Quantifying Processes Governing Nutrient Concentrations in a Coastal Aquifer via Principal Component Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Approach
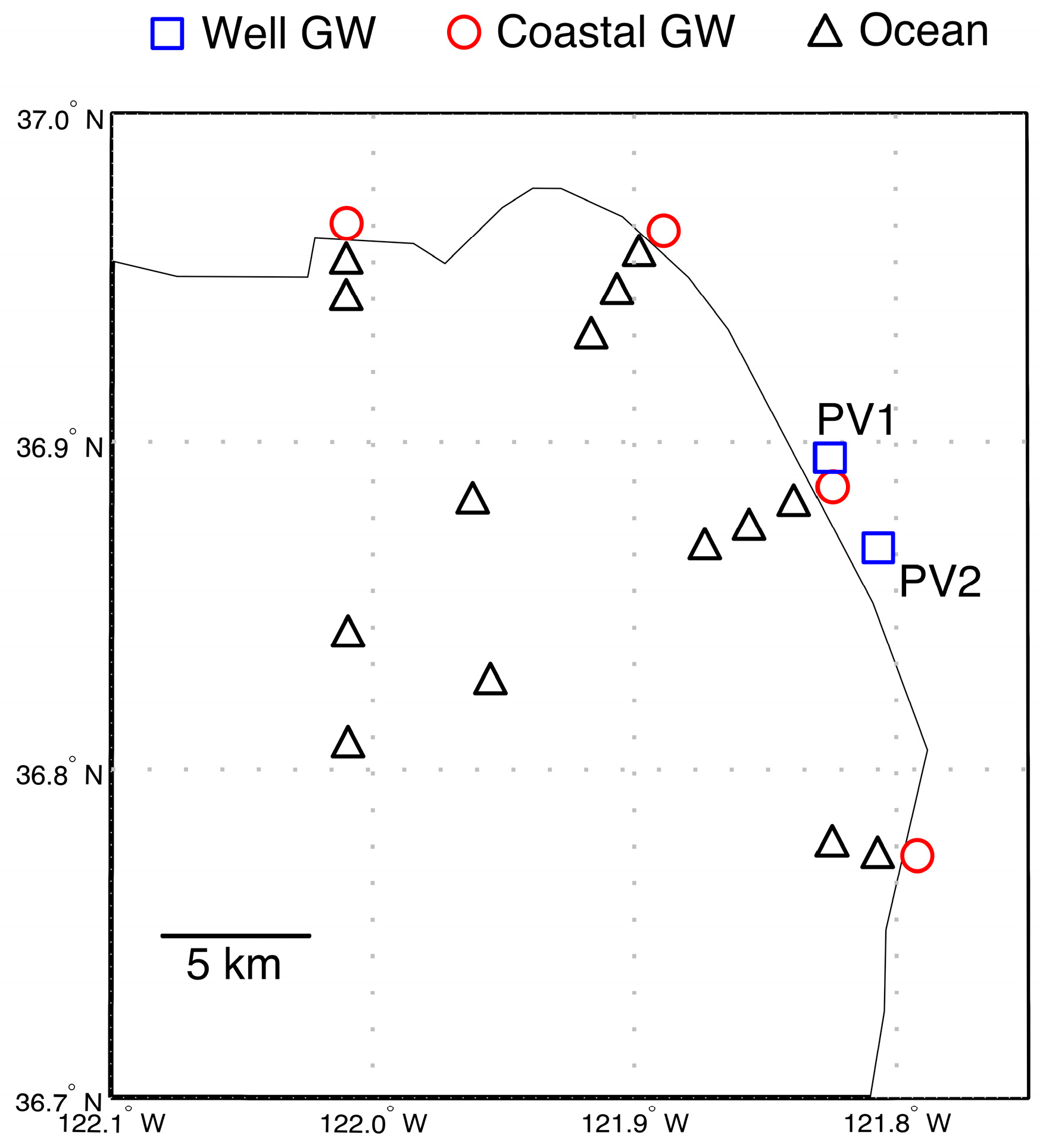
2.2. Sample Collection
2.3. Analytical Methods
2.4. Statistical Analyses
3. Results
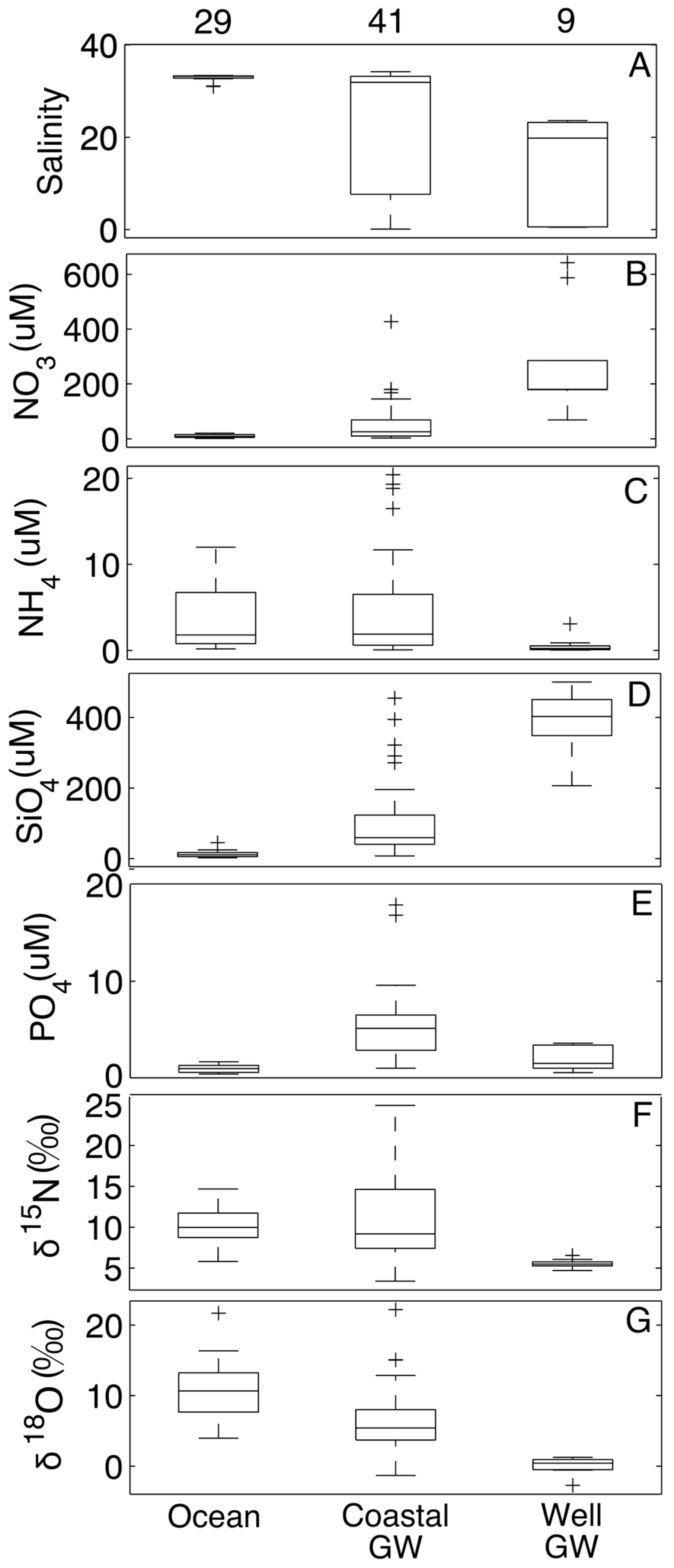
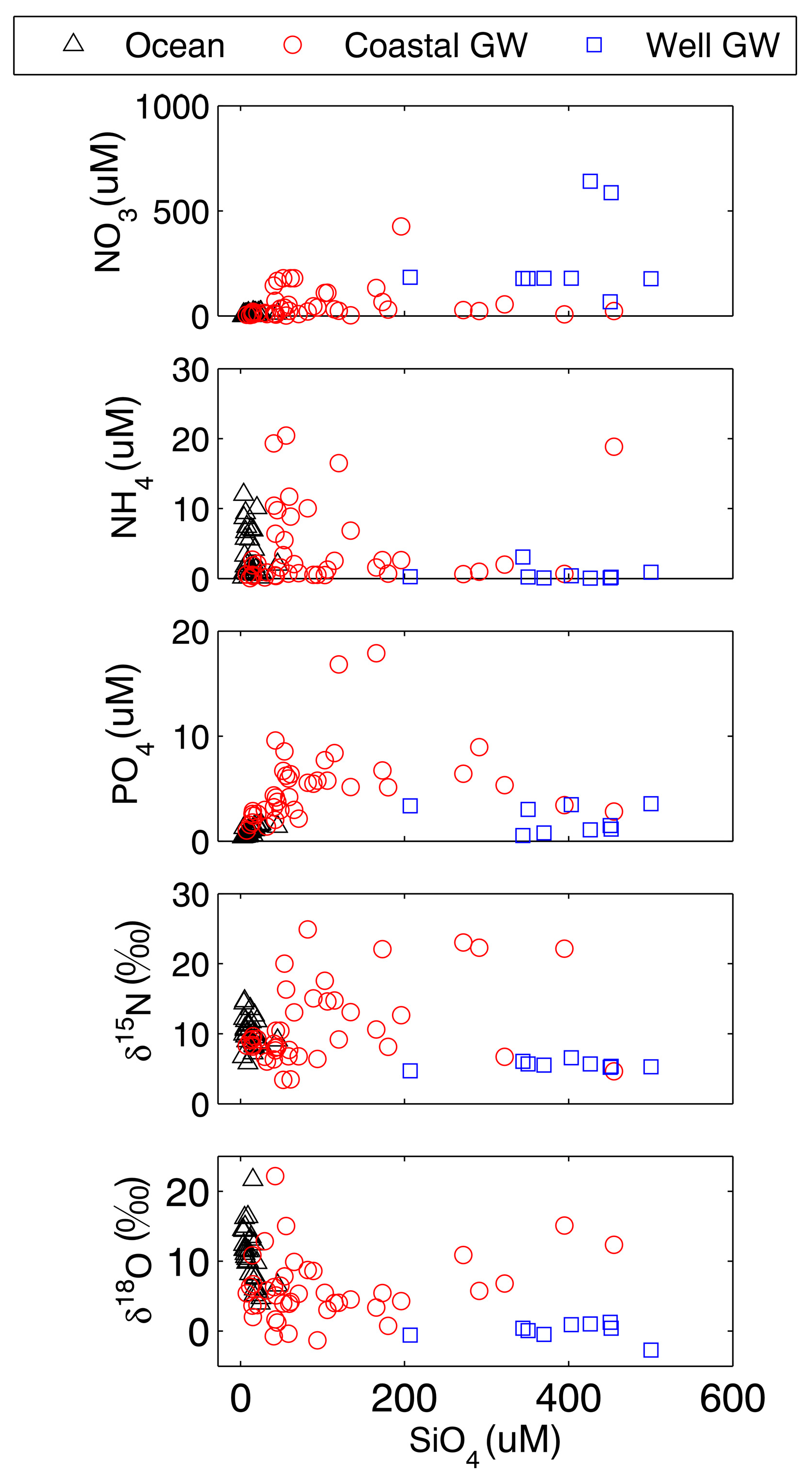
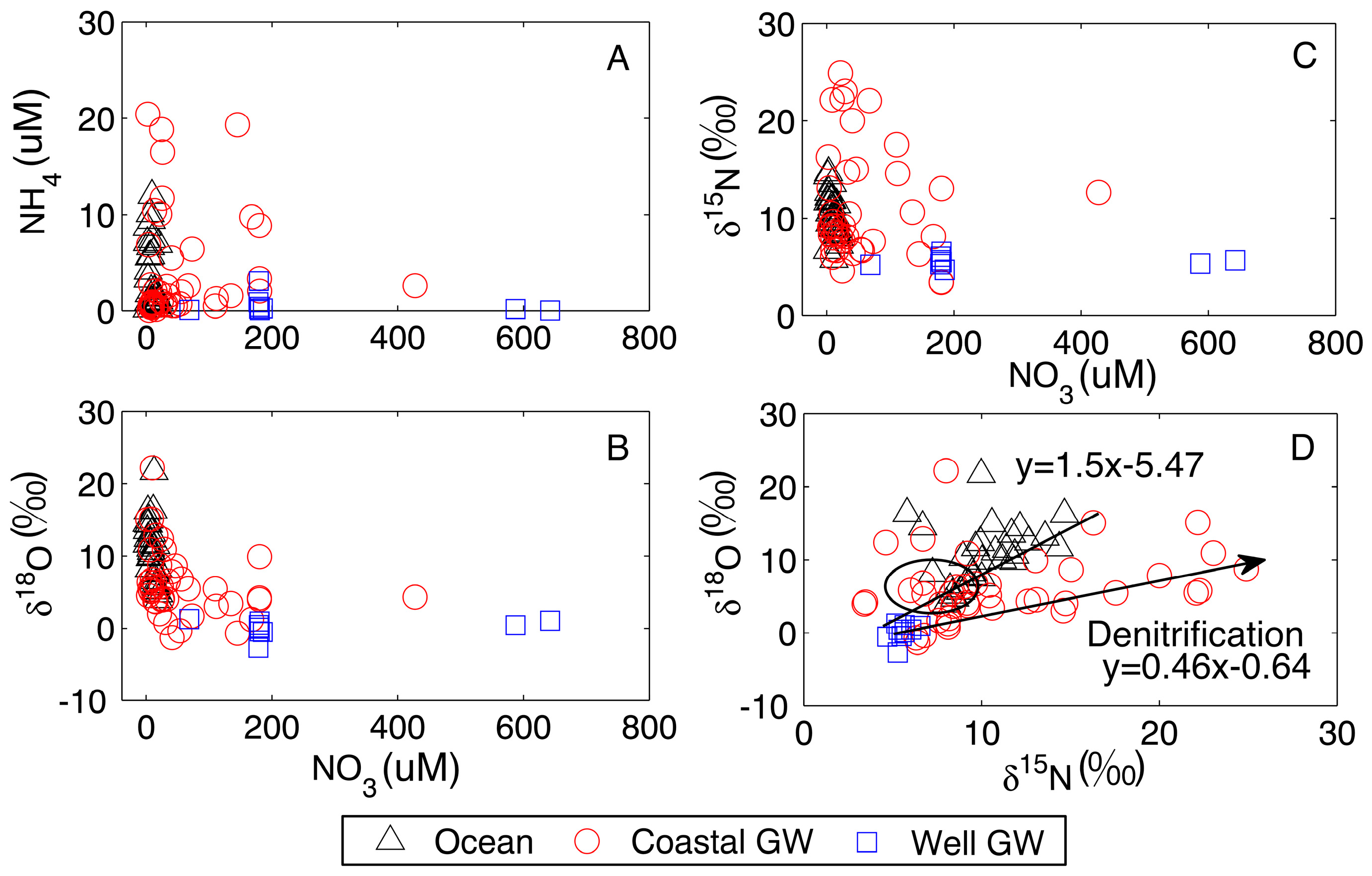
4. Discussion
4.1. Processes Observed in the Coastal Aquifer
4.2. Principal Component Analysis
4.3. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kwon, E.; Kim, G.; Primeau, F.; Moore, W.; Cho, H.-M.; DeVries, T.; Sarmiento, J.; Charette, M.; Cho, Y.-K. Global Estimate of Submarine Groundwater Discharge Based on an Observationally Constrained Radium Isotope Model. Geophys. Res. Lett. 2014, 62–68. [Google Scholar] [CrossRef]
- Lecher, A.L. Groundwater Discharge in the Arctic: A Review of Studies and Implications for Biogeochemistry. Hydrology 2017, 4, 41. [Google Scholar] [CrossRef]
- Taniguchi, M.; Burnett, W.C.; Cable, J.E.; Turner, J.V. Investigation of submarine groundwater discharge. Hydrol. Process. 2002, 16, 2115–2129. [Google Scholar] [CrossRef]
- Moore, W.S.; Sarmiento, J.L.; Key, R.M. Submarine groundwater discharge revealed by 228Ra distribution in the upper Atlantic Ocean. Nat. Geosci. 2008, 1, 309–311. [Google Scholar] [CrossRef]
- Hosono, T.; Ono, M.; Burnett, W.C.; Tokunaga, T.; Taniguchi, M.; Akimichi, T. Spatial distribution of submarine groundwater discharge and associated nutrients within a local coastal area. Environ. Sci. Technol. 2012, 46, 5319–5326. [Google Scholar] [CrossRef] [PubMed]
- Lecher, A.L.; Mackey, K.R.M.; Kudela, R.; Ryan, J.; Fisher, A.; Murray, J.; Paytan, A. Nutrient Loading through Submarine Groundwater Discharge and Phytoplankton Growth in Monterey Bay, CA. Environ. Sci. Technol. 2015, 49, 6665–6673. [Google Scholar] [CrossRef] [PubMed]
- Valiela, I.; Foreman, K.; LaMontagne, M.; Hersh, D.; Costa, J.; Peckol, P.; DeMeo-Andreson, B.; D’Avanzo, C.; Babione, M.; Sham, C.-H.; et al. Couplings of Watersheds and Coastal Waters: Sources and Consequences of Nutrient Enrichment in Waquoit Bay, Massachusetts. Estuaries 1992, 15, 443. [Google Scholar] [CrossRef]
- Carruthers, T.J.B.; Van Tussenbroek, B.I.; Dennison, W.C. Influence of submarine springs and wastewater on nutrient dynamics of Caribbean seagrass meadows. Estuar. Coast. Shelf Sci. 2005, 64, 191–199. [Google Scholar] [CrossRef]
- Lecher, A.L.; Mackey, K.R.M.; Paytan, A. River and submarine groundwater discharge effects on diatom phytoplankton abundance in the Gulf of Alaska. Hydrology 2017, 4, 61. [Google Scholar] [CrossRef]
- Knee, K.; Paytan, A. 4.08 Submarine Groundwater Discharge: A Source of Nutrients, Metals, and Pollutants to the Coastal Ocean. Treatise Estuar. Coast. Sci. 2011, 4, 205–234. [Google Scholar] [CrossRef]
- Moore, W.S. The subterranean estuary: A reaction zone of ground water and sea water. Mar. Chem. 1999, 65, 111–125. [Google Scholar] [CrossRef]
- Lecher, A.L.; Chien, C.; Paytan, A. Submarine groundwater discharge as a source of nutrients to the North Pacific and Arctic coastal ocean. Mar. Chem. 2016, 186, 167–177. [Google Scholar] [CrossRef]
- Slomp, C.P.; Van Cappellen, P. Nutrient inputs to the coastal ocean through submarine groundwater discharge: Controls and potential impact. J. Hydrol. 2004, 295, 64–86. [Google Scholar] [CrossRef]
- Erler, D.V.; Santos, I.R.; Zhang, Y.; Tait, D.R.; Befus, K.M.; Hidden, A.; Li, L.; Eyre, B.D. Nitrogen transformations within a tropical subterranean estuary. Mar. Chem. 2014, 164, 38–47. [Google Scholar] [CrossRef]
- Kroeger, K.D.; Charette, M.A. Nitrogen biogeochemistry of submarine groundwater discharge. Limnol. Oceanogr. 2008, 53, 1025–1039. [Google Scholar] [CrossRef]
- Charette, M.A.; Sholkovitz, E.R. Trace element cycling in a subterranean estuary: Part 2. Geochemistry of the pore water. Geochim. Cosmochim. Acta 2006, 70, 811–826. [Google Scholar] [CrossRef]
- Charette, M.A.; Sholkovitz, E.R.; Hansel, C.M. Trace element cycling in a subterranean estuary: Part 1. Geochemistry of the permeable sediments. Geochim. Cosmochim. Acta 2005, 69, 2095–2109. [Google Scholar] [CrossRef]
- Kim, J.; Cho, H.-M.; Kim, G. 228Ra flux in the northwestern Pacific marginal seas: Implications for disproportionally large submarine groundwater discharge. Ocean Sci. J. 2015, 50, 195–202. [Google Scholar] [CrossRef]
- Rodellas, V.; Garcia-Orellana, J.; Masqué, P.; Feldman, M.; Weinstein, Y. Submarine groundwater discharge as a major source of nutrients to the Mediterranean Sea. Proc. Natl. Acad. Sci. USA 2015, 112, 3926–3930. [Google Scholar] [CrossRef] [PubMed]
- Le Gland, G.; Mémery, L.; Aumont, O.; Resplandy, L. Improving the inverse modeling of a trace isotope: How precisely can radium-228 fluxes toward the ocean and submarine groundwater discharge be estimated? Biogeosciences 2017, 14, 3171–3189. [Google Scholar] [CrossRef]
- Sánchez-Martos, F.; Jiménez-Espinosa, R.; Pulido-Bosch, A. Mapping groundwater quality variables using PCA and geostatistics: A case study of Bajo Andarax, southeastern Spain. Hydrol. Sci. J. 2001, 46, 227–242. [Google Scholar] [CrossRef]
- Guggenmos, M.R.; Daughney, C.J.; Jackson, B.M.; Morgenstern, U. Regional-scale identification of groundwater-surface water interaction using hydrochemistry and multivariate statistical methods, Wairarapa Valley, New Zealand. Hydrol. Earth Syst. Sci. 2011, 15, 3383–3398. [Google Scholar] [CrossRef]
- Chen, K.; Jiao, J.J.; Huang, J.; Huang, R. Multivariate statistical evaluation of trace elements in groundwater in a coastal area in Shenzhen, China. Environ. Pollut. 2007, 147, 771–780. [Google Scholar] [CrossRef] [PubMed]
- De Andrade, E.M.; Palácio, H.A.Q.; Souza, I.H.; de Oliveira Leão, R.A.; Guerreiro, M.J. Land use effects in groundwater composition of an alluvial aquifer (Trussu River, Brazil) by multivariate techniques. Environ. Res. 2008, 106, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, V.; Lefebvre, R.; Therrien, R.; Savard, M.M. Multivariate statistical analysis of geochemical data as indicative of the hydrogeochemical evolution of groundwater in a sedimentary rock aquifer system. J. Hydrol. 2008, 353, 294–313. [Google Scholar] [CrossRef]
- Lee, J.M.; Woo, N.C.; Lee, C.J.; Yoo, K. Characterising bedrock aquifer systems in Korea using paired water-level monitoring data. Water 2017, 9, 420. [Google Scholar] [CrossRef]
- Abou Zakhem, B.; Al-Charideh, A.; Kattaa, B. Using principal component analysis in the investigation of groundwater hydrochemistry of Upper Jezireh Basin, Syria. Hydrol. Sci. J. 2017, 62, 2266–2279. [Google Scholar] [CrossRef]
- Paliy, O.; Shankar, V. Application of multivariate statistical techniques in microbial ecology. Mol. Ecol. 2016, 25, 1032–1057. [Google Scholar] [CrossRef] [PubMed]
- Lecher, A.L.; Fisher, A.T.; Paytan, A. Submarine groundwater discharge in Northern Monterey Bay, California: Evaluation by mixing and mass balance models. Mar. Chem. 2016, 179, 44–55. [Google Scholar] [CrossRef]
- Hanson, R. Geohydrologic Framework of Recharge and Seawater Intrusion in the Pajaro Valley, Santa Cruz and Monterey Counties, California; US Geological Survey: Washington, DC, USA, 2003.
- Hansen, H.P.; Koroleff, F. Determination of nutrients. In Methods of Seawater Analysis: Third, Completely Revised and Extended Edition; Minister of Supply and Services Canada: Ottawa, ON, Canada, 2007; pp. 159–228. ISBN 9783527613984. [Google Scholar]
- McIlvin, M.R.; Altabet, M.A. Chemical conversion of nitrate and nitrite to nitrous oxide for nitrogen and oxygen isotopic analysis in freshwater and seawater. Anal. Chem. 2005, 77, 5589–5595. [Google Scholar] [CrossRef] [PubMed]
- Ryabenko, E. Effect of chloride on the chemical conversion of nitrate to nitrous oxide for δ 15 N analysis. Limnol. Oceanogr. Methods 2009, 7, 545–552. [Google Scholar] [CrossRef]
- Street, J.H.; Knee, K.L.; Grossman, E.E.; Paytan, A. Submarine groundwater discharge and nutrient addition to the coastal zone and coral reefs of leeward Hawai′i. Mar. Chem. 2008, 109, 355–376. [Google Scholar] [CrossRef]
- Wankel, S.D.; Kendall, C.; Pennington, J.T.; Chavez, F.P.; Paytan, A. Nitrification in the euphotic zone as evidenced by nitrate dual isotopic composition: Observations from Monterey Bay, California. Glob. Biogeochem. Cycles 2007, 21. [Google Scholar] [CrossRef]
- Sigman, D.M.; Granger, J.; DiFiore, P.J.; Lehmann, M.M.; Ho, R.; Cane, G.; van Geen, A. Coupled nitrogen and oxygen isotope measurements of nitrate along the eastern North Pacific margin. Glob. Biogeochem. Cycles 2005, 19. [Google Scholar] [CrossRef]
- Chen, D.J.Z.; MacQuarrie, K.T.B. Correlation of delta N-15 and delta O-18 in NO3- during denitrification in groundwater. J. Environ. Eng. Sci. 2005, 4, 221–226. [Google Scholar] [CrossRef]
- Knee, K.; Street, J.H.; Grossman, E.G.; Paytan, A. Nutrient inputs to the coastal ocean from submarine groundwater discharge in a groundwater-dominated system: Relation to land use (Kona coast, Hawaii, U.S.A.). Limnol. Oceanogr. 2010, 55, 1105–1122. [Google Scholar] [CrossRef]


| Process | Nutrients Affected | Indicators of Presence |
|---|---|---|
| Mixing | All | Distinct end-members in the ocean and inland groundwater and good correlation with a conservative tracer like salinity or silicate |
| Denitrification | Nitrate | Concentrations of nitrate decrease as δ15NNO3 and δ18ONO3 increase |
| Nitrification | Nitrate | Concentrations of nitrate increase as δ15NNO3 and δ18ONO3 decrease |
| Decay of Organic Material | Phosphate and Ammonium | Increasing concentrations of phosphate and ammonium |
| Salinization | Phosphate | Increased salinity and phosphate concentrations as cations cause desorption from aquifer substrate |
| Precipitation of Oxides | Phosphate | Decreasing phosphate concentrations under oxic conditions |
| Nutrient Uptake | All | Decreasing concentrations of nutrients paired with increasing isotopic signatures |
| Reduction of Iron Oxides | Iron and Phosphate | Increasing phosphate concentrations and dissolved iron and manganese under anoxic conditions |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lecher, A.L.; Murray, J.; Paytan, A. Quantifying Processes Governing Nutrient Concentrations in a Coastal Aquifer via Principal Component Analysis. Hydrology 2018, 5, 15. https://doi.org/10.3390/hydrology5010015
Lecher AL, Murray J, Paytan A. Quantifying Processes Governing Nutrient Concentrations in a Coastal Aquifer via Principal Component Analysis. Hydrology. 2018; 5(1):15. https://doi.org/10.3390/hydrology5010015
Chicago/Turabian StyleLecher, Alanna L., Joseph Murray, and Adina Paytan. 2018. "Quantifying Processes Governing Nutrient Concentrations in a Coastal Aquifer via Principal Component Analysis" Hydrology 5, no. 1: 15. https://doi.org/10.3390/hydrology5010015
APA StyleLecher, A. L., Murray, J., & Paytan, A. (2018). Quantifying Processes Governing Nutrient Concentrations in a Coastal Aquifer via Principal Component Analysis. Hydrology, 5(1), 15. https://doi.org/10.3390/hydrology5010015





