Effects of Diaphragmatic Breathing on Health: A Narrative Review
Abstract
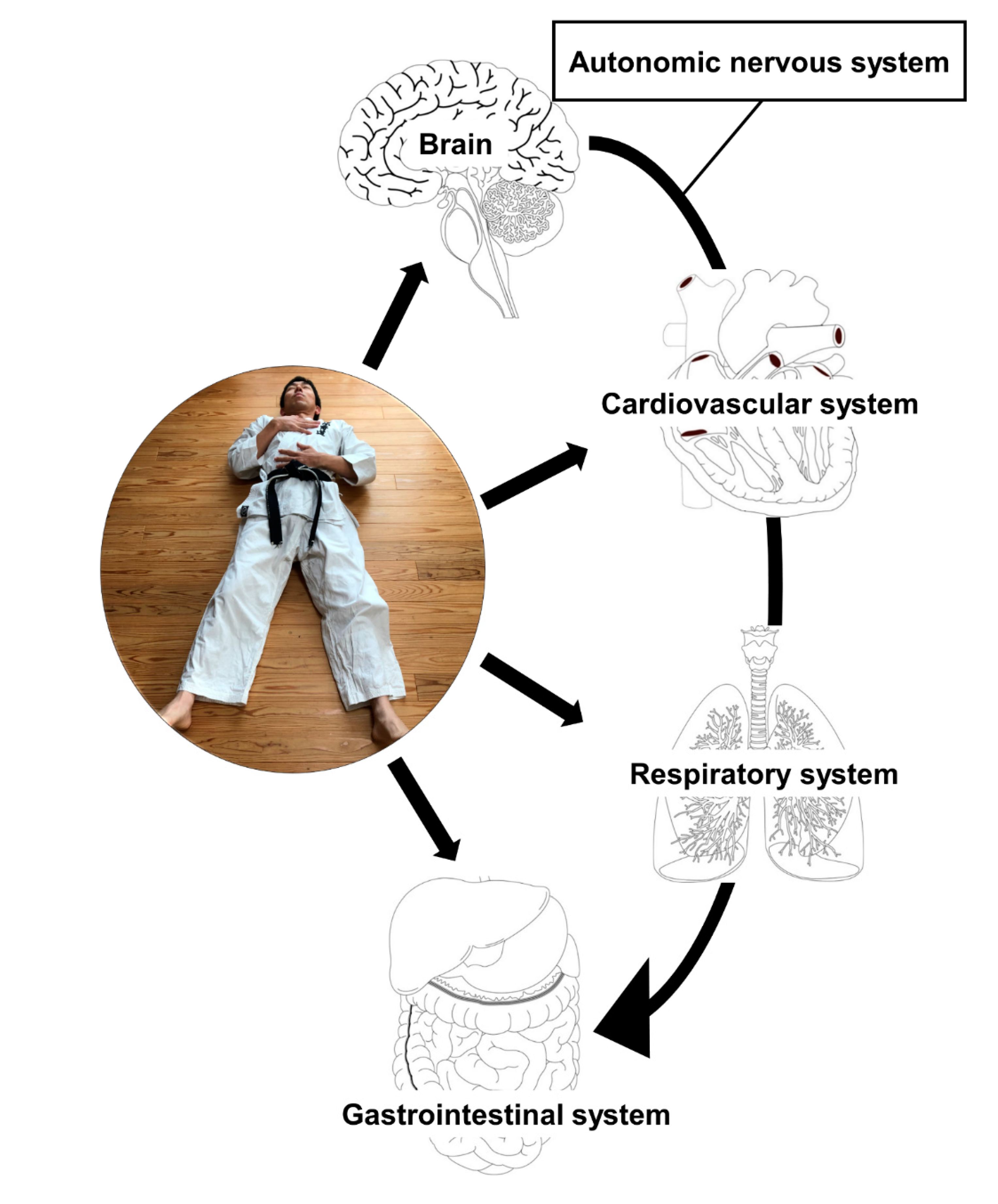
1. Introduction
2. Methods
3. Results
3.1. Systematic Reviews
3.2. Randomized Controlled Trials
3.2.1. COPD and Asthma
3.2.2. Cancer
3.2.3. Other Diseases
3.2.4. Healthy Individuals
4. Discussion
5. Conclusions
Funding
Conflicts of Interest
References
- Sewall, H.; Pollard, M.E. On the Relations of Diaphragmatic and Costal Respiration, with particular reference to Phonation. J. Physiol. 1890, 11, 159–264. [Google Scholar] [CrossRef]
- Fogarty, M.J.; Mantilla, C.B.; Sieck, G.C. Breathing: Motor control of diaphragm muscle. Physiology 2018, 33, 113–126. [Google Scholar] [CrossRef]
- Ricoy, J.; Rodríguez-Núñez, N.; Álvarez-Dobaño, J.M.; Toubes, M.E.; Riveiro, V.; Valdés, L. Diaphragmatic dysfunction. Pulmonology 2019, 25, 223–235. [Google Scholar] [CrossRef]
- Kocjan, J.; Adamek, M.; Gzik-Zroska, B.; Czyżewski, D.; Rydel, M. Network of breathing. Multifunctional role of the diaphragm: A review. Adv. Respir. Med. 2017, 85, 224–232. [Google Scholar] [CrossRef]
- Bordoni, B.; Purgol, S.; Bizzarri, A.; Modica, M.; Morabito, B. The Influence of Breathing on the Central Nervous System. Cureus 2018, 10, e2724. [Google Scholar] [CrossRef]
- Bordoni, B.; Zanier, E. The continuity of the body: Hypothesis of treatment of the five diaphragms. J. Altern. Complement. Med. 2015, 21, 237–242. [Google Scholar] [CrossRef]
- Bruton, A.; Garrod, R.; Thomas, M. Respiratory physiotherapy: Towards a clearer definition of terminology. Physiotherapy 2011, 97, 345–349. [Google Scholar] [CrossRef]
- Rama, S.; Ballentine, R.; Hymes, A. Science of Breath: A Practical Guide; Himalayan Institute Press: Honesdale, PA, USA, 1998; pp. 26–44. [Google Scholar]
- Zou, L.; Sasaki, J.E.; Wei, G.X.; Huang, T.; Yeung, A.S.; Neto, O.B.; Chen, K.W.; Hui, S.S. Effects of Mind⁻Body Exercises (Tai Chi/Yoga) on Heart Rate Variability Parameters and Perceived Stress: A Systematic Review with Meta-Analysis of Randomized Controlled Trials. J. Clin. Med. 2018, 7, 404. [Google Scholar] [CrossRef]
- Martarelli, D.; Cocchioni, M.; Scuri, S.; Pompei, P. Diaphragmatic breathing reduces exercise-induced oxidative stress. Evid. Based Complement. Altern. Med. 2011, 2011, 932430. [Google Scholar] [CrossRef]
- Ambrosino, N.; Paggiaro, P.L.; Macchi, M.; Filieri, M.; Toma, G.; Lombardi, F.A.; Del Cesta, F.; Parlanti, A.; Loi, A.M.; Baschieri, L. A study of short-term effect of rehabilitative therapy in chronic obstructive pulmonary disease. Respiration 1981, 41, 40–44. [Google Scholar] [CrossRef]
- Tiep, B.L.; Burns, M.; Kao, D.; Madison, R.; Herrera, J. Pursed lips breathing training using ear oximetry. Chest 1986, 90, 218–221. [Google Scholar] [CrossRef]
- Collins, E.G.; Fehr, L.; Bammert, C.; O’Connell, S.; Laghi, F.; Hanson, K.; Hagarty, E.; Langbein, W.E. Effect of ventilation-feedback training on endurance and perceived breathlessness during constant work-rate leg-cycle exercise in patients with COPD. J. Rehabil. Res. Dev. 2003, 40, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.Y.; Dean, E.; Chow, C.C. Comparison of the oxygen cost of breathing exercises and spontaneous breathing in patients with stable chronic obstructive pulmonary disease. Phys. Ther. 2003, 83, 424–431. [Google Scholar] [CrossRef]
- Garrod, R.; Dallimore, K.; Cook, J.; Davies, V.; Quade, K. An evaluation of the acute impact of pursed lips breathing on walking distance in nonspontaneous pursed lips breathing chronic obstructive pulmonary disease patients. Chron. Respir. Dis. 2005, 2, 67–72. [Google Scholar] [CrossRef]
- Spahija, J.; de Marchie, M.; Grassino, A. Effects of imposed pursed-lips breathing on respiratory mechanics and dyspnea at rest and during exercise in COPD. Chest 2005, 128, 640–650. [Google Scholar] [CrossRef]
- Nield, M.A.; Soo Hoo, G.W.; Roper, J.M.; Santiago, S. Efficacy of pursed-lips breathing: A breathing pattern retraining strategy for dyspnea reduction. J. Cardiopulm. Rehabil. Prev. 2007, 27, 237–244. [Google Scholar] [CrossRef]
- Collins, E.G.; Langbein, W.E.; Fehr, L.; O’Connell, S.; Jelinek, C.; Hagarty, E.; Edwards, L.; Reda, D.; Tobin, M.J.; Laghi, F. Can ventilation-feedback training augment exercise tolerance in patients with chronic obstructive pulmonary disease? Am. J. Respir. Crit. Care Med. 2008, 177, 844–852. [Google Scholar] [CrossRef]
- Faager, G.; Stâhle, A.; Larsen, F.F. Influence of spontaneous pursed lips breathing on walking endurance and oxygen saturation in patients with moderate to severe chronic obstructive pulmonary disease. Clin. Rehabil. 2008, 22, 675–683. [Google Scholar] [CrossRef]
- Bonilha, A.G.; Onofre, F.; Vieira, M.L.; Prado, M.Y.; Martinez, J.A. Effects of singing classes on pulmonary function and quality of life of COPD patients. Int. J. Chron. Obstruct. Pulmon. Dis. 2009, 4, 1–8. [Google Scholar]
- Lord, V.M.; Cave, P.; Hume, V.J.; Flude, E.J.; Evans, A.; Kelly, J.L.; Polkey, M.I.; Hopkinson, N.S. Singing teaching as a therapy for chronic respiratory disease—A randomised controlled trial and qualitative evaluation. BMC Pulm. Med. 2010, 10, 41. [Google Scholar] [CrossRef]
- Lin, W.C.; Yuan, S.C.; Chien, J.Y.; Weng, S.C.; Chou, M.C.; Kuo, H.W. The effects of respiratory training for chronic obstructive pulmonary disease patients: A randomised clinical trial. J. Clin. Nurs. 2012, 21, 2870–2878. [Google Scholar] [CrossRef] [PubMed]
- Lord, V.M.; Hume, V.J.; Kelly, J.L.; Cave, P.; Silver, J.; Waldman, M.; White, C.; Smith, C.; Tanner, R.; Sanchez, M.; et al. Singing classes for chronic obstructive pulmonary disease: A randomized controlled trial. BMC Pulm. Med. 2012, 12, 69. [Google Scholar] [CrossRef]
- van Gestel, A.J.; Kohler, M.; Steier, J.; Teschler, S.; Russi, E.W.; Teschler, H. The effects of controlled breathing during pulmonary rehabilitation in patients with COPD. Respiration 2012, 83, 115–124. [Google Scholar] [CrossRef]
- Yamaguti, W.P.; Claudino, R.C.; Neto, A.P.; Chammas, M.C.; Gomes, A.C.; Salge, J.M.; Moriya, H.T.; Cukier, A.; Carvalho, C.R. Diaphragmatic breathing training program improves abdominal motion during natural breathing in patients with chronic obstructive pulmonary disease: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2012, 93, 571–577. [Google Scholar] [CrossRef]
- Bhatt, S.P.; Luqman-Arafath, T.K.; Gupta, A.K.; Mohan, A.; Stoltzfus, J.C.; Dey, T.; Nanda, S.; Guleria, R. Volitional pursed lips breathing in patients with stable chronic obstructive pulmonary disease improves exercise capacity. Chron. Respir. Dis. 2013, 10, 5–10. [Google Scholar] [CrossRef]
- de Araujo, C.L.; Karloh, M.; Dos Reis, C.M.; Palú, M.; Mayer, A.F. Pursed-lips breathing reduces dynamic hyperinflation induced by activities of daily living test in patients with chronic obstructive pulmonary disease: A randomized cross-over study. J. Rehabil. Med. 2015, 47, 957–962. [Google Scholar]
- Cabral, L.F.; D’Elia, T.C.; Marins, D.S.; Zin, W.A.; Guimarães, F.S. Pursed lip breathing improves exercise tolerance in COPD: A randomized crossover study. Eur. J. Phys. Rehabil. Med. 2015, 51, 79–88. [Google Scholar]
- Borge, C.R.; Mengshoel, A.M.; Omenaas, E.; Moum, T.; Ekman, I.; Lein, M.P.; Mack, U.; Wahl, A.K. Effects of guided deep breathing on breathlessness and the breathing pattern in chronic obstructive pulmonary disease: A double-blind randomized control study. Patient Educ. Couns. 2015, 98, 182–190. [Google Scholar] [CrossRef]
- Thomas, M.; McKinley, R.K.; Freeman, E.; Foy, C.; Prodger, P.; Price, D. Breathing retraining for dysfunctional breathing in asthma: A randomised controlled trial. Thorax 2003, 58, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.; McKinley, R.K.; Mellor, S.; Watkin, G.; Holloway, E.; Scullion, J.; Shaw, D.E.; Wardlaw, A.; Price, D.; Pavord, I. Breathing exercises for asthma: A randomised controlled trial. Thorax 2009, 64, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Grammatopoulou, E.P.; Skordilis, E.K.; Stavrou, N.; Myrianthefs, P.; Katrteroliotis, K.; Baltopoulos, G.; Koutsouki, D. The effect of physiotherapy-based breathing retraining on asthma control. J. Asthma 2011, 48, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Asher, M.I.; Douglas, C.; Airy, M.; Andrews, D.; Trenholme, A. Effects of chest physical therapy on lung function in children recovering from acute severe asthma. Pediatr. Pulmonol. 1990, 9, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Bingöl, G.K.; Yilmaz, M.; Sur, S.; Ufuk, D.A.; Sarpel, T.; Güneter, S.K. The effects of daily pulmonary rehabilitation program at home on childhood asthma. Allergol. Immunopathol. 2000, 28, 12–14. [Google Scholar]
- Lima, E.V.N.C.L.; Lima, W.L.; Nobre, A.; dos Santos, A.M.; Brito, L.M.O.; do Rosário da Silva Ramos Costa, M. Inspiratory muscle training and respiratory exercises in children with asthma. J. Bras. Pneumol. 2008, 34, 552–558. [Google Scholar] [CrossRef]
- Manzano, R.M.; Carvalho, C.R.; Saraiva-Romanholo, B.M.; Vieira, J.E. Chest physiotherapy during immediate postoperative period among patients undergoing upper abdominal surgery: Randomized clinical trial. Sao Paulo Med. J. 2008, 126, 269–273. [Google Scholar] [CrossRef]
- Roukema, J.A.; Carol, E.J.; Prins, J.G. The prevention of pulmonary complications after upper abdominal surgery in patients with noncompromised pulmonary status. Arch. Surg. 1988, 123, 30–34. [Google Scholar] [CrossRef]
- Ribeiro, S.; Gastaldi, A.C.; Fernandes, C. The effect of respiratory kinesiotherapy in patients undergoing upper abdominal surgery. Einstein 2008, 6, 166–169. [Google Scholar]
- Gastaldi, A.C.; Magalhães, C.M.B.; Baraúna, M.A.; Silva, E.M.C.; Souza, H.C.D. Benefícios da cinesioterapia respiratória no pós-operatório de colecistectomia laparoscópica. Rev. Bras. Fisioter. 2008, 12, 100–106. [Google Scholar] [CrossRef][Green Version]
- Forgiarini, L.A., Jr.; Carvalho, A.T.; Ferreira, T.S.; Monteiro, M.B.; Bosco, A.D.; Gonçalves, M.P.; Dias, A.S. Atendimento fisioterapêutico no pós-operatório imediato de pacientes submetidos à cirurgia abdominal. J. Bras. Pneumol. 2009, 35, 445–459. [Google Scholar] [CrossRef]
- Ali, L.A.; Pingitore, A.; Piaggi, P.; Brucini, F.; Passera, M.; Marotta, M.; Cadoni, A.; Passino, C.; Catapano, G.; Festa, P. Respiratory Training Late After Fontan Intervention: Impact on Cardiorespiratory Performance. Pediatr. Cardiol. 2018, 39, 695–704. [Google Scholar] [CrossRef]
- Holland, A.E.; Hill, C.J.; Jones, A.Y.; McDonald, C.F. Breathing exercises for chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 2012, 10, CD008250. [Google Scholar] [CrossRef] [PubMed]
- Lausin, G.; Gouilly, P. Étude des effets de la ventilation dirigée abdomino-diaphragmatique (Vdad) chez des patients BPCO de stade I et II: Study of the effects of controlled abdominodiaphragmatic ventilation in patients with level I and II COPD. Kinésithér. Revue 2009, 9, 29–38. [Google Scholar] [CrossRef]
- Ubolnuar, N.; Tantisuwat, A.; Thaveeratitham, P.; Lertmaharit, S.; Kruapanich, C.; Mathiyakom, W. Effects of Breathing Exercises in Patients with Chronic Obstructive Pulmonary Disease: Systematic Review and Meta-Analysis. Ann. Rehabil. Med. 2019, 43, 509–523. [Google Scholar] [CrossRef] [PubMed]
- Santino, T.A.; Chaves, G.S.; Freitas, D.A.; Fregonezi, G.A.; Mendonça, K.M. Breathing exercises for adults with asthma. Cochrane Database Syst. Rev. 2020, 3, CD001277. [Google Scholar] [CrossRef]
- Girodo, M.; Ekstrand, K.A.; Metivier, G.J. Deep diaphragmatic breathing: Rehabilitation exercises for the asthmatic patient. Arch. Phys. Med. Rehabil. 1992, 73, 717–720. [Google Scholar]
- Prem, V.; Sahoo, R.C.; Adhikari, P. Effect of diaphragmatic breathing exercise on quality of life in subjects with asthma: A systematic review. Physiother. Theory Pract. 2013, 29, 271–277. [Google Scholar] [CrossRef]
- Macêdo, T.M.; Freitas, D.A.; Chaves, G.S.; Holloway, E.A.; Mendonça, K.M. Breathing exercises for children with asthma. Cochrane Database Syst. Rev. 2016, 4, CD011017. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.; McKinley, R.K.; Freeman, E.; Foy, C. Prevalence of dysfunctional breathing in patients treated for asthma in primary care: Cross sectional survey. BMJ 2001, 322, 1098–1100. [Google Scholar] [CrossRef]
- Gridina, I.; Bidat, E.; Chevallier, B.; Stheneur, C. Prevalence of chronic hyperventilation syndrome in children and teenagers. Arch. Pediatr. Organe Off. Soc. Fr. Pediatr. 2013, 20, 265–268. [Google Scholar] [CrossRef]
- Barker, N.J.; Jones, M.; O’Connell, N.E.; Everard, M.L. Breathing exercises for dysfunctional breathing/hyperventilation syndrome in children. Cochrane Database Syst. Rev. 2013, 12, CD010376. [Google Scholar]
- Jones, M.; Harvey, A.; Marston, L.; O’Connell, N.E. Breathing exercises for dysfunctional breathing/hyperventilation syndrome in adults. Cochrane Database Syst. Rev. 2013, 5, CD009041. [Google Scholar] [CrossRef] [PubMed]
- Lindeboom, I.; Vlaander-van der Giessen, G. Hyperventilation and therapy [Hyperventilatie en therapie]. Ned. Tijdschr. Voor Fysiother. 1980, 90, 18–21. [Google Scholar]
- Lewis, L.K.; Williams, M.T.; Olds, T. Short-term effects on outcomes related to the mechanism of intervention and physiological outcomes but insufficient evidence of clinical benefits for breathing control: A systematic review. Aust. J. Physiother. 2007, 53, 219–227. [Google Scholar] [CrossRef]
- Mcneill, R.S.; Mckenzie, J.M. An assessment of the value of breathing exercises in chronic bronchitis and asthma. Thorax 1955, 10, 250–252. [Google Scholar] [CrossRef][Green Version]
- Wiens, M.E.; Reimer, M.A.; Guyn, H.L. Music therapy as a treatment method for improving respiratory muscle strength in patients with advanced multiple sclerosis: A pilot study. Rehabil. Nurs. 1999, 24, 74–80. [Google Scholar] [CrossRef]
- Sackner, M.A.; Silva, G.; Banks, J.M.; Watson, D.D.; Smoak, W.M. Distribution of ventilation during diaphragmatic breathing in obstructive lung disease. Am. Rev. Respir. Dis. 1974, 109, 331–337. [Google Scholar]
- Brach, B.B.; Chao, R.P.; Sgroi, V.L.; Minh, V.D.; Ashburn, W.L.; Moser, K.M. 133Xenon washout patterns during diaphragmatic breathing. Studies in normal subjects and patients with chronic obstructive pulmonary disease. Chest 1977, 71, 735–739. [Google Scholar] [CrossRef]
- Sackner, M.A.; Gonzalez, H.F.; Jenouri, G.; Rodriguez, M. Effects of abdominal and thoracic breathing on breathing pattern components in normal subjects and in patients with chronic obstructive pulmonary disease. Am. Rev. Respir. Dis. 1984, 130, 584–587. [Google Scholar]
- Becklake, M.R.; Mcgregor, M.; Goldman, H.I.; Braudo, J.L. A study of the effects of physiotherapy in chronic hypertrophic emphysema using lung function tests. Dis. Chest 1954, 26, 180–191. [Google Scholar] [CrossRef]
- Blaney, F.; Sawyer, T. Sonographic measurement of diaphragmatic motion after upper abdominal surgery: A comparison of three breathing manoeuvres. Physiother. Theo. Pract. 1997, 13, 207–215. [Google Scholar] [CrossRef]
- Campbell, E.J.; Friend, J. Action of breathing exercises in pulmonary emphysema. Lancet 1955, 265, 325–329. [Google Scholar] [CrossRef]
- Chuter, T.A.; Weissman, C.; Mathews, D.M.; Starker, P.M. Diaphragmatic breathing maneuvers and movement of the diaphragm after cholecystectomy. Chest 1990, 97, 1110–1114. [Google Scholar] [CrossRef]
- Gosselink, R.A.; Wagenaar, R.C.; Rijswijk, H.; Sargeant, A.J.; Decramer, M.L. Diaphragmatic breathing reduces efficiency of breathing in patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 1995, 151, 1136–1142. [Google Scholar] [PubMed]
- Grimby, G.; Oxhoj, H.; Bake, B. Effects of abdominal breathing on distribution of ventilation in obstructive lung disease. Clin. Sci. Mol. Med. 1975, 48, 193–199. [Google Scholar] [CrossRef]
- Ito, M.; Kakizaki, F.; Tsuzura, Y.; Yamada, M. Immediate effect of respiratory muscle stretch gymnastics and diaphragmatic breathing on respiratory pattern. Respiratory Muscle Conditioning Group. Intern. Med. 1999, 38, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Mckinley, H.; Gersten, J.W.; Speck, L. Pressure-volume relationships in emphysema patients before and after breathing exercises. Arch. Phys. Med. Rehabil. 1961, 42, 513–517. [Google Scholar]
- Miller, W.F. A physiologic evaluation of the effects of diaphragmatic breathing training in patients with chronic pulmonary emphysema. Am. J. Med. 1954, 17, 471–477. [Google Scholar] [CrossRef]
- Shearer, M.O.; Banks, J.M.; Silva, G.; Sackner, M.A. Lung ventilation during diaphragmatic breathing. Phys. Ther. 1972, 52, 139–148. [Google Scholar] [CrossRef]
- Sinclair, J.D. The effect of breathing exercises in pulmonary emphysema. Thorax 1955, 10, 246–249. [Google Scholar] [CrossRef][Green Version]
- Vitacca, M.; Clini, E.; Bianchi, L.; Ambrosino, N. Acute effects of deep diaphragmatic breathing in COPD patients with chronic respiratory insufficiency. Eur. Respir. J. 1998, 11, 408–415. [Google Scholar] [CrossRef]
- Williams, I.P.; Smith, C.M.; McGavin, C.R. Diaphragmatic breathing training and walking performance in chronic airways obstruction. Br. J. Dis. Chest 1982, 76, 164–166. [Google Scholar] [CrossRef]
- Willeput, R.; Vachaudez, J.P.; Lenders, D.; Nys, A.; Knoops, T.; Sergysels, R. Thoracoabdominal motion during chest physiotherapy in patients affected by chronic obstructive lung disease. Respiration 1983, 44, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Grams, S.T.; Ono, L.M.; Noronha, M.A.; Schivinski, C.I.; Paulin, E. Breathing exercises in upper abdominal surgery: A systematic review and meta-analysis. Rev. Bras. Fisioter. 2012, 16, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Hallböök, T.; Lindblad, B.; Lindroth, B.; Wolff, T. Prophylaxis against pulmonary complications in patients undergoing gall-bladder surgery. A comparison between early mobilization, physiotherapy with and without bronchodilatation. Ann. Chir. Gynaecol. 1984, 73, 55–58. [Google Scholar] [PubMed]
- Hopper, S.I.; Murray, S.L.; Ferrara, L.R.; Singleton, J.K. Effectiveness of diaphragmatic breathing for reducing physiological and psychological stress in adults: A quantitative systematic review. JBI Database Syst. Rev. Implement. Rep. 2019, 17, 1855–1876. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Yue, Z.Q.; Gong, Z.Q.; Zhang, H.; Duan, N.Y.; Shi, Y.T.; Wei, G.X.; Li, Y.F. The Effect of Diaphragmatic Breathing on Attention, Negative Affect and Stress in Healthy Adults. Front. Psychol. 2017, 8, 874. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Kiran, R.; Singla, H.K.; Sah, A.N. Stress management through regulation of blood pressure among college students. Work 2016, 54, 745–752. [Google Scholar] [CrossRef]
- Sundram, B.M.; Dahlui, M.; Chinna, K. “Taking my breath away by keeping stress at bay”—An employee assistance program in the automotive assembly plant. Iran. J. Public Health 2014, 43, 263–272. [Google Scholar]
- Yekefallah, L.; Zohal, M.A.; Keshavarzsarkar, O.; Barikani, A.; Gheraati, M. Comparing the effects of upper limb and breathing exercises on six-minute walking distance among patients with chronic obstructive pulmonary disease: A three-group randomized controlled clinical trial. Adv. Respir. Med. 2019, 87, 77–82. [Google Scholar] [CrossRef]
- Shaw, B.S.; Shaw, I. Pulmonary function and abdominal and thoracic kinematic changes following aerobic and inspiratory resistive diaphragmatic breathing training in asthmatics. Lung 2011, 189, 131–139. [Google Scholar] [CrossRef]
- Campbell, D.F.; Dixon, J.K.; Sanderford, L.D.; Denicola, M.A. Relaxation: Its effect on the nutritional status and performance status of clients with cancer. J. Am. Diet. Assoc. 1984, 84, 201–204. [Google Scholar]
- Shahriari, M.; Dehghan, M.; Pahlavanzadeh, S.; Hazini, A. Effects of progressive muscle relaxation, guided imagery and deep diaphragmatic breathing on quality of life in elderly with breast or prostate cancer. J. Educ. Health Promot. 2017, 6, 1. [Google Scholar]
- Silva, C.A.; Motta, M.E. The use of abdominal muscle training, breathing exercises and abdominal massage to treat paediatric chronic functional constipation. Colorectal. Dis. 2013, 15, e250–e255. [Google Scholar] [CrossRef]
- Wang, S.Z.; Li, S.; Xu, X.Y.; Lin, G.P.; Shao, L.; Zhao, Y.; Wang, T.H. Effect of slow abdominal breathing combined with biofeedback on blood pressure and heart rate variability in prehypertension. J. Altern. Complement. Med. 2010, 16, 1039–1045. [Google Scholar] [CrossRef]
- Nolan, R.P.; Kamath, M.V.; Floras, J.S.; Stanley, J.; Pang, C.; Picton, P.; Young, Q.R. Heart rate variability biofeedback as a behavioral neurocardiac intervention to enhance vagal heart rate control. Am. Heart J. 2005, 149, 1137. [Google Scholar] [CrossRef]
- Seo, Y.; Yates, B.; LaFramboise, L.; Pozehl, B.; Norman, J.F.; Hertzog, M. A Home-Based Diaphragmatic Breathing Retraining in Rural Patients with Heart Failure. West. J. Nurs. Res. 2016, 38, 270–291. [Google Scholar] [CrossRef]
- Sutbeyaz, S.T.; Koseoglu, F.; Inan, L.; Coskun, O. Respiratory muscle training improves cardiopulmonary function and exercise tolerance in subjects with subacute stroke: A randomized controlled trial. Clin. Rehabil. 2010, 24, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Eherer, A.J.; Netolitzky, F.; Högenauer, C.; Pusching, G.; Hinterleitner, T.A.; Scheidl, S.; Kraxner, W.; Krejs, G.J.; Hoffmann, K.M. Positive effect of abdominal breathing exercise on gastroesophageal reflux disease: A randomized, controlled study. Am. J. Gastroenterol. 2012, 107, 372–378. [Google Scholar] [CrossRef]
- Kaushik, R.; Kaushik, R.M.; Mahajan, S.K.; Rajesh, V. Biofeedback assisted diaphragmatic breathing and systematic relaxation versus propranolol in long term prophylaxis of migraine. Complement. Ther. Med. 2005, 13, 165–174. [Google Scholar] [CrossRef]
- Chen, Y.F.; Huang, X.Y.; Chien, C.H.; Cheng, J.F. The Effectiveness of Diaphragmatic Breathing Relaxation Training for Reducing Anxiety. Perspect. Psychiatr. Care 2017, 53, 329–336. [Google Scholar] [CrossRef]
- Kavan, M.G.; Elsasser, G.; Barone, E.J. Generalized anxiety disorder: Practical assessment and management. Am. Fam Physician 2009, 79, 785–791. [Google Scholar] [PubMed]
- Liu, C.J.; Tu, G.Y.; Lin, S.L.; Yeh, C.C. Comparisons of different breathing ratios of slow breathing among outpatients with anxiety disorders. J. Med. Health 2014, 3, 57–66. [Google Scholar]
- Russell, M.E.; Hoffman, B.; Stromberg, S.; Carlson, C.R. Use of controlled diaphragmatic breathing for the management of motion sickness in a virtual reality environment. Appl. Psychophysiol. Biofeedback 2014, 39, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Gimenez, M.; Servera, E.; Abril, E.; Saavedra, P.; Darias, M.; Gomez, A.; Hannhart, B. Comprehensive directed breathing retraining improves exertional dyspnea for men with spirometry within normal limits. Am. J. Phys. Med. Rehabil. 2010, 89, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Cahalin, L.P.; Braga, M.; Matsuo, Y.; Hernandez, E.D. Efficacy of diaphragmatic breathing in persons with chronic obstructive pulmonary disease: A review of the literature. J. Cardiopulm. Rehabil. 2002, 22, 7–21. [Google Scholar] [CrossRef] [PubMed]
- Dechman, G.; Wilson, C.R. Evidence underlying cardiopulmonary physical therapy in stable COPD. Cardiopulmonary Phys. Ther. J. 2002, 13, 20–22. [Google Scholar] [CrossRef]
- Han, J.W.; Kim, Y.M. Effect of breathing exercises combined with dynamic upper extremity exercises on the pulmonary function of young adults. J. Back Musculoskelet. Rehabil. 2018, 31, 405–409. [Google Scholar] [CrossRef]
- Bahenský, P.; Malátová, R.; Bunc, V. Changed dynamic ventilation parameters as a result of a breathing exercise intervention program. J. Sports Med. Phys. Fit. 2019, 59, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- Sperlich, B.; Fricke, H.; de Marées, M.; Linville, J.W.; Mester, J. Does respiratory muscle training increase physical performance? Mil. Med. 2009, 174, 977–982. [Google Scholar] [CrossRef]
- Riganas, C.S.; Vrabas, I.S.; Christoulas, K.; Mandroukas, K. Specific inspiratory muscle training does not improve performance or VO2max levels in well trained rowers. J. Sports Med. Phys. Fit. 2008, 48, 285–292. [Google Scholar]
- Kolar, P.; Neuwirth, J.; Sanda, J.; Suchanek, V.; Svata, Z.; Volejnik, J.; Pivec, M. Analysis of diaphragm movement during tidal breathing and during its activation while breath holding using MRI synchronized with spirometry. Physiol. Res. 2009, 58, 383–392. [Google Scholar]
- Russo, M.A.; Santarelli, D.M.; O’Rourke, D. The physiological effects of slow breathing in the healthy human. Breathe 2017, 13, 298–309. [Google Scholar] [CrossRef]
- Bernardi, L.; Gabutti, A.; Porta, C.; Spicuzza, L. Slow breathing reduces chemoreflex response to hypoxia and hypercapnia, and increases baroreflex sensitivity. J. Hypertens. 2001, 19, 2221–2229. [Google Scholar] [CrossRef]
- Bilo, G.; Revera, M.; Bussotti, M.; Bonacina, D.; Styczkiewicz, K.; Caldara, G.; Giglio, A.; Faini, A.; Giuliano, A.; Lombardi, C.; et al. Effects of slow deep breathing at high altitude on oxygen saturation, pulmonary and systemic hemodynamics. PLoS ONE 2012, 7, e49074. [Google Scholar] [CrossRef]
- Billman, G.E. Heart rate variability—A historical perspective. Front. Physiol. 2011, 2, 86. [Google Scholar] [CrossRef] [PubMed]
- Dick, T.E.; Mims, J.R.; Hsieh, Y.H.; Morris, K.F.; Wehrwein, E.A. Increased cardio-respiratory coupling evoked by slow deep breathing can persist in normal humans. Respir. Physiol. Neurobiol. 2014, 204, 99–111. [Google Scholar] [CrossRef]
- Lee, J.S.; Lee, M.S.; Lee, J.Y.; Cornélissen, G.; Otsuka, K.; Halberg, F. Effects of diaphragmatic breathing on ambulatory blood pressure and heart rate. Biomed. Pharmacother. 2003, 57 (Suppl. S1), 87s–91s. [Google Scholar] [CrossRef]
- Chang, Q.; Liu, R.; Shen, Z. Effects of slow breathing rate on blood pressure and heart rate variabilities. Int. J. Cardiol. 2013, 169, e6–e8. [Google Scholar] [CrossRef] [PubMed]
- Thayer, J.F.; Yamamoto, S.S.; Brosschot, J.F. The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. Int. J. Cardiol. 2010, 141, 122–131. [Google Scholar] [CrossRef]
- Villemure, C.; Čeko, M.; Cotton, V.A.; Bushnell, M.C. Neuroprotective effects of yoga practice: Age-, experience-, and frequency-dependent plasticity. Front. Hum. Neurosci. 2015, 9, 281. [Google Scholar] [CrossRef]
- Jerath, R.; Crawford, M.W.; Barnes, V.A.; Harden, K. Self-regulation of breathing as a primary treatment for anxiety. Appl. Psychophysiol. Biofeedback 2015, 40, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Roi, G.S.; Mosconi, G.; Totti, V.; Angelini, M.L.; Brugin, E.; Sarto, P.; Merlo, L.; Sgarzi, S.; Stancari, M.; Todeschini, P.; et al. Renal function and physical fitness after 12-mo supervised training in kidney transplant recipients. World J. Transplant. 2018, 8, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Wickerson, L.; Rozenberg, D.; Janaudis-Ferreira, T.; Deliva, R.; Lo, V.; Beauchamp, G.; Helm, D.; Gottesman, C.; Mendes, P.; Vieira, L.; et al. Physical rehabilitation for lung transplant candidates and recipients: An evidence-informed clinical approach. World J. Transplant. 2016, 6, 517–531. [Google Scholar] [CrossRef] [PubMed]
| Authors, Year | Subjects | Included Studies | Primary Outcomes | Results |
|---|---|---|---|---|
| Holland et al., 2012 [42] | COPD | 16 RCTs | Dyspnea, exercise capacity, and health-related quality of life | Dyspnea↑ 6-min walk distance↑ |
| Ubolnuar et al., 2019 [44] | COPD | 19 RCTs | Ventilation, exercise capacity, dyspnea, and quality of life | Respiratory rate↓ Quality of life→ |
| Santino et al., 2020 [45] | Asthma | 22 RCTs | Quality of life | Unknown due to insufficient data |
| Prem et al., 2013 [47] | Asthma | 3 RCTs | Quality of life | Quality of life↑? (meta-analysis was not performed) |
| Macêdo et al., 2016 [48] | Asthma (children) | 3 RCTs | Quality of life, asthma symptoms, and adverse effects | Unknown due to insufficient data |
| Barker et al., 2013 [51] | Dysfunctional/hyperventilation syndrome (children) | No eligible studies | Quality of life | Unknown |
| Jones et al., 2013 [52] | Dysfunctional/hyperventilation syndrome (adults) | 1 RCT | Quality of life and adverse effects | Unknown due to insufficient data |
| Lewis et al., 2007 [54] | Chronic respiratory disease, post-surgical, or asymptomatic individuals | 2 RCTs, 3 non-RCTs, and 15 quasi-experimental studies | Short-term clinical outcomes (not specified) | Abdominal excursion↑, diaphragm excursion↑, respiratory rate↓, tidal volume↑, arterial oxygen saturation↑, percutaneous oxygen↑ Work of breathing↑, dyspnea↑ |
| Grams et al., 2012 [74] | Post upper abdominal surgery | 6 RCTs or quasi-RCTs | Respiratory function and postoperative complications | Respiratory muscle strength↑ Respiratory function→ |
| Hopper et al., 2019 [76] | 1 RCT and 2 quasi-experimental studies | Stress | Stress↓ (meta-analysis was not performed) |
| Authors, Year | Subjects | Intervention Study Duration | Results |
|---|---|---|---|
| Yekefallah et al., 2019 [80] | 75 patients with COPD | Breathing exercise (DB and pursed-lip breathing) and upper limb exercise One month | 6-min walking distance↑ |
| Shaw and Shaw, 2011 [81] | 88 patients with asthma | DB, aerobic exercise, and aerobic exercise plus DB 8 weeks | FVC↑, FEV1↑, FEV1/FVC ratio↓, PEF↑, FEF rate→, MVV→, VT→ |
| Campbell et al., 1984 [82] | 22 patients with cancer (except breast cancer) | Relaxation technique including DB 6 weeks | Desirable weight gain Improvement in Performance Status |
| Shahirai et al., 2017 [83] | 50 elderly patients with breast or prostate cancer | Muscle relaxation, guided imagery, and DB 6weeks | Quality of life↑ |
| Silva and Motta, 2013 [84] | 72 pediatric patients with chronic functional constipation | Isometric training of the abdominal muscle, DB, and abdominal massage 6 weeks | Defecation frequency↑ Fecal incontinence→ |
| Wang et al., 2010 [85] | 22 postmenopausal women with prehypertention | DB and DB with frontal electromyographic biofeedback training | Systolic blood pressure↓ |
| Seo et al., 2016 [87] | 29 patients with heart failure | Home-based DB retraining 8 weeks | Dyspnea↓ Daily physical activity↑ Functional status↑ |
| Sutbeyaz et al., 2010 [88] | 45 inpatients with subacute stroke | DB combined with pursed-lip breathing and inspiratory muscle training 6 weeks | PEF ↑, VC→, FVC→, FEV1→, FEF25–75%→, MVV→ |
| Eherer et al., 2012 [89] | 19 patients with non-erosive gastroesophageal reflux disease | DB 4 weeks | Time with a pH < 4.0↓ Quality of life↑ |
| Kaushik et al., 2005 [90] | 167 patients with migraine | DB with biofeedback and 80 mg/day of propranolol 6 months | Resurgence of migraine↓ |
| Chen et al., 2017 [91] | 30 patients with anxiety | DB 8 weeks | Anxiety score↓, peripheral temperature↑, heart rate↓, breathing rate↓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamasaki, H. Effects of Diaphragmatic Breathing on Health: A Narrative Review. Medicines 2020, 7, 65. https://doi.org/10.3390/medicines7100065
Hamasaki H. Effects of Diaphragmatic Breathing on Health: A Narrative Review. Medicines. 2020; 7(10):65. https://doi.org/10.3390/medicines7100065
Chicago/Turabian StyleHamasaki, Hidetaka. 2020. "Effects of Diaphragmatic Breathing on Health: A Narrative Review" Medicines 7, no. 10: 65. https://doi.org/10.3390/medicines7100065
APA StyleHamasaki, H. (2020). Effects of Diaphragmatic Breathing on Health: A Narrative Review. Medicines, 7(10), 65. https://doi.org/10.3390/medicines7100065





