Abstract
Four distinct types of multiple emulsions were synthesized using xanthan gum and pectin through two distinct manufacturing processes. The assessment encompassed the examination of morphology, stability, and rheological properties for the resulting water-in-oil-in-water (W/O/W) double emulsions. Formulations were meticulously crafted with emulsifiers that were compatible with varying compositions. Remarkably stable multiple emulsions were achieved with a 0.5 wt% xanthan concentration, demonstrating resilience for nearly two months across diverse storage temperatures. In contrast, multiple emulsions formulated with a higher pectin concentration (2.75 wt%) exhibited instability within a mere three days. All multiple emulsions displayed shear-thinning behavior, characterized by a decline in apparent viscosity with escalating shear rates. Comparatively, multiple emulsions incorporating xanthan gum showcased elevated viscosity at low shear rates in contrast to those formulated with pectin. These results underscore the pivotal role of the stepwise process over the direct approach and emphasize the direct correlation between biopolymer concentration and emulsion stability. This present investigation demonstrated the potential use of pectin and xanthan gum as stabilizers of multiple emulsions with potential application in the pharmaceutical industry for the formulation of topical dosage forms.
Keywords:
multiple emulsions; xanthan gum; pectin; stability; rheology; morphology; interaction effects 1. Introduction
Emulsions, consisting of two typically immiscible liquids, such as oil and water, manifest as a composite with a continuous phase enveloping a dispersed phase. In this intricate structure, non-soluble small droplets disperse within the continuous phase [1].
Emulsions are categorized based on the arrangement of the oil and aqueous phases, with prevalent examples being oil-in-water (O/W) emulsions like mayonnaise, milk, cream, soups, and sauces, or water-in-oil (W/O) emulsions found in margarine, butter, and spreads [2]. A contemporary development involves the creation of multiple emulsions [3,4]. These complex multiphase structures, holding nonsoluble small droplets within a continuous phase, have garnered substantial interest in the food, cosmetic, and pharmaceutical industries [5,6]. Double emulsions, despite being thermodynamically unstable, are integral to this innovation, often relying on synthetic surfactants for stabilization [7,8,9]. The two principal categories of multiple emulsions are water-in-oil-in-water (W1/O/W2) and oil-in-water-in-oil (O1/W/O2) double emulsions [10,11]. While the first report of a W/O/W multiple emulsion dates back to 1925 by Seifriz [12,13], it is only in the last fifty years that their diverse applications have been explored. Currently, these systems have varied applications in several domains, especially the pharmaceutical field, for the administration of various therapeutic molecules. Herbert [14] emerged as a pioneer in 1965, utilizing multiple emulsions for the controlled release of drugs in pharmaceuticals [15,16]. Double emulsions can be used for the encapsulation of drugs like vitamins [17], vaccines [18], or even certain bioactive compounds extracted from plants [19]. The conception and development of hydrophilic drug distribution systems is an ongoing process in pharmaceutical research. Multiple emulsions, especially W/O/W emulsions, are admirable candidates for the controlled and sustained release of such drugs [20]. The release of a water-soluble drug from the multiple W/O/W emulsions occurs by a mechanism of swelling followed by a breakdown of the oil globules, in which the lipophilic surfactant is a decisive factor, and the increasing of lipophilic surfactant concentration causes amelioration instability of the multiple emulsions [20]. The main advantage of such systems is that they increase the solubility and bioavailability of therapeutic drugs as well as the ability to favor the topical transport of hydrophilic drugs. Multiple emulsions are used as an alternative to liposomes as a delivery system. In addition, W/O/W emulsions are generally employed for intravenous routes for the administration of vaccines or other ingredients like anticancer drugs [21,22]. Double emulsions drug delivery systems were used to enhance the intestinal permeability of drugs classified under the Biopharmaceutics Classification System (BCS) class III, which exhibit high solubility and low permeability like zanamivir, a hydrophilic antiviral drug. The permeability of such drugs was enhanced by dissolving the drug in the inner aqueous phase and increasing its lipophilicity through the oil phase [23]. In cancer therapy, W/O/W emulsions were used for the encapsulation of Rhodamine B in the treatment of brain cancer glioblastoma multiforme (GBM). The encapsulation of the drug in the internal droplets of emulsions leads to low cytotoxicity and allows the creation of a patient-friendly drug delivery system [24]. Wang et al. demonstrated the higher encapsulation efficiency of resveratrol in w/o/w emulsions Compared with liposomes and lipid nanoparticles. A possible reason is that W/O/W emulsions have two oil-water interface films, which provide better protection to the resveratrol core [10]. The encapsulation of 5-FU (a strong chemotherapeutic drug) into double emulsions allowed a decrease in skin cancer cell multiplication and greater cell viability compared to the pure 5-FU formulation [25]. Furthermore, the double emulsion method enabled the simultaneous loading of hydrophilic and lipophilic drugs, as reported by Lee et al., where hydrophilic doxorubicin and hydrophobic erlotinib were encapsulated in a W/O/W system. They demonstrated that this system allowed a fast cellular uptake and successful tumor suppression by the retention of more doxorubicin molecules inside tumor cells, which was also observed [26].
The stabilization of multiple emulsions commonly involves the synergistic use of hydrophilic and hydrophobic surfactants [27,28,29]. Surfactants with a low Hydrophilic Lipophilic Balance (HLB) are employed to stabilize the internal emulsion [30], while those with a high HLB are employed for the outer emulsion [6]. In the pharmaceutical and cosmetic industries, chemical synthesis products are prevalent emulsifiers and stabilizers [31,32]. However, these synthetic surfactants are often used conventionally and may pose potential irritations. Addressing the growing preference for natural and biodegradable products, there is a growing interest in replacing these synthetic surfactants with more natural molecules, aligning with the contemporary trend outlined by K. Ezzroug (2019) [33]. Polysaccharides, primarily hydrophilic polymers, are commonly utilized in emulsion formulation due to their stabilizing properties [34,35]. They enhance the viscosity of the aqueous continuous phase [36] or induce gelation [37]. Despite their typically low surface activity, certain polysaccharides exhibit surface activity and can stabilize emulsions by adsorbing at the liquid-liquid interface. Well-known examples include Arabic gum, acetylated beet pectin, and modified starch, subject to recent studies [38,39]. In contrast to traditional surfactants, polymers introduce strong steric repulsion between droplets, facilitating emulsion stabilization at low emulsifier concentrations over extended periods [40,41]. Moreover, their higher molecular mass generally renders them less toxic than surfactants [15]. Stabilization by biomolecules involves interaction with hydrophobic surfactants, forming a complex film that fortifies the elastic interface, thereby impeding breakage [42]. This film enhances mechanical resistance and diminishes coalescence through steric hindrance [43]. Four conventional processes are used for the manufacture of multiple emulsions [15]. The most common and most often used is the two-step process [44]. In addition, studies show that it is possible to formulate multiple emulsions with good long-term stability in a single emulsification step (direct process) using a single emulsifier of the particle or amphiphilic copolymer type [45,46,47,48,49,50]. Therefore, Emulsions can be kinetically stable over a long period, which determines their lifespan. This stability can be achieved either by surfactant molecules, colloidal particles, or using natural polymers (biopolymers). It is this last approach that we have chosen to develop in this work.
The chosen polysaccharide is xanthan gum, a biological macromolecule derived from Xanthomonas campestris. Widely applied in food products, this anionic polysaccharide boasts distinctive physical attributes such as viscosity and pseudo-plasticity, along with chemical properties like water solubility and pH stability [51,52]. In emulsions, it serves as a crucial thickening and stabilizing agent [53,54]. Despite its non-adsorptive nature at interfaces, xanthan gum effectively impedes creaming by modifying the rheological characteristics of the continuous phase [36,55]. This polysaccharide demonstrates commendable stability against variations in temperature, pH, salts, and enzymatic degradation. Typically incorporated into the aqueous phase, it enhances the stability of oil-in-water (O/W) emulsions by forming a robust three-dimensional gel-like network within the aqueous medium [56].
Similar to xanthan gum, pectin is a high-molecular-weight heteropolysaccharide extensively employed as a thickening, emulsifying, and stabilizing agent in the food and pharmaceutical industries [57]. Pectin creates a pseudo-viscous solution with water, solidifying upon boiling [55,58].
This study was undertaken with the principal objective of delving into the intricate dynamics governing the stability of multiple emulsions. Specifically, the investigation centered on double emulsions of the water-in-oil-in-water (W/O/W) variety, where the stabilization was orchestrated through the incorporation of xanthan gum and pectin. Notably, the study sought to discern the nuanced influence of polysaccharide concentration on the overall stability of these emulsions. The rigorous stability assessment of the meticulously prepared double emulsions spanned a comprehensive timeframe of three months, exposing them to diverse storage temperatures to capture a holistic understanding of their endurance under varied conditions. Complementing this temporal evaluation, an exhaustive rheological study was systematically executed for each formulated composition, offering insights into the emulsions’ dynamic properties. In conjunction with these analytical approaches, microscopic analyses were intricately performed on the distinct formulations. This multifaceted methodology aimed to provide a thorough and enriched exploration of the stability mechanisms governing W/O/W double emulsions, offering valuable insights into their rheological behavior and microscopic morphology.
This study introduces a significant innovation in the preparation and characterization of multiple emulsions, specifically water-in-oil-in-water (W/O/W) emulsions, using two different biopolymers, xanthan gum, and pectin, along with two distinct manufacturing processes. The study comprehensively evaluates the morphology, stability, and rheological properties of these four types of W/O/W emulsions, formulated with compatible emulsifiers. One of the key findings of this research is the successful creation of highly stable multiple emulsions employing a 0.5% weight concentration of xanthan gum, which maintained its stability for approximately two months at varying storage temperatures. Conversely, emulsions prepared with a higher concentration of pectin (2.75% weight) exhibited signs of instability within just three days. Additionally, all multiple emulsions in the study exhibited shear-thinning behavior, wherein their apparent viscosity decreased with increasing shear rate. Notably, emulsions containing xanthan gum displayed the highest viscosity at low shear rates compared to those with pectin. Moreover, this study underscores that the stepwise manufacturing process is more suitable than the direct process, and it highlights the direct influence of biopolymer concentration on emulsion stability. These findings hold substantial promise for enhancing emulsion formulation and stability, with potential implications across various industries, including the food and pharmaceutical sectors. The study’s insights into optimizing multiple emulsion composition and production processes provide valuable contributions to this field of research.
2. Materials and Methods
2.1. Materials
The organic phase, composed of vaseline oil, was thoughtfully sourced from a local supermarket in Algeria, imparting a regional touch to the composition. The lipophilic surfactant employed, Sorbitan Monolaurate (Span 60) with an HLB of 4.3, and the hydrophilic surfactant (Tween 80 with an HLB of 15), were supplied by the esteemed Sigma-Aldrich, ensuring high-quality standards. The food-grade xanthan gum, sourced from Rhodia (Algeria), adds a crucial functional dimension to the formulation. Regarding the thickening agent, pectin, selected for its unique properties (a notably high degree of esterification, between 70 and 75%), was also provided by Rhodia, introducing a specific aspect to emulsion stabilization. Quality ingredients, such as sodium hydroxide (NaOH) and magnesium sulfate heptahydrate (MgSO4·7H2O), were meticulously chosen from Prolabo in France, ensuring the reliability and consistency of experimental results. This meticulous approach in selecting components reflects a commitment to scientific excellence throughout this study.
2.2. Analytical Methods
The meticulous examination of emulsion properties was carried out using cutting-edge instrumentation. The LF 191 conductometer from WTW, Germany, was employed with precision to gauge emulsion conductivity. pH measurements were executed using the state-of-the-art Mark Metrhom AGCH-9100 Hrisan pH meter, ensuring accuracy in the assessment. To unravel the intricate microstructure of the scrutinized emulsions and validate their multiplicity, a crucial role was played by the optical microscope (Olympus® BX50, Olympus Optical, Tokyo, Japan), seamlessly connected to a camera and offering an impressive ×1000 enlargement for a detailed analysis. Additionally, the viscosity analysis was conducted using the Bohlin Visco 88 from Malvern Instruments, which is equipped with a rotating mobile component for nuanced measurements. Simultaneously, the estimation of particle size distribution was accomplished through the sophisticated utilization of ImageJ software (version 1.52a), demonstrating a holistic and multifaceted approach to comprehensively characterize the diverse properties of the emulsions.
3. Results
3.1. W/O/W Multiple Emulsions Formulation Using the Stepwise Process
This method represents the most prevalent, straightforward, and easily graspable approach in the realm of emulsion preparation. The preparation of multiple emulsions took place through a two-step procedure, which is a method consistently advocated by various authors in the field. In the initial stage, a primary water-in-oil (W/O) emulsion was meticulously formulated. Following this, in the subsequent step, the primary emulsion underwent dispersion into an aqueous solution. The development of the foundational W/O emulsion entailed the careful introduction of an aqueous phase (Phase B) into the oil phase (Phase A), culminating in the creation of a sophisticated multiple water-in-oil-in-water (W/O/W) emulsion.
3.1.1. Primary Emulsion (W/O) Formulation
The procedure commenced by elevating the temperature of the aqueous phase, which included the salt and the oily phase, hosting the lipophilic surfactant, to 75 °C to ensure complete solubility. Post-cooling, a meticulous blending process ensued, with the gradual infusion of the aqueous phase into the oily phase achieved through a homogenizer operating at 11,000 rpm. This controlled integration method facilitated the creation of various formulations, each meticulously detailed in Table 1 for comprehensive reference.

Table 1.
Composition of the formulated primary emulsions.
Characteristics of Primary Emulsions Formulated
- Physico-chemical characterization
The immediate visual examination of all formulated compositions yielded valuable insights into their macroscopic characteristics right after preparation. A noteworthy observation was the immediate phase separation in the case of the first formulation (F1). However, for formulations F2, F3, and F4, a nuanced interplay emerged, highlighting the substantial influence of the lipophilic surfactant and the oily phase proportions on the resulting emulsions. It was particularly intriguing to observe that a higher concentration of the lipophilic agent (3–5%) coupled with a lower proportion of the oily phase (24%) led to a discernible enhancement in emulsion consistency. This phenomenon can be rationalized by the intricate interplay of surfactant molecules with the water-in-oil (W/O) interface, covering the entire interfacial film. The heightened concentration of the lipophilic agent contributed to increased rigidity in the interfacial film, thereby elevating emulsion consistency. Conversely, as the oil amount increased while maintaining the same concentration of the lipophilic agent, a reduction in consistency ensued, resulting in a less viscous system. In this context, the lower concentration of surfactant molecules on the interfacial film, owing to the expanded interface, led to diminished stiffness in the interfacial film and, consequently, reduced emulsion consistency. Furthermore, the observation that the pH value remained relatively constant across formulations underscores its resilience to variations in the concentration of emulsion constituents.
The identification of emulsion type (W/O or O/W) can be carried out through three distinct methods: washability with water, the dye method, and conductimetry, with the latter being the most commonly employed and considered as the standard. Conductimetry involves measuring the conductivity of the continuous phase, where an aqueous phase containing an ionic surfactant typically exhibits a conductance greater than or equal to 1 mS, while that of a lipophilic phase approaches zero [44]. The determination of the emulsion type is further confirmed when the emulsion floats on water, signifying its classification as W/O. This is substantiated by consistently low conductivity measurements across all emulsions, indicating the oily phase as the continuous phase.
Moreover, the stability of the emulsion was scrutinized in both real-time and accelerated time. Real-time measurements involved monitoring the dispersion’s endurance under standard storage conditions to observe its natural aging. Conversely, the accelerated time study subjected the solution to stress conditions (temperature variations, centrifugal acceleration, etc.) to expedite the observation of demixing [44]. The centrifugation results for formulations F2, F3, and F4 demonstrate the absence of instability phenomena at various speeds, except for formulation F2, which exhibits slight separation at high rotation speeds (6000 rpm).
- Viscosity study
Variation of viscosity as a function of shear rate has been studied η = f (). The expression of the shear rate is the same as that of a coaxial cylinder rheometer, with an infinite radius of the outer cylinder (Re) [59].
The formula for shear rate () as the following relation gives a function of speed of rotation:
with : rotation mobile speed.
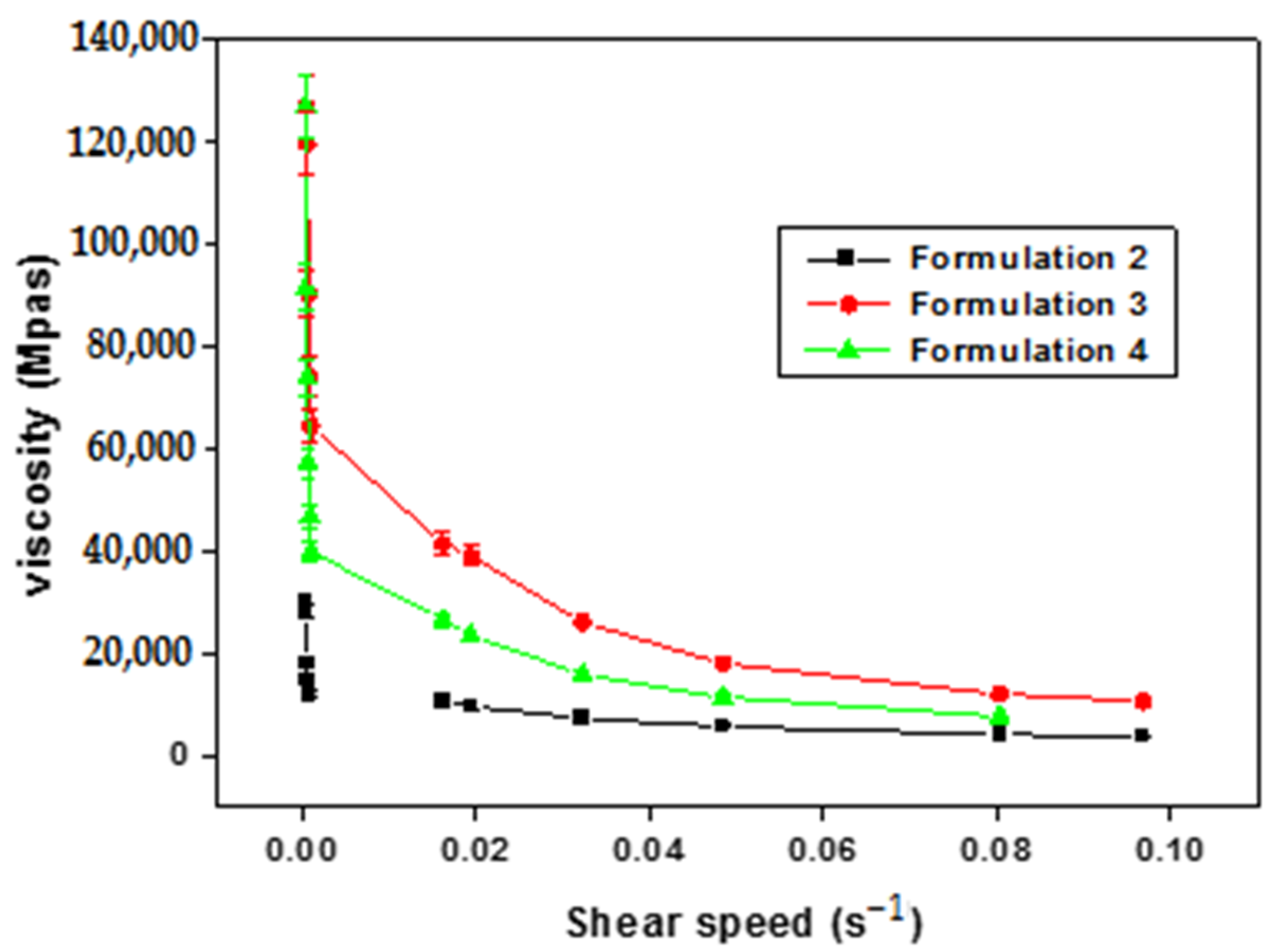
The viscosimetric study presented in Figure 1 reveals an inverse relationship between viscosity and shear rate. Consequently, it can be deduced that the primary emulsions formulated (F2–F4) exhibit non-Newtonian behavior characteristic of pseudo-plastic and shear-thinning fluids.
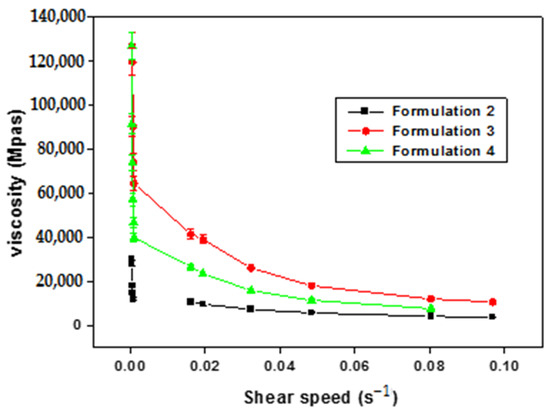
Figure 1.
Evolution of apparent viscosity as a function of shear rate of primary emulsions.
Shear thinning behavior is a desired characteristic in the pharmaceutical field, both to facilitate the administration and packaging of these systems, because this character allows the characteristics of the product to be preserved after use or processing (packaging in the form of a tube, box, or bottle). This pseudo-plastic character is also interesting, on the one hand, for the conservation and reuse of multidose forms, as this behavior will keep the formulation stable after use, where it always recognizes its initial form after solicitation. On the other hand, shear thinning systems are easily spreadable, especially in topical formulations when applied to the skin or in contact with mucous membranes, like ocular mucosa [60,61].
- Microscopical (morphological) study
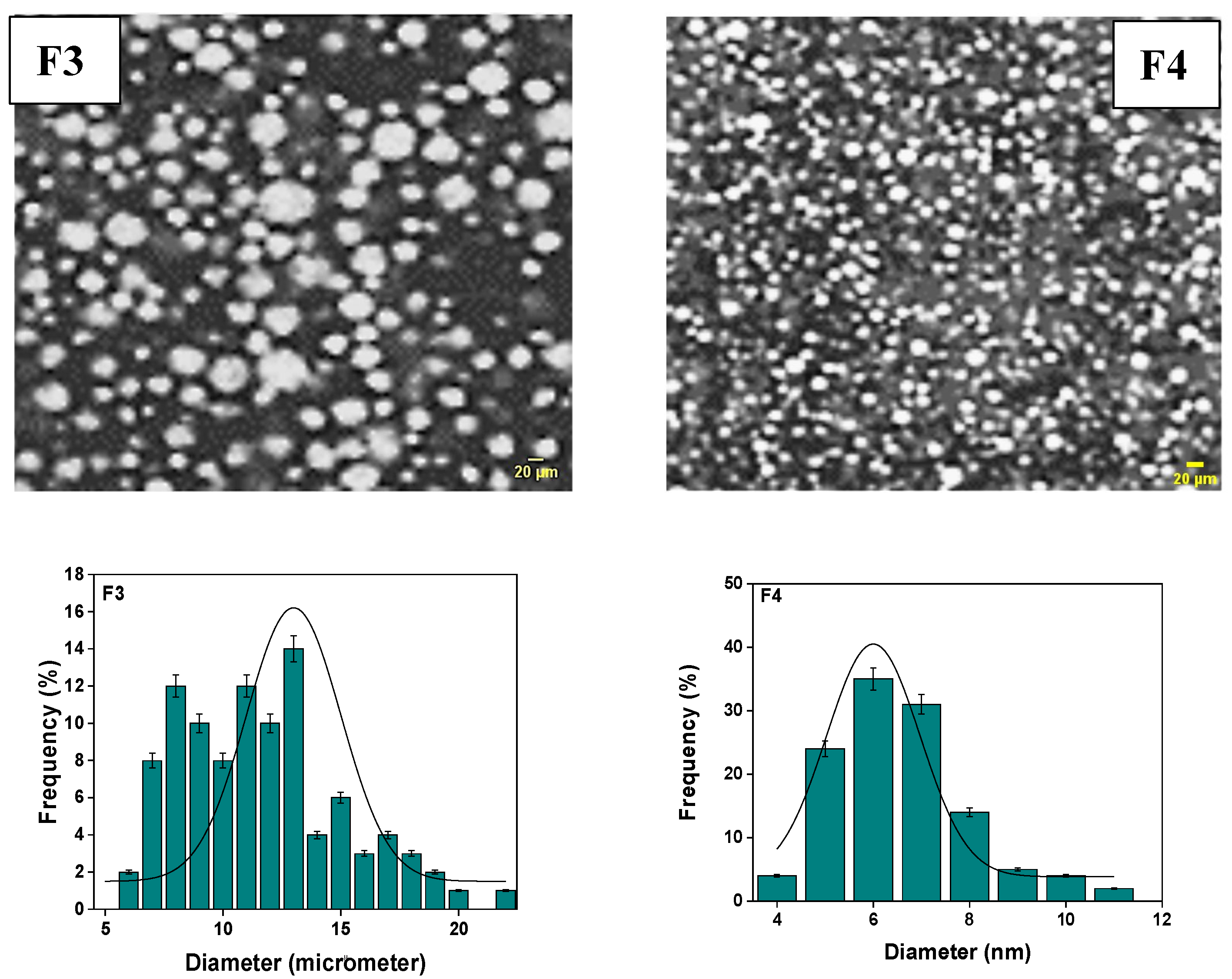
The obtained optical microscopy images for different formulations (F2, F3, and F4) were produced using optical microscopy with an enlargement (×1000) endowed with a photomicrograph (Figure 2).
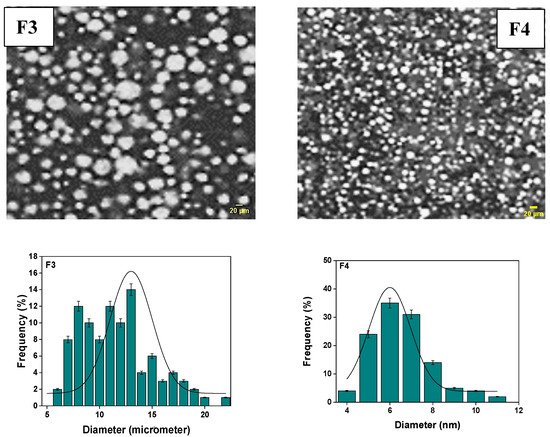
Figure 2.
The size distribution of particles in the primary emulsion (F3 and F4) was determined through analysis of optical microscopy images.
Upon a meticulous microscopic examination, the primary emulsions reveal a distinctive dispersion pattern of aqueous globules within the oily phase, a pattern influenced by the proportion of the lipophilic agent. Notably, a higher quantity of the lipophilic agent results in a finer dispersion, contributing to the formation of a more rigid and stable emulsion. Conversely, an increase in the lipophilic agent, coupled with a rise in vaseline oil content, produces a more rigid film with fewer dispersed globules. Nevertheless, this adjustment results in a stable yet less rigid emulsion. The observable particle size becomes more pronounced, highlighting the varied diameters of globules formed due to the emulsion’s unique consistency. Furthermore, the average diameter of the emulsions formulated as F3 and F4 was precisely measured using ImageJ software, yielding results of approximately (13.0 ± 0.7) µm for F3 and (07.00 ± 1.75) µm for F4. In the realm of emulsions, a common challenge surfaces in the form of droplet aggregation, often leading to a substantial increase in emulsion viscosity. This rapid creaming phenomenon is attributed to alterations in droplet size and droplet size distribution, which, in turn, affect the emulsion’s rheology by modifying interactions and dispersed-phase volume fraction. However, it’s important to note that for dilute emulsions, droplet size exerts no significant effect on viscosity in the absence of long-distance attractive or repulsive interactions between the droplets [62,63,64,65].
3.1.2. Formulation and Characterization of Multiple Emulsions W/O/W Stabilized by Xanthan Gum
The precise formulation of primary emulsions (W/O) followed by a comprehensive battery of characterization tests has pinpointed formulation (F4) as the optimum primary emulsion for integration into the composition of the multiple emulsion (W/O/W). This choice is anchored in its outstanding stability and fine dispersion, setting it apart as a superior candidate among the array of emulsions evaluated. The creation of a multiple W/O/W emulsion involves encapsulating the chosen primary emulsion (W/O) within an external aqueous phase. To facilitate this, xanthan gum is pre-dissolved in the aqueous phase through gentle stirring, a step initiated 24 h prior to actual use. Subsequently, the hydrophilic surfactant (Tween 80) is introduced into the aqueous phase. Throughout this process, a delicate stirring approach (approximately 500 rpm) is employed to prevent the rupture of the primary emulsion. Simultaneously, the primary emulsion is gradually incorporated over a 15-min duration at room temperature.
Comprehensive details regarding the various formulated systems are meticulously outlined in Table 2.

Table 2.
Composition of the formulated multiple emulsions based on xanthan gum.
Characterizations of Multiple Emulsions Formulated
- Physico-chemical characterizations
The macroscopic analysis involves visually examining the appearance of the formulated emulsions (F1–F10) with the naked eye. It becomes apparent that the consistency of the emulsions is intricately linked to the proportion of the biopolymer; a lower amount of xanthan gum results in a less viscous emulsion, while a higher amount increases viscosity. pH measurements reveal varying acidic values, reflecting the acidity of the xanthan gum integrated into the emulsions. It is noteworthy that emulsions with acidic pH have been neutralized (pH ≈ 7) with NaOH (10%) for improved preservation. Furthermore, the pH value of the emulsion plays a pivotal role in its preservation and determines potential incompatibilities with other components, such as active ingredients and excipients. pH can alter the solubility of an excipient and change its electrical state. Generally, an excipient in its neutral form is less irritating than in its ionized form. Ensuring the optimal pH of the emulsion is crucial to maintaining the excipient in its neutral form. The pH is also a critical parameter that requires adjustment based on the route of administration. For example, certain dosage forms like emulsions for ophthalmic use necessitate a specific pH closely aligned with the physiological pH of the administration site. Similarly, conductivity measurements indicate that the external phase of all the formulated multiple emulsions is an aqueous phase (current-conducting phase) with high electrical conductivity values, reaching up to 2.2 mS/cm. This confirms the dispersion of the primary emulsion in water, resulting in a W/O/W type system.
This result is in agreement with the value cited in the literature for a W/O/W type emulsion, which is of the order of 2.8 mS/cm [66].
On the other hand, the rate encapsulation of the formulated systems was achieved by an assay of MgSO4·7H2O initially incorporated into the internal aqueous phase directly on the multiple emulsion using a chemical method (Compléxometric), and the yield was calculated by the relation:
Degree encapsulation of the various emulsions formulated varied between 72% and 92%, which means that the primary emulsion is actually encapsulated in the external phase.
- Stability at different storage temperatures
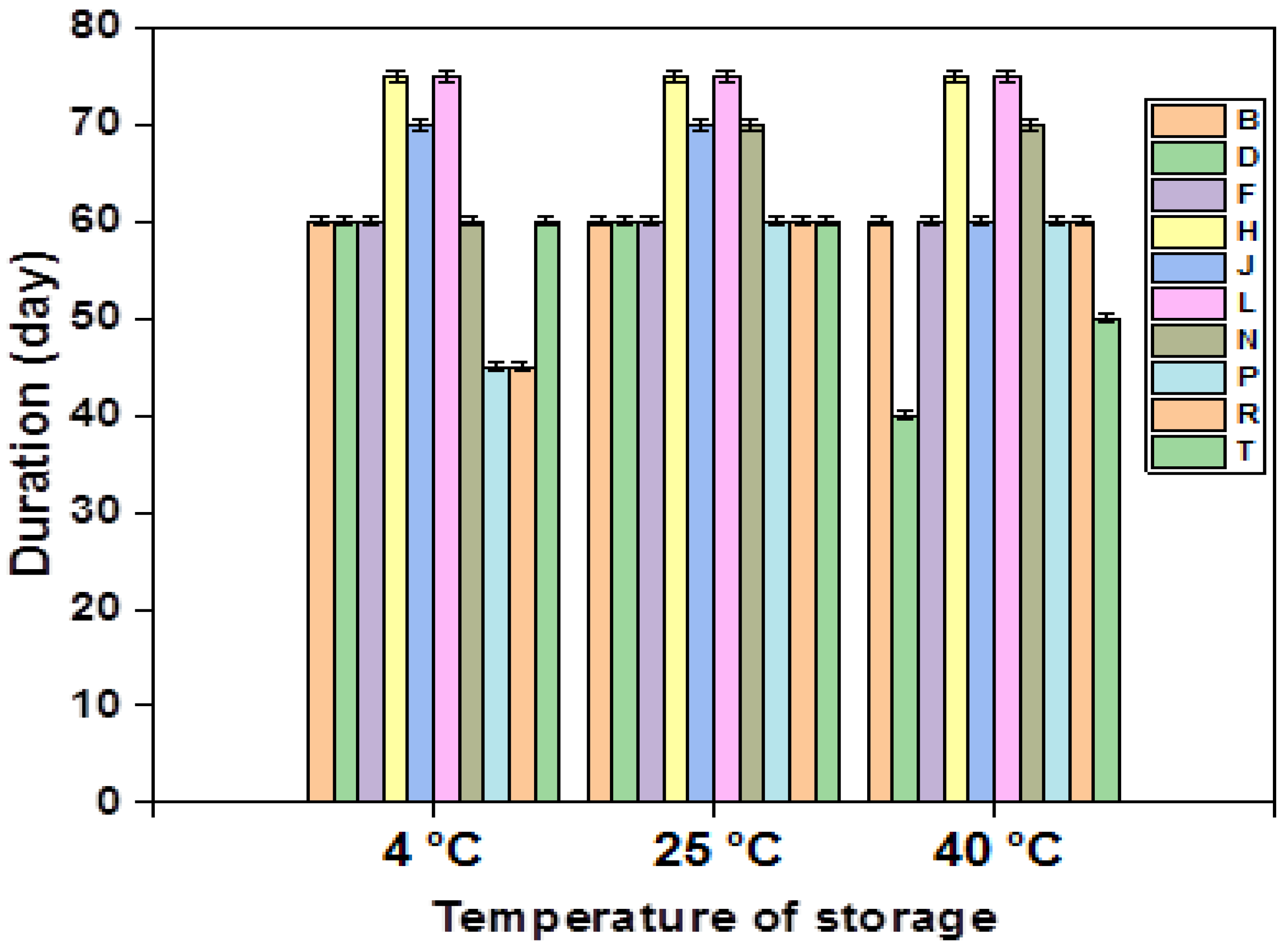
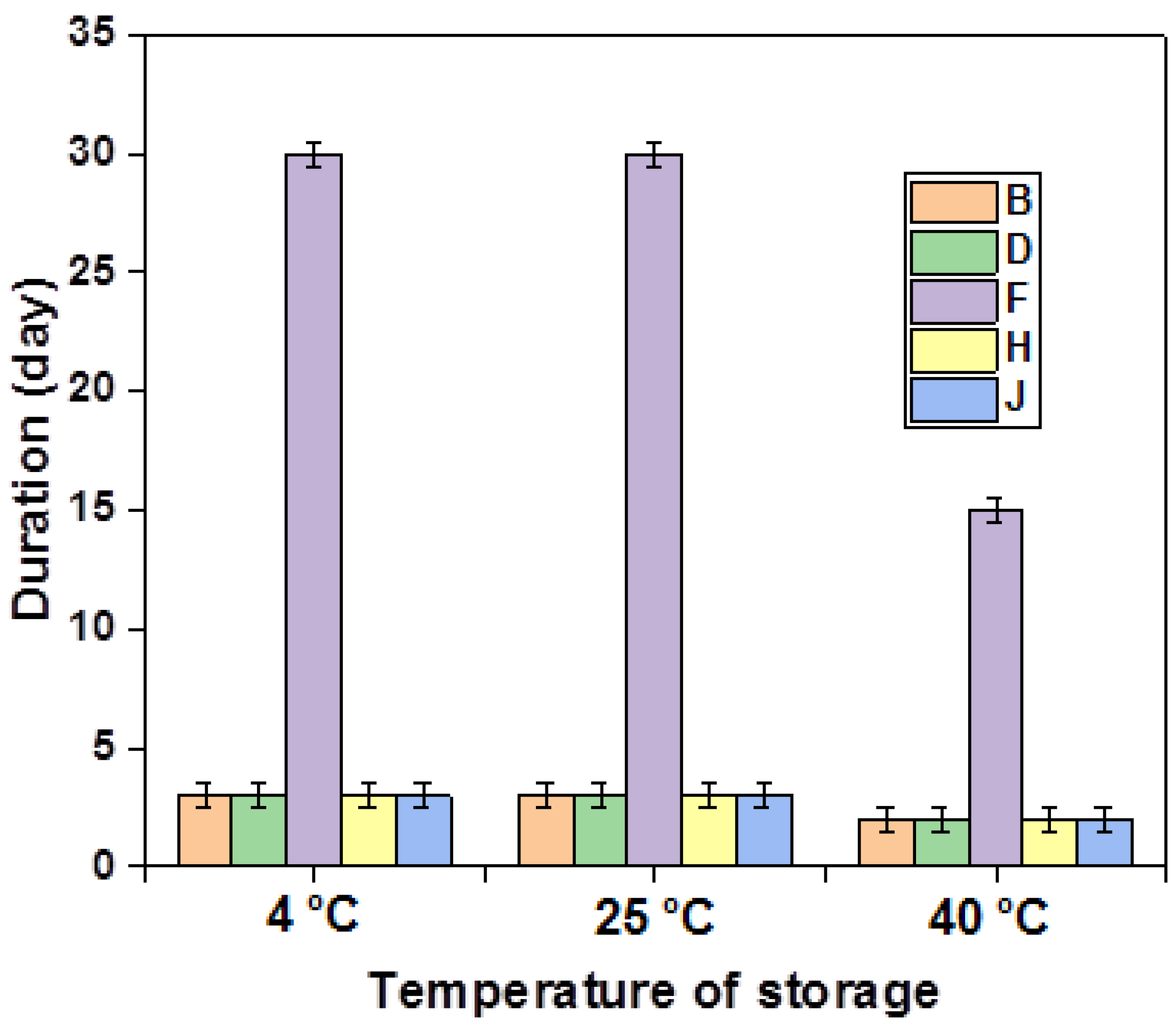
All multiple emulsions formulations (F1–F10) were subjected to stability tests at different temperatures (4 °C, 25 °C, and 40 °C); the results are presented in the figure (Figure 3).
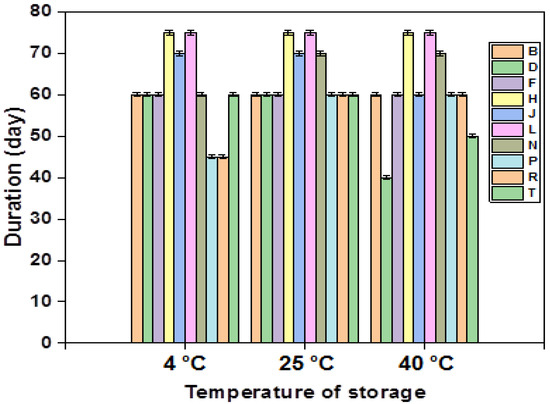
Figure 3.
Stability of the different emulsions stabilized with XG over time.
An in-depth analysis of the provided figure underscores the notable stability exhibited by formulations F4 and F6 over a three-month period at various storage temperatures. Notably, these two formulations share a commonality in containing the same concentration of primary emulsion (40%) and xanthan gum (1.5%). On the contrary, formulations F2, F8, and F9, characterized by a reduced concentration of the primary emulsion (60%), manifest instability phenomena after 2 months at 25 °C and 4 °C, and after 45 days at 40 °C. This highlights the pivotal role of an increased amount of the gelling agent in the external phase, enhancing the stability of multiple emulsions by elevating the viscosity of the continuous phase and facilitating the formation of aggregates through macromolecule adsorption on neighboring particle surfaces. Subsequently, these aggregates are linked together by hydrocarbon bridges.
It is crucial to emphasize that the influence of the concentration of the lipophilic surfactant on stability surpasses that of the hydrophilic surfactant. This is primarily attributed to the former’s integral role in sustaining the stability of the primary emulsion and contributing to the increased rigidity of the second interface.
- Viscosity study
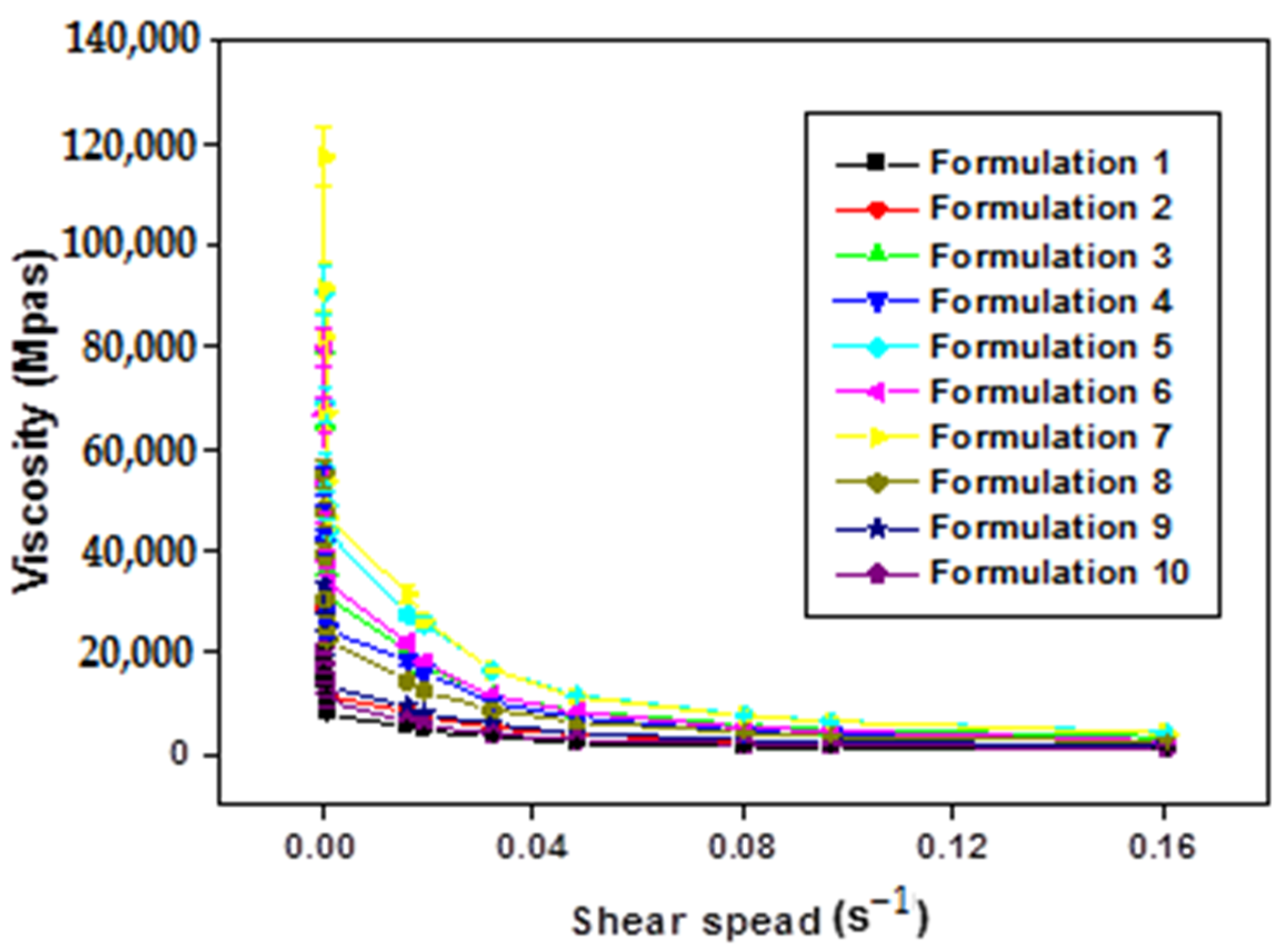
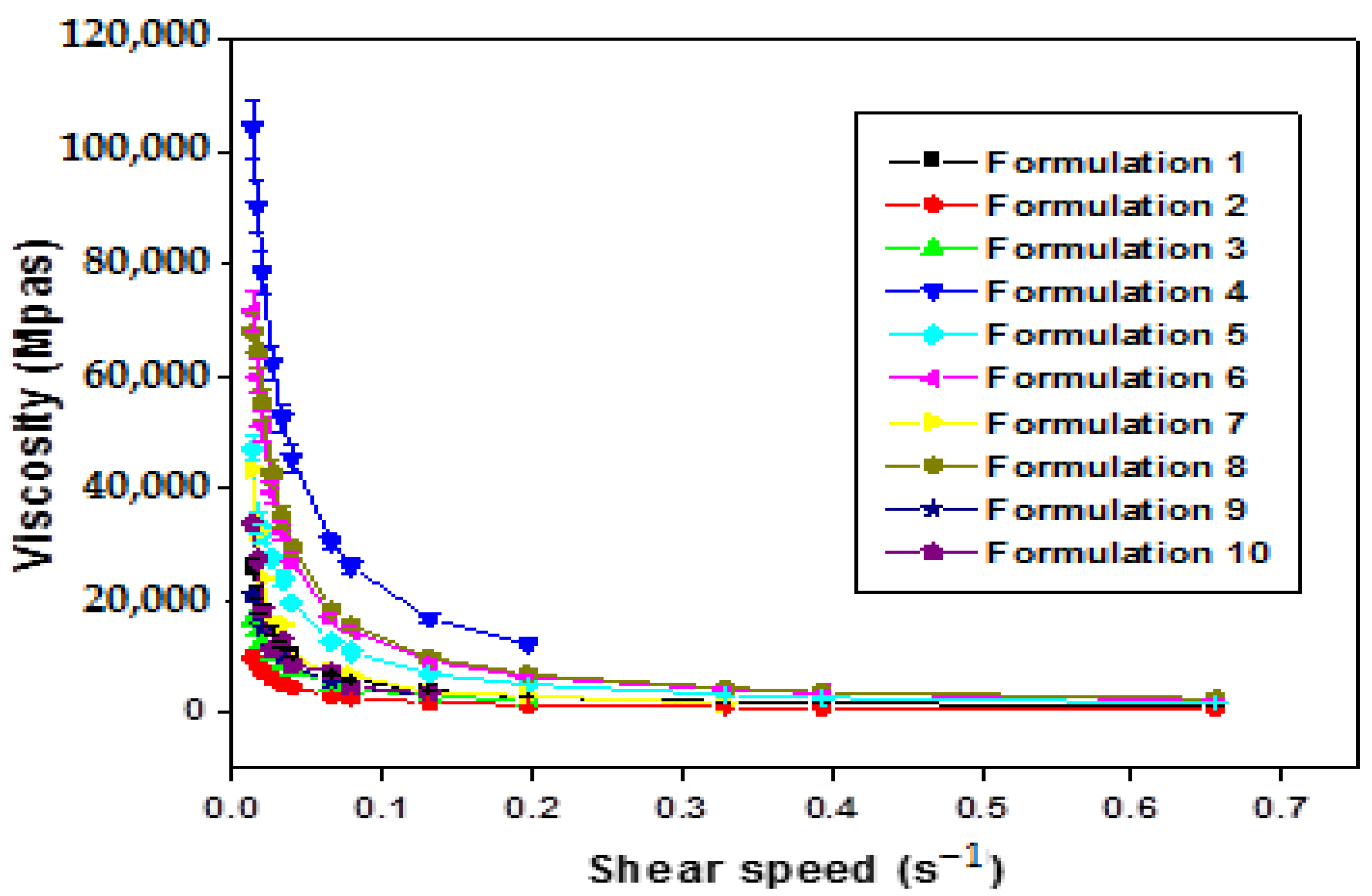
The increase in the shear rate of all formulations (F1–F10) leads to a gradual decrease in viscosity (Figure 4). This confirms that all the formulations obtained are of non-Newtonian pseudo-plastic fluid type with a shear-thinning character.
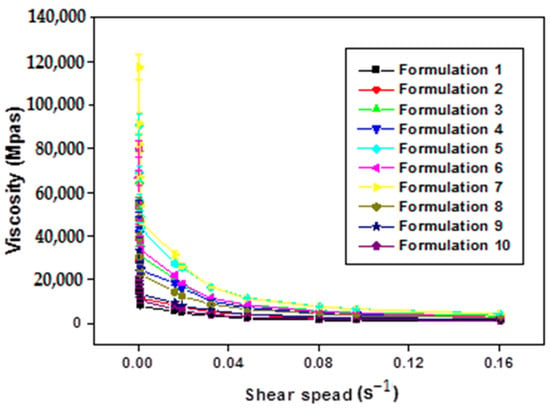
Figure 4.
Variation in apparent viscosity as a function of shear rate of xanthan gum based multiple emulsions.
The comprehensive analysis of Figure 4 illuminates a consistent trend where the apparent viscosity decreases with an escalating shear rate, indicative of the shear-thinning behavior exhibited by the formulated emulsions. This pseudo-plastic behavior, inherent in polymers with long chains, can be elucidated as follows: under rest or weak constraints, molecular chains are arranged randomly and entangled. With an increasing shear rate, these chains align and disperse in the direction of the force, resulting in a subsequent reduction in apparent viscosity.
Significantly, the rheological behavior of the obtained emulsions is notably responsive to the concentration of the biopolymer. Apparent viscosity values exhibit discernible variations in direct correlation with changes in polysaccharide concentration. Specifically, at low shear rates, an augmentation in xanthan gum (XG) concentration corresponds to higher apparent viscosity values. It is pivotal to underscore that the viscosity of double emulsions intricately ties into that of the outer phase, itself influenced by the concentration of XG. The observed robust stability in these intricate systems can be attributed to the heightened concentration of XG [8].
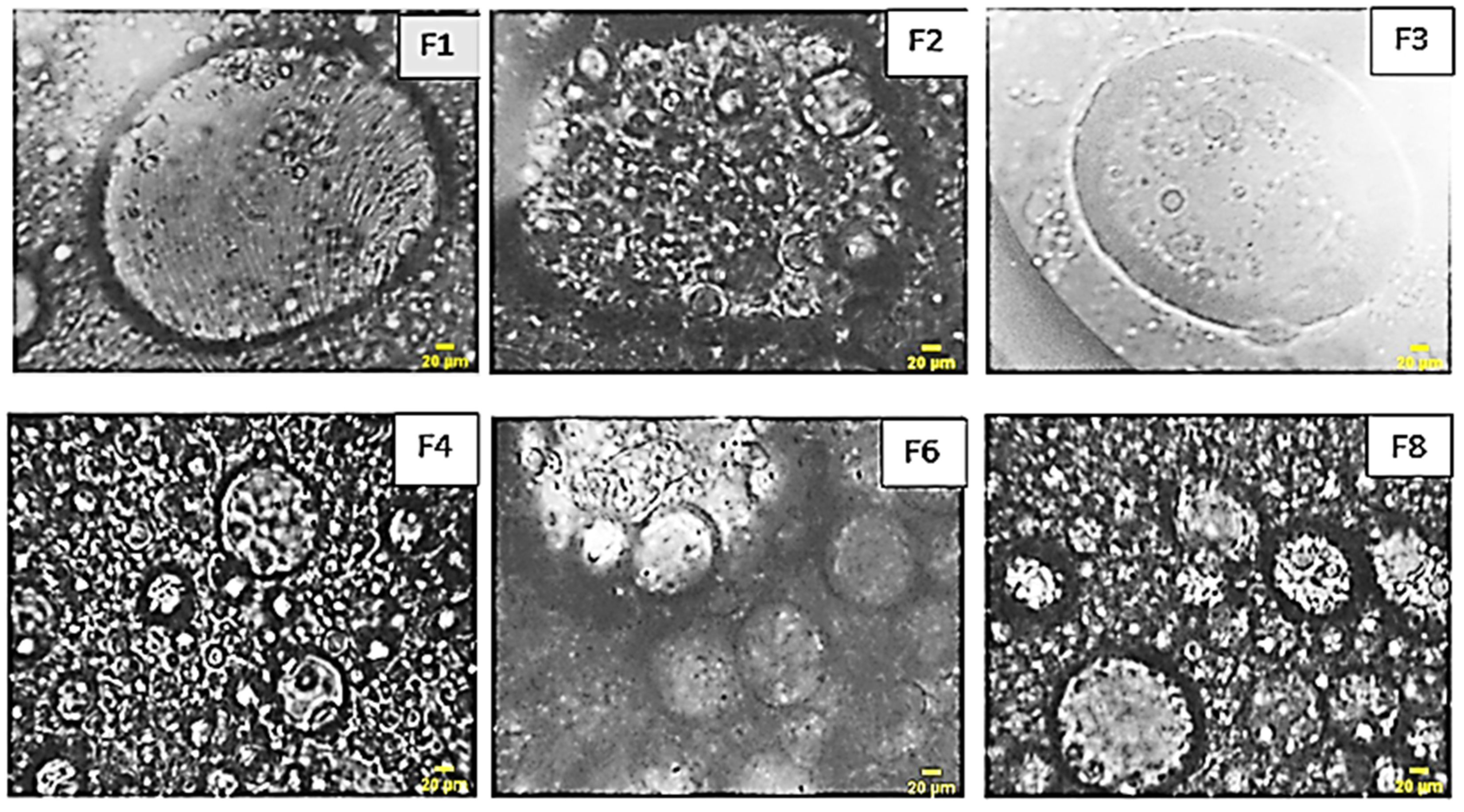
- Microscopic study
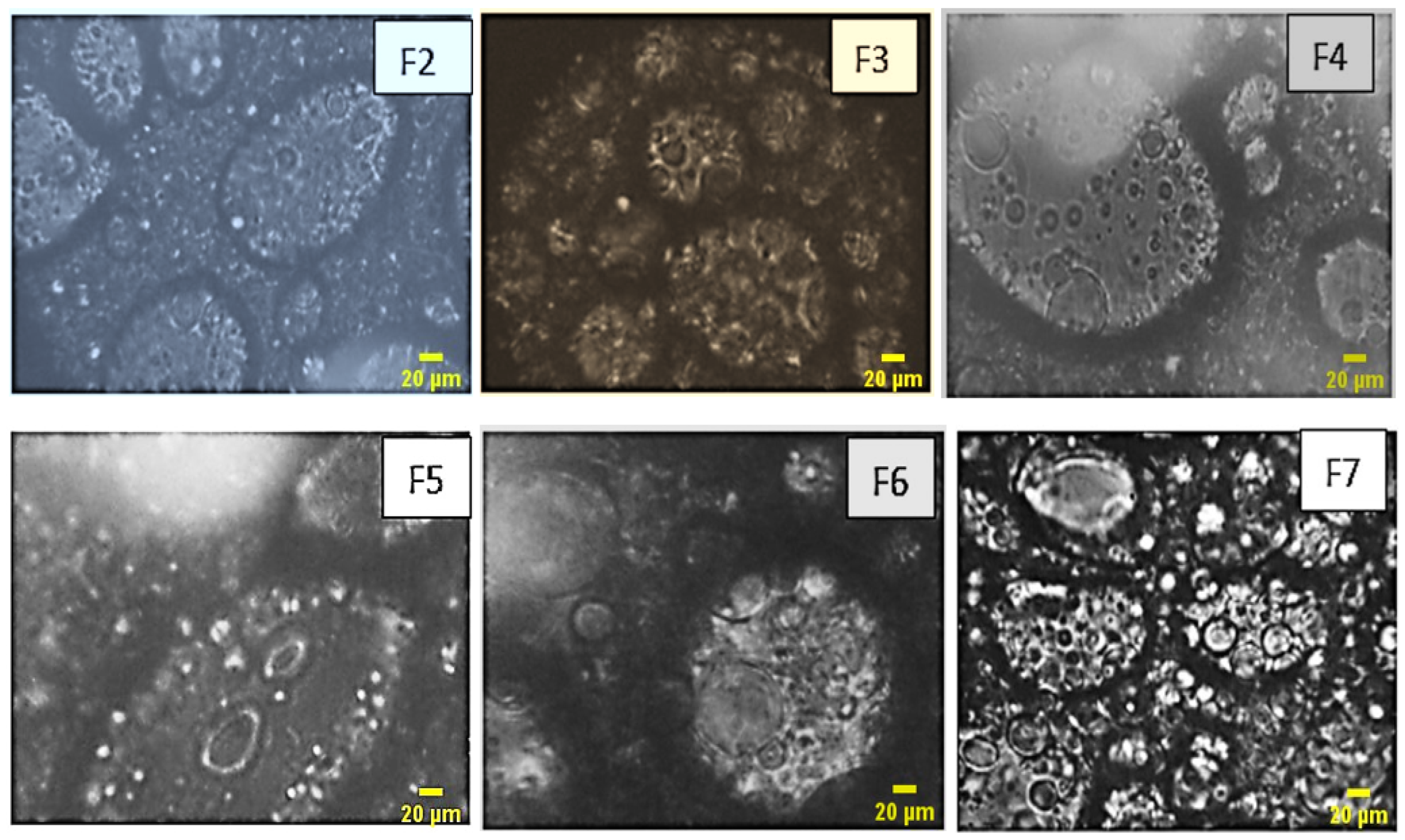
The microscopic analysis of the formulated emulsions vividly reveals the presence of multiple emulsions, showcasing a diverse array of internal aqueous globules. The distinct micrographs presented in Figure 5 offer a clear delineation of the interfacial films formed by both the primary and secondary emulsifiers, providing valuable insights into the intricate structure of these emulsion systems.
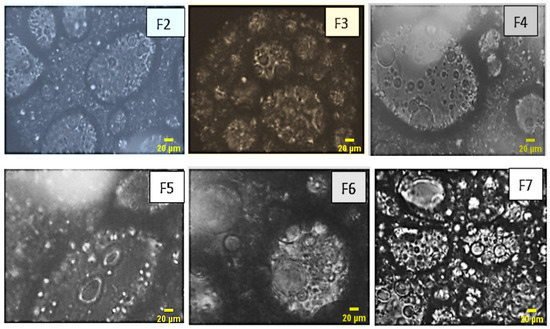
Figure 5.
Optical microscopy images of the formulas of multiples emulsion W/O/W were obtained by a stepwise process.
As a result, the emulsions formulated exhibit a distinct multiple emulsion structure. Notably, the increase in the quantity of the primary emulsion is directly associated with a heightened number of encapsulated globules, as observed in formulations F2 and F5. Additionally, it becomes apparent that an elevated concentration of the hydrophilic agent correlates with an increased abundance of globules, as evidenced in formulations F3 and F7. This observation enriches our understanding of the intricate relationship between formulation components and the resulting emulsion morphology.
3.1.3. Formulation and Characterization of Multiple W/O/W Emulsions Stabilized by Pectin
The same procedure was considered for the case of Xanthan gum (Section 3.1.2), where xanthan is replaced by pectin. Table 3 summarizes the composition of the various formulations prepared.

Table 3.
Composition of the formulated multiple emulsions stabilized with pectin.
Characterizations of Multiple Emulsions Formulated
- Physico-chemical characterizations
The initial assessment of emulsion quality involves a macroscopic examination, offering insights into visual characteristics like appearance, consistency, and homogeneity. The specific results for different formulations (F1–F10) are elaborated in Table 3. Significantly, the measured pH values point to an acidic pH, attributed to the acidic nature of the included pectin. This aligns with the high conductivity values observed across all formulations, confirming the aqueous continuous phase and classifying them as W/O/W type emulsions.
In addition, formulations F3, F6, F7, and F9 showcase notable stability, lasting for almost one (01) month at 4 °C and 25 °C and for 15 days at 40 °C. These formulations stand out for their substantial content of primary emulsion (>40%) and a high concentration of pectin (5%). On the contrary, other formulations exhibit instability phenomena, typically within a maximum of 3 days. Interestingly, the consistently superior stability observed during cold storage holds true for all tested emulsions, as depicted in Figure 6. This enriched understanding contributes to a more comprehensive evaluation of emulsion performance.
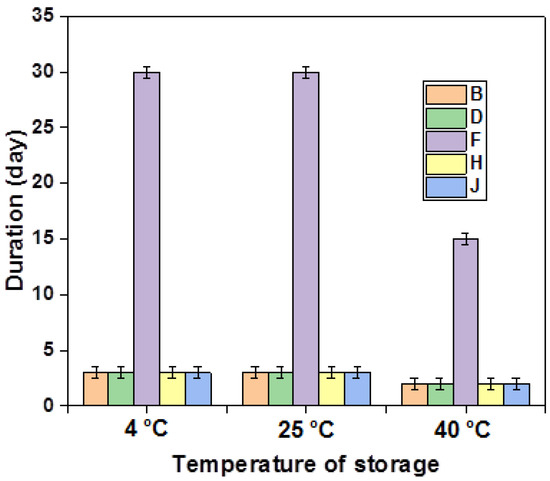
Figure 6.
Stability of the different emulsions stabilized with pectin over time.
The stability study also showed that the stability of the double emulsions formulated with xanthan is more stable than that of those formulated and stabilized with pectin. These findings may be explained as follows: On the one hand, pectin is subjected during thermal processing to degradation due to the acceleration of depolymerization reactions (acid hydrolysis, β-elimination, and demethoxylation), which is one of the main causes of texture deterioration. Furthermore, when the pectin is heated, the sensitivity of each single chain to depolymerization depends on its degree of methoxylation, which leads to the fact that some chains are degraded while others remain unaffected [67,68]. On the other hand, Due to its higher molecular weight and branched structure, the xanthan gum could reduce the interfacial tension and tend to form a cohesive interfacial film around the emulsion droplets by improving the viscosity of the external phase, thus improving the stability of the system [61]. In solution, the Xanthan molecule exists as a rod-like fivefold helical ordered conformation. This macromolecule is well-known to present a reversible conformational transition (helix–coil) under conditions of elevated temperatures and low ionic strength, where the helical structure may be converted to a coil conformation with a lower viscosity [69]. The midpoint of this transition™ has been quoted by Craig et al., 1997, as being in the region of 55–70 °C. Native GX solution progressively undergoes the order/disorder transition from a partially ordered random broken helix to a disordered random coil conformation at this temperature. Therefore, at 25 °C, the XG exists in the form of single chain helical conformation, while at the temperature of 45 °C, the gx partly retained its ordered double helix conformation but was not completely destructured. This leads to a drop in viscosity and not to complete destructuration. Then, at the temperature of 45 °C, the XG solution becomes less viscous, leading to slightly reduced stability [70,71].
In fact, keeping fresh is a condition found on the labels of emulsions, which allows them to be stored longer. Decreased instability is observed at high temperatures, explained by Stok’s law: in addition, the rise in temperature decreases the viscosity, which leads to an increase in the speed of sedimentation or creaming. Likewise, the yield encapsulation for stable formulations exceeds 60%, which is significant that about 60% of primary emulsion is encapsulated in the external phase.
Furthermore, the presence of xanthan gum and pectin increases the resistance of emulsions to flow by increasing their viscosity. At elevated storage temperatures, pectin undergoes a depolymerization phenomenon, and xanthan undergoes a sol-gel transition [70,71]. However, at cold temperatures (4 °C) and moderate ambient temperatures (25 °C), the viscosity of the emulsions increases. This implies that a firm structure is formed to overcome the Brownian motion, and thus, a larger disruptive force is needed to make the formulation flow.
- Viscosity study
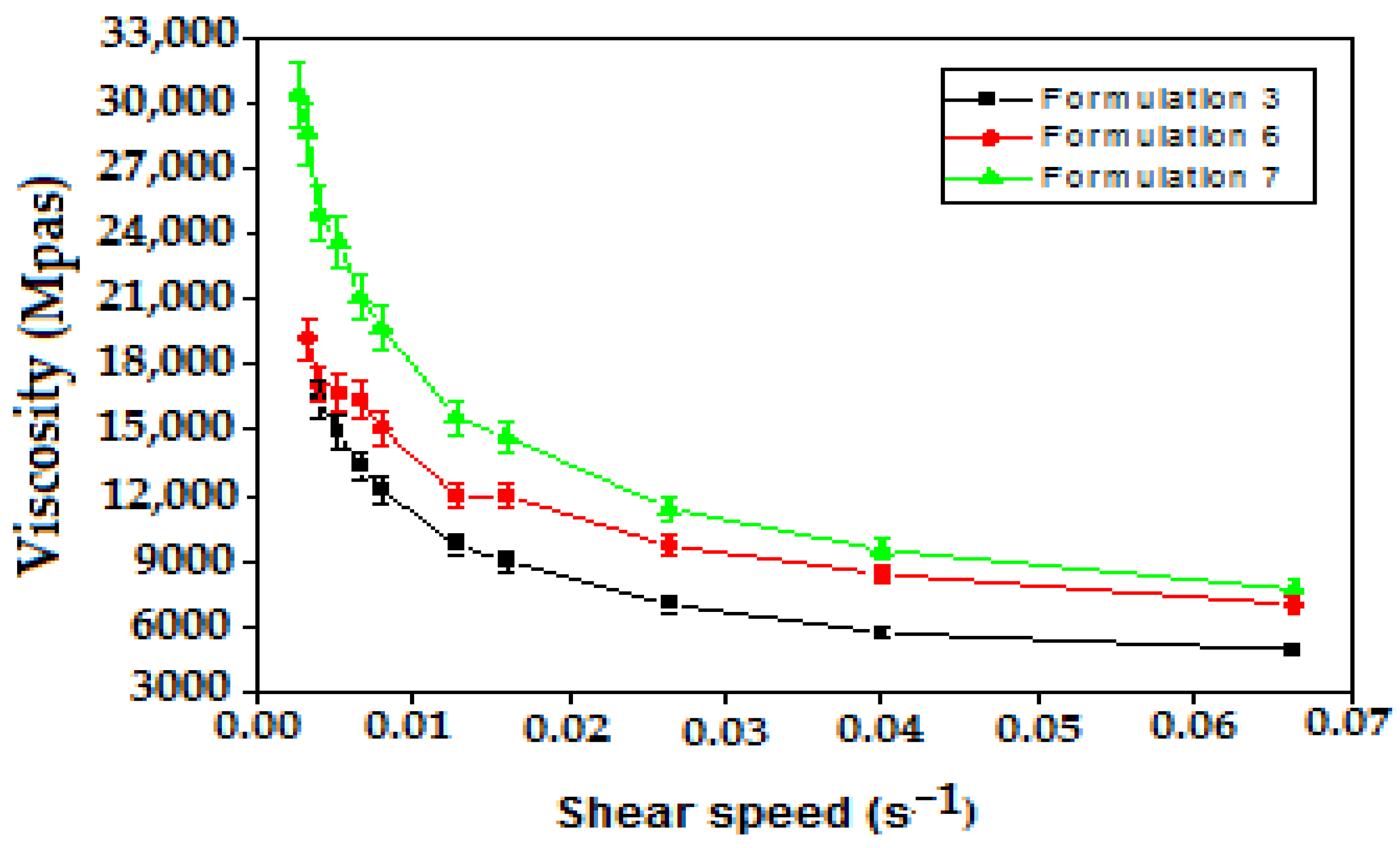
The variation in the apparent viscosity as a function of the shear rates of the formulated emulsions is shown (Figure 7), indicating that all emulsions tested are of the non-Newtonian type (shear thinning).
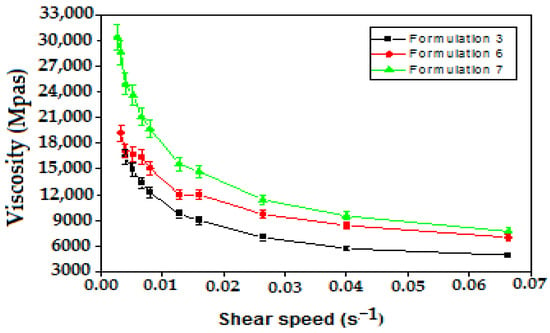
Figure 7.
Variation in apparent viscosity as a function of shear rate of Pectin based multiple emulsions.
- Microscopical Study
The observation of different optical microscopy images shows the presence of a multiple emulsion in most of the formulations obtained (Figure 8).
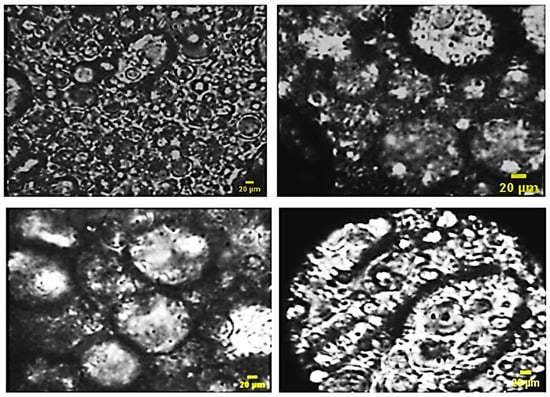
Figure 8.
Optical microscopy images of the formulas of multiples emulsion W/O/W obtained by stepwise process.
Figure 8 shows the presence of multiple globules as well as other simple ones; the reason is not clear but can be justified by the bursting of a few globules under the effect of intense agitation.
3.2. Formulation and Characterization of Multiple W/O/W Emulsions Stabilized by Xanthan Using the Direct Process
3.2.1. Formulation Procedure
This intricate process, conducted at 25 °C with a rotational speed of 15,000 rpm, involves the meticulous blending of equal parts of the aqueous phase—containing a small quantity of hydrophilic emulsifying agent—and the oily phase, enriched with a substantial amount of lipophilic emulsifying agent. In our specific application, the aqueous phase comprises distilled water, incorporating Tween 80 as the hydrophilic surfactant, while the oily phase consists of vaseline oil, with Span 60 serving as the lipophilic surfactant. The diverse formulations resulting from this process are detailed in Table 4.

Table 4.
Experimental matrix of different formulas obtained by direct process stabilized with XG.
3.2.2. Characterization of Multiple W/O/W Emulsions
The comprehensive assessment of the formulations’ macroscopic appearance reveals a generally homogeneous aspect, except for emulsions F4, F6, and F8, which exhibit heightened consistency. The pH values across the emulsions (F1–F10) indicate acidity attributable to the presence of xanthan gum. Meanwhile, elevated conductivity values affirm the aqueous nature of the continuous phase, confirming the W/O/W classification.
Examining the stability over 30 days at various storage temperatures (4 °C, 25 °C, and 40 °C) emphasizes the crucial role of xanthan gum in the external phase for stability. As a viscosifying agent, xanthan contributes to increased system rigidity.
Additionally, the presence of a high concentration of lipophilic surfactant, particularly in emulsions rich in Span 60, extends the stability duration. This is attributed to Span 60 reinforcing rigidity in the interfacial film, thereby enhancing overall emulsion stability.
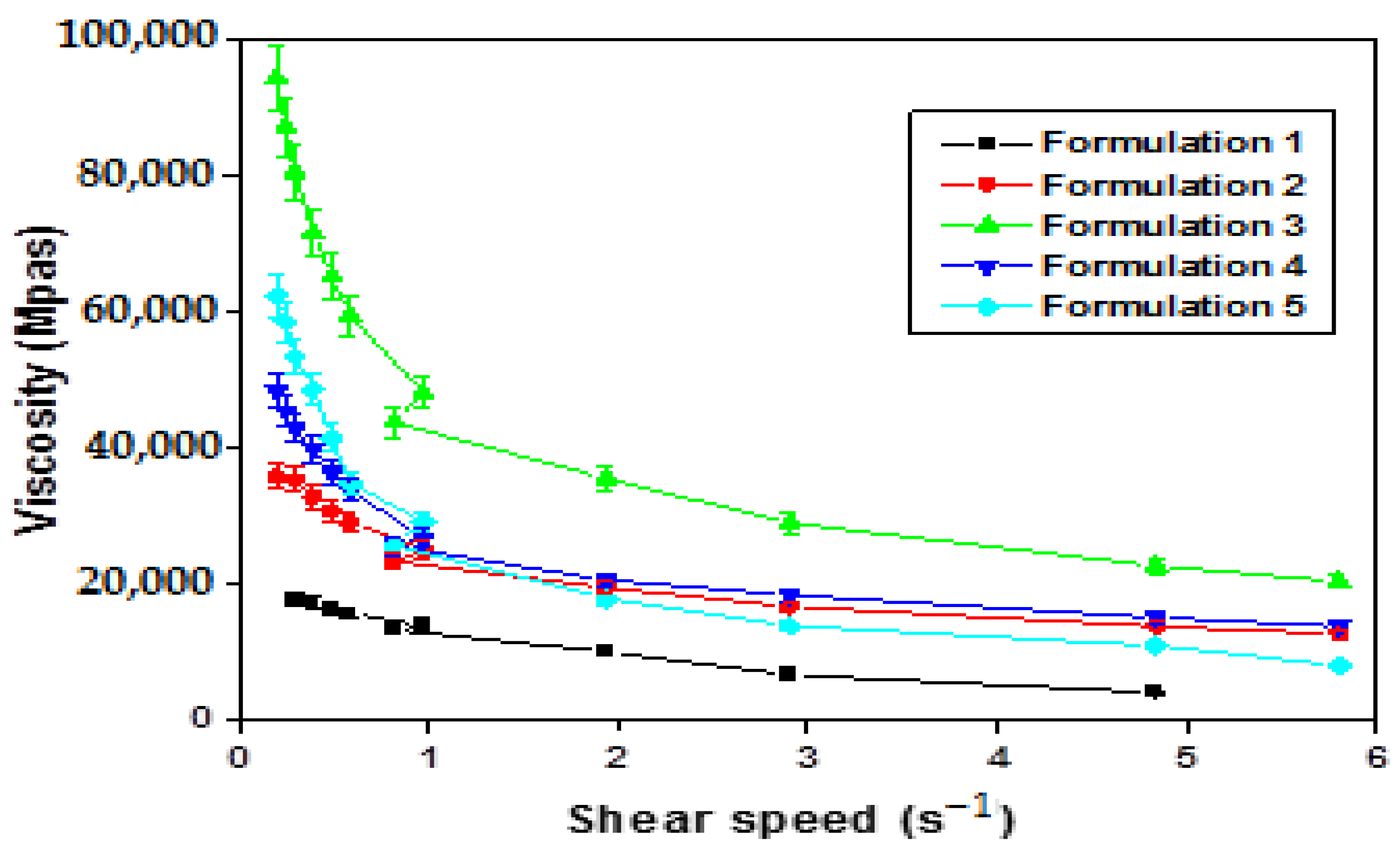
The viscosity evolution in relation to the shear rate for all formulations is visually depicted in Figure 9.
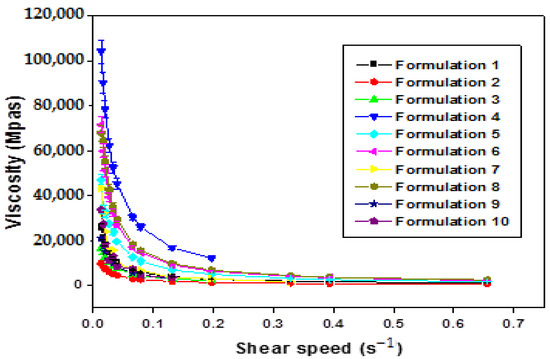
Figure 9.
Variation in apparent viscosity as a function of shear rate of W/O/W emulsions based on GX obtained by direct process.
Figure 9 provides insights into the rheological behavior of the formulated emulsions (F1–F10), revealing a pseudo-plastic, non-Newtonian fluid type.
The morphological examination, as depicted in Figure 10, distinctly showcases the dispersion of internal globules characterized by a relatively uniform size. The quantity of these globules varies in direct correlation with the concentration of each constituent, offering a visual representation of the emulsion’s internal structure.
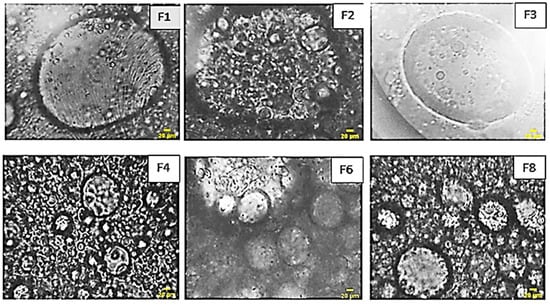
Figure 10.
Optical microscopy images of the formulas of XG based multiple emulsions. W/O/W obtained by direct process.
3.3. Formulation and Characterization of Multiple W/O/W Emulsions Stabilized by Pectin Using Direct Process
As previously mentioned (Section 3.2), multiple emulsions of W/O/W type, stabilized by pectin, were obtained by direct process, and the various formulations produced are presented in Table 5.

Table 5.
Experimental matrix of different formulas obtained by direct process stabilized with Pectin.
Characterization of W/O/W Multiple Emulsions
The formulated emulsions (F3, F5, F6, F7, and F9) present a uniform and consistent appearance, distinct from emulsions (F1, F2, F4, F8, and F10) that exhibit immediate phase separation. This observed separation is likely linked to the quantity of the stabilizing agent (pectin), suggesting that the presence of pectin mitigates instability phenomena in the formulated emulsions.
The visually consistent formulations (F3, F5, F6, F7, and F9) display heightened conductivity values, clearly indicating that the continuous phase is aqueous, affirming their classification as W/O/W type emulsions. Moreover, the observed acidity of pectin aligns with the measured low pH values.
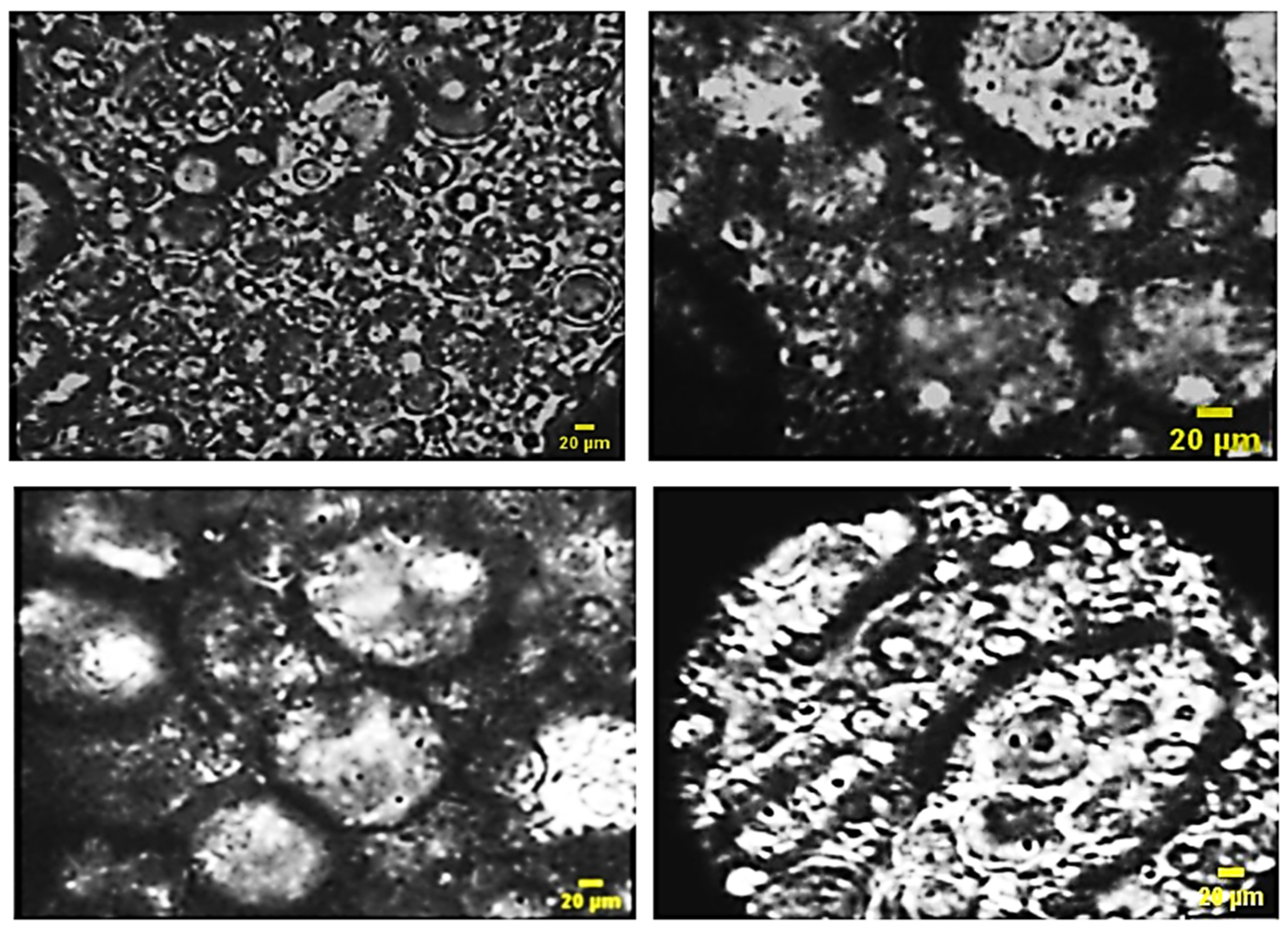
Microscopic examination offers valuable insights into the obtained systems, enabling the observation of their stability over time. The provided microscopic images (Figure 11) vividly illustrate the multiplicity of globules, confirming the formulation of multiple emulsions.
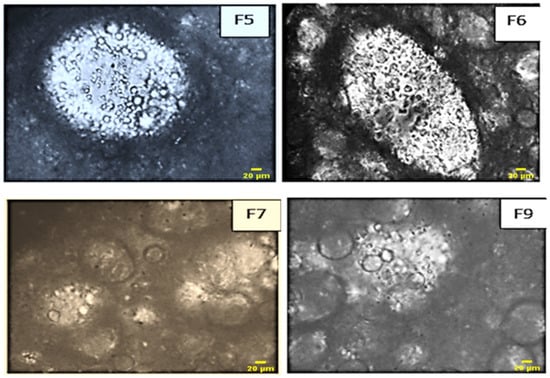
Figure 11.
Microscopic images of the formulas of multiples emulsion W/O/W obtained by direct process with pectin.
The viscosity study shows that apparent viscosity decreases when the shear rate increases (Figure 12), demonstrating that all the homogeneous emulsions obtained are of the non-Newtonian type with pseudo-plastic behavior.
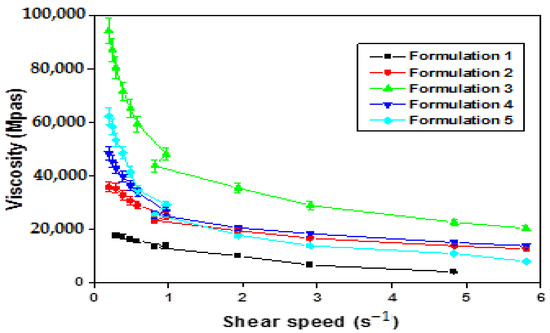
Figure 12.
Variation in apparent viscosity as a function of shear rate of W/O/W emulsions obtained by direct process with pectin.
4. Comparison of Results
In delving deeper into the intricacies of the formulation processes, it becomes evident that the stepwise approach stands out as the more favorable method, showcasing heightened viscosities and extended stability for multiple W/O/W emulsions compared to the direct process. The nuances between these methods encompass surfactant concentration differentials, the mode of phase introduction, and the vigor and duration of agitation. These pivotal factors play a direct role in shaping the nature, structure, and rheological properties of the emulsions.
Furthermore, the strategic placement of each surfactant within its designated phase, coupled with the sequence of introducing aqueous and oily phases, emerges as a critical determinant of globule size and overall emulsion stability. Notably, emulsions crafted with each emulsifier meticulously added to its respective phase yield finer globules with a more uniform distribution, a notable improvement over those generated through the uniform emulsifier addition in the same phase (direct process). The intensity and duration of agitation further dictate emulsion stability, with disparities in liquid velocities favoring droplet rupture in the direct process.
In summation, the stepwise process emerges as the more judicious choice in comparison to the direct process. Additionally, the stability of multiple emulsions is significantly influenced by the presence of polysaccharides, with xanthan gum displaying noteworthy and superior stability in contrast to pectin. Emulsions with meager xanthan gum content (0.5%) exhibit stability for an impressive period of almost 2 months at varying storage temperatures. Conversely, those laden with a substantial amount of pectin (2.75%) demonstrate instability within a mere 3 days.
5. Conclusions
In the pharmaceutical sector, double emulsions hold significant promise, particularly due to their unique ability, similar to liposomes and unlike microparticles, to transport hydrophilic active ingredients within their internal aqueous compartments.
The research findings underscore that multiple emulsions with lower concentrations of xanthan exhibit superior stability compared to those formulated with higher concentrations of pectin. Despite the inherent instability associated with multiple emulsions, the primary objective of this study is to formulate stable W/O/W type multiple emulsions with xanthan gum, showcasing stability for over two months. Characterization methods encompass physico-chemical studies, viscosity assessments, and microscopic analyses. The evaluation of stability at various storage temperatures indicates that optimal stability, characterized by the absence of instability phenomena, is achieved during cold storage.
The successful development of stable multiple emulsions with xanthan gum underscores a direct influence of biopolymer concentration on emulsion stability.
Highlights: Stable multiple emulsions formulated with xanthan gum/pectin using a direct process were developed
- -
- Stable multiple emulsions formulated with xanthan gum/pectin using process-by-step were developed.
- -
- Emulsions containing xanthan gum present viscoelastic properties higher than pectin.
- -
- The stepwise process is the most suitable compared to that of the direct process.
- -
- The stability obtained by xanthan gum is significant and better compared to that obtained once pectin is used.
Author Contributions
Conceptualization, M.B., M.M.Y., K.E., S.T., S.L., N.M.-M., A.N.E.H.S., H.T., M.K., A.A., B.J. and J.Z.; Data curation, M.B.; Formal analysis, M.B., M.M.Y., K.E., S.T., S.L., N.M.-M., A.N.E.H.S., H.T., M.K., A.A., B.J. and J.Z.; Investigation, M.B., M.M.Y., K.E., S.T., S.L., N.M.-M., A.N.E.H.S., H.T., M.K., A.A., B.J. and J.Z.; Methodology, M.B., M.M.Y., K.E., S.T., S.L., N.M.-M., A.N.E.H.S., H.T., M.K., A.A., B.J. and J.Z.; Project administration, H.T., A.A., B.J. and J.Z.; Resources, S.L., N.M.-M., H.T., M.K., A.A. and J.Z.; Software, M.B., M.M.Y., K.E., S.T., S.L., N.M.-M., H.T., A.A. and J.Z.; Supervision, M.M.Y., S.L., N.M.-M., A.A., B.J. and J.Z.; Validation, M.B., M.M.Y., K.E., S.T., S.L., N.M.-M., A.N.E.H.S., H.T., M.K., A.A., B.J. and J.Z.; Visualization, M.B., M.M.Y., K.E., S.T., S.L., N.M.-M., A.N.E.H.S., H.T., M.K., A.A., B.J. and J.Z.; Writing—original draft, K.E. and H.T.; Writing—review & editing, M.B., M.M.Y., S.T., S.L., N.M.-M., A.N.E.H.S., H.T., M.K., A.A., B.J. and J.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The data presented in this study are available in the manuscript.
Acknowledgments
The authors extend their thanks to the SAIDAL Pharmaceutical Company of Medea for the technical support and to Yamina Zouambia (University of Medea) for their contribution in conducting some of the characterization techniques.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Nooshkam, M.; Varidi, M. Maillard Conjugate-Based Delivery Systems for the Encapsulation, Protection, and Controlled Release of Nutraceuticals and Food Bioactive Ingredients: A Review. Food Hydrocoll. 2020, 100, 105389. [Google Scholar] [CrossRef]
- Khalid, N.; Kobayashi, I.; Neves, M.A.; Uemura, K.; Nakajima, M. Preparation and Characterization of Water-in-Oil Emulsions Loaded with High Concentration of l-Ascorbic Acid. LWT Food Sci. Technol. 2013, 51, 448–454. [Google Scholar] [CrossRef]
- Zamouche, M.; Tahraoui, H.; Laggoun, Z.; Mechati, S.; Chemchmi, R.; Kanjal, M.I.; Amrane, A.; Hadadi, A.; Mouni, L. Optimization and Prediction of Stability of Emulsified Liquid Membrane (ELM): Artificial Neural Network. Processes 2023, 11, 364. [Google Scholar] [CrossRef]
- Tatar, B.; Şahin, S. Advances in Food Rheology and Its Applications. In Rheology of Emulsion; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Yahoum, M.M.; Toumi, S.; Hentabli, S.; Tahraoui, H.; Lefnaoui, S.; Hadjsadok, A.; Amrane, A.; Kebir, M.; Moula, N.; Assadi, A.A. Experimental Analysis and Neural Network Modeling of the Rheological Behavior of Xanthan Gum and Its Derivatives. Materials 2023, 16, 2565. [Google Scholar] [CrossRef]
- Luhede, L.; Wollborn, T.; Fritsching, U. Stability of Multiple Emulsions under Shear Stress. Can. J. Chem. Eng. 2020, 98, 186–193. [Google Scholar] [CrossRef]
- Stasse, M.; Laurichesse, E.; Ribaut, T.; Anthony, O.; Héroguez, V.; Schmitt, V. Formulation of Concentrated Oil-in-Water-in-Oil Double Emulsions for Fragrance Encapsulation. Colloids Surf. A Physicochem. Eng. Asp. 2020, 592, 124564. [Google Scholar] [CrossRef]
- Seddari, S.; Moulai-Mostefa, N. Double Emulsions Stabilized by Xanthan in the Absence of Hydrophilic Surfactant. J. Dispers. Sci. Technol. 2016, 37, 530–535. [Google Scholar] [CrossRef]
- Nedjhioui, M.; Nasrallah, N.; Kebir, M.; Tahraoui, H.; Bouallouche, R.; Assadi, A.A.; Amrane, A.; Jaouadi, B.; Zhang, J.; Mouni, L. Designing an Efficient Surfactant–Polymer–Oil–Electrolyte System: A Multi-Objective Optimization Study. Processes 2023, 11, 1314. [Google Scholar] [CrossRef]
- Wang, J.; Shi, A.; Agyei, D.; Wang, Q. Formulation of Water-in-Oil-in-Water (W/O/W) Emulsions Containing Trans-Resveratrol. RSC Adv. 2017, 7, 35917–35927. [Google Scholar] [CrossRef]
- Leong, T.S.; Zhou, M.; Kukan, N.; Ashokkumar, M.; Martin, G.J. Preparation of Water-in-Oil-in-Water Emulsions by Low Frequency Ultrasound Using Skim Milk and Sunflower Oil. Food Hydrocoll. 2017, 63, 685–695. [Google Scholar] [CrossRef]
- Seifriz, W. Studies in Emulsions III–V. J. Phys. Chem. 2002, 29, 738–749. [Google Scholar] [CrossRef]
- Yan, J.; Bauer, W.-A.C.; Fischlechner, M.; Hollfelder, F.; Kaminski, C.F.; Huck, W.T. Monodisperse Water-in-Oil-in-Water (W/O/W) Double Emulsion Droplets as Uniform Compartments for High-Throughput Analysis via Flow Cytometry. Micromachines 2013, 4, 402–413. [Google Scholar] [CrossRef]
- Dams, S.S.; Walker, I.M. [5] Multiple Emulsions as Targetable Delivery Systems. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 1987; Volume 149, pp. 51–64. ISBN 0076-6879. [Google Scholar]
- Protat, M. Formation d’Emulsions Multiples Stimulables en une Seule Étape d’Emulsification: Effet du Sel et Évolution Vers des Architectures Biocompatibles. Ph.D. Thesis, Université Pierre et Marie Curie, Paris, France, 2016. [Google Scholar]
- Raina, N.; Rani, R.; Thakur, V.K.; Gupta, M. New Insights in Topical Drug Delivery for Skin Disorders: From a Nanotechnological Perspective. ACS Omega 2023, 8, 19145–19167. [Google Scholar] [CrossRef]
- Swamy, M.V.; Metta, S.; Kumar, A.A.; Reddy, C.; Dharani, G.; Ramya, K. Formulation and Characterization of a Multiple Emulsion Containing Vitamin-C. Indian J. 2023, 15, 1–6. [Google Scholar] [CrossRef]
- Deepak, S.; Sanjay, K.; Piyush, A. Recent Advancement, Technology & Applications of the Multiple Emulsions. Innovare J. Health Sci. 2013, 1, 19–23. [Google Scholar]
- Mudrić, J.; Šavikin, K.; Ibrić, S.; Đuriš, J. Double Emulsions (W/O/W Emulsions): Encapsulation of Plant Bioactives. Lek. Sirovine 2019, 39, 76–83. [Google Scholar] [CrossRef]
- Moussaoui, N.; Hammadi, L.; Boudjenane, N.-E.; Denine, R.R. Development of Multiple W/O/W Emulsions Used in Pharmaceutical Field: Effect of Additives and Insulin on Physicochemical and Rheological Stability of Emulsions. Colloid Polym. Sci. 2017, 295, 125–133. [Google Scholar] [CrossRef]
- Dhadde, G.S.; Mali, H.S.; Raut, I.D.; Nitalikar, M.M. An Overview on Multiple Emulsions. Asian J. Pharm. Technol. 2021, 11, 156–162. [Google Scholar] [CrossRef]
- Khan, B.A.; Akhtar, N.; Khan, H.M.S.; Waseem, K.; Mahmood, T.; Rasul, A.; Iqbal, M.; Khan, H. Basics of Pharmaceutical Emulsions: A Review. Afr. J. Pharm. Pharmacol. 2011, 5, 2715–2725. [Google Scholar]
- Ifrah, S.; Dahan, A.; Debotton, N. Towards Effective Antiviral Oral Therapy: Development of a Novel Self-Double Emulsifying Drug Delivery System for Improved Zanamivir Intestinal Permeability. Pharmaceutics 2023, 15, 2518. [Google Scholar] [CrossRef] [PubMed]
- Dluska, E.; Markowska-Radomska, A.; Metera, A.; Ordak, M. Multiple Emulsions as a Biomaterial-Based Delivery System for the Controlled Release of an Anti-Cancer Drug. J. Phys. Conf. Ser. 2020, 1681, 012021. [Google Scholar] [CrossRef]
- Jain, U.; Jaiswal, H. Designing 5-fluorouracil loaded Lipid Nanoparticles Using Double Emulsion and Solvent Evaporation for Skin Cancer Therapy. Oncol. Radiother. 2023, 17, 935–942. [Google Scholar]
- Lee, Y.; Lee, D.; Park, E.; Jang, S.; Cheon, S.Y.; Han, S.; Koo, H. Rhamnolipid-Coated W/O/W Double Emulsion Nanoparticles for Efficient Delivery of Doxorubicin/Erlotinib and Combination Chemotherapy. J. Nanobiotechnol. 2021, 19, 411. [Google Scholar]
- Yahoum, M.M.; Toumi, S.; Tahraoui, H.; Lefnaoui, S.; Hadjsadok, A.; Amrane, A.; Kebir, M.; Zhang, J.; Assadi, A.A.; Mouni, L. Evaluation of Physicochemical and Amphiphilic Properties of New Xanthan Gum Hydrophobically Functionalized Derivatives. Sustainability 2023, 15, 6345. [Google Scholar] [CrossRef]
- Jiao, J.; Burgess, D.J. Rheology and Stability of Water-in-Oil-in-Water Multiple Emulsions Containing Span 83 and Tween 80. Aaps Pharmsci 2003, 5, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Yahoum, M.M.; Toumi, S.; Tahraoui, H.; Lefnaoui, S.; Kebir, M.; Amrane, A.; Assadi, A.A.; Zhang, J.; Mouni, L. Formulation and Evaluation of Xanthan Gum Microspheres for the Sustained Release of Metformin Hydrochloride. Micromachines 2023, 14, 609. [Google Scholar] [CrossRef] [PubMed]
- Salager, J.-L.; Antón, R.; Bullón, J.; Forgiarini, A.; Marquez, R. How to Use the Normalized Hydrophilic-Lipophilic Deviation (HLDN) Concept for the Formulation of Equilibrated and Emulsified Surfactant-Oil-Water Systems for Cosmetics and Pharmaceutical Products. Cosmetics 2020, 7, 57. [Google Scholar] [CrossRef]
- Mellou, F.; Varvaresou, A.; Papageorgiou, S. Renewable Sources: Applications in Personal Care Formulations. Int. J. Cosmet. Sci. 2019, 41, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Schreiner, T.B.; Dias, M.M.; Barreiro, M.F.; Pinho, S.P. Saponins as Natural Emulsifiers for Nanoemulsions. J. Agric. Food Chem. 2022, 70, 6573–6590. [Google Scholar] [CrossRef]
- Ezzroug, K.; Bouaziz, M.N. Formulation et Évaluation de Vecteurs Particulaires à Base de Polysaccharides Natifs et Modifiés. Applications Pharmaceutiques. Ph.D. Thesis, University of Medea, Medea, Algeria, February 2019. [Google Scholar]
- Campos, P.; Melo, M.O.; Camargo Junior, F.B. Effects of Polysaccharide-Based Formulations on Human Skin. In Polysaccharides; Springer: Cham, Switzerland, 2014; pp. 1–8. [Google Scholar]
- Lim, X.-Y.; Li, J.; Yin, H.-M.; He, M.; Li, L.; Zhang, T. Stabilization of Essential Oil: Polysaccharide-Based Drug Delivery System with Plant-like Structure Based on Biomimetic Concept. Polymers 2023, 15, 3338. [Google Scholar] [CrossRef]
- Dickinson, E. Hydrocolloids as Emulsifiers and Emulsion Stabilizers. Food Hydrocoll. 2009, 23, 1473–1482. [Google Scholar] [CrossRef]
- Weiss, J.; Scherze, I.; Muschiolik, G. Polysaccharide Gel with Multiple Emulsion. Food Hydrocoll. 2005, 19, 605–615. [Google Scholar] [CrossRef]
- Dokić, L.; Krstonošić, V.; Nikolić, I. Physicochemical Characteristics and Stability of Oil-in-Water Emulsions Stabilized by OSA Starch. Food Hydrocoll. 2012, 29, 185–192. [Google Scholar] [CrossRef]
- Krstonošić, V.; Dokić, L.; Nikolić, I.; Milanović, M. Influence of Xanthan Gum on Oil-in-Water Emulsion Characteristics Stabilized by OSA Starch. Food Hydrocoll. 2015, 45, 9–17. [Google Scholar] [CrossRef]
- Wulff-Pérez, M.; de Vicente, J.; Martín-Rodríguez, A.; Gálvez-Ruiz, M.J. Controlling Lipolysis through Steric Surfactants: New Insights on the Controlled Degradation of Submicron Emulsions after Oral and Intravenous Administration. Int. J. Pharm. 2012, 423, 161–166. [Google Scholar] [CrossRef]
- Costa, C.; Medronho, B.; Filipe, A.; Mira, I.; Lindman, B.; Edlund, H.; Norgren, M. Emulsion Formation and Stabilization by Biomolecules: The Leading Role of Cellulose. Polymers 2019, 11, 1570. [Google Scholar] [CrossRef] [PubMed]
- Sharma, T.; Dohare, N.; Kumari, M.; Singh, U.K.; Khan, A.B.; Borse, M.S.; Patel, R. Comparative Effect of Cationic Gemini Surfactant and Its Monomeric Counterpart on the Conformational Stability and Activity of Lysozyme. RSC Adv. 2017, 7, 16763–16776. [Google Scholar] [CrossRef]
- Garti, N.; Bisperink, C. Double Emulsions: Progress and Applications. Curr. Opin. Colloid Interface Sci. 1998, 3, 657–667. [Google Scholar] [CrossRef]
- Stauffer, F. La Préparation D’émulsions Doubles Par Un Système Microfluidique. Sciences Pharmaceutiques. 26 March 2014. Available online: https://www.researchgate.net/profile/Fanny-Stauffer/publication/323795638_La_preparation_d’emulsions_doubles_par_un_systeme_microfluidique/links/5e9575bf92851c2f529f5099/La-preparation-demulsions-doubles-par-un-systeme-microfluidique.pdf (accessed on 3 February 2024).
- Besnard, L.; Protat, M.; Malloggi, F.; Daillant, J.; Cousin, F.; Pantoustier, N.; Guenoun, P.; Perrin, P. Breaking of the Bancroft Rule for Multiple Emulsions Stabilized by a Single Stimulable Polymer. Soft Matter 2014, 10, 7073–7087. [Google Scholar] [CrossRef]
- Besnard, L.; Marchal, F.; Paredes, J.F.; Daillant, J.; Pantoustier, N.; Perrin, P.; Guenoun, P. Multiple Emulsions Controlled by Stimuli-Responsive Polymers. Adv. Mater. 2013, 25, 2844–2848. [Google Scholar] [CrossRef]
- Hanson, J.A.; Chang, C.B.; Graves, S.M.; Li, Z.; Mason, T.G.; Deming, T.J. Nanoscale Double Emulsions Stabilized by Single-Component Block Copolypeptides. Nature 2008, 455, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Tu, F.; Lee, D. One-Step Encapsulation and Triggered Release Based on Janus Particle-Stabilized Multiple Emulsions. Chem. Commun. 2014, 50, 15549–15552. [Google Scholar] [CrossRef]
- Sun, G.; Liu, M.; Zhou, X.; Hong, L.; Ngai, T. Influence of Asymmetric Ratio of Amphiphilic Diblock Copolymers on One-Step Formation and Stability of Multiple Emulsions. Colloids Surf. A Physicochem. Eng. Asp. 2014, 454, 16–22. [Google Scholar] [CrossRef]
- Hong, L.; Sun, G.; Cai, J.; Ngai, T. One-Step Formation of w/o/w Multiple Emulsions Stabilized by Single Amphiphilic Block Copolymers. Langmuir 2012, 28, 2332–2336. [Google Scholar] [CrossRef] [PubMed]
- Kobori, T.; Matsumoto, A.; Sugiyama, S. pH-Dependent Interaction between Sodium Caseinate and Xanthan Gum. Carbohydr. Polym. 2009, 75, 719–723. [Google Scholar] [CrossRef]
- López, M.J.; Vargas-Garcıa, M.C.; Suarez-Estrella, F.; Moreno, J. Properties of Xanthan Obtained from Agricultural Wastes Acid Hydrolysates. J. Food Eng. 2004, 63, 111–115. [Google Scholar] [CrossRef]
- Bouyer, E.; Mekhloufi, G.; Huang, N.; Rosilio, V.; Agnely, F. β-Lactoglobulin, Gum Arabic, and Xanthan Gum for Emulsifying Sweet Almond Oil: Formulation and Stabilization Mechanisms of Pharmaceutical Emulsions. Colloids Surf. A Physicochem. Eng. Asp. 2013, 433, 77–87. [Google Scholar] [CrossRef]
- Garcıa-Ochoa, F.; Santos, V.; Casas, J.; Gómez, E. Xanthan Gum: Production, Recovery, and Properties. Biotechnol. Adv. 2000, 18, 549–579. [Google Scholar] [CrossRef]
- Yin, M.; Yang, D.; Lai, S.; Yang, H. Rheological Properties of Xanthan-Modified Fish Gelatin and Its Potential to Replace Mammalian Gelatin in Low-Fat Stirred Yogurt. LWT 2021, 147, 111643. [Google Scholar] [CrossRef]
- Santos, J.; Alcaide-González, M.; Trujillo-Cayado, L.; Carrillo, F.; Alfaro-Rodríguez, M. Development of Food-Grade Pickering Emulsions Stabilized by a Biological Macromolecule (Xanthan Gum) and Zein. Int. J. Biol. Macromol. 2020, 153, 747–754. [Google Scholar] [CrossRef]
- Lutz, R.; Aserin, A.; Wicker, L.; Garti, N. Release of Electrolytes from W/O/W Double Emulsions Stabilized by a Soluble Complex of Modified Pectin and Whey Protein Isolate. Colloids Surf. B Biointerfaces 2009, 74, 178–185. [Google Scholar] [CrossRef]
- Huang, X.; Tu, R.; Song, H.; Dong, K.; Geng, F.; Chen, L.; Huang, Q.; Wu, Y. Fabrication of Gelatin-EGCG-Pectin Ternary Complex Stabilized W/O/W Double Emulsions by Ultrasonic Emulsification: Physicochemical Stability, Rheological Properties and Structure. J. Food Eng. 2023, 338, 111259. [Google Scholar] [CrossRef]
- Mezger, T. The Rheology Handbook: For Users of Rotational and Oscillatory Rheometers; European Coatings: Nuremberg, Germany, 2020; ISBN 3-86630-532-X. [Google Scholar]
- Manral, K. Viscoelastic Properties and Rheological Characterization of Carbomers. Int. J. Res. Eng. Technol. 2015, 1, 17–30. [Google Scholar]
- Saharudin, S.H.; Ahmad, Z.; Basri, M. Role of Xanthan Gum on Physicochemical and Rheological Properties of Rice Bran Oil Emulsion. Int. Food Res. J. 2016, 23, 1361. [Google Scholar]
- Pal, R. Rheology of Emulsions Containing Polymeric Liquids. Encycl. Emuls. Technol. 1996, 4, 93–263. [Google Scholar]
- Frank, D.; Appelqvist, I.; Piyasiri, U.; Wooster, T.J.; Delahunty, C. Proton Transfer Reaction Mass Spectrometry and Time Intensity Perceptual Measurement of Flavor Release from Lipid Emulsions Using Trained Human Subjects. J. Agric. Food Chem. 2011, 59, 4891–4903. [Google Scholar] [CrossRef]
- Van Aken, G.A.; Vingerhoeds, M.H.; De Wijk, R.A. Textural Perception of Liquid Emulsions: Role of Oil Content, Oil Viscosity and Emulsion Viscosity. Food Hydrocoll. 2011, 25, 789–796. [Google Scholar] [CrossRef]
- McClements, D.J. Food Emulsions: Principles, Practices, and Techniques; CRC Press: Boca Raton, FL, USA, 2004; ISBN 0-429-12389-2. [Google Scholar]
- André, P.; Aubry, J.-M.; Berteina-Raboin, S.; Bouix-Peter, C.; Del Bino, S.; Humbert, P.; Leclaire, J.; Marrot, L.; Masson, C.; Pélisson, I. Formulation Cosmétique Matières Premières, Concepts et Procédés Innovants; EDP: Les Ulis Cedex, France, 2005. [Google Scholar]
- Guo, T.; Li, J.; Guo, M.; Yang, Q.; Dai, X.; Qiao, X.; Song, Z.; Tian, C.; Li, Y.; Ge, H. Low Temperature Inhibits Pectin Degradation by PpCBFs to Prolong Peach Storage Time. J. Food Sci. 2023, 88, 3725–3736. [Google Scholar] [CrossRef] [PubMed]
- Fraeye, I.; De Roeck, A.; Duvetter, T.; Verlent, I.; Hendrickx, M.; Van Loey, A. Influence of Pectin Properties and Processing Conditions on Thermal Pectin Degradation. Food Chem. 2007, 105, 555–563. [Google Scholar] [CrossRef]
- Craig, D.Q.; Kee, A.; Tamburic, S.; Barnes, D. An Investigation into the Temperature Dependence of the Rheological Synergy between Xanthan Gum and Locust Bean Gum Mixtures. J. Biomater. Sci. Polym. Ed. 1997, 8, 377–389. [Google Scholar] [CrossRef] [PubMed]
- Reinoso, D.; Martín-Alfonso, M.J.; Luckham, P.F.; Martinez-Boza, F.J. Flow Behavior and Thermal Resistance of Xanthan Gum in Formate Brine. J. Pet. Sci. Eng. 2020, 188, 106881. [Google Scholar] [CrossRef]
- Pelletier, E.; Viebke, C.; Meadows, J.; Williams, P.A. A Rheological Study of the Order–Disorder Conformational Transition of Xanthan Gum. Biopolym. Orig. Res. Biomol. 2001, 59, 339–346. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).