1. Introduction
The most critical properties characterizing the performance of cement are early and typical compressive strength and setting time. Calcium sulfate addition affects these characteristics by simultaneously acting as a modifier and a regulator of their values [
1]. The cement must contain clinker and calcium sulfate, while the standards permit the usage of other components, like limestone, pozzolans, fly ash, and slag. Gypsum is the primary source of sulfates (SO
3) composed mainly of calcium sulfate dihydrate (CASO
4.2H
2O or Cs.2H) and a small content of anhydrite (CaSO
4 or Cs). During cement grinding, elevated temperatures partially dehydrate gypsum into hemihydrate form or bassanite (CaSO
4.0.5H
2O or Cs.0.5H) [
2]. Copeland et al. [
3] and a recent systematic review [
4] clarify the solubility of the three phases of CaSO
4.
Clinker is mainly composed of four mineral phases: tricalcium silicate (3CaO ∙ SiO
2 or C
3S), dicalcium silicate (2CaO ∙ SiO
2 or C
2S), tricalcium aluminate (3CaO ∙ Al
2O
3 or C
3A), and tetra calcium aluminoferrite (4CaO ∙ Al
2O
3 ∙ Fe
2O
3 or C
4AF). Bogue established the mathematical formulae for calculating the four clinker compounds as a function of the percentages of the four basic oxides CaO, SiO
2, Al
2O
3, Fe
2O
3, or C, S, A, F, as well as the free lime CaO
f [
5] (pp. 245–250).
The reactions of these four mineral components with water (H) give rise to various compounds, in which the dominant phases are calcium silicate hydrates (C-S-H) and calcium hydroxide (CH). Equations (5) and (6) express generalized forms of the hydration reactions of C
3S and C
2S, including some small decimal numbers m and n [
1].
Gypsum addition also obstructs the fast exothermic reaction of C
3A by forming ettringite (C
3A.3Cs.32H), according to Equation (7).
Ettringite formation regulates uncontrolled C
3A hydration, preventing flash set during concrete production, transfer, and placement [
6,
7,
8]. Conversely, adding too much gypsum leads to a drop in strength and detrimental expansion of concrete and mortar, meaning there is a sulfate optimum. Cement standards, such as EN 197-1:2011 [
9] and ASTM C150 [
10], prohibit sulfates above a certain level per type of cement, though they do not suggest an optimal value because that figure depends on the various physical, chemical, and mechanical properties of cement and clinker produced in the specific conditions of each production unit. Furthermore, the property to be optimized impacts the location of the optimum. Some standards, such as ASTM C563-16 [
11], define a method for determining the optimal SO
3, albeit only for a particular property of cement, i.e., 1-day compressive strength.
Historically, determining the optimal sulfates in cement was subjected to intensive and continuous research. In recent years, a primary goal of the cement industry was the reduction in its carbon footprint by decreasing CO
2 emissions in clinker production and clinker consumption per ton of product. Optimizing the composition of raw materials for clinker production can achieve the above goal [
12,
13]. Optimizing SO
3 is essential for reducing clinker incorporation into the cement while retaining or improving product performance. Lerch [
14] conducted the first thorough study of sulfate optimization in cement past and mortar. According to the researcher, the optimal sulfate content in cement is closely related to hydration heat, length changes of mortar specimens cured in water, alkalis, C
3A content, and cement fineness. Fincan [
15] mentions that Lerch was a pioneer and inspired many studies on sulfate optimization in cement and cementitious systems.
Several researchers investigated the impact of SO
3 on the hydration of the clinker mineral phases [
16,
17,
18,
19,
20,
21,
22,
23,
24,
25]. Bentur [
16] found that gypsum influenced the quantity and quality of the hydrated products by accelerating the hydration process and, at the same time, lowering the intrinsic strength of the gel. He interpreted the effect of gypsum on strength in terms of its influence on the extent of hydration and the chemical composition of the gel. Soroka et al. [
18] concluded that gypsum accelerates the rate of hydration when its addition is below the optimum SO
3 content. Nevertheless, if the gypsum added exceeds the optimal level, considerable obstruction occurs. The authors did not observe any effect of the SO
3 content on the density of the hydration products and pore-size distribution. They pointed out that further study is required to explain the impact of SO
3 on strength. On the contrary, Sersale et al. [
19] found that gypsum addition modified the microstructure of Portland cement mortars. They correlated these modifications with the mechanical behavior of cement mortars by examining Portland cement with sulfate content ranging between 1.5 and 4.5%. A SO
3 content of 2–3.5% promotes a shifting in the pore size distribution to lower values, ranging between 100 and 1000 Å, and a variation in total porosity. They concluded that this issue is likely the main factor governing the influence of SO
3 on the compressive strength of Portland cement. Gunay [
22], in his thesis, studied the influence of aluminates hydration in the presence of calcium sulfate on C
3S hydration and its consequences on cement optimum sulfate. He observed the SO
3 optimum when the hydration of C
3S, during the accelerated period, takes place simultaneously or slightly before the exothermic peak due to the dissolution of C
3A and the precipitation of mono-substituted Al
2O
3 and Fe
2O
3 (AFm). He concluded that the presence of AFm during the accelerated period of C
3S hydration would be the cause of the observed modification of the microstructure of the cement paste: the porosity increases with the addition of calcium sulfate, though the assembly of hydrates is denser. This effect of the sulfate level is the source of the optimal compressive strength observed by Gunay. Zunino et al. [
24] investigated the influence of sulfate addition on hydration kinetics and the C-S-H morphology of C
3S and C
3S/C
3A systems at an early age. Adding gypsum changed the needle length of C-S-H and increased the nucleation density. In C
3S/C
3A systems, they did not observe any difference in C-S-H morphology before and after the aluminate peak. Andrade Neto et al. [
25] studied the hydration and interactions between pure and doped C
3S and C
3A in the presence of different calcium sulfates. Except for sulfates of gypsum, Miller et al. [
26], Taylor [
27], and Horkoss et al. [
28] investigated the importance of the amount and phases of SO
3 incorporated into the clinker. Mohammed et al. [
29] optimized the SO
3 content of a CEM I cement via grinding clinker with a ball mill until a fineness of 3270 cm
2/g was achieved, before preparing mixes with ground gypsum ranging from 0 to 9%. According to their results, the water demanded for normal consistency, setting times, compressive strength, the heat of hydration, swelling, drying shrinkage, and hydration degree are adversely affected by gypsum addition above or below the optimal. For this cement, which was composed only of clinker and gypsum, they found that the optimal sulfate for 2-, 7-, and 28-day strength was 3%.
Nowadays, with the reduction in CO
2 emissions being imperative, composite types of cement constitute the bulk of the products of the cement industry, rendering the optimization of sulfates in such cement more relevant. During the last few decades, numerous researchers conducted deep research investigating the optimal sulfate content of cement containing supplementary cementitious materials (SCM) [
30,
31,
32,
33,
34,
35,
36]. Yamashita et al. [
30] investigated the influence of limestone powder (LSP) on the optimum SO
3 for Portland cement samples with different Al
2O
3 contents, which was not negligible. Analyses showed that if SO
3 is less than optimal, an increase in sulfate promotes hydration in C
3A and increases compressive strength. In the presence of higher SO
3 content, excess formation of expansive ettringite introduced more pores, and compressive strength decreased. After adding LSP, a lower sulfate content was adequate to obtain the maximum compressive strength. Liu et al. [
32] examined the effect of gypsum content on cementitious mixtures containing limestone, fly ash, and slag by studying several properties: initial and final setting time, past fluidity, water demand, and strength. Adu-Amankwah et al. [
33] conducted detailed research into the consequence of sulfate additions on hydration and the performance of ternary slag–limestone composite cement using complementary techniques. Their results showed that the presence of sulfate influenced the early-age reaction kinetics of the clinker phases and supplementary cementitious materials. These changes impacted the total porosity and cement strength in opposing ways: porosity was reduced with increasing ettringite fraction, while the lower water content of the C-S-H reduced the space-filling capacity of the C-S-H. Han et al. [
34] investigated the effect of gypsum on the properties of composite binders containing high-volume slag and iron tailing powder using multiple measuring techniques. They concluded that although incorporating gypsum promoted early hydration of cement and slag, it limited their further hydration at later ages. Added gypsum formed a large amount of ettringite and densified early-age pore structure, though it coarsened later-age pore structure. Fiscan [
15] optimized sulfate in cement–slag blends based on calorimetry and early strength results, investigating the influence of fineness, C
3A, C
4AF of cement, and the Al
2O
3 content of slag on the optimum SO
3. Niemuth [
7] examined the effect of fly ash on the optimum sulfate content in Portland cement, providing experimental data on strength development and heat release during early hydration for cement–fly ash systems with different SO
3 levels. He demonstrated that some fly ash samples achieve their sulfate demand. When a cement contains these samples, there is effectively an increase in the optimal SO
3 level compared to the corresponding CEM I Portland cement. In his research into the optimal SO
3 content of Portland and pozzolanic cement types, Tsamatsoulis et al. [
35] reached the same conclusion.
Increasingly, cement manufacturers use grinding aids/strength improvers to reduce energy consumption and improve cement quality. According to Magistri et al. [
37], hydration in the presence of such additives proceeds differently than in cement produced without them, remarkably improving cement properties. Hirsch et al. [
38] examined the impact of triethanolamine (TEA) on the sulfate balance of Portland cement with mixed sulfate carriers (Cs.2H, Cs.0.5H, CS). Their results indicated that TEA influenced the balance of sulfate and aluminate-containing clinker phases. These effects were closely related to the types and amounts of the sulfate carriers present in the cement. The very practical, from a process point of view, research of Recchi et al. [
39] investigates the influences of the type (Cs.2H, Cs.0.5H, Cs) and amount of calcium sulfate on the reactivity of TEA and tri-isopropanolamine (TIPA) strength improvers, which are today’s most widely used alkanolamines. TEA is an early compressive strength enhancer, while TIPA increases the late strengths. The authors concluded that if the sulfate carrier is bassanite, the early strength increase obtained with the addition of TEA is more evident due to the higher solubility of Cs.0.5H. On the other hand, TIPA is significantly less efficient at increasing late strength when bassanite is the source of SO
3, i.e., Cs.0.5H is not preferable when the objective of ΤIPA use is to enhance 28-day strength. Andrade Neto et al. [
40] recently conducted a detailed review of the effect of SO
3 on cement hydration, noticing that despite many years of research, questions regarding sulfate optimization remain. Further investigation into the influence of clinker and CaSO
4 characteristics using different supplementary cementitious materials and chemical admixtures is needed.
Some researchers provided or applied equations correlating optimal SO
3 with the cement’s chemical and physical characteristics. For achieving maximum 28-day strength with C
3A, K
2O, Na
2O, and fineness of cement, Schade et al. [
41] presented three equations (Haskell, Jawed and Skalny, and Ost). They utilized the third equation to perform a design of experiments (DoE) to model the sulfate amounts in ultra-finely ground and fast-hardening clinker. Kurdowski [
42] reported four equations for approximating the optimal SO
3 content (Ost, Lerch, Jirku, and Haskell), before noticing that these empirical equations have limited accuracy, even if they include the main factors affecting the optimum gypsum addition. He considered that using experimentation to determine the most convenient sulfate addition is the best approach. Andrade Neto et al. [
40] summarized the laboratory methods for determining the correct SO
3 content in conjunction with their advantages and disadvantages. From an industrial perspective, strength measurement is the most utilized method, since compressive strength is a key performance criterion for producers and customers. ASTM C563-16 [
11] describes the determination of approximate optimum SO
3 for maximum compressive strength by measuring the change in this property of cement mortar as a result of substituting calcium sulfate for a portion of the cement. The standard suggests a parabolic equation between strength and sulfate that optimally fits the experimental points when assuming a symmetric distribution. The optimal SO
3 approximation corresponds to the value providing the vertex of the parabola. The standard clearly states that in cases of a skewed function of the strength versus sulfate to the right or left of the peak, an asymmetric distribution function may provide a better fit. Niemuth [
7], Tsamatsoulis [
35], and Fincan [
15] applied the parabolic formula in their attempts to find an adequate approximation of SO
3 optimal content. Tsamatsoulis [
35], when trying to determine a unified function for several cement types with variable clinker content, used the ratio of sulfate amount to clinker percent (%SO
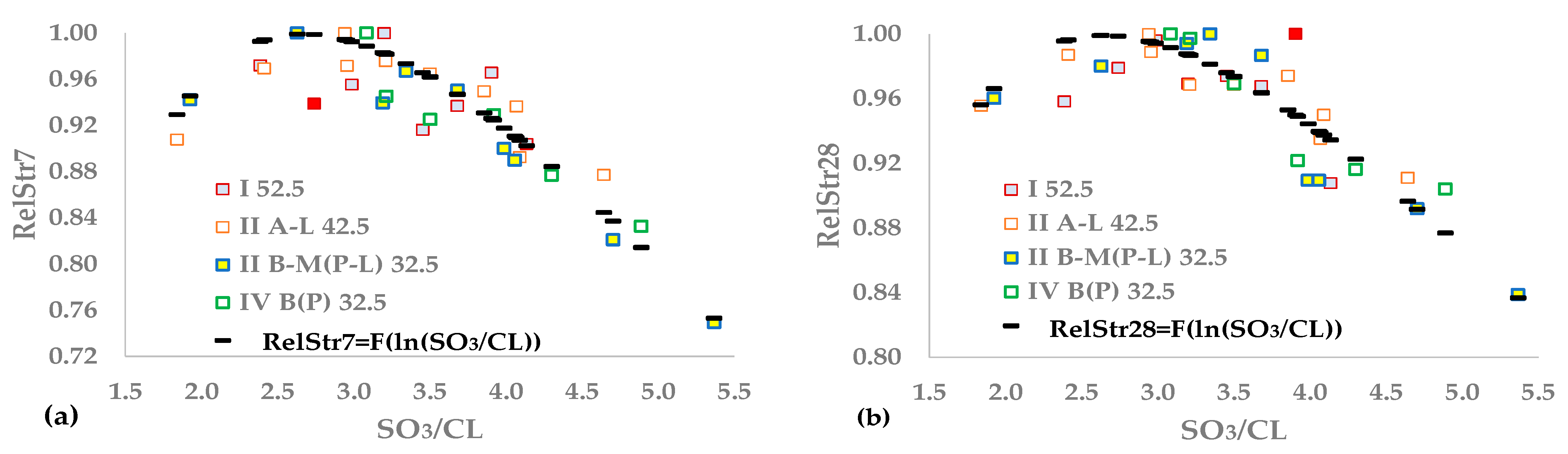
3/CL) as an independent variable.
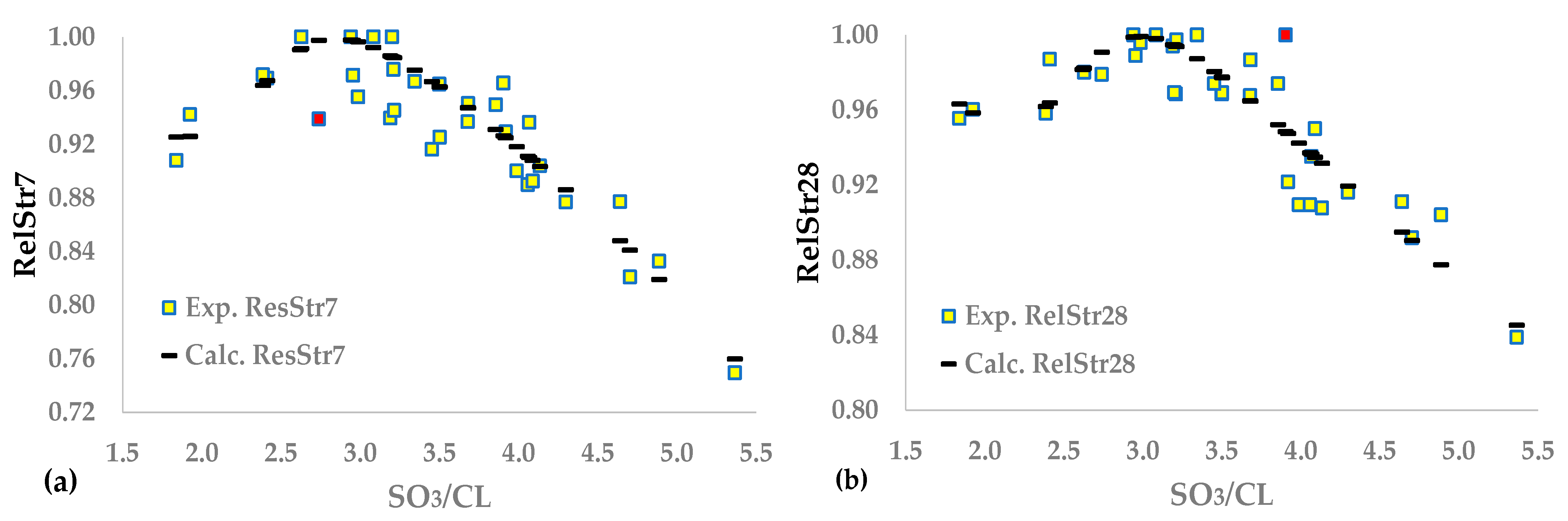
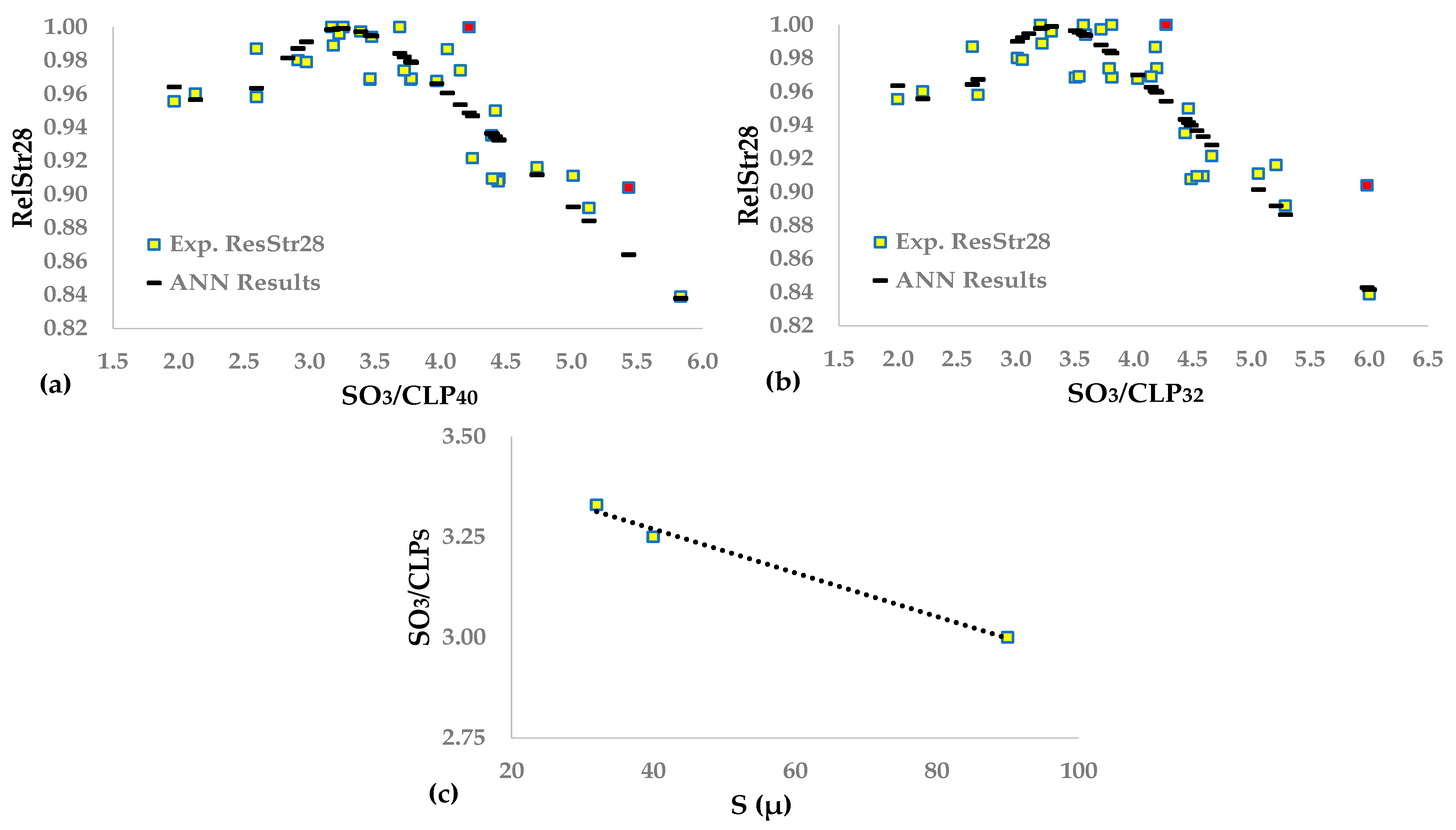
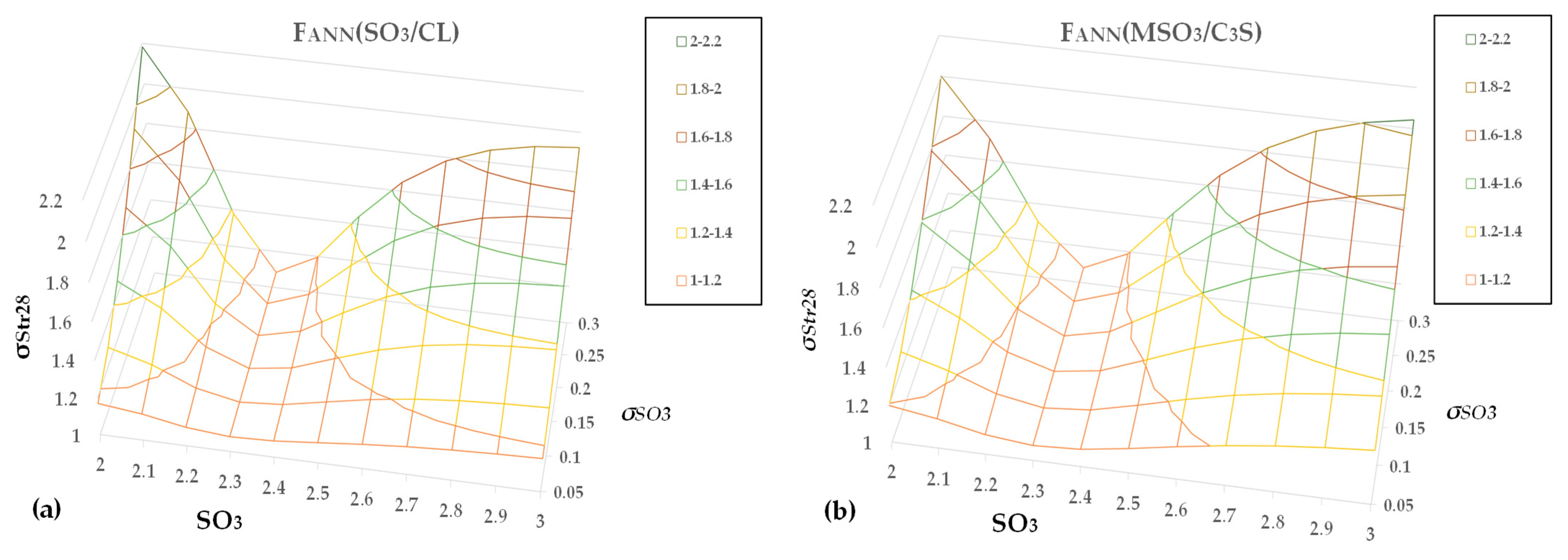
This study aims to approximate the optimal SO
3 content of cement using the maximization of compressive strength as a criterion for cement produced in industrial mills. The experimental design includes tests on four types of cement containing up to three main components, except gypsum, and belonging to three strength classes. We developed several relationships correlating the 7- and 28-day strengths of the sulfate and clinker content of the cement and the clinker mineral composition. We normalized the results to obtain unique functions for all experimental data, using an approach similar to the method presented in [
35]. This study proved that a shallow artificial neural network [
43,
44] fits the data distribution better than a parabolic function. Finally, we focused on the impact of SO
3 uncertainty on the 28-day strength variance using the error propagation method. The structure of the paper is as follows:
Section 2 includes the sampling procedure, experimental methods, and test results;
Section 3 describes the implemented algorithms for data processing, including a detailed discussion of each set of results; and, finally,
Section 4 summarizes the main conclusions of this research.
2. Materials and Methods
In this study, we chose to optimize the sulfate amount of the basic cement types produced according to EN 197: 2011 [
9] in the Halyps cement plant, using the compressive strength criterion as a guide.
Table 1 shows the CEM types investigated and the norm requirements as regards composition and strength limits. The nominal composition did not contain the gypsum. The producer was responsible for adjusting its content by respecting the maximum SO
3 limit, which was 4% for CEM I 52.5 N and 3.5% for the other three CEM types. We observed that the research covered a wide range of Portland (CEM I, CEM II) and pozzolanic (CEM IV) types. The study was, therefore, general for the cement products of this specific cement plant. The optimal value of cement sulfate depended on various factors, which were summarized by Andrade Neto et al. [
40] as follows: (a) clinker mineralogy (C
3S, C
3A) and alkali content, as well as cement and clinker fineness; (b) form of SO
3 carriers (Cs.2H, Cs.0.5H, Cs or alkali sulfates), as well as mineral or chemical gypsum; (c) intergrinding or separate grinding of clinker and gypsum; (d) content and type of SCM; (e) grinding aid/strength improver type; (f) hydration age; (g) water/binder ratio; and (h) curing conditions. Using the standard EN 196-1 [
45] to create mortars, we attempted to find the SO
3 contents for maximum 7- and 28-day strength and examined the optimum position as a function of hydration age, fulfilling the conditions (f)–(h).
The goal of the experimentation was to obtain cement samples as close as possible to industrially produced cement in terms of fineness, chemical characteristics, forms of the sulfate carriers, and interaction of the grinding aid with the solids. Grinding in the Halyps cement plant was performed through co-grinding the raw materials in closed-circuit mills equipped with high-performance separators and co-feeding a 28-day strength enhancer into the mill feed. In an earlier study [
46], we found that for a step change in gypsum, the mill circuit required around 1 h to reach the steady state regarding SO
3. For each CEM type, by operating the mill in automatic mode and after stabilizing the circuit around the operating set points and the desired fineness, we suddenly decreased the gypsum to 2%. There was no change in the percentage of clinker, while another material increased in proportion (limestone or pozzolan). Sampling of around 20 kg of the final product followed after 1.5 h. The second step was an increase in gypsum to 7.5% and a proportional decrease in limestone or pozzolan to maintain the %clinker constant. Further sampling took place 1.5 h after this change. During this process, with an operation kept as constant as possible, the laboratory sampled clinker at the mill inlet. In this way, the lab created two samples of industrial cement with low and high gypsum for each CEM type. Mixing them in proper proportions yielded samples with suitable SO
3 values usable to correlate strength and sulfate. The industrial tests of the four CEM types were realized within two months to allow the clinker composition to incorporate the actual production variances.
The samples’ analysis comprised several mechanical, physical, and chemical tests: (i) fineness with air-jet sieving and specific surface with Blaine apparatus, using EN 196-6 [
47]; (ii) loss on ignition (LOI), insoluble residue (InsRes), and oxides analysis via XRF by applying EN 196-2 [
48]; (iii) laser particle size analysis; (iv) compressive strength measurement using EN 196-1; and (v) clinker and cement phases using XRD. Halyps lab conducted the analyses (i)–(iv), while the Devnya Cement lab performed the XRD analysis.
2.1. Clinker and Raw Materials Analysis
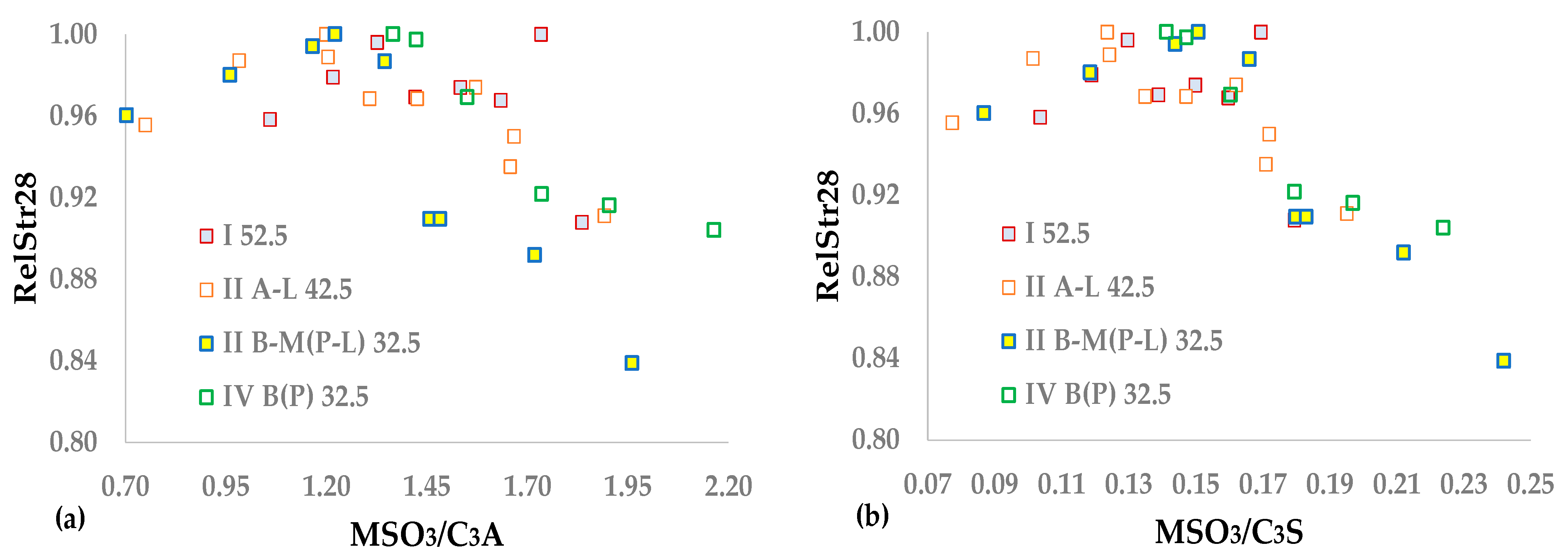
Aside from the experiment illustration, clinker, gypsum, limestone, and pozzolan analyses used during the test are necessary for estimating the CEM composition.
Table 2 demonstrates the XRF and XRD analyses of the four clinkers. The mean values of the loss on ignition and insoluble residue were equal to 0.3. Equation (8) presents the molecular ratio between SO
3 and alkalies, i.e., MSO
3.
Table 2 presents the mean values and standard deviations of SO
3, C
3S, and MSO
3. The corresponding annual values for 2022 of the Halyps clinker were as follows: SO
3 = 0.86 ± 0.25, C
3S = 64.7 ± 2.5, and MSO
3 = 0.88 ± 0.24; these results mean that the variance in the four clinkers’ properties covers a significant amount of the actual variability in the plant clinker in terms of quality. The mineral phases measured via XRD differ from those computed with Bogue formulae, though the C
3S values fit relatively well. XRD did not detect anhydrite and langbeinite because the MSO
3 < 1.
Table 3 presents the average analysis of the three raw materials. Assuming that CaO, MgO, SO
3, and LOI exist in gypsum in the form of calcium and magnesium carbonates, gypsum dihydrate, and anhydrite, we computed these four components by solving the corresponding linear system. The results show that gypsum is mainly dihydrate, containing a small percentage of anhydrite.
2.2. Cement Analyses
The laboratories conducted chemical, physical, and mechanical tests on the cement samples to reliably correlate the variables and minimize uncertainties.
Table 4 provides both the chemical analysis of the eight samples taken for the CEM types under investigation and the gypsum phases within the cement measured via XRD. We also took advantage of the automatic recording of process data in the plant database to find the cement temperature at the mill outlet for each of the eight tests.
Table 4 shows the mean value of this temperature (T
CEM) for each of the eight tests. Although the detection of gypsum phases via XRD could be approximated, the main conclusion was that hemihydrate was null or negligible. This issue arose because the cement temperature at the mill outlet never reached 100 °C, as dihydrate starts to dehydrate to Cs.0.5H [
49]. Krause et al. [
50] experimentally found that the transformation of gypsum into hemihydrate could take place at a temperature of 50 °C. However, our industrial results do not verify these findings.
Table 5 shows the fineness measurements of these samples expressed as %residues in 40, 32, and 20 microns (R40, R32, R20) and obtained via air-jet sieving and Blaine. All samples had 100% passing in the 90 microns sieve (R90 = 0). The same table demonstrates several fineness modules obtained via laser particle size analysis: diameters of 50%, 90%, and 10% passing (D50, D90, D10), as well as percentages between 32 and 3 microns (P32–R3).
The solution of a linear system, as given in Equations (9)–(12) and expressing the mass balances, provided the dry cement composition of all samples using chemical analysis of cement, clinker, and raw materials. This estimation avoided the uncertainties of the mill feeders where the raw materials had a certain percentage of moisture.
CL,
G,
Lim, and
Pz are the clinker, gypsum, limestone, and pozzolan percentages With
SO3 X,
LOIX, and
InsResX, we denoted the sulfate, loss on ignition, and insoluble residue of the material
X (
X =
CL,
G,
Lim,
Pz,
CEM). Solving the system of (9)–(11) produced the composition of CEM I 52.5 N and CEM II A-L 42.5 N. The calculation of the
Pz requires the solution of the system (9)–(12) for CEM II B-M(P-L) 32.5 N. For the CEM IV(P) 32.5 N with high pozzolan content, our algorithm used three additional equations that expressed the mass balances of SiO
2, Al
2O
3, and CaO. The composition results from the error minimization between the actual and calculated chemical analysis of cement were realized using the Generalized Reduced Gradient non-linear regression method. The lab conducted a chemical analysis, except for the eight samples, of the residues at 40, 32, and 20 microns of each of them. By determining the composition of the total sample and the material retained in each sieve, we calculated the %constituents of the material that passes through that sieve. The above approach made it feasible to calculate the composition in each fraction and investigate the grindability of the materials during co-grinding.
Table 6 depicts the %components of the samples and passings through the three mentioned sieves and the residues of each material in the three sieves. Equations (13) and (14) provide these values.
The j indexes from 1 to 3 correspond to the sieves at 40, 32, and 20 microns, while the k indexes 1 to 4 correspond to the four materials (clinker, gypsum, limestone, pozzolan). Rj is the residue of a sample at the sieve j, and RMk,j is the residue of material k at the sieve j. CompSamplek is the percentage of the component k within the sample, while CompResk,j and CompPassk,j are the percentages of the component k in the residue and passing in the sieve j.
These results indicate the higher grindability of gypsum and limestone compared to the other compounds. We observed that clinker and pozzolan had similar grindability. Therefore, approximating the optimal SO
3 content using industrially produced cement in a closed-circuit mill was crucial. It was difficult to obtain the same particle size per constituent and the distribution of the grinding aid via grinding in an open circuit lab apparatus or interblending the previously ground materials. Tang et al. [
51] concluded that when testing to determine the optimum sulfate content, it was significant to co-grind the calcium sulfate with clinker because co-grinding resulted in lower sulfate demand than interblending. Thus, the results obtained via the latter method would not be representative.
2.3. Cement Samples Mixing and Related Tests
The lab mixed the cement samples of low and high gypsum to create new samples at regular and proper intervals of SO
3 values. Complete chemical analysis of these samples, composition calculation—as described in
Section 2.2—mortars preparation, and measuring the 7- and 28-day compressive strengths followed.
Table 7 depicts the proportions of low and high gypsum samples, the SO
3 and %Clinker values, the %ratio of sulfates to clinker (%SO
3/CL), and the 7- and 28-day compressive strengths of each mix for the four CEM types investigated. SO
3 values stem from XRF measurement, while %Clinker results stem from composition calculation.
In
Appendix A, we provide a list of raw materials suppliers and the manufacturers of the main equipment used.