Evaluation of Stabilization and Physical–Chemical Properties of CNT Antifreeze Nanofluid Prepared in 50:50 EG/Water by Modified Strategy
Abstract
:1. Introduction
2. Material and Method
2.1. Materials
2.2. Methodology
2.2.1. Preparation of Nanofluid by Conventional Method (Method 1)
2.2.2. Preparation of Nanofluid by Modified Method (Method 2)
2.3. Characterization
3. Result and Discussion
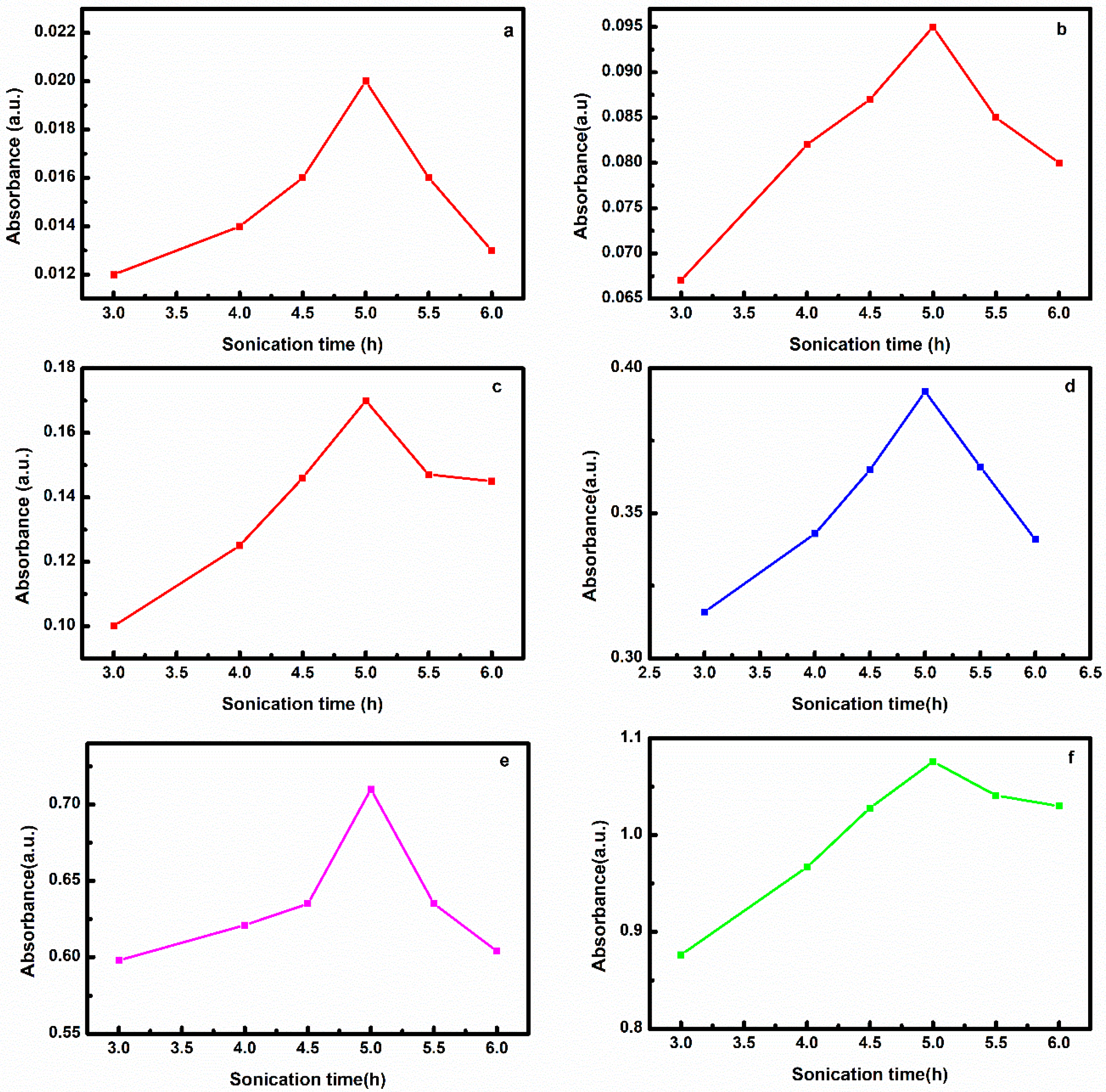
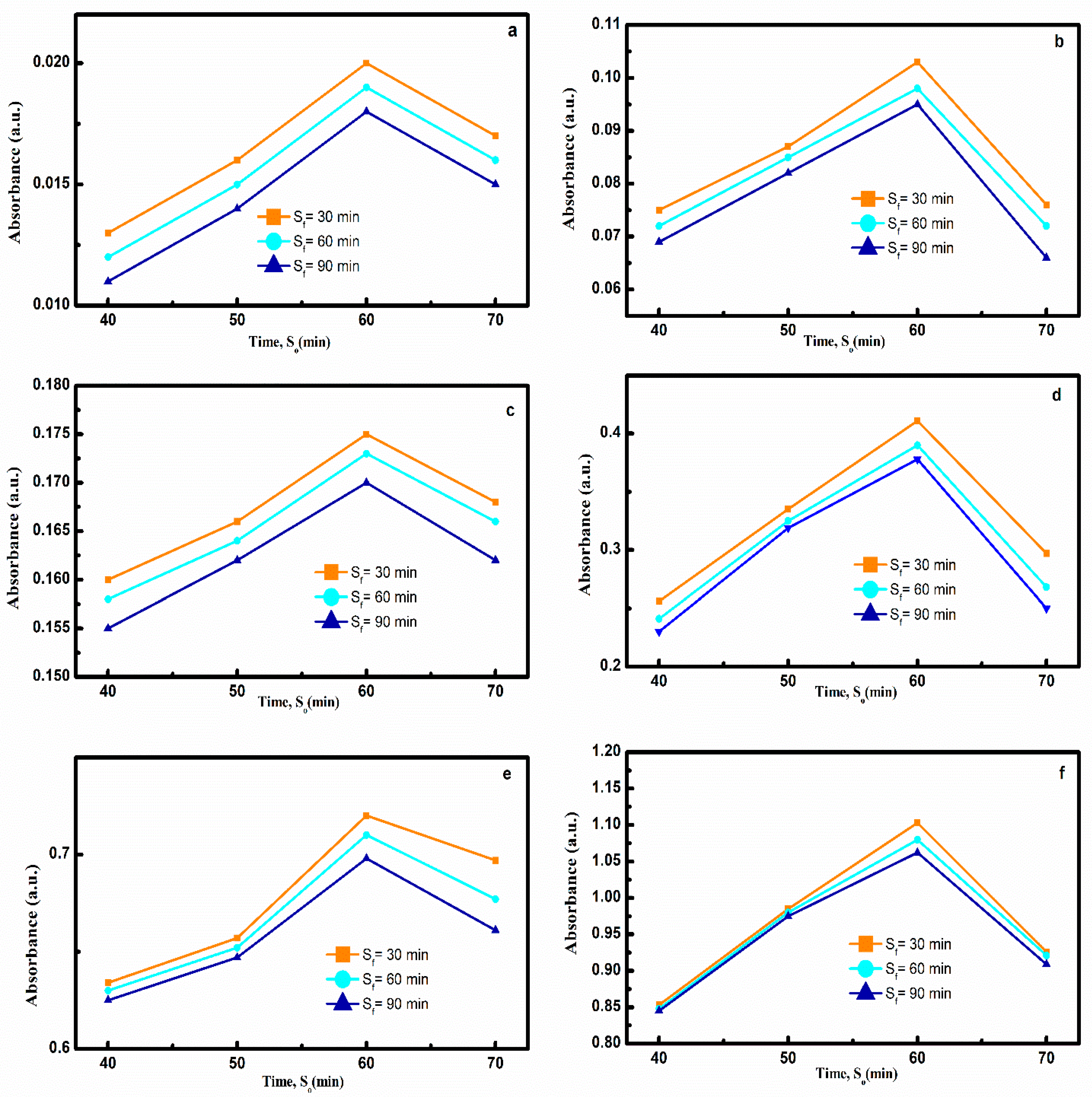
3.1. Effect of Sonication Time
3.1.1. Effect of Sonication Time on Nanofluid Prepared by Method 1
3.1.2. Effect of Sonication Time on Nanofluid Prepared by Method 2
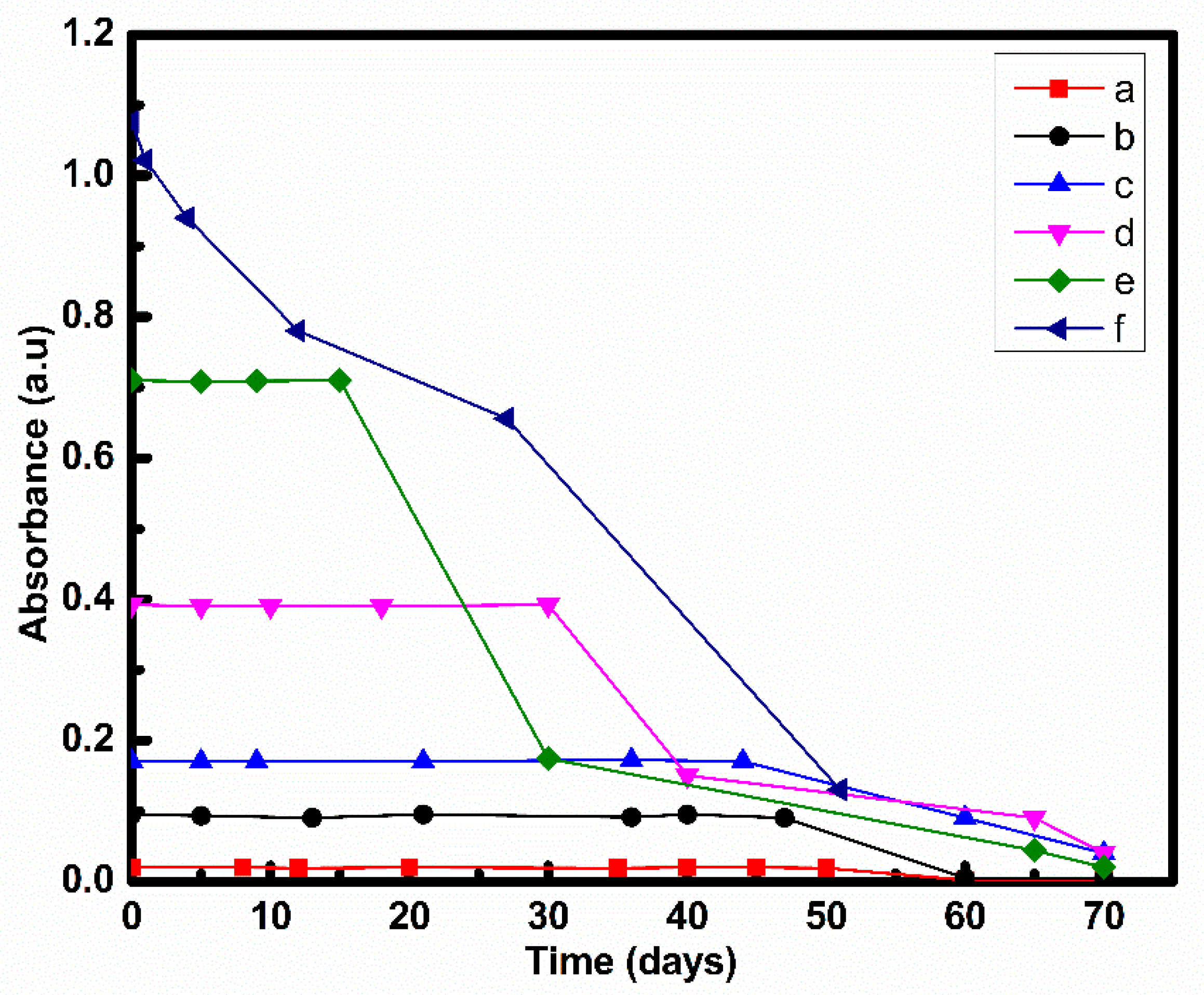
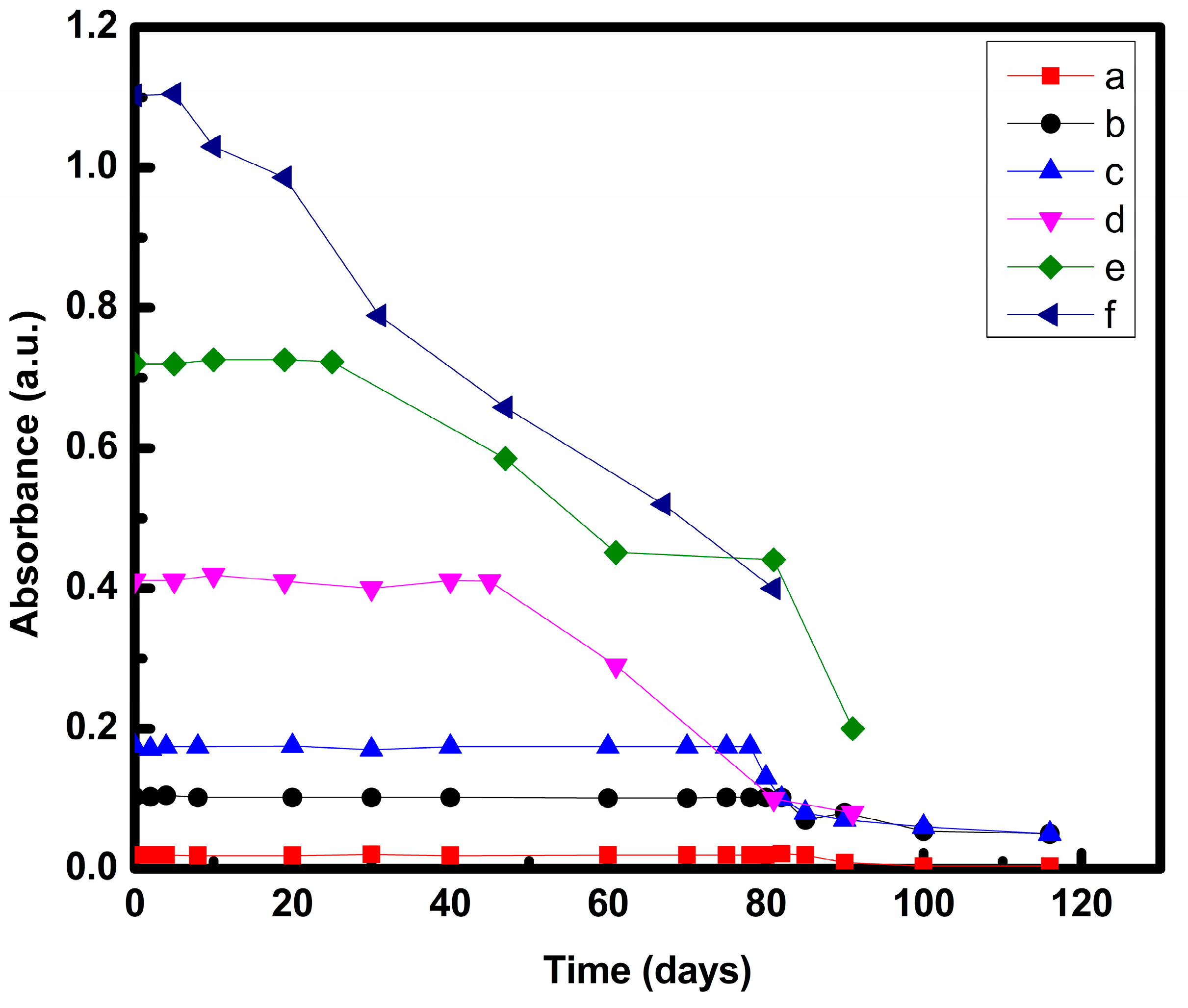
3.2. Stability Analysis
3.2.1. UV-Vis Spectroscopy
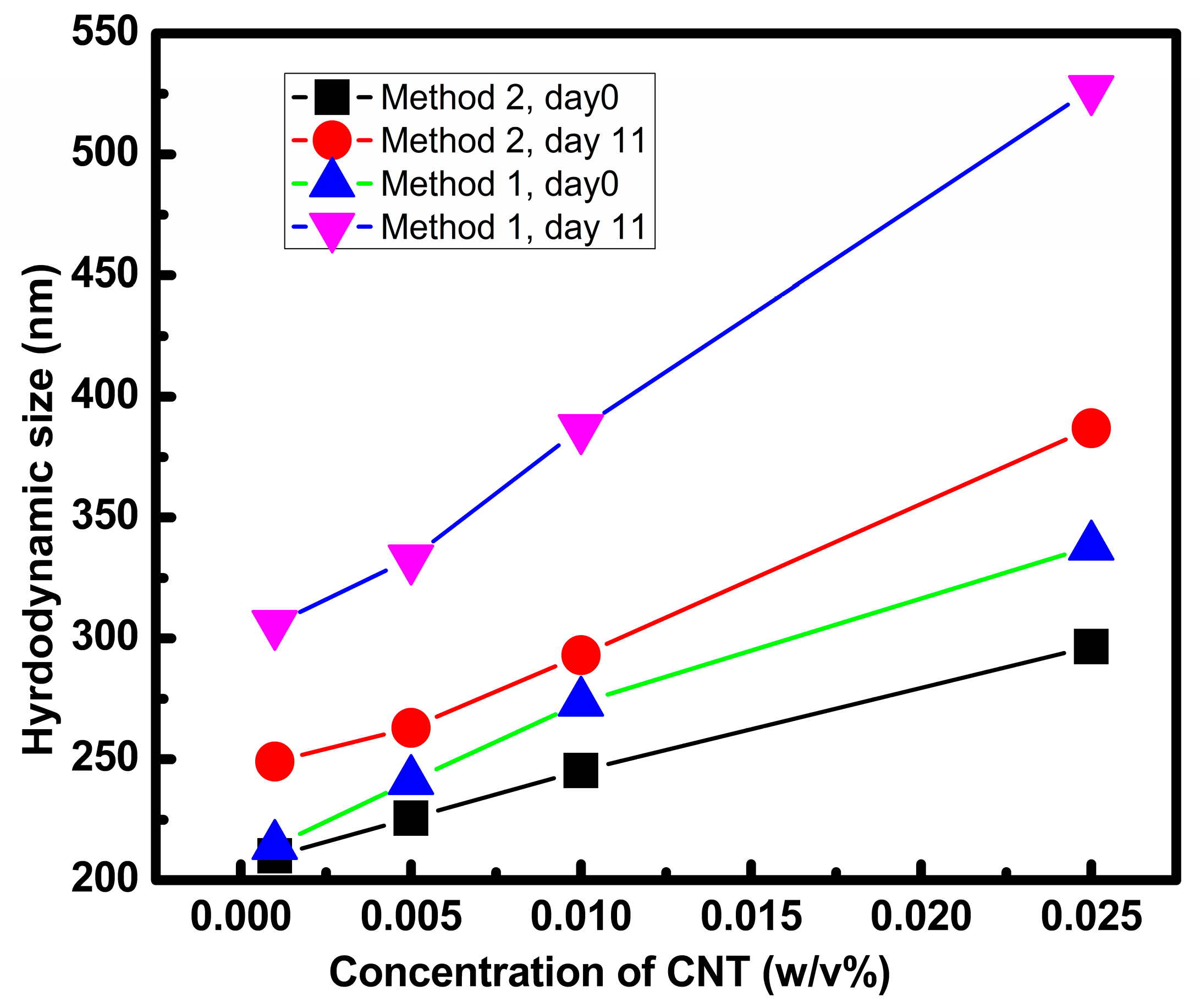
3.2.2. Dynamic Light Scattering
3.3. Study of Interactions between CNT and Base Fluid
3.3.1. FTIR Spectroscopy
3.3.2. NMR Spectroscopy
3.4. Physical–Chemical Properties
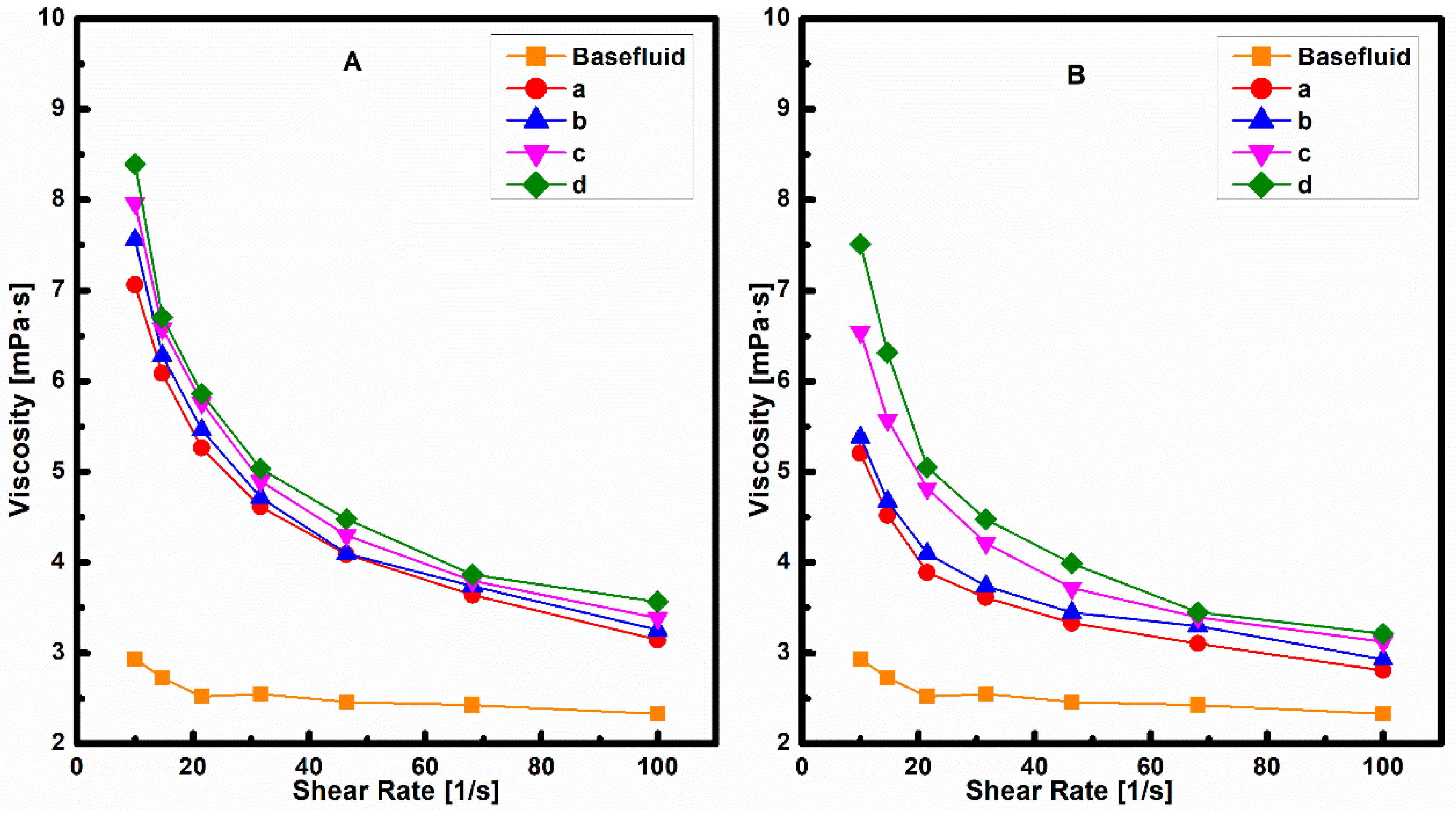
3.4.1. Viscosity
3.4.2. Density
3.4.3. Thermal Conductivity
4. Comparison of Present Work with Work Reported in Literature
5. Conclusions
- ▪
- Method 2 requires less time (1.5 h) for preparation of CNT antifreeze nanofluid in EG/water as compared to Method 1 (5 h).
- ▪
- The preparation method plays an important role in stabilization of nanofluid. It was found that noncovalent interactions are responsible for stabilization of CNT nanofluid.
- ▪
- A higher enhancement in thermal conductivity was observed in the case of Method 2 (20%) in comparison to Method 1 (17%).
- ▪
- A 7.4% greater increase in viscosity was seen in the case of nanofluid prepared by Method 1 in comparison to Method 2 at 100/s shear rate.
- ▪
- In both nanofluids, a marginal difference is seen in density as compared with the base fluid.
- ▪
- This study provides an alternate way of producing CNT antifreeze nanofluid in EG/water system, without the use of surfactant or the need of long term sonication. The better way of preparing such type of nanofluids is to first disperse CNT in EG, which has a higher capability of dispersing CNT, followed by the addition of water.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| CNT | Carbon Nanotube |
| EG | Ethylene Glycol |
| Φ | CNT amount (V%) |
| ρ(bf) | Base fluid density (Kg/m3) |
| ρ(nf) | Nanofluid Density (Kg/m3) |
| M(nf) | Nanofluid mass (Kg) |
| M(bf) | Base fluid mass (Kg) |
| V(nf) | Nanofluid volume (m3) |
| S0 | Sonication time prior to addition of water (min) |
| Sf | Sonication time after addition of water (min) |
References
- American Society of Heating, Refrigerating and Air-Conditioning Engineers. Ashrae Handbook: Fundamentals; ASHRAE: Atlanta, GA, USA, 2009. [Google Scholar]
- Roohani, E.; Toghraie, D. Heat transfer improvement of antifreeze by changing it to hybrid nanofluid: Effects of hybrid magnesium oxide–graphene oxide nanopowders. J. Therm. Anal. Calorim. 2022, 147, 6777–6791. [Google Scholar] [CrossRef]
- Afrand, M. Experimental study on thermal conductivity of ethylene glycol containing hybrid nano-additives and development of a new correlation. Appl. Therm. Eng. 2017, 110, 1111–1119. [Google Scholar] [CrossRef]
- Izadi, F.; Ranjbarzadeh, R.; Kalbasi, R.; Afrand, M. A new experimental correlation for non-Newtonian behavior of COOH-DWCNTs/antifreeze nanofluid. Phys. E Low Dimens. Syst. Nanostruct. 2018, 98, 83–89. [Google Scholar] [CrossRef]
- Murshed, S.M.S.; de Castro, C.A.N. Conduction and convection heat transfer characteristics of ethylene glycol based nanofluids—A review. Appl. Energy 2016, 184, 681–695. [Google Scholar] [CrossRef]
- Azmi, W.H.; Hamid, K.A.; Usri, N.A.; Mamat, R.; Sharma, K.V. Heat transfer augmentation of ethylene glycol: Water nanofluids and applications—A review. Int. Commun. Heat Mass Transf. 2016, 75, 13–23. [Google Scholar] [CrossRef]
- Ahmadi, M.H.; Mirlohi, A.; Nazari, M.A.; Ghasempour, R. A review of thermal conductivity of various nanofluids. J. Mol. Liq. 2018, 265, 181–188. [Google Scholar] [CrossRef]
- Kumar, L.H.; Kazi, S.N.; Masjuki, H.H.; Zubir, M.N.M. A review of recent advances in green nanofluids and their application in thermal systems. Chem. Eng. J. 2022, 429, 132321. [Google Scholar] [CrossRef]
- Said, Z.; Sundar, L.S.; Tiwari, A.K.; Ali, H.M.; Sheikholeslami, M.; Bellos, E.; Babar, H. Recent advances on the fundamental physical phenomena behind stability, dynamic motion, thermophysical properties, heat transport, applications, and challenges of nanofluids. Phys. Rep. 2022, 946, 1–94. [Google Scholar] [CrossRef]
- Rudyak, V.Y.; Tret’yakov, D.S. Rheological Properties of Water- and Ethylene-Glycol-Based Nanofluids with Single-Walled Carbon Nanotubes. J. Eng. Phys. Thermophys. 2021, 94, 1208–1216. [Google Scholar] [CrossRef]
- Kumaresan, V.; Velraj, R. Experimental investigation of the physical-chemical properties of water-ethylene glycol mixture based CNT nanofluids. Thermochim Acta 2012, 545, 180–186. [Google Scholar] [CrossRef]
- Sandhu, H.; Gangacharyulu, D. An experimental study on stability and some thermophysical properties of multiwalled carbon nanotubes with water–ethylene glycol mixtures. Part. Sci. Technol. 2017, 35, 547–554. [Google Scholar] [CrossRef]
- Ganeshkumar, J.; Kathirkaman, D.; Raja, K.; Kumaresan, V.; Velraj, R. Experimental study on density, thermal conductivity, specific heat, and viscosity of water-ethylene glycol mixture dispersed with carbon nanotubes. Therm. Sci. 2017, 21 Pt A, 255–265. [Google Scholar] [CrossRef]
- Soltanimehr, M.; Afrand, M. Thermal conductivity enhancement of COOH-functionalized MWCNTs/ethylene glycol–water nanofluid for application in heating and cooling systems. Appl. Therm. Eng. 2016, 105, 716–723. [Google Scholar] [CrossRef]
- Mirbagheri, M.H.; Akbari, M.; Mehmandoust, B. Proposing a new experimental correlation for thermal conductivity of nanofluids containing of functionalized multiwalled carbon nanotubes suspended in a binary base fluid. Int. Commun. Heat Mass Transf. 2018, 98, 216–222. [Google Scholar] [CrossRef]
- Yan, S.R.; Toghraie, D.; Abdulkareem, L.A.; Alizadeh, A.; Barnoon, P.; Afrand, M. The rheological behavior of MWCNTs–ZnO/Water–Ethylene glycol hybrid non-Newtonian nanofluid by using of an experimental investigation. J. Mater. Res. Technol. 2020, 9, 8401–8406. [Google Scholar] [CrossRef]
- Esfe, M.H.; Alidoust, S.; Tamrabad, S.N.H.; Toghraie, D.; Hatami, H. Thermal conductivity of MWCNT-TiO2/Water-EG hybrid nanofluids: Calculating the price performance factor (PPF) using statistical and experimental methods (RSM). Case Stud. Therm. Eng. 2023, 48, 103094. [Google Scholar] [CrossRef]
- Shahsavani, E.; Afrand, M.; Kalbasi, R. Experimental study on rheological behavior of water–ethylene glycol mixture in the presence of functionalized multi-walled carbon nanotubes: A novel correlation for the non-Newtonian nanofluid. J. Therm. Anal. Calorim. 2018, 131, 1177–1185. [Google Scholar] [CrossRef]
- Dehkordi, R.A.; Esfe, M.H.; Afrand, M. Effects of functionalized single walled carbon nanotubes on thermal performance of antifreeze: An experimental study on thermal conductivity. Appl. Therm. Eng. 2017, 120, 358–366. [Google Scholar] [CrossRef]
- Bagheri, H.; Nadooshan, A.A. The effects of hybrid nano-powder of zinc oxide and multi walled carbon nanotubes on the thermal conductivity of an antifreeze. Phys. E Low Dimens. Syst. Nanostruct. 2018, 103, 361–366. [Google Scholar] [CrossRef]
- Moradi, A.; Zareh, M.; Afrand, M.; Khayat, M. Effects of temperature and volume concentration on thermal conductivity of TiO2-MWCNTs (70-30)/EG-water hybrid nano-fluid. Powder Technol. 2020, 362, 578–585. [Google Scholar] [CrossRef]
- Eshgarf, H.; Sina, N.; Esfe, M.H.; Izadi, F.; Afrand, M. Prediction of rheological behavior of MWCNTs–SiO2/EG–water non-Newtonian hybrid nanofluid by designing new correlations and optimal artificial neural networks. J. Therm. Anal. Calorim. 2018, 132, 1029–1038. [Google Scholar] [CrossRef]
- Yadav, P.; Gupta, S.M.; Sharma, S.K. A review on stabilization of carbon nanotube nanofluid. J. Therm. Anal. Calorim. 2022, 147, 6537–6561. [Google Scholar] [CrossRef]
- Yadav, P.; Gupta, S.M.; Sharma, S.K. Preparation and characterization of surfactant-free CNT based nanofluid in EG/water (60:40 ratio) basefluid for refrigerant application. J. Therm. Anal. Calorim. 2023, 148, 10037–10050. [Google Scholar] [CrossRef]
- Pak, B.C.; Cho, Y.I. Hydrodynamic and heat transfer study of dispersed fluids with submicron metallic oxide particles. Exp. Heat Transf. 1998, 11, 151–170. [Google Scholar] [CrossRef]
- Islam, M.M.; Chong, T.H.; Shah, S.K.; Shahrul, I.M.; Rahman, S.; Bui, L.D.; Ma, A. Effect of ultrasonication duration on colloidal structure and viscosity of alumina-water nanofluid. Ind. Eng. Chem. Res. 2014, 53, 6677–6684. [Google Scholar] [CrossRef]
- Javadian, S.; Motaee, A.; Sharifi, M.; Aghdastinat, H.; Taghavi, F. Dispersion stability of multi-walled carbon nanotubes in catanionic surfactant mixtures. Colloids Surf. A Physicochem. Eng. Asp. 2017, 531, 141–149. [Google Scholar] [CrossRef]
- Pantzali, M.N.; Mouza, A.A.; Paras, S.V. Investigating the efficacy of nanofluids as coolants in plate heat exchangers (PHE). Chem. Eng. Sci. 2009, 64, 3290–3300. [Google Scholar] [CrossRef]
- Zhao, M.; Lv, W.; Li, Y.; Dai, C.; Zhou, H.; Song, X.; Wu, Y. A study on preparation and stabilizing mechanism of hydrophobic silica nanofluids. Materials 2018, 11, 1385. [Google Scholar] [CrossRef]
- Haghighi, E.B.; Nikkam, N.; Saleemi, M.; Behi, M.; Mirmohammadi, S.A.; Poth, H.; Khodabandeh, R.; Toprak, M.S.; Muhammed, M.; Palm, B. Shelf stability of nanofluids and its effect on thermal conductivity and viscosity. Meas. Sci. Technol. 2013, 24, 105301. [Google Scholar] [CrossRef]
- Chakraborty, S.; Panigrahi, P.K. Stability of nanofluid: A review. Appl. Therm. Eng. 2020, 174, 115259. [Google Scholar] [CrossRef]
- Witharana, S.; Palabiyik, I.; Musina, Z.; Ding, Y. Stability of glycol nanofluids—The theory and experiment. Powder Technol. 2013, 239, 72–77. [Google Scholar] [CrossRef]
- Suganthi, K.S.; Rajan, K.S. A formulation strategy for preparation of ZnO-Propylene glycol-water nanofluids with improved transport properties. Int. J. Heat Mass Transf. 2014, 71, 653–663. [Google Scholar] [CrossRef]
- Ilyas, S.U.; Pendyala, R.; Marneni, N. Stability of Nanofluids. In Topics in Mining, Metallurgy and Materials Engineering; Springer Science and Business Media Deutschland GmbH: Berlin/Heidelberg, Germany, 2017; pp. 1–31. [Google Scholar] [CrossRef]
- Gupta, N.; Gupta, S.M.; Sharma, S.K. Synthesis, characterization and dispersion stability of water-based Cu–CNT hybrid nanofluid without surfactant. Microfluid. Nanofluidics 2021, 25, 14. [Google Scholar] [CrossRef]
- Guo, Y.C.; Cai, C.; Zhang, Y.H. Observation of conformational changes in ethylene glycol-water complexes by FTIR-ATR spectroscopy and computational studies. AIP Adv. 2018, 8, 055308. [Google Scholar] [CrossRef]
- Krishnan, B.K.; Krishnan, D.R.S. Raman and infrared spectra of ethylene glycol. Proc. Indian Acad. Sci. Sect. A 1966, 64, 111–122. [Google Scholar] [CrossRef]
- Balamurugan, K.; Baskar, P.; Kumar, R.M.; Das, S.; Subramanian, V. Interaction of carbon nanotube with ethylene glycol-water binary mixture: A molecular dynamics and density functional theory investigation. J. Phys. Chem. C 2012, 116, 4365–4373. [Google Scholar] [CrossRef]
- Babij, N.R.; McCusker, E.O.; Whiteker, G.T.; Canturk, B.; Choy, N.; Creemer, L.C.; De Amicis, C.V.; Hewlett, N.M.; Johnson, P.L.; Knobelsdorf, J.A.; et al. NMR Chemical Shifts of Trace Impurities: Industrially Preferred Solvents Used in Process and Green Chemistry. Org. Process Res. Dev. 2016, 20, 661–667. [Google Scholar] [CrossRef]
- Jo, B.; Banerjee, D. Viscosity measurements of multi-walled carbon nanotubes-based high temperature nanofluids. Mater. Lett. 2014, 122, 212–215. [Google Scholar] [CrossRef]
- Amiri, A.; Sadri, R.; Ahmadi, G.; Chew, B.T.; Kazi, S.N.; Shanbedi, M.; Alehashem, M.S. Synthesis of polyethylene glycol-functionalized multi-walled carbon nanotubes with a microwave-assisted approach for improved heat dissipation. RSC Adv. 2015, 5, 35425–35434. [Google Scholar] [CrossRef]
- Lee, J.; Lee, H.; Baik, Y.J.; Koo, J. Quantitative analyses of factors affecting thermal conductivity of nanofluids using an improved transient hot-wire method apparatus. Int. J. Heat Mass Transf. 2015, 89, 116–123. [Google Scholar] [CrossRef]
- Hays, A.; Marsh, C.P.; Alvarado, J.; Franks, R. The Effect of Nanoparticle Agglomeration on Enhanced Nanofluidic Thermal Conductivity. 2006. Available online: http://docs.lib.purdue.edu/iracc/829 (accessed on 8 November 2022).
- Kumar, R.M.; Baskar, P.; Balamurugan, K.; Das, S.; Subramanian, V. Interaction of ethylene glycol-water clusters with aromatic surfaces. RSC Adv. 2013, 3, 7798–7807. [Google Scholar] [CrossRef]
- Kakavandi, A.; Akbari, M. Experimental investigation of thermal conductivity of nanofluids containing of hybrid nanoparticles suspended in binary base fluids and propose a new correlation. Int. J. Heat Mass Transf. 2018, 124, 742–751. [Google Scholar] [CrossRef]
- Irani, M.; Afrand, M.; Mehmandoust, B. Curve fitting on experimental data of a new hybrid nano-antifreeze viscosity: Presenting new correlations for non-Newtonian nanofluid. Phys. A Stat. Mech. Its Appl. 2019, 531, 120837. [Google Scholar] [CrossRef]
- Eshgarf, H.; Afrand, M. An experimental study on rheological behavior of non-Newtonian hybrid nano-coolant for application in cooling and heating systems. Exp. Therm. Fluid Sci. 2016, 76, 221–227. [Google Scholar] [CrossRef]



| Concentration of CNT (w/v%) | Stability of Nanofluid (Days) | |
|---|---|---|
| Method 1 | Method 2 | |
| 0.001 | 50 | 85 |
| 0.005 | 47 | 82 |
| 0.01 | 44 | 78 |
| 0.025 | 30 | 45 |
| 0.05 | 15 | 25 |
| 0.075 | 0 | 5 |
| Shear Rate (1/s) | % Enhancement in Comparison to Base Fluid | |||
|---|---|---|---|---|
| 0.001 w/v% | 0.005 w/v% | 0.01 w/v% | 0.025 w/v% | |
| 10 | 140.83 | 157.88 | 171.52 | 186.40 |
| 14.7 | 123.10 | 130.44 | 141.44 | 145.87 |
| 21.5 | 108.71 | 116.64 | 128.54 | 132.43 |
| 31.6 | 81.22 | 85.14 | 92.33 | 97.63 |
| 46.4 | 66.28 | 66.69 | 74.82 | 82.14 |
| 68.1 | 50.00 | 54.12 | 56.56 | 59.28 |
| 100 | 35.03 | 39.75 | 45.33 | 53.06 |
| Shear Rate (1/s) | % Enhancement in Comparison to Base Fluid | |||
|---|---|---|---|---|
| 0.001 w/v% | 0.005 w/v% | 0.01 w/v% | 0.025 w/v% | |
| 10 | 77.55 | 83.49 | 123.03 | 156.18 |
| 14.7 | 65.73 | 71.34 | 104.27 | 131.46 |
| 21.5 | 54.29 | 62.59 | 91.00 | 100.27 |
| 31.6 | 41.85 | 46.70 | 65.39 | 75.72 |
| 46.4 | 35.46 | 40.16 | 51.15 | 62.25 |
| 68.1 | 27.94 | 35.97 | 39.88 | 42.18 |
| 100 | 20.58 | 25.69 | 34.20 | 37.89 |
| Concentration of CNT (w/v%) | Experimental Density (Kg/cc) of Nanofluid Prepared by Method 1 | Experimental Density (Kg/cc) of Nanofluid Prepared by Method 2 | Theoretical Density (Kg/cc) |
|---|---|---|---|
| 0 (base fluid) | 1058 | 1058 | |
| 0.001 | 1058.16 | 1058.20 | 1058.48 |
| 0.005 | 1059.67 | 1060 | 1060.39 |
| 0.01 | 1060.67 | 1061.34 | 1062.89 |
| 0.025 | 1066.01 | 1067.35 | 1070.50 |
| (EG/Water) | Nanoparticle Used | Technique Used for Dispersion | Stability | Reference |
|---|---|---|---|---|
| 50:50 | COOH-DWCNT | 5–6 h sonication | - | [4] |
| 50:50 | MWCNT-SiC | 1 h stirring and 45 min sonication | - | [45] |
| 50:50 | Al2O3-MWCNT | 45 min stirring and 1.5 h sonication | - | [46] |
| 50:50 | COOH-SWCNT | 2.30 h stirring and 6 h sonication | 10 days | [19] |
| 50:50 | MWCNT-SiO2 | 2 h stirring and 5–6 h sonication | - | [47] |
| 50:50 | MWCNT-TiO2 | 3 h stirring and 5 h sonication | 2 weeks | [17] |
| 50:50 | COOH-MWCNT | 5 h sonication | 50 days | Present study |
| 50:50 | COOH-MWCNT | 90 min sonication | 85 days | Present study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yadav, P.; Gupta, S.M.; Sharma, S.K. Evaluation of Stabilization and Physical–Chemical Properties of CNT Antifreeze Nanofluid Prepared in 50:50 EG/Water by Modified Strategy. ChemEngineering 2023, 7, 117. https://doi.org/10.3390/chemengineering7060117
Yadav P, Gupta SM, Sharma SK. Evaluation of Stabilization and Physical–Chemical Properties of CNT Antifreeze Nanofluid Prepared in 50:50 EG/Water by Modified Strategy. ChemEngineering. 2023; 7(6):117. https://doi.org/10.3390/chemengineering7060117
Chicago/Turabian StyleYadav, Priyanka, Shipra Mital Gupta, and Surendra Kumar Sharma. 2023. "Evaluation of Stabilization and Physical–Chemical Properties of CNT Antifreeze Nanofluid Prepared in 50:50 EG/Water by Modified Strategy" ChemEngineering 7, no. 6: 117. https://doi.org/10.3390/chemengineering7060117
APA StyleYadav, P., Gupta, S. M., & Sharma, S. K. (2023). Evaluation of Stabilization and Physical–Chemical Properties of CNT Antifreeze Nanofluid Prepared in 50:50 EG/Water by Modified Strategy. ChemEngineering, 7(6), 117. https://doi.org/10.3390/chemengineering7060117





