In Vitro Effect of the Traditional Medicine Hainosan (Painongsan) on Porphyromonas gingivalis
Abstract
1. Introduction
2. Materials and Methods
2.1. Crude Drugs and Chemicals
2.2. Bacterial Strains and Culture Conditions
2.3. Growth-Inhibitory Analysis
2.4. Time-Kill Analysis
2.5. Morphological Analysis
2.6. Statistical Analysis
3. Results
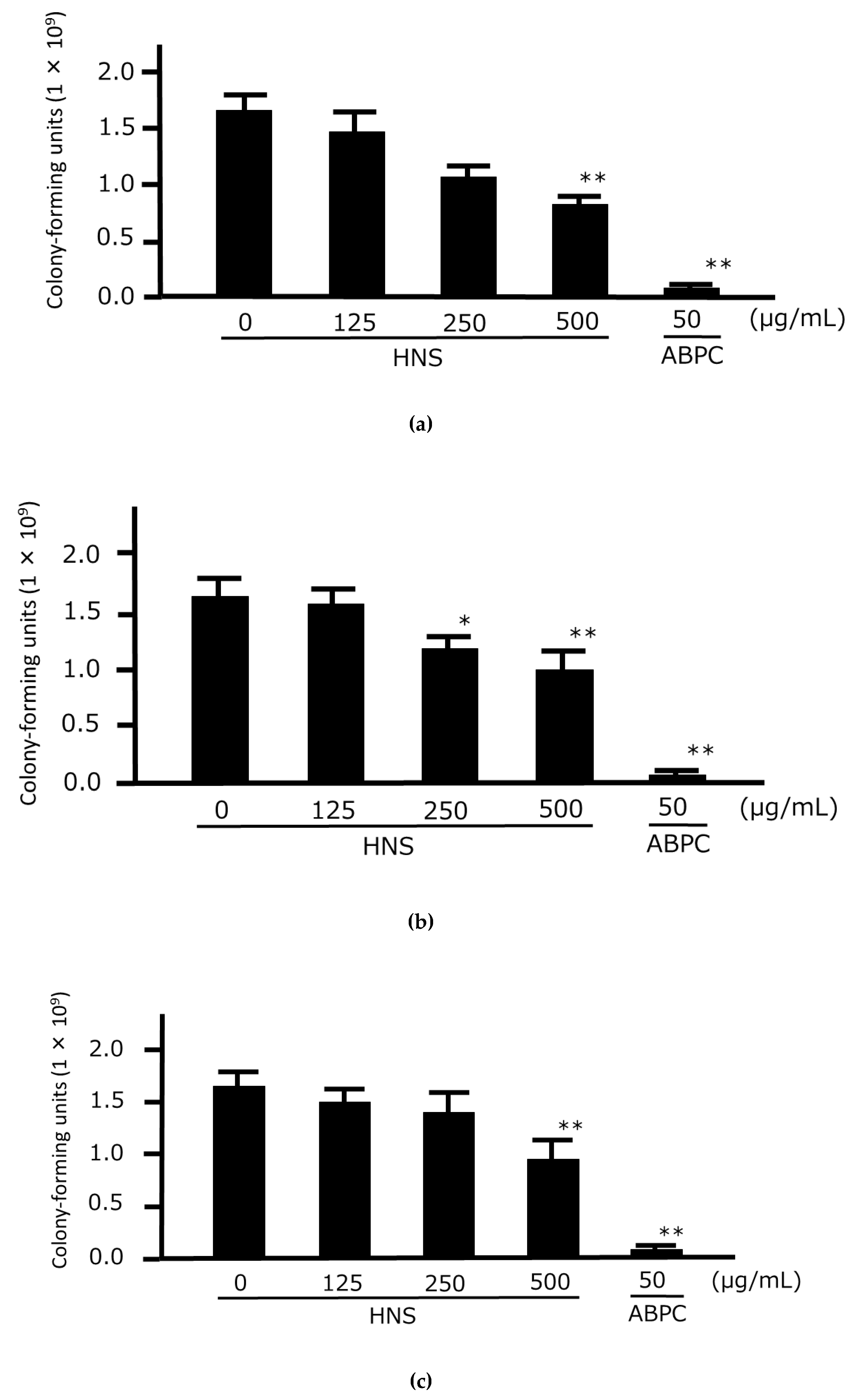
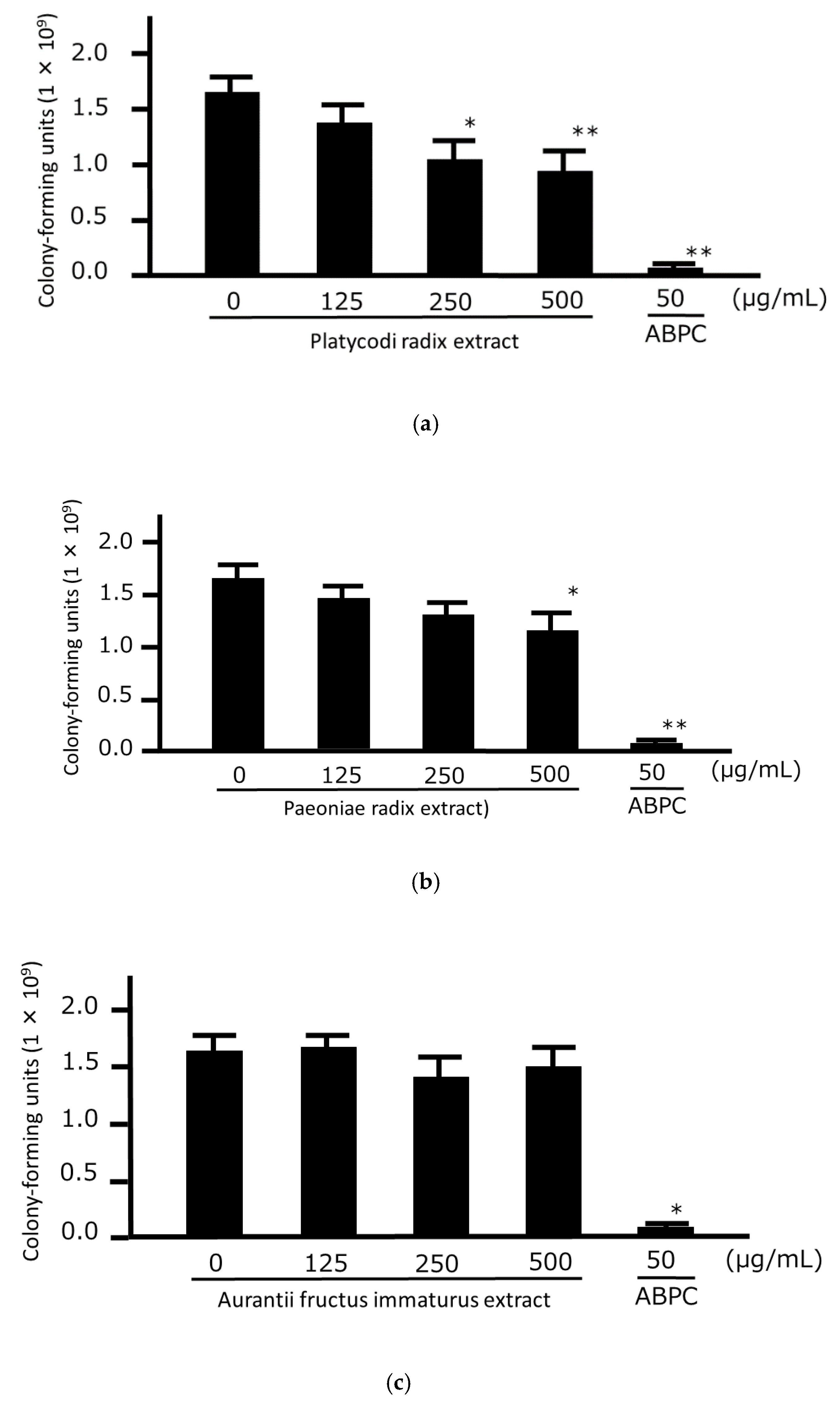
3.1. Growth-Inhibitory Analysis of HNS and the Crude Drug Extracts
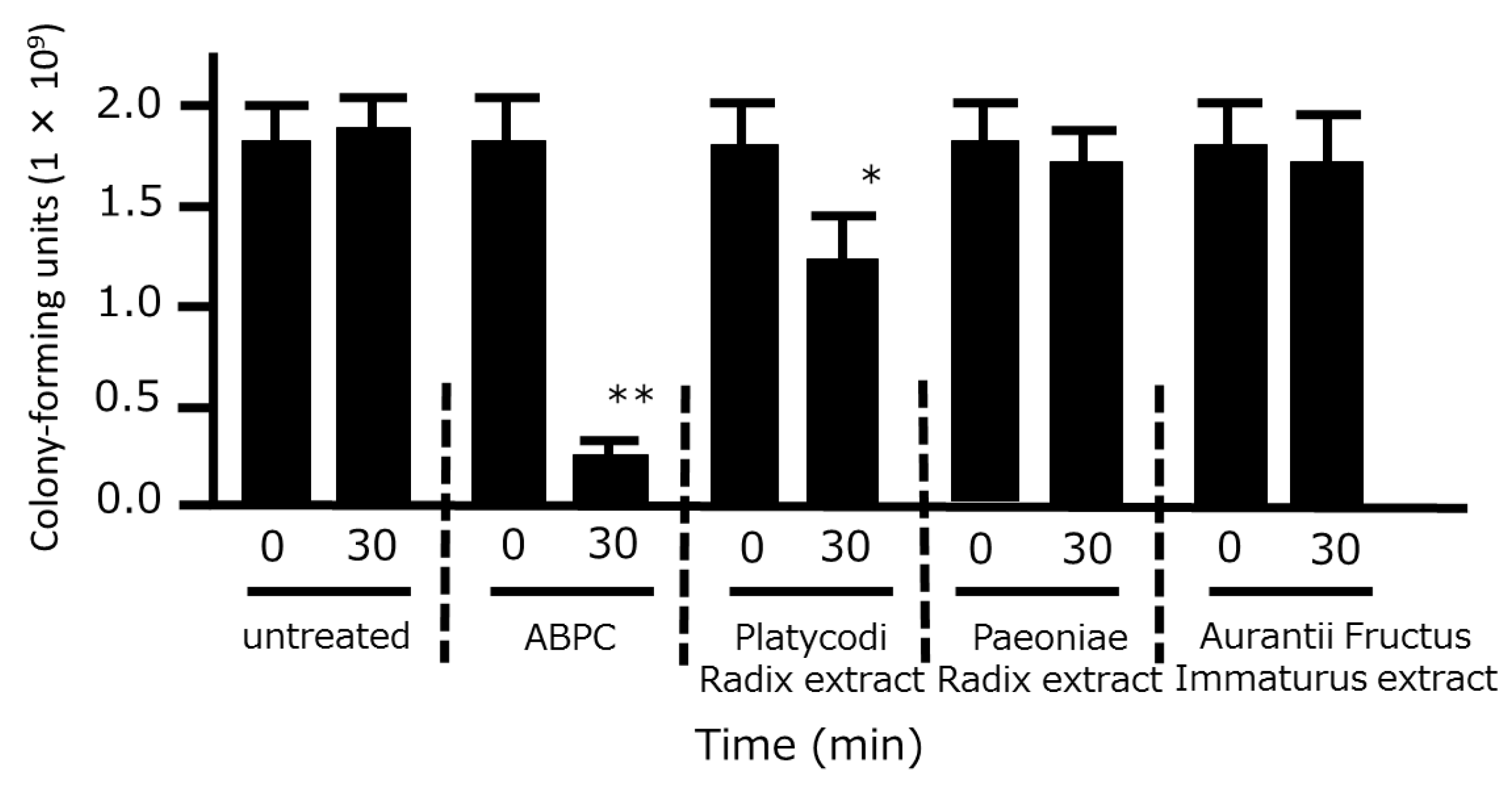
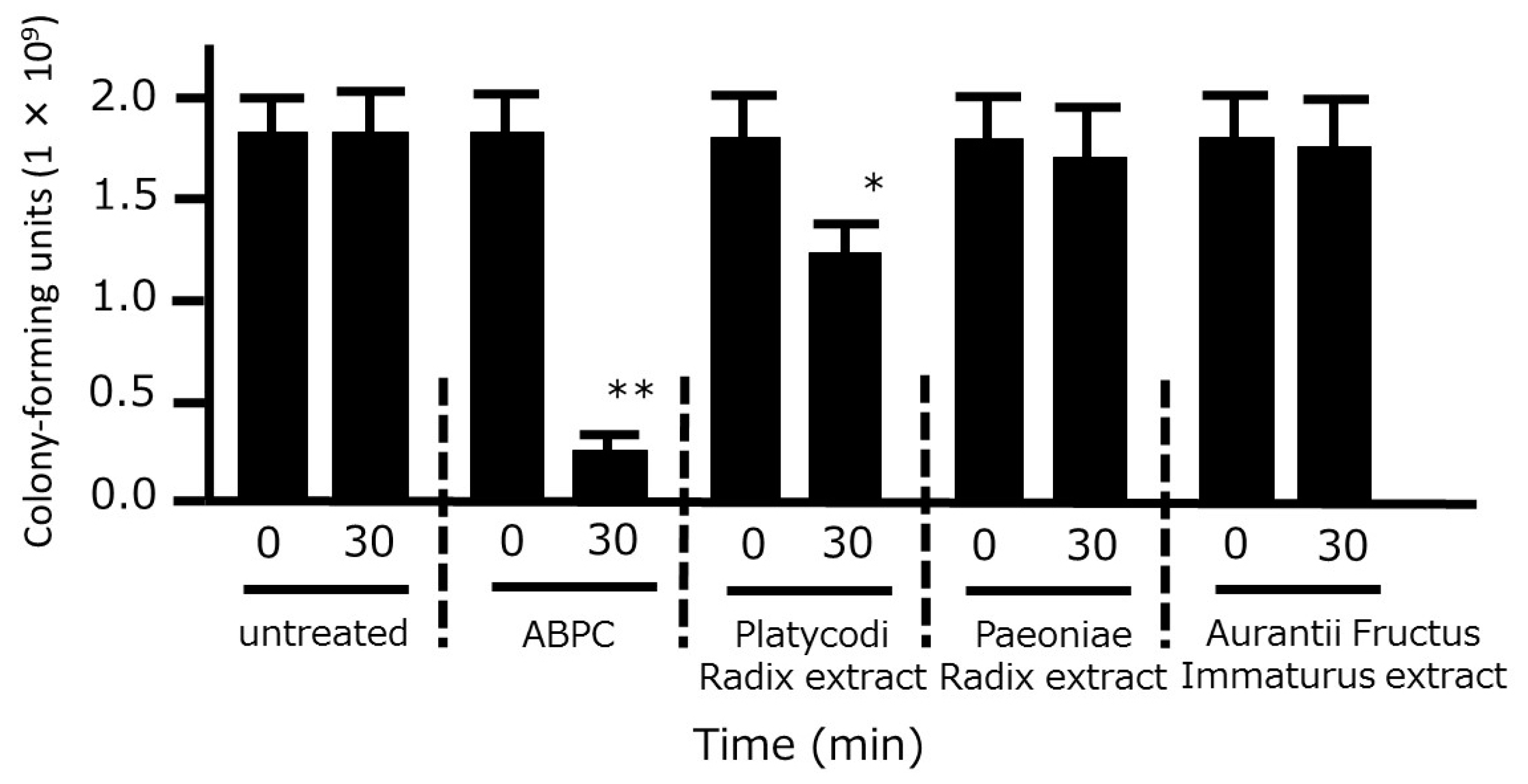
3.2. Time-Kill Analysis of the Crude Drug Extracts
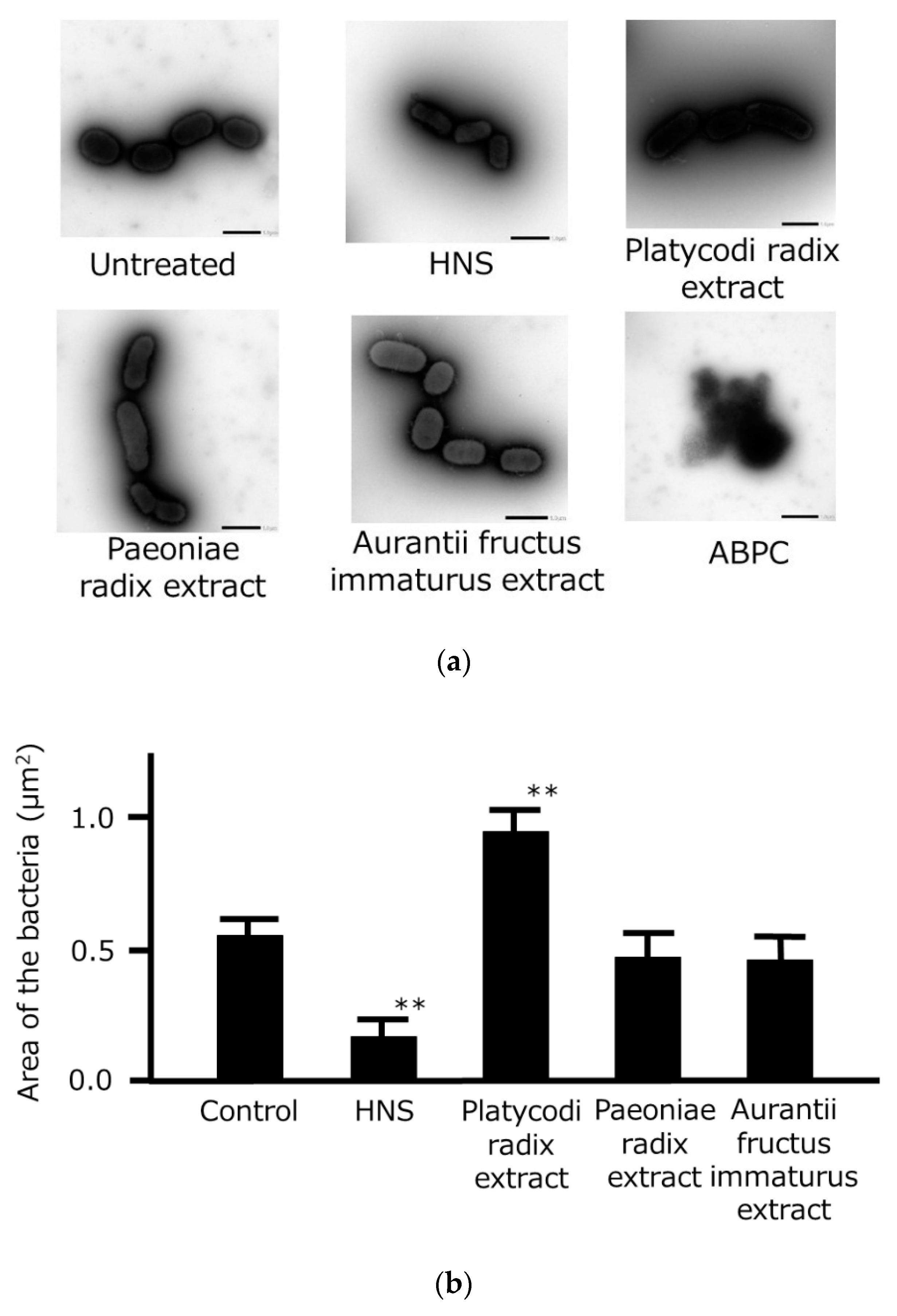
3.3. Morphological Effects of the Crude Drug Extracts
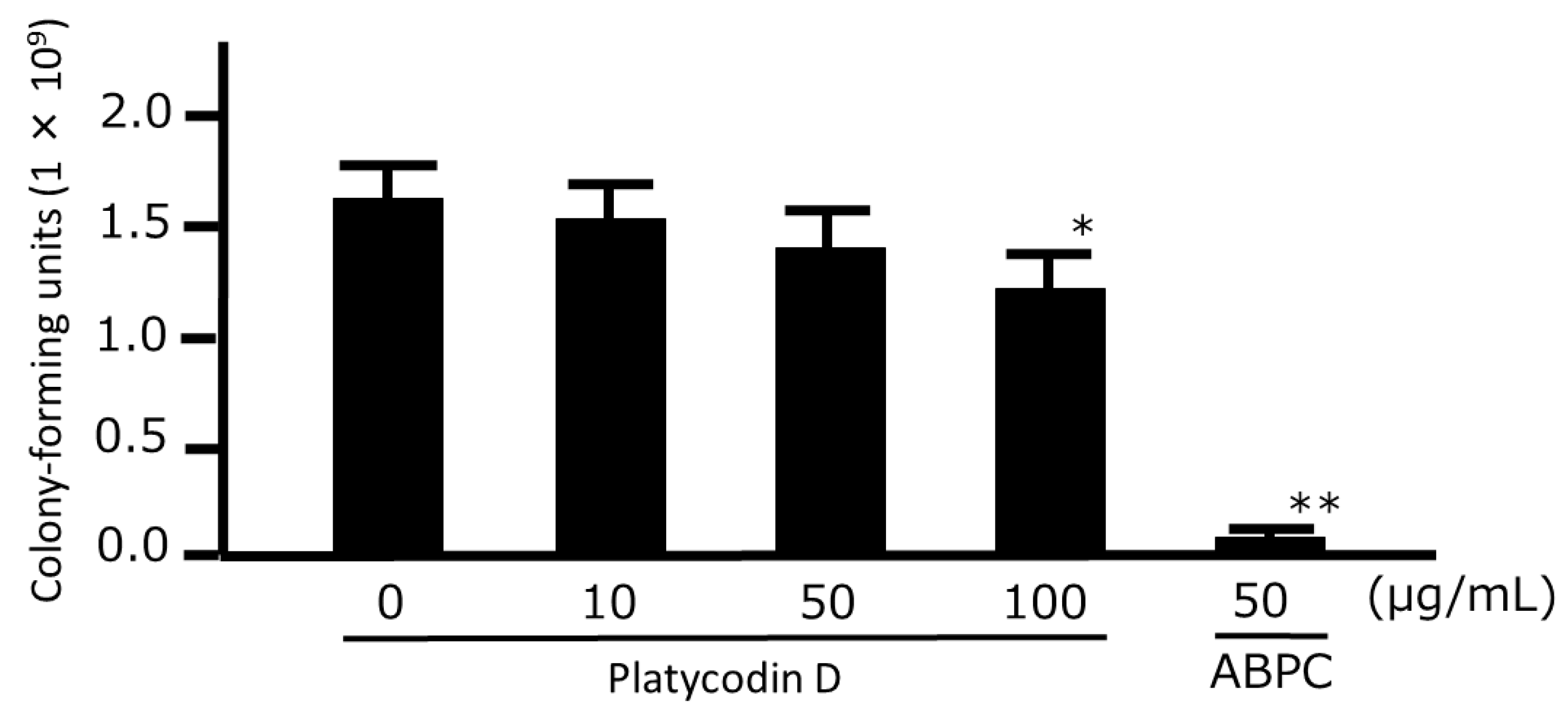
3.4. Growth-Inhibitory and Time-Kill Analyses of Platycodin D
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ikai, R.; Hasegawa, Y.; Izumigawa, M.; Nagano, K.; Yoshida, Y.; Kitai, N.; Lamont, R.J.; Yoshimura, F.; Murakami, Y. Mfa4, an accessory protein of Mfa1 fimbriae, modulates fimbrial biogenesis, cell auto-aggregation, and biofilm formation in Porphyromonas gingivalis. PLoS ONE 2015, 10, e0139454. [Google Scholar] [CrossRef]
- Barbosa, G.M.; Colombo, A.V.; Rodrigues, P.H.; Simionato, M.R. Intraspecies variability affects heterotypic biofilms of Porphyromonas gingivalis and Prevotella intermedia: Evidences of strain-dependence biofilm modulation by physical contact and by released soluble factors. PLoS ONE 2015, 10, e0138687. [Google Scholar] [CrossRef]
- Ramsey, M.M.; Rumbaugh, K.P.; Whiteley, M. Metabolite cross-feeding enhances virulence in a model polymicrobial infection. PLoS Pathog. 2011, 7, e1002012. [Google Scholar] [CrossRef] [PubMed]
- Ozaki, Y. Studies on antiinflammatory effect of Japanese Oriental medicines (kampo medicines) used to treat inflammatory diseases. Biol. Pharm. Bull. 1995, 18, 559–562. [Google Scholar] [CrossRef][Green Version]
- Okamura, N.; Maki, T.; Ishida, S.; Uraguchi, C.; Onishi, Y.; Sadasue, E.; Tsuruta, Y.; Yagi, A. Dissolution profiles of principal ingredients in Kampo medicinal powders by high-performance liquid chromatography. Chem. Pharm. Bull. 2000, 48, 1782–1785. [Google Scholar] [CrossRef] [PubMed]
- Brook, I. Antimicrobials therapy of anaerobic infections. J. Chemother. 2016, 28, 143–150. [Google Scholar] [CrossRef]
- Montagner, F.; Jacinto, R.C.; Correa Signoretti, F.G.; Scheffer de Mattos, V.; Grecca, F.S.; Gomes, B.P. β-lactamic resistance profiles in Porphyromonas, Prevotella, and Parvimonas species isolated from acute endodontic infections. J. Endod. 2014, 40, 339–344. [Google Scholar] [CrossRef]
- Bahar, H.; Torun, M.M.; Demirci, M.; Kocazeybek, B. Antimicrobial resistance and β-lactamase production of clinical isolates of prevotella and porphyromonas species. Chemotherapy 2005, 51, 9–14. [Google Scholar] [CrossRef]
- Shi, W.; Wen, D.; Chen, C.; Yuan, L.; Gao, W.; Tang, P.; Cheng, X.; Yao, K. β-lactamase production and antibiotic susceptibility pattern of Moraxella catarrhalis isolates collected from two county hospitals in China. BMC Microbiol. 2018, 18, 77. [Google Scholar] [CrossRef]
- Sugano, N. Biological plaque control: Novel therapeutic approach to periodontal disease. J. Oral. Sci. 2012, 54, 1–5. [Google Scholar] [CrossRef]
- Japan Kampo Medicines Manufactures Association (Ed.) Under the Super Vision of National Institute of Health Sciences, the Affiliated Institutions of the Ministry of Health, Labour and Welfare of Japan; Handbook on OTC Medicinal Product in Kampo; Jiho Inc.: Tokyo, Japan, 2013; ISBN 484074498X. (In Japanese)
- Minami, M.; Takase, H.; Taira, M.; Makino, T. Hainosan (painongsan) suppresses the biofilm formation of Porphyromonas gingivalis and Prevotella intermedia in vitro. Tradit. Kampo Med. 2019, (in press). [CrossRef]
- Pharmaceutical and Medical Device Regulatory Science Society of Japan. Japanese Pharmacopoeia, 17th ed.; Jiho: Tokyo, Japan, 2016. [Google Scholar]
- Watanabe, T.; Maruyama, F.; Nozawa, T.; Aoki, A.; Okano, S.; Shibata, Y.; Oshima, K.; Kurokawa, K.; Hattori, M.; Nakagawa, I.; et al. Complete genome sequence of the bacterium Porphyromonas gingivalis TDC60, which causes periodontal disease. J. Bacteriol. 2011, 193, 4259–4260. [Google Scholar] [CrossRef]
- Fukamachi, H.; Matsumoto, C.; Omiya, Y.; Arimoto, T.; Morisaki, H.; Kataoka, H.; Kadena, M.; Funatsu, T.; Fukutake, M.; Kase, Y.; et al. Effects of hangeshashinto on growth of oral microorganisms. Evid. Based Complement. Alternat. Med. 2015, 2015, 512947. [Google Scholar] [CrossRef]
- Minami, M.; Konishi, T.; Takase, H.; Makino, T. Shin’iseihaito (Xinyiqingfeitang) suppresses the biofilm formation of Streptococcus pneumoniae in vitro. Biomed. Res. Int. 2017, 2017, 4575709. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to Image J: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Saeki, T.; Nikaido, T. Evaluations of saponin properties of HPLC analysis of Platycodon grandiflorum A.DC. Yakugaku Zasshi. 2003, 123, 431–441. [Google Scholar] [CrossRef]
- Lee, K.-Y.; Cha, J.-D.; Choi, S.-M.; Jang, E.-J.; Ko, E.-S.; Cha, S.-M.; Yun, S.-I. Antibacterial activity of the ethanol extract of Paeonia lactiflora on growth of oral bacteria. J. Oral Health Dent. 2018, 2, 201. [Google Scholar] [CrossRef]
- Tsui, V.W.; Wong, R.W.; Rabie, A.B. The inhibitory effects of naringin on the growth of periodontal pathogens in vitro. Phytother. Res. 2008, 22, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Ara, T.; Maeda, Y.; Fujinami, Y.; Imamura, Y.; Hattori, T.; Wang, P.L. Preventive effects of a kampo medicine, shosaikoto, on inflammatory responses in LPS-treated human gingival fibroblasts. Biol. Pharm. Bull. 2008, 31, 1141–1144. [Google Scholar] [CrossRef]
- Ara, T.; Honjo, K.; Fujinami, Y.; Hattori, T.; Imamura, Y.; Wang, P.L. Preventive effects of a kampo medicine, orento on inflammatory responses in lipopolysaccharide treated human gingival fibroblasts. Biol. Pharm. Bull. 2010, 33, 611–616. [Google Scholar] [CrossRef]
- Takeda, O.; Toyama, T.; Watanabe, K.; Sato, T.; Sasaguri, K.; Akimoto, S.; Sato, S.; Kawata, T.; Hamada, N. Ameliorating effects of Juzentaihoto on restraint stress and P. gingivalis-induced alveolar bone loss. Arch. Oral Biol. 2014, 59, 1130–1138. [Google Scholar] [CrossRef]
- Liao, J.; Azelmat, J.; Zhao, L.; Yoshioka, M.; Hinode, D.; Grenier, D. The kampo medicine rokumigan possesses antibiofilm, anti-inflammatory and wound healing properties. BioMed Res. Int. 2014, 2014, 436206. [Google Scholar] [CrossRef]
- Liao, J.; Zhao, L.; Yoshioka, M.; Hinode, D.; Grenier, D. Japanese traditional herbal medicines (Kampo) on growth and virulence properties of Porphyromonas gingivalis and viability of oral epithelial cells. Pharm. Biol. 2013, 51, 1538–1544. [Google Scholar] [CrossRef]
- Patra, J.K.; Kim, E.S.; Oh, K.; Kim, H.J.; Dhakal, R.; Kim, Y.; Baek, K.H. Bactericidal effect of extracts and metabolites of Robinia pseudoacacia L. on Streptococcus mutans and Porphyromonas gingivalis causing dental plaque and periodontal inflammatory diseases. Molecules 2015, 20, 6128–6139. [Google Scholar] [CrossRef] [PubMed]
- Patra, J.K.; Kim, E.S.; Oh, K.; Kim, H.J.; Kim, Y.; Baek, K.H. Antibacterial effect of crude extract and metabolites of Phytolacca americana on pathogens responsible for periodontal inflammatory diseases and dental caries. BMC Complement. Altern. Med. 2014, 14, 343. [Google Scholar] [CrossRef] [PubMed]
- Lien, H.M.; Tseng, C.J.; Huang, C.L.; Lin, Y.T.; Chen, C.C.; Lai, Y.Y. Antimicrobial activity of Antrodia camphorata extracts against oral bacteria. PLoS ONE 2014, 9, e105286. [Google Scholar] [CrossRef]
- Cai, L.; Wu, C.D. Compounds from Syzygium aromaticum possessing growth inhibitory activity against oral pathogens. J. Nat. Prod. 1996, 59, 987–990. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Minami, M.; Takase, H.; Taira, M.; Makino, T. In Vitro Effect of the Traditional Medicine Hainosan (Painongsan) on Porphyromonas gingivalis. Medicines 2019, 6, 58. https://doi.org/10.3390/medicines6020058
Minami M, Takase H, Taira M, Makino T. In Vitro Effect of the Traditional Medicine Hainosan (Painongsan) on Porphyromonas gingivalis. Medicines. 2019; 6(2):58. https://doi.org/10.3390/medicines6020058
Chicago/Turabian StyleMinami, Masaaki, Hiroshi Takase, Masayo Taira, and Toshiaki Makino. 2019. "In Vitro Effect of the Traditional Medicine Hainosan (Painongsan) on Porphyromonas gingivalis" Medicines 6, no. 2: 58. https://doi.org/10.3390/medicines6020058
APA StyleMinami, M., Takase, H., Taira, M., & Makino, T. (2019). In Vitro Effect of the Traditional Medicine Hainosan (Painongsan) on Porphyromonas gingivalis. Medicines, 6(2), 58. https://doi.org/10.3390/medicines6020058





