Preparation of Layered Double Hydroxides toward Precisely Designed Hierarchical Organization
Abstract
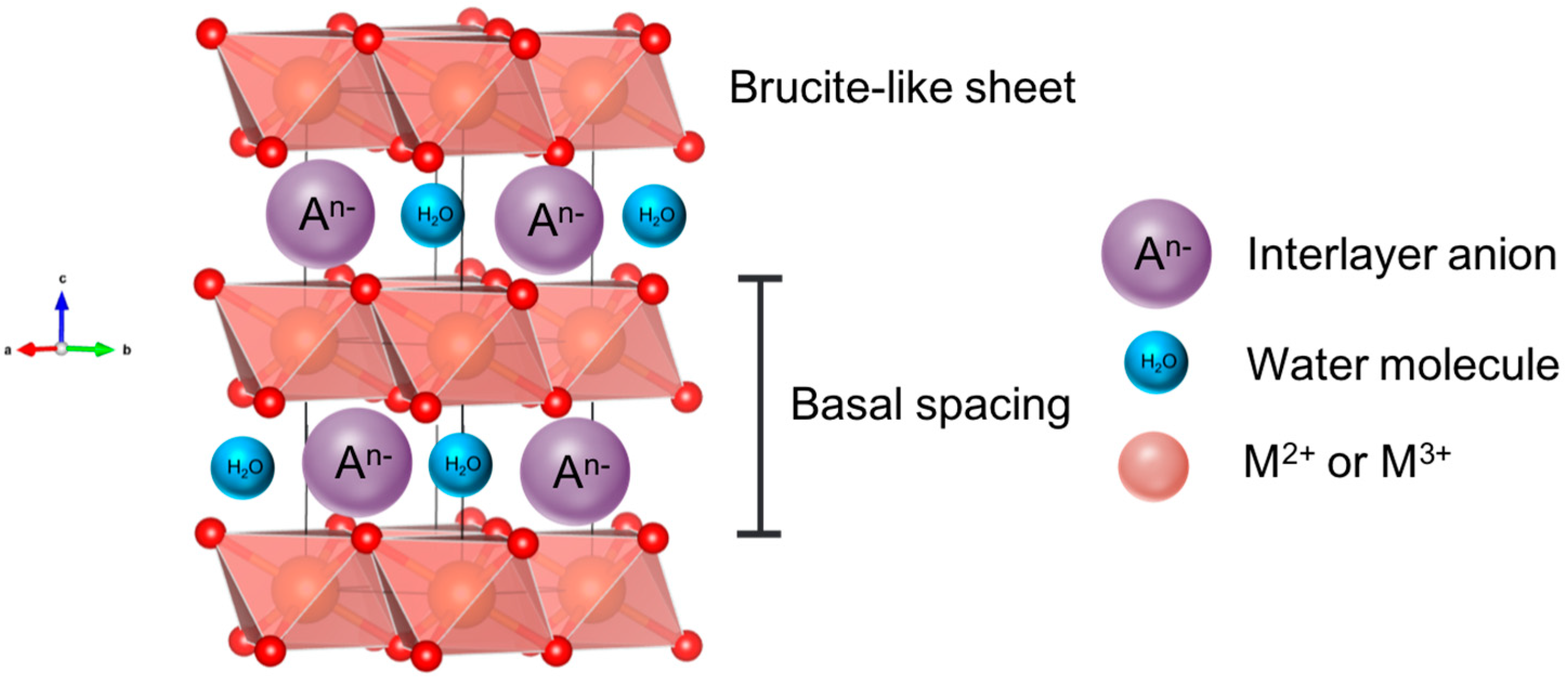
:1. Introduction
2. Co-Precipitation from Homogeneous Solution
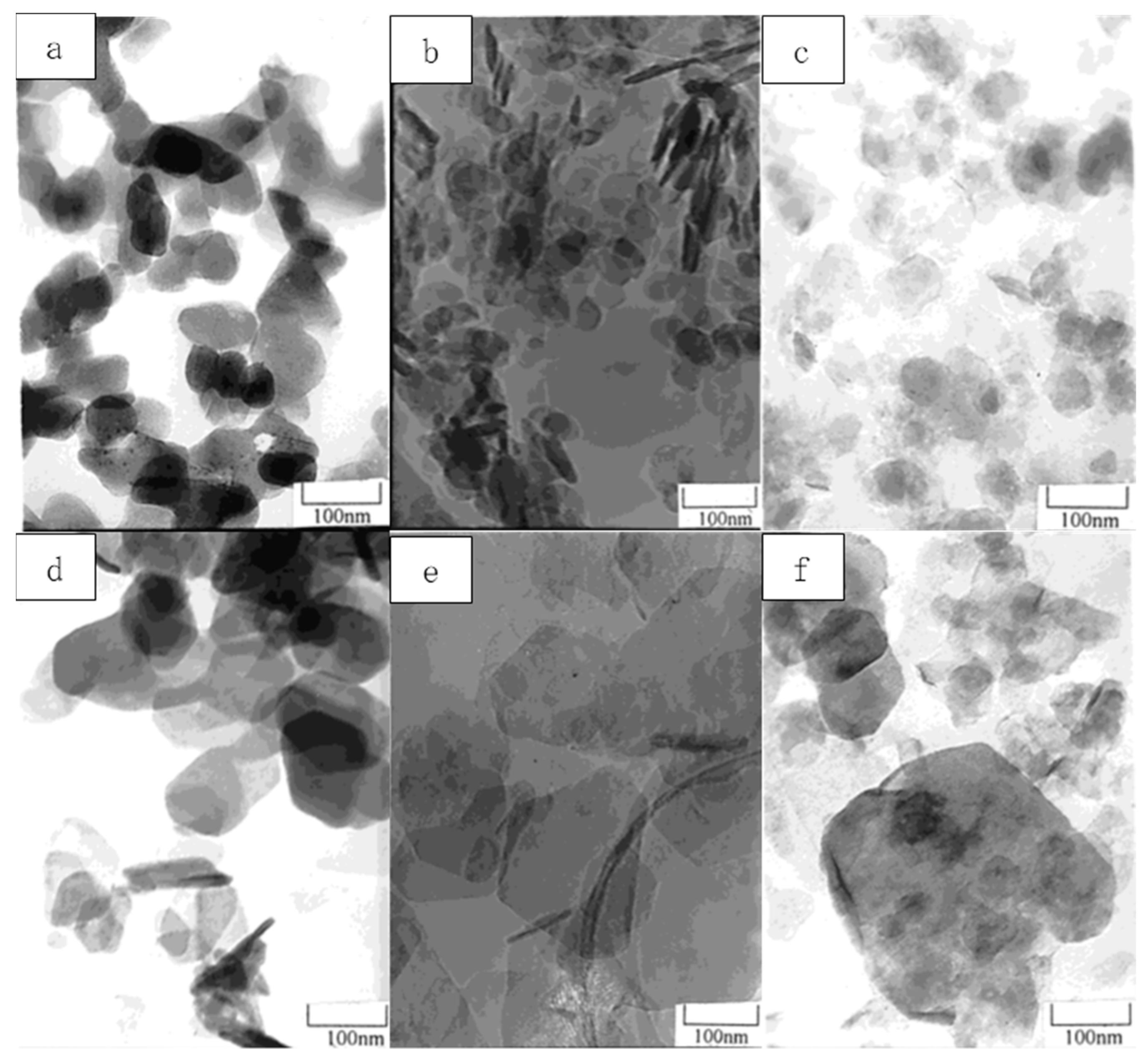
2.1. Co-Precipitation by the Addition of Base
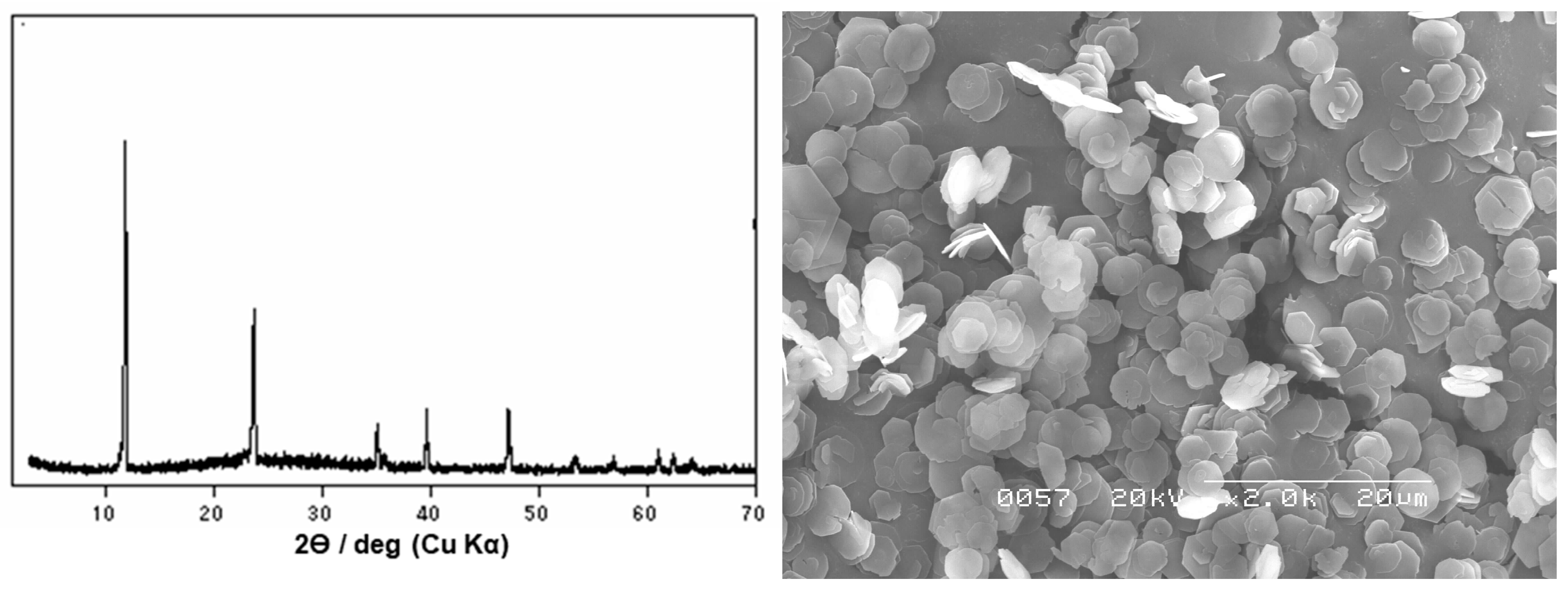
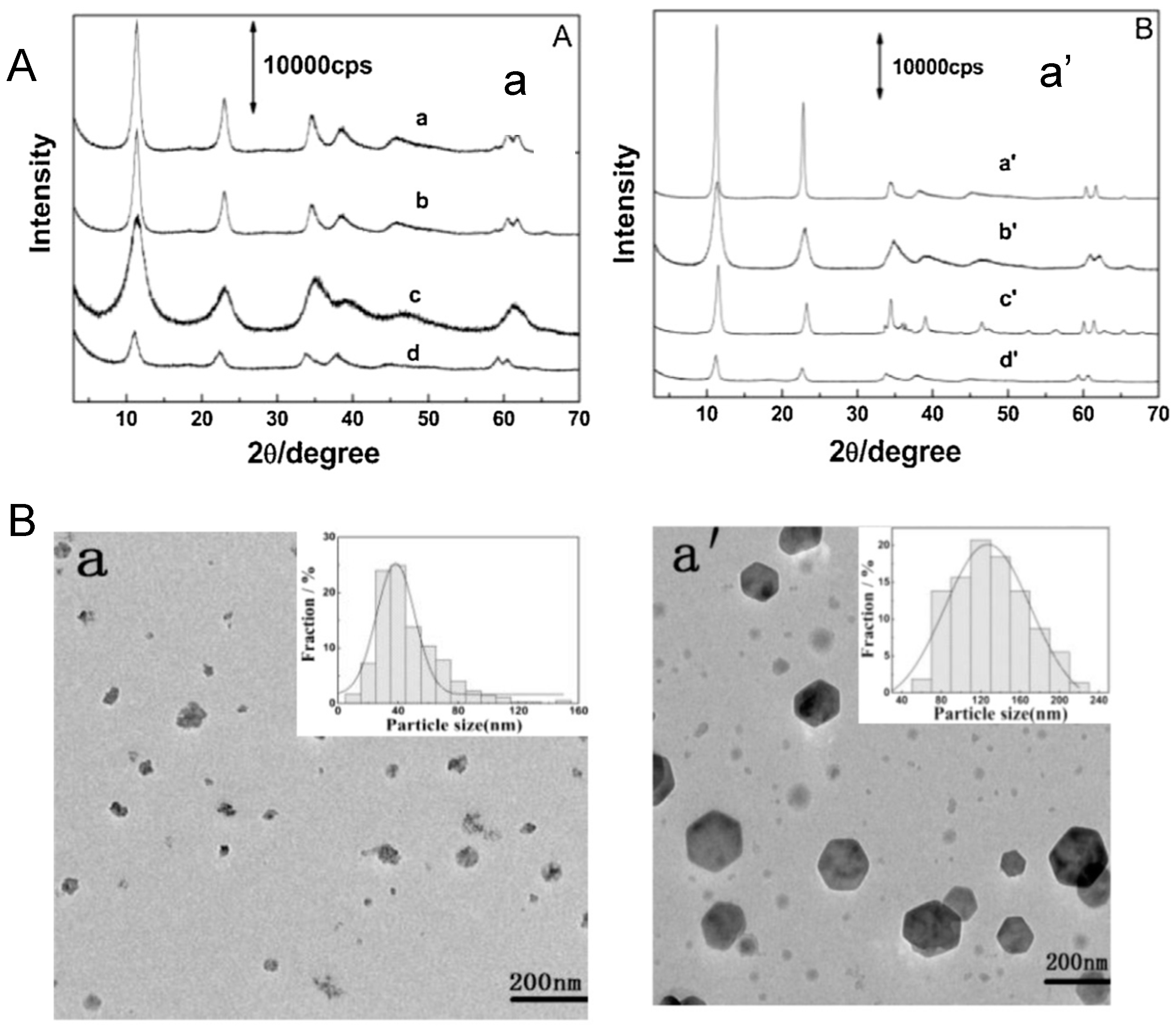
2.2. Homogeneous Precipitation
2.3. LDH Precipitation from the Solution Containing Additives
3. Preparation from Slurry of Metal Oxides/Hydroxides
4. Mechanochemical Syntheses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- He, J.; Wei, M.; Li, B.; Kang, Y.; Evans, D.G.; Duan, X. Preparation of layered double hydroxides. Struct. Bond. 2006, 119, 89–119. [Google Scholar]
- Evans, D.G.; Slade, R.C.T. Structural aspects of layered double hydroxides. Struct. Bond. 2006, 119, 1–87. [Google Scholar]
- Rives, V. Layered Double Hydroxides: Present and Future; Nova Science Publishers Inc.: New York, NY, USA, 2001. [Google Scholar]
- Cavani, F.; Trifiró, F.; Vaccari, A. Hydrotalcite-type anionic clays: Preparation, properties and applications. Catal. Today 1991, 11, 173–301. [Google Scholar] [CrossRef]
- Newman, S.P.; Jones, W. Synthesis, characterization and applications of layered double hydroxides containing organic guests. New J. Chem. 1998, 22, 105–115. [Google Scholar] [CrossRef]
- Kuang, Y.; Zhao, L.; Zhang, S.; Zhang, F.; Dong, M.; Xu, S. Morphologies, preparations and applications of layered double hydroxide micro-/nanostructures. Materials 2010, 3, 5220–5235. [Google Scholar] [CrossRef]
- Nishimura, S.; Takagaki, A.; Ebitani, K. Characterization, synthesis and catalysis of hydrotalcite-related materials for highly efficient materials transformations. Green Chem. 2013, 15, 2026–2042. [Google Scholar] [CrossRef]
- Theiss, F.L.; Ayoko, G.A.; Frost, R.L. Synthesis of layered double hydroxides containing Mg2+, Zn2+, Ca2+ and Al3+ layer cations by co-precipitation methods—A review. Appl. Surf. Sci. 2016, 383, 200–213. [Google Scholar] [CrossRef]
- Conterosito, E.; Gianotti, V.; Palin, L.; Boccaleri, E.; Viterbo, D.; Milanesio, M. Facile preparation methods of hydrotalcite layered materials and their structural characterization by combined techniques. Inorg. Chim. Acta. 2018, 470, 36–50. [Google Scholar] [CrossRef]
- Sels, B.; Vos, D.D.; Buntinx, M.; Pierard, F.; Mesmaeker, A.K.; Jacobs, P. Layered double hydroxides exchanged with tungstate as biomimetic catalysts for mild oxidative bromination. Nature 1999, 400, 855–857. [Google Scholar] [CrossRef]
- Choundary, B.M.; Madhi, S.N.; Chowdari, M.; Kantam, M.L.; Sreedhar, B. Layered double hydroxide supported nanopalladium catalyst for Heck-, Suzuki-, Sonogashira-, and Stille-type coupling reactions of chloroarenes. J. Am. Chem. Soc. 2002, 124, 14127–14136. [Google Scholar] [CrossRef]
- Prevot, V.; Casal, B.; Ruiz-Hitzky, E. Intracrystalline alkylation of benzoate ions into layered double hydroxides. J. Mater. Chem. 2001, 11, 554–560. [Google Scholar] [CrossRef]
- Yu, J.J.; Wang, X.P.; Li, L.D.; Hao, Z.P.; Xu, Z.P.; Lu, G.Q. Novel multi-functional mixed-oxide catalysts for effective NOx capture, decomposition, and reduction. Adv. Func. Mater. 2007, 17, 3598–3606. [Google Scholar] [CrossRef]
- Kaneda, K.; Yamashita, T.; Matsushita, T.; Ebitani, K. Heterogeneous oxidation of allylic and benzylic alcohols catalyzed by Ru-Al-Mg hydrotalcites in the presence of molecular oxygen. J. Org. Chem. 1998, 63, 1750–1751. [Google Scholar] [CrossRef]
- Costantino, U.; Leroux, F.; Nocchetti, M.; Mousty, C. LDH in physical, chemical, biochemical, and life sciences. Devel. Clay Sci. 2013, 5, 765–791. [Google Scholar]
- Fan, G.; Li, F.; Evans, D.G.; Duan, X. Catalytic applications of layered double hydroxides: Recent advances and perspectives. Chem. Soc. Rev. 2014, 43, 7040–7066. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Cheng, J.P.; Fang, J.H.; Yang, Y.; Liu, F.; Zhang, X.B. NiAl-layered double hydroxide/reduced graphene oxide composite: Microwave-assisted synthesis and supercapacitive properties. Electrochim. Acta 2014, 134, 309–318. [Google Scholar] [CrossRef]
- Li, F.; Duan, X. Applications of layered double hydroxides. Struct. Bond. 2006, 119, 193–223. [Google Scholar]
- Hoyo, D.C. Layered double hydroxides and human health: An overview. Appl. Clay Sci. 2007, 36, 103–121. [Google Scholar]
- Zhao, M.; Zhao, Q.; Li, B.; Xue, H.; Pang, H.; Chen, C. Recent progress in layered double hydroxide based materials for electrochemical capacitors: Design, synthesis and performance. Nanoscale 2017, 9, 15206–15225. [Google Scholar] [CrossRef]
- Inomata, K.; Ogawa, M. Preparation and properties of Mg/Al layered double hydroxide oleate and stearate intercalation compounds. Bull. Chem. Soc. Jpn. 2006, 79, 336–342. [Google Scholar] [CrossRef]
- Goodman-Gilman, A.; Goodman, L.S.; Gilman, A. The Pharmacological Basis of Therapeutics, VI; MacMillan Publishing Co, Inc.: New York, NY, USA, 1980; p. 995. [Google Scholar]
- Hashimoto, Y.; Shiozawa, H.; Kishimoto, H.; Setoguchi, Y. Drug Composition: PCT—The International Patent System. Patent Number 95,307,512, 1997. [Google Scholar]
- Tomohisa, M.; Mitsuo, H. Enteric Granule-Containing Tablets. U.S. Patent No. 5,798,120, 25 August 1998. [Google Scholar]
- Woo, J.S.; Yi, H.G. Antifungal Oral Composition Containing Itraconazole and Process for Preparing Same. U.S. Patent No. 6,039,981, 21 March 2000. [Google Scholar]
- Ambrogi, V.; Fardella, G.; Grandolini, G.; Perioli, L. Intercalation compounds of hydrotalcite-like anionic clays with antiinflammatory agents—I. Intercalation and in vitro release of ibuprofen. Int. J. Pharm. 2001, 220, 23–32. [Google Scholar] [CrossRef]
- Khan, A.I.; Lei, L.; Norquist, A.J.; O’Hare, D. Intercalation and controlled release of pharmaceutically active compounds from a layered double hydroxide. Chem. Commun. 2001, 22, 2342–2343. [Google Scholar] [CrossRef]
- Choy, J.H.; Oh, J.M.; Park, M.; Sohn, K.M.; Kim, J.W. Inorganic–biomolecular hybrid nanomaterials as a genetic molecular code system. Adv. Mater. 2004, 16, 1181–1184. [Google Scholar] [CrossRef]
- Oh, J.M.; Park, M.; Kim, S.T.; Jung, J.Y.; Kang, Y.G.; Chong, J.H. Efficient delivery of anticancer drug MTX through MTX- LDH nanohybrid system. J. Phys. Chem. Solids 2006, 67, 1024–1027. [Google Scholar] [CrossRef]
- Rojas, R.; Palena, M.C.; Jimenez-Kairuz, A.F.; Manzo, R.H.; Giacomelli, C.E. Modeling drug release from a layered double hydroxide–ibuprofen complex. Appl. Clay Sci. 2012, 62–63, 15–20. [Google Scholar] [CrossRef]
- Choy, J.H.; Kwak, S.Y.; Park, S.J.; Jeong, Y.J.; Portier, J. Intercalative nanohybrids of nucleoside monophosphates and DNA in layered metal hydroxide. J. Am. Chem. Soc. 1999, 121, 1399–1400. [Google Scholar] [CrossRef]
- Choy, J.H.; Kwak, S.Y.; Park, S.J.; Jeong, Y.J.; Portier, J. Inorganic layered double hydroxides as nonviral vectors. Angew. Chem. Int. Ed. 2000, 39, 4042–4045. [Google Scholar] [CrossRef]
- Del Arco, M.; Cebadera, E.; Gutierrez, S.C.; Martin, M.; Montero, J.; Rives, V.; Rocha, J.; Sevilla, M.A. Mg,Al layered double hydroxides with intercalated indomethacin: Synthesis, characterization, and pharmacological study. J. Pharm. Sci. 2004, 93, 1649–1658. [Google Scholar] [CrossRef]
- Nakayama, H.; Wada, N.; Tsuhako, M. Intercalation of amino acids and peptides into Mg-Al layered double hydroxide by reconstruction method. Int. J. Pharm. 2004, 269, 469–478. [Google Scholar] [CrossRef]
- Aisawa, S.; Kudo, H.; Hoshi, T.; Takahashi, S.; Hirahara, H.; Umetsu, Y.; Narita, E. Intercalation behavior of amino acids into Zn-Al-layered double hydroxide by calcination-rehydration reaction. J. Solid State Chem. 2004, 177, 3987–3994. [Google Scholar] [CrossRef]
- Choi, S.J.; Oh, J.M.; Choy, J.H. Anticancer drug-layered hydroxide nanohybrids as potent cancer chemotherapy agents. J. Phys. Chem. Solids 2008, 69, 1528–1532. [Google Scholar] [CrossRef]
- Oh, J.M.; Choi, S.J.; Lee, G.E.; Kim, J.E.; Choy, J.H. Inorganic metal hydroxide nanoparticles for targeted cellular uptake through clathrin-mediated endocytosis. Chem. Asian J. 2009, 4, 67–73. [Google Scholar] [CrossRef]
- Moujahid, E.M.; Besse, J.P.; Leroux, F. Poly(styrenesulfonate) layered double hydroxide nanocomposites. Stability and subsequent structural transformation with changes in temperature. J. Mater. Chem. 2003, 13, 258–264. [Google Scholar] [CrossRef]
- Kottegoda, N.S.; Jones, W. Preparation and characterisation of Li-Al-glycine layered double hydroxides (LDHs)-polymer nanocomposites. Macromol. Symp. 2005, 222, 65–71. [Google Scholar] [CrossRef]
- Lee, W.F.; Chen, Y.C. Superabsorbent polymeric materials. XIV. Preparation and water absorbency of nanocomposite superabsorbents containing intercalated hydrotalcite. J. Appl. Polym. Sci. 2004, 94, 2417–2424. [Google Scholar] [CrossRef]
- Shichi, T.; Minamikawa, Y.; Yasuda, N.; Yamada, N.; Okamoto, Y.; Takagi, K. Enantioselective photopinacolization of benzophenone and benzhydrol intercalated in Mg-Al LDH interlayers. Chem. Lett. 2003, 32, 240–241. [Google Scholar] [CrossRef]
- Takagi, K.; Shichi, T.; Usami, H.; Sawaki, Y. Controlled photocycloaddition of unsaturated carboxylates intercalated in hydrotalcite clay interlayers. J. Am. Chem. Soc. 1993, 115, 4339–4344. [Google Scholar] [CrossRef]
- Liu, J.; Li, Y.; Huang, X.; Li, G.; Li, Z. Layered double hydroxide nano- and microstructures grown directly on metal substrates and their calcined products for application as li-ion battery electrodes. Adv. Funct. Mater. 2008, 18, 1448–1458. [Google Scholar] [CrossRef]
- Yao, K.; Taniguchi, M.; Nakata, M.; Takahashi, M.; Yamagishi, A. Electrochemical scanning tunneling microscopy observation of ordered surface layers on an anionic clay-modified electrode. Langmuir 1998, 14, 2890–2895. [Google Scholar] [CrossRef]
- Morigi, M.; Scavetta, E.; Berrettoni, M.; Giorgetti, M.; Tonelli, D. Sulfate-selective electrodes based on hydrotalcites. Anal. Chim. Acta. 2011, 439, 265–272. [Google Scholar] [CrossRef]
- Scavetta, E.; Mignani, A.; Prandstraller, D.; Tonelli, D. Electrosynthesis of thin films of Ni, Al hydrotalcite like compounds. Chem. Mater. 2007, 19, 4523–4529. [Google Scholar] [CrossRef]
- Trifiro, F.; Vaccari, A. Handbook of Layered Materials; Marcel Dekker: New York, NY, USA, 2004; p. 251. [Google Scholar]
- Reichle, W.T. Synthesis of anionic clay-minerals (mixed metal-hydroxides, hydrotalcite). Solid States Ionics 1986, 22, 135–141. [Google Scholar] [CrossRef]
- Intasa-Ard, G.S.; Bureekaew, S.; Ogawa, M. Efficient production of MgAl layered double hydroxide nanoparticle. J. Ceram. Soc. Jpn. 2019, 127, 11–17. [Google Scholar] [CrossRef]
- Yun, S.K.; Pinnavaia, T.J. Water content and particle texture of synthetic hydrotalcite-like layered double hydroxides. Chem. Mater. 1995, 7, 348–354. [Google Scholar] [CrossRef]
- Aisawa, S.; Takahashi, S.; Ogasawara, W.; Umetsu, Y.; Narita, E. Direct intercalation of amino acids into layered double hydroxides by coprecipitation. J. Solid State Chem. 2001, 162, 52–62. [Google Scholar] [CrossRef]
- Li, Y.; Gao, B.; Wu, T.; Sun, D.; Li, X.; Wang, B.; Lu, F. Hexavalent chromium removal from aqueous solution by adsorption on aluminum magnesium mixed hydroxide. Water Res. 2009, 43, 3067–3075. [Google Scholar] [CrossRef]
- Zhang, H.; Zou, K.; Guo, S.; Duan, X. Nanostructural drug-inorganic clay composites: Structure, thermal property and in vitro release of captopril-intercalated Mg–Al-layered double hydroxides. J. Solid State Chem. 2006, 179, 1792–1801. [Google Scholar] [CrossRef]
- Zhang, H.; Pan, D.; Zou, K.; He, J.; Duan, X. A novel core-shell structured magnetic organic-inorganic nanohybrid involving drug-intercalated layered double hydroxides coated on a magnesium ferrite core for magnetically controlled drug release. J. Mater. Chem. 2009, 19, 3069–3077. [Google Scholar] [CrossRef]
- Choi, S.J.; Oh, J.M.; Choy, J.H. Safety aspect of inorganic layered nanoparticles: Size-dependency in vitro and in vivo. J. Nanosci. Nanotechnol. 2008, 8, 5297–5301. [Google Scholar] [CrossRef]
- Chakraborty, M.; Dasgupta, S.; Sengupta, S.; Chakraborty, J.; Ghosh, S.; Ghosh, J.; Basu, D. A facile synthetic strategy for Mg–Al layered double hydroxide material as nanocarrier for methotrexate. Ceram. Int. 2012, 38, 941–949. [Google Scholar] [CrossRef]
- Huang, W.; Zhang, H.; Pan, D. Study on the release behavior and mechanism by monitoring the morphology changes of the large-sized drug-LDH nanohybrids. AIChE J. 2011, 57, 1936–1946. [Google Scholar] [CrossRef]
- Parida, K.M.; Mohapatra, L. Carbonate intercalated Zn/Fe layered double hydroxide: A novel photocatalyst for the enhanced photo degradation of azo dyes. Chem. Eng. J. 2012, 179, 131–139. [Google Scholar] [CrossRef]
- Cantrell, D.G.; Gillie, L.J.; Lee, A.F.; Wilson, K. Structure-reactivity correlations in MgAl hydrotalcite catalysts for biodiesel synthesis. Appl. Catal. A-GEN 2005, 287, 183–190. [Google Scholar] [CrossRef]
- Darder, M.; López-Blanco, M.; Aranda, P.; Leroux, F.; Ruiz-Hitzky, E. Bio-nanocomposites based on layered double hydroxides. Chem. Mater. 2005, 17, 1969–1977. [Google Scholar] [CrossRef]
- Oh, J.M.; Hwang, S.H.; Choy, J.H. The effect of synthetic conditions on tailoring the size of hydrotalcite particles. Solid State Ionics 2002, 151, 285–291. [Google Scholar] [CrossRef]
- Carja, G.; Nakamura, R.; Aida, T.; Niiyama, H. Textural properties of layered double hydroxides: Effect of magnesium substitution by copper or iron. Microporous Mesoporous Mater. 2001, 47, 275–284. [Google Scholar] [CrossRef]
- Shan, R.R.; Yan, L.G.; Yang, Y.M.; Yang, K.; Yu, S.J.; Yu, H.Q.; Du, B. Highly efficient removal of three red dyes by adsorption onto Mg–Al-layered double hydroxide. J. Ind. Eng. Chem. 2015, 21, 561–568. [Google Scholar] [CrossRef]
- Leroux, F.; Gachon, J.; Besse, J.P. Biopolymer immobilization during the crystalline growth of layered double hydroxide. J. Solid State Chem. 2004, 177, 245–250. [Google Scholar] [CrossRef]
- Gao, P.; Li, F.; Zhan, H.; Zhao, N.; Xiao, F.; Wei, W.; Sun, Y. Influence of Zr on the performance of Cu/Zn/Al/Zr catalysts via hydrotalcite-like precursors for CO2 hydrogenation to methanol. J. Catal. 2013, 298, 51–60. [Google Scholar] [CrossRef]
- Cao, F.; Wang, Y.; Ping, Q.; Liao, Z. Zn–Al–NO3-layered double hydroxides with intercalated diclofenac for ocular delivery. Int. J. Pharm. 2011, 404, 250–256. [Google Scholar] [CrossRef]
- Xu, Z.P.; Stevenson, G.; Lu, C.Q.; Lu, G.Q. Dispersion and size control of layered double hydroxide nanoparticles in aqueous solutions. J. Phys. Chem. B. 2006, 110, 16923–16929. [Google Scholar] [CrossRef]
- Choy, J.H.; Kwak, S.Y.; Park, J.S.; Jeong, Y.J. Cellular uptake behavior of [γ-32P] labeled ATP–LDH nanohybrids. J. Mater. Chem. 2001, 11, 1671–1674. [Google Scholar] [CrossRef]
- Gu, Z.; Thomas, A.C.; Xu, Z.P.; Campbell, J.H.; Lu, G.Q. In vitro sustained release of LMWH from MgAl-layered double hydroxide nanohybrids. Chem. Mater. 2008, 20, 3715–3722. [Google Scholar] [CrossRef]
- Ulibarri, M.A.; Pavlovic, I.; Barriga, C.; Hermosın, M.C.; Cornejo, J. Adsorption of anionic species on hydrotalcite-like compounds: Effect of interlayer anion and crystallinity. Appl. Clay Sci. 2001, 18, 17–27. [Google Scholar] [CrossRef]
- Kloprogge, J.T.; Hickey, L.; Frost, R.L. The effects of synthesis pH and hydrothermal treatment on the formation of zinc aluminum hydrotalcites. J. Solid State Chem. 2004, 177, 4047–4057. [Google Scholar] [CrossRef] [Green Version]
- Pan, D.; Zhang, H.; Fan, T.; Chen, J.; Duan, X. Nearly monodispersed core–shell structural Fe3O4@ DFUR–LDH submicro particles for magnetically controlled drug delivery and release. Chem. Commun. 2011, 47, 908–910. [Google Scholar] [CrossRef]
- Raki, L.; Beaudoin, J.J.; Mitchell, L. Layered double hydroxide-like materials: Nanocomposites for use in concrete. Cement Concrete Res. 2004, 34, 1717–1724. [Google Scholar] [CrossRef]
- Li, Y.W.; Yao, J.H.; Liu, C.J.; Zhao, W.M.; Deng, W.X.; Zhong, S.K. Effect of interlayer anions on the electrochemical performance of Al-substituted α-type nickel hydroxide electrodes. Int. J. Hydrog. Energy 2010, 35, 2539–2545. [Google Scholar] [CrossRef]
- Géraud, E.; Rafqah, S.; Sarakha, M.; Forano, C.; Prevot, V.; Leroux, F. Three dimensionally ordered macroporous layered double hydroxides: Preparation by templated impregnation/coprecipitation and pattern stability upon calcination. Chem. Mater. 2007, 20, 1116–1125. [Google Scholar] [CrossRef]
- Li, B.; He, J.; Evans, D.G.; Duan, X. Inorganic layered double hydroxides as a drug delivery system—Intercalation and in vitro release of fenbufen. Appl. Clay Sci. 2004, 27, 199–207. [Google Scholar] [CrossRef]
- Kang, D.; Yu, X.; Tong, S.; Ge, M.; Zuo, J.; Cao, C.; Song, W. Performance and mechanism of Mg/Fe layered double hydroxides for fluoride and arsenate removal from aqueous solution. Chem. Eng. J. 2013, 228, 731–740. [Google Scholar] [CrossRef]
- Nitoh, K.; Ayral, A.; Ogawa, M. Preparation of well-defined nanometer-sized layered double hydroxides by novel pH adjustment method using ion-exchange resin. Chem. Lett. 2010, 39, 1018–1019. [Google Scholar] [CrossRef]
- Naito, S.; Nitoh, K.; Ayral, A.; Ogawa, M. Preparation of finite particles of nitrate forms of layered double hydroxides by pH adjustment with anion exchange resin. Ind. Eng. Chem. Res. 2012, 51, 14414–14418. [Google Scholar] [CrossRef]
- Li, K.; Kumada, N.; Yonesaki, Y.; Takei, T.; Kinomura, N.; Wang, H.; Wang, C. The pH effects on the formation of Ni/Al nitrate form layered double hydroxides (LDHs) by chemical precipitation and hydrothermal method. Mater. Chem. Phys. 2010, 121, 223–229. [Google Scholar] [CrossRef]
- Ogawa, M.; Inomata, K. Preparation of layered double hydroxides. Clay Sci. 2011, 15, 131–137. [Google Scholar]
- Reichle, W.T. Catalytic reactions by thermally activated, synthetic, anionic clay minerals. J. Catal. 1985, 94, 547–557. [Google Scholar] [CrossRef]
- Miyata, S. The syntheses of hydrotalcite-like compounds and their structures and physico-chemical properties-I: The Systems Mg2+-Al3+-NO3−, Mg2+-Al3+-Cl−, Mg2+-Al 3+-ClO4−, Ni2+-Al3+-Cl− and Zn2+-Al3+-Cl−. Clays Clay Miner. 1975, 23, 369–375. [Google Scholar] [CrossRef]
- Miyata, S.; Akira, O. Synthesis of hydrotalcite-like compounds and their physico-chemical properties-the systems Mg2+-Al3+-SO42− and Mg2+-Al3+-CrO42−. Clays Clay Miner. 1977, 25, 14–18. [Google Scholar] [CrossRef]
- Miyata, S. Physico-chemical properties of synthetic hydrotalcites in relation to composition. Clays Clay miner. 1980, 28, 50–56. [Google Scholar] [CrossRef]
- Zhao, S.; Yi, H.; Tang, X.; Gao, F.; Yu, Q.; Zhou, Y.; Yang, Z. Enhancement effects of ultrasound assisted in the synthesis of NiAl hydrotalcite for carbonyl sulfide removal. Ultrason. Sonochem. 2016, 32, 336–342. [Google Scholar] [CrossRef]
- Climent, J.M.; Corma, A.; Iborra, S.; Epping, K.; Velty, A. Increasing the basicity and catalytic activity of hydrotalcites by different synthesis procedures. J. Catal. 2004, 225, 316–326. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, F.; Zhang, R.; Evans, D.G.; Duan, X. Preparation of layered double-hydroxide nanomaterials with a uniform crystallite size using a new method involving separate nucleation and aging steps. Chem. Mater. 2002, 14, 4286–4291. [Google Scholar] [CrossRef]
- Feng, Y.; Li, D.; Li, C.; Wang, Z.; Evans, D.G.; Duan, X. Synthesis of Cu-containing layered double hydroxides with a narrow crystallite-size distribution. Clays Clay Miner. 2003, 51, 566–569. [Google Scholar] [CrossRef]
- Chang, Z.; Evans, D.G.; Duan, X.; Vial, C.; Ghanbaja, J.; Prevot, V.; Forano, C. Synthesis of [Zn–Al–CO3] layered double hydroxides by a coprecipitation method under steady-state conditions. J. Solid State Chem. 2005, 178, 2766–2777. [Google Scholar] [CrossRef]
- Shiba, K.; Ogawa, M. Precise Synthesis of well-defined inorganic-organic hybrid particles. Chem. Rec. 2018, 18, 950–968. [Google Scholar] [CrossRef]
- Pang, X.; Sun, M.; Ma, X.; Hou, W. Synthesis of layered double hydroxide nanosheets by coprecipitation using a T-type microchannel reactor. J. Solid State Chem. 2014, 210, 111–115. [Google Scholar] [CrossRef]
- Labajos, F.M.; Rives, V.; Ulibarri, M.A. Effect of hydrothermal and thermal treatments on the physicochemical properties of Mg-Al hydrotalcite-like materials. J. Mater. Sci. 1992, 27, 1546–1552. [Google Scholar] [CrossRef]
- Benito, P.; Guinea, I.; Herrero, M.; Labajos, M.F.; Rives, V. Incidence of microwave hydrothermal treatments on the crystallinity properties of hydrotalcite-like compounds. Z. Anorg. Allg. Chem. 2007, 633, 1815–1819. [Google Scholar] [CrossRef]
- Benito, P.; Labajos, F.M.; Rives, V. Microwaves and layered double hydroxides: A smooth understanding. Pure Appl. Chem. 2009, 81, 1459–1471. [Google Scholar] [CrossRef]
- Sugimoto, T. Monodispersed Particles; Elsevier: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Costantino, U.; Marmottini, F.; Nocchetti, M.; Vivan, R. New synthetic routes to hydrotalcite-like compounds–Characterization and properties of the obtained materials. Eur. J. Inorg. Chem. 1998, 10, 1439–1446. [Google Scholar] [CrossRef]
- Inayat, A.; Klumpp, M.; Schwieger, W. The urea method for the direct synthesis of ZnAl layered double hydroxides with nitrate as the interlayer anion. Appl. Clay Sci. 2011, 51, 452–459. [Google Scholar] [CrossRef]
- Staal, L.B.; Pushparaj, S.S.C.; Forano, C.; Prevot, V.; Ravnsbæk, D.B.; Bjerring, M.; Nielsen, U.G. Competitive reactions during synthesis of zinc aluminum layered double hydroxides by thermal hydrolysis of urea. J. Mater. Chem. A 2017, 5, 21795–21806. [Google Scholar] [CrossRef]
- Narita, E.; Takahashi, Y.; Umetsu, Y. Synthesis of highly crystalline Al-Li layered double hydroxide by homogeneous precipitation method. Nippon Kagaku Kaishi 2001, 5, 273–279. [Google Scholar] [CrossRef]
- Liu, Z.; Ma, R.; Osada, M.; Iyi, N.; Ebina, Y.; Takada, K.; Sasaki, T. Synthesis, anion exchange, and delamination of Co-Al layered double hydroxide: Assembly of the exfoliated nanosheet/polyanion composite films and magneto-optical studies. J. Am. Chem. Soc. 2006, 128, 4872–4880. [Google Scholar] [CrossRef]
- Adachi-Pagano, M.; Forano, C.; Besse, J.-P. Synthesis of Al-rich hydrotalcite-like compounds by using the urea hydrolysis reaction-control of size and morphology. J. Mater. Chem. 2003, 13, 1988–1993. [Google Scholar] [CrossRef]
- Iyi, N.; Tamura, K.; Yamada, H. One-pot synthesis of organophilic layered double hydroxides (LDHs) containing aliphatic carboxylates: Extended “homogeneous precipitation” method. J. Colloid Interface Sci. 2009, 340, 67–73. [Google Scholar] [CrossRef]
- Perioli, L.; Ambrogi, V.; Rossi, C.; Latterini, L.; Nocchetti, M.; Costantino, U. Use of anionic clays for photoprotection and sunscreen photostability: Hydrotalcites and phenylbenzimidazole sulfonic acid. J. Phys. Chem. Solids 2006, 67, 1079–1083. [Google Scholar] [CrossRef]
- Iyi, N.; Matsumoto, T.; Kaneko, Y.; Kitamura, K. A novel synthetic route to layered double hydroxides using hexamethylenetetramine. Chem. Lett. 2004, 33, 1122–1123. [Google Scholar] [CrossRef]
- Ogawa, M.; Kaiho, H. Homogeneous precipitation of uniform hydrotalcite particles. Langmuir 2002, 18, 4240–4242. [Google Scholar] [CrossRef]
- Kayano, M.; Ogawa, M. Preparation of large platy particles of Co-Al layered double hydroxides. Clays Clay Miner. 2006, 54, 385–392. [Google Scholar] [CrossRef]
- Kayano, M.; Ogawa, M. Controlled particle size and size distribution of Co-Al layered double hydroxide via the hydrothermal urea method in aqueous alcohols. Bull. Chem. Soc. Jpn. 2006, 79, 1988–1990. [Google Scholar] [CrossRef]
- Arai, Y.; Ogawa, M. Preparation of Co-Al layered double hydroxides by the hydrothermal urea method for controlled particle size. Appl. Clay Sci. 2009, 42, 601–604. [Google Scholar] [CrossRef]
- Pérez-Ramírez, J.; Mul, G.; Kapteijn, F.; Moulijn, J.A. In situ investigation of the thermal decomposition of Co–Al hydrotalcite in different atmospheres. J. Mater. Chem. 2001, 11, 821–830. [Google Scholar] [CrossRef]
- Kloprogge, J.T.; Frost, R.L. Fourier transform and Raman spectroscopic study of the local structure of Mg-, Ni-, and Co-hydrotalcites. J. Solid State Chem. 1999, 146, 506–515. [Google Scholar] [CrossRef]
- Shaw, W.H.R.; Bordeaux, J.J. The decomposition of urea in aqueous media. J. Am. Chem. Soc. 1955, 77, 4729–4733. [Google Scholar] [CrossRef]
- He, J.; Li, B.; Evans, D.G.; Duan, X. Synthesis of layered double hydroxides in an emulsion solution. Colloids Surf. A Physicochem. Eng. Aspects 2004, 251, 191–196. [Google Scholar] [CrossRef]
- Hu, G.; O’Hare, D. Unique layered double hydroxide morphologies using reverse microemulsion synthesis. J. Am. Chem. Soc. 2005, 127, 17808–17813. [Google Scholar] [CrossRef]
- Hu, G.; Wang, N.; O’Hare, D.; Davis, J. Synthesis of magnesium aluminium layered double hydroxides in reverse microemulsions. J. Mater. Chem. 2007, 17, 2257–2266. [Google Scholar] [CrossRef]
- Bellezza, F.; Cipiciani, A.; Costantino, U.; Nocchetti, N.; Posati, T. Hydrotalcite-Like Nanocrystals from Water-in-Oil Microemulsions. Eur. J. Inorg. Chem. 2009, 18, 2603–2611. [Google Scholar] [CrossRef]
- Bellezza, F.; Nocchetti, M.; Posati, T.; Giovagnoli, S.; Cipiciani, A. Synthesis of colloidal dispersions of NiAl, ZnAl, NiCr, ZnCr, NiFe, and MgFe hydrotalcite-like nanoparticles. J. Colloid Interface Sci. 2012, 376, 20–27. [Google Scholar] [CrossRef]
- Ogawa, M.; Asai, S. Hydrothermal synthesis of layered double hydroxide-deoxycholate intercalation compounds. Chem. Mater. 2000, 12, 3253–3255. [Google Scholar] [CrossRef]
- Ogawa, M.; Saito, F. Easily oxidizable polysulfide anion occluded in the interlayer space of Mg/Al layered double hydroxide. Chem. Lett. 2004, 33, 1030–1031. [Google Scholar] [CrossRef]
- Ogawa, M.; Sugiyama, Y. Facile synthesis of Zn-Al layered double hydroxide from aqueous suspension of zinc oxide and aluminum hydroxide. J. Ceram. Soc. Jpn. 2009, 117, 179–184. [Google Scholar] [CrossRef] [Green Version]
- Newman, S.P.; Jones, W.; O’Corner, P.; Stamires, D.N. Synthesis of the 3R2 polytype of a hydrotalcite-like mineral. J. Mater. Chem. 2002, 12, 153–155. [Google Scholar] [CrossRef]
- Xu, P.Z.; Lu, Q.G. Hydrothermal synthesis of Layered Double Hydroxides (LDHs) from mixed MgO and Al2O3: LDH formation mechanism. Chem. Mater. 2005, 17, 1055–1062. [Google Scholar] [CrossRef]
- Mitchell, S.; Biswick, T.; Jones, W.; Williams, G.; O’Hare, D. A synchrotron radiation study of the hydrothermal synthesis of layered double hydroxides from MgO and Al2O3 slurries. Green Chem. 2007, 9, 373–378. [Google Scholar] [CrossRef]
- Chitrakar, R.; Tezuka, S.; Sonoda, A.; Sakane, K.; Ooi, K.; Hirotsu, T. A solvent-free synthesis of Zn-Al layered double hydroxides. Chem. Lett. 2007, 36, 446–447. [Google Scholar] [CrossRef]
- Senna, M. Chemical powder technology—A new insight into atomic processes on the surface of fine particles. Adv. Powder. Tech. 2002, 3, 115–138. [Google Scholar] [CrossRef]
- Braga, D.; Giaffreda, S.L.; Grepioni, F.; Pettersen, A.; Maini, L.; Curzi, M.; Polito, M. Mechanochemical preparation of molecular and supramolecular organometallic materials and coordination networks. Dalton trans. 2006, 10, 1249–1263. [Google Scholar] [CrossRef]
- Pichon, A.; Lazuen-Garay, A.; James, S.L. Solvent-free synthesis of a microporous metal–organic framework. Cryst. Eng. Comm. 2006, 8, 211–214. [Google Scholar] [CrossRef]
- Baláž, P.; Achimovičová, M.; Baláž, M.; Billik, P.; Cherkezova-Zheleva, Z.; Criado, J.M.; Delogu, F.; Dutková, E.; Gaffet, E.; Gotor, F.J.; et al. Hallmarks of mechanochemistry: From nanoparticles to technology. Chem. Soc. Rev. 2013, 42, 7571–7637. [Google Scholar] [CrossRef]
- Takacs, L. The historical development of mechanochemistry. Chem. Soc. Rev. 2013, 42, 7649–7659. [Google Scholar] [CrossRef]
- Wang, G.W. Mechanochemical organic synthesis. Chem. Soc. Rev. 2013, 42, 7668–7700. [Google Scholar] [CrossRef]
- Alex, T.C.; Kumar, R.; Roy, S.K.; Mehrotra, S.P. Mechanical activation of Al-oxyhydroxide minerals–a review. Miner. Process. Extr. Metall. Rev. 2016, 37, 1–26. [Google Scholar] [CrossRef]
- Zhang, P.; Dai, S. Mechanochemical synthesis of porous organic materials. J. Mater. Chem. A 2017, 5, 16118–16127. [Google Scholar] [CrossRef]
- Intasa-Ard, G.S.; Imwiset, J.K.; Bureekaew, S.; Ogawa, M. Mechanochemical methods for the preparation of intercalation compounds, from intercalation to the formation of layered double hydroxides. Dalton Trans. 2018, 47, 2896–2916. [Google Scholar] [CrossRef]
- Poeppelmeier, K.R.; Hwu, S.J. Synthesis of lithium dialuminate by salt imbibition. Inorg. Chem. 1987, 26, 3297–3302. [Google Scholar] [CrossRef]
- Ay, A.N.; Zümreoglu-Karan, B.; Mafra, L. A Simple Mechanochemical Route to Layered Double Hydroxides: Synthesis of Hydrotalcite-Like Mg-Al-NO3-LDH by Manual Grinding in a Mortar. Z. Anorg. Allg. Chem. 2009, 635, 1470–1475. [Google Scholar] [CrossRef]
- Ferencz, Z.; Szabados, M.; Ádok-Sipiczki, M.; Kukovecz, Á.; Kónya, Z.; Sipos, P.; Pálinkó, I. Mechanochemically assisted synthesis of pristine Ca (II) Sn (IV)-layered double hydroxides and their amino acid intercalated nanocomposites. J. Mater. Sci. 2014, 49, 8478–8486. [Google Scholar] [CrossRef]
- Conterosito, E.; Van Beek, W.; Palin, L.; Croce, G.; Perioli, L.; Viterbo, D.; Gatti, G.; Milanesio, M. Development of a fast and clean intercalation method for organic molecules into layered double hydroxides. Cryst. Growth Des. 2013, 13, 1162–1169. [Google Scholar] [CrossRef]
- Jia, C.; Zhang, X.; Li, S. Studies on mechanochemical method to synthesize LDH nanoparticles. Chin. J. Chem. 2012, 30, 277–282. [Google Scholar] [CrossRef]
- Zhang, X.; Li, S. Mechanochemical approach for synthesis of layered double hydroxides. Appl. Surf. Sci. 2013, 274, 158–163. [Google Scholar] [CrossRef]
- Isupov, V.P.; Chupakhina, L.E.; Mitrofanova, R.P. Mechanochemical synthesis of double hydroxides. J. Mater. Synth. Process. 2000, 8, 251–253. [Google Scholar] [CrossRef]
- Khusnutdinov, V.P.; Isupov, V.P. Mechanochemical synthesis of a hydroxycarbonate form of layered magnesium aluminum hydroxides. Inorg. Mater. 2008, 44, 263–267. [Google Scholar] [CrossRef]
- Tongamp, W.; Zhang, Q.; Saito, F. Mechanochemical route for synthesizing nitrate form of layered double hydroxide. Powder Technol. 2008, 185, 43–48. [Google Scholar] [CrossRef]
- Tongamp, W.; Zhang, Q.; Saito, F. Generation of hydrogen gas from polyethylene mechanically milled with Ni-doped layered double hydroxide. Fuel Process. Technol. 2009, 90, 909–913. [Google Scholar] [CrossRef]
- Kuramoto, K.; Ogawa, M. Preparation of layered double hydroxide–organic intercalation compounds by solid–solid reactions. Bull. Chem. Soc. Jpn. 2011, 84, 675–677. [Google Scholar] [CrossRef]
- Kuramoto, K.; Intasa-Ard, G.S.; Bureekaew, S.; Ogawa, M. Mechanochemical synthesis of finite particle of layered double hydroxide-acetate intercalation compound: Swelling, thin film and ion exchange. J. Solid State Chem. 2017, 253, 147–151. [Google Scholar] [CrossRef]
- Meyn, M.; Beneke, K.; Lagaly, G. Anion-exchange reactions of layered double hydroxides. Inorg. Chem. 1990, 29, 5201–5207. [Google Scholar] [CrossRef]
- Kelkar, C.P.; Schutz, A.A. Ni-, Mg- and Co-containing hydrotalcite-like materials with a sheet-like morphology: Synthesis and characterization. Microporous Mater. 1997, 10, 163–172. [Google Scholar] [CrossRef]
- Greenwell, H.C.; Jones, W.; Stamires, D.N.; O’Connor, P.; Brady, M.F. A one-pot synthesis of hybrid organo-layered double hydroxide catalyst precursors. Green Chem. 2006, 8, 1067–1072. [Google Scholar] [CrossRef]
- Wang, B.; Qu, J.; Li, X.; He, X.; Zhang, Q. Precursor Preparation to Promote the Adsorption of Mg-Al Layered Double Hydroxide. J. Am. Ceram. Soc. 2016, 99, 2882–2885. [Google Scholar] [CrossRef]
- Qu, J.; He, X.; Chen, M.; Hu, H.; Zhang, Q.; Liu, X. Mechanochemical synthesis of Cu-Al and methyl orange intercalated Cu-Al layered double hydroxides. Mater. Chem. Phys. 2017, 191, 173–180. [Google Scholar] [CrossRef]
- Qu, J.; He, X.; Chen, M.; Huang, P.; Zhang, Q.; Liu, X. A facile mechanochemical approach to synthesize Zn-Al layered double hydroxide. J. Solid State Chem. 2017, 250, 1–5. [Google Scholar] [CrossRef]
- Szabados, M.; Bús, C.; Adok-Sipiczki, M.; Kónya, Z.; Kukovecz, Á.; Sipos, P.; Pálinkó, I. Ultrasound-enhanced milling in the synthesis of phase-pure, highly crystalline ZnAl-layered double hydroxide of low Zn (II) content. Particuology 2016, 27, 29–33. [Google Scholar] [CrossRef]







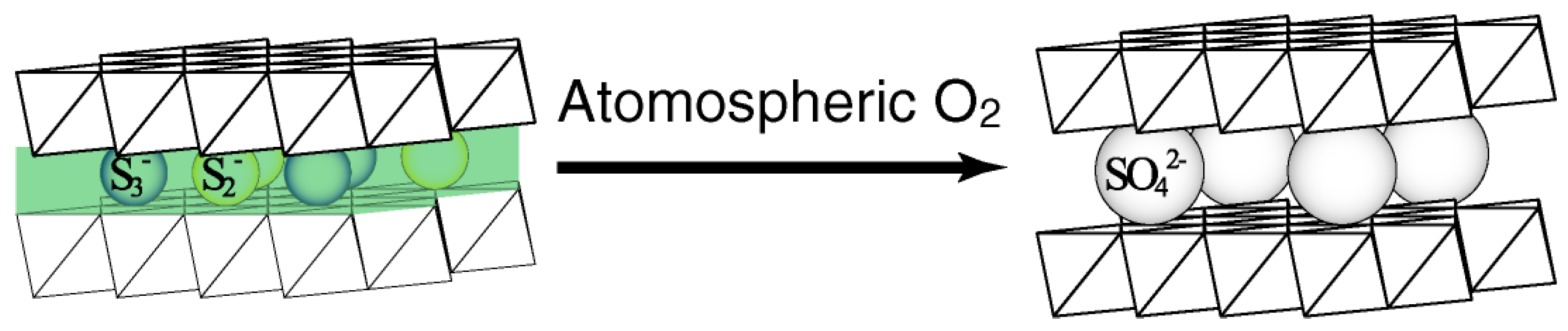


| Synthetic Condition | LDH Characteristic | Ref | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [Mg2+ + Al3+] (M) | M2+/M3+ Ratio | Base | [Base] (M) | Mixing Condition | Aging Condition | Product Amount (g from 100 mL) | Interlayer Anion | Particle Morphology | |||||
| T (°C) | Atmosphere | T (°C) | Time (h) | Atmosphere | Shape | Size | |||||||
| 0.9 | 2 | NaOH and Na2CO3 | 2.0 | <35 | Air | 65 | 18 | Air | 5.9–7.1 | CO32− | Poorly dispersed unshaped aggregates | 0.5 µm | [82] |
| 3 | 25 | 200 | Air (closed container) | 5.9 | Poorly dispersed well-defined hexagonal plate | 1 µm | [48] | ||||||
| 300 | 5.9 | Well-defined hexagonal plate | 15 µm | ||||||||||
| 1.0 | 3 | NaOH | 2.0 | 25 | N2 | without aging | Not given | NO3− | Poorly dispersed spherical aggregates | Aggregates size 10–50 µm | [83] | ||
| Cl− | |||||||||||||
| ClO3− | |||||||||||||
| 3.5 | 30 | Air | without aging | Not given | SO42− | Aggregates | >10 µm | [84] | |||||
| 2.0 | Cr2O42− | ||||||||||||
| 2 | NaOH and Na2CO3 | 2.0 | 40 | Air | 150 | 24 | Air (closed container) | Not given | CO32− | Hexagonal aggregates | Not given | [85] | |
| 3 | Spherical aggregates | Not given | |||||||||||
| 3 | NH3 gas | Not given | 25 | N2 | without aging | Not given | Cl− | Poorly dispersed spherical aggregates | Aggregates size 10–50 µm | [83] | |||
| 3 | NH4OH | 6.4 | 25 | Air | without aging | 9.7 | Cl− | Uniform platy particle | 50 nm | [49] | |||
| 25 | 24 | Air | 10.6 | ||||||||||
| 80 | 24 | Air | 10.0 | ||||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wijitwongwan, R.; Intasa-ard, S.; Ogawa, M. Preparation of Layered Double Hydroxides toward Precisely Designed Hierarchical Organization. ChemEngineering 2019, 3, 68. https://doi.org/10.3390/chemengineering3030068
Wijitwongwan R, Intasa-ard S, Ogawa M. Preparation of Layered Double Hydroxides toward Precisely Designed Hierarchical Organization. ChemEngineering. 2019; 3(3):68. https://doi.org/10.3390/chemengineering3030068
Chicago/Turabian StyleWijitwongwan, Rattanawadee (Ploy), Soontaree (Grace) Intasa-ard, and Makoto Ogawa. 2019. "Preparation of Layered Double Hydroxides toward Precisely Designed Hierarchical Organization" ChemEngineering 3, no. 3: 68. https://doi.org/10.3390/chemengineering3030068





