A Novel, Low-Cost, 3D-Printed Motorized Injector for Retinal Sheet Transplantation
Abstract
1. Introduction
2. Materials and Methods
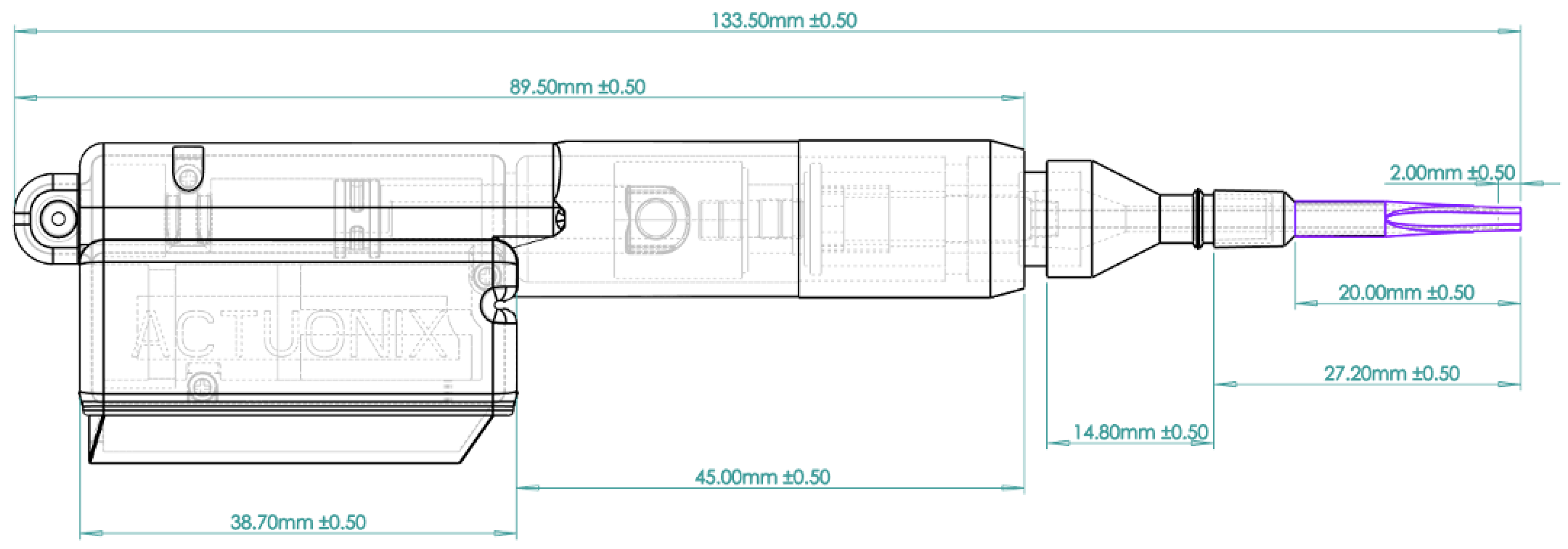
2.1. Design of Motorized Injector System
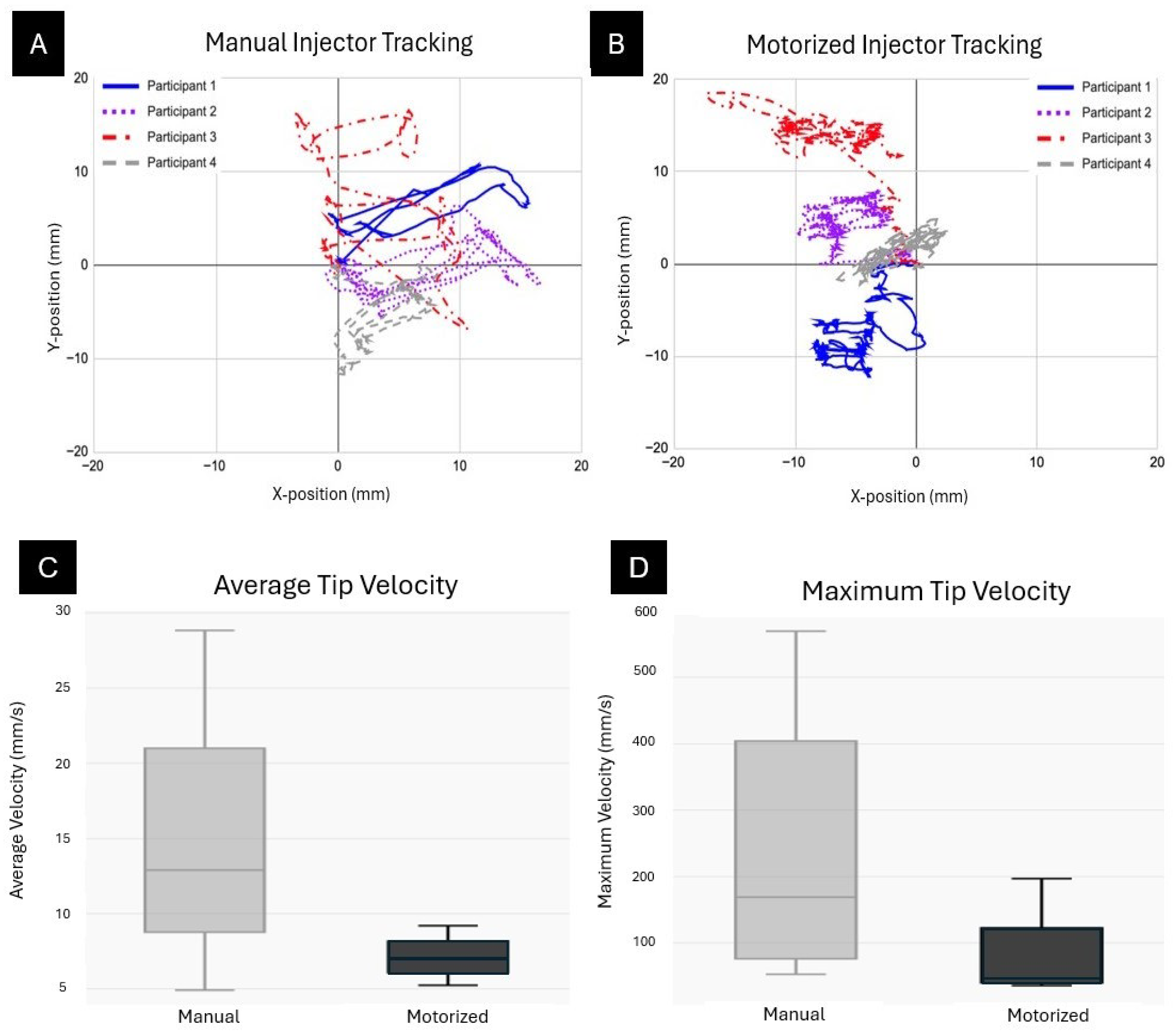
2.2. Measurement of Injector Tip Movement
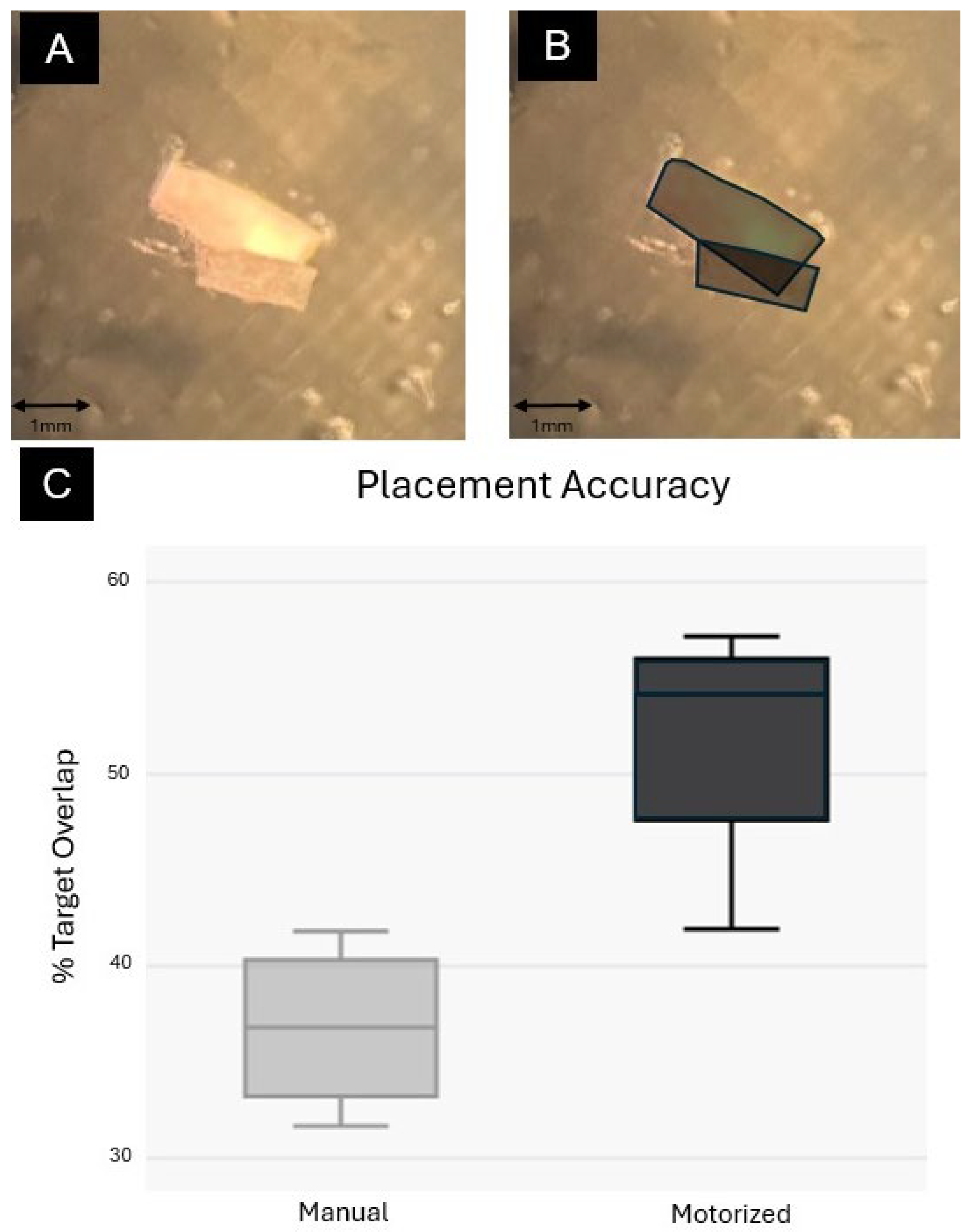
2.3. Accuracy of Retinal Sheet Placement
3. Results
3.1. Measurement of Injector Tip Movement
3.2. Accuracy of Retinal Sheet Placement
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AMD | Age-related macular degeneration |
| RPE | Retinal pigmented epithelium |
| hESC | Human embryonic stem cell |
References
- Rachitskaya, A.; Yuan, A.; Davidson, S.; Streicher, M.; DeBenedictis, M.; Rosenfeldt, A.B.; Alberts, J. Computer-Assisted Immersive Visual Rehabilitation in Argus II Retinal Prosthesis Recipients. Ophthalmol. Retin. 2020, 4, 613–619. [Google Scholar] [CrossRef]
- Humayun, M.S.; Lee, S.Y. Advanced Retina Implants. Ophthalmol. Retin. 2022, 6, 899–905. [Google Scholar] [CrossRef] [PubMed]
- Gogliettino, A.R.; Madugula, S.S.; Grosberg, L.E.; Vilkhu, R.S.; Brown, J.; Nguyen, H.; Kling, A.; Hottowy, P.; Dąbrowski, W.; Sher, A.; et al. High-Fidelity Reproduction of Visual Signals by Electrical Stimulation in the Central Primate Retina. J. Neurosci. Off. J. Soc. Neurosci. 2023, 43, 4625–4641. [Google Scholar] [CrossRef] [PubMed]
- Muqit, M.M.K.; Le Mer, Y.; Olmos de Koo, L.; Holz, F.G.; Sahel, J.A.; Palanker, D. Prosthetic Visual Acuity with the PRIMA Subretinal Microchip in Patients with Atrophic Age-Related Macular Degeneration at 4 Years Follow-Up. Ophthalmol. Sci. 2024, 4, 100510. [Google Scholar] [CrossRef]
- Schwartz, S.D.; Tan, G.; Hosseini, H.; Nagiel, A. Subretinal Transplantation of Embryonic Stem Cell-Derived Retinal Pigment Epithelium for the Treatment of Macular Degeneration: An Assessment at 4 Years. Investig. Ophthalmol. Vis. Sci. 2016, 57, ORSFc1-9. [Google Scholar] [CrossRef]
- Kashani, A.H.; Lebkowski, J.S.; Rahhal, F.M.; Avery, R.L.; Salehi-Had, H.; Dang, W.; Lin, C.-M.; Mitra, D.; Zhu, D.; Thomas, B.B.; et al. A Bioengineered Retinal Pigment Epithelial Monolayer for Advanced, Dry Age-Related Macular Degeneration. Sci. Transl. Med. 2018, 10, eaao4097. [Google Scholar] [CrossRef]
- Radtke, N.D.; Aramant, R.B.; Petry, H.M.; Green, P.T.; Pidwell, D.J.; Seiler, M.J. Vision Improvement in Retinal Degeneration Patients by Implantation of Retina Together with Retinal Pigment Epithelium. Am. J. Ophthalmol. 2008, 146, 172–182.e1. [Google Scholar] [CrossRef]
- Seiler, M.J.; Aramant, R.B. Cell Replacement and Visual Restoration by Retinal Sheet Transplants. Prog. Retin. Eye Res. 2012, 31, 661–687. [Google Scholar] [CrossRef]
- Seiler, M.J.; Lin, R.E.; McLelland, B.T.; Mathur, A.; Lin, B.; Sigman, J.; De Guzman, A.T.; Kitzes, L.M.; Aramant, R.B.; Thomas, B.B. Vision Recovery and Connectivity by Fetal Retinal Sheet Transplantation in an Immunodeficient Retinal Degenerate Rat Model. Investig. Ophthalmol. Vis. Sci. 2017, 58, 614–630. [Google Scholar] [CrossRef]
- Lin, B.; McLelland, B.T.; Mathur, A.; Aramant, R.B.; Seiler, M.J. Sheets of Human Retinal Progenitor Transplants Improve Vision in Rats with Severe Retinal Degeneration. Exp. Eye Res. 2018, 174, 13–28. [Google Scholar] [CrossRef]
- Lin, B.; McLelland, B.T.; Aramant, R.B.; Thomas, B.B.; Nistor, G.; Keirstead, H.S.; Seiler, M.J. Retina Organoid Transplants Develop Photoreceptors and Improve Visual Function in RCS Rats with RPE Dysfunction. Investig. Ophthalmol. Vis. Sci. 2020, 61, 34. [Google Scholar] [CrossRef]
- McLelland, B.T.; Lin, B.; Mathur, A.; Aramant, R.B.; Thomas, B.B.; Nistor, G.; Keirstead, H.S.; Seiler, M.J. Transplanted hESC-Derived Retina Organoid Sheets Differentiate, Integrate, and Improve Visual Function in Retinal Degenerate Rats. Investig. Ophthalmol. Vis. Sci. 2018, 59, 2586–2603. [Google Scholar] [CrossRef]
- Lin, B.; Singh, R.K.; Seiler, M.J.; Nasonkin, I.O. Survival and Functional Integration of Human Embryonic Stem Cell–Derived Retinal Organoids After Shipping and Transplantation into Retinal Degeneration Rats. Stem Cells Dev. 2024, 33, 201–213. [Google Scholar] [CrossRef]
- Watari, K.; Yamasaki, S.; Tu, H.-Y.; Shikamura, M.; Kamei, T.; Adachi, H.; Tochitani, T.; Kita, Y.; Nakamura, A.; Ueyama, K.; et al. Self-Organization, Quality Control, and Preclinical Studies of Human iPSC-Derived Retinal Sheets for Tissue-Transplantation Therapy. Commun. Biol. 2023, 6, 164. [Google Scholar] [CrossRef]
- Iraha, S.; Tu, H.-Y.; Yamasaki, S.; Kagawa, T.; Goto, M.; Takahashi, R.; Watanabe, T.; Sugita, S.; Yonemura, S.; Sunagawa, G.A.; et al. Establishment of Immunodeficient Retinal Degeneration Model Mice and Functional Maturation of Human ESC-Derived Retinal Sheets after Transplantation. Stem Cell Rep. 2018, 10, 1059–1074. [Google Scholar] [CrossRef]
- Assawachananont, J.; Mandai, M.; Okamoto, S.; Yamada, C.; Eiraku, M.; Yonemura, S.; Sasai, Y.; Takahashi, M. Transplantation of Embryonic and Induced Pluripotent Stem Cell-Derived 3D Retinal Sheets into Retinal Degenerative Mice. Stem Cell Rep. 2014, 2, 662–674. [Google Scholar] [CrossRef]
- Matsuyama, T.; Tu, H.-Y.; Sun, J.; Hashiguchi, T.; Akiba, R.; Sho, J.; Fujii, M.; Onishi, A.; Takahashi, M.; Mandai, M. Genetically Engineered Stem Cell-Derived Retinal Grafts for Improved Retinal Reconstruction after Transplantation. iScience 2021, 24, 102866. [Google Scholar] [CrossRef]
- Thomas, B.B.; Lin, B.; Martinez-Camarillo, J.C.; Zhu, D.; McLelland, B.T.; Nistor, G.; Keirstead, H.S.; Humayun, M.S.; Seiler, M.J. Co-Grafts of Human Embryonic Stem Cell Derived Retina Organoids and Retinal Pigment Epithelium for Retinal Reconstruction in Immunodeficient Retinal Degenerate Royal College of Surgeons Rats. Front. Neurosci. 2021, 15, 752958. [Google Scholar] [CrossRef]
- Brown, D. Tracker Video Analysis and Modeling Tool. Available online: https://opensourcephysics.github.io/tracker-website/ (accessed on 13 October 2024).
- Taylor, R.; Jensen, P.; Whitcomb, L.; Barnes, A.; Kumar, R.; Stoianovici, D.; Gupta, P.; Wang, Z.; deJuan, E.; Kavoussi, L. A Steady-Hand Robotic System for Microsurgical Augmentation. In Medical Image Computing and Computer-Assisted Intervention—MICCAI’99; Taylor, C., Colchester, A., Eds.; Lecture Notes in Computer Science; Springer: Berlin/Heidelberg, Germany, 1999; Volume 1679, pp. 1031–1041. [Google Scholar] [CrossRef]
- Riemann, C. Automated Subretinal Injection: Greater Accuracy, Precision, and Reliability. Retin. Today 2021, 1–2. Available online: https://medone.com/wp-content/uploads/2021/10/2021RT_MedOne_FINAL-PDF-Reprint.pdf (accessed on 10 January 2025).
- Łajczak, P.M.; Nawrat, Z. Sharper Vision, Steady Hands: Can Robots Improve Subretinal Drug Delivery? Systematic Review. J. Robot. Surg. 2024, 18, 235. [Google Scholar] [CrossRef] [PubMed]
- Xue, K.; Edwards, T.L.; Meenink, H.C.M.; Beelen, M.J.; Naus, G.J.L.; Simunovic, M.P.; De Smet, M.D.; MacLaren, R.E. Robot-Assisted Retinal Surgery: Overcoming Human Limitations. In Surgical Retina; Ohji, M., Ed.; Retina Atlas; Springer: Singapore, 2019; pp. 109–114. [Google Scholar] [CrossRef]
- Singh, S.P.N.; Riviere, C.N. Physiological Tremor Amplitude during Retinal Microsurgery. In Proceedings of the IEEE 28th Annual Northeast Bioengineering Conference (IEEE Cat. No.02CH37342), Philadelphia, PA, USA, 21 April 2002; IEEE: New York, NY, USA, 2002; pp. 171–172. [Google Scholar] [CrossRef]
- Parolini, B.; Di Salvatore, A.; Pinackatt, S.J.; Baldi, A.; Besozzi, G.; Finzi, A.; Cardillo, D.; Sallam, A.B.; Frisina, R. Long-term results of autologous retinal pigment epithelium and choroid transplantation for the treatment of exudative and atrophic maculopathies. Retina 2020, 40, 507–520. [Google Scholar] [CrossRef] [PubMed]
- Falkner-Radler, C.I.; Krebs, I.; Glittenberg, C.; Povazay, B.; Drexler, W.; Graf, A.; Binder, S. Human Retinal Pigment Epithelium (RPE) Transplantation: Outcome after Autologous RPE-Choroid Sheet and RPE Cell-Suspension in a Randomised Clinical Study. Br. J. Ophthalmol. 2011, 95, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.A.B.; Stefanini, F.R.; Falabella, P.; Koss, M.J.; Wells, T.; Diniz, B.; Ribeiro, R.; Schor, P.; Maia, M.; Penha, F.M.; et al. Development of a New Tissue Injector for Subretinal Transplantation of Human Embryonic Stem Cell Derived Retinal Pigmented Epithelium. Int. J. Retin. Vitr. 2017, 3, 41. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lim, J.; Ung, F.; Malhotra, S.; Diaz, J.C.; Hamilton, A.; Chen, C.; Tang, W.C.; Seiler, M.J.; Browne, A.W. A Novel, Low-Cost, 3D-Printed Motorized Injector for Retinal Sheet Transplantation. Bioengineering 2026, 13, 188. https://doi.org/10.3390/bioengineering13020188
Lim J, Ung F, Malhotra S, Diaz JC, Hamilton A, Chen C, Tang WC, Seiler MJ, Browne AW. A Novel, Low-Cost, 3D-Printed Motorized Injector for Retinal Sheet Transplantation. Bioengineering. 2026; 13(2):188. https://doi.org/10.3390/bioengineering13020188
Chicago/Turabian StyleLim, Jerald, Francis Ung, Samir Malhotra, Jacob C. Diaz, Austen Hamilton, Clare Chen, William C. Tang, Magdalene J. Seiler, and Andrew W. Browne. 2026. "A Novel, Low-Cost, 3D-Printed Motorized Injector for Retinal Sheet Transplantation" Bioengineering 13, no. 2: 188. https://doi.org/10.3390/bioengineering13020188
APA StyleLim, J., Ung, F., Malhotra, S., Diaz, J. C., Hamilton, A., Chen, C., Tang, W. C., Seiler, M. J., & Browne, A. W. (2026). A Novel, Low-Cost, 3D-Printed Motorized Injector for Retinal Sheet Transplantation. Bioengineering, 13(2), 188. https://doi.org/10.3390/bioengineering13020188







