Transition Metal–Nitrogen–Carbon (M–N–C) Catalysts for Oxygen Reduction Reaction. Insights on Synthesis and Performance in Polymer Electrolyte Fuel Cells
Abstract
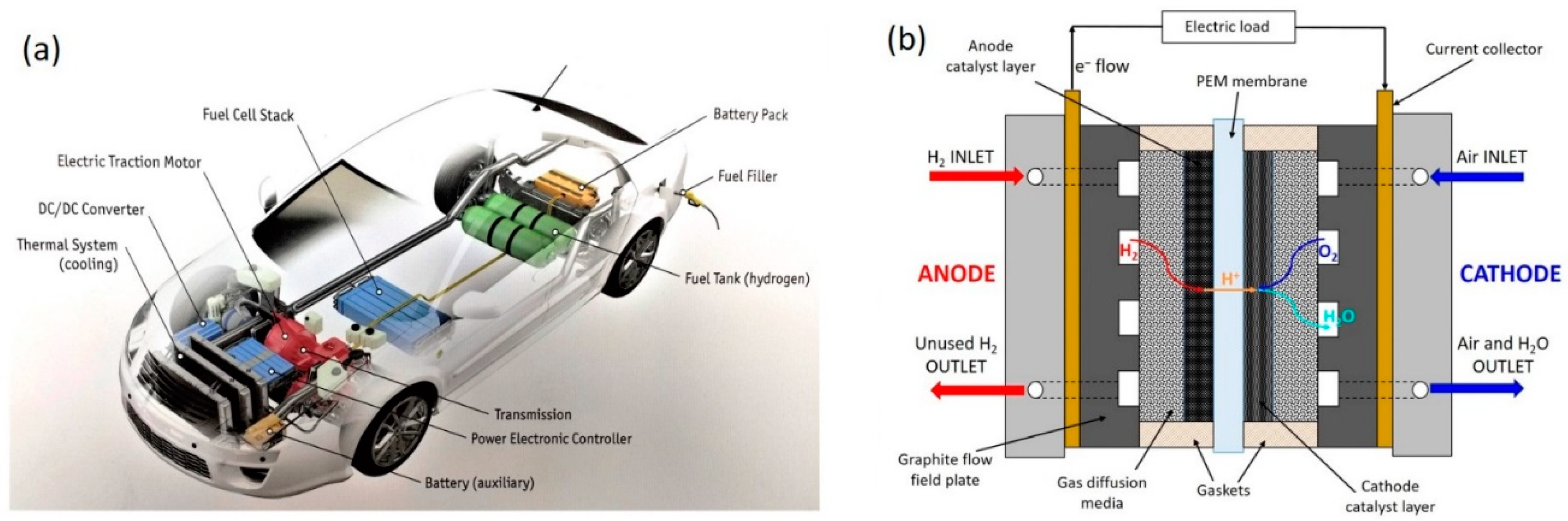
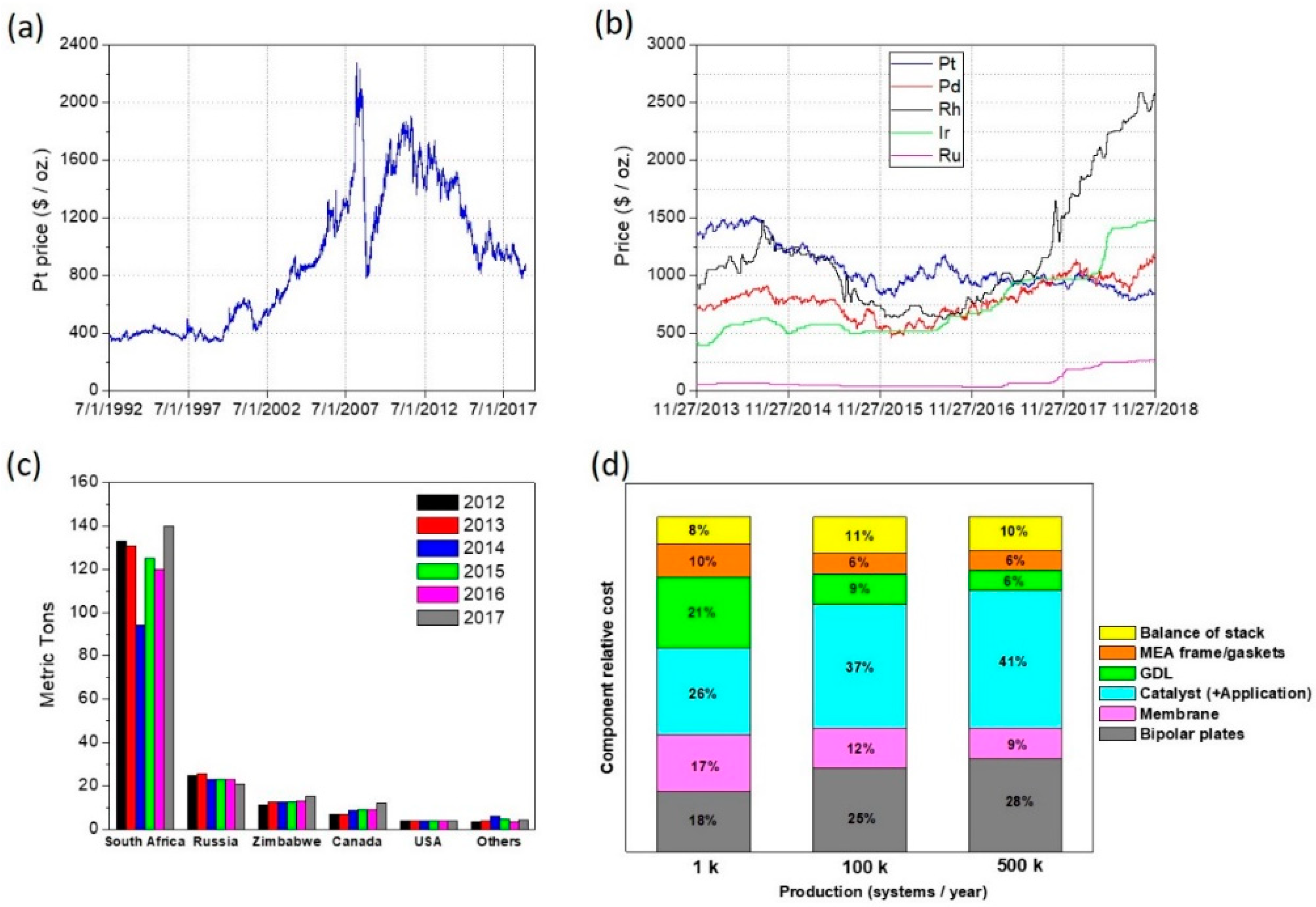
1. Introduction
2. Different Approaches for M–N–C Catalysts Synthesis
- Method 1: catalysts derived from carbon support and nitrogen containing molecule (Section 2.1);
- Method 2: catalysts derived from nitrogen containing polymer (Section 2.2);
- Method 3: catalysts derived from silica template and organic precursors (Hard Template Method) (Section 2.3);
- Method 4: catalysts derived from metal organic frameworks (MOFs) (Section 2.4);
- Method 5: catalysts derived from MOFs and additional precursors (Section 2.5).
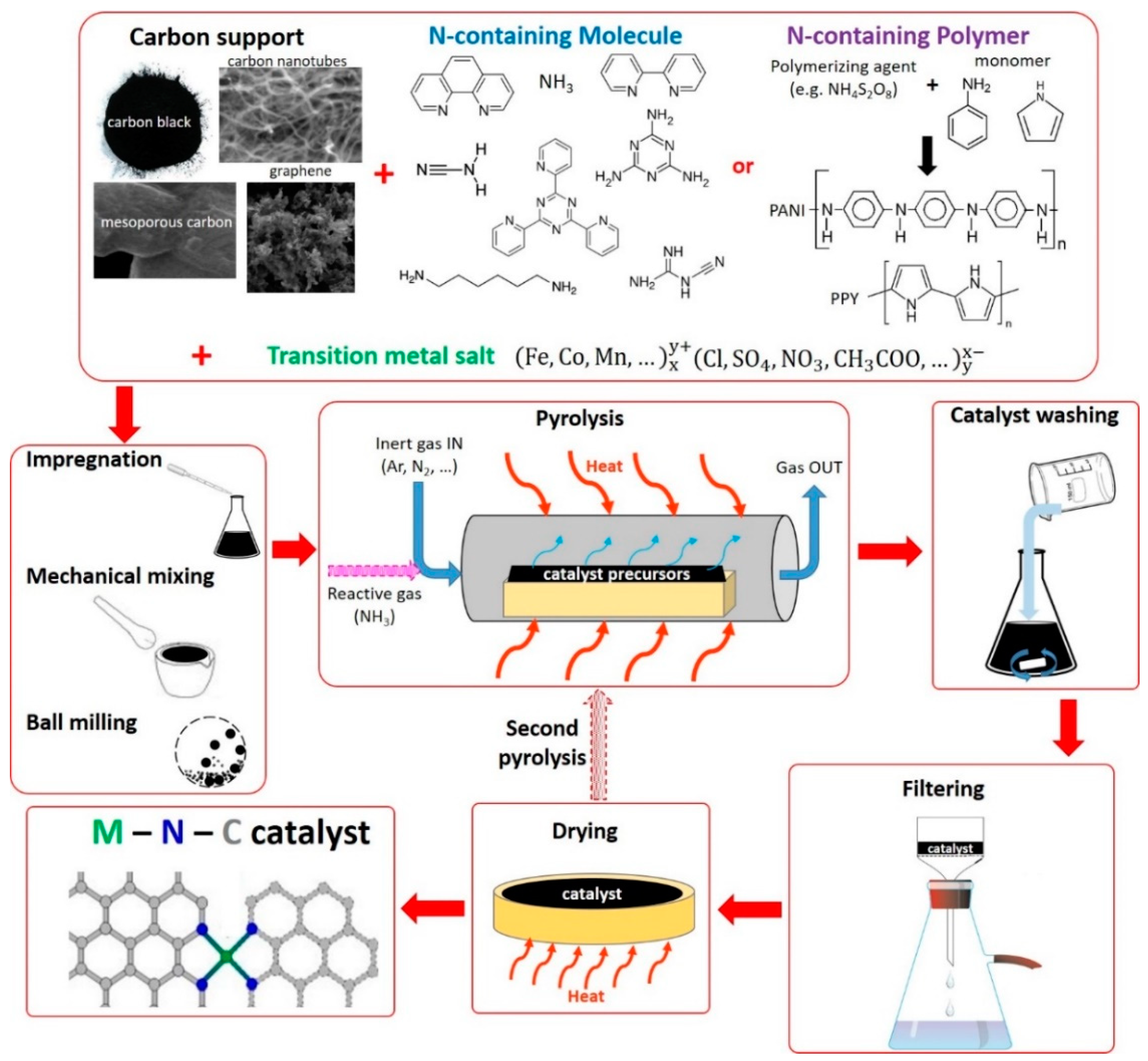
2.1. Method 1: Catalysts Derived from Carbon Support and Nitrogen Containing Molecule
2.2. Method 2: Catalysts Derived from Nitrogen Containing Polymer
- -
- -
- -
- electroconductive polymers.
- -
- -
- using the electroconductive polymer as N (and C) precursor material to prepare M–N–C catalysts via a high-temperature heat treatment, as described in Section 2.1 [104,105].
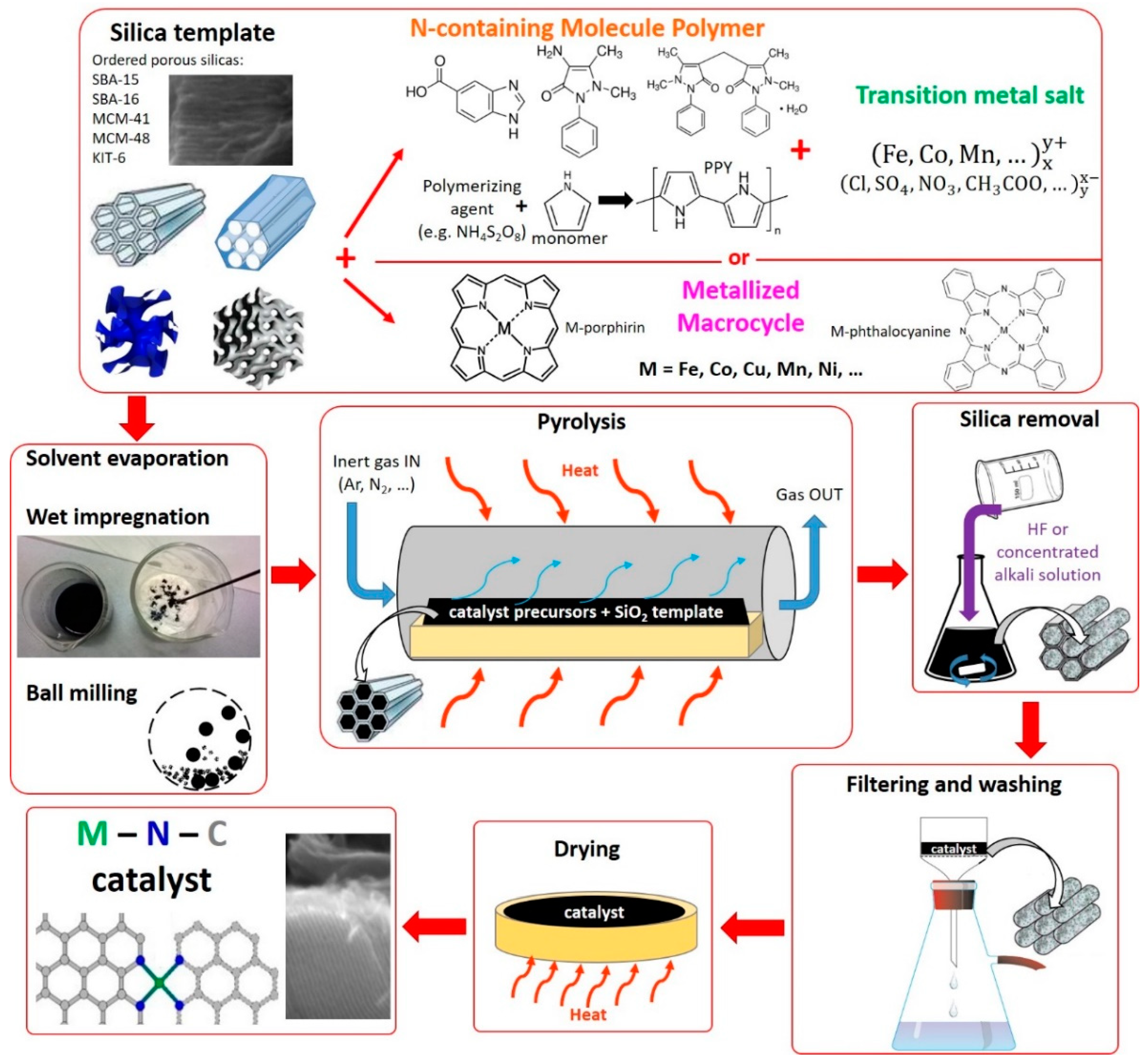
2.3. Method 3: Catalysts Derived from Silica Template and Organic Precursors (Hard Template Method)
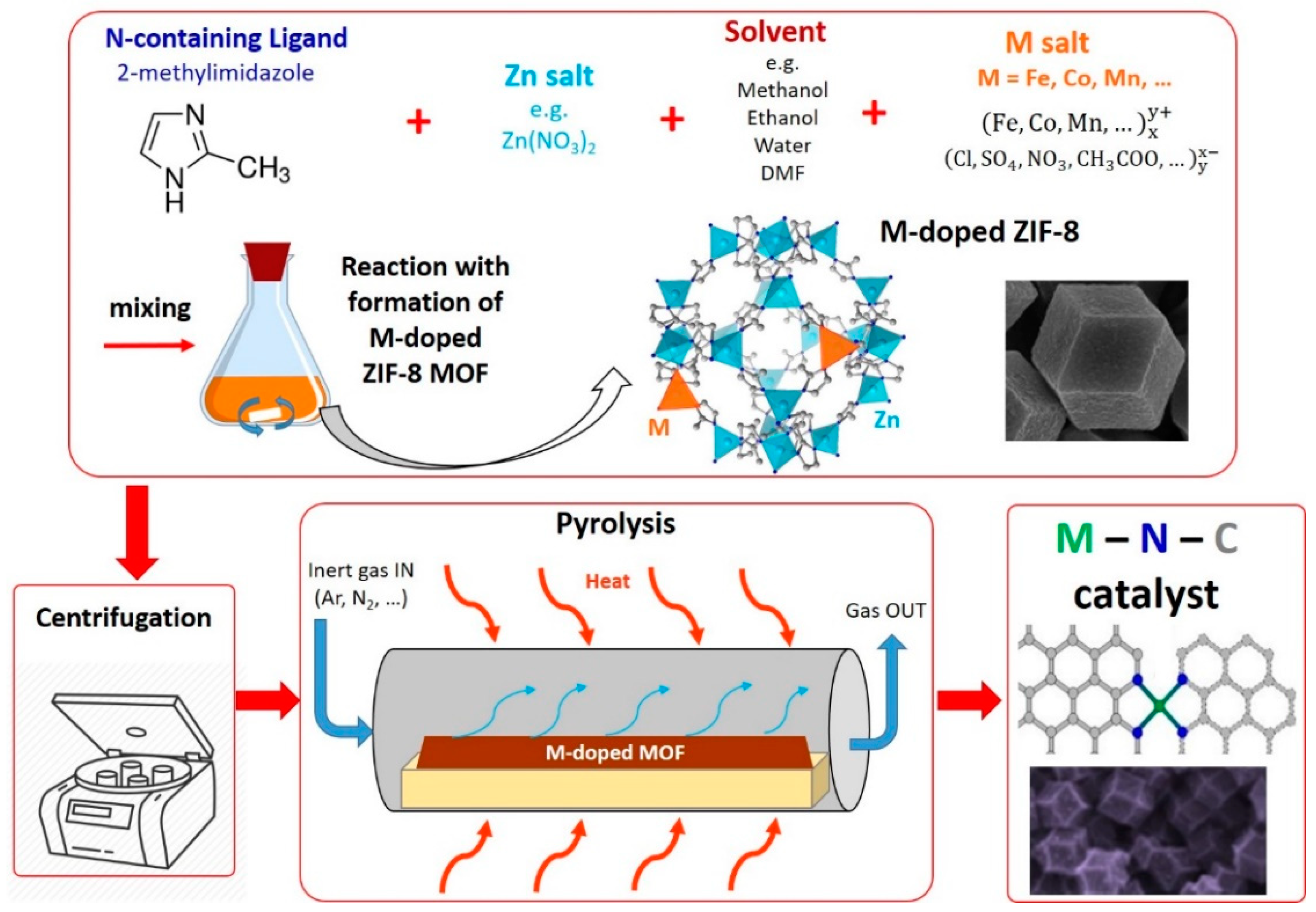
2.4. Method 4: Catalysts Derived from Metal Organic Frameworks (MOFs)
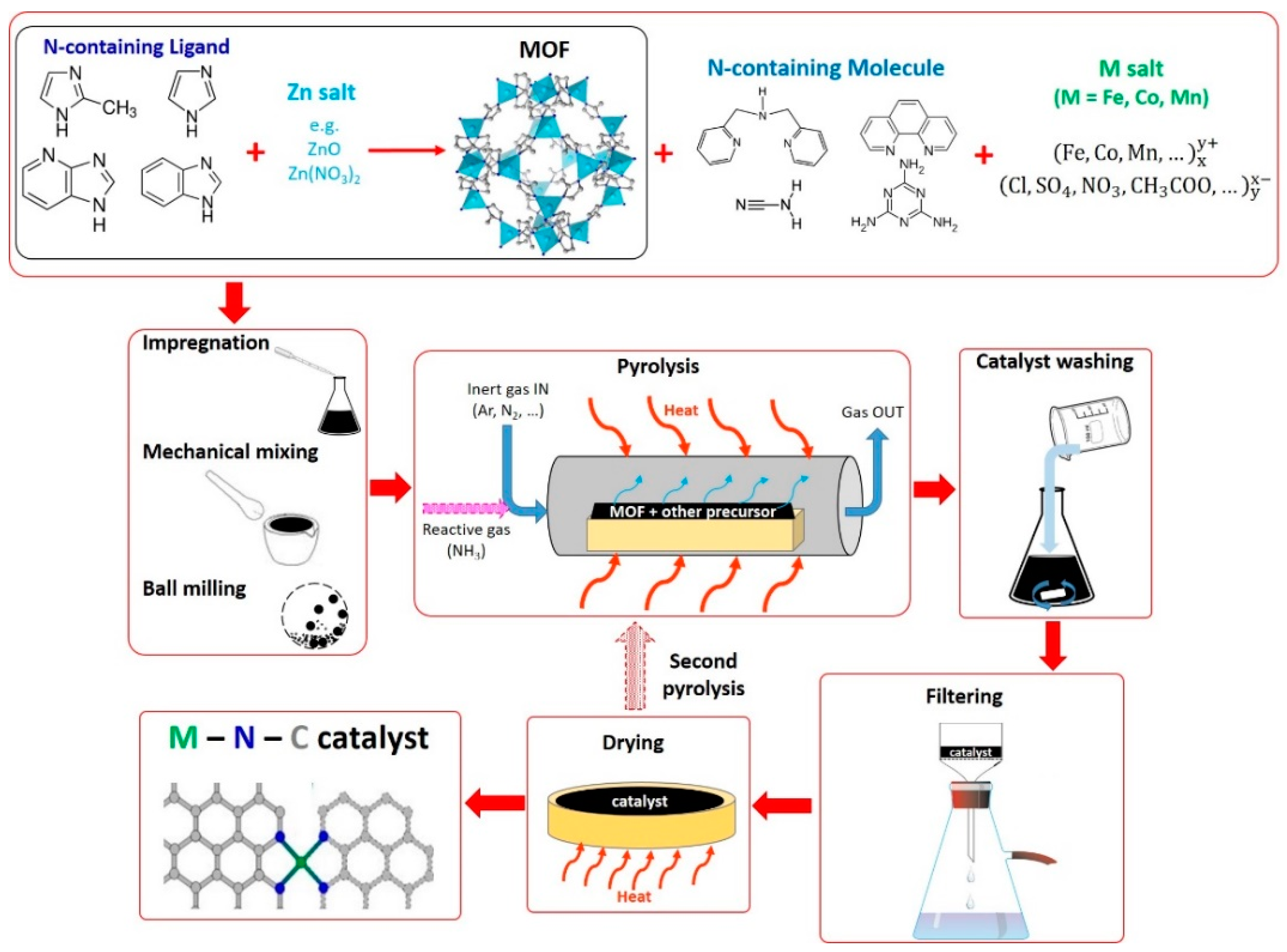
2.5. Method 5: Catalysts Derived from MOFs and Additional Precursors
3. Catalysts Performance Analysis and Comparison
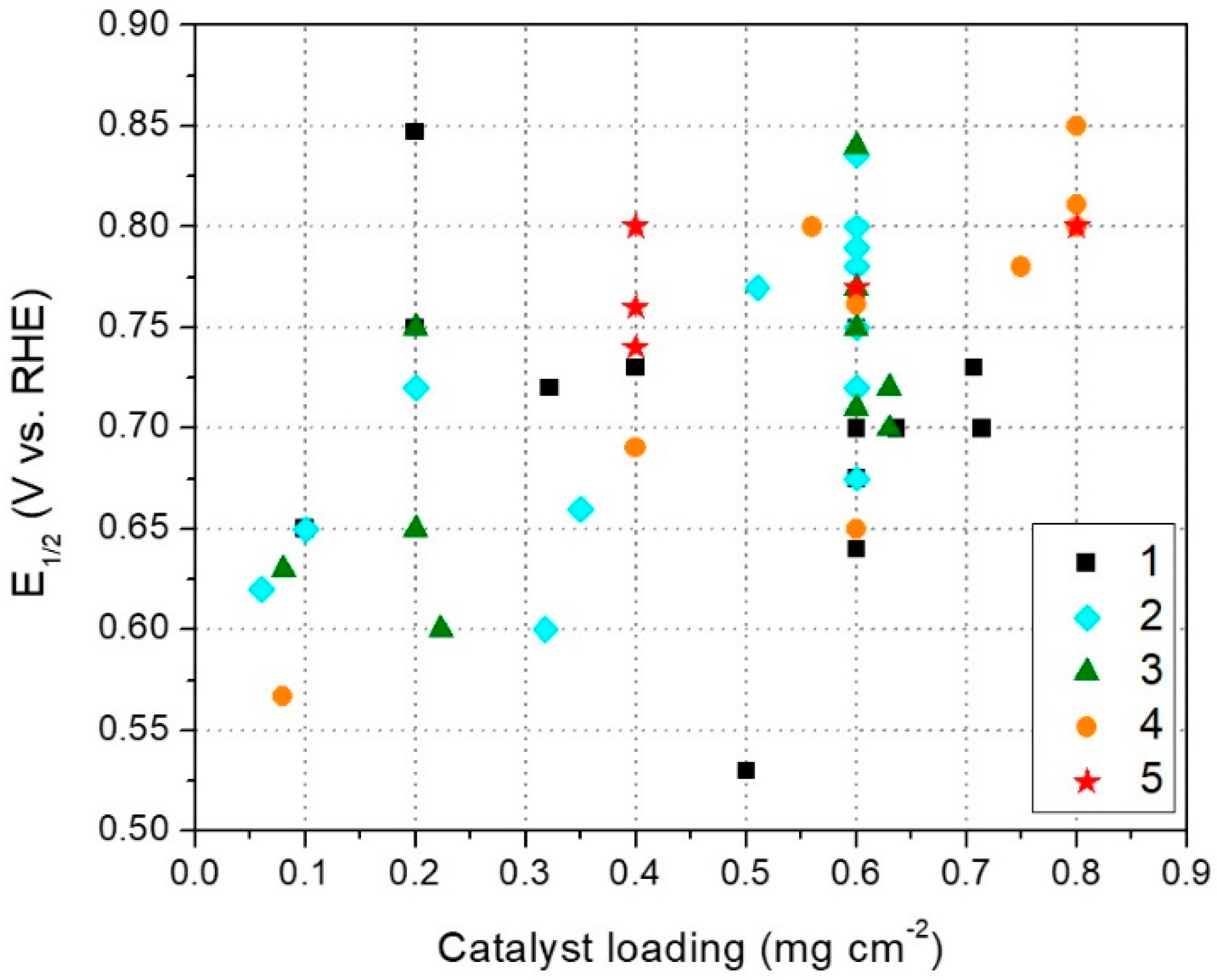
3.1. RDE Performance
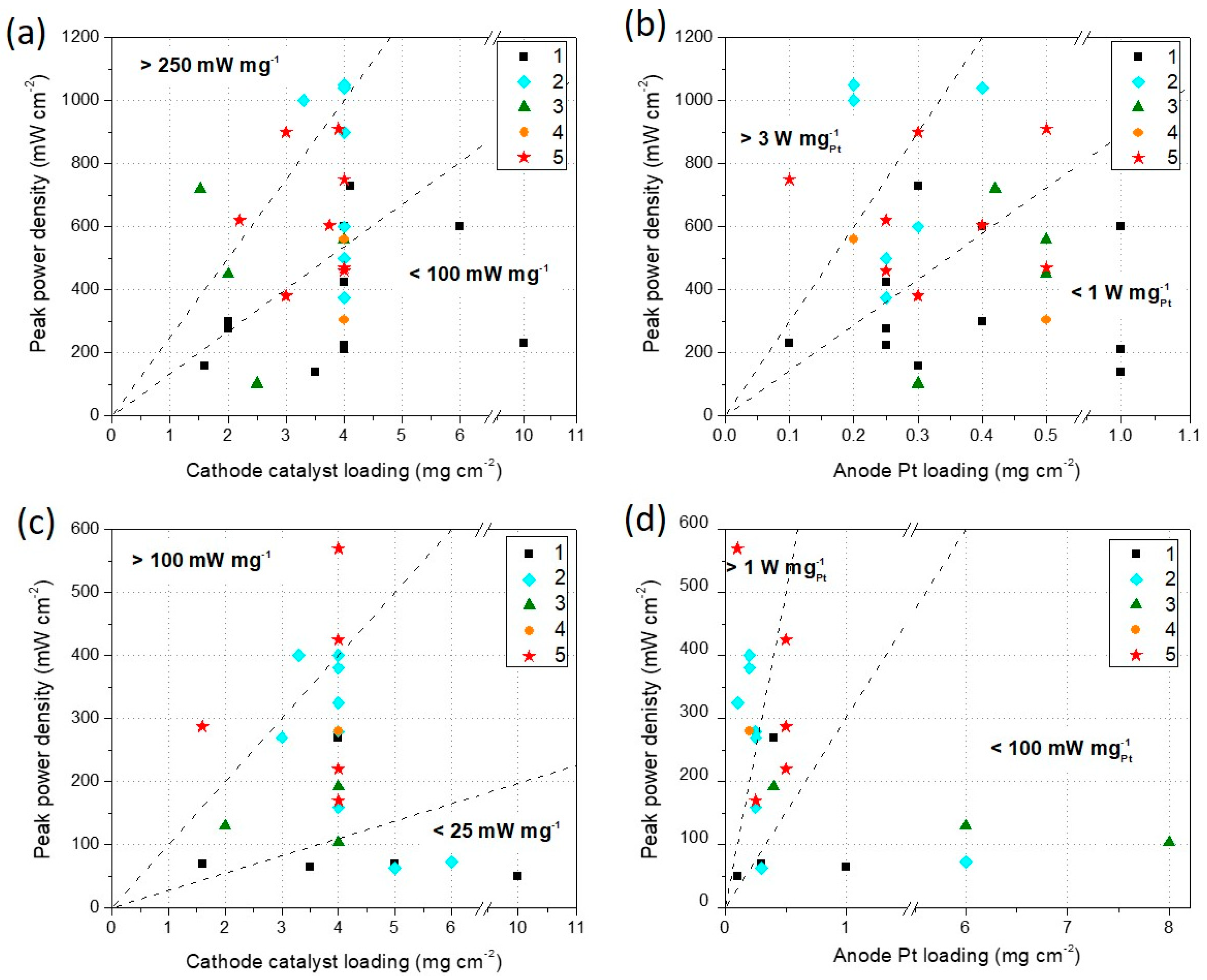
3.2. H2/O2 and H2/Air PEFC Performance
4. Conclusions and Perspectives
- Method 1: catalysts derived from carbon support and nitrogen containing molecule;
- Method 2: catalysts derived from nitrogen containing polymer;
- Method 3: catalysts derived from silica template and organic precursors (Hard Template Method);
- Method 4: catalysts derived from metal organic frameworks (MOFs);
- Method 5: catalysts derived from MOFs and additional precursors.
Funding
Conflicts of Interest
Appendix A
| Synthesis Method | E1/2 (V vs. RHE) | Catalyst Loading on RDE Tip (mg·cm−2) | Reference | Durability Test Done |
|---|---|---|---|---|
| 1 | 0.68 | 0.60 | [186] | X |
| 1 | 0.70 | 0.64 | [46] | |
| 1 | 0.73 | 0.40 | [187] | |
| 1 | 0.85 | 0.20 | [84] | X |
| 1 | 0.75 | 0.20 | [188] | X |
| 1 | 0.75 | 0.20 | [189] | |
| 1 | 0.64 | 0.60 | [73] | |
| 1 | 0.65 | 0.10 | [72] | |
| 1 | 0.53 | 0.50 | [47] | X |
| 1 | 0.70 | 0.71 | [79] | |
| 1 | 0.73 | 0.71 | [190] | |
| 1 | 0.72 | 0.32 | [191] | |
| 1 | 0.75 | 0.60 | [77] | |
| 1 | 0.70 | 0.60 | [192] | X |
| 2 | 0.65 | 0.10 | [116] | |
| 2 | 0.60 | 0.32 | [50] | |
| 2 | 0.77 | 0.51 | [36] | X |
| 2 | 0.72 | 0.20 | [94] | |
| 2 | 0.80 | 0.60 | [105] | X |
| 2 | 0.80 | 0.60 | [193] | X |
| 2 | 0.75 | 0.60 | [106] | X |
| 2 | 0.68 | 0.60 | [194] | |
| 2 | 0.80 | 0.60 | [89] | |
| 2 | 0.78 | 0.60 | [91] | |
| 2 | 0.62 | 0.06 | [195] | |
| 2 | 0.72 | 0.60 | [111] | |
| 2 | 0.79 | 0.60 | [108] | X |
| 2 | 0.84 | 0.60 | [100] | |
| 2 | 0.66 | 0.35 | [113] | |
| 3 | 0.77 | 0.60 | [121] | X |
| 3 | 0.65 | 0.20 | [121] | X |
| 3 | 0.60 | 0.22 | [127] | |
| 3 | 0.84 | 0.60 | [123] | X |
| 3 | 0.70 | 0.63 | [22] | X |
| 3 | 0.72 | 0.63 | [126] | |
| 3 | 0.75 | 0.20 | [196] | |
| 3 | 0.71 | 0.60 | [197] | |
| 3 | 0.75 | 0.60 | [128] | |
| 3 | 0.63 | 0.08 | [124] | X |
| 4 | 0.57 | 0.08 | [152] | |
| 4 | 0.80 | 0.56 | [150] | X |
| 4 | 0.85 | 0.80 | [138] | X |
| 4 | 0.81 | 0.80 | [145] | |
| 4 | 0.76 | 0.60 | [147] | X |
| 4 | 0.65 | 0.60 | [149] | |
| 4 | 0.69 | 0.40 | [141] | |
| 4 | 0.78 | 0.75 | [148] | X |
| 4 | 0.80 | 0.80 | [151] | X |
| 5 | 0.80 | 0.80 | [161] | X |
| 5 | 0.80 | 0.40 | [156] | X |
| 5 | 0.76 | 0.40 | [160] | X |
| 5 | 0.77 | 0.60 | [153] | X |
| 5 | 0.74 | 0.40 | [154] |
| Synthesis Method | Maximum Power Density H2/O2 (mW·cm−2) | Maximum Power Density H2/Air (mW·cm−2) | Cathode Catalyst Loading (mg·cm−2) | Anode Pt Loading (mg·cm−2) | Reference | Durability Test Done |
|---|---|---|---|---|---|---|
| 1 | 730 | 4.1 | 0.30 | [187] | ||
| 1 | 600 | - | 6.0 | 1.00 | [84] | |
| 1 | 600 | 270 | 4.0 | 0.40 | [189] | X |
| 1 | 230 | 50 | 10.0 | 0.10 | [198] | X |
| 1 | 160 | 70 | 1.6 | 0.30 | [73] | |
| 1 | 300 | - | 2.0 | 0.40 | [80] | X |
| 1 | 210 | - | 4.0 | 1.00 | [79] | X |
| 1 | 225 | - | 4.0 | 0.25 | [190] | X |
| 1 | 140 | 65 | 3.5 | 1.00 | [191] | X |
| 1 | 425 | - | 4.0 | 0.25 | [77] | X |
| 1 | 275 | - | 2.0 | 0.25 | [86] | X |
| 1 | - | 70 | 5.0 | 0.30 | [72] | |
| 2 | 1000 | 400 | 3.3 | 0.20 | [94] | X |
| 2 | 500 | 4.0 | 0.25 | [105] | X | |
| 2 | 900 | 400 | 4.0 | 2.00 | [193] | |
| 2 | 375 | 4.0 | 0.25 | [106] | X | |
| 2 | 600 | 4.0 | 0.30 | [111] | ||
| 2 | 1050 | 380 | 4.0 | 0.20 | [108] | X |
| 2 | 1040 | 4.0 | 0.40 | [100] | X | |
| 2 | 62.5 | 5.0 | 0.30 | [116] | ||
| 2 | 72.5 | 6.0 | 6.00 | [103] | X | |
| 2 | 325 | 4.0 | 0.10 | [36] | X | |
| 2 | 160 | 4.0 | 0.25 | [199] | X | |
| 2 | 270 | 3.0 | 0.25 | [194] | X | |
| 2 | 280 | 4.0 | 0.25 | [89] | X | |
| 3 | 560 | 4.0 | 0.50 | [121] | ||
| 3 | 450 | 2.0 | 0.50 | [200] | X | |
| 3 | 720 | 1.5 | 0.42 | [123] | X | |
| 3 | 100 | 2.5 | 0.30 | [22] | ||
| 3 | 105 | 2.5 | 0.30 | [126] | ||
| 3 | 104 | 4.0 | 8.00 | [127] | X | |
| 3 | 130 | 2.0 | 6.00 | [201] | X | |
| 3 | 192 | 4.0 | 0.40 | [128] | ||
| 4 | 305 | 4.0 | 0.50 | [145] | ||
| 4 | 560 | 280 | 4.0 | 0.20 | [151] | X |
| 5 | 460 | 170 | 4.0 | 0.25 | [161] | X |
| 5 | 900 | 3.0 | 0.30 | [156] | ||
| 5 | 603 | 3.8 | 0.40 | [159] | ||
| 5 | 750 | 570 | 4.0 | 0.10 | [162] | X |
| 5 | 910 | 3.9 | 0.50 | [158] | X | |
| 5 | 620 | 2.2 | 0.25 | [160] | X | |
| 5 | 380 | 3.0 | 0.30 | [153] | X | |
| 5 | 470 | 220 | 4.0 | 0.5 | [202] | X |
| 5 | - | 425 | 4.9 | 0.5 | [155] | X |
| 5 | - | 287 | 1.6 | 0.5 | [154] | X |
References
- Espeland, E.K.; Kettenring, K.M. Strategic plant choices can alleviate climate change impacts: A review. J. Environ. Manag. 2018, 222, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Krajačić, G.; Duić, N.; Zmijarević, Z.; Mathiesen, B.V.; Vučinić, A.A.; Da Graa Carvalho, M. Planning for a 100% independent energy system based on smart energy storage for integration of renewables and CO2 emissions reduction. Appl. Therm. Eng. 2011, 31, 2073–2083. [Google Scholar] [CrossRef]
- Zhu, Y.P.; Guo, C.; Zheng, Y.; Qiao, S.-Z. Surface and Interface Engineering of Noble-Metal-Free Electrocatalysts for Efficient Energy Conversion Processes. Acc. Chem. Res. 2017, 50, 915–923. [Google Scholar] [CrossRef] [PubMed]
- Coralli, A.; Sarruf, B.J.M.; de Miranda, P.E.V.; Osmieri, L.; Specchia, S.; Minh, N.Q. Fuel Cells. In Science and Engineering of Hydrogen-Based Energy Technologies; de Miranda, P.E.V., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; ISBN 9780128142516. [Google Scholar]
- Lemke, C.; Grueger, F.; Arnhold, O. MELY: Market Model for Water Electrolysis—Electrolysis’ Economic Potential given its Technological Feasibility. Energy Procedia 2015, 73, 59–68. [Google Scholar] [CrossRef]
- Speers, P. Hydrogen Mobility Europe (H2ME): Vehicle and Hydrogen Refuelling Station Deployment Results. World Electr. Veh. J. 2018, 1. [Google Scholar] [CrossRef]
- EG&G Technical Services, Inc. Fuel Cell Handbook, 7th ed.; U.S. Department of Energy: Morgantown, WV, USA, 2004.
- Vignarooban, K.; Lin, J.; Arvay, A.; Kolli, S.; Kruusenberg, I.; Tammeveski, K.; Munukutla, L.; Kannan, A.M. Nano-electrocatalyst materials for low temperature fuel cells: A review. Chin. J. Catal. 2015, 36, 458–472. [Google Scholar] [CrossRef]
- Durst, J.; Simon, C.; Hasche, F.; Gasteiger, H.A. Hydrogen Oxidation and Evolution Reaction Kinetics on Carbon Supported Pt, Ir, Rh, and Pd Electrocatalysts in Acidic Media. J. Electrochem. Soc. 2014, 162, F190–F203. [Google Scholar] [CrossRef]
- Sebastián, D.; Serov, A.; Artyushkova, K.; Gordon, J.; Atanassov, P.; Aricò, A.S.; Baglio, V. High Performance and Cost-Effective Direct Methanol Fuel Cells: Fe-N-C Methanol-Tolerant Oxygen Reduction Reaction Catalysts. ChemSusChem 2016, 9, 1986–1995. [Google Scholar] [CrossRef]
- Johnson Matthey. Available online: http://www.platinum.matthey.com/prices (accessed on 27 November 2018).
- US Geological Survey © Statista 2018. Available online: https://www.statista.com/statistics/273645/global-mine-production-of-platinum/ (accessed on 27 November 2018).
- Kongkanand, A.; Mathias, M.F. The Priority and Challenge of High-Power Performance of Low-Platinum Proton-Exchange Membrane Fuel Cells. J. Phys. Chem. Lett. 2016, 7, 1127–1137. [Google Scholar] [CrossRef]
- Banham, D.; Ye, S. Current Status and Future Development of Catalyst Materials and Catalyst Layers for Proton Exchange Membrane Fuel Cells: An Industrial Perspective. ACS Energy Lett. 2017, 2, 629–638. [Google Scholar] [CrossRef]
- Jaouen, F.; Proietti, E.; Lefèvre, M.; Chenitz, R.; Dodelet, J.-P.; Wu, G.; Chung, H.T.; Johnston, C.M.; Zelenay, P. Recent advances in non-precious metal catalysis for oxygen-reduction reaction in polymer electrolyte fuel cells. Energy Environ. Sci. 2011, 4, 114–130. [Google Scholar] [CrossRef]
- Wilson, A.; Kleen, G.; Papageorgopoulos, D. DOE Hydrogen and Fuel Cells Program Record #17007—Fuel Cell System Cost; 2017. Available online: https://www.hydrogen.energy.gov/pdfs/17007_fuel_cell_system_cost_2017.pdf (accessed on 2 February 2019).
- Li, J.; Jaouen, F. Structure and activity of metal-centered coordination sites in pyrolyzed metal–nitrogen–carbon catalysts for the electrochemical reduction of O2. Curr. Opin. Electrochem. 2018. [Google Scholar] [CrossRef]
- Kramm, U.I.; Lefèvre, M.; Larouche, N.; Schmeisser, D.; Dodelet, J.P. Correlations between mass activity and physicochemical properties of Fe/N/C catalysts for the ORR in PEM fuel cell via 57Fe Mössbauer spectroscopy and other techniques. J. Am. Chem. Soc. 2014, 136, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Jia, Q.; Ramaswamy, N.; Hafiz, H.; Tylus, U.; Strickland, K.; Wu, G.; Barbiellini, B.; Bansil, A.; Holby, E.F.; Zelenay, P.; et al. Experimental Observation of Redox-Induced Fe-N Switching Behavior as a Determinant Role for Oxygen Reduction Activity. ACS Nano 2015, 9, 12496–12505. [Google Scholar] [CrossRef] [PubMed]
- Jasinski, R. A New Fuel Cell Cathode Catalyst. Nature 1964, 201, 1212–1213. [Google Scholar] [CrossRef]
- Kiros, Y. Metal Porphyrins for Oxygen Reduction in PEMFC. Int. J. Electrochem. Sci. 2007, 2, 285–300. [Google Scholar]
- Osmieri, L.; Escudero-Cid, R.; Armandi, M.; Ocón, P.; Monteverde Videla, A.H.A.; Specchia, S. Effects of using two transition metals in the synthesis of non-noble electrocatalysts for oxygen reduction reaction in direct methanol fuel cell. Electrochim. Acta 2018, 266, 220–232. [Google Scholar] [CrossRef]
- Osmieri, L.; Monteverde Videla, A.H.A.; Armandi, M.; Specchia, S. Influence of different transition metals on the properties of Me-N-C (Me = Fe, Co, Cu, Zn) catalysts synthesized using SBA-15 as tubular nano-silica reactor for oxygen reduction reaction. Int. J. Hydrogen Energy 2016, 41, 22570–22588. [Google Scholar] [CrossRef]
- Othman, R.; Dicks, A.L.; Zhu, Z. Non precious metal catalysts for the PEM fuel cell cathode. Int. J. Hydrogen Energy 2012, 37, 357–372. [Google Scholar] [CrossRef]
- Chen, Z.; Higgins, D.; Yu, A.; Zhang, L.; Zhang, J. A review on non-precious metal electrocatalysts for PEM fuel cells. Energy Environ. Sci. 2011, 4, 3167–3192. [Google Scholar] [CrossRef]
- Monteverde Videla, A.H.A.; Osmieri, L.; Specchia, S. Non-noble metal (NNM) catalysts for fuel cells: Tuning the activity by a rational step-by-step single variable evolution. In Electrochemistry of N4 Macrocyclic Metal Complexes: Volume 1: Energy, 2nd ed.; Zagal, J.H., Bedioui, F., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; ISBN 9783319311722. [Google Scholar]
- Xu, J.; Zhao, Y.; Shen, C.; Guan, L. Sulfur- and nitrogen-doped, ferrocene-derived mesoporous carbons with efficient electrochemical reduction of oxygen. ACS Appl. Mater. Interfaces 2013, 5, 12594–12601. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Cao, R.; Cho, J.; Wu, G. Nanocarbon electrocatalysts for oxygen reduction in alkaline media for advanced energy conversion and storage. Adv. Energy Mater. 2014, 4. [Google Scholar] [CrossRef]
- Osmieri, L.; Escudero-Cid, R.; Monteverde Videla, A.H.A.; Ocón, P.; Specchia, S. Application of a non-noble Fe-N-C catalyst for oxygen reduction reaction in an alkaline direct ethanol fuel cell. Renew. Energy 2017, 115, 226–237. [Google Scholar] [CrossRef]
- Wei, W.; Tao, Y.; Lv, W.; Su, F.-Y.; Ke, L.; Li, J.; Wang, D.-W.; Li, B.; Kang, F.; Yang, Q.-H. Unusual High Oxygen Reduction Performance in All-Carbon Electrocatalysts. Sci. Rep. 2014, 4, 6289. [Google Scholar] [CrossRef] [PubMed]
- Osmieri, L.; Zafferoni, C.; Wang, L.; MonteverdeVidela, A.H.A.; Lavacchi, A.; Specchia, S. Polypyrrole-Derived Fe-Co-N-C Catalyst for the Oxygen Reduction Reaction: Performance in Alkaline Hydrogen and Ethanol Fuel Cells. ChemElectroChem 2018. [Google Scholar] [CrossRef]
- Li, X.; Popov, B.N.; Kawahara, T.; Yanagi, H. Non-precious metal catalysts synthesized from precursors of carbon, nitrogen, and transition metal for oxygen reduction in alkaline fuel cells. J. Power Sources 2011, 196, 1717–1722. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, L.; Preuß, K.; Qiao, M.; Titirici, M.M.; Varcoe, J.; Cai, Q. Halloysite-derived nitrogen doped carbon electrocatalysts for anion exchange membrane fuel cells. J. Power Sources 2017, 372, 82–90. [Google Scholar] [CrossRef]
- Gottesfeld, S.; Dekel, D.R.; Page, M.; Bae, C.; Yan, Y.; Zelenay, P.; Kim, Y.S. Anion exchange membrane fuel cells: Current status and remaining challenges. J. Power Sources 2018, 375, 170–184. [Google Scholar] [CrossRef]
- Dekel, D.R. Review of cell performance in anion exchange membrane fuel cells. J. Power Sources 2018, 375, 158–169. [Google Scholar] [CrossRef]
- Peng, H.; Mo, Z.; Liao, S.; Liang, H.; Yang, L.; Luo, F.; Song, H.; Zhong, Y.; Zhang, B. High performance Fe- and N- Doped carbon catalyst with graphene structure for oxygen reduction. Sci. Rep. 2013, 3, 1–7. [Google Scholar] [CrossRef]
- Ramaswamy, N.; Mukerjee, S. Influence of inner- and outer-sphere electron transfer mechanisms during electrocatalysis of oxygen reduction in alkaline media. J. Phys. Chem. C 2011, 115, 18015–18026. [Google Scholar] [CrossRef]
- Ramaswamy, N.; Mukerjee, S. Fundamental Mechanistic Understanding of Electrocatalysis of Oxygen Reduction on Pt and Non-Pt Surfaces: Acid versus Alkaline Media. Adv. Phys. Chem. 2012, 2012, 1–17. [Google Scholar] [CrossRef]
- Trogadas, P.; Fuller, T.F.; Strasser, P. Carbon as catalyst and support for electrochemical energy conversion. Carbon 2014, 75, 5–42. [Google Scholar] [CrossRef]
- Shao, M.; Chang, Q.; Dodelet, J.-P.; Chenitz, R. Recent Advances in Electrocatalysts for Oxygen Reduction Reaction. Chem. Rev. 2016, 116, 3594–3657. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-J.; Wilkinson, D.P.; Zhang, J. Noncarbon support materials for polymer electrolyte membrane fuel cell electrocatalysts. Chem. Rev. 2011, 111, 7625–7651. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Ma, L.; Yadav, R.M.; Yang, Y.; Zhang, X.; Vajtai, R.; Lou, J.; Ajayan, P.M. Nitrogen-Doped Graphene with Pyridinic Dominance as a Highly Active and Stable Electrocatalyst for Oxygen Reduction. ACS Appl. Mater. Interfaces 2015, 7, 14763–14769. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Song, Y.; He, S.; Tjiu, W.W.; Pan, J.; Xia, Y.Y.; Liu, T. Nitrogen-doped graphene nanoribbons as efficient metal-free electrocatalysts for oxygen reduction. ACS Appl. Mater. Interfaces 2014, 6, 4214–4222. [Google Scholar] [CrossRef] [PubMed]
- Masa, J.; Zhao, A.; Wei, X.; Muhler, M.; Schuhmann, W. Metal-free catalysts for oxygen reduction in alkaline electrolytes: Influence of the presence of Co, Fe, Mn and Ni inclusions. Electrochim. Acta 2014, 128, 271–278. [Google Scholar] [CrossRef]
- Zhang, C.; Hao, R.; Liao, H.; Hou, Y. Synthesis of amino-functionalized graphene as metal-free catalyst and exploration of the roles of various nitrogen states in oxygen reduction reaction. Nano Energy 2013, 2, 88–97. [Google Scholar] [CrossRef]
- Osmieri, L.; Escudero-Cid, R.; Armandi, M.; Monteverde Videla, A.H.A.; Fierro, J.L.G.; Ocón, P.; Specchia, S. Fe-N/C catalysts for oxygen reduction reaction supported on different carbonaceous materials. Performance in acidic and alkaline direct alcohol fuel cells. Appl. Catal. B Environ. 2017, 205, 637–653. [Google Scholar] [CrossRef]
- Monteverde Videla, A.H.A.; Ban, S.; Specchia, S.; Zhang, L.; Zhang, J. Non-noble Fe–NX electrocatalysts supported on the reduced graphene oxide for oxygen reduction reaction. Carbon 2014, 76, 386–400. [Google Scholar] [CrossRef]
- Tran, T.N.; Song, M.Y.; Kang, T.H.; Samdani, J.; Park, H.Y.; Kim, H.; Jhung, S.H.; Yu, J.S. Iron Phosphide Incorporated into Iron-Treated Heteroatoms-Doped Porous Bio-Carbon as Efficient Electrocatalyst for the Oxygen Reduction Reaction. ChemElectroChem 2018, 5, 1944–1953. [Google Scholar] [CrossRef]
- Martínez Millán, W.; Toledano Thompson, T.; Arriaga, L.; Smit, M.A. Characterization of composite materials of electroconductive polymer and cobalt as electrocatalysts for the oxygen reduction reaction. Int. J. Hydrogen Energy 2009, 34, 694–702. [Google Scholar] [CrossRef]
- Osmieri, L.; Monteverde Videla, A.H.A.; Specchia, S. Optimization of a Fe-N-C electrocatalyst supported on ordered mesoporous carbon functionalized with polypyrrole for oxygen reduction reaction. Int. J. Hydrogen Energy 2016, 41, 19610–19628. [Google Scholar] [CrossRef]
- Jaouen, F.; Dodelet, J.-P. O2 Reduction Mechanism on Non-Noble Metal Catalysts for PEM Fuel Cells. Part I: Experimental Rates of O2 Electroreduction, H2O2 Electroreduction, and H2O2 Disproportionation. J. Phys. Chem. C 2009, 113, 15422–15432. [Google Scholar] [CrossRef]
- Qiao, J.; Xu, L.; Ding, L.; Zhang, L.; Baker, R.; Dai, X.; Zhang, J. Using pyridine as nitrogen-rich precursor to synthesize Co-N-S/C non-noble metal electrocatalysts for oxygen reduction reaction. Appl. Catal. B Environ. 2012, 125, 197–205. [Google Scholar] [CrossRef]
- Matter, P.H.; Zhang, L.; Ozkan, U.S. The role of nanostructure in nitrogen-containing carbon catalysts for the oxygen reduction reaction. J. Catal. 2006, 239, 83–96. [Google Scholar] [CrossRef]
- Subramanian, N.P.; Li, X.; Nallathambi, V.; Kumaraguru, S.P.; Colon-Mercado, H.; Wu, G.; Lee, J.W.; Popov, B.N. Nitrogen-modified carbon-based catalysts for oxygen reduction reaction in polymer electrolyte membrane fuel cells. J. Power Sources 2009, 188, 38–44. [Google Scholar] [CrossRef]
- Jaouen, F.; Charreteur, F.; Dodelet, J.P. Fe-Based Catalysts for Oxygen Reduction in PEMFCs. J. Electrochem. Soc. 2006, 153, A689. [Google Scholar] [CrossRef]
- Lefèvre, M.; Proietti, E.; Jaouen, F.; Dodelet, J.-P. Iron-based catalysts with improved oxygen reduction activity in polymer electrolyte fuel cells. Science 2009, 324, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Jaouen, F.; Marcotte, S.; Dodelet, J.-P.; Lindbergh, G. Oxygen Reduction Catalysts for Polymer Electrolyte Fuel Cells from the Pyrolysis of Iron Acetate Adsorbed on Various Carbon Supports. J. Phys. Chem. B 2003, 107, 1376–1386. [Google Scholar] [CrossRef]
- Schilling, T.; Bron, M. Oxygen reduction at Fe–N-modified multi-walled carbon nanotubes in acidic electrolyte. Electrochim. Acta 2008, 53, 5379–5385. [Google Scholar] [CrossRef]
- Wang, X.; Wang, B.; Zhong, J.; Zhao, F.; Han, N.; Huang, W.; Zeng, M.; Fan, J.; Li, Y. Iron polyphthalocyanine sheathed multiwalled carbon nanotubes: A high-performance electrocatalyst for oxygen reduction reaction. Nano Res. 2016, 9, 1497–1506. [Google Scholar] [CrossRef]
- Osmieri, L.; Monteverde Videla, A.H.A.; Specchia, S. Activity of Co–N multi walled carbon nanotubes electrocatalysts for oxygen reduction reaction in acid conditions. J. Power Sources 2015, 278, 296–307. [Google Scholar] [CrossRef]
- Byon, H.R.; Suntivich, J.; Crumlin, E.J.; Shao-Horn, Y. Fe-N-modified multi-walled carbon nanotubes for oxygen reduction reaction in acid. Phys. Chem. Chem. Phys. 2011, 13, 21437–21445. [Google Scholar] [CrossRef] [PubMed]
- Negro, E.; Monteverde Videla, A.H.A.; Baglio, V.; Aricò, A.S.; Specchia, S.; Koper, G.J.M. Fe–N supported on graphitic carbon nano-networks grown from cobalt as oxygen reduction catalysts for low-temperature fuel cells. Appl. Catal. B Environ. 2015, 166–167, 75–83. [Google Scholar] [CrossRef]
- Byon, H.R.; Suntivich, J.; Shao-Horn, Y. Graphene-Based Non-Noble-Metal Catalysts for Oxygen Reduction Reaction in Acid. Chem. Mater. 2011, 23, 3421–3428. [Google Scholar] [CrossRef]
- Sebastián, D.; Baglio, V.; Sun, S.; Tavares, A.C.; Aricò, A.S. Graphene-supported substoichiometric sodium tantalate as a methanol-tolerant, non-noble-metal catalyst for the electroreduction of oxygen. ChemCatChem 2015, 7, 911–915. [Google Scholar] [CrossRef]
- Tsai, C.-W.; Tu, M.-H.; Chen, C.-J.; Hung, T.-F.; Liu, R.-S.; Liu, W.-R.; Lo, M.-Y.; Peng, Y.-M.; Zhang, L.; Zhang, J.; et al. Nitrogen-doped graphene nanosheet-supported non-precious iron nitride nanoparticles as an efficient electrocatalyst for oxygen reduction. RSC Adv. 2011, 1, 1349. [Google Scholar] [CrossRef]
- Park, J.C.; Choi, C.H. Graphene-derived Fe/Co-N-C catalyst in direct methanol fuel cells: Effects of the methanol concentration and ionomer content on cell performance. J. Power Sources 2017, 358, 76–84. [Google Scholar] [CrossRef]
- Zhang, L.; Kim, J.; Dy, E.; Ban, S.; Tsay, K.C.; Kawai, H.; Shi, Z.; Zhang, J. Synthesis of novel mesoporous carbon spheres and their supported Fe-based electrocatalysts for PEM fuel cell oxygen reduction reaction. Electrochim. Acta 2013, 108, 480–485. [Google Scholar] [CrossRef]
- Monteverde Videla, A.H.A.; Zhang, L.; Kim, J.; Zeng, J.; Francia, C.; Zhang, J.; Specchia, S. Mesoporous carbons supported non-noble metal Fe-N X electrocatalysts for PEM fuel cell oxygen reduction reaction. J. Appl. Electrochem. 2013, 43, 159–169. [Google Scholar] [CrossRef]
- Herrmann, I.; Kramm, U.I.; Fiechter, S.; Bogdanoff, P. Oxalate supported pyrolysis of CoTMPP as electrocatalysts for the oxygen reduction reaction. Electrochim. Acta 2009, 54, 4275–4287. [Google Scholar] [CrossRef]
- Tian, J.; Morozan, A.; Sougrati, M.T.; Lefèvre, M.; Chenitz, R.; Dodelet, J.-P.; Jones, D.; Jaouen, F. Optimized synthesis of Fe/N/C cathode catalysts for PEM fuel cells: A matter of iron-ligand coordination strength. Angew. Chemie Int. Ed. 2013, 52, 6867–6870. [Google Scholar] [CrossRef] [PubMed]
- Velázquez-Palenzuela, A.; Zhang, L.; Wang, L.; Cabot, P.L.; Brillas, E.; Tsay, K.; Zhang, J. Carbon-Supported Fe–N x Catalysts Synthesized by Pyrolysis of the Fe(II)–2,3,5,6-Tetra(2-pyridyl)pyrazine Complex: Structure, Electrochemical Properties, and Oxygen Reduction Reaction Activity. J. Phys. Chem. C 2011, 115, 12929–12940. [Google Scholar] [CrossRef]
- Bezerra, C.W.B.; Zhang, L.; Lee, K.; Liu, H.; Zhang, J.; Shi, Z.; Marques, A.L.B.; Marques, E.P.; Wu, S.; Zhang, J. Novel carbon-supported Fe-N electrocatalysts synthesized through heat treatment of iron tripyridyl triazine complexes for the PEM fuel cell oxygen reduction reaction. Electrochim. Acta 2008, 53, 7703–7710. [Google Scholar] [CrossRef]
- Velázquez-Palenzuela, A.; Zhang, L.; Wang, L.; Cabot, P.L.; Brillas, E.; Tsay, K.; Zhang, J. Fe-Nx/C electrocatalysts synthesized by pyrolysis of Fe(II)-2,3,5,6-tetra(2-pyridyl)pyrazine complex for PEM fuel cell oxygen reduction reaction. Electrochim. Acta 2011, 56, 4744–4752. [Google Scholar] [CrossRef]
- Zhong, L.; Hu, Y.; Cleemann, L.N.; Pan, C.; Sværke, J.; Jensen, J.O.; Li, Q. Encapsulated iron-based oxygen reduction electrocatalysts by high pressure pyrolysis. Int. J. Hydrogen Energy 2017, 42, 22887–22896. [Google Scholar] [CrossRef]
- Zhong, H.; Zhang, H.; Liu, S.; Deng, C.; Wang, M. Nitrogen-enriched carbon from melamine resins with superior oxygen reduction reaction activity. ChemSusChem 2013, 6, 807–812. [Google Scholar] [CrossRef]
- Bayram, E.; Yilmaz, G.; Mukerjee, S. A solution-based procedure for synthesis of nitrogen doped graphene as an efficient electrocatalyst for oxygen reduction reactions in acidic and alkaline electrolytes. Appl. Catal. B Environ. 2016, 192, 26–34. [Google Scholar] [CrossRef]
- Wu, G.; Nelson, M.; Ma, S.; Meng, H.; Cui, G.; Shen, P.K. Synthesis of nitrogen-doped onion-like carbon and its use in carbon-based CoFe binary non-precious-metal catalysts for oxygen-reduction. Carbon 2011, 49, 3972–3982. [Google Scholar] [CrossRef]
- Chung, H.T.; Johnston, C.M.; Artyushkova, K.; Ferrandon, M.; Myers, D.J.; Zelenay, P. Cyanamide-derived non-precious metal catalyst for oxygen reduction. Electrochem. commun. 2010, 12, 1792–1795. [Google Scholar] [CrossRef]
- Choi, C.H.; Lim, H.K.; Chung, M.W.; Park, J.C.; Shin, H.; Kim, H.; Woo, S.I. Long-range electron transfer over graphene-based catalyst for high-performing oxygen reduction reactions: Importance of size, n-doping, and metallic impurities. J. Am. Chem. Soc. 2014, 136, 9070–9077. [Google Scholar] [CrossRef] [PubMed]
- Nallathambi, V.; Leonard, N.; Kothandaraman, R.; Calabrese Barton, S. Nitrogen Precursor Effects in Iron-Nitrogen-Carbon Oxygen Reduction Catalysts. Electrochem. Solid-State Lett. 2011, 14, B55–B58. [Google Scholar] [CrossRef]
- Charreteur, F.; Jaouen, F.; Dodelet, J.P. Iron porphyrin-based cathode catalysts for PEM fuel cells: Influence of pyrolysis gas on activity and stability. Electrochim. Acta 2009, 54, 6622–6630. [Google Scholar] [CrossRef]
- Meng, H.; Larouche, N.; Lefvre, M.; Jaouen, F.; Stansfield, B.; Dodelet, J.P. Iron porphyrin-based cathode catalysts for polymer electrolyte membrane fuel cells: Effect of NH3 and Ar mixtures as pyrolysis gases on catalytic activity and stability. Electrochim. Acta 2010, 55, 6450–6461. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhang, B.; Feng, Z.; Su, D.S. Synthesis-structure-performance correlation for poly(phenylenediamine)s/iron/carbon non-precious metal catalysts for oxygen reduction reaction. Catal. Today 2016, 260, 112–118. [Google Scholar] [CrossRef]
- Shu, C.; Chen, Y.; Yang, X.-D.; Liu, Y.; Chong, S.; Fang, Y.; Liu, Y.; Yang, W.-H. Enhanced Fe dispersion via “pinning” effect of thiocyanate ion on ferric ion in Fe-N-S-doped catalyst as an excellent oxygen reduction reaction electrode. J. Power Sources 2018, 376, 161–167. [Google Scholar] [CrossRef]
- Thompson, S.T.; Wilson, A.R.; Zelenay, P.; Myers, D.J.; More, K.L.; Neyerlin, K.C.; Papageorgopoulos, D. ElectroCat: DOE’s approach to PGM-free catalyst and electrode R&D. Solid State Ionics 2018, 319, 68–76. [Google Scholar] [CrossRef]
- Huang, H.C.; Shown, I.; Chang, S.T.; Hsu, H.C.; Du, H.Y.; Kuo, M.C.; Wong, K.T.; Wang, S.F.; Wang, C.H.; Chen, L.C.; et al. Pyrolyzed cobalt corrole as a potential non-precious catalyst for fuel cells. Adv. Funct. Mater. 2012, 22, 3500–3508. [Google Scholar] [CrossRef]
- Ramavathu, L.N.; Maniam, K.K.; Gopalram, K.; Chetty, R. Effect of pyrolysis temperature on cobalt phthalocyanine supported on carbon nanotubes for oxygen reduction reaction. J. Appl. Electrochem. 2012, 42, 945–951. [Google Scholar] [CrossRef]
- Banham, D.; Ye, S.; Pei, K.; Ozaki, J.; Kishimoto, T.; Imashiro, Y. A review of the stability and durability of non-precious metal catalysts for the oxygen reduction reaction in proton exchange membrane fuel cells. J. Power Sources 2015, 285, 334–348. [Google Scholar] [CrossRef]
- Wu, G.; Johnston, C.M.; Mack, N.H.; Artyushkova, K.; Ferrandon, M.; Nelson, M.; Lezama-Pacheco, J.S.; Conradson, S.D.; More, K.L.; Myers, D.J.; et al. Synthesis–structure–performance correlation for polyaniline–Me–C non-precious metal cathode catalysts for oxygen reduction in fuel cells. J. Mater. Chem. 2011, 21, 11392–11405. [Google Scholar] [CrossRef]
- Pérez-Alonso, F.J.; Salam, M.A.; Herranz, T.; Gómez de la Fuente, J.L.; Al-Thabaiti, S.A.; Basahel, S.N.; Peña, M.A.; Fierro, J.L.G.; Rojas, S. Effect of carbon nanotube diameter for the synthesis of Fe/N/multiwall carbon nanotubes and repercussions for the oxygen reduction reaction. J. Power Sources 2013, 240, 494–502. [Google Scholar] [CrossRef]
- Ferrandon, M.; Kropf, A.J.; Myers, D.J.; Kramm, U.; Bogdanoff, P.; Wu, G.; Johnston, C.M.; Zelenay, P. Multitechnique Characterization of a Polyaniline-Iron-Carbon Oxygen Reduction Catalyst. J. Phys. Chem. C 2012, 116, 16001–16013. [Google Scholar] [CrossRef]
- Wu, G.; More, K.L.; Xu, P.; Wang, H.-L.; Ferrandon, M.; Kropf, A.J.; Myers, D.J.; Ma, S.; Johnston, C.M.; Zelenay, P. A carbon-nanotube-supported graphene-rich non-precious metal oxygen reduction catalyst with enhanced performance durability. Chem. Commun. 2013, 49, 3291. [Google Scholar] [CrossRef] [PubMed]
- Chokai, M.; Taniguchi, M.; Moriya, S.; Matsubayashi, K.; Shinoda, T.; Nabae, Y.; Kuroki, S.; Hayakawa, T.; Kakimoto, M.; Ozaki, J.; et al. Preparation of carbon alloy catalysts for polymer electrolyte fuel cells from nitrogen-containing rigid-rod polymers. J. Power Sources 2010, 195, 5947–5951. [Google Scholar] [CrossRef]
- Chokai, M.; Daidou, T.; Nabae, Y. Development of Pt-Free Carbon-Based Catalyst for PEFC Cathode Prepared from Polyacrylonitrile. ECS Trans. 2014, 64, 261–270. [Google Scholar] [CrossRef]
- Wang, M.; Woo, K.; Lou, T.; Zhai, Y.; Kim, D. Defining catalyst layer ingredients in PEMFC by orthogonal test and C–V method. Int. J. Hydrogen Energy 2005, 30, 381–384. [Google Scholar] [CrossRef]
- Ding, S.-Y.; Wang, W. Covalent organic frameworks (COFs): From design to applications. Chem. Soc. Rev. 2013, 42, 548–568. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Jin, Y.; Gu, H.; Zhang, W. Recent development of efficient electrocatalysts derived from porous organic polymers for oxygen reduction reaction. Sci. China Chem. 2017, 60, 999–1006. [Google Scholar] [CrossRef]
- Day, N.U.; Wamser, C.C.; Walter, M.G. Porphyrin polymers and organic frameworks. Polym. Int. 2015, 64, 833–857. [Google Scholar] [CrossRef]
- Wang, Q.; Zhou, Z.Y.; Lai, Y.J.; You, Y.; Liu, J.G.; Wu, X.L.; Terefe, E.; Chen, C.; Song, L.; Rauf, M.; et al. Phenylenediamine-based FeNx/C catalyst with high activity for oxygen reduction in acid medium and its active-site probing. J. Am. Chem. Soc. 2014, 136, 10882–10885. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-C.; Lai, Y.-J.; Song, L.; Zhou, Z.-Y.; Liu, J.-G.; Wang, Q.; Yang, X.-D.; Chen, C.; Shi, W.; Zheng, Y.-P.; et al. S-Doping of an Fe/N/C ORR Catalyst for Polymer Electrolyte Membrane Fuel Cells with High Power Density. Angew. Chemie Int. Ed. 2015, 54, 9907–9910. [Google Scholar] [CrossRef] [PubMed]
- Rita Sulub, S.; Martínez Millán, W.; Smit, M.A. Study of the Catalytic Activity for Oxygen Reduction of Polythiophene Modified with Cobalt or Nickel. Int. J. Electrochem. Sci. 2009, 4, 1015–1027. [Google Scholar]
- Millán, W.M.; Smit, M.A. Study of electrocatalysts for oxygen reduction based on electroconducting polymer and nickel. J. Appl. Polym. Sci. 2009, 112, 2959–2967. [Google Scholar] [CrossRef]
- Bashyam, R.; Zelenay, P. A class of non-precious metal composite catalysts for fuel cells. Nature 2006, 443, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Zhang, L.; Lui, H.; Hui, R.; Shi, Z.; Zhang, J. Oxygen reduction reaction (ORR) catalyzed by carbon-supported cobalt polypyrrole (Co-PPy/C) electrocatalysts. Electrochim. Acta 2009, 54, 4704–4711. [Google Scholar] [CrossRef]
- Wu, G.; More, K.L.; Johnston, C.M.; Zelenay, P. High-performance electrocatalysts for oxygen reduction derived from polyaniline, iron, and cobalt. Science 2011, 332, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Artyushkova, K.; Ferrandon, M.; Kropf, A.J.; Myers, D.; Zelenay, P. Performance Durability of Polyaniline-derived Non-precious Cathode Catalysts. ECS Trans. 2009, 25, 1299–1311. [Google Scholar] [CrossRef]
- Sha, H.-D.; Yuan, X.; Hu, X.-X.; Lin, H.; Wen, W.; Ma, Z.-F. Effects of Pyrrole Polymerizing Oxidant on the Properties of Pyrolysed Carbon-Supported Cobalt-Polypyrrole as Electrocatalysts for Oxygen Reduction Reaction. J. Electrochem. Soc. 2013, 160, F507–F513. [Google Scholar] [CrossRef]
- Fu, X.; Zamani, P.; Choi, J.Y.; Hassan, F.M.; Jiang, G.; Higgins, D.C.; Zhang, Y.; Hoque, M.A.; Chen, Z. In Situ Polymer Graphenization Ingrained with Nanoporosity in a Nitrogenous Electrocatalyst Boosting the Performance of Polymer-Electrolyte-Membrane Fuel Cells. Adv. Mater. 2017, 29, 1604456. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.-S.; Oh, J.-G.; Roh, B.; Hwang, I.; Kim, H. Development of highly active and stable non-precious oxygen reduction catalysts for PEM fuel cells using polypyrrole and a chelating agent. Electrochem. commun. 2011, 13, 879–881. [Google Scholar] [CrossRef]
- Domínguez, C.; Pérez-Alonso, F.J.; Abdel Salam, M.; Gómez De La Fuente, J.L.; Al-Thabaiti, S.A.; Basahel, S.N.; Peña, M.A.; Fierro, J.L.G.; Rojas, S. Effect of transition metal (M: Fe, Co or Mn) for the oxygen reduction reaction with non-precious metal catalysts in acid medium. Int. J. Hydrogen Energy 2014, 39, 5309–5318. [Google Scholar] [CrossRef]
- Ding, W.; Li, L.; Xiong, K.; Wang, Y.; Li, W.; Nie, Y.; Chen, S.; Qi, X.; Wei, Z. Shape fixing via salt recrystallization: A morphology-controlled approach to convert nanostructured polymer to carbon nanomaterial as a highly active catalyst for oxygen reduction reaction. J. Am. Chem. Soc. 2015, 137, 5414–5420. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Cao, S.; Qiu, Y.; Zhao, L. Bimetallic Fe-Co promoting one-step growth of hierarchical nitrogen- doped carbon nanotubes/nanofibers for highly efficient oxygen reduction reaction. Mater. Sci. Eng. B 2017, 223, 159–166. [Google Scholar] [CrossRef]
- Oh, H.S.; Kim, H. The role of transition metals in non-precious nitrogen-modified carbon-based electrocatalysts for oxygen reduction reaction. J. Power Sources 2012, 212, 220–225. [Google Scholar] [CrossRef]
- Zeng, J.; Francia, C.; Gerbaldi, C.; Dumitrescu, M.A.; Specchia, S.; Spinelli, P. Smart synthesis of hollow core mesoporous shell carbons (HCMSC) as effective catalyst supports for methanol oxidation and oxygen reduction reactions. J. Solid State Electrochem. 2012, 16, 3087–3096. [Google Scholar] [CrossRef]
- Shrestha, S.; Mustain, W.E. Properties of Nitrogen-Functionalized Ordered Mesoporous Carbon Prepared Using Polypyrrole Precursor. J. Electrochem. Soc. 2010, 157, B1665–B1672. [Google Scholar] [CrossRef]
- Liu, H.; Shi, Z.; Zhang, J.; Zhang, L.; Zhang, J. Ultrasonic spray pyrolyzed iron-polypyrrole mesoporous spheres for fuel cell oxygen reduction electrocatalysts. J. Mater. Chem. 2009, 19, 468–470. [Google Scholar] [CrossRef]
- Thielemann, J.P.; Girgsdies, F.; Schlögl, R.; Hess, C. Pore structure and surface area of silica SBA-15: Influence of washing and scale-up. Beilstein J. Nanotechnol. 2011, 2, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Fulvio, P.F.; Jaroniec, M.; Liang, C.; Dai, S. Polypyrrole-Based Nitrogen-Doped Carbon Replicas of SBA-15 and SBA-16 Containing Magnetic Nanoparticles. J. Phys. Chem. C 2008, 112, 13126–13133. [Google Scholar] [CrossRef]
- Zhao, D.; Feng, J.; Huo, Q.; Melosh, N.; Fredrickson, G.H.; Chmelka, B.F.; Stucky, G.D. Triblock Copolymer Syntheses of Mesoporous Silica with Periodic 50 to 300 Angstrom Pores. Science 1998, 279, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Janarthanan, R.; Serov, A.; Kishore, S.; Gamarra, D.A.; Atanassov, P.; Hibbs, M.R.; Herring, A.M. Direct Methanol Anion Exchange Membrane Fuel Cell with a Non-Platinum Group Metal Cathode based on Iron-Aminoantipyrine Catalyst. Electrochim. Acta 2015, 175, 202–208. [Google Scholar] [CrossRef]
- Serov, A.; Artyushkova, K.; Atanassov, P. Fe-N-C Oxygen Reduction Fuel Cell Catalyst Derived from Carbendazim: Synthesis, Structure, and Reactivity. Adv. Energy Mater. 2014, 4, 1301735. [Google Scholar] [CrossRef]
- Serov, A.; Artyushkova, K.; Niangar, E.; Wang, C.; Dale, N.; Jaouen, F.; Sougrati, M.T.; Jia, Q.; Mukerjee, S.; Atanassov, P. Nano-structured non-platinum catalysts for automotive fuel cell application. Nano Energy 2015, 16, 293–300. [Google Scholar] [CrossRef]
- Cheon, J.Y.; Kim, T.; Choi, Y.; Jeong, H.Y.; Kim, M.G.; Sa, Y.J.; Kim, J.; Lee, Z.; Yang, T.-H.; Kwon, K.; et al. Ordered mesoporous porphyrinic carbons with very high electrocatalytic activity for the oxygen reduction reaction. Sci. Rep. 2013, 3, 2715. [Google Scholar] [CrossRef]
- Monteverde Videla, A.H.A.; Osmieri, L.; Armandi, M.; Specchia, S. Varying the morphology of Fe-N-C electrocatalysts by templating Iron Phthalocyanine precursor with different porous SiO2 to promote the Oxygen Reduction Reaction. Electrochim. Acta 2015, 177, 43–50. [Google Scholar] [CrossRef]
- Lee, K.T.; Ji, X.; Rault, M.; Nazar, L.F. Simple synthesis of graphitic ordered mesoporous carbon materials by a solid-state method using metal phthalocyanines. Angew. Chemie Int. Ed. 2009, 48, 5661–5665. [Google Scholar] [CrossRef]
- Osmieri, L.; Escudero-Cid, R.; Monteverde Videla, A.H.A.; Ocón, P.; Specchia, S. Performance of a Fe-N-C catalyst for the oxygen reduction reaction in direct methanol fuel cell: Cathode formulation optimization and short-term durability. Appl. Catal. B Environ. 2017, 201, 253–265. [Google Scholar] [CrossRef]
- Ziegelbauer, J.M.; Olson, T.S.; Pylypenko, S.; Alamgir, F.; Jaye, C.; Atanassov, P.; Mukerjee, S. Direct spectroscopic observation of the structural origin of peroxide generation from Co-based pyrolyzed porphyrins for ORR applications. J. Phys. Chem. C 2008, 112, 8839–8849. [Google Scholar] [CrossRef]
- Serov, A.; Artyushkova, K.; Andersen, N.I.; Stariha, S.; Atanassov, P. Original Mechanochemical Synthesis of Non-Platinum Group Metals Oxygen Reduction Reaction Catalysts Assisted by Sacrificial Support Method. Electrochim. Acta 2015, 179, 154–160. [Google Scholar] [CrossRef]
- Mun, Y.; Kim, M.J.; Park, S.A.; Lee, E.; Ye, Y.; Lee, S.; Kim, Y.T.; Kim, S.; Kim, O.H.; Cho, Y.H.; et al. Soft-template synthesis of mesoporous non-precious metal catalyst with Fe-Nx/C active sites for oxygen reduction reaction in fuel cells. Appl. Catal. B Environ. 2018, 222, 191–199. [Google Scholar] [CrossRef]
- Jun, S.; Joo, S.H.; Ryoo, R.; Kruk, M.; Jaroniec, M.; Liu, Z.; Ohsuna, T.; Terasaki, O. Synthesis of New, Nanoporous Carbon with Hexagonally Ordered Mesostructure. J. Am. Chem. Soc. 2000, 122, 10712–10713. [Google Scholar] [CrossRef]
- Ren, Y.; Chia, G.H.; Gao, Z. Metal-organic frameworks in fuel cell technologies. Nano Today 2013, 8, 577–597. [Google Scholar] [CrossRef]
- Furukawa, H.; Cordova, K.E.; O’Keeffe, M.; Yaghi, O.M. The chemistry and applications of metal-organic frameworks. Science 2013, 341, 1230444. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.-H.; Filler, R.; Lee, J.; Li, J.; Mandal, B.K. Synthesis and hydrogen adsorption properties of a new phthalocyanine-based metal–organic framework. Renew. Energy 2010, 35, 1592–1595. [Google Scholar] [CrossRef]
- Kornienko, N.; Zhao, Y.; Kley, C.S.; Zhu, C.; Kim, D.; Lin, S.; Chang, C.J.; Yaghi, O.M.; Yang, P. Metal-Organic Frameworks for Electrocatalytic Reduction of Carbon Dioxide. J. Am. Chem. Soc. 2015, 137, 14129–14135. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Osgood, H.; Xie, X.; Shao, Y.; Wu, G. Engineering nanostructures of PGM-free oxygen-reduction catalysts using metal-organic frameworks. Nano Energy 2017, 31, 331–350. [Google Scholar] [CrossRef]
- Zhao, Y.; Song, Z.; Li, X.; Sun, Q.; Cheng, N.; Lawes, S.; Sun, X. Metal Organic Frameworks for Energy Storage and Conversion. Energy Storage Mater. 2015, 2, 35–62. [Google Scholar] [CrossRef]
- Wu, G. Current challenge and perspective of PGM-free cathode catalysts for PEM fuel cells. Front. Energy 2017, 11, 286–298. [Google Scholar] [CrossRef]
- Zhang, H.; Hwang, S.; Wang, M.; Feng, Z.; Karakalos, S.; Luo, L.; Qiao, Z.; Xie, X.; Wang, C.; Su, D.; et al. Single Atomic Iron Catalysts for Oxygen Reduction in Acidic Media: Particle Size Control and Thermal Activation. J. Am. Chem. Soc. 2017, 139, 14143–14149. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zeng, X.; Wang, W.; Cao, D. Recent Progress in MOF-Derived, Heteroatom-Doped Porous Carbons as Highly Efficient Electrocatalysts for Oxygen Reduction Reaction in Fuel Cells. Adv. Funct. Mater. 2018, 28, 1–21. [Google Scholar] [CrossRef]
- Barkholtz, H.M.; Liu, D.-J. Advancements in rationally designed PGM-free fuel cell catalysts derived from metal–organic frameworks. Mater. Horizons 2017, 4, 20–37. [Google Scholar] [CrossRef]
- Xia, W.; Zhu, J.; Guo, W.; An, L.; Xia, D.; Zou, R. Well-defined carbon polyhedrons prepared from nano metal-organic frameworks for oxygen reduction. J. Mater. Chem. A 2014, 2, 11606–11613. [Google Scholar] [CrossRef]
- Park, K.S.; Ni, Z.; Cote, A.P.; Choi, J.Y.; Huang, R.; Uribe-Romo, F.J.; Chae, H.K.; O’Keeffe, M.; Yaghi, O.M. Exceptional chemical and thermal stability of zeolitic imidazolate frameworks. Proc. Natl. Acad. Sci. USA 2006, 103, 10186–10191. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.R.; Jang, M.S.; Cho, H.Y.; Kwon, H.J.; Kim, S.; Ahn, W.S. ZIF-8: A comparison of synthesis methods. Chem. Eng. J. 2015, 271, 276–280. [Google Scholar] [CrossRef]
- Wang, X.; Fan, X.; Lin, H.; Fu, H.; Wang, T.; Zheng, J.; Li, X. An efficient Co-N-C oxygen reduction catalyst with highly dispersed Co sites derived from a ZnCo bimetallic zeolitic imidazolate framework. RSC Adv. 2016, 6, 37965–37973. [Google Scholar] [CrossRef]
- Afsahi, F.; Kaliaguine, S. Non-precious electrocatalysts synthesized from metal-organic frameworks. J. Mater. Chem. A 2014, 2, 12270–12279. [Google Scholar] [CrossRef]
- Zhao, S.; Yin, H.; Du, L.; He, L.; Zhao, K.; Chang, L.; Yin, G.; Zhao, H.; Liu, S.; Tang, Z. Carbonized nanoscale metal-organic frameworks as high performance electrocatalyst for oxygen reduction reaction. ACS Nano 2014, 8, 12660–12668. [Google Scholar] [CrossRef] [PubMed]
- You, B.; Jiang, N.; Sheng, M.; Drisdell, W.S.; Yano, J.; Sun, Y. Bimetal-Organic Framework Self-Adjusted Synthesis of Support-Free Nonprecious Electrocatalysts for Efficient Oxygen Reduction. ACS Catal. 2015, 5, 7068–7076. [Google Scholar] [CrossRef]
- Liu, T.; Zhao, P.; Hua, X.; Luo, W.; Chen, S.; Cheng, G. An Fe-N-C hybrid electrocatalyst derived from a bimetal-organic framework for efficient oxygen reduction. J. Mater. Chem. A 2016, 4, 11357–11364. [Google Scholar] [CrossRef]
- Ma, S.; Goenaga, G.A.; Call, A.V.; Liu, D.J. Cobalt imidazolate framework as precursor for oxygen reduction reaction electrocatalysts. Chem. A Eur. J. 2011, 17, 2063–2067. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, H.; Lin, H.; Gupta, S.; Wang, C.; Tao, Z.; Fu, H.; Wang, T.; Zheng, J.; Wu, G.; et al. Directly Converting Fe–doped Metal-Organic Frameworks into Highly Active and Stable Fe–N-C Catalysts for Oxygen Reduction in Acid. Nano Energy 2016, 25, 110–119. [Google Scholar] [CrossRef]
- Wang, X.X.; Cullen, D.A.; Pan, Y.-T.; Hwang, S.; Wang, M.; Feng, Z.; Wang, J.; Engelhard, M.H.; Zhang, H.; He, Y.; et al. Nitrogen-Coordinated Single Cobalt Atom Catalysts for Oxygen Reduction in Proton Exchange Membrane Fuel Cells. Adv. Mater. 2018, 30, 1706758. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Xia, W.; Lin, C.; Sun, J.; Mahmood, A.; Wang, Q.; Qiu, B.; Tabassum, H.; Zou, R. A pore-expansion strategy to synthesize hierarchically porous carbon derived from metal-organic framework for enhanced oxygen reduction. Carbon 2017, 114, 284–290. [Google Scholar] [CrossRef]
- Strickland, K.; Miner, E.; Jia, Q.; Tylus, U.; Ramaswamy, N.; Liang, W.; Sougrati, M.-T.; Jaouen, F.; Mukerjee, S. Highly active oxygen reduction non-platinum group metal electrocatalyst without direct metal–nitrogen coordination. Nat. Commun. 2015, 6, 7343. [Google Scholar] [CrossRef]
- Zhao, D.; Shui, J.-L.; Chen, C.; Chen, X.; Reprogle, B.M.; Wang, D.; Liu, D.-J. Iron imidazolate framework as precursor for electrocatalysts in polymer electrolyte membrane fuel cells. Chem. Sci. 2012, 3, 3200–3205. [Google Scholar] [CrossRef]
- Chenitz, R.; Kramm, U.I.; Lefevre, M.; Glibin, V.; Zhang, G.; Sun, S.; Dodelet, J.-P. A specific demetalation of Fe-N4 catalytic sites in the micropores of NC_Ar + NH3 is at the origin of the initial activity loss of this highly active Fe/N/C catalyst used for the reduction of oxygen in PEM fuel cell. Energy Environ. Sci. 2017, 365–382. [Google Scholar] [CrossRef]
- Shui, J.; Chen, C.; Grabstanowicz, L.; Zhao, D.; Liu, D.-J. Highly efficient nonprecious metal catalyst prepared with metal-organic framework in a continuous carbon nanofibrous network. Proc. Natl. Acad. Sci. USA 2015, 112, 10629–10634. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Jia, Q.; Ghoshal, S.; Liang, W.; Mukerjee, S. Highly Active and Stable Fe-N-C Catalyst for Oxygen Depolarized Cathode Applications. Langmuir 2017, 33, 9246–9253. [Google Scholar] [CrossRef] [PubMed]
- Proietti, E.; Jaouen, F.; Lefèvre, M.; Larouche, N.; Tian, J.; Herranz, J.; Dodelet, J.-P. Iron-based cathode catalyst with enhanced power density in polymer electrolyte membrane fuel cells. Nat. Commun. 2011, 2, 416. [Google Scholar] [CrossRef]
- Barkholtz, H.M.; Chong, L.; Kaiser, Z.B.; Xu, T.; Liu, D.-J. Highly Active Non-PGM Catalysts Prepared from Metal Organic Frameworks. Catalysts 2015, 5, 955–965. [Google Scholar] [CrossRef]
- Zhao, D.; Shui, J.-L.; Grabstanowicz, L.R.; Chen, C.; Commet, S.M.; Xu, T.; Lu, J.; Liu, D.-J. Highly Efficient Non-Precious Metal Electrocatalysts Prepared from One-Pot Synthesized Zeolitic Imidazolate Frameworks. Adv. Mater. 2014, 26, 1093–1097. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, M.; Cullen, D.A.; Hwang, S.; Wang, M.; Li, B.; Liu, K.; Karakalos, S.; Lucero, M.; Zhang, H.; et al. Atomically dispersed manganese catalysts for oxygen reduction in proton-exchange membrane fuel cells. Nat. Catal. 2018, 1. [Google Scholar] [CrossRef]
- Banham, D.; Kishimoto, T.; Zhou, Y.; Sato, T.; Bai, K.; Ozaki, J.; Imashiro, Y.; Ye, S. Critical advancements in achieving high power and stable nonprecious metal catalyst—Based MEAs for real-world proton exchange membrane fuel cell applications. Sci. Adv. 2018, 4, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhao, X.; Sun, J.; Li, D.; Yang, X. A versatile strategy to fabricate MOFs/carbon material integrations and their derivatives for enhanced electrocatalysis. RSC Adv. 2016, 6, 7728–7735. [Google Scholar] [CrossRef]
- Ge, L.; Yang, Y.; Wang, L.; Zhou, W.; De Marco, R.; Chen, Z.; Zou, J.; Zhu, Z. High activity electrocatalysts from metal-organic framework-carbon nanotube templates for the oxygen reduction reaction. Carbon 2015, 82, 417–424. [Google Scholar] [CrossRef]
- Zhong, H.X.; Wang, J.; Zhang, Y.W.; Xu, W.L.; Xing, W.; Xu, D.; Zhang, Y.F.; Zhang, X.B. ZIF-8 derived graphene-based nitrogen-doped porous carbon sheets as highly efficient and durable oxygen reduction electrocatalysts. Angew. Chemie Int. Ed. 2014, 53, 14235–14239. [Google Scholar] [CrossRef]
- Zelenay, P.; Myers, D. ElectroCat (Electrocatalysis Consortium) DOE Annual Merit Review; 2018. Available online: https://www.hydrogen.energy.gov/pdfs/review18/fc160_myers_2018_o.pdf (accessed on 2 February 2019).
- CatCost—Free and Public Catalyst Cost Estimation Tool. Available online: https://catcost.chemcatbio.org/ (accessed on 20 January 2019).
- Rotating Electrode Methods and Oxygen Reduction Electrocatalysts; Xing, W., Yin, G., Zhang, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 978-0-444-63278-4. [Google Scholar]
- Frackowiak, E.; Béguin, F. Carbon materials for the electrochemical storage of energy in capacitors. Carbon 2001, 39, 937–950. [Google Scholar] [CrossRef]
- Kim, T.; Lim, S.; Kwon, K.; Hong, S.H.; Qiao, W.; Rhee, C.K.; Yoon, S.H.; Mochida, I. Electrochemical capacitances of well-defined carbon surfaces. Langmuir 2006, 22, 9086–9088. [Google Scholar] [CrossRef] [PubMed]
- Osmieri, L.; Monteverde Videla, A.H.A.; Specchia, S. The use of different types of reduced graphene oxide in the preparation of Fe-N-C electrocatalysts: Capacitive behavior and oxygen reduction reaction activity in alkaline medium. J. Solid State Electrochem. 2016, 20, 3507–3523. [Google Scholar] [CrossRef]
- Chlistunoff, J. RRDE and Voltammetric Study of ORR on Pyrolyzed Fe/Polyaniline Catalyst. On the Origins of Variable Tafel Slopes. J. Phys. Chem. C 2011, 115, 6496–6507. [Google Scholar] [CrossRef]
- Osmieri, L.; Monteverde Videla, A.H.A.; Ocón, P.; Specchia, S. Kinetics of Oxygen Electroreduction on Me-N-C (Me = Fe, Co, Cu) Catalysts in Acidic Medium. Insights on the Effect of the Transition Metal. J. Phys. Chem. C 2017, 121, 17796–17817. [Google Scholar] [CrossRef]
- Bonakdarpour, A.; Lefevre, M.; Yang, R.; Jaouen, F.; Dahn, T.; Dodelet, J.-P.; Dahn, J.R. Impact of Loading in RRDE Experiments on Fe–N–C Catalysts: Two- or Four-Electron Oxygen Reduction? Electrochem. Solid-State Lett. 2008, 11, B105. [Google Scholar] [CrossRef]
- Zhou, R.; Zheng, Y.; Jaroniec, M.; Qiao, S.-Z. Determination of the Electron Transfer Number for the Oxygen Reduction Reaction: From Theory to Experiment. ACS Catal. 2016, 6, 4720–4728. [Google Scholar] [CrossRef]
- Brouzgou, A.; Song, S.Q.; Tsiakaras, P. Low and non-platinum electrocatalysts for PEMFCs: Current status, challenges and prospects. Appl. Catal. B Environ. 2012, 127, 371–388. [Google Scholar] [CrossRef]
- Choi, J.-Y.; Yang, L.; Kishimoto, T.; Fu, X.; Ye, S.; Chen, Z.; Banham, D. Is the rapid initial performance loss of Fe/N/C non precious metal catalysts due to micropore flooding? Energy Environ. Sci. 2017, 10, 296–305. [Google Scholar] [CrossRef]
- Choi, C.H.; Lim, H.K.; Chung, M.W.; Chon, G.; Ranjbar Sahraie, N.; Altin, A.; Sougrati, M.T.; Stievano, L.; Oh, H.S.; Park, E.S.; et al. The Achilles’ heel of iron-based catalysts during oxygen reduction in an acidic medium. Energy Environ. Sci. 2018, 11, 3176–3182. [Google Scholar] [CrossRef]
- Serov, A.; Atanassov, P. Carbendazim-Based Catalytic Materals. U.S. Patent No. US9425464B2, 23 August 2016. [Google Scholar]
- Serov, A.; Atanassov, P. Materials with Atomically Dispersed Chemical Moieties. International Patent No. WO 2016/133921 Al, 25 August 2016. [Google Scholar]
- Calabrese Barton, S.; Ramanujam, K.; Nallathambi, V. Catalyst for Oxygen Reduction Reaction in Fuel Cells. U.S. Patent No. 9,379,388, 28 June 2016. [Google Scholar]
- Armel, V.; Bennet, S.; Jaouen, F.; Jones, D.; Hindocha, S.; Salles, F. Oxygen Reduction Reaction Catalyst. U.S. Patent No. US 2018/0294485 A1, 11 October 2018. [Google Scholar]
- Serov, A.; Atanassov, P.; Halevi, B.; Short, P. Non-PGM Catalyst for Orr Based on Pyrolysed Poly-Complexes. European Patent No. EP 2 906 347 B1, 28 November 2018. [Google Scholar]
- Serov, A.; Halevi, B.; Artyushkova, K.; Atanassov, P.; Martinez, U. Non-PGM Cathode Catalysts for Fuel Cell Application Derived from Heat Treated Heteroatomic Amines Precursors. European Patent No. EP 2 720 793 B1, 20 December 2012. [Google Scholar]
- Imashiro, Y.; Kishimoto, T.; Sato, T.; Ozaki, J.; Maie, T.; Kusadokoro, S. Carbon Catalyst, Electrode, and Battery. U.S. Patent No. 2017/0194653A1, 6 July 2017. [Google Scholar]
- Li, Q.; Wan, G.; Zhao, H.; Pan, L.; Wang, N.; Zhao, W.; Zhou, X.; Cui, X.; Chen, H. Nitrogen-Doped Carbon Vesicles with Dual Iron-Based Sites for Efficient Oxygen Reduction. ChemSusChem 2017, 10, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.; Shui, J.L.; Grabstanowicz, L.; Chen, C.; Commet, S.; Reprogle, B.; Xu, T.; Yu, L.; Liu, D.J. A highly active and support-free oxygen reduction catalyst prepared from ultrahigh-surface-area porous polyporphyrin. Angew. Chemie Int. Ed. 2013, 52, 8349–8353. [Google Scholar] [CrossRef] [PubMed]
- Muthukrishnan, A.; Nabae, Y.; Hayakawa, T.; Okajima, T.; Ohsaka, T. Fe-containing polyimide-based high-performance ORR catalysts in acidic medium: A kinetic approach to study the durability of catalysts. Catal. Sci. Technol. 2015, 5, 475–483. [Google Scholar] [CrossRef]
- Nabae, Y.; Kuang, Y.; Chokai, M.; Ichihara, T.; Isoda, A.; Hayakawa, T.; Aoki, T. High performance Pt-free cathode catalysts for polymer electrolyte membrane fuel cells prepared from widely available chemicals. J. Mater. Chem. A 2014, 2, 11561–11564. [Google Scholar] [CrossRef]
- Vinayan, B.P.; Diemant, T.; Behm, R.J.; Ramaprabhu, S. Iron encapsulated nitrogen and sulfur co-doped few layer graphene as a non-precious ORR catalyst for PEMFC application. RSC Adv. 2015, 5, 66494–66501. [Google Scholar] [CrossRef]
- Maruyama, J.; Hasegawa, T.; Iwasaki, S.; Kanda, H.; Kishimoto, H. Heat treatment of carbonized hemoglobin with ammonia for enhancement of pore development and oxygen reduction activity. ACS Sustain. Chem. Eng. 2014, 2, 493–499. [Google Scholar] [CrossRef]
- Hu, Y.; Jensen, J.O.; Zhang, W.; Martin, S.; Chenitz, R.; Pan, C.; Xing, W.; Bjerrum, N.J.; Li, Q. Fe3C-based oxygen reduction catalysts: Synthesis, hollow spherical structures and applications in fuel cells. J. Mater. Chem. A 2015, 3, 1752–1760. [Google Scholar] [CrossRef]
- Chung, H.T.; Cullen, D.A.; Higgins, D.; Sneed, B.T.; Holby, E.F.; More, K.L.; Zelenay, P. Direct atomic-level insight into the active sites of a high-performance PGM-free ORR catalyst. Science 2017, 357, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.T.; Wu, G.; Li, Q.; Zelenay, P. Role of two carbon phases in oxygen reduction reaction on the Co-PPy-C catalyst. Int. J. Hydrogen Energy 2013, 39, 15887–15893. [Google Scholar] [CrossRef]
- Wang, G.; Jiang, K.; Xu, M.; Min, C.; Ma, B.; Yang, X. Short communication A high activity nitrogen-doped carbon catalyst for oxygen reduction reaction derived from polyaniline-iron coordination polymer. J. Power Sources 2014, 266, 222–225. [Google Scholar] [CrossRef]
- Robson, M.H.; Serov, A.; Artyushkova, K.; Atanassov, P. A mechanistic study of 4-aminoantipyrine and iron derived non-platinum group metal catalyst on the oxygen reduction reaction. Electrochim. Acta 2013, 90, 656–665. [Google Scholar] [CrossRef]
- Serov, A.; Robson, M.H.; Smolnik, M.; Atanassov, P. Tri-metallic transition metal–nitrogen–carbon catalysts derived by sacrificial support method synthesis. Electrochim. Acta 2013, 109, 433–439. [Google Scholar] [CrossRef]
- Maruyama, J.; Okamura, J.; Miyazaki, K.; Uchimoto, Y.; Abe, I. Hemoglobin pyropolymer used as a precursor of a noble-metal-free fuel cell cathode catalyst. J. Phys. Chem. C 2008, 112, 2784–2790. [Google Scholar] [CrossRef]
- Ferrandon, M.; Wang, X.; Kropf, A.J.; Myers, D.J.; Wu, G.; Johnston, C.M.; Zelenay, P. Stability of iron species in heat-treated polyaniline–iron–carbon polymer electrolyte fuel cell cathode catalysts. Electrochim. Acta 2013, 110, 282–291. [Google Scholar] [CrossRef]
- Liu, G.; Li, X.; Ganesan, P.; Popov, B.N. Studies of oxygen reduction reaction active sites and stability of nitrogen-modified carbon composite catalysts for PEM fuel cells. Electrochim. Acta 2010, 55, 2853–2858. [Google Scholar] [CrossRef]
- Piela, B.; Olson, T.S.; Atanassov, P.; Zelenay, P. Highly methanol-tolerant non-precious metal cathode catalysts for direct methanol fuel cell. Electrochim. Acta 2010, 55, 7615–7621. [Google Scholar] [CrossRef]
- Larouche, N.; Chenitz, R.; Lefèvre, M.; Proietti, E.; Dodelet, J.P. Activity and stability in proton exchange membrane fuel cells of iron-based cathode catalysts synthesized with addition of carbon fibers. Electrochim. Acta 2014, 115, 170–182. [Google Scholar] [CrossRef]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osmieri, L. Transition Metal–Nitrogen–Carbon (M–N–C) Catalysts for Oxygen Reduction Reaction. Insights on Synthesis and Performance in Polymer Electrolyte Fuel Cells. ChemEngineering 2019, 3, 16. https://doi.org/10.3390/chemengineering3010016
Osmieri L. Transition Metal–Nitrogen–Carbon (M–N–C) Catalysts for Oxygen Reduction Reaction. Insights on Synthesis and Performance in Polymer Electrolyte Fuel Cells. ChemEngineering. 2019; 3(1):16. https://doi.org/10.3390/chemengineering3010016
Chicago/Turabian StyleOsmieri, Luigi. 2019. "Transition Metal–Nitrogen–Carbon (M–N–C) Catalysts for Oxygen Reduction Reaction. Insights on Synthesis and Performance in Polymer Electrolyte Fuel Cells" ChemEngineering 3, no. 1: 16. https://doi.org/10.3390/chemengineering3010016
APA StyleOsmieri, L. (2019). Transition Metal–Nitrogen–Carbon (M–N–C) Catalysts for Oxygen Reduction Reaction. Insights on Synthesis and Performance in Polymer Electrolyte Fuel Cells. ChemEngineering, 3(1), 16. https://doi.org/10.3390/chemengineering3010016





