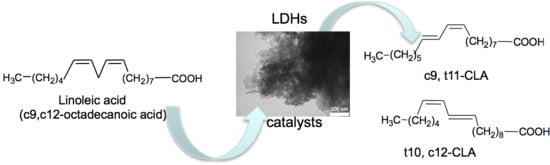
Layered Double Hydroxides for the Catalytic Isomerization of Linoleic Acid to Conjugated Linoleic Acids (CLAs)
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Characterization of the Catalysts
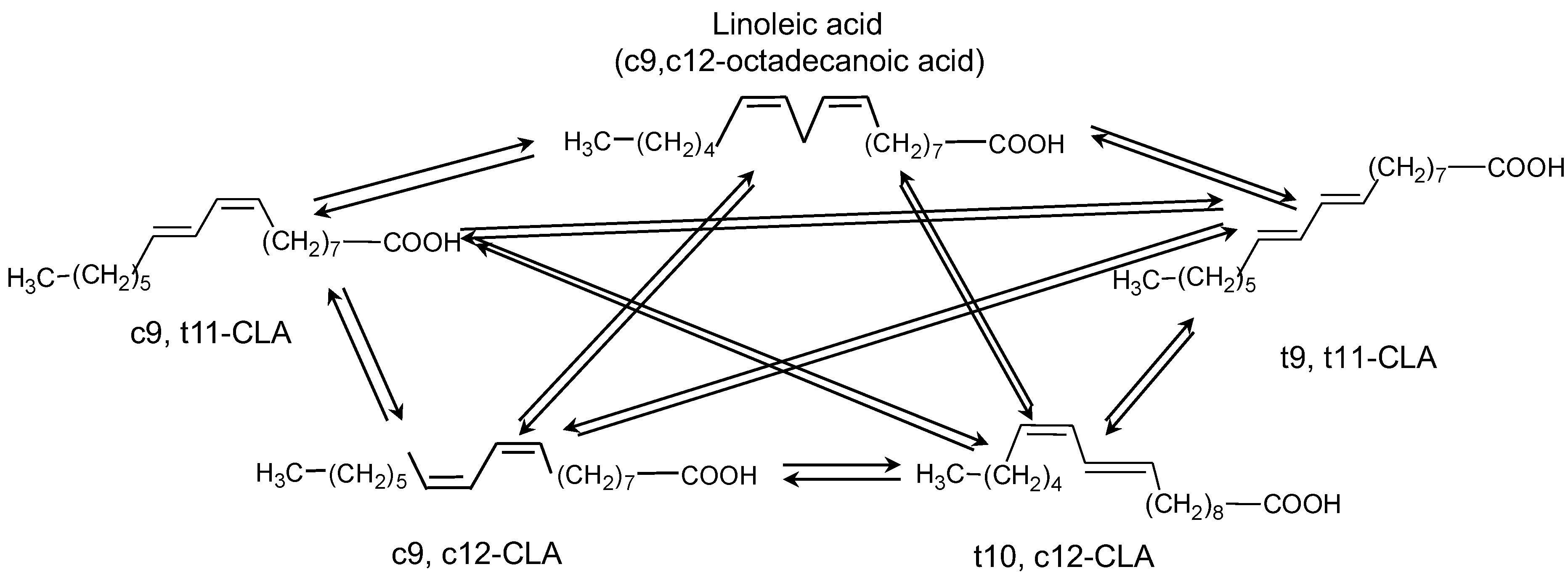
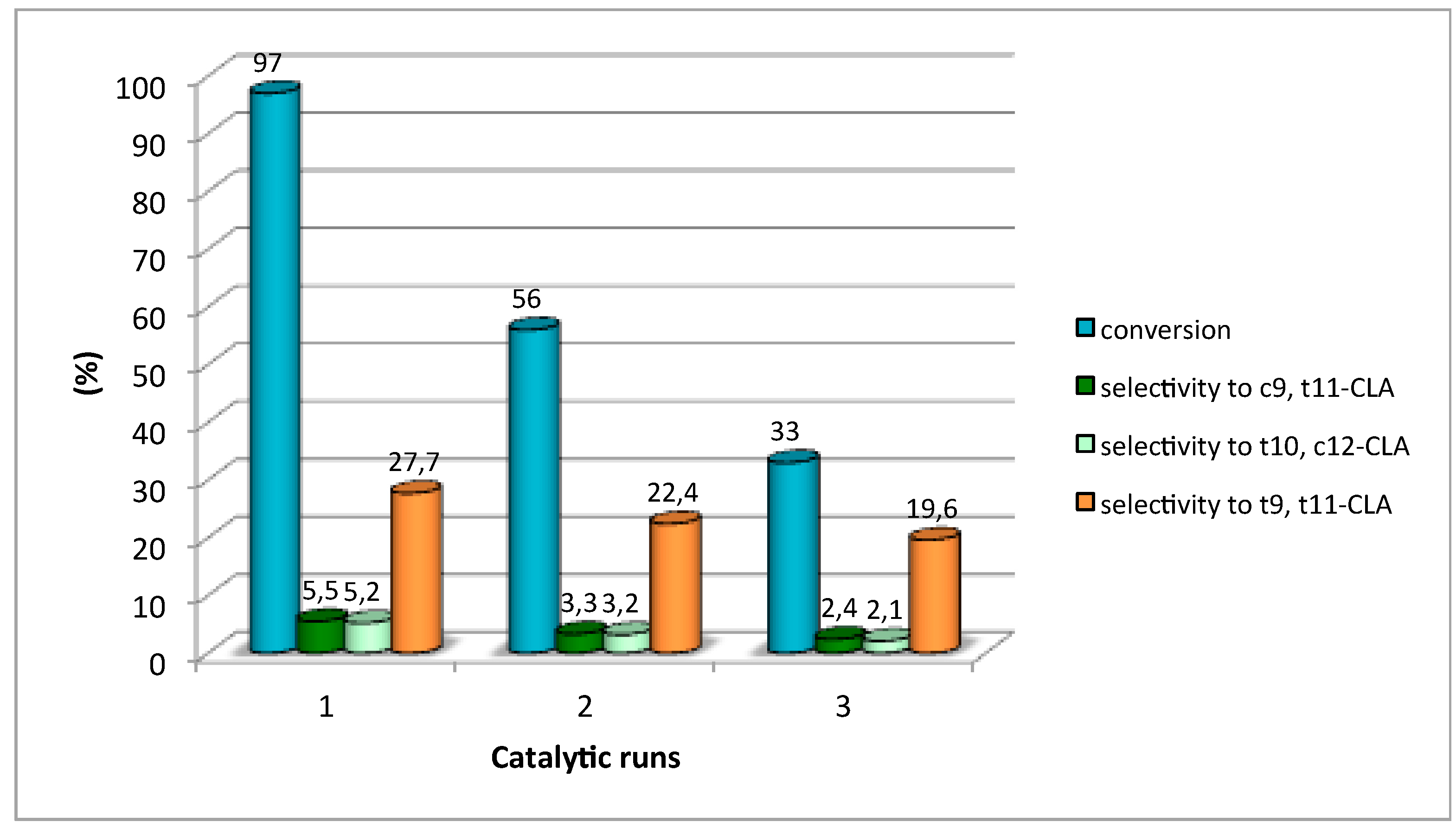
3.2. Catalytic Activity
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- O’Quinn, P.R.; Nelssen, J.L.; Goodband, R.D.; Tokach, M.D. Conjugated linoleic acid. Anim. Health Res. Rev. 2000, 1, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Whigham, L.D.; Cook, M.E.; Atkinson, R.L. Conjugated linoleic acid: Implications for human health. Pharmacol. Res. 2000, 42, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Mougios, V.; Matsakas, A.; Petridou, A.; Ring, S.; Sagredos, A.; Melissopoulou, A.; Tsigilis, N.; Nikolaidis, M.J. Effect of supplementation with conjugated linoleic acid on human serum lipids and body fat. Nutr. Biochem. 2001, 12, 585–594. [Google Scholar] [CrossRef]
- Palombo, J.D.; Ganguly, A.; Bistrian, B.R.; Menard, M.P. The antiproliferative effects of biologically active isomers of conjugated linoleic acid on human colorectal and prostatic cancer cells. Cancer Lett. 2002, 177, 163–172. [Google Scholar] [CrossRef]
- Busch, S.; Zander, L.; Albiez, W.; Horlacher, P.; Westfechtel, A. Method for the production of conjugated linoleic acids. EP1527153A1, 4 May 2005. [Google Scholar]
- Bernas, A.; Kumar, N.; Mäki-Arvela, P.; Laine, E.; Holmbom, B.; Salmi, T.; Murzin, D. Conjugation of linoleic acid over a hydrogen pre-activated heterogeneous catalyst. Chem. Commun. 2002, 10, 1142–1143. [Google Scholar] [CrossRef]
- Bernas, A.; Laukkanen, P.; Kumar, N.; Mäki-Arvela, P.; Väyrynen, J.; Laine, E.; Holmbom, B.; Salmi, T.; Murzin, D. A new heterogeneously catalytic pathway for isomerization of linoleic acid over Ru/C and Ni/H–MCM-41 catalysts. J. Catal. 2002, 210, 354–366. [Google Scholar] [CrossRef]
- Bernas, A.; Kumar, N.; Mäki-Arvela, P.; Kul’kova, N.V.; Holmbom, B.; Salmi, T.; Murzin, D. Isomerization of linoleic acid over supported metal catalysts. Appl. Catal. A 2003, 245, 257–275. [Google Scholar] [CrossRef]
- Bernas, A.; Mäki-Arvela, P.; Kumar, N.; Holmbom, B.; Salmi, T.; Murzin, D. Heterogeneously catalytic isomerization of linoleic acid over supported ruthenium catalysts for production of anticarcinogenic food constituents. Ind. Eng. Chem. Res. 2003, 42, 718–727. [Google Scholar] [CrossRef]
- Bernas, A.; Murzin, D. Influence of hydrogen preactivation on the linoleic acid isomerization properties of supported ruthenium catalyst. React. Kinet. Catal. Lett. 2003, 78, 3–10. [Google Scholar] [CrossRef]
- Bernas, A.; Kumar, N.; Laukkanen, P.; Väyrynen, J.; Salmi, T.; Murzin, D. Influence of ruthenium precursor on catalytic activity of Ru/Al2O3 catalyst in selective isomerization of linoleic acid to cis-9,trans-11- andtrans-10,cis-12-conjugated linoleic acid. Appl. Catal. A 2004, 267, 121–133. [Google Scholar] [CrossRef]
- Bernas, A.; Kumar, N.; Mäki-Arvela, P.; Holmbom, B.; Salmi, T.; Murzin, D. Heterogeneous catalytic production of conjugated linoleic acid. Org. Process. Res. Dev. 2004, 8, 341–352. [Google Scholar] [CrossRef]
- Sim, K.S.; Hilaire, L.; Le Normand, F.; Touroude, R.; Paul-Boncour, V.; Percheron-Guegan, A. Catalysis by palladium–rare-earth-metal (REPd3) intermetallic compounds: hydrogenation of but-1-ene, buta-1,3-diene and but-1-yne. J. Chem. Soc. Faraday Trans. 1991, 87, 1453–1460. [Google Scholar] [CrossRef]
- Kreich, M.; Claus, P. Direct Conversion of Linoleic Acid over Silver Catalysts in the Presence of H2: An Unusual Way towards Conjugated Linoleic Acids. Angew. Chem. Int. Ed. 2005, 44, 7800–7804. [Google Scholar] [CrossRef] [PubMed]
- Bauer, P.; Horlacher, P.; Claus, P. Direct isomerization of linoleic acid to conjugated linoleic acids (CLA) using gold catalysts. Chem. Eng. Technol. 2009, 32, 2005–2010. [Google Scholar] [CrossRef]
- Simakova, O.; Lleino, A.; Campo, B.; Maki-Arvela, P.; Kordas, K.; Mikkola, J.; Murzin, D. Linoleic acid isomerization over mesoporous carbon supported gold catalysts. Catal. Today 2010, 150, 32–36. [Google Scholar] [CrossRef]
- Tanabe, K.; Hölderich, W. Industrial application of solid acid–base catalysts. Appl. Catal. A Gen. 1999, 181, 399–434. [Google Scholar] [CrossRef]
- Cardó, X.; Bergadà, O.; Cesteros, Y.; Salagre, P. Effect of catalyst acidity and porosity on the catalytic isomerization of linoleic acid to obtain conjugated linoleic acids (CLAs). Chem. Eng. J. 2012, 183, 459–465. [Google Scholar] [CrossRef]
- Oukaci, R.; Wu, J.C.S. Secondary reactions during CO hydrogenation on zeolite-supported metal catalysts: Influence of alkali cations. J. Catal. 1987, 107, 471–481. [Google Scholar] [CrossRef]
- Chen, J.; Chen, X.; Zheng, Y.; Li, Q. Synergetic effect of ruthenium and basicity sites in the Ru–MgAl catalyst for hydrogen-free production of conjugated linoleic acids. RSC Adv. 2015, 5, 20248–20255. [Google Scholar] [CrossRef]
- Vaccari, A. Clays and catalysis: A promising future. Appl. Clay Sci. 1999, 14, 161–198. [Google Scholar] [CrossRef]
- Rives, V.; Ulibarri, M.A. Layered double hydroxides (LDH) intercalated with metal coordination compounds and oxometalates. Coord. Chem. Rev. 1999, 181, 61–120. [Google Scholar] [CrossRef]
- Cavani, F.; Trifiro, F.; Vaccari, A. Hydrotalcite-type anionic clays: Preparation, properties and applications. Catal. Today 1991, 11, 173–302. [Google Scholar] [CrossRef]
- Cesteros, Y.; Salagre, P.; Medina, F.; Sueiras, J.E.; Tichit, D.; Coq, B. Hydrodechlorination of 1,2,4-trichlorobenzene on nickel-based catalysts prepared from several Ni/Mg/Al hydrotalcite-like precursors. Appl. Catal. B Environ. 2001, 32, 25–35. [Google Scholar] [CrossRef]
- Basile, F.; Fornasari, G.; Rosetti, V.; Trifirò, F.; Vaccari, A. Effect of the Mg/Al ratio of the hydrotalcite-type precursor on the dispersion and activity of Rh and Ru catalysts for the partial oxidation of methane. Catal. Today 2004, 91–92, 293–297. [Google Scholar] [CrossRef]
- Abelló, S.; Medina, F.; Tichit, D.; Pérez-Ramírez, J.; Groen, J.C.; Sueiras, J.E.; Salagre, P.; Cesteros, Y. Aldol Condensations Over Reconstructed Mg–Al Hydrotalcites: Structure–Activity Relationships Related to the Rehydration Method. Chem. Eur. J. 2005, 11, 728–739. [Google Scholar] [CrossRef] [PubMed]
- Debecker, D.P.; Gaigneaux, E.M.; Busca, G. Exploring, Tuning, and Exploiting the Basicity of Hydrotalcites for Applications in Heterogeneous Catalysis. Chemistry 2009, 15, 3920–3935. [Google Scholar] [CrossRef] [PubMed]
- Climent, M.J.; Corma, A.; Iborra, S.; Epping, K.; Velty, A. Increasing the basicity and catalytic activity of hydrotalcites by different synthesis procedures. J. Catal. 2004, 225, 316–326. [Google Scholar] [CrossRef]
- Di Cosimo, J.I.; Apesteguia, C.R.; Ginés, M.J.L.; Iglesia, E. Structural Requirements and Reaction Pathways in Condensation Reactions of Alcohols on MgyAlOx Catalysts. J. Catal. 2000, 190, 261–275. [Google Scholar] [CrossRef]
- Corma, A. Inorganic Solid Acids and Their Use in Acid-Catalyzed Hydrocarbon Reactions. Chem. Rev. 1995, 95, 559–614. [Google Scholar] [CrossRef]
- Bruce, S.M.; Zong, Z.; Chatzidimitriou, A.; Avci, L.E.; Bond, J.Q.; Carreon, M.A.; Wettstein, S.G. Small pore zeolite catalysts for furfural synthesis from xylose and switchgrass in a γ-valerolactone/water solvent. J. Mol. Catal. A Chem. 2016, 422, 18–22. [Google Scholar] [CrossRef]
- Jones, A.J.; Iglesia, E. The strength of Brønsted acid sites in microporous aluminosilicates. ACS Catal. 2015, 5, 5741–5755. [Google Scholar] [CrossRef]
- Hattori, H. Heterogeneous basic catalysis. Chem. Rev. 1995, 95, 537–558. [Google Scholar] [CrossRef]
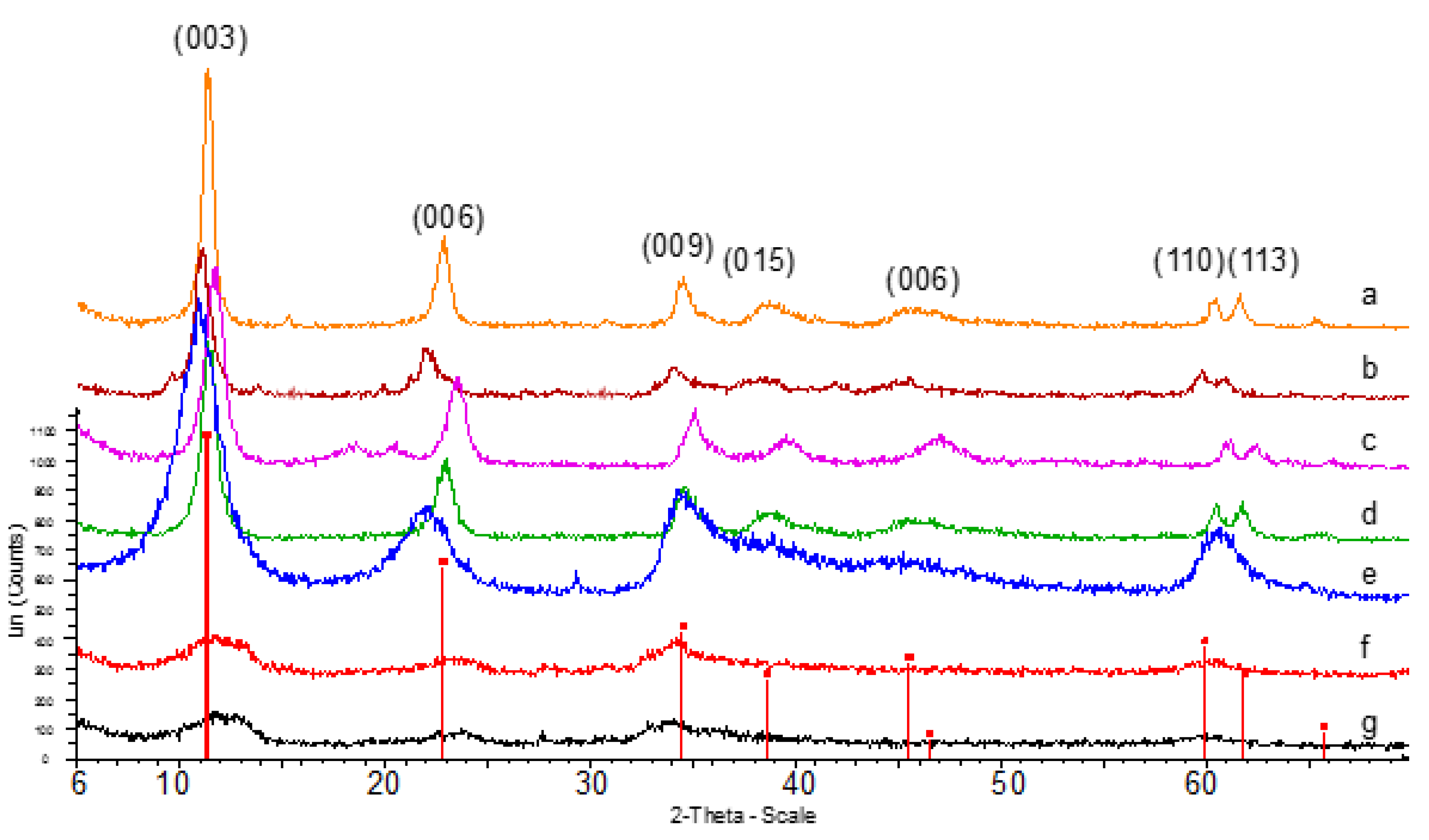
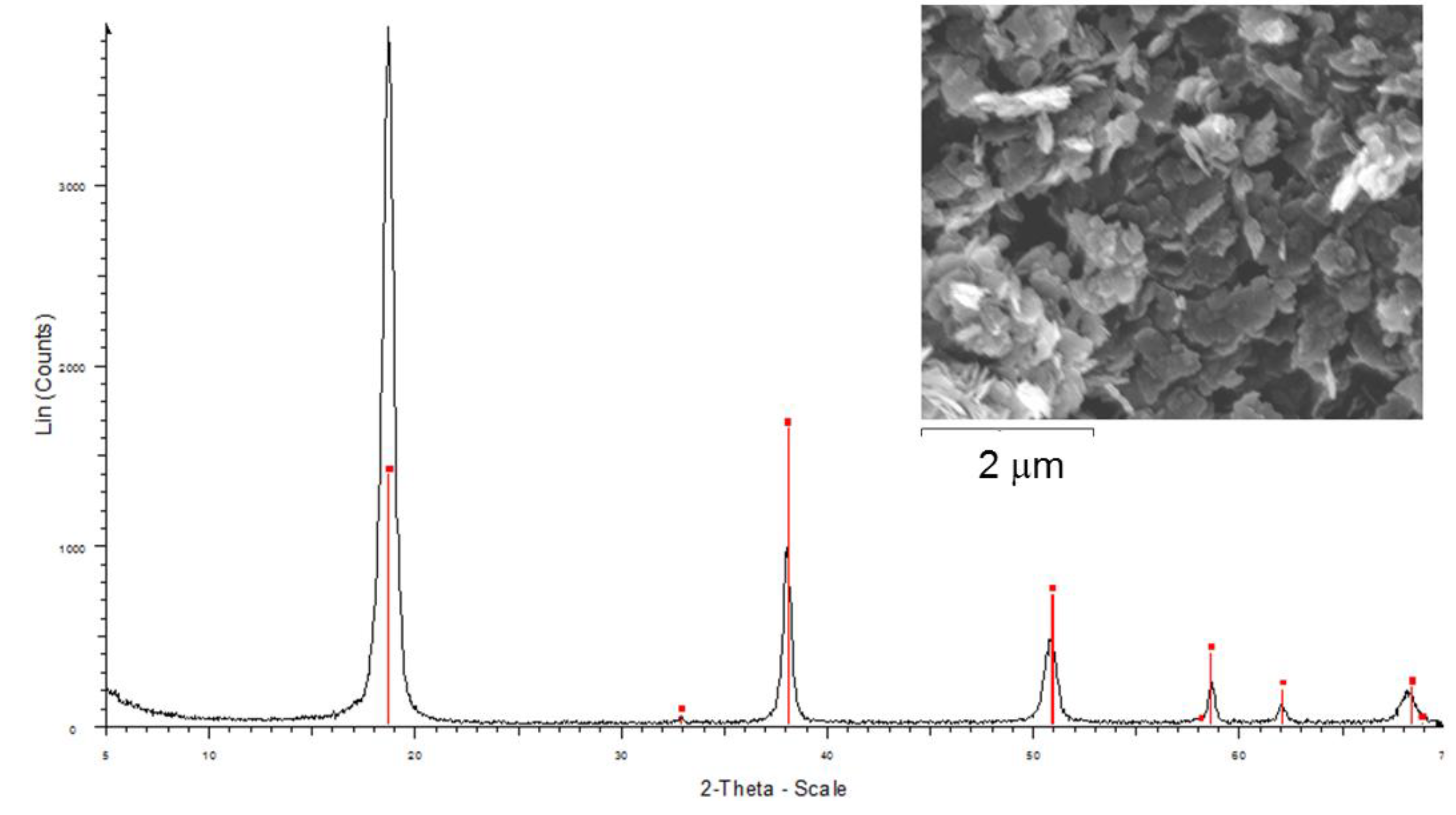
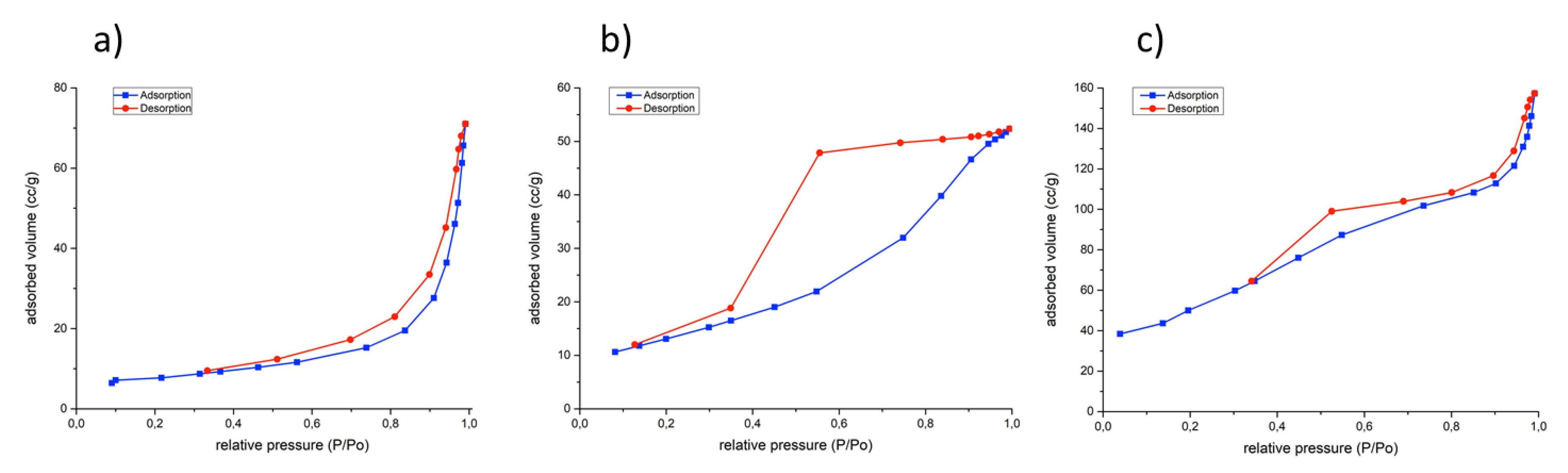

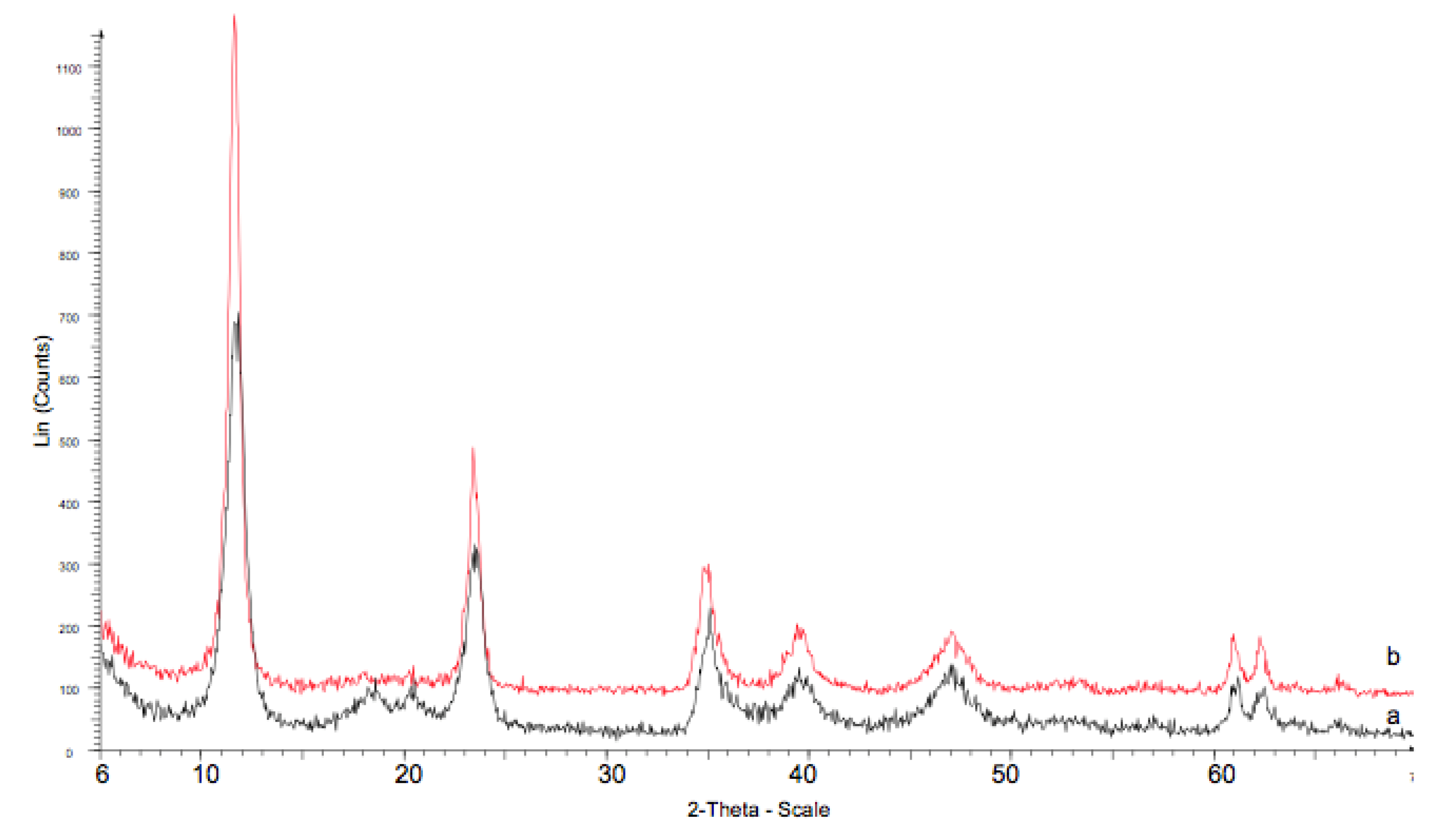
| Catalysts | Crystalline Phases (XRD) | a (nm) | c (nm) | FWHM (003) (°) | Crystallite Size (nm) (XRD) a | B.E.T. Surface Area (m2/g) |
|---|---|---|---|---|---|---|
| HTMgAl1 | Hydrotalcite | 0.306 | 2.340 | 0.562 | 39.9 | 35 |
| HTMgAl2 | Hydrotalcite | 0.309 | 2.405 | 0.766 | 35.0 | 57 |
| HTMgAl3 | Hydrotalcite | 0.305 | 2.425 | 0.685 | 32.8 | 48 |
| HTNiMgAl1 | Hydrotalcite | 0.305 | 2.321 | 0.805 | 25.8 | 37 |
| HTNiMgAl2 | Hydrotalcite | 0.304 | 2.375 | 2.051 | 5.5 | 58 |
| HTCuZnCr | Hydrotalcite | 0.308 | 2.341 | 2.618 | - | 151 |
| HTCuCr | Hydrotalcite | 0.309 | 2.309 | 1.412 | - | 88 |
| cHTMgAl1 | MgO | - | - | - | 3.1 | 220 |
| cHTNiMgAl1 | MgO | - | - | - | 5.4 | 201 |
| MgO | MgO | - | - | - | 16.9 | 104 |
| B | Mg(OH)2 | - | - | - | 14.6 | 42 |
| Catalysts | Mg/Al | Ni/Al | Ni/Mg | Cu/Cr | Cu/Zn | Zn/Cr |
|---|---|---|---|---|---|---|
| HTMgAl1 | 7.30 (9) | - | - | - | - | - |
| HTMgAl2 | 8.82 (9) | - | - | - | - | - |
| HTMgAl3 | 2.91 (3) | - | - | - | - | - |
| HTNiMgAl1 | 0.19 (0.24) | 2.34 (2.4) | 9.58 (10) | - | - | - |
| HTNiMgAl2 | 2.31 (2.4) | 0.59 (0.6) | 0.26 (0.25) | - | - | - |
| HTCuZnCr | - | - | - | 0.87 (1) | 0.99 (1) | 0.97 (1) |
| HTCuCr | - | - | - | 3.94 (4) | - | - |
| Catalysts | Conversion (%) | Selectivity to c9, t11-CLA (%) | Selectivity to t10, c12-CLA (%) | Selectivity to t9, t11-CLA (%) | Other Products (%) |
|---|---|---|---|---|---|
| HTMgAl1 | 92 | 0.1 | 0.3 | 3.5 | 96.1 |
| HTMgAl2 | 87 | 2.4 | 2.5 | 8.7 | 86.4 |
| HTMgAl3 | 97 | 5.5 | 5.2 | 27.7 | 61.6 |
| HTNiMgAl1 | 73 | 0.3 | 0.3 | 2.9 | 96.5 |
| HTNiMgAl2 | 81 | 8.1 | 7.5 | 13.2 | 71.2 |
| HTCuZnCr | 84 | 0.4 | 0.7 | 1.2 | 97.7 |
| HTCuCr | 97 | - | - | - | 100 |
| cHTMgAl1 | 94 | 0.5 | 0.5 | 1.4 | 97.6 |
| cHTNiMgAl1 | 78 | 0.2 | 0.1 | 1.3 | 98.4 |
| MgO | 98 | 0 | 0 | 3.2 | 96.8 |
| B | 100 | 0 | 0 | 0 | 100 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardó, X.; Salagre, P.; Cesteros, Y. Layered Double Hydroxides for the Catalytic Isomerization of Linoleic Acid to Conjugated Linoleic Acids (CLAs). ChemEngineering 2019, 3, 30. https://doi.org/10.3390/chemengineering3010030
Cardó X, Salagre P, Cesteros Y. Layered Double Hydroxides for the Catalytic Isomerization of Linoleic Acid to Conjugated Linoleic Acids (CLAs). ChemEngineering. 2019; 3(1):30. https://doi.org/10.3390/chemengineering3010030
Chicago/Turabian StyleCardó, Xavier, Pilar Salagre, and Yolanda Cesteros. 2019. "Layered Double Hydroxides for the Catalytic Isomerization of Linoleic Acid to Conjugated Linoleic Acids (CLAs)" ChemEngineering 3, no. 1: 30. https://doi.org/10.3390/chemengineering3010030
APA StyleCardó, X., Salagre, P., & Cesteros, Y. (2019). Layered Double Hydroxides for the Catalytic Isomerization of Linoleic Acid to Conjugated Linoleic Acids (CLAs). ChemEngineering, 3(1), 30. https://doi.org/10.3390/chemengineering3010030