Biocompatibility Issues of Wound Dressings
Abstract
1. Introduction
2. Results and Discussion
2.1. In Vitro Biocompatibility of WDs with Cultured DFs (2D-Model)
2.2. In Vitro Biocompatibility of WDs with BSE “The Dermal Equivalent, ED” (3D Model)
2.3. In Vitro 2D and 3D Models of WD Biocompatibility as a Basis for Rational Choice
3. Materials and Methods
3.1. Test Wound Dressings
3.2. Cell Culture
3.3. Design and Research Methods
3.4. Extraction Method
3.5. Determination of Cell Viability Under 2D Conditions
3.6. Determination of Cell Viability Under 3D Conditions
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zavrazhnov, A.A.; Gvozdev, M.Y.; Krutova, V.A.; Ordokova, A.A. Wounds and Wound Process; KubSMU: Krasnodar, Russia, 2016. [Google Scholar]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef]
- Sen, C.K.; Gordillo, G.M.; Roy, S.; Kirsner, R.; Lambert, L.; Hunt, T.K.; Gottrup, F.; Gurtner, G.C.; Longaker, M.T. Human skin wounds: A major and snowballing threat to public health and the economy. Wound Repair Regen. 2009, 17, 763–771. [Google Scholar] [CrossRef]
- Li, J.; Chen, J.; Kirsner, R. Pathophysiology of acute wound healing. Clin. Dermatol. 2007, 25, 9–18. [Google Scholar] [CrossRef]
- Borbolla-Jiménez, F.V.; Peña-Corona, S.I.; Farah, S.J.; Jiménez-Valdés, M.T.; Pineda-Pérez, E.; Romero-Montero, A.; Del Prado-Audelo, M.L.; Bernal-Chávez, S.A.; Magaña, J.J.; Leyva-Gómez, G. Films for Wound Healing Fabricated Using a Solvent Casting Technique. Pharmaceutics 2023, 15, 1914. [Google Scholar] [CrossRef] [PubMed]
- Rezvani Ghomi, E.; Khalili, S.; Nouri Khorasani, S.; Esmaeely Neisiany, R.; Ramakrishna, S. Wound dressings: Current advances and future directions. J. Appl. Polym. Sci. 2019, 136, 47738. [Google Scholar] [CrossRef]
- Kammona, O.; Tsanaktsidou, E.; Kiparissides, C. Recent Developments in 3D-(Bio)printed Hydrogels as Wound Dressings. Gels 2024, 10, 147. [Google Scholar] [CrossRef]
- Rezvani Ghomi, E.; Niazi, M.; Ramakrishna, S. The evolution of wound dressings: From traditional to smart dressings. Polym. Adv. Technol. 2023, 34, 520–530. [Google Scholar] [CrossRef]
- Kashtanov, A.D.; Vasiliev, Y.U.L.; Bayrashevskaya, A.V. Overview of modern materials used to cover wound surfaces. Russ. J. Oper. Surg. Clin. Anat. 2020, 4, 49–56. [Google Scholar] [CrossRef]
- Mirhaj, M.; Labbaf, S.; Tavakoli, M.; Seifalian, A.M. Emerging treatment strategies in wound care. Int. Wound J. 2022, 19, 1934–1954. [Google Scholar] [CrossRef] [PubMed]
- Brumberg, V.; Astrelina, T.; Malivanova, T.; Samoilov, A. Modern Wound Dressings: Hydrogel Dressings. Biomedicines 2021, 9, 1235. [Google Scholar] [CrossRef] [PubMed]
- Broussard, K.C.; Powers, J.G. Wound dressings: Selecting the most appropriate type. Am. J. Clin. Dermatol. 2013, 14, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wu, W.; Yeo, J.C.C.; Soo, X.Y.D.; Thitsartarn, W.; Liu, S.; Tan, B.H.; Suwardi, A.; Li, Z.; Zhu, Q.; et al. Responsive hydrogel dressings for intelligent wound management. BMEMat 2023, 1, 12021. [Google Scholar] [CrossRef]
- Moradifar, F.; Sepahdoost, N.; Tavakoli, P.; Mirzapoor, A. Multi-functional dressings for recovery and screenable treatment of wounds: A review. Heliyon 2025, 11, e41465. [Google Scholar] [CrossRef] [PubMed]
- Mirhaj, M.; Labbaf, S.; Tavakoli, M.; Seifalian, A. An Overview on the Recent Advances in the Treatment of Infected Wounds: Antibacterial Wound Dressings. Macromol. Biosci. 2022, 22, e2200014. [Google Scholar] [CrossRef]
- Ahmad, N. In Vitro and In Vivo Characterization Methods for Evaluation of Modern Wound Dressings. Pharmaceutics 2022, 15, 42. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, G.K.; Martinez-Rodriguez, S.; Md Fadilah, N.I.; Looi Qi Hao, D.; Markey, G.; Shukla, P.; Fauzi, M.B.; Panetsos, F. Progress in wound-healing products based on natural compounds, stem cells, and microRNA-based biopolymers in the European, USA, and Asian markets: Opportunities, barriers, and regulatory issues. Polymers 2024, 16, 1280. [Google Scholar] [CrossRef]
- Laurano, R.; Boffito, M.; Ciardelli, G.; Chiono, V. Wound dressing products: A translational investigation from the bench to the market. Eng. Regen. 2022, 3, 182–200. [Google Scholar] [CrossRef]
- Heras, K.L.; Igartua, M.; Santos-Vizcaino, E.; Hernandez, R.M. Cell-based dressings: A journey through chronic wound management. Biomater. Adv. 2022, 135, 212738. [Google Scholar] [CrossRef]
- Alberts, A.; Tudorache, D.-I.; Niculescu, A.-G.; Grumezescu, A.M. Advancements in Wound Dressing Materials: Highlighting Recent Progress in Hydrogels, Foams, and Antimicrobial Dressings. Gels 2025, 11, 123. [Google Scholar] [CrossRef]
- Blinova, M.I.; Yudintseva, N.M.; Alexandrova, O.I.; Balluzek, M.F.; Khabarova, I.G.; Markin, S.M.; Chagunava, O.L. Clinical experience of healing trophic ulcers using the cellular product “The Dermal Equivalent ED”. Health—Basis Hum. Potential Probl. Solut. 2015, 10, 690–695. [Google Scholar]
- Blinova, M.I.; Yudintseva, N.M.; Alexander-Sinclair, E.I. Equivalent Dermal (DE). Development and Application Practice; Publishing and Printing Association of Higher Educational Institutions: St. Petersburg, Russia, 2022. [Google Scholar]
- Guo, W.; Ding, X.; Zhang, H.; Liu, Z.; Han, Y.; Wei, Q.; Okoro, O.V.; Shavandi, A.; Nie, L. Recent Advances of Chitosan-Based Hydrogels for Skin-Wound Dressings. Gels 2024, 10, 175. [Google Scholar] [CrossRef]
- Demeter, M.; Scărișoreanu, A.; Călina, I. State of the Art of Hydrogel Wound Dressings Developed by Ionizing Radiation. Gels 2023, 9, 55. [Google Scholar] [CrossRef]
- Nguyen, H.M.; Ngoc Le, T.T.; Nguyen, A.T.; Thien Le, H.N.; Pham, T.T. Biomedical materials for wound dressing: Recent advances and applications. RSC Adv. 2023, 13, 5509–5528. [Google Scholar] [CrossRef] [PubMed]
- Nasra, S.; Patel, M.; Shukla, H.; Bhatt, M.; Kumar, A. Functional hydrogel-based wound dressings: A review on biocompatibility and therapeutic efficacy. Life Sci. 2023, 334, 122232. [Google Scholar] [CrossRef] [PubMed]
- ISO 10993-5; Biological Evaluation of Medical Devices-Part 5: Tests for In Vitro Cytotoxicity. ISO: Geneve, Switzerland, 2009.
- View. Available online: https://cr.minzdrav.gov.ru/view-cr/687_3 (accessed on 28 April 2025).
- Order of the Ministry of Health of the Russian Federation No. 559n Dated 9 June 2020 “On Approval of the Procedure for Providing Medical Care to the Population in the Field of SURGERY (Kombustiology)” (The Document Has Not Entered Into Force). Available online: https://www.garant.ru/products/ipo/prime/doc/74619234/ (accessed on 9 June 2020).
- Ibrahim, M.A.; Nasr, G.M.; Ahmed, R.M.; Kelany, N.A. Physical characterization, biocompatibility, and antimicrobial activity of polyvinyl alcohol/sodium alginate blend doped with TiO2 nanoparticles for wound dressing applications. Sci. Rep. 2024, 14, 5391. [Google Scholar] [CrossRef] [PubMed]
- HARTMANN Homepage. Available online: https://www.hartmann.info (accessed on 28 April 2025).
- Biotekpharm: Innovative Wound Dressings and Patches. Available online: https://www.voscopran.ru (accessed on 28 April 2025).
- L&R Russia: Lohmann & Rauscher Russia. Available online: https://www.lohmann-rauscher.com (accessed on 28 April 2025).
- Convatec—Pioneering Solutions for Management of Chronic Conditions. Available online: https://convatec.com (accessed on 28 April 2025).
- Napoli—The Official Website of LLC “Napoli”. Available online: http://napoly.info/ru (accessed on 28 April 2025).
- Morshed, K.M.; Jain, S.K.; McMartin, K.E. Propylene glycol-mediated cell injury in a primary culture of human proximal tubule cells. Toxicol. Sci. 1998, 46, 410–417. [Google Scholar] [CrossRef]
- Morshed, K. Acute Toxicity of Propylene Glycol: An Assessment Using Cultured Proximal Tubule Cells of Human Origin. Fundam. Appl. Toxicol. 1994, 23, 38–43. [Google Scholar] [CrossRef]
- Aleksandrova, O.I.; Okolov, I.N.; Khorolskaya, J.I.; Mikhailova, N.A.; Darvish, D.M.; Blinova, M.I. In Vitro Cytotoxicity Screening as a Criterion for the Rational Selection of Tear Substitutes. In Cytotoxicity—Definition, Identification, and Cytotoxic Compounds; Istifli, E.S., Ila, H.B., Eds.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Yang, D.; Chen, H.; Wei, H.; Liu, A.; Wei, D.X.; Chen, J. Hydrogel wound dressings containing bioactive compounds originated from traditional Chinese herbs: A review. Smart Mater. Med. 2024, 5, 153–165. [Google Scholar] [CrossRef]
- Kukhareva, L.V.; Paramonov, B.A.; Shamolina, I.I.; Semenova, E.G. A Method for Obtaining Collagen for the Treatment of Pathologies of Body Tissues. Patent RU 2214827, 27 October 2003. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]

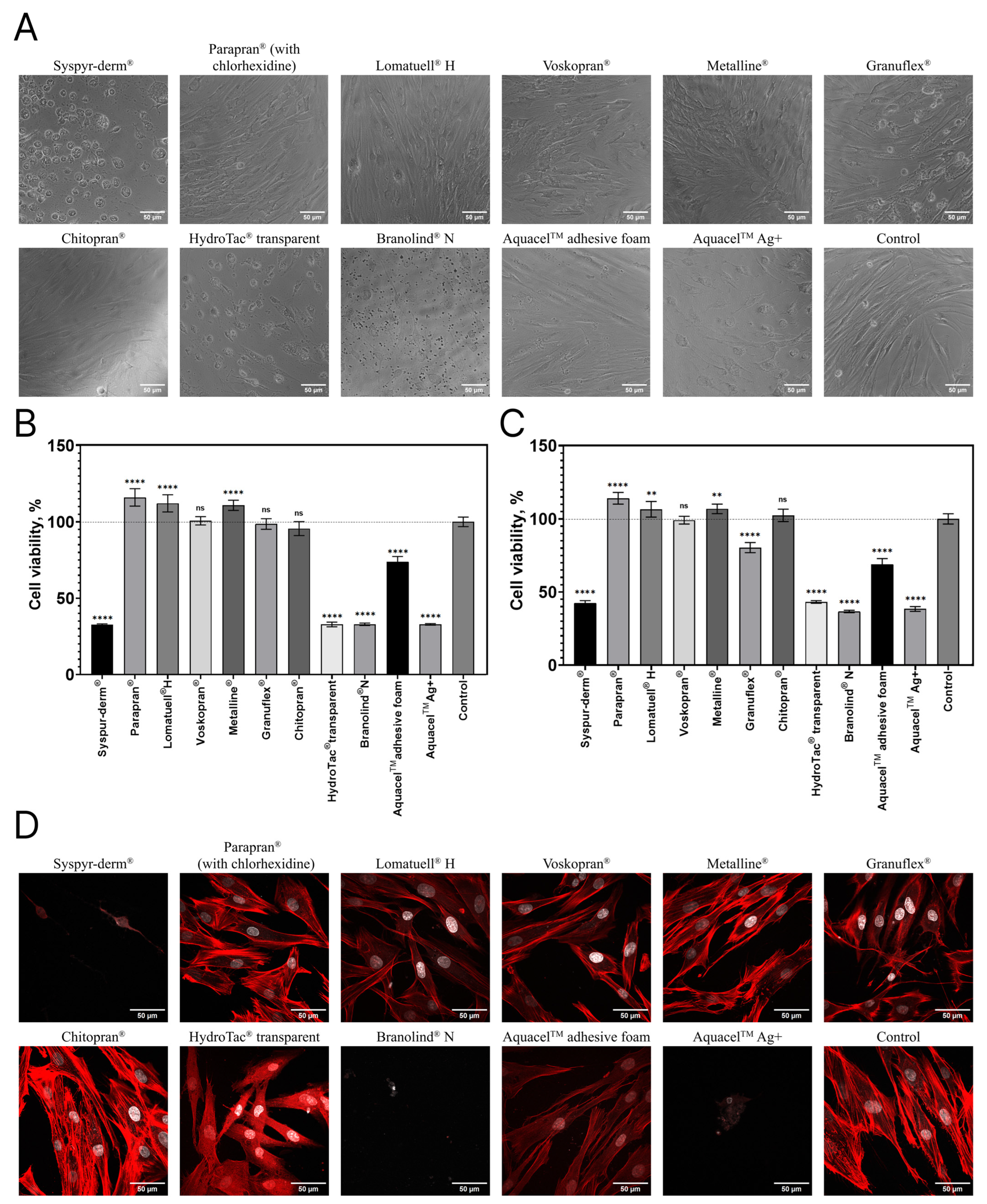


| In Vitro Biocompatibility WDs | ||||||||
|---|---|---|---|---|---|---|---|---|
| High | Requires Clarification | Low | ||||||
| WD | M1, % | M2, % | WD | M1, % | M2, % | WD | M1, % | M2, % |
| Parapran® (with chlorhexidine) | 116.0 ± 5.7 | 114.1 ± 4.0 | Granuflex® | 98.6 ± 3.5 | 80.4 ± 3.5 | Syspyr-derm® | 32.7 ± 0.5 | 42.5 ± 1.7 |
| Lomatuell® H | 112.1 ± 5.6 | 106.6 ± 5.4 | HydroTac® transparent | 32.8 ± 1.5 | 43.2 ± 0.9 | |||
| Voskopran® | 100.7 ± 2.7 | 99.1 ± 2.7 | Branolind® N | 32.9 ± 0.8 | 36.7 ± 0.8 | |||
| Metalline® | 110.8 ± 3.4 | 106.9 ± 3.3 | AquacelTM adhesive foam | 73.9 ± 3.4 | 69.0 ± 3.9 | |||
| Chitopran® | 95.6 ± 4.6 | 102.4 ± 4.2 | AquacelTM Ag+ | 32.9 ± 0.5 | 38.4 ± 1.7 | |||
| WD’s Type | Description | Main Characteristics | Application | Advantages | Disadvantages | ||
|---|---|---|---|---|---|---|---|
| 1 | Syspur-derm® 1 | Hydro-cellular dressings | Double-layer polyurethane sponge material | The wound dressing promotes intensive and effective cleaning of the infected wound surface thanks to the adhesion of wound discharge. The inner layer of the sponge serves as a matrix for the formation of new cell tissues during the formation of granulation tissue in clean wounds. The outer film helps reduce drying of the wound and acts as an external barrier against infection. The outer layer also has a high degree of oxygen permeability to maintain temperature parameters. | Cleansing of wounds of thermal and mechanical origin, stimulation of healing, and formation of new granulation tissue in chronic or acute wounds with a slow recovery process, such as bedsores, radiation injuries, diabetic ulcers, and trophic changes. | Effective and fast wound cleansing thanks to the sorption capacity of the product. Acceleration of the reparative process in tissues and stimulation of granulation tissue formation. Painlessness and pleasant aroma during bandaging. Protection of the wound from secondary infection and drying out. Possibility of modeling the bandage in different planes during wound closure. No maceration or allergic reaction around the skin. | Monitoring of the wound dressing should be performed daily. If any fluid accumulation or signs of infection are noted, the dressing should be changed. To ensure complete cleaning of the wound, it is recommended to replace the compress every 12–24 h. |
| 2 | Parapran® 2 | Knitted mesh made of 100% cotton, paraffin, chlorhexidine bigluconate | Antimicrobial atraumatic dressing with chlorhexidine is a local therapeutic and prophylactic antiseptic that is applied to wounds. The dressing contains chlorhexidine in a paraffin composition that gradually releases the antiseptic over time, providing a long-lasting antimicrobial effect on the wound. | Purulent and purulent-necrotic wounds of various etiologies: burns, bedsores, trophic ulcers, frostbite, purulent-inflammatory diseases of soft tissues, abscesses, and phlegmon. | Atraumatic and painless bandaging. Effective prevention of secondary infections. Creation of a humid environment. The ability to adjust the bandage in different directions. | Individual intolerance to the constituent components. | |
| 3 | Lomatuell® H 3 | Atraumatic | Cotton bandage with paraffin impregnation | A sterile, coarse-mesh cotton bandage that is impregnated with a neutral, water-repellent (hydrophobic) ointment. The mesh structure allows for free absorption of any secretions from the wound and directs them to a secondary, absorbable bandage. | First and second degree burns, superficial wounds, abrasions, cracks, radiation-induced wounds, trophic ulcers on the shin, and donor and recipient sites during transplantation. | The wound dressing provides good ventilation for the wound and has analgesic, antibacterial, and wound healing effects. It can be used in conjunction with any antibiotics or antiseptic medications. | Daily dressings. Individual intolerance to the constituent components. |
| 4 | Voskopran® 2 | Anti-inflammatory ointment dressing | Knitted polyester mesh, beeswax, propolis, vitamin E, Levomekol ointment (dioxomethyltetrahydropyrimidine, chloramphenicol) | A combined drug that contains the antibiotic chloramphenicol and the immunostimulating agent methyluracil. This medication provides an anti-inflammatory and anti-microbial effect on the wound. Due to its polyethylene glycol base, “Voskopran” ointment has a long-lasting absorption effect. | Wounds with signs of infection, including purulent wounds and trophic ulcers. Bedsores, diabetic foot ulcers, and wounds from prolonged pressure “crash” syndrome. Gangrene, purulent inflammatory skin diseases, such as boils and phlegmons. Burns of I-III degrees, lacerations, and animal and insect bites. Gunshot wounds and dermatitis. | High antimicrobial activity not only against aerobic Gram-positive and Gram-negative bacteria, but also against anaerobic microorganisms. Long-lasting sorption effect. Optimization of the processes of wound repair. | Individual intolerance to the constituent components. |
| 5 | Metalline® 3 | Sorbing | The wound layer is a non-woven viscose material coated with aluminum. The absorbent layer is non–woven fabric. The outer layer is a thin, breathable non-woven fabric | It consists of three layers. The outer layer is made of a non-woven fabric. The middle layer is made from a fibrous, absorbent non-woven material. And the inner layer, which is in contact with the wound, is made from thin, perforated aluminum foil. This ensures the atraumatic properties of the medication. | This product is intended for use in various medical fields, including traumatology, surgery, dermatology, and phlebology. It is especially suitable for the treatment of extensive wounds, abrasions, and burns. It can also be used as a first aid bandage and for securing drains and tracheostomy tubes. | It does not stick to the wound surface. A painless bandage change. Gentle wound care. Air and vapor permeability. Soft and plastic structure. Optimal drainage capacity. It is waterproof, (the patient can take a bath or shower with a bandage applied.) | Individual intolerance to the constituent components. |
| 6 | Granuflex® 4 | Hydrocolloid | A hydrocolloid dressing consists of an inner layer that is in contact with the wound (gelatin, pectin, sodium carboxymethylcellulose) and an outer layer (polyurethane film) | A wound dressing consists of an inner layer in contact with the wound, which is made of hydrocolloids, an intermediate layer of polyurethane foam, and an outer layer made of polyurethane film. The wound dressing interacts with wound exudate to create a moist environment, which supports the healing process. It also absorbs exudate and promotes autolytic cleansing of the wound. | Wound dressing is used to treat chronic, exudating wounds such as leg ulcers, pressure sores, minor burns, donor sites (after hemostasis has been achieved) and other types of wounds that are granulating. When applied to wounds with dry, peeling, or necrotic tissue, the dressing helps prevent the loss of moisture from the skin’s surface. This helps rehydrate dead tissue, which can then be removed by autolysis. | Adhesive foam with a thin layer of hydrocolloid extends beyond the central hydrocolloid mass, creating a border with low-profile edges for added safety in hard-to-reach areas. | Individual intolerance to the constituent components. It is important to carefully monitor the condition of the wound and all treatment should be performed under the supervision of a doctor. The use of Granuflex in the presence of an anaerobic infection is not recommended. Improper use or frequent changes in bandages can lead to skin irritation or peeling. |
| 7 | Chitopran® 5 | Bioplastic coating | Biopolymer dressing based on chitosan nanofibers in the form of a non-woven fabric | It is a bioplastic material composed of randomly oriented chitosan fibers with a diameter between 300 and 400 nanometers. In the treatment of patients, wound coverings measuring 10 × 10 cm were used. | Local treatment of superficial granulating, slow-healing, long-lasting wounds in the regenerative stage, I–III degree burns, trophic ulcers, pressure sores, and frostbite. Temporary closure of III degree burn wounds to prepare them for autografting, closure of donor sites. Healing of oral wound surfaces. | It provides the necessary level of moisture, air circulation, and painless removal. It can be easily removed from the wound or absorbed by itself. It can also be used as a gauze, as the sterile Chitopran wound healing bandage (5 × 7.5 cm) is easy to shape. Suitable for sensitive, delicate skin. | Individual intolerance to the constituent components. |
| 8 | HydroTac ® transparent 1 | Hydrogel | A hydrogel dressing made of a polymer based on a hybrid of polyurethane and polyurea containing propylene glycol with a water content of about 60%. | Due to its high water content of about 60%, the HydroTac transparent bandage instantly creates and maintains a moist wound environment. This helps to rapidly reject necrotic masses and stimulates the processes of granulation and epithelialization. | The bandage is suitable for treating weakly exudating and dry, chronic wounds of various origins and any location, without clinical signs of infection, such as trophic venous ulcers, diabetic ulcers, pressure sores, and other poorly healing wounds, at the stages of granulation and epithelialization. It can also be used to treat grade II burns and donor sites. | A hydrogel dressing that can be used to treat dry, chronic, and poorly healing wounds, as well as wounds with low levels of exudate during the epithelialization phase. This dressing helps create and maintain an optimal moist wound environment, providing long-term hydration. It actively stimulates epithelialization and has a firm fit to the wound. The dressing can stay on the wound for up to seven days without the need for bandage changes. | HydroTac transparent dressing should not be used if the patient has a hypersensitivity to any of its ingredients. It should also not be used on clinically infected wounds or third-degree burns. |
| 9 | Branolind® N 1 | Coarse cotton fabric impregnated with an ointment composition: Peruvian balm (benzoic and cinnamic acid, vanillin, etc.), white petroleum jelly, cetomacrogol, glycerin monostearate, hydrogenated fat, medium-chain triglycerides. | The bandage has a soft and flexible structure, which allows it to tightly fit around the damaged area without causing pain or discomfort. The Peruvian balm in the dressing contains strong anti-inflammatory properties and helps accelerate the process of tissue regeneration. The Branolind N ointment with Peruvian balsam dressing provides reliable protection for wounds from infection and improves blood microcirculation. The bandage is made from coarse-mesh cotton fabric, which has a high degree of air permeability and secret permeability. | Abrasions, wounds, ulcers, scuffs and other superficial skin lesions; burns within the skin; donor wounds during skin transplantation; venous trophic ulcers; wounds in the treatment of diabetic foot; wounds with a high risk of scarring contractures. | Atraumatic and painless during bandages. Protection of the wound from secondary infection. Prevention of wound drying out. Possibility of adjusting bandages in different directions when closing wounds. | It is possible to develop dermatitis, contact sensitivity, and photosensitization as well as allergic reactions from Peruvian balsam. It is necessary to avoid contact of the product with the eyes, mucous membranes, and serous membranes. | |
| 10 | AquacelTM adhesive foam 4 | The non-woven layer that comes into contact with the wound surface is made of sodium carboxymethylcellulose. On top of that, there is a layer of polyurethane foam. | It comes into direct contact with the wound and absorbs a large amount of wound exudate and bacteria. It forms a soft gel, which creates the effect of micro-contouring the wound surface, creating and maintaining a moist environment for faster healing. The foam layer absorbs and retains a large amount of wound content. The outer film provides a barrier against viruses and bacteria and is waterproof, protecting the wound from external contamination. The silicone adhesive edge promotes a strong, skin-friendly adhesion, promoting an atraumatic change in the wound coating. Silver has a bactericidal effect on a wide range of bacteria, including antibiotic-resistant hospital strains. | Ulcers of the lower extremities (trophic, diabetic), infected wounds and wounds with an increased risk of infection, pressure sores with partial or complete skin damage to the entire depth, and post-traumatic wounds such as lacerations and abrasions. | The wound dressing absorbs a large amount of wound discharge, including bacteria. It helps remove non-viable tissue from the wound and cleans it, promoting faster healing. Silicone technology provides less trauma to the skin around the wound, and the dressing can remain in place for up to 7 days unless there are clinical indications for removing it. | Individual intolerance to the constituent components. | |
| 11 | AquacelTM Ag+ 4 | A non-woven bandage consists of two layers of sodium carboxymethylcellulose, with 1.2% silver and ethylenediaminetetraacetic acid. It also contains benzetonium chloride and is stitched together with a thread made from regenerated cellulose. | It contains EDTA and benzethonium chloride, thanks to which it is able to remove formed bacterial biofilms and prevent their formation, as well as increase the effectiveness of silver delivery to microorganisms. | Infected wounds or wounds with a high risk of infection, such as trophic ulcers in the lower extremities due to venous insufficiency or ischemia, diabetic foot ulcers, bedsores with partial or complete damage to tissue layers, surgical wounds, traumatic wounds, bleeding wounds after mechanical or surgical treatment, exudating wounds from cancerous skin tumors like fungoid tumors, granulomatous carcinoma, skin metastases, and Kaposi’s sarcoma. Wounds where the presence of bacteria may be a suspected cause of chronic non-healing. | Reduces the severity of pain syndrome when the bandage is applied to the wound; absorbs and retains excess fluid and toxic substances (e.g., bacteria) within the fibers of the bandage, thus reducing the risk of skin maceration and damage around the wound; precisely replicates the features of wound relief, reducing the dead space in the wound (in contact with the therapeutic bandage); and provides controlled release of silver ions as exudate accumulates on the bandage, with rapidly developing and stable antibacterial activity. | Allergic reaction to silver or sodium carboxymethylcellulose. |
| Biocompatibility WDs with BSE “The Dermal Equivalent, ED” in Direct Contact In Vitro | ||
|---|---|---|
| WD | M1, % | M2, % |
| Parapran® | 125.0 ± 6.0 | 97.4 ± 1.0 |
| Metalline® | 127.5 ± 5.6 | 88.9 ± 0.5 |
| Lomatuell®H | 117.7 ± 5.7 | 87.0 ± 1.0 |
| HydroTac® transparent | 98.4 ± 2.6 | 86.8 ± 0.3 |
| Voskopran® | 117.3 ± 1.9 | 84.2 ± 4.9 |
| Chitopran® | 99.2 ± 9.8 | 75.9 ± 1.2 |
| Granuflex® | 105.0 ± 4.9 | 74.8 ± 0.7 |
| Branolind®N | 91.5 ± 1.3 | 63.4 ± 0.6 |
| Syspur-derm® | 97.2 ± 2.3 | 62.1 ± 2.3 |
| AquacelTM adhesive foam | 90.7 ± 3.2 | 56.9 ± 0.8 |
| AquacelTMAg+ | 97.5 ± 2.8 | 55.4 ± 1.2 |
| Biocompatibility WDs In Vitro | ||||
|---|---|---|---|---|
| WD | M1, % | M2, % | M3, % | M4, % |
| Parapran® | 116.0 ± 5.7 | 114.1 ± 4.0 | 125.0 ± 6.0 | 97.4 ± 1.0 |
| Metalline® | 110.8 ± 3.4 | 106.9 ± 3.3 | 127.5 ± 5.6 | 88.9 ± 0.5 |
| Lomatuell®H | 112.1 ± 5.6 | 106.6 ± 5.4 | 117.7 ± 5.7 | 87.0 ± 1.0 |
| HydroTac® transparent | 32.8 ± 1.5 | 43.2 ± 0.9 | 98.4 ± 2.6 | 86.8 ± 0.3 |
| Voskopran® | 100.7 ± 2.7 | 99.1 ± 2.7 | 117.3 ± 1.9 | 84.2 ± 4.9 |
| Chitopran® | 95.6 ± 4.6 | 102.4 ± 4.2 | 99.2 ± 9.8 | 75.9 ± 1.2 |
| Granuflex® | 98.6 ± 3.5 | 80.4 ± 3.5 | 105.0 ± 4.9 | 74.8 ± 0.7 |
| Branolind®N | 32.9 ± 0.8 | 36.7 ± 0.8 | 91.5 ± 1.3 | 63.4 ± 0.6 |
| Syspur-derm® | 32.7 ± 0.5 | 42.5 ± 1.7 | 97.2 ± 2.3 | 62.1 ± 2.3 |
| AquacelTM adhesive foam | 73.9 ± 3.4 | 69.0 ± 3.9 | 90.7 ± 3.2 | 56.9 ± 0.8 |
| AquacelTMAg+ | 32.9 ± 0.5 | 38.4 ± 1.7 | 97.5 ± 2.8 | 55.4 ± 1.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alexander-Sinclair, E.I.; Lapina, E.S.; Edomenko, N.V.; Kostyakov, D.V.; Zinoviev, E.V.; Blinova, M.I.; Mikhailova, N.A. Biocompatibility Issues of Wound Dressings. Bioengineering 2025, 12, 1196. https://doi.org/10.3390/bioengineering12111196
Alexander-Sinclair EI, Lapina ES, Edomenko NV, Kostyakov DV, Zinoviev EV, Blinova MI, Mikhailova NA. Biocompatibility Issues of Wound Dressings. Bioengineering. 2025; 12(11):1196. https://doi.org/10.3390/bioengineering12111196
Chicago/Turabian StyleAlexander-Sinclair, Elga I., Ekaterina S. Lapina, Nikita V. Edomenko, Denis V. Kostyakov, Evgeniy V. Zinoviev, Miralda I. Blinova, and Natalia A. Mikhailova. 2025. "Biocompatibility Issues of Wound Dressings" Bioengineering 12, no. 11: 1196. https://doi.org/10.3390/bioengineering12111196
APA StyleAlexander-Sinclair, E. I., Lapina, E. S., Edomenko, N. V., Kostyakov, D. V., Zinoviev, E. V., Blinova, M. I., & Mikhailova, N. A. (2025). Biocompatibility Issues of Wound Dressings. Bioengineering, 12(11), 1196. https://doi.org/10.3390/bioengineering12111196







