Synthesizing the Effects of Submarine Groundwater Discharge on Marine Biota
Abstract
1. Introduction
2. Phytoplankton and Bacteria
3. Macrophytes
4. Animals
5. Ecosystem Studies
6. Research Priorities
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lecher, A.L. Groundwater Discharge in the Arctic: A Review of Studies and Implications for Biogeochemistry. Hydrology 2017, 4, 41. [Google Scholar] [CrossRef]
- Taniguchi, M.; Burnett, W.C.; Cable, J.E.; Turner, J.V. Investigation of submarine groundwater discharge. Hydrol. Process. 2002, 16, 2115–2129. [Google Scholar] [CrossRef]
- Moore, W.S. The effect of submarine groundwater discharge on the ocean. Ann. Rev. Mar. Sci. 2010, 2, 59–88. [Google Scholar] [CrossRef] [PubMed]
- Moore, W.S. The subterranean estuary: A reaction zone of ground water and sea water. Mar. Chem. 1999, 65, 111–125. [Google Scholar] [CrossRef]
- Slomp, C.P.; Van Cappellen, P. Nutrient inputs to the coastal ocean through submarine groundwater discharge: Controls and potential impact. J. Hydrol. 2004, 295, 64–86. [Google Scholar] [CrossRef]
- Lecher, A.L.; Chien, C.; Paytan, A. Submarine groundwater discharge as a source of nutrients to the North Pacific and Arctic coastal ocean. Mar. Chem. 2016, 186, 167–177. [Google Scholar] [CrossRef]
- Hwang, D.W.; Lee, Y.W.; Kim, G. Large submarine groundwater discharge and benthic eutrophication on Bangdu Bay on volcanic Jeju Island, Korea. Limnol. Oceanogr. 2005, 50, 1393–1403. [Google Scholar] [CrossRef]
- Shellenbarger, G.; Monismith, S.; Genin, A.; Paytan, A. The importance of submarine groundwater discharge to the nearshore nutrient supply in the Gulf of Aqaba (Israel). Limnol. Oceanogr. 2006, 51, 1876–1886. [Google Scholar] [CrossRef]
- Kwon, E.; Kim, G.; Primeau, F.; Moore, W.; Cho, H.-M.; DeVries, T.; Sarmiento, J.; Charette, M.; Cho, Y.-K. Global Estimate of Submarine Groundwater Discharge Based on an Observationally Constrained Radium Isotope Model. Geophys. Res. Lett. 2014, 41, 8438–8444. [Google Scholar] [CrossRef]
- Moore, W.S.; Sarmiento, J.L.; Key, R.M. Submarine groundwater discharge revealed by 228Ra distribution in the upper Atlantic Ocean. Nat. Geosci. 2008, 1, 309–311. [Google Scholar] [CrossRef]
- Black, F.J.; Paytan, A.; Knee, K.L.; De Sieyes, N.R.; Ganguli, P.M.; Gray, E.; Flegal, A.R. Submarine groundwater discharge of total mercury and monomethylmercury to central California coastal waters. Environ. Sci. Technol. 2009, 43, 5652–5659. [Google Scholar] [CrossRef] [PubMed]
- Georg, R.B.; West, A.J.; Basu, A.R.; Halliday, A.N. Silicon fluxes and isotope composition of direct groundwater discharge into the Bay of Bengal and the effect on the global ocean silicon isotope budget. Earth Planet. Sci. Lett. 2009, 283, 67–74. [Google Scholar] [CrossRef]
- Moosdorf, N.; Oehler, T. Societal use of fresh submarine groundwater discharge: An overlooked water resource. Earth-Sci. Rev. 2017, 171, 338–348. [Google Scholar] [CrossRef]
- Shaw, R.D.; Prepas, E.E. Groundwater-lake interactions: I. Accuracy of seepage meter estimates of lake seepage. J. Hydrol. 1990, 119, 105–120. [Google Scholar] [CrossRef]
- Sholkovitz, E.R.; Herbold, C.W.; Charette, M.A. An automated dye-dilution based seepage meter for the time-series measurement of submarine groundwater discharge. Limnol. Oceanogr. Methods 2011, 1, 16–28. [Google Scholar] [CrossRef]
- Taniguchi, M.; Burnett, W.C.; Smith, C.F.; Paulsen, R.J.; O’Rourke, D.; Krupa, S.L.; Christoff, J.L. Spatial and temporal distributions of submarine groundwater discharge rates obtained from various types of seepage meters at a site in the Northeastern Gulf of Mexico. Biogeochemistry 2003, 66, 35–53. [Google Scholar] [CrossRef]
- Moore, W.S. Fifteen years experience in measuring 224Ra and 223Ra by delayed-coincidence counting. Mar. Chem. 2008, 109, 188–197. [Google Scholar] [CrossRef]
- Burnett, W.C.; Peterson, R.; Moore, W.S.; de Oliveira, J. Radon and radium isotopes as tracers of submarine groundwater discharge—Results from the Ubatuba, Brazil SGD assessment intercomparison. Estuar. Coast. Shelf Sci. 2008, 76, 501–511. [Google Scholar] [CrossRef]
- Oehler, T.; Mogollón, J.M.; Moosdorf, N.; Winkler, A.; Kopf, A.; Pichler, T. Submarine groundwater discharge within a landslide scar at the French Mediterranean coast. Estuar. Coast. Shelf Sci. 2017, 198. [Google Scholar] [CrossRef]
- Cook, P.G.; Rodellas, V.; Andrisoa, A.; Stieglitz, T.C. Exchange across the sediment-water interface quantified from porewater radon profiles. J. Hydrol. 2018, 559, 873–883. [Google Scholar] [CrossRef]
- Dulaiova, H.; Camilli, R.; Henderson, P.B.; Charette, M.A. Coupled radon, methane and nitrate sensors for large-scale assessment of groundwater discharge and non-point source pollution to coastal waters. J. Environ. Radioact. 2010, 101, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Paytan, A.; Lecher, A.L.; Dimova, N.; Sparrow, K.J.; Kodovska, F.G.-T.; Murray, J.; Tulaczyk, S.; Kessler, J.D. Methane transport from the active layer to lakes in the Arctic using Toolik Lake, Alaska, as a case study. Proc. Natl. Acad. Sci. USA 2015, 112, 3636–3640. [Google Scholar] [CrossRef] [PubMed]
- Lecher, A.L.; Fisher, A.T.; Paytan, A. Submarine groundwater discharge in Northern Monterey Bay, California: Evaluation by mixing and mass balance models. Mar. Chem. 2016, 179, 44–55. [Google Scholar] [CrossRef]
- Young, M.B.; Gonneea, M.E.; Fong, D.A.; Moore, W.S.; Herrera-Silveira, J.; Paytan, A. Characterizing sources of groundwater to a tropical coastal lagoon in a karstic area using radium isotopes and water chemistry. Mar. Chem. 2008, 109, 377–394. [Google Scholar] [CrossRef]
- Wilson, J.; Rocha, C. Regional scale assessment of Submarine Groundwater Discharge in Ireland combining medium resolution satellite imagery and geochemical tracing techniques. Remote Sens. Environ. 2012, 119, 21–34. [Google Scholar] [CrossRef]
- Amato, D.W.; Bishop, J.M.; Glenn, C.R.; Dulai, H.; Smith, C.M. Impact of submarine groundwater discharge on marine water quality and reef biota of Maui. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Darnell, K.M.; Dunton, K.H. Plasticity in turtle grass (Thalassia testudinum) flower production as a response to porewater nitrogen availability. Aquat. Bot. 2017, 138, 100–106. [Google Scholar] [CrossRef]
- Umezawa, Y.; Miyajima, T.; Yamamuro, M.; Kayanne, H.; Koike, I. Fine-scale mapping of land-derived nitrogen in coral reefs by δ15N in macroalgae. Limnol. Oceanogr. 2002, 47, 1405–1416. [Google Scholar] [CrossRef]
- Lecher, A.L.; Mackey, K.; Kudela, R.; Ryan, J.; Fisher, A.; Murray, J.; Paytan, A. Nutrient loading through submarine groundwater discharge and phytoplankton growth in Monterey bay, CA. Environ. Sci. Technol. 2015, 49. [Google Scholar] [CrossRef] [PubMed]
- Lecher, A.L.; Mackey, K.R.M.; Paytan, A. River and submarine groundwater discharge effects on diatom phytoplankton abundance in the Gulf of Alaska. Hydrology 2017, 4. [Google Scholar] [CrossRef]
- Garcés, E.; Basterretxea, G.; Tovar-Sánchez, A. Changes in microbial communities in response to submarine groundwater input. Mar. Ecol. Prog. Ser. 2011, 438, 47–58. [Google Scholar] [CrossRef]
- Blanco, A.C.; Nadaoka, K.; Yamamoto, T. Planktonic and benthic microalgal community composition as indicators of terrestrial influence on a fringing reef in Ishigaki Island, Southwest Japan. Mar. Environ. Res. 2008, 66, 520–535. [Google Scholar] [CrossRef] [PubMed]
- Gobler, C.J.; Boneillo, G.E. Impacts of anthropogenically influenced groundwater seepage on water chemistry and phytoplankton dynamics within a coastal marine system. Mar. Ecol. Prog. Ser. 2003, 255, 101–114. [Google Scholar] [CrossRef]
- Sugimoto, R.; Kitagawa, K.; Nishi, S.; Honda, H.; Yamada, M.; Kobayashi, S.; Shoji, J.; Ohsawa, S.; Taniguchi, M.; Tominaga, O. Phytoplankton primary productivity around submarine groundwater discharge in nearshore coasts. Mar. Ecol. Prog. Ser. 2017, 563, 25–33. [Google Scholar] [CrossRef]
- Paerl, H.W. Coastal eutrophication and harmful algal blooms: Importance of atmospheric deposition and groundwater as “new” nitrogen and other nutrient sources. Limnol. Oceanogr. 1997, 42, 1154–1165. [Google Scholar] [CrossRef]
- Laroche, J.; Nuzzi, R.; Waters, R.; Wyman, K.; Falkowski, P.; Wallace, D. Brown Tide blooms in Long Island’s coastal waters linked to interannual variability in groundwater flow. Glob. Chang. Biol. 1997, 3, 397–410. [Google Scholar] [CrossRef]
- Su, N.; Burnett, W.C.; Eller, K.T.; MacIntyre, H.L.; Mortazavi, B.; Leifer, J.; Novoveska, L. Radon and radium isotopes, groundwater discharge and harmful algal blooms in Little Lagoon, Alabama. In Interdisciplinary Studies on Environmental Chemistry; Terrapub: Tokyo, Japan, 2012; Volume 6, pp. 329–338. [Google Scholar]
- Liefer, J.D.; MacIntyre, H.L.; Novoveská, L.; Smith, W.L.; Dorsey, C.P. Temporal and spatial variability in Pseudo-nitzschia spp. in Alabama coastal waters: A “hot spot” linked to submarine groundwater discharge? Harmful Algae 2009, 8, 706–714. [Google Scholar] [CrossRef]
- Lee, E.; Shin, D.; Hyun, S.P.; Ko, K.-S.; Moon, H.S.; Koh, D.-C.; Ha, K.; Kim, B.-Y. Periodic change in coastal microbial community structure associated with submarine groundwater discharge and tidal fluctuation. Limnol. Oceanogr. 2017, 62, 437–451. [Google Scholar] [CrossRef]
- Troccoli-Ghinaglia, L.; Herrera-Silveira, J.A.; Comín, F.A.; Díaz-Ramos, J.R. Phytoplankton community variations in tropical coastal area affected where submarine groundwater occurs. Cont. Shelf Res. 2010, 30, 2082–2091. [Google Scholar] [CrossRef]
- Short, F.T.; Burdick, D.M. Quantifying Eelgrass Habitat Loss in Relation to Housing Development and Nitrogen Loading in Waquoit Bay, Massachusetts. Estuaries 1996, 19, 730–739. [Google Scholar] [CrossRef]
- Crook, E.D.; Cohen, A.L.; Rebolledo-Vieyra, M.; Hernandez, L.; Paytan, A. Reduced calcification and lack of acclimatization by coral colonies growing in areas of persistent natural acidification. Proc. Natl. Acad. Sci. USA 2013, 110, 11044–11049. [Google Scholar] [CrossRef] [PubMed]
- Pironet, F.N.; Jones, J.B. Treatments for ectoparasites and diseases in captive Western Australian dhufish. Aquac. Int. 2000, 8, 349–361. [Google Scholar] [CrossRef]
- Wang, X.; Li, H.; Zheng, C.; Yang, J.; Zhang, Y.; Zhang, M.; Qi, Z.; Xiao, K.; Zhang, X. Submarine groundwater discharge as an important nutrient source influencing nutrient structure in coastal water of Daya Bay, China. Geochim. Cosmochim. Acta 2018, 225, 52–65. [Google Scholar] [CrossRef]
- de Sieyes, N.; Yamahara, K.; Layton, B.; Joyce, E.; Boehm, A. Submarine discharge of nutrient-enriched fresh groundwater at Stinson Beach, California is enhanced during neap tides. Limnol. Ocean. 2008, 53, 1434–1445. [Google Scholar] [CrossRef]
- Ryan, J.P.; Gower, J.F.R.; King, S.A.; Bissett, W.P.; Fischer, A.M.; Kudela, R.M.; Kolber, Z.; Mazzillo, F.; Rienecker, E.V.; Chavez, F.P. A coastal ocean extreme bloom incubator. Geophys. Res. Lett. 2008, 35, L12602. [Google Scholar] [CrossRef]
- Peterson, B.J.; Stubler, A.D.; Wall, C.C.; Gobler, C.J. Nitrogen-rich groundwater intrusion affects productivity, but not herbivory, of the tropical seagrass thalassia testudinum. Aquat. Biol. 2012, 15, 1–9. [Google Scholar] [CrossRef]
- Futch, J.C.; Griffin, D.W.; Lipp, E.K. Human enteric viruses in groundwater indicate offshore transport of human sewage to coral reefs of the Upper Florida Keys. Environ. Microbiol. 2010, 12, 964–974. [Google Scholar] [CrossRef] [PubMed]
- Perry, M.J. Alkaline phosphatase activity in subtropical Central North Pacific waters using a sensitive fluorometric method. Mar. Biol. 1972, 15, 113–119. [Google Scholar] [CrossRef]
- Pichard, S.L.; Paul, J.H. Detection of gene expression in genetically engineered microorganisms and natural phytoplankton populations in the marine environment by mRNA analysis. Appl. Environ. Microbiol. 1991, 57, 1721–1727. [Google Scholar] [PubMed]
- Nielsen, E.S. The use of radio-active carbon (c14) for measuring organic production in the sea. ICES J. Mar. Sci. 1952, 18, 117–140. [Google Scholar] [CrossRef]
- Grande, K.D.; Williams, P.J.L.; Marra, J.; Purdie, D.A.; Heinemann, K.; Eppley, R.W.; Bender, M.L. Primary production in the North Pacific gyre: A comparison of rates determined by the 14C, O2 concentration and 18O methods. Deep Sea Res. Part A Oceanogr. Res. Pap. 1989, 36, 1621–1634. [Google Scholar] [CrossRef]
- Vieira, S.; Ribeiro, L.; Jesus, B.; Cartaxana, P.; Silva, J.M. Da Photosynthesis assessment in microphytobenthos using conventional and imaging pulse amplitude modulation fluorometry. Photochem. Photobiol. 2013, 89, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Schlitzer, R. Ocean Data View. 2018. odv.awi.de.
- Lapointe, B.E. Nutrient thresholds for bottom-up control of macroalgal blooms on coral reefs in Jamaica and southeast Florida. Limnol. Oceanogr. 1997, 42, 1119–1131. [Google Scholar] [CrossRef]
- Lee, Y.-W.; Kim, G.; Lim, W.-A.; Hwang, D.-W. A relationship between submarine groundwater borne nutrients traced by Ra isotopes and the intensity of dinoflagellate red-tides occurring in the southern sea of Korea. Limnol. Oceanogr. 2010, 55, 1–10. [Google Scholar] [CrossRef]
- Waska, H.; Kim, G. Differences in microphytobenthos and macrofaunal abundances associated with groundwater discharge in the intertidal zone. Mar. Ecol. Prog. Ser. 2010, 407, 159–172. [Google Scholar] [CrossRef]
- Waska, H.; Kim, G. Submarine groundwater discharge (SGD) as a main nutrient source for benthic and water-column primary production in a large intertidal environment of the Yellow Sea. J. Sea Res. 2011, 65, 103–113. [Google Scholar] [CrossRef]
- Lee, Y.W.; Kim, G. Linking groundwater-borne nutrients and dinoflagellate red-tide outbreaks in the southern sea of Korea using a Ra tracer. Estuar. Coast. Shelf Sci. 2007, 71, 309–317. [Google Scholar] [CrossRef]
- Derse, E.; Knee, K.L.; Wankel, S.D.; Kendall, C.; Berg, C.J.; Paytan, A. Identifying sources of nitrogen to Hanalei Bay, Kauai, utilizing the nitrogen isotope signature of macroalgae. Environ. Sci. Technol. 2007, 41, 5217–5223. [Google Scholar] [CrossRef] [PubMed]
- Richardson, C.M.; Dulai, H.; Popp, B.N.; Ruttenberg, K.; Fackrell, J.K. Submarine groundwater discharge drives biogeochemistry in two Hawaiian reefs. Limnol. Oceanogr. 2017, 62, S348–S363. [Google Scholar] [CrossRef]
- Knee, K.L.; Layton, B.A.; Street, J.H.; Boehm, A.B.; Paytan, A. Sources of Nutrients and Fecal Indicator Bacteria to Nearshore Waters on the North Shore of Kaua`i (Hawai`i, USA). Estuaries Coasts 2008, 31, 607–622. [Google Scholar] [CrossRef]
- Lubarsky, K.A.; Silbiger, N.J.; Donahue, M.J. Effects of submarine groundwater discharge on coral accretion and bioerosion on two shallow reef flats. Limnol. Oceanogr. 2018, 63, 1660–1672. [Google Scholar] [CrossRef]
- Prouty, N.G.; Cohen, A.; Yates, K.K.; Storlazzi, C.D.; Swarzenski, P.W.; White, D. Vulnerability of Coral Reefs to Bioerosion From Land-Based Sources of Pollution. J. Geophys. Res. Ocean. 2017, 122, 9319–9331. [Google Scholar] [CrossRef]
- Prouty, N.G.; Yates, K.K.; Smiley, N.; Gallagher, C.; Cheriton, O.; Storlazzi, C.D. Carbonate system parameters of an algal-dominated reef along West Maui. Biogeosciences 2018, 15, 2467–2480. [Google Scholar] [CrossRef]
- McMahon, A.; Santos, I.R. Nitrogen enrichment and speciation in a coral reef lagoon driven by groundwater inputs of bird guano. J. Geophys. Res. Ocean. 2017, 122, 7218–7236. [Google Scholar] [CrossRef]
- Boehm, A.B.; Shellenbarger, G.G.; Paytan, A. Groundwater discharge: Potential association with fecal indicator bacteria in the surf zone. Environ. Sci. Technol. 2004, 38, 3558–3566. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A.E.; Boehm, A.B.; Francis, C.A. Denitrifier community composition along a nitrate and salinity gradient in a coastal aquifer. Appl. Environ. Microbiol. 2006, 72, 2102–2109. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A.E.; Francis, C.A.; De Sieyes, N.R.; Boehm, A.B. Shifts in the relative abundance of ammonia-oxidizing bacteria and archaea across physicochemical gradients in a subterranean estuary. Environ. Microbiol. 2008, 10, 1068–1079. [Google Scholar] [CrossRef] [PubMed]
- Blanco, A.C.; Watanabe, A.; Nadaoka, K.; Motooka, S.; Herrera, E.C.; Yamamoto, T. Estimation of nearshore groundwater discharge and its potential effects on a fringing coral reef. Mar. Pollut. Bull. 2011, 62, 770–785. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.K.; Kang, H.; Oh, Y.H.; Park, S.R.; Kim, G. Green tide development associated with submarine groundwater discharge in a coastal harbor, Jeju, Korea. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Lapointe, B.E.; Barile, P.J.; Littler, M.M.; Littler, D.S. Macroalgal blooms on southeast Florida coral reefs: II. Cross-shelf discrimination of nitrogen sources indicates widespread assimilation of sewage nitrogen. Harmful Algae 2005, 4, 1106–1122. [Google Scholar] [CrossRef]
- Hu, C.; Muller-Karger, F.E.; Swarzenski, P.W. Hurricanes, submarine groundwater discharge, and Florida’s red tides. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Lirman, D.; Orlando, B.; Maciá, S.; Manzello, D.; Kaufman, L.; Biber, P.; Jones, T. Coral communities of Biscayne Bay, Florida and adjacent offshore areas: Diversity, abundance, distribution, and environmental correlates. Aquat. Conserv. Mar. Freshw. Ecosyst. 2003, 13, 121–135. [Google Scholar] [CrossRef]
- Kohout, F.A.; Kolipinski, M.C. Biological zonation related to groundwater discharge along the shore of Biscayne Bay, Miami, Florida. In Estuaries; Lauff, G., Ed.; AAAS: Washington, DC, USA, 1967; pp. 488–499. [Google Scholar]
- Kotwicki, L.; Grzelak, K.; Czub, M.; Dellwig, O.; Gentz, T.; Szymczycha, B.; Böttcher, M.E. Submarine groundwater discharge to the Baltic coastal zone: Impacts on the meiofaunal community. J. Mar. Syst. 2014, 129, 118–126. [Google Scholar] [CrossRef]
- Donis, D.; Janssen, F.; Liu, B.; Wenzhöfer, F.; Dellwig, O.; Escher, P.; Spitzy, A.; Böttcher, M.E. Biogeochemical impact of submarine ground water discharge on coastal surface sands of the southern Baltic Sea. Estuar. Coast. Shelf Sci. 2017, 189, 131–142. [Google Scholar] [CrossRef]
- Gobler, C.J.; Sañudo-Wilhelmy, S.A. Temporal variability of groundwater seepage and brown tide blooms in a Long Island embayment. Mar. Ecol. Prog. Ser. 2001, 217, 299–309. [Google Scholar] [CrossRef]
- Grzelak, K.; Tamborski, J.; Kotwicki, L.; Bokuniewicz, H. Ecostructuring of marine nematode communities by submarine groundwater discharge. Mar. Environ. Res. 2018, 136, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, M.; Tominaga, O.; Sugimoto, R.; Kitagawa, K.; Yamanda, M.; Honda, H.; Shoki, J.; Kobayashi, S.; Ikuta, K.; Taniguchi, M. Evaluation of the environmental condition of Submarine Groundwater Discharge (SGD) by using nitrate stable isotope ratio recorded in sea lettuce (Ulva sp.). In Proceedings of the JSFS 85th Anniversary-Commemorative International Symposim “Fisheries Science for Future Generations”, Tokyo, Japan, 22–24 September 2017; p. 5005. [Google Scholar]
- Honda, H.; Sugimoto, R.; Kobayashi, S. Submarine Groundwater Discharge and its Influence on Primary Production in Japanese Coasts: Case Study in Obama Bay. In The Water-Energy-Food Nexus; Springer: Singapore, 2018; pp. 101–115. [Google Scholar]
- Utsunomiya, T.; Hata, M.; Sugimoto, R.; Honda, H.; Kobayashi, S.; Miyata, Y.; Yamada, M.; Tominaga, O.; Shoji, J.; Taniguchi, M. Higher species richness and abundance of fish and benthic invertebrates around submarine groundwater discharge in Obama Bay, Japan. J. Hydrol. Reg. Stud. 2017, 11, 139–146. [Google Scholar] [CrossRef]
- Hata, M.; Sugimoto, R.; Hori, M.; Tomiyama, T.; Shoji, J. Occurrence, distribution and prey items of juvenile marbled sole Pseudopleuronectes yokohamae around a submarine groundwater seepage on a tidal flat in southwestern Japan. J. Sea Res. 2016, 111, 47–53. [Google Scholar] [CrossRef]
- Su, N.; Burnett, W.C.; MacIntyre, H.L.; Liefer, J.D.; Peterson, R.N.; Viso, R. Natural Radon and Radium Isotopes for Assessing Groundwater Discharge into Little Lagoon, AL: Implications for Harmful Algal Blooms. Estuaries Coasts 2014, 37, 893–910. [Google Scholar] [CrossRef]
- Welti, N.; Gale, D.; Hayes, M.; Kumar, A.; Gasparon, M.; Gibbes, B.; Lockington, D. Intertidal diatom communities reflect patchiness in groundwater discharge. Estuar. Coast. Shelf Sci. 2015, 163, 116–124. [Google Scholar] [CrossRef]
- Sewell, P.L. Urban groundwater as a possible nutrient source for an estuarine benthic algal bloom. Estuar. Coast. Shelf Sci. 1982, 15, 569–576. [Google Scholar] [CrossRef]
- Machado, D.A.; Imberger, J. Modeling the impact of natural and anthropogenic nutrient sources on phytoplankton dynamics in a shallow coastal domain, Western Australia. Environ. Fluid Mech. 2014, 14, 87–111. [Google Scholar] [CrossRef]
- Sospedra, J.; Niencheski, L.F.H.; Falco, S.; Andrade, C.F.F.; Attisano, K.K.; Rodilla, M. Identifying the main sources of silicate in coastal waters of the Southern Gulf of Valencia (Western Mediterranean Sea). Oceanologia 2018, 60, 52–64. [Google Scholar] [CrossRef]
- Valiela, I.; Foreman, K.; LaMontagne, M.; Hersh, D.; Costa, J.; Peckol, P.; DeMeo-Andreson, B.; D’Avanzo, C.; Babione, M.; Sham, C.-H.; et al. Couplings of Watersheds and Coastal Waters: Sources and Consequences of Nutrient Enrichment in Waquoit Bay, Massachusetts. Estuaries 1992, 15, 443–457. [Google Scholar] [CrossRef]
- McClelland, J.W.; Valiela, I.; Michener, R.H. Nitrogen-stable isotope signatures in estuarine food webs: A record of increasing urbanization in coastal watersheds. Limnol. Oceanogr. 1997, 42, 930–937. [Google Scholar] [CrossRef]
- Rogers, D.R.; Casciotti, K.L. Abundance and diversity of archaeal ammonia oxidizers in a coastal groundwater system. Appl. Environ. Microbiol. 2010, 76, 7938–7948. [Google Scholar] [CrossRef] [PubMed]
- Carruthers, T.J.B.; Van Tussenbroek, B.I.; Dennison, W.C. Influence of submarine springs and wastewater on nutrient dynamics of Caribbean seagrass meadows. Estuar. Coast. Shelf Sci. 2005, 64, 191–199. [Google Scholar] [CrossRef]
- Álvarez-Góngora, C.; Herrera-Silveira, J.A. Variations of phytoplankton community structure related to water quality trends in a tropical karstic coastal zone. Mar. Pollut. Bull. 2006, 52, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Crook, E.D.; Potts, D.; Rebolledo-Vieyra, M.; Hernandez, L.; Paytan, A. Calcifying coral abundance near low-pH springs: Implications for future ocean acidification. Coral Reefs 2012, 31, 239–245. [Google Scholar] [CrossRef]
- Martinez, A.; Hernandez-Terrones, L.; Rebolledo-Vieyra, M.; Paytan, A. Impact of carbonate saturation on large Caribbrean benthic foraminifera assemblages. Biogeosci. Discuss. 2018. [Google Scholar] [CrossRef]
- Carvalho, L.F.; Rocha, C.; Fleming, A.; Veiga-Pires, C.; Aníbal, J. Interception of nutrient rich submarine groundwater discharge seepage on European temperate beaches by the acoel flatworm, Symsagittifera roscoffensis. Mar. Pollut. Bull. 2013, 75, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Encarnação, J.; Leitão, F.; Range, P.; Piló, D.; Chícharo, M.A.; Chícharo, L. The influence of submarine groundwater discharges on subtidal meiofauna assemblages in south Portugal (Algarve). Estuar. Coast. Shelf Sci. 2013, 130, 202–208. [Google Scholar] [CrossRef]
- Encarnação, J.; Leitão, F.; Range, P.; Piló, D.; Chícharo, M.A.; Chícharo, L. Local and temporal variations in near-shore macrobenthic communities associated with submarine groundwater discharges. Mar. Ecol. 2015, 36, 926–941. [Google Scholar] [CrossRef]
- Silva, A.C.F.; Tavares, P.; Shapouri, M.; Stigter, T.Y.; Monteiro, J.P.; Machado, M.; Cancela da Fonseca, L.; Ribeiro, L. Estuarine biodiversity as an indicator of groundwater discharge. Estuar. Coast. Shelf Sci. 2012, 97, 38–43. [Google Scholar] [CrossRef]
- Piló, D.; Barbosa, A.B.; Teodósio, M.A.; Encarnação, J.; Leitão, F.; Range, P.; Krug, L.A.; Cruz, J.; Chícharo, L. Are submarine groundwater discharges affecting the structure and physiological status of rocky intertidal communities? Mar. Environ. Res. 2018, 136, 158–173. [Google Scholar] [CrossRef] [PubMed]
- Gagan, M.K.; Ayliffe, L.K.; Opdyke, B.N.; Hopley, D.; Scott-Gagan, H.; Cowley, J. Coral oxygen isotope evidence for recent groundwater fluxes to the Australian Great Barrier Reef. Geophys. Res. Lett. 2002, 29, 43-1–43-4. [Google Scholar] [CrossRef]
- Miller, D.C.; Ullman, W.J. Ecological Consequences of Ground Water Discharge to Delaware Bay, United States. Ground Water 2004, 42, 959–970. [Google Scholar] [CrossRef]
- Dale, R.K.; Miller, D.C. Hydrologic interactions of infaunal polychaetes and intertidal groundwater discharge. Mar. Ecol. Prog. Ser. 2008, 363, 205–215. [Google Scholar] [CrossRef]
- Ouisse, V.; Riera, P.; Migné, A.; Leroux, C.; Davoult, D. Freshwater seepages and ephemeral macrolagae proliferation in an intertidal bay: I Effect on benthic community structure and food web. Estuar. Coast. Shelf Sci. 2011, 91, 272–281. [Google Scholar] [CrossRef]
- Migné, A.; Ouisse, V.; Hubas, C.; Davoult, D. Freshwater seepages and ephemeral macroalgae proliferation in an intertidal bay: II. Effect on benthic biomass and metabolism. Estuar. Coast. Shelf Sci. 2011, 92, 161–168. [Google Scholar] [CrossRef]
- Zipperle, A.; Reise, K. Freshwater springs on intertidal sand flats cause a switch in dominance among polychaete worms. J. Sea Res. 2005, 54, 143–150. [Google Scholar] [CrossRef]
- Bussmann, I.; Dando, P.R.; Niven, S.J.; Suess, E. Groundwater seepage in the marine environment: Role for mass flux and bacterial activity. Mar. Ecol. Prog. Ser. 1999, 178, 169–177. [Google Scholar] [CrossRef]
- Erler, D.V.; Shepherd, B.O.; Linsley, B.K.; Lough, J.M.; Cantin, N.E. Coral skeletons record increasing agricultural related groundwater nitrogen inputs to a South Pacific reef over the past century. Geophys. Res. Lett. 2018. [Google Scholar] [CrossRef]
- Kamermans, P.; Hemminga, M.A.; Tack, J.F.; Mateo, M.A.; Marbà, N.; Mtolera, M.; Stapel, J.; Verheyden, A.; Van Daele, T. Groundwater effects on diversity and abundance of lagoonal seagrasses in Kenya and on Zanzibar Island (East Africa). Mar. Ecol. Prog. Ser. 2002, 231, 75–83. [Google Scholar] [CrossRef]
- Jiang, W.; Yu, K.; Song, Y.; Zhao, J.X.; Feng, Y.X.; Wang, Y.; Xu, S. Coral geochemical record of submarine groundwater discharge back to 1870 in the northern South China Sea. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2018, 507, 30–38. [Google Scholar] [CrossRef]
- Sekerci, Y.; Petrovskii, S. Mathematical Modelling of Plankton–Oxygen Dynamics Under the Climate Change. Bull. Math. Biol. 2015, 77, 2325–2353. [Google Scholar] [CrossRef] [PubMed]
- Lomas, M.W.; Gilbert, P.M. Comparisons of nitrate uptake, storage, and reduction in marine diatoms and flagellates. J. Phycol. 2000, 36, 903–913. [Google Scholar] [CrossRef]
- Kim, G.; Ryu, J.W.; Yang, H.S.; Yun, S.T. Submarine groundwater discharge (SGD) into the Yellow Sea revealed by228Ra and226Ra isotopes: Implications for global silicate fluxes. Earth Planet. Sci. Lett. 2005, 237, 156–166. [Google Scholar] [CrossRef]
- Brzezinski, M.A. The Si:C:N of marine diatoms: Interspecific variability and the effect of some environmental variables. J. Phycol. 1985, 21, 347–357. [Google Scholar] [CrossRef]
- Spillane, T. Diatom Frustules as a Mechanical Defense against Predation by Heterotrophic Dinoflagellates. Master’s Thesis, Western Washington University, Bellingham, WA, USA, February 2016. [Google Scholar]
- Anschutz, P.; Smith, T.; Mouret, A.; Deborde, J.; Bujan, S.; Poirier, D.; Lecroart, P. Tidal sands as biogeochemical reactors. Estuar. Coast. Shelf Sci. 2009, 84, 84–90. [Google Scholar] [CrossRef]
- Tréguer, P.J.; De La Rocha, C.L. The World Ocean Silica Cycle. Ann. Rev. Mar. Sci. 2013, 5, 477–501. [Google Scholar] [CrossRef] [PubMed]
- Trainer, V.L.; Bates, S.S.; Lundholm, N.; Thessen, A.E.; Cochlan, W.P.; Adams, N.G.; Trick, C.G. Pseudo-nitzschia physiological ecology, phylogeny, toxicity, monitoring and impacts on ecosystem health. Harmful Algae 2012, 14, 271–300. [Google Scholar] [CrossRef]
- Bates, S.; Garrison, D.; Horner, R. Bloom dynamics and physiology producing pseudo-nitzschia species. In Physioligical Ecology of Harmful Algal Blooms; Springer: Heidelberg, Germany, 1998; pp. 267–292. [Google Scholar]
- McCabe, R.M.; Hickey, B.M.; Kudela, R.M.; Lefebvre, K.A.; Adams, N.G.; Bill, B.D.; Gulland, F.M.D.; Thomson, R.E.; Cochlan, W.P.; Trainer, V.L. An unprecedented coastwide toxic algal bloom linked to anomalous ocean conditions. Geophys. Res. Lett. 2016, 43, 10366–10376. [Google Scholar] [CrossRef] [PubMed]
- Schnetzer, A.; Miller, P.E.; Schaffner, R.A.; Stauffer, B.A.; Jones, B.H.; Weisberg, S.B.; DiGiacomo, P.M.; Berelson, W.M.; Caron, D.A. Blooms of Pseudo-nitzschia and domic acid in the San Pedro Channel and Low Angeles harbor areas of the Southern California Bight, 2003–2004. Harmful Algae 2007, 6, 371–387. [Google Scholar] [CrossRef]
- Mackey, K.R.M.; Mioni, C.E.; Ryan, J.P.; Paytan, A. Phosphorus cycling in the red tide incubator region of monterey bay in response to upwelling. Front. Microbiol. 2012, 3, 33. [Google Scholar] [CrossRef] [PubMed]
- Hibberd, D.J.; Greenwood, A.D.; Bronwen Griffiths, H. Observations on the ultrastructure of the flagella and periplast in the cryptophyceae. Br. Phycol. J. 1971, 6, 61–72. [Google Scholar] [CrossRef]
- Patterson, K.L.; Porter, J.W.; Ritchie, K.B.; Polson, S.W.; Mueller, E.; Peters, E.C.; Santavy, D.L.; Smith, G.W. The etiology of white pox, a lethal disease of the Caribbean elkhorn coral, Acropora palmata. Proc. Natl. Acad. Sci. USA 2002, 99, 8725–8730. [Google Scholar] [CrossRef] [PubMed]
- Teichberg, M.; Fox, S.E.; Olsen, Y.S.; Valiela, I.; Martinetto, P.; Iribarne, O.; Muto, E.Y.; Petti, M.A.V.; Corbisier, T.N.; Soto-Jiménez, M.; et al. Eutrophication and macroalgal blooms in temperate and tropical coastal waters: Nutrient enrichment experiments with Ulva spp. Glob. Chang. Biol. 2010, 16, 2624–2637. [Google Scholar] [CrossRef]
- Giere, O. Meiobenthology: The Microscopic Motile Fauna of Aquatic Sediments; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 9783540686613. [Google Scholar]
- Richardson, C.M.; Dulai, H.; Whittier, R.B. Sources and spatial variability of groundwater-delivered nutrients in Maunalua Bay, O‘ahu, Hawai‘i. J. Hydrol. Reg. Stud. 2017, 11, 178–193. [Google Scholar] [CrossRef]
- Bishop, J.M.; Glenn, C.R.; Amato, D.W.; Dulai, H. Effect of land use and groundwater flow path on submarine groundwater discharge nutrient flux. J. Hydrol. Reg. Stud. 2017, 11, 194–218. [Google Scholar] [CrossRef]
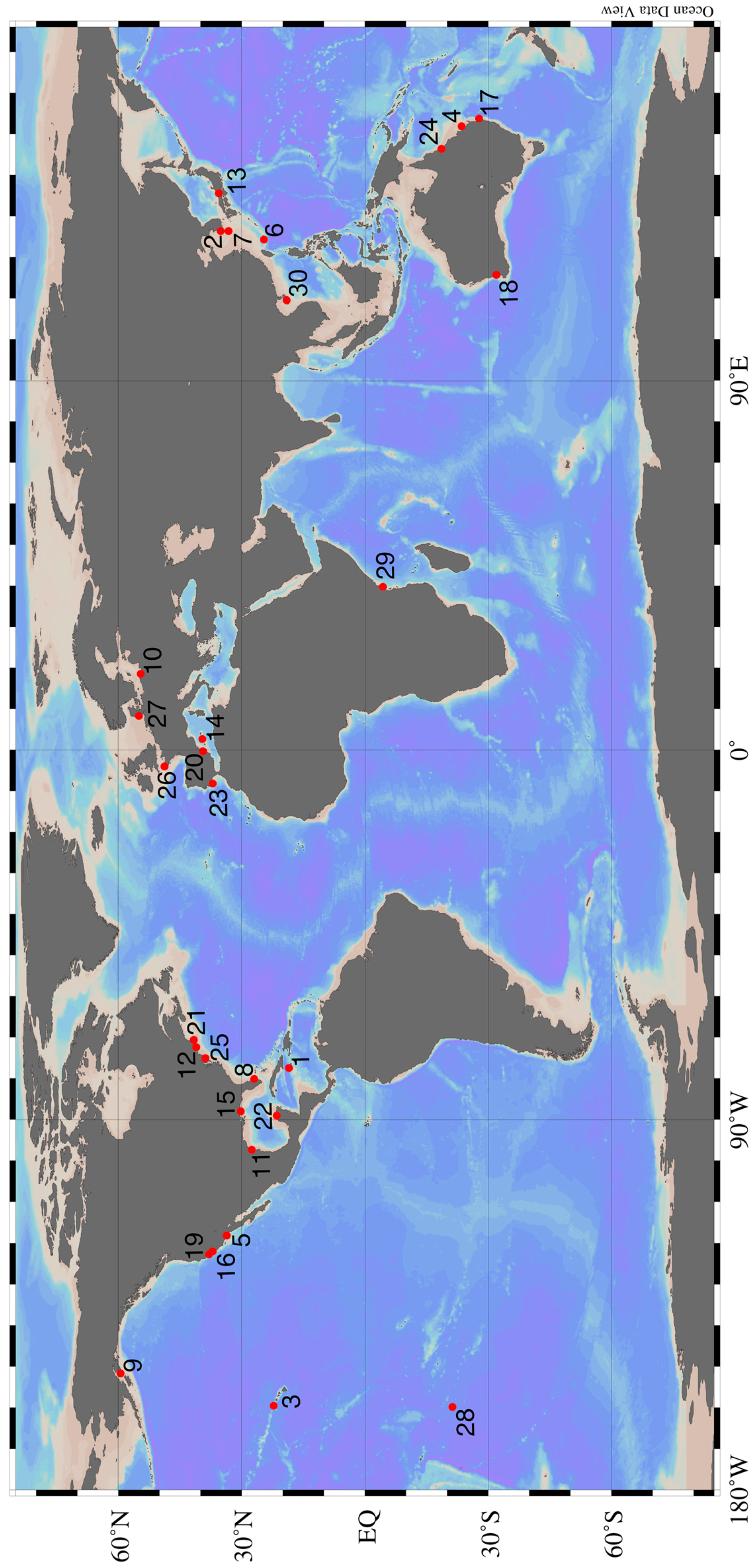

| Figure 1 Number | Study Site | References | Biota Studied |
|---|---|---|---|
| 1 | Discovery Bay, Jamaica | [47,55] | Macrophytes |
| 2 | Various sites on the Korean Peninsula | [56,57,58,59] | Phytoplankton & Bacteria, Animals |
| 3 | Various sites around Hawaii | [26,60,61,62,63,64,65] | Phytoplankton & Bacteria, Macrophytes, Animals, Ecosystem Studies |
| 4 | Heron Island, Australia | [66] | Ecosystem Study |
| 5 | Huntington Beach, California | [67,68,69] | Phytoplankton & Bacteria |
| 6 | Ishigaki Island, Japan | [28,32,70] | Macrophytes, Phytoplankton & Bacteria |
| 7 | Jeju Island, Korea | [7,39,71] | Macrophytes, Phytoplankton & Bacteria |
| 8 | Various sites on the Florida Peninsula | [48,55,72,73,74,75] | Macrophytes, Phytoplankton & Bacteria, Animals |
| 9 | Kasitsna Bay, Alaska | [30] | Phytoplankton & Bacteria |
| 10 | Puck Bay, Poland | [76,77] | Animals, Ecosystem Study |
| 11 | Laguna Madre, Texas | [27] | Macrophytes |
| 12 | Long Island, New York | [33,36,78,79] | Phytoplankton & Bacteria, Animals |
| 13 | Various sites around mainland Japan | [80,81,82,83] | Phytoplankton & Bacteria, Macrophytes, Animals, Ecosystem Study |
| 14 | Majorca, Spain | [31] | Phytoplankton & Bacteria |
| 15 | Mobile Bay, Alabama | [37,38,84] | Phytoplankton & Bacteria |
| 16 | Monterey Bay, California | [29] | Phytoplankton & Bacteria |
| 17 | North Stadbroke Island, Australia | [85] | Phytoplankton & Bacteria |
| 18 | Perth, Australia | [86,87] | Macrophytes, Phytoplankton & Bacteria |
| 19 | Stinson Beach, California | [45] | Phytoplankton & Bacteria |
| 20 | Valencia, Spain | [88] | Phytoplankton & Bacteria |
| 21 | Waquoit Bay, Massachusetts | [41,89,90,91] | Phytoplankton & Bacteria, Macrophytes, Animals, Ecosystem Study |
| 22 | Various sites on the Yucatán Peninsula | [40,42,92,93,94,95] | Macrophytes, Phytoplankton & Bacteria, Animals |
| 23 | Various sites around Portugal | [96,97,98,99,100] | Animals |
| 24 | Orpheus Island, Australia | [101] | Animals |
| 25 | Delaware Bay, USA | [102,103] | Animals |
| 26 | Roscoff Aber Bay, France | [104,105] | Animals |
| 27 | Various sites Around Germany | [106,107] | Animals, Phytoplankton & Bacteria |
| 28 | Rarotonga, Cook Islands | [108] | Animals |
| 29 | Various sites in southern Kenya and Zanzibar Island | [109] | Macrophytes |
| 30 | South China Sea | [110] | Animals |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lecher, A.L.; Mackey, K.R.M. Synthesizing the Effects of Submarine Groundwater Discharge on Marine Biota. Hydrology 2018, 5, 60. https://doi.org/10.3390/hydrology5040060
Lecher AL, Mackey KRM. Synthesizing the Effects of Submarine Groundwater Discharge on Marine Biota. Hydrology. 2018; 5(4):60. https://doi.org/10.3390/hydrology5040060
Chicago/Turabian StyleLecher, Alanna L., and Katherine R. M. Mackey. 2018. "Synthesizing the Effects of Submarine Groundwater Discharge on Marine Biota" Hydrology 5, no. 4: 60. https://doi.org/10.3390/hydrology5040060
APA StyleLecher, A. L., & Mackey, K. R. M. (2018). Synthesizing the Effects of Submarine Groundwater Discharge on Marine Biota. Hydrology, 5(4), 60. https://doi.org/10.3390/hydrology5040060





