics. Bioengineering 2026, 13, 189. https://doi.org/10.3390/bioengineering13020189
Mutlu O, Rugonyi S. A Parametric Finite Element Analysis of Chick Embryo Aortic Valve Leaflet Biomechanics. Bioengineering. 2026; 13(2):189. https://doi.org/10.3390/bioengineering13020189
Chicago/Turabian StyleMutlu, Onur, and Sandra Rugonyi. 2026. "A Parametric Finite Element Analysis of Chick Embryo Aortic Valve Leaflet Biomechanics" Bioengineering 13, no. 2: 189. https://doi.org/10.3390/bioengineering13020189
APA StyleMutlu, O., & Rugonyi, S. (2026). A Parametric Finite Element Analysis of Chick Embryo Aortic Valve Leaflet Biomechanics. Bioengineering, 13(2), 189. https://doi.org/10.3390/bioengineering13020189







