Individualized PEEP without Recruitment Maneuvers Improves Intraoperative Oxygenation: A Randomized Controlled Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Approval
2.2. Inclusion Criteria
- Patients undergoing elective robot-assisted prostate surgery under general anesthesia;
- American Society of Anesthesiologists (ASA) grade I~III;
- BMI < 30 kg/m2.
2.3. Exclusion Criteria
- Untreated ischemic heart disease;
- Acute or chronic respiratory failure and moderate to severe obstructive or restrictive pulmonary diseases, including chronic obstructive pulmonary disease and asthma;
- Previous lung or airway surgery;
- Neuromuscular diseases;
- Preoperative SpO2 (in room air) < 95%.
2.4. Anesthesia Management
2.5. Intraoperative Ventilation
2.6. Measurements
2.7. The Individual PEEP Titration by EIT
2.8. Outcome Measures
- (1)
- Intraoperative driving pressure, plateau pressure, and Cdyn;
- (2)
- Ventral over dorsal ventilation distribution ratio;
- (3)
- Intraoperative hemodynamics;
- (4)
- The incidence of postoperative hypoxemia (SpO2 < 92% were on room air after extubation) in the PACU.
2.9. Statistical Analysis
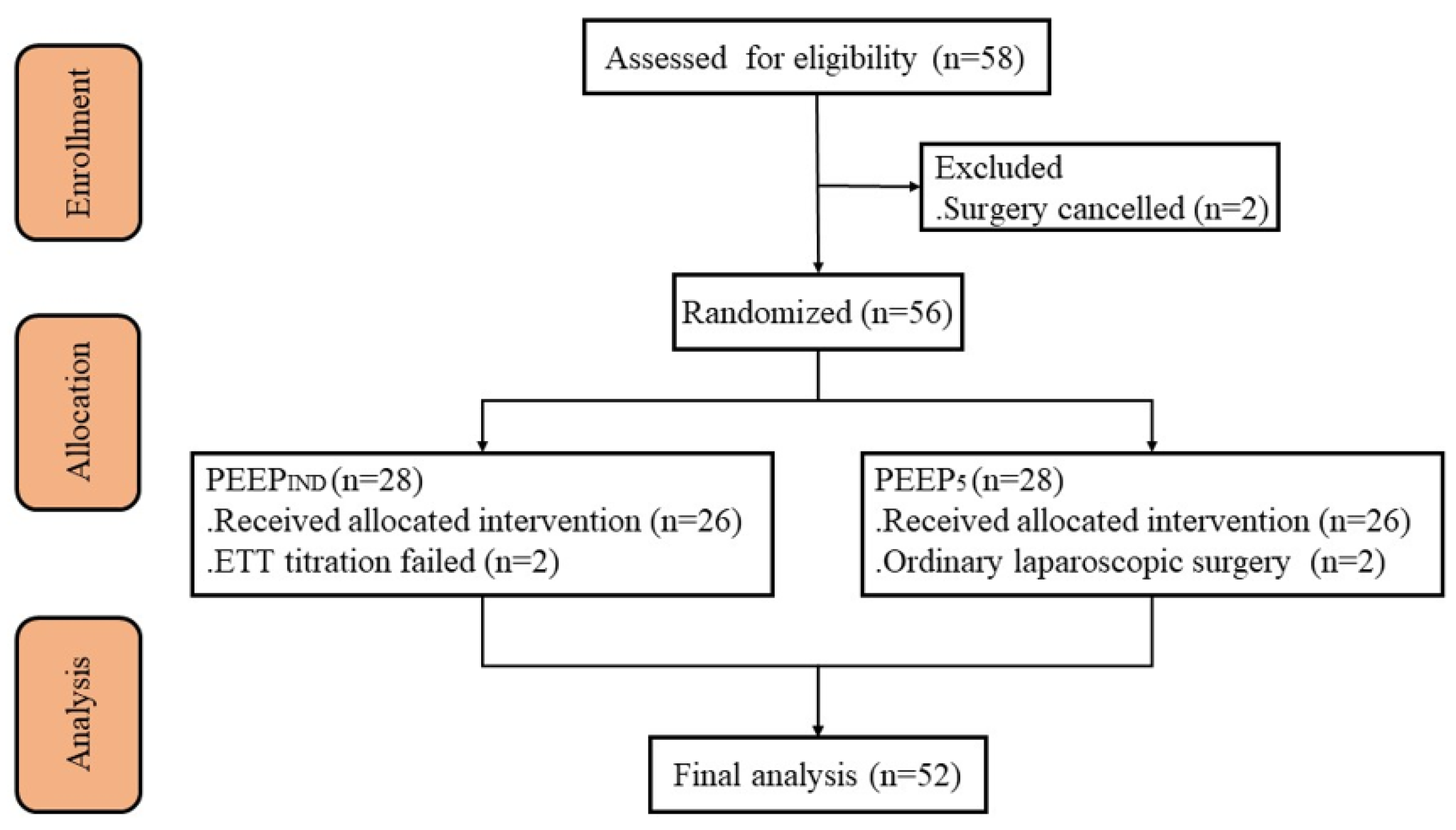
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stolzenburg, J.; Kallidonis, P.; Qazi, H.; Ho Thi, P.; Dietel, A.; Liatsikos, E.N.; Do, M. Extraperitoneal approach for robotic-assisted simple prostatectomy. Urology 2014, 84, 1099–1105. [Google Scholar] [PubMed]
- Andersson, L.E.; Beth, M.; Thorne, A.; Aspelin, P.; Odeberg-Wernerman, S. Effect of carbon dioxide pneumoperitoneum on development of atelectasis during anesthesia, examined by spiral computed tomography. Anesthesiology 2005, 102, 293–299. [Google Scholar] [PubMed]
- Sahetya, S.K. Searching for the optimal positive end-expiratory pressure for lung protective ventilation. Curr. Opin. Crit. Care 2020, 26, 53–58. [Google Scholar]
- Girrbach, F.; Petroff, D.; Schulz, S.; Hempel, G.; Lange, M.; Klotz, C.; Scherz, S.; Giannella-Neto, A.; Beda, A.; Jardim-Neto, A.; et al. Individualised positive end-expiratory pressure guided by electrical impedance tomography for robot-assisted laparoscopic radical prostatectomy: A prospective, randomised controlled clinical trial. Br. J. Anaesth. 2020, 125, 373–382. [Google Scholar] [PubMed]
- Pelosi, P.; Croci, M.; Ravagnan, I.; Tredici, S.; Pedoto, A.; Lissoni, A.; Gattinoni, L. The effects of body mass on lung volumes, respiratory mechanics, and gas exchange during general anesthesia. Anesth. Analg. 1998, 87, 654–660. [Google Scholar]
- Qian, M.; Yang, F.; Zhao, L.; Shen, J.; Xie, Y. Individualized positive end-expiratory pressure titration on respiration and circulation in elderly patients undergoing spinal surgery in prone position under general anesthesia. Am. J. Transl. Res. 2021, 13, 13835–13844. [Google Scholar]
- Maisch, S.; Reissmann, H.; Fuellekrug, B.; Weismann, D.; Rutkowski, T.; Tusman, G.; Bohm, S.H. Compliance and dead space fraction indicate an optimal level of positive End-Expiratory pressure after recruitment in anesthetized patients. Anesth. Analg. 2008, 106, 175–181. [Google Scholar]
- Blankman, P.; Shono, A.; Hermans, B.J.M.; Wesselius, T.; Hasan, D.; Gommers, D. Detection of optimal PEEP for equal distribution of tidal volume by volumetric capnography and electrical impedance tomography during decreasing levels of PEEP in post cardiac-surgery patients. Br. J. Anaesth. 2016, 116, 862–869. [Google Scholar]
- Bian, W.; Chen, W.; Chao, Y.; Wang, L.; Li, L.; Guan, J.; Zang, X.; Zhen, J.; Sheng, B.; Zhu, X. Application of dead space fraction to titrate optimal positive end-expiratory pressure in an ARDS swine model. Exp. Ther. Med. 2017, 13, 1572–1577. [Google Scholar]
- Malbouisson, L.M.; Muller, J.; Constantin, J.; Lu, Q.; Puybasset, L.; Rouby, J.; Ct, S.A.S.G.; The, C.S.A.S. Computed tomography assessment of positive end-expiratory pressure-induced alveolar recruitment in patients with acute respiratory distress syndrome. Am. J. Resp. Crit. Care 2001, 163, 1444–1450. [Google Scholar]
- Beitler, J.R.; Sarge, T.; Banner-Goodspeed, V.M.; Gong, M.N.; Cook, D.; Novack, V.; Loring, S.H.; Talmor, D.; Epvent-, S.G. Effect of titrating positive End-Expiratory pressure (PEEP) with an esophageal pressure–guided strategy vs an empirical high PEEP-Fio2 strategy on death and days free from mechanical ventilation among patients with acute respiratory distress syndrome: A randomized clinical trial. JAMA 2019, 321, 846–857. [Google Scholar] [PubMed]
- Hsu, H.J.; Chang, H.T.; Zhao, Z.; Wang, P.H.; Zhang, J.H.; Chen, Y.S.; Frerichs, I.; Moller, K.; Fu, F.; Hsu, H.S.; et al. Positive end-expiratory pressure titration with electrical impedance tomography and pressure-volume curve: A randomized trial in moderate to severe ARDS. Physiol. Meas. 2021, 42, 14002. [Google Scholar]
- Zhao, Z.; Chen, T.F.; Teng, H.C.; Wang, Y.C.; Chang, M.Y.; Chang, H.T.; Frerichs, I.; Fu, F.; Moller, K. Is there a need for individualized adjustment of electrode belt position during EIT-guided titration of positive end-expiratory pressure? Physiol. Meas. 2022, 43, 064001. [Google Scholar]
- Pereira, S.M.; Tucci, M.R.; Morais, C.C.A.; Simões, C.M.; Tonelotto, B.F.F.; Pompeo, M.S.; Kay, F.U.; Pelosi, P.; Vieira, J.E.; Amato, M.B.P. Individual positive end-expiratory pressure settings optimize intraoperative mechanical ventilation and reduce postoperative atelectasis. Anesthesiology 2018, 129, 1070–1081. [Google Scholar]
- Reinius, H.; Borges, J.B.; Engstrom, J.; Ahlgren, O.; Lennmyr, F.; Larsson, A.; Freden, F. Optimal PEEP during one-lung ventilation with capnothorax: An experimental study. Acta Anaesthesiol. Scand. 2019, 63, 222–231. [Google Scholar] [PubMed]
- Nestler, C.; Simon, P.; Petroff, D.; Hammermüller, S.; Kamrath, D.; Wolf, S.; Dietrich, A.; Camilo, L.M.; Beda, A.; Carvalho, A.R.; et al. Individualized positive end-expiratory pressure in obese patients during general anaesthesia: A randomized controlled clinical trial using electrical impedance tomography. Br. J. Anaesth. 2017, 119, 1194–1205. [Google Scholar] [PubMed]
- Suzumura, E.A.; Figueiró, M.; Normilio-Silva, K.; Laranjeira, L.; Oliveira, C.; Buehler, A.M.; Bugano, D.; Passos Amato, M.B.; Ribeiro Carvalho, C.R.; Berwanger, O.; et al. Effects of alveolar recruitment maneuvers on clinical outcomes in patients with acute respiratory distress syndrome: A systematic review and meta-analysis. Intensive Care Med. 2014, 40, 1227–1240. [Google Scholar] [PubMed]
- Costa, E.L.; Borges, J.B.; Melo, A.; Suarez-Sipmann, F.; Toufen, C.J.; Bohm, S.H.; Amato, M.B. Bedside estimation of recruitable alveolar collapse and hyperdistension by electrical impedance tomography. Intensive Care Med. 2009, 35, 1132–1137. [Google Scholar]
- Liu, K.; Huang, C.; Xu, M.; Wu, J.; Frerichs, I.; Moeller, K.; Zhao, Z. PEEP guided by electrical impedance tomography during one-lung ventilation in elderly patients undergoing thoracoscopic surgery. Ann. Transl. Med. 2019, 7, 757. [Google Scholar]
- Akand, M.; Erdogru, T.; Avci, E.; Ates, M. Transperitoneal versus extraperitoneal robot-assisted laparoscopic radical prostatectomy: A prospective single surgeon randomized comparative study. Int. J. Urol. 2015, 22, 916–921. [Google Scholar]
- Dal Moro, F.; Crestani, A.; Valotto, C.; Guttilla, A.; Soncin, R.; Mangano, A.; Zattoni, F. Anesthesiologic effects of transperitoneal versus extraperitoneal approach during robot-assisted radical prostatectomy: Results of a prospective randomized study. Int. Braz. J. Urol. 2015, 41, 466–472. [Google Scholar] [PubMed]
- Cochetti, G.; Del Zingaro, M.; Ciarletti, S.; Paladini, A.; Felici, G.; Stivalini, D.; Cellini, V.; Mearini, E. New evolution of robotic radical prostatectomy: A single center experience with PERUSIA technique. Appl. Sci. 2021, 11, 1513. [Google Scholar]
- Di Pierro, G.B.; Wirth, J.G.; Ferrari, M.; Danuser, H.; Mattei, A. Impact of a single-surgeon learning curve on complications, positioning injuries, and renal function in patients undergoing robot-assisted radical prostatectomy and extended pelvic lymph node dissection. Urology 2014, 84, 1106–1111. [Google Scholar] [PubMed]
- Paladini, A.; Cochetti, G.; Felici, G.; Russo, M.; Saqer, E.; Cari, L.; Bordini, S.; Mearini, E. Complications of extraperitoneal robot-assisted radical prostatectomy in high-risk prostate cancer: A single high-volume center experience. Front. Surg. 2023, 10, 1157528. [Google Scholar] [PubMed]
- Rohloff, M.; Cicic, A.; Christensen, C.; Maatman, T.K.; Lindberg, J.; Maatman, T.J. Reduction in postoperative ileus rates utilizing lower pressure pneumoperitoneum in robotic-assisted radical prostatectomy. J. Robot. Surg. 2019, 13, 671–674. [Google Scholar] [PubMed]
- Kobylianskii, J.; Murray, A.; Brace, D.; Goligher, E.; Fan, E. Electrical impedance tomography in adult patients undergoing mechanical ventilation: A systematic review. J. Crit. Care 2016, 35, 33–50. [Google Scholar] [PubMed]
- Zitzmann, A.; Pulletz, S.; Gonzales-Rios, P.; Frenkel, P.; Teschendorf, P.; Kremeier, P.; Loser, B.; Krukewitt, L.; Reuter, D.A.; Bohm, S.H.; et al. Regional ventilation in spontaneously breathing COVID-19 patients during postural maneuvers assessed by electrical impedance tomography. Acta Anaesthesiol. Scand. 2023, 67, 185–194. [Google Scholar]
- Futier, E.; Jaber, S. Lung-protective ventilation in abdominal surgery. Curr. Opin. Crit. Care 2014, 20, 426–430. [Google Scholar]
- Lagier, D.; Velly, L.J.; Guinard, B.; Bruder, N.; Guidon, C.; Vidal Melo, M.F.; Alessi, M. Perioperative open-lung approach, regional ventilation, and lung injury in cardiac surgery. Anesthesiology 2020, 133, 1029–1045. [Google Scholar]
- Young, C.C.; Harris, E.M.; Vacchiano, C.; Bodnar, S.; Bukowy, B.; Elliott, R.R.D.; Migliarese, J.; Ragains, C.; Trethewey, B.; Woodward, A.; et al. Lung-protective ventilation for the surgical patient: International expert panel-based consensus recommendations. Br. J. Anaesth. 2019, 123, 898–913. [Google Scholar]
- Muders, T.; Luepschen, H.; Zinserling, J.; Greschus, S.; Fimmers, R.; Guenther, U.; Buchwald, M.; Grigutsch, D.; Leonhardt, S.; Putensen, C.; et al. Tidal recruitment assessed by electrical impedance tomography and computed tomography in a porcine model of lung injury. Crit. Care Med. 2012, 40, 903–911. [Google Scholar]
- PROVE Network Investigators. High versus low positive end-expiratory pressure during general anaesthesia for open abdominal surgery (PROVHILO trial): A multicentre randomised controlled trial. Lancet 2014, 384, 495–503. [Google Scholar]
- Shono, A.; Katayama, N.; Fujihara, T.; Böhm, S.H.; Waldmann, A.D.; Ugata, K.; Nikai, T.; Saito, Y. Positive end-expiratory pressure and distribution of ventilation in pneumoperitoneum combined with steep trendelenburg position. Anesthesiology 2020, 132, 476–490. [Google Scholar] [PubMed]
- Luecke, T.; Pelosi, P. Clinical review: Positive end-expiratory pressure and cardiac output. Crit. Care 2005, 9, 607–621. [Google Scholar] [PubMed]
- Vieillard-Baron, A.; Matthay, M.; Teboul, J.L.; Bein, T.; Schultz, M.; Magder, S.; Marini, J.J. Experts’ opinion on management of hemodynamics in ARDS patients: Focus on the effects of mechanical ventilation. Intensive Care Med. 2016, 42, 739–749. [Google Scholar]
- Serpa, N.A.; Hemmes, S.N.; Barbas, C.S.; Beiderlinden, M.; Fernandez-Bustamante, A.; Futier, E.; Hollmann, M.W.; Jaber, S.; Kozian, A.; Licker, M.; et al. Incidence of mortality and morbidity related to postoperative lung injury in patients who have undergone abdominal or thoracic surgery: A systematic review and meta-analysis. Lancet Respir. Med. 2014, 2, 1007–1015. [Google Scholar]
- Sribar, A.; Merc, V.; Persec, Z.; Persec, J.; Milas, I.; Husedzinovic, S. Influence of different PEEP levels on electrical impedance tomography findings in patients under general anesthesia ventilated in the lateral decubitus position. J. Clin. Monit. Comput. 2020, 34, 311–318. [Google Scholar]
- Hedenstierna, G. Gas exchange during anaesthesia. Br. J. Anaesth. 1990, 64, 507–514. [Google Scholar] [CrossRef]
- Miskovic, A.; Lumb, A.B. Postoperative pulmonary complications. Br. J. Anaesth. 2017, 118, 317–334. [Google Scholar]
| Characteristic | PEEP5 (n = 26) | PEEPIND (n = 26) |
|---|---|---|
| Age (yr) | 65 (52–83) | 67.8 (53–80) |
| <65 [n (%)] | 13 (50) | 9 (34.6) |
| BMI (kg m−2) | 24.4 (2.1) | 24.1 (2.2) |
| ASA physical status | ||
| I | 3 (11.5) | 4 (15.4) |
| II | 22 (84.6) | 22 (84.6) |
| III | 1 (3.8) | 0 (0) |
| Hypertension (%) | 12 (46.2) | 7 (26.9) |
| Diabetes (%) | 3 (11.5) | 3 (11.5) |
| Total fluid infusion (mL) | 2063.5 ± 257.1 | 1932.7 ± 342 |
| Blood loss (mL) | 124.2 ± 50.5 | 123.1 ± 45.2 |
| Urinary output (mL) | 357.7 ± 177 | 330.8 ± 228.1 |
| Vasoactive injections | ||
| Ephedrine (mg) | 6.7 ± 4.7 | 7.4 ± 6.6 |
| Phenylephrine (μg) | 204.6 ± 223.7 | 127.7 ± 176.5 |
| Anesthesia duration (min) | 227.4 ± 35.1 | 218 ± 34.1 |
| Surgery duration (min) | 169.4 ± 29.3 | 169.6 ± 27.4 |
| PEEP (cm H2O) | 5 (5–5) | 16 (12–18) |
| Parameter | Time Points | PEEP5 (n = 26) | PEEPIND (n = 26) | p-Value |
|---|---|---|---|---|
| PaO2/FiO2 (kPa) | PEEP 5 min | 56.6 ± 9.0 | 63.8 ± 14.2 | 0.033 |
| PEEP 60 min | 57.3 ± 14.4 | 65.5 ± 13.1 | 0.036 | |
| Pre-extubation | 56.8 ± 14.1 | 71.6 ± 10.7 | 0.003 | |
| Plateau pressure (cm H2O) | PEEP 5 min | 21.3 ± 5.0 | 27.9 ± 4.33 | 0.000 |
| PEEP 60 min | 21.3 ± 3.0 | 27.6 ± 4.2 | 0.000 | |
| Pre-extubation | 20.0 ± 4.4 | 27.5 ± 3.4 | 0.000 | |
| Driving pressure (cm H2O) | PEEP 5 min | 14.2 ± 3.0 | 13.7 ± 3.6 | 0.586 |
| PEEP 60 min | 15.8 ± 2.6 | 13.4 ± 4.1 | 0.014 | |
| Pre-extubation | 15.0 ± 4.4 | 12.0 ± 3.0 | 0.044 | |
| Cdyn (mL/cm H2O) | PEEP 5 min | 32.6 ± 6.1 | 35.9 ± 7.0 | 0.076 |
| PEEP 60 min | 29.6 ± 5.2 | 37.5 ± 9.2 | 0.000 | |
| Pre-extubation | 33.6 ± 9.1 | 44.5 ± 12.8 | 0.017 | |
| HR | PEEP 5 min | 63.0 ± 8.9 | 64.4 ± 8.7 | 0.562 |
| PEEP 60 min | 66.2 ± 10.0 | 61.4 ± 8.9 | 0.076 | |
| Pre-extubation | 66.2 ± 8.1 | 60.2 ± 8.1 | 0.080 | |
| SBP | PEEP 5 min | 110.6 ± 13.3 | 113.8 ± 12.0 | 0.363 |
| PEEP 60 min | 114.9 ± 23.7 | 117.5 ± 12.6 | 0.632 | |
| Pre-extubation | 117.9 ± 12.3 | 123.8 ± 17.8 | 0.321 | |
| DBP | PEEP 5 min | 59.8 ± 8.9 | 61.0 ± 6.9 | 0.579 |
| PEEP 60 min | 60.3 ± 9.6 | 65.0 ± 10.2 | 0.088 | |
| Pre-extubation | 62.5 ± 8.3 | 65.9 ± 9.1 | 0.315 |
| PEEP | Pw | Pb | ||
|---|---|---|---|---|
| PEEP5 (n = 26) | PEEPIND (n = 26) | |||
| Ventral/dorsal distribution ratio | ||||
| T0 | 1.09 (0.97–1.21) | 1.08 (0.96–1.19) | 0.004 | 0.000 |
| T1 | 1.39 (1.26–1.52) | 0.99 (0.86–1.12) | ||
| T2 | 2.00 (1.53–2.47) | 1.02 (0.57–1.47) | ||
| T3 | 1.27 (1.06–1.47) | 1.17 (0.97–1.37) | ||
| Total (n) | Hypoxemia [n (%)] | Non-Hypoxemia [n (%)] | p-Value | |
|---|---|---|---|---|
| PEEP5 | 26 | 7 (26.9) | 19 (73.1) | 0.021 |
| PEEPIND | 26 | 1 (3.8) | 25 (96.2) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, L.; Yang, L.; Gao, L.; Zhao, Z.; Zhang, J. Individualized PEEP without Recruitment Maneuvers Improves Intraoperative Oxygenation: A Randomized Controlled Study. Bioengineering 2023, 10, 1172. https://doi.org/10.3390/bioengineering10101172
Pan L, Yang L, Gao L, Zhao Z, Zhang J. Individualized PEEP without Recruitment Maneuvers Improves Intraoperative Oxygenation: A Randomized Controlled Study. Bioengineering. 2023; 10(10):1172. https://doi.org/10.3390/bioengineering10101172
Chicago/Turabian StylePan, Lili, Li Yang, Lingling Gao, Zhanqi Zhao, and Jun Zhang. 2023. "Individualized PEEP without Recruitment Maneuvers Improves Intraoperative Oxygenation: A Randomized Controlled Study" Bioengineering 10, no. 10: 1172. https://doi.org/10.3390/bioengineering10101172
APA StylePan, L., Yang, L., Gao, L., Zhao, Z., & Zhang, J. (2023). Individualized PEEP without Recruitment Maneuvers Improves Intraoperative Oxygenation: A Randomized Controlled Study. Bioengineering, 10(10), 1172. https://doi.org/10.3390/bioengineering10101172







