Integration of Shear-Wave Elastography and Inertial Motion Sensing for Quantitative Monitoring of Tendon Remodeling After Shockwave Therapy in Greater Trochanteric Pain Syndrome
Abstract
1. Introduction
2. Materials and Methods
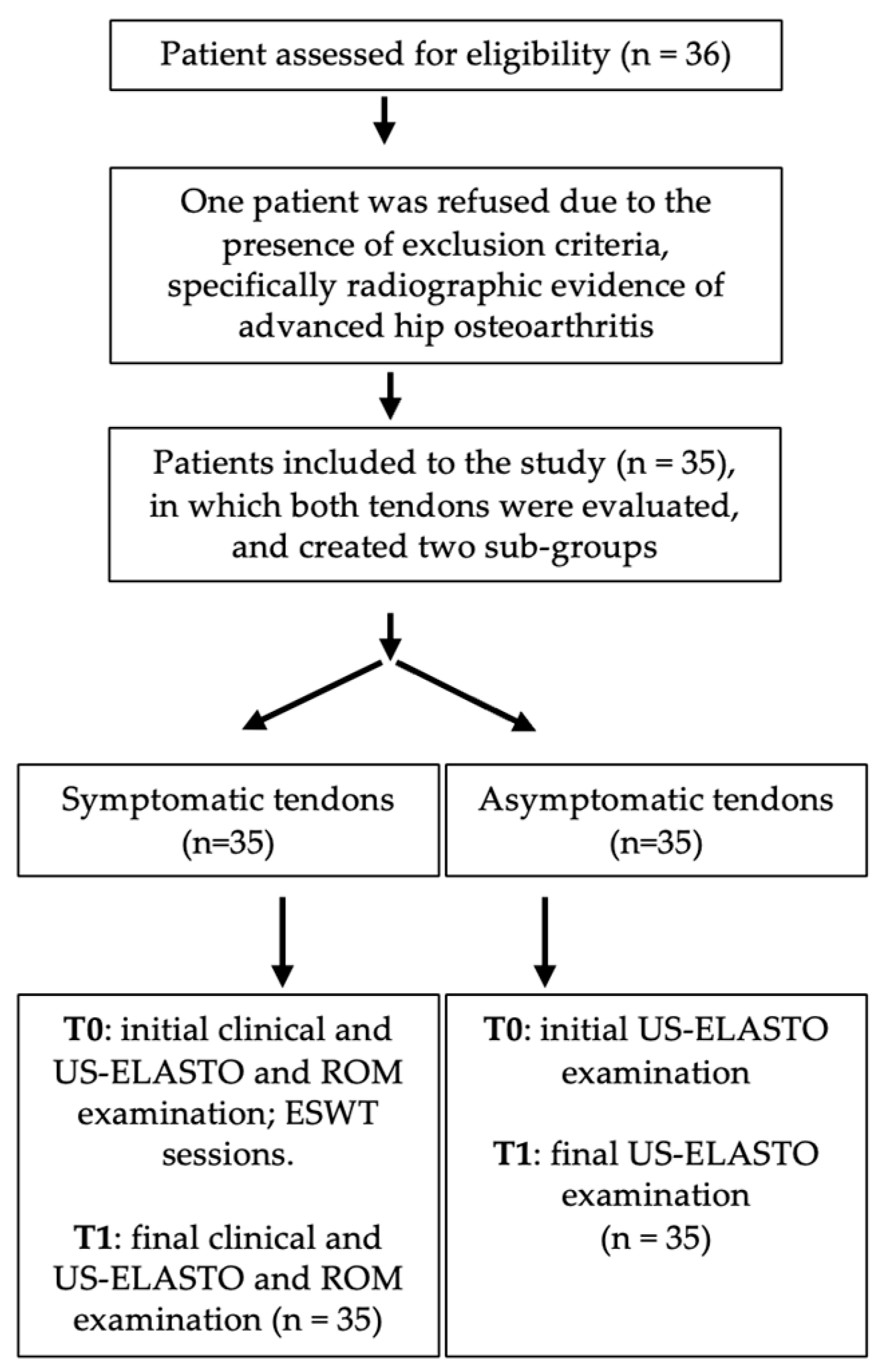
2.1. Study Design and Population
2.2. Intervention
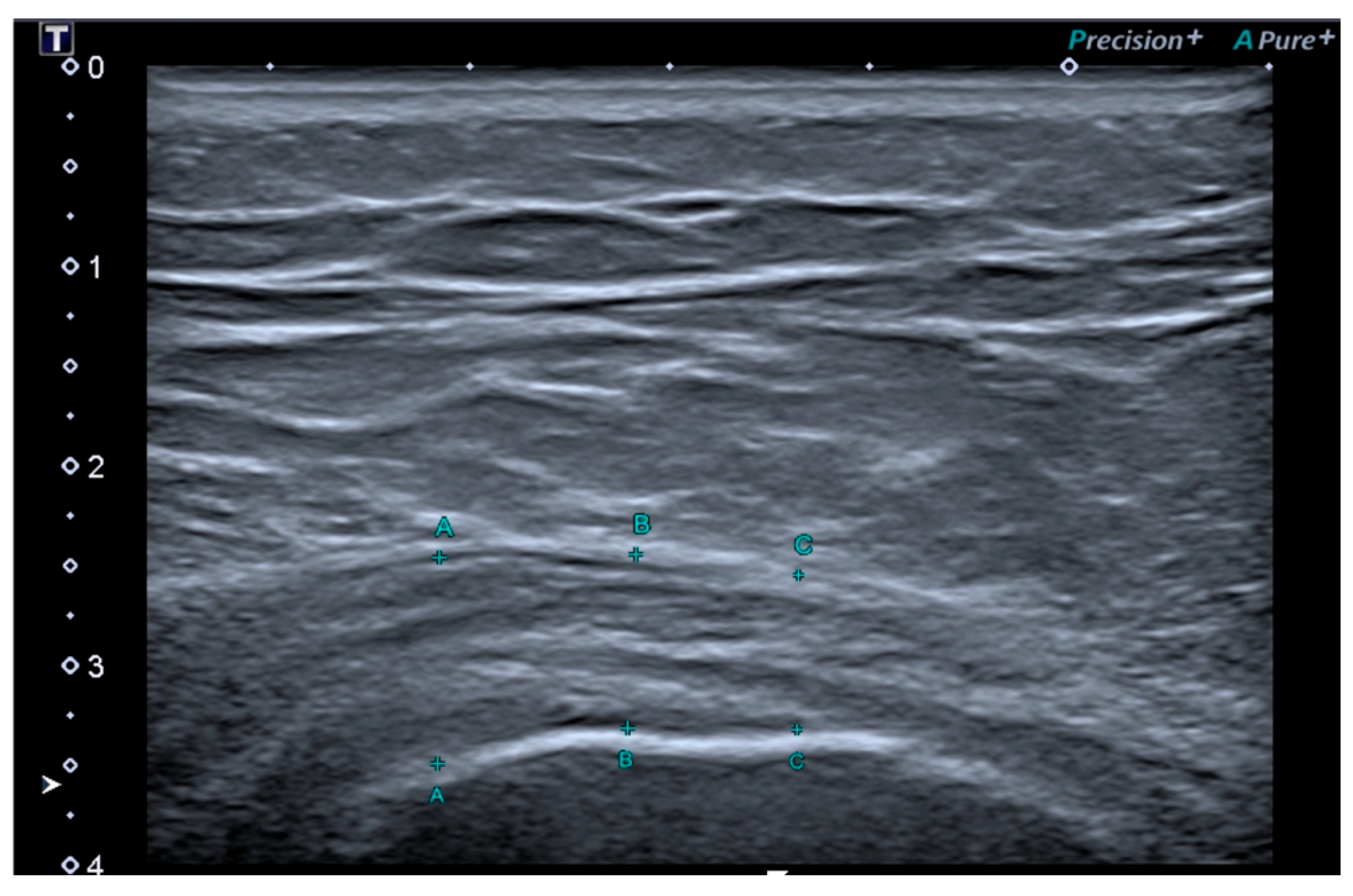
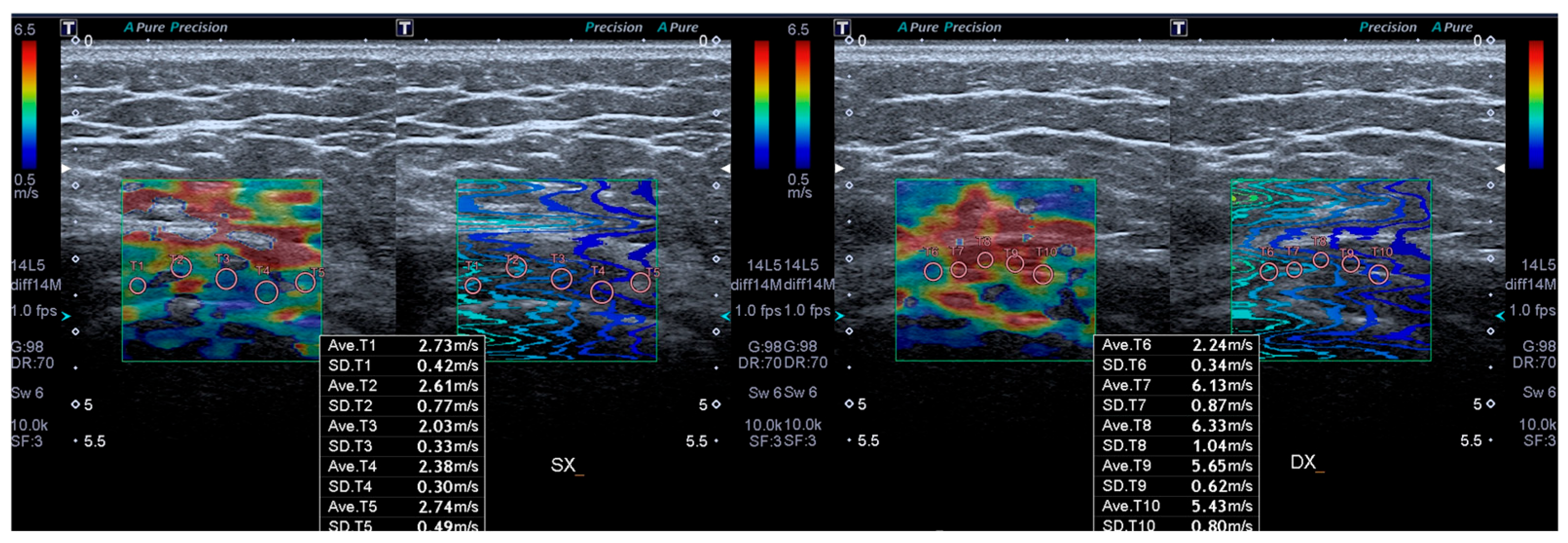
2.3. Outcome Measures
2.4. Statistical Analysis
3. Results
3.1. Patient Demographic and Clinical Characteristics
3.2. Outcome Measures Results
4. Discussion
5. Limitations and Future Perspectives
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Albers, I.S.; Zwerver, J.; Diercks, R.L.; Dekker, J.H.; Van den Akker-Scheek, I. Incidence and prevalence of lower extremity tendinopathy in a Dutch general practice population: A cross sectional study. BMC Musculoskelet. Disord. 2016, 17, 16. [Google Scholar] [CrossRef]
- Riel, H.; Lindstrøm, C.F.; Rathleff, M.S.; Jensen, M.B.; Olesen, J.L. Prevalence and incidence rate of lower-extremity tendinopathies in a Danish general practice: A registry-based study. BMC Musculoskelet. Disord. 2019, 20, 239. [Google Scholar] [CrossRef] [PubMed]
- Fearon, A.M.; Cook, J.L.; Scarvell, J.M.; Neeman, T.; Cormick, W.; Smith, P.N. Greater trochanteric pain syndrome negatively affects work, physical activity and quality of life: A case control study. J. Arthroplast. 2014, 29, 383–386. [Google Scholar] [CrossRef] [PubMed]
- Allison, K.; Vicenzino, B.; Wrigley, T.V.; Grimaldi, A.; Hodges, P.W.; Bennell, K.L. Hip Abductor Muscle Weakness in Individuals with Gluteal Tendinopathy. Med. Sci. Sports Exerc. 2016, 48, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Fearon, A.; Neeman, T.; Smith, P.; Scarvell, J.; Cook, J. Pain, not structural impairments may explain activity limitations in people with gluteal tendinopathy or hip osteoarthritis: A cross sectional study. Gait Posture 2017, 52, 237–243. [Google Scholar] [CrossRef]
- Plinsinga, M.; Coombes, B.; Mellor, R.; Vicenzino, B. The multimodal nature of persistent greater trochanteric pain syndrome. J. Sci. Med. Sport 2019, 22, S24. [Google Scholar] [CrossRef]
- Connell, D.A.; Bass, C.; Sykes, C.J.; Young, D.; Edwards, E. Sonographic evaluation of gluteus medius and minimus tendinopathy. Eur. Radiol. 2003, 13, 1339–1347. [Google Scholar] [CrossRef]
- Fearon, A.M.; Scarvell, J.M.; Cook, J.L.; Smith, P.N. Does ultrasound correlate with surgical or histologic findings in greater trochanteric pain syndrome? A pilot study. Clin. Orthop. Relat. Res. 2010, 468, 1838–1844. [Google Scholar] [CrossRef]
- Kingzett-Taylor, A.; Tirman, P.F.; Feller, J.; McGann, W.; Prieto, V.; Wischer, T.; Cameron, J.A.; Cvitanic, O.; Genant, H.K. Tendinosis and tears of gluteus medius and minimus muscles as a cause of hip pain: MR imaging findings. AJR Am. J. Roentgenol. 1999, 173, 1123–1126. [Google Scholar] [CrossRef]
- Kong, A.; Van der Vliet, A.; Zadow, S. MRI and US of gluteal tendinopathy in greater trochanteric pain syndrome. Eur. Radiol. 2007, 17, 1772–1783. [Google Scholar] [CrossRef]
- Fearon, A.; Scarvell, J.; Cook, J.; Neeman, T.; Smith, P. Greater trochanteric pain syndrome is as painful and functionally debilitating as severe OA of the hip, a prospective case control study. Orthop. Proc. 2012, 94-B, 127. [Google Scholar]
- Lievense, A.; Bierma-Zeinstra, S.; Schouten, B.; Bohnen, A.; Verhaar, J.; Koes, B. Prognosis of trochanteric pain in primary care. Br. J. Gen. Pract. 2005, 55, 199–204. [Google Scholar] [PubMed]
- Grimaldi, A.; Fearon, A. Gluteal Tendinopathy: Integrating Pathomechanics and Clinical Features in Its Management. J. Orthop. Sports Phys. Ther. 2015, 45, 910–922. [Google Scholar] [CrossRef] [PubMed]
- Mellor, R.; Grimaldi, A.; Wajswelner, H.; Hodges, P.; Abbott, J.H.; Bennell, K.; Vicenzino, B. Exercise and load modification versus corticosteroid injection versus ‘wait and see’ for persistent gluteus medius/minimus tendinopathy (the LEAP trial): A protocol for a randomised clinical trial. BMC Musculoskelet. Disord. 2016, 17, 196. [Google Scholar] [CrossRef]
- Ferreira, V.M.L.M.; Pinto, R.Z.; Simoneau, G.; Pogetti, L.S.; Resende, R.A.; Ocarino, J.M. Achilles tendinopathy physical impairments evaluated through clinician-friendly measures: A systematic review with meta-analysis and GRADE recommendations. Braz. J. Phys. Ther. 2025, 29, 101212. [Google Scholar] [CrossRef]
- Feng, R.; Best, T.M.; Wang, L.; Gao, W.; Liu, H.; Yu, B. Knee Movement Characteristics of Basketball Players in Landing Tasks Before Onset of Patellar Tendinopathy: A Prospective Study. Front. Sports Act. Living 2022, 4, 847945. [Google Scholar] [CrossRef]
- Allison, K.; Wrigley, T.V.; Vicenzino, B.; Bennell, K.L.; Grimaldi, A.; Hodges, P.W. Kinematics and kinetics during walking in individuals with gluteal tendinopathy. Clin. Biomech. 2016, 32, 56–63. [Google Scholar] [CrossRef]
- Hilligsøe, M.; Rathleff, M.S.; Olesen, J.L. Ultrasound Definitions and Findings in Greater Trochanteric Pain Syndrome: A Systematic Review. Ultrasound Med. Biol. 2020, 46, 1584–1598. [Google Scholar] [CrossRef]
- Brinks, A.; van Rijn, R.M.; Willemsen, S.P.; Bohnen, A.M.; Verhaar, J.A.; Koes, B.W.; Bierma-Zeinstra, S.M. Corticosteroid injections for greater trochanteric pain syndrome: A randomized controlled trial in primary care. Ann. Fam. Med. 2011, 9, 226–234. [Google Scholar] [CrossRef]
- Fitzpatrick, J.; Bulsara, M.K.; O’Donnell, J.; McCrory, P.R.; Zheng, M.H. The Effectiveness of Platelet-Rich Plasma Injections in Gluteal Tendinopathy: A Randomized, Double-Blind Controlled Trial Comparing a Single Platelet-Rich Plasma Injection with a Single Corticosteroid Injection. Am. J. Sports Med. 2018, 46, 933–939. [Google Scholar] [CrossRef]
- Ramon, S.; Russo, S.; Santoboni, F.; Lucenteforte, G.; Di Luise, C.; de Unzurrunzaga, R.; Vetrano, M.; Albano, M.; Baldini, R.; Cugat, R.; et al. Focused Shockwave Treatment for Greater Trochanteric Pain Syndrome: A Multicenter, Randomized, Controlled Clinical Trial. J. Bone Jt. Surg. 2020, 102, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhang, S.-X.; Yang, Q.; Li, B.-L.; Meng, Q.-G.; Guo, Z.-G. Effect of extracorporeal shock-wave therapy for treating patients with chronic rotator cuff tendonitis. Medicine 2017, 96, e7940. [Google Scholar] [CrossRef] [PubMed]
- Testa, G.; Vescio, A.; Perez, S.; Consoli, A.; Costarella, L.; Sessa, G.; Pavone, V. Extracorporeal Shockwave Therapy Treatment in Upper Limb Diseases: A Systematic Review. J. Clin. Med. 2020, 9, 453. [Google Scholar] [CrossRef] [PubMed]
- Delia, C.; Santilli, G.; Colonna, V.; Di Stasi, V.; Latini, E.; Ciccarelli, A.; Taurone, S.; Franchitto, A.; Santoboni, F.; Trischitta, D.; et al. Focal Versus Combined Focal Plus Radial Extracorporeal Shockwave Therapy in Lateral Elbow Tendinopathy: A Retrospective Study. J. Funct. Morphol. Kinesiol. 2024, 9, 201. [Google Scholar] [CrossRef]
- Santilli, G.; Ioppolo, F.; Mangone, M.; Agostini, F.; Bernetti, A.; Forleo, S.; Cazzolla, S.; Mannino, A.C.; Fricano, A.; Franchitto, A.; et al. High Versus Low-Energy Extracorporeal Shockwave Therapy for Chronic Lateral Epicondylitis: A Retrospective Study. J. Funct. Morphol. Kinesiol. 2024, 9, 173. [Google Scholar] [CrossRef]
- Galasso, O.; Amelio, E.; Riccelli, D.A.; Gasparini, G. Short-term outcomes of extracorporeal shock wave therapy for the treatment of chronic non-calcific tendinopathy of the supraspinatus: A double-blind, randomized, placebo-controlled trial. BMC Musculoskelet. Disord. 2012, 13, 86. [Google Scholar] [CrossRef]
- Hutchinson, J.L.; Gusberti, D.; Saab, G. Changing appearance of intraosseous calcific tendinitis in the shoulder with time: A case report. Radiol. Case Rep. 2019, 14, 1267–1271. [Google Scholar] [CrossRef]
- Heller, G.Z.; Manuguerra, M.; Chow, R. How to analyze the Visual Analogue Scale: Myths, truths and clinical relevance. Scand. J. Pain 2016, 13, 67–75. [Google Scholar] [CrossRef]
- Harris, W.H. Traumatic arthritis of the hip after dislocation and acetabular fractures: Treatment by mold arthroplasty. An end-result study using a new method of result evaluation. J. Bone Jt. Surg. Am. 1969, 51, 737–755. [Google Scholar] [CrossRef] [PubMed]
- Chow, I.H.W.; Cheing, G.L. Comparison of different energy densities of extracorporeal shock wave therapy (ESWT) for the management of chronic heel pain. Clin. Rehabil. 2007, 21, 131–141. [Google Scholar] [CrossRef]
- Rompe, J.D.; Kirkpatrick, C.J.; Küllmer, K.; Schwitalle, M.; Krischek, O. Dose-related effects of shock waves on rabbit tendo Achillis. A sonographic and histological study. J. Bone Jt. Surg. Br. 1998, 80, 546–552. [Google Scholar] [CrossRef]
- Cacchio, A.; De Blasis, E.; Necozione, S.; Rosa, F.; Riddle, D.L.; di Orio, F.; De Blasis, D.; Santilli, V. The Italian version of the lower extremity functional scale was reliable, valid, and responsive. J. Clin. Epidemiol. 2010, 63, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.I.; Donatelli, R.A.; Schmitz, C.; Hellman, M.A.; Buxbaum, F. Chronic Plantar Fasciitis Treated with Two Sessions of Radial Extracorporeal Shock Wave Therapy. Foot Ankle Int. 2010, 31, 391–397. [Google Scholar] [CrossRef]
- Ebert, J.R.; Retheesh, T.; Mutreja, R.; Janes, G.C. The Clinical, Functional and Biomechanical Presentation of Patients with Symptomatic Hip Abductor Tendon Tears. Int. J. Sports Phys. Ther. 2016, 11, 725–737. [Google Scholar] [PubMed]
- Yelverton, C.; Rama, S.; Zipfel, B. Manual therapy interventions in the treatment of plantar fasciitis: A comparison of three approaches. Health SA 2019, 24, 1244. [Google Scholar] [CrossRef]
- Cerfoglio, S.; Capodaglio, P.; Rossi, P.; Conforti, I.; D’Angeli, V.; Milani, E.; Galli, M.; Cimolin, V. Evaluation of Upper Body and Lower Limbs Kinematics through an IMU-Based Medical System: A Comparative Study with the Optoelectronic System. Sensors 2023, 23, 6156. [Google Scholar] [CrossRef]
- Whittaker, J.L.; Emery, C.A. Sonographic measures of the gluteus medius, gluteus minimus, and vastus medialis muscles. J. Orthop. Sports Phys. Ther. 2014, 44, 627–632. [Google Scholar] [CrossRef]
- Mifsud, T.; Gatt, A.; Micallef-Stafrace, K.; Chockalingam, N.; Padhiar, N. Elastography in the assessment of the Achilles tendon: A systematic review of measurement properties. J. Foot Ankle Res. 2023, 16, 23. [Google Scholar] [CrossRef]
- Fitzpatrick, J.; Bulsara, M.K.; O’Donnell, J.; Zheng, M.H. Leucocyte-Rich Platelet-Rich Plasma Treatment of Gluteus Medius and Minimus Tendinopathy: A Double-Blind Randomized Controlled Trial with 2-Year Follow-up. Am. J. Sports Med. 2019, 47, 1130–1137. [Google Scholar] [CrossRef]
- Ratner, B. The correlation coefficient: Its values range between +1/−1, or do they? J. Target. Meas. Anal. Mark. 2009, 17, 139–142. [Google Scholar] [CrossRef]
- Chen, X.-M.; Cui, L.-G.; He, P.; Shen, W.-W.; Qian, Y.-J.; Wang, J.-R. Shear Wave Elastographic Characterization of Normal and Torn Achilles Tendons: A pilot study. J. Ultrasound Med. 2013, 32, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Hackett, L.; Aveledo, R.; Lam, P.H.; Murrell, G.A. Reliability of shear wave elastography ultrasound to assess the supraspinatus tendon: An intra and inter-rater in vivo study. Shoulder Elb. 2020, 12, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Torgutalp, Ş.Ş.; Babayeva, N.; Taş, S.; Dönmez, G.; Korkusuz, F. Effects of hyperlipidemia on patellar tendon stiffness: A shear wave elastography study. Clin. Biomech. 2020, 75, 104998. [Google Scholar] [CrossRef] [PubMed]
- Ito, N.; Sigurðsson, H.B.; Pohlig, R.T.; Cortes, D.H.; Grävare Silbernagel, K.; Sprague, A.L. Reliability of Continuous Shear Wave Elastography in the Pathological Patellar Tendon. J. Ultrasound Med. 2023, 42, 1047–1055. [Google Scholar] [CrossRef]
- Vetrano, M.; d’Alessandro, F.; Torrisi, M.R.; Ferretti, A.; Vulpiani, M.C.; Visco, V. Extracorporeal shock wave therapy promotes cell proliferation and collagen synthesis of primary cultured human tenocytes. Knee Surg. Sports Traumatol. Arthrosc. 2011, 19, 2159–2168. [Google Scholar] [CrossRef]
- Wang, C.-J.; Huang, H.-Y.; Pai, C.-H. Shock wave-enhanced neovascularization at the tendon-bone junction: An experiment in dogs. J. Foot Ankle Surg. 2002, 41, 16–22. [Google Scholar] [CrossRef]
- Wang, F.S.; Yang, K.D.; Chen, R.F.; Wang, C.J.; Sheen-Chen, S.M. Extracorporeal shock wave promotes growth and differentiation of bone-marrow stromal cells towards osteoprogenitors associated with induction of TGF-β1. J. Bone Jt. Surg. Br. 2002, 84, 457–461. [Google Scholar] [CrossRef]
- Kobsar, D.; Charlton, J.M.; Tse, C.; Esculier, J.-F.; Graffos, A.; Krowchuk, N.M.; Thatcher, D.; Hunt, M.A. Validity and reliability of wearable inertial sensors in healthy adult walking: A systematic review and meta-analysis. J. NeuroEngineering Rehabil. 2020, 17, 62. [Google Scholar] [CrossRef]
- Lin, C.-L.; Yang, M.-T.; Lee, Y.-H.; Chen, Y.-W.; Vitoonpong, T.; Huang, S.-W. Comparison of Clinical and Ultrasound Imaging Outcomes Between Corticosteroid and Hypertonic Dextrose Injections for Chronic Supraspinatus Tendinopathy. Orthop. J. Sports Med. 2022, 10, 23259671221129603. [Google Scholar] [CrossRef]
- Liu, C.-P.; Lu, T.-Y.; Wang, H.-C.; Chang, C.-Y.; Hsieh, C.-Y.; Chan, C.-T. Inertial Measurement Unit-Based Frozen Shoulder Identification from Daily Shoulder Tasks Using Machine Learning Approaches. Sensors 2024, 24, 6656. [Google Scholar] [CrossRef]
- Hassan, S.M.; Hafez, A.R.; Seif, H.E.; Kachanathu, S.J. The effect of deep friction massage versus stretching of wrist extensor muscles in the treatment of patients with tennis elbow. Open J. Ther. Rehabil. 2016, 4, 48–54. [Google Scholar] [CrossRef]
- Kanwal, R.; Khan, J.; Awan, W.A.; Khan, R.; Malik, S. Stretching exercises versus deep friction massage for the management of piriformis syndrome. Rehabil. J. 2018, 2, 65–69. [Google Scholar] [CrossRef]
- Abbas, S.; Riaz, R.; Khan, A.; Javed, A.; Raza, S. Effects of mulligan and cyriax approach in patients with subacute lateral epicondylitis. Rehabil. J. 2019, 3, 107–115. [Google Scholar] [CrossRef]
- Mellor, R.; Bennell, K.; Grimaldi, A.; Nicolson, P.; Kasza, J.; Hodges, P.; Wajswelner, H.; Vicenzino, B. Education plus exercise versus corticosteroid injection use versus a wait and see approach on global outcome and pain from gluteal tendinopathy: Prospective, single blinded, randomised clinical trial. BMJ 2018, 361, k1662. [Google Scholar] [CrossRef] [PubMed]
- Sigurðsson, H.B.; Couppé, C.; Silbernagel, K.G. Data driven model of midportion achilles tendinopathy health created with factor analysis. BMC Musculoskelet. Disord. 2022, 23, 744. [Google Scholar] [CrossRef]
- Murphy, M.C.; Debenham, J.; Bulsara, C.; Chivers, P.; Rio, E.K.; Docking, S.; Travers, M.; Gibson, W. Assessment and monitoring of Achilles tendinopathy in clinical practice: A qualitative descriptive exploration of the barriers clinicians face. BMJ Open Sport Exerc. Med. 2022, 8, e001355. [Google Scholar] [CrossRef]
- Macdermid, J.C.; Silbernagel, K.G. Outcome Evaluation in Tendinopathy: Foundations of Assessment and a Summary of Selected Measures. J. Orthop. Sports Phys. Ther. 2015, 45, 950–964. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, Z.; Li, X.; Lei, Y.; Omalley, O. Advancing Biomedical Engineering Through a Multi-Modal Sensor Fusion System for Enhanced Physical Training. AIMS Bioeng. 2023, 10, 364–383. [Google Scholar] [CrossRef]
- Ahn, K.-S.; Kang, C.H.; Hong, S.-J.; Jeong, W.-K. Ultrasound Elastography of Lateral Epicondylosis: Clinical Feasibility of Quantitative Elastographic Measurements. Am. J. Roentgenol. 2014, 202, 1094–1099. [Google Scholar] [CrossRef]
- Hall, T.J. AAPM/RSNA Physics Tutorial for Residents: Topics in US: Beyond the basics: Elasticity imaging with US. Radiographics 2003, 23, 1657–1671. [Google Scholar] [CrossRef]
- Elsayed, M.; Hafez, M.R.M.; Ibrahim, M.A.H. Ultrasound with shear wave elastography in diagnosis and follow-up of common extensor tendinopathy in cases with lateral epicondylitis: A cross-sectional analytic study. Egypt. J. Radiol. Nucl. Med. 2022, 53, 236. [Google Scholar] [CrossRef]
- Alanen, A.; Räisänen, A.; Benson, L.; Pasanen, K. The use of inertial measurement units for analyzing change of direction movement in sports: A scoping review. Int. J. Sports Sci. Coach. 2021, 16, 1332–1353. [Google Scholar] [CrossRef]
- Parvin, N.; Joo, S.W.; Jung, J.H.; Mandal, T.K. Multimodal AI in Biomedicine: Pioneering the Future of Biomaterials, Diagnostics, and Personalized Healthcare. Nanomaterials 2025, 15, 895. [Google Scholar] [CrossRef]
- Santilli, G.; Vetrano, M.; Mangone, M.; Agostini, F.; Bernetti, A.; Coraci, D.; Paoloni, M.; de Sire, A.; Paolucci, T.; Latini, E.; et al. Predictive Prognostic Factors in Non-Calcific Supraspinatus Tendinopathy Treated with Focused Extracorporeal Shock Wave Therapy: An Artificial Neural Network Approach. Life 2024, 14, 681. [Google Scholar] [CrossRef]
- Zhou, J.; Yang, D.-B.; Wang, J.; Li, H.-Z.; Wang, Y.-C. Role of shear wave elastography in the evaluation of the treatment and prognosis of supraspinatus tendinitis. World J. Clin. Cases 2020, 8, 2977–2987. [Google Scholar] [CrossRef]
- Wang, T.; He, Y.; Yi, W.; Xie, H.; Wang, D.; Zeng, J.; Liang, J.; Chen, Y.; Mo, Q.; Zhou, M.; et al. Shear wave elastography of the supraspinatus tendon with early degeneration in asymptomatic type II diabetes mellitus patients: A multicenter study. BMC Musculoskelet. Disord. 2025, 26, 637. [Google Scholar] [CrossRef]
- Santilli, G.; Mangone, M.; Agostini, F.; Paoloni, M.; Bernetti, A.; Diko, A.; Tognolo, L.; Coraci, D.; Vigevano, F.; Vetrano, M.; et al. Evaluation of Rehabilitation Outcomes in Patients with Chronic Neurological Health Conditions Using a Machine Learning Approach. J. Funct. Morphol. Kinesiol. 2024, 9, 176. [Google Scholar] [CrossRef]
- Cerasa, A.; Tartarisco, G.; Bruschetta, R.; Ciancarelli, I.; Morone, G.; Calabrò, R.S.; Pioggia, G.; Tonin, P.; Iosa, M. Predicting Outcome in Patients with Brain Injury: Differences between Machine Learning versus Conventional Statistics. Biomedicines 2022, 10, 2267. [Google Scholar] [CrossRef]

| Variable | Value |
|---|---|
| Age (years) | 67.3 ± 9.9 |
| Gender (Male/Female) | 5/30 |
| BMI | 26.4 ± 4.9 |
| HHS T0 | 61.6 ± 10.1 |
| HHS T1 | 78.7 ± 14.1 |
| VAS T0 | 6.4 ± 1 |
| VAS T1 | 3.8 ± 2.1 |
| LEFS T0 | 46.5 ± 10.9 |
| LEFS T1 | 55.3 ± 9.4 |
| RM T0 | 3.2 ± 0.7 |
| RM T1 | 2.1 ± 0.8 |
| Hip Abduction T0 | 33.6 ± 1.8 |
| Hip Abduction T1 | 35.2 ± 2 |
| Thickness affected side T0 | 9.6 ± 1.5 |
| Thickness affected side T1 | 8.6 ± 1.1 |
| Thickness healthy side T0 | 8.1 ± 1 |
| SWEv affected side T0 | 1.8 ± 0.3 |
| SWEv affected side T1 | 3.2 ± 0.8 |
| SWEv healthy side T0 | 4.2 ± 0.7 |
| Symptomatic GMT T0 | Symptomatic GMT T1 | Healthy GMT T0 | p-Value | |
|---|---|---|---|---|
| Gluteus Medius Tendon Thickness | 9.6 ± 1.4 | 8.6 ± 1.1 | 8.1 ± 1 | <0.05 |
| SWEv (m/s) | 1.8 ± 0.3 | 3.2 ± 0.8 | 4.2 ± 0.7 | <0.05 |
| Hip Abduction | VAS Score | Harris Hip Score | Lower Extremity Functional Scale | Roles and Maudsley | |
|---|---|---|---|---|---|
| Baseline T0 | 33.6 ± 1.8 | 6.4 ± 1.4 | 61.6 ± 10.1 | 46.5 ± 10.9 | 2.3 ± 0.6 |
| Follow-up T1 | 35.1 ± 2 | 3.9 ± 2.1 | 78.7 ± 14 | 55.2 ± 9.4 | 1.5 ± 0.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Santilli, G.; Ciccarelli, A.; Agostini, F.; Bernetti, A.; Vetrano, M.; Nusca, S.M.; Latini, E.; Mangone, M.; Taurone, S.; Coraci, D.; et al. Integration of Shear-Wave Elastography and Inertial Motion Sensing for Quantitative Monitoring of Tendon Remodeling After Shockwave Therapy in Greater Trochanteric Pain Syndrome. Bioengineering 2026, 13, 83. https://doi.org/10.3390/bioengineering13010083
Santilli G, Ciccarelli A, Agostini F, Bernetti A, Vetrano M, Nusca SM, Latini E, Mangone M, Taurone S, Coraci D, et al. Integration of Shear-Wave Elastography and Inertial Motion Sensing for Quantitative Monitoring of Tendon Remodeling After Shockwave Therapy in Greater Trochanteric Pain Syndrome. Bioengineering. 2026; 13(1):83. https://doi.org/10.3390/bioengineering13010083
Chicago/Turabian StyleSantilli, Gabriele, Antonello Ciccarelli, Francesco Agostini, Andrea Bernetti, Mario Vetrano, Sveva Maria Nusca, Eleonora Latini, Massimiliano Mangone, Samanta Taurone, Daniele Coraci, and et al. 2026. "Integration of Shear-Wave Elastography and Inertial Motion Sensing for Quantitative Monitoring of Tendon Remodeling After Shockwave Therapy in Greater Trochanteric Pain Syndrome" Bioengineering 13, no. 1: 83. https://doi.org/10.3390/bioengineering13010083
APA StyleSantilli, G., Ciccarelli, A., Agostini, F., Bernetti, A., Vetrano, M., Nusca, S. M., Latini, E., Mangone, M., Taurone, S., Coraci, D., Felzani, G., Paoloni, M., & Santilli, V. (2026). Integration of Shear-Wave Elastography and Inertial Motion Sensing for Quantitative Monitoring of Tendon Remodeling After Shockwave Therapy in Greater Trochanteric Pain Syndrome. Bioengineering, 13(1), 83. https://doi.org/10.3390/bioengineering13010083









