Radiomics-Based Machine Learning Model for Predicting Overall and Progression-Free Survival in Rare Cancer: A Case Study for Primary CNS Lymphoma Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Dataset Description
2.2. Image Pre-Processing
- for each patient, all available MRI acquisitions were registered on the T1-gd image (sequence where segmentation was performed);
- normalization methods were applied for MRI intensities normalization (described in detail in Section 2.2.1);
- all sequences were resampled (voxels 1 mm) [44].
2.2.1. Intensity Normalization of MR Images
2.3. Segmentation VOI (Volume of Interest) and Features Extraction
2.4. Machine Learning Model Building
2.5. Experiments
2.5.1. Feature Robustness
2.5.2. Overall and Progression Free Survival Prediction
3. Results
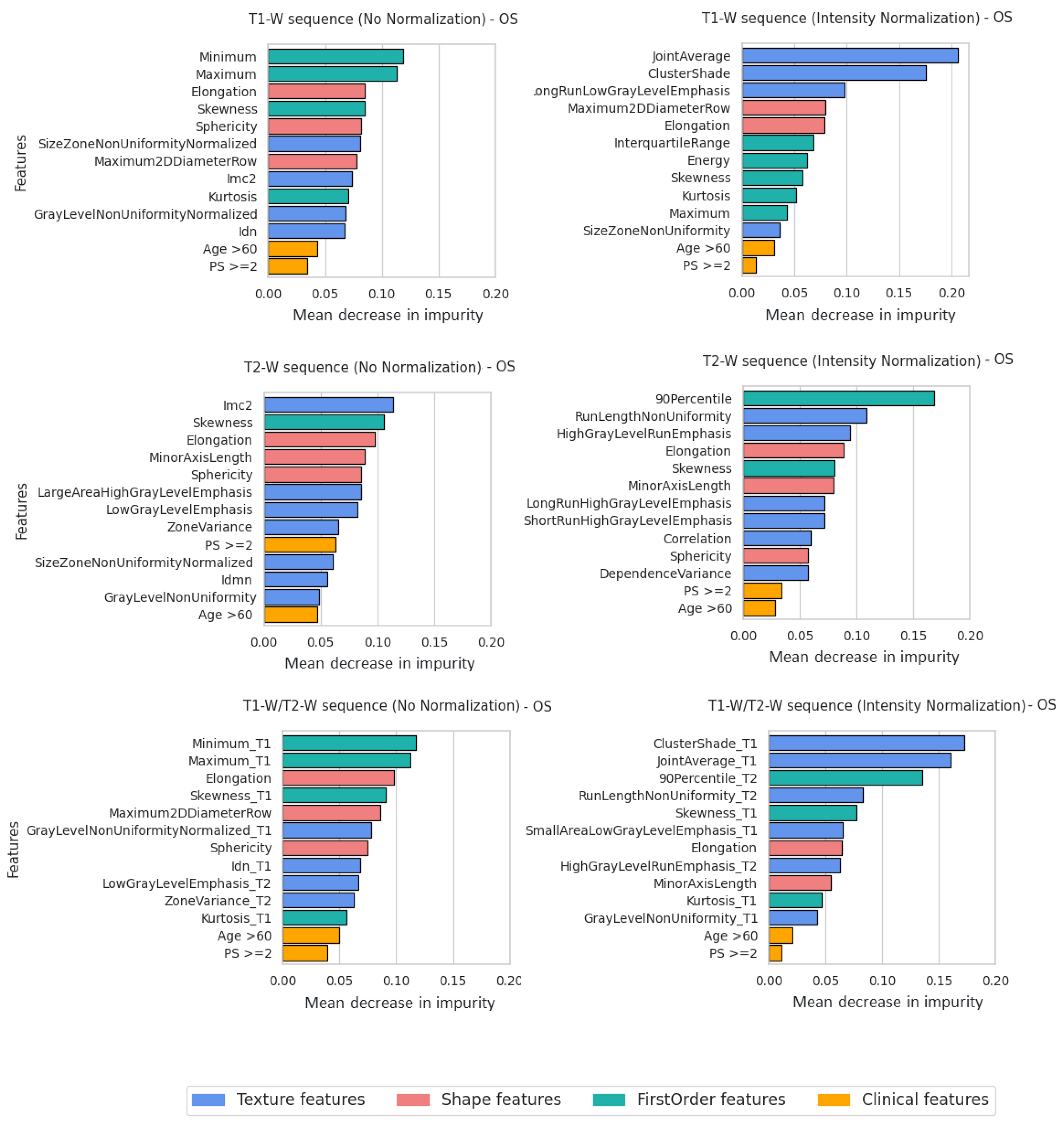
3.1. Impact of the Intensity Normalization Method on Radiomics Feature
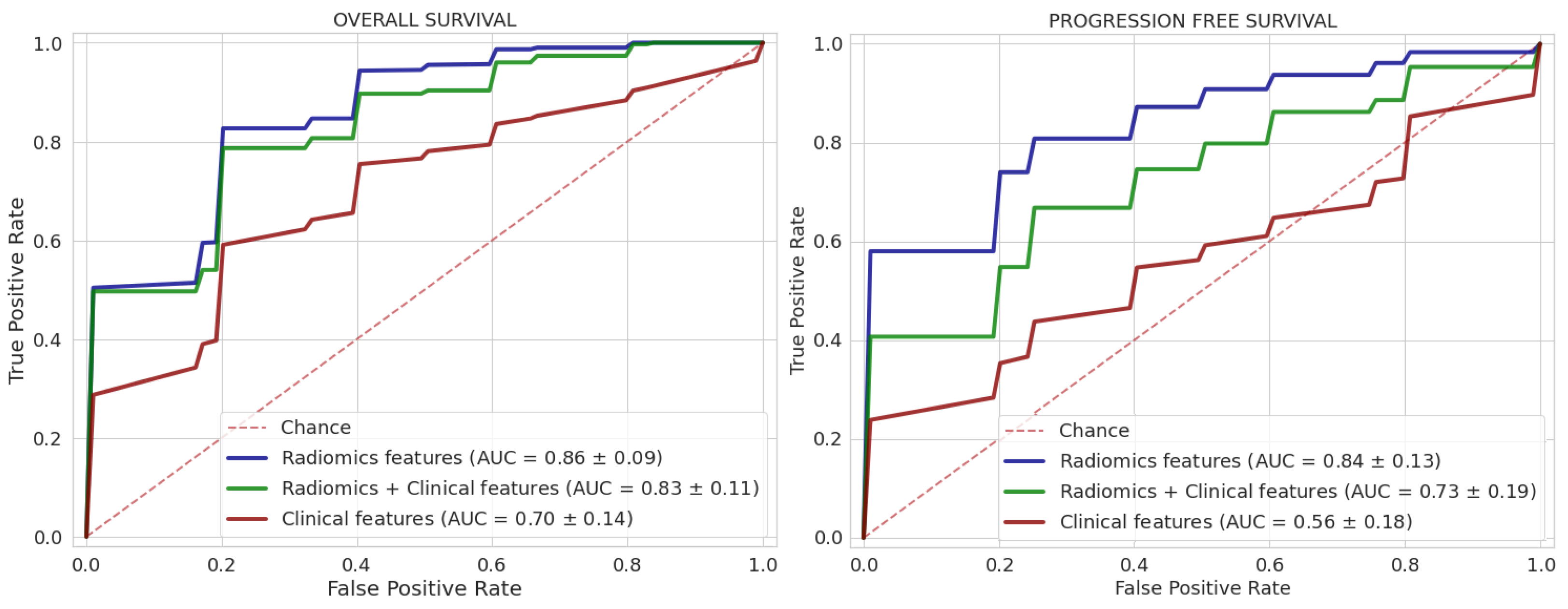
3.2. Performance Comparison of Classification Models
3.2.1. OS Classification Task
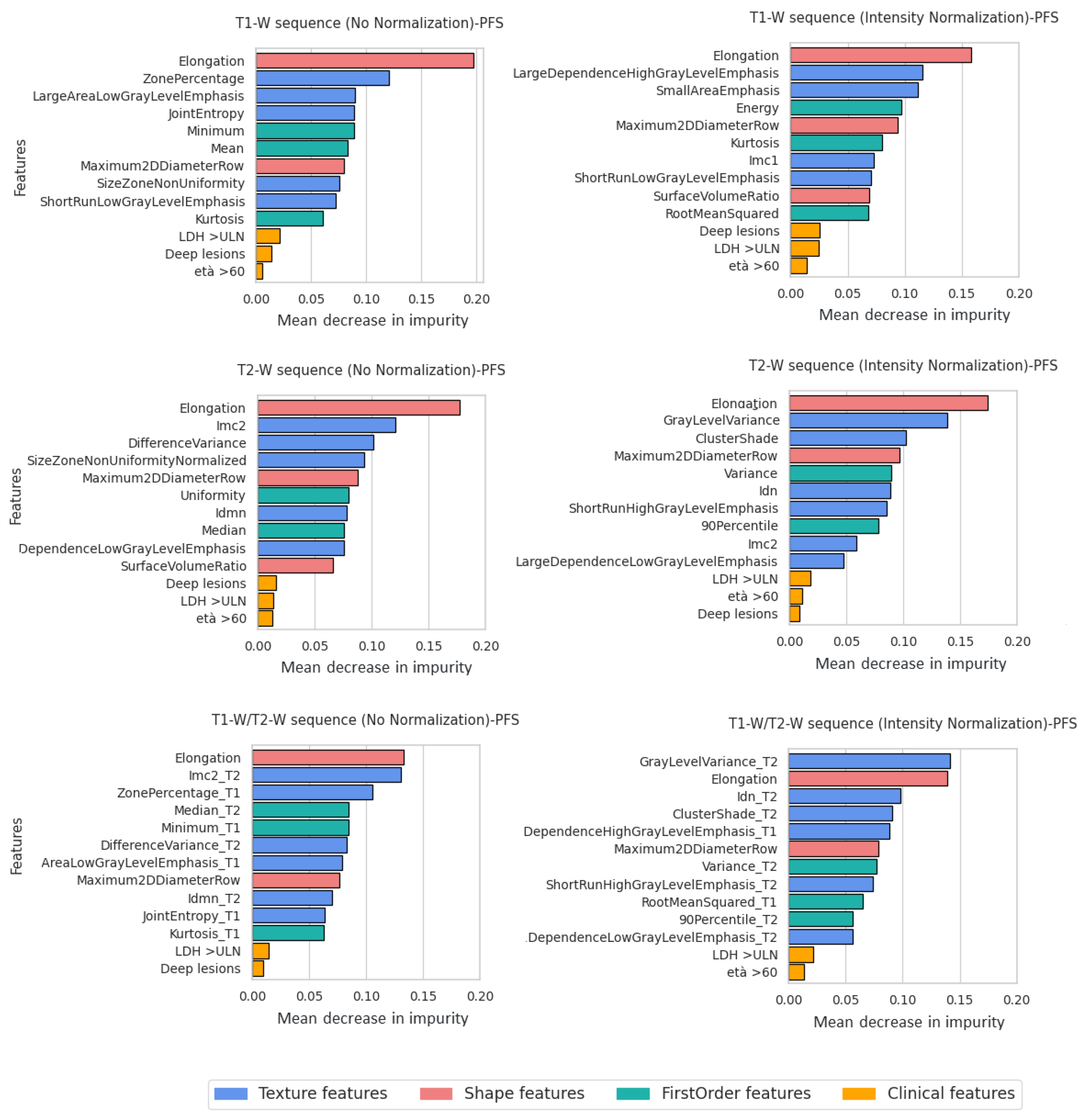
3.2.2. PFS Classification Task
3.3. Feature Importance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kluin, P. Primary diffuse large B-cell lymphoma of the CNS. In World Health Organization: Pathology and Genetics of Tumors of Haematopoietic and Lymphoid Tissues; World Health Organization: Geneva, Switzerland, 2008; pp. 240–241. [Google Scholar]
- Ferreri, A.J.; Holdhoff, M.; Nayak, L.; Rubenstein, J.L. Evolving Treatments for Primary Central Nervous System Lymphoma. In American Society of Clinical Oncology Educational Book; American Society of Clinical Oncology: Alexandria, VA, USA, 2019; Volume 39, pp. 454–466. [Google Scholar]
- Grommes, C.; Rubenstein, J.L.; DeAngelis, L.M.; Ferreri, A.J.; Batchelor, T.T. Comprehensive approach to diagnosis and treatment of newly diagnosed primary CNS lymphoma. Neuro-Oncology 2019, 21, 296–305. [Google Scholar] [CrossRef]
- Calimeri, T.; Steffanoni, S.; Gagliardi, F.; Chiara, A.; Ferreri, A. How we treat primary central nervous system lymphoma. ESMO Open 2021, 6, 100213. [Google Scholar] [CrossRef]
- Ferreri, A.J.; Cwynarski, K.; Pulczynski, E.; Fox, C.P.; Schorb, E.; La Rosée, P.; Binder, M.; Fabbri, A.; Torri, V.; Minacapelli, E.; et al. Whole-brain radiotherapy or autologous stem-cell transplantation as consolidation strategies after high-dose methotrexate-based chemoimmunotherapy in patients with primary CNS lymphoma: Results of the second randomisation of the International Extranodal Lymphoma Study Group-32 phase 2 trial. Lancet Haematol. 2017, 4, e510–e523. [Google Scholar]
- Houillier, C.; Taillandier, L.; Dureau, S.; Lamy, T.; Laadhari, M.; Chinot, O.; Moluçon-Chabrot, C.; Soubeyran, P.; Gressin, R.; Choquet, S.; et al. Radiotherapy or autologous stem-cell transplantation for primary CNS lymphoma in patients 60 years of age and younger: Results of the intergroup ANOCEF-GOELAMS randomized phase II PRECIS study. J. Clin. Oncol. 2019, 37, 823–833. [Google Scholar] [CrossRef]
- Batchelor, T.; Giri, S.; Ruppert, A.S.; Bartlett, N.L.; Hsi, E.D.; Cheson, B.D.; Nayak, L.; Leonard, J.P.; Rubenstein, J.L. Myeloablative versus non-myeloablative consolidative chemotherapy for newly diagnosed primary central nervous system lymphoma: Results of CALGB 51101 (Alliance). J. Clin. Oncol. 2021, 39, 7506. [Google Scholar] [CrossRef]
- Houillier, C.; Soussain, C.; Ghesquières, H.; Soubeyran, P.; Chinot, O.; Taillandier, L.; Lamy, T.; Choquet, S.; Ahle, G.; Damaj, G.; et al. Management and outcome of primary CNS lymphoma in the modern era: An LOC network study. Neurology 2020, 94, e1027–e1039. [Google Scholar] [CrossRef]
- Ambady, P.; Holdhoff, M.; Bonekamp, D.; Wong, F.; Grossman, S.A. Late relapses in primary CNS lymphoma after complete remissions with high-dose methotrexate monotherapy. Cns Oncl. 2015, 4, 393–398. [Google Scholar] [CrossRef]
- Gillies, R.J.; Kinahan, P.E.; Hricak, H. Radiomics: Images are more than pictures, they are data. Radiology 2016, 278, 563–577. [Google Scholar] [CrossRef]
- Zhou, M.; Scott, J.; Chaudhury, B.; Hall, L.; Goldgof, D.; Yeom, K.W.; Iv, M.; Ou, Y.; Kalpathy-Cramer, J.; Napel, S.; et al. Radiomics in brain tumor: Image assessment, quantitative feature descriptors, and machine-learning approaches. Am. J. Neuroradiol. 2018, 39, 208–216. [Google Scholar] [CrossRef]
- Khemchandani, M.A.; Jadhav, S.M.; Iyer, B. Brain Tumor Segmentation and Identification Using Particle Imperialist Deep Convolutional Neural Network in MRI Images. Int. J. Interact. Multimed. Artif. Intell. 2022, 7, 7. [Google Scholar] [CrossRef]
- Hassan, L.; Saleh, A.; Abdel-Nasser, M.; Omer, O.A.; Puig, D. Promising deep semantic nuclei segmentation models for multi-institutional histopathology images of different organs. Int. J. Interact. Multimed. Artif. Intell. 2021, 6, 6. [Google Scholar] [CrossRef]
- Tomaszewski, M.R.; Gillies, R.J. The biological meaning of radiomic features. Radiology 2021, 298, 505–516. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, S.; Di Dong, J.W.; Fang, C.; Zhou, X.; Sun, K.; Li, L.; Li, B.; Wang, M.; Tian, J. The applications of radiomics in precision diagnosis and treatment of oncology: Opportunities and challenges. Theranostics 2019, 9, 1303. [Google Scholar] [CrossRef]
- Luo, H.; Zhuang, Q.; Wang, Y.; Abudumijiti, A.; Shi, K.; Rominger, A.; Chen, H.; Yang, Z.; Tran, V.; Wu, G.; et al. A novel image signature-based radiomics method to achieve precise diagnosis and prognostic stratification of gliomas. Lab. Investig. 2021, 101, 450–462. [Google Scholar] [CrossRef]
- Rizzo, S.; Botta, F.; Raimondi, S.; Origgi, D.; Fanciullo, C.; Morganti, A.G.; Bellomi, M. Radiomics: The facts and the challenges of image analysis. Eur. Radiol. Exp. 2018, 2, 1–8. [Google Scholar] [CrossRef]
- Mayerhoefer, M.E.; Materka, A.; Langs, G.; Häggström, I.; Szczypiński, P.; Gibbs, P.; Cook, G. Introduction to radiomics. J. Nucl. Med. 2020, 61, 488–495. [Google Scholar] [CrossRef]
- Thawani, R.; McLane, M.; Beig, N.; Ghose, S.; Prasanna, P.; Velcheti, V.; Madabhushi, A. Radiomics and radiogenomics in lung cancer: A review for the clinician. Lung Cancer 2018, 115, 34–41. [Google Scholar] [CrossRef]
- Valdora, F.; Houssami, N.; Rossi, F.; Calabrese, M.; Tagliafico, A.S. Rapid review: Radiomics and breast cancer. Breast Cancer Res. Treat. 2018, 169, 217–229. [Google Scholar] [CrossRef]
- Staal, F.C.; van der Reijd, D.J.; Taghavi, M.; Lambregts, D.M.; Beets-Tan, R.G.; Maas, M. Radiomics for the prediction of treatment outcome and survival in patients with colorectal cancer: A systematic review. Clin. Color. Cancer 2021, 20, 52–71. [Google Scholar] [CrossRef]
- Kang, D.; Park, J.E.; Kim, Y.H.; Kim, J.H.; Oh, J.Y.; Kim, J.; Kim, Y.; Kim, S.T.; Kim, H.S. Diffusion radiomics as a diagnostic model for atypical manifestation of primary central nervous system lymphoma: Development and multicenter external validation. Neuro-Oncology 2018, 20, 1251–1261. [Google Scholar] [CrossRef]
- Chen, C.; Zheng, A.; Ou, X.; Wang, J.; Ma, X. Comparison of radiomics-based machine-learning classifiers in diagnosis of glioblastoma from primary central nervous system lymphoma. Front. Oncol. 2020, 10, 1151. [Google Scholar] [CrossRef]
- Xia, W.; Hu, B.; Li, H.; Geng, C.; Wu, Q.; Yang, L.; Yin, B.; Gao, X.; Li, Y.; Geng, D. Multiparametric-MRI-based radiomics model for differentiating primary central nervous system lymphoma from glioblastoma: Development and cross-vendor validation. J. Magn. Reson. Imaging 2021, 53, 242–250. [Google Scholar] [CrossRef]
- Yun, J.; Park, J.E.; Lee, H.; Ham, S.; Kim, N.; Kim, H.S. Radiomic features and multilayer perceptron network classifier: A robust MRI classification strategy for distinguishing glioblastoma from primary central nervous system lymphoma. Sci. Rep. 2019, 9, 5746. [Google Scholar] [CrossRef]
- Eisenhut, F.; Schmidt, M.A.; Putz, F.; Lettmaier, S.; Fröhlich, K.; Arinrad, S.; Coras, R.; Luecking, H.; Lang, S.; Fietkau, R.; et al. Classification of primary cerebral lymphoma and glioblastoma featuring dynamic susceptibility contrast and apparent diffusion coefficient. Brain Sci. 2020, 10, 886. [Google Scholar] [CrossRef]
- Kunimatsu, A.; Kunimatsu, N.; Kamiya, K.; Watadani, T.; Mori, H.; Abe, O. Comparison between glioblastoma and primary central nervous system lymphoma using MR image-based texture analysis. Magn. Reson. Med. Sci. 2018, 17, 50. [Google Scholar] [CrossRef]
- Kim, Y.; Cho, H.h.; Kim, S.T.; Park, H.; Nam, D.; Kong, D.S. Radiomics features to distinguish glioblastoma from primary central nervous system lymphoma on multi-parametric MRI. Neuroradiology 2018, 60, 1297–1305. [Google Scholar] [CrossRef]
- Wang, H.; Zhou, Y.; Li, L.; Hou, W.; Ma, X.; Tian, R. Current status and quality of radiomics studies in lymphoma: A systematic review. Eur. Radiol. 2020, 30, 6228–6240. [Google Scholar] [CrossRef]
- Chen, C.; Zhuo, H.; Wei, X.; Ma, X. Contrast-enhanced MRI texture parameters as potential prognostic factors for primary central nervous system lymphoma patients receiving high-dose methotrexate-based chemotherapy. Contrast Media Mol. Imaging 2019, 2019, 5481491. [Google Scholar] [CrossRef]
- Ali, O.M.; Nalawade, S.S.; Xi, Y.; Wagner, B.; Mazal, A.; Ahlers, S.; Rizvi, S.M.; Awan, F.T.; Kumar, K.A.; Desai, N.B.; et al. A Radiomic Machine Learning Model to Predict Treatment Response to Methotrexate and Survival Outcomes in Primary Central Nervous System Lymphoma (PCNSL). Blood 2020, 136, 29–30. [Google Scholar] [CrossRef]
- Villano, J.; Koshy, M.; Shaikh, H.; Dolecek, T.; McCarthy, B. Age, gender, and racial differences in incidence and survival in primary CNS lymphoma. Br. J. Cancer 2011, 105, 1414–1418. [Google Scholar] [CrossRef]
- Scalco, E.; Belfatto, A.; Mastropietro, A.; Rancati, T.; Avuzzi, B.; Messina, A.; Valdagni, R.; Rizzo, G. T2w-MRI signal normalization affects radiomics features reproducibility. Med. Phys. 2020, 47, 1680–1691. [Google Scholar] [CrossRef]
- Isaksson, L.J.; Raimondi, S.; Botta, F.; Pepa, M.; Gugliandolo, S.G.; De Angelis, S.P.; Marvaso, G.; Petralia, G.; De Cobelli, O.; Gandini, S.; et al. Effects of MRI image normalization techniques in prostate cancer radiomics. Phys. Medica 2020, 71, 7–13. [Google Scholar] [CrossRef]
- Hoebel, K.V.; Patel, J.B.; Beers, A.L.; Chang, K.; Singh, P.; Brown, J.M.; Pinho, M.C.; Batchelor, T.T.; Gerstner, E.R.; Rosen, B.R.; et al. Radiomics Repeatability Pitfalls in a Scan-Rescan MRI Study of Glioblastoma. Radiol. Artif. Intell. 2020, 3, e190199. [Google Scholar] [CrossRef]
- Schwier, M.; van Griethuysen, J.; Vangel, M.G.; Pieper, S.; Peled, S.; Tempany, C.; Aerts, H.J.; Kikinis, R.; Fennessy, F.M.; Fedorov, A. Repeatability of multiparametric prostate MRI radiomics features. Sci. Rep. 2019, 9, 9441. [Google Scholar] [CrossRef]
- Crombé, A.; Kind, M.; Fadli, D.; Le Loarer, F.; Italiano, A.; Buy, X.; Saut, O. Intensity harmonization techniques influence radiomics features and radiomics-based predictions in sarcoma patients. Sci. Rep. 2020, 10, 15496. [Google Scholar] [CrossRef]
- Shenkier, T.N.; Blay, J.Y.; O’Neill, B.P.; Poortmans, P.; Thiel, E.; Jahnke, K.; Abrey, L.E.; Neuwelt, E.; Tsang, R.; Batchelor, T.; et al. Primary CNS lymphoma of T-cell origin: A descriptive analysis from the international primary CNS lymphoma collaborative group. J. Clin. Oncol. 2005, 23, 2233–2239. [Google Scholar] [CrossRef]
- Moradmand, H.; Aghamiri, S.M.R.; Ghaderi, R. Impact of image preprocessing methods on reproducibility of radiomic features in multimodal magnetic resonance imaging in glioblastoma. J. Appl. Clin. Med. Phys. 2020, 21, 179–190. [Google Scholar] [CrossRef]
- Fedorov, A.; Beichel, R.; Kalpathy-Cramer, J.; Finet, J.; Fillion-Robin, J.C.; Pujol, S.; Bauer, C.; Jennings, D.; Fennessy, F.; Sonka, M.; et al. 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn. Reson. Imaging 2012, 30, 1323–1341. [Google Scholar] [CrossRef]
- Sled, J.G.; Zijdenbos, A.P.; Evans, A.C. A nonparametric method for automatic correction of intensity nonuniformity in MRI data. IEEE Trans. Med. Imaging 1998, 17, 87–97. [Google Scholar] [CrossRef]
- Kalavathi, P.; Prasath, V.S. Methods on skull stripping of MRI head scan images—A review. J. Digit. Imaging 2016, 29, 365–379. [Google Scholar] [CrossRef]
- Bauer, S.; Fejes, T.; Reyes, M. A skull-stripping filter for ITK. Insight J. 2013, 2012, 1–7. [Google Scholar] [CrossRef]
- Aganj, I.; Yeo, B.T.T.; Sabuncu, M.R.; Fischl, B. On removing interpolation and resampling artifacts in rigid image registration. IEEE Trans. Image Process 2012, 22, 816–827. [Google Scholar] [CrossRef]
- Shinohara, R.T.; Sweeney, E.M.; Goldsmith, J.; Shiee, N.; Mateen, F.J.; Calabresi, P.A.; Jarso, S.; Pham, D.L.; Reich, D.S.; Crainiceanu, C.M.; et al. Statistical normalization techniques for magnetic resonance imaging. Neuroimage Clin. 2014, 6, 9–19. [Google Scholar] [CrossRef]
- Nyúl, L.G.; Udupa, J.K. On standardizing the MR image intensity scale. Magn. Reson. Med. Off. J. Int. Soc. Magn. Reson. Med. 1999, 42, 1072–1081. [Google Scholar] [CrossRef]
- Van Griethuysen, J.J.; Fedorov, A.; Parmar, C.; Hosny, A.; Aucoin, N.; Narayan, V.; Beets-Tan, R.G.; Fillion-Robin, J.C.; Pieper, S.; Aerts, H.J. Computational radiomics system to decode the radiographic phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef]
- Liang, Z.G.; Tan, H.Q.; Zhang, F.; Rui Tan, L.K.; Lin, L.; Lenkowicz, J.; Wang, H.; Wen Ong, E.H.; Kusumawidjaja, G.; Phua, J.H.; et al. Comparison of radiomics tools for image analyses and clinical prediction in nasopharyngeal carcinoma. Br. J. Radiol. 2019, 92, 20190271. [Google Scholar] [CrossRef]
- Shrout, P.E.; Fleiss, J.L. Intraclass correlations: Uses in assessing rater reliability. Psychol. Bull. 1979, 86, 420. [Google Scholar] [CrossRef]
- Kasenda, B.; Ferreri, A.J.; Marturano, E.; Forst, D.; Bromberg, J.; Ghesquieres, H.; Ferlay, C.; Blay, J.Y.; Hoang-Xuan, K.; Pulczynski, E.; et al. First-line treatment and outcome of elderly patients with primary central nervous system lymphoma (PCNSL)—A systematic review and individual patient data meta-analysis. Ann. Oncol. 2015, 26, 1305–1313. [Google Scholar] [CrossRef]
- Ferreri, A.J.; Blay, J.Y.; Reni, M.; Pasini, F.; Spina, M.; Ambrosetti, A.; Calderoni, A.; Rossi, A.; Vavassori, V.; Conconi, A.; et al. Prognostic scoring system for primary CNS lymphomas: The International Extranodal Lymphoma Study Group experience. J. Clin. Oncol. 2003, 21, 266–272. [Google Scholar] [CrossRef]
- Abrey, L.E.; Ben-Porat, L.; Panageas, K.S.; Yahalom, J.; Berkey, B.; Curran, W.; Schultz, C.; Leibel, S.; Nelson, D.; Mehta, M.; et al. Primary central nervous system lymphoma: The Memorial Sloan-Kettering Cancer Center prognostic model. J. Clin. Oncol. 2006, 24, 5711–5715. [Google Scholar] [CrossRef]
- Carré, A.; Klausner, G.; Edjlali, M.; Lerousseau, M.; Briend-Diop, J.; Sun, R.; Ammari, S.; Reuzé, S.; Alvarez-Andres, E.; Estienne, T.; et al. Standardization of Brain MRI across Machines and Protocols: Bridging the Gap for MRI-Based Radiomics. In Proceedings of the Radiotherapy and Oncology, Online, 28 November–1 December 2020; Elsevier Ireland Ltd. Elsevier House: East Park Shannon, UK, 2020; Volume 152, p. S294. [Google Scholar]
- Barajas, R.F., Jr.; Politi, L.S.; Anzalone, N.; Schöder, H.; Fox, C.P.; Boxerman, J.L.; Kaufmann, T.J.; Quarles, C.C.; Ellingson, B.M.; Auer, D.; et al. Consensus recommendations for MRI and PET imaging of primary central nervous system lymphoma: Guideline statement from the International Primary CNS Lymphoma Collaborative Group (IPCG). Neuro-Oncology 2021, 23, 1056–1071. [Google Scholar] [CrossRef]
- Li, Y.; Ammari, S.; Balleyguier, C.; Lassau, N.; Chouzenoux, E. Impact of Preprocessing and Harmonization Methods on the Removal of Scanner Effects in Brain MRI Radiomic Features. Cancers 2021, 13, 3000. [Google Scholar] [CrossRef]
- Fujima, N.; Homma, A.; Harada, T.; Shimizu, Y.; Tha, K.K.; Kano, S.; Mizumachi, T.; Li, R.; Kudo, K.; Shirato, H. The utility of MRI histogram and texture analysis for the prediction of histological diagnosis in head and neck malignancies. Cancer Imaging 2019, 19, 5. [Google Scholar] [CrossRef]
- Meyer, H.J.; Schob, S.; Höhn, A.K.; Surov, A. MRI texture analysis reflects histopathology parameters in thyroid cancer–a first preliminary study. Transl. Oncol. 2017, 10, 911–916. [Google Scholar] [CrossRef]
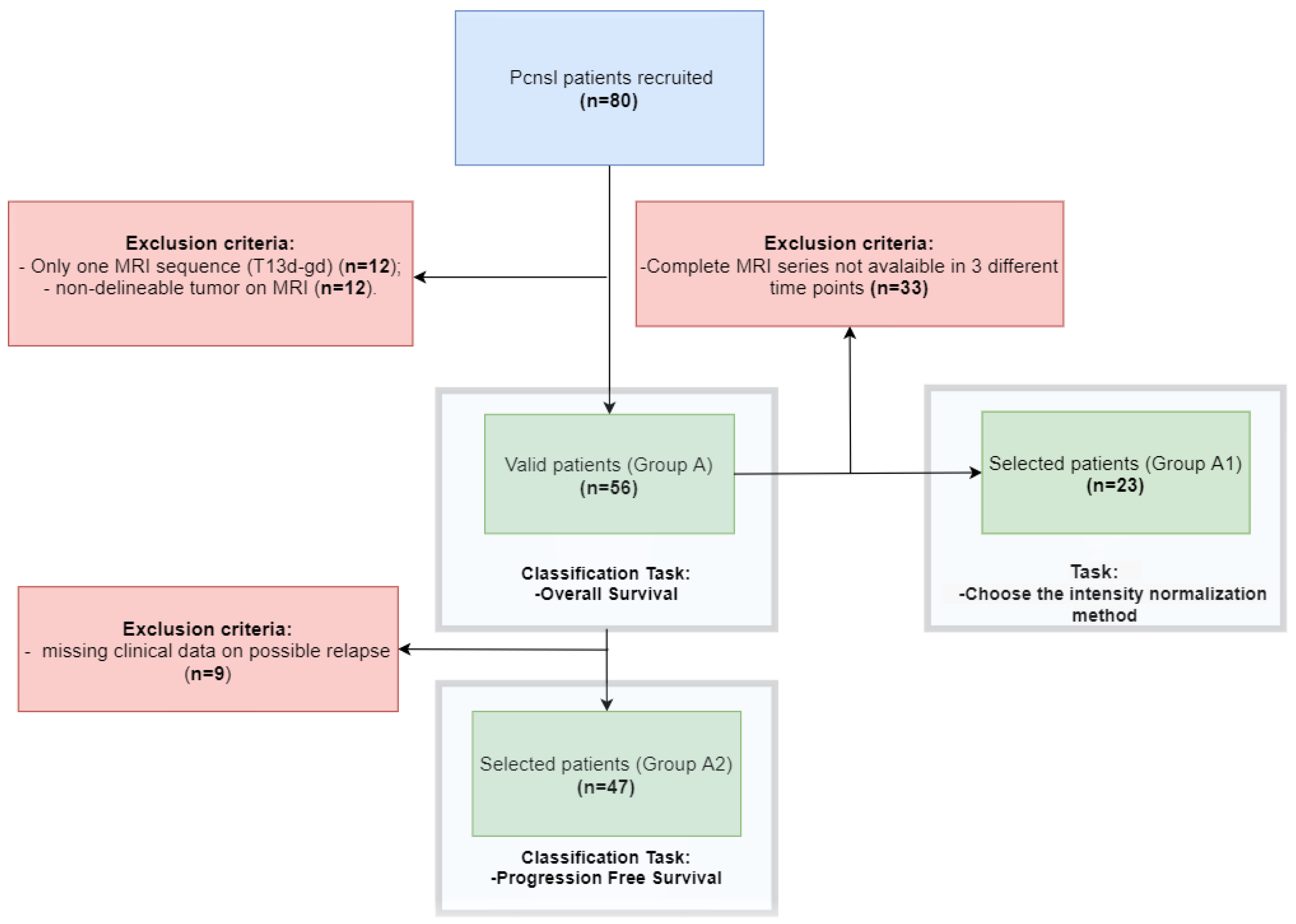


| Eligible Patients (#) | 56/80 (70%) |
| Male:Female | 0.56 |
| Median Age | 69 (41–85) |
| Multiple lesions | 32 (58%) |
| Involvement of deep areas § | 45 (80%) |
| Lactic dehydrogenase serum level >ULN | 35 (52%) |
| Cerebrospinal-fluid protein concentration >ULN * | 34(60%) |
| ECOG—Performance Status >2 | 30 (53%) |
| IELSG risk score | |
| -Low | 5 (9%) |
| -Intermediate | 28 (50%) |
| -High | 23 (41%) |
| Sites of disease | |
| -Brain parenchyma | 56 (100%) |
| Treatment details | |
| Induction | |
| MATRix | 37 (66%) |
| MAT | 2 (3%) |
| HD-MTX + HD-ARAC | 10 (17%) |
| HD-MTX + Alkylators | 4 (7%) |
| WBRT ± TMZ | 4 (7%) |
| Rituximab | 43 (77%) |
| Consolidations | |
| ASCT | 15 (27%) |
| WBRT | 6 (11%) |
| DeVIC | 5 (9%) |
| Oral Maintenance | 3 (5%) |
| None | 26 (46%) |
| Unknown | 1(2%) |
| Treatment delay >20 gg | 40 (71%) |
| Refractory to first line @ | 22 (39%) |
| 1-year PFS | 24/47 (51%) |
| 1-year OS | 30/56 (54%) |
| OS | Radiomics Features | ETC | SVM | LR | RF | KN |
| T1-W | No Normalizazion | 0.67 (0.61–0.79) | 0.71 (0.70–0.71) | 0.71 (0.67–0.71) | 0.67 (0.61–0.72) | 0.67 (0.61–0.73) |
| Intensity Normalization | 0.75 (0.67–0.83) | 0.77 (0.68–0.83) | 0.77 (0.73–0.83) | 0.73 (0.67–0.83) | 0.73 (0.63–0.80) | |
| T2-W | No Normalization | 0.67 (0.55–0.73) | 0.67 (0.57–0.71) | 0.71 (0.67–0.71) | 0.59 (0.50–0.71) | 0.57 (0.44–0.70) |
| Intensity Normalization | 0.79 (0.73–0.86) | 0.80 (0.77–0.86) | 0.80 (0.75–0.86) | 0.73 (0.67–0.830) | 0.77 (0.72–0.80) | |
| T1-W/T2-W | No Normalization | 0.67 (0.57–0.72) | 0.67 (0.55–0.76) | 0.67 (0.60–0.76) | 0.61 (0.54–0.71) | 0.61 (0.54–0.70) |
| Intensity Normalization | 0.80 (0.77–0.86) | 0.80 (0.72–0.83) | 0.80 (0.73–0.83) | 0.83 (0.77–0.86) | 0.80 (0.72–0.83) | |
| OS | Radiomics + Clinical Features | ETC | SVM | LR | RF | KN |
| T1-W | No Normalizazion | 0.72 (0.67–0.80) | 0.73 (0.60–0.80) | 0.73 (0.60–0.80) | 0.67 (0.61–0.75) | 0.73 (0.60–0.77) |
| Intensity Normalization | 0.80 (0.73–0.83) | 0.79 (0.68–0.83) | 0.80 (0.68–0.83) | 0.80 (0.71–0.83) | 0.82 (0.73–0.86) | |
| T2-W | No Normalization | 0.73 (0.66–0.80) | 0.72 (0.60–0.825) | 0.72 (0.66–0.77) | 0.72 (0.60–0.77) | 0.67 (0.60–0.77) |
| Intensity Normalization | 0.77 (0.66–0.86) | 0.77 (0.68–0.83) | 0.77 (0.66–0.83) | 0.73 (0.68–0.80) | 0.77 (0.67–0.77) | |
| T1-W/T2-W | No Normalization | 0.77 (0.67–0.86) | 0.73 (0.66–0.83) | 0.73 (0.66–0.83) | 0.67 (0.60–0.72) | 0.73 (0.60–0.80) |
| Intensity Normalization | 0.80 (0.73–0.86) | 0.80 (0.73–0.83) | 0.80 (0.72–0.83) | 0.77 (0.68–0.83) | 0.80 (0.72–0.83) | |
| OS | Clinical Features | ETC | SVM | LR | RF | KN |
| 0.60 (0.44–0.67) | 0.71 (0.66–0.79) | 0.71 (0.66–0.77) | 0.60 (0.54–0.67) | 0.67 (0.60–0.77) | ||
| PFS | Radiomics Features | ETC | SVM | LR | RF | KN |
| T1-W | No Normalizazion | 0.67 (0.54–0.72) | 0.68 (0.58–0.75) | 0.71 (0.66–0.79) | 0.60 (0.50–0.72) | 0.60 (0.54–0.73) |
| Intensity Normalization | 0.60 (0.50–0.66) | 0.68 (0.60–0.68) | 0.68 (0.66–0.73) | 0.60 (0.50–0.66) | 0.67 (0.55–0.66) | |
| T2-W | No Normalization | 0.67 (0.55–0.75) | 0.67 (0.61–0.68) | 0.67 (0.61–0.68) | 0.60 (0.50–0.73) | 0.67 (0.51–0.73) |
| Intensity Normalization | 0.68 (0.57–0.76) | 0.80 (0.67–0.88) | 0.80 (0.67–0.86) | 0.68 (0.55–0.76) | 0.73 (0.67–0.83) | |
| T1-W/T2-W | No Normalization | 0.67 (0.50–0.74) | 0.67 (0.60–0.73) | 0.67 (0.58–0.73) | 0.60 (0.46–0.67) | 0.67 (0.58–0.73) |
| Intensity Normalization | 0.63 (0.50–0.75) | 0.70 (0.60–0.80) | 0.73 (0.62–0.80) | 0.67 (0.58–0.75) | 0.68 (0.60–0.75) | |
| PFS | Radiomics + Clinical Features | ETC | SVM | LR | RF | KN |
| T1-W | No Normalizazion | 0.60 (0.44–0.67) | 0.67 (0.60–0.71) | 0.67 (0.55–0.71) | 0.60 (0.51–0.73) | 0.60 (0.47–0.72) |
| Intensity Normalization | 0.60 (0.45–0.67) | 0.68 (0.61–0.67) | 0.68 (0.60–0.67) | 0.60 (0.50–0.72) | 0.60 (0.45–0.67) | |
| T2-W | No Normalization | 0.60 (0.48–0.71) | 0.61 (0.55–0.67) | 0.67 (0.55–0.76) | 0.60 (0.50–0.70) | 0.62 (0.55–0.67) |
| Intensity Normalization | 0.68 (0.50–0.76) | 0.72 (0.60–0.80) | 0.69 (0.60–0.75) | 0.70 (0.60–0.77) | 0.69 (0.60–0.73) | |
| T1-W/T2-W | No Normalization | 0.64 (0.55–0.68) | 0.67 (0.66–0.71) | 0.67 (0.61–0.77) | 0.61 (0.50–0.73) | 0.61 (0.50–0.67) |
| Intensity Normalization | 0.61 (0.44–0.68) | 0.69 (0.60–0.73) | 0.65 (0.55–0.68) | 0.60 (0.50–0.67) | 0.60 (0.45–0.62) | |
| PFS | Clinical Features | ETC | SVM | LR | RF | KN |
| 0.55 (0.41–0.60) | 0.62 (0.51–0.67) | 0.67 (0.63–0.71) | 0.57(0.47–0.65) | 0.55 (0.40–0.61) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Destito, M.; Marzullo, A.; Leone, R.; Zaffino, P.; Steffanoni, S.; Erbella, F.; Calimeri, F.; Anzalone, N.; De Momi, E.; Ferreri, A.J.M.; et al. Radiomics-Based Machine Learning Model for Predicting Overall and Progression-Free Survival in Rare Cancer: A Case Study for Primary CNS Lymphoma Patients. Bioengineering 2023, 10, 285. https://doi.org/10.3390/bioengineering10030285
Destito M, Marzullo A, Leone R, Zaffino P, Steffanoni S, Erbella F, Calimeri F, Anzalone N, De Momi E, Ferreri AJM, et al. Radiomics-Based Machine Learning Model for Predicting Overall and Progression-Free Survival in Rare Cancer: A Case Study for Primary CNS Lymphoma Patients. Bioengineering. 2023; 10(3):285. https://doi.org/10.3390/bioengineering10030285
Chicago/Turabian StyleDestito, Michela, Aldo Marzullo, Riccardo Leone, Paolo Zaffino, Sara Steffanoni, Federico Erbella, Francesco Calimeri, Nicoletta Anzalone, Elena De Momi, Andrés J. M. Ferreri, and et al. 2023. "Radiomics-Based Machine Learning Model for Predicting Overall and Progression-Free Survival in Rare Cancer: A Case Study for Primary CNS Lymphoma Patients" Bioengineering 10, no. 3: 285. https://doi.org/10.3390/bioengineering10030285
APA StyleDestito, M., Marzullo, A., Leone, R., Zaffino, P., Steffanoni, S., Erbella, F., Calimeri, F., Anzalone, N., De Momi, E., Ferreri, A. J. M., Calimeri, T., & Spadea, M. F. (2023). Radiomics-Based Machine Learning Model for Predicting Overall and Progression-Free Survival in Rare Cancer: A Case Study for Primary CNS Lymphoma Patients. Bioengineering, 10(3), 285. https://doi.org/10.3390/bioengineering10030285











