Simulated Wildfire Smoke Significantly Alters Sperm DNA Methylation Patterns in a Murine Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.1.1. Ethics Statement
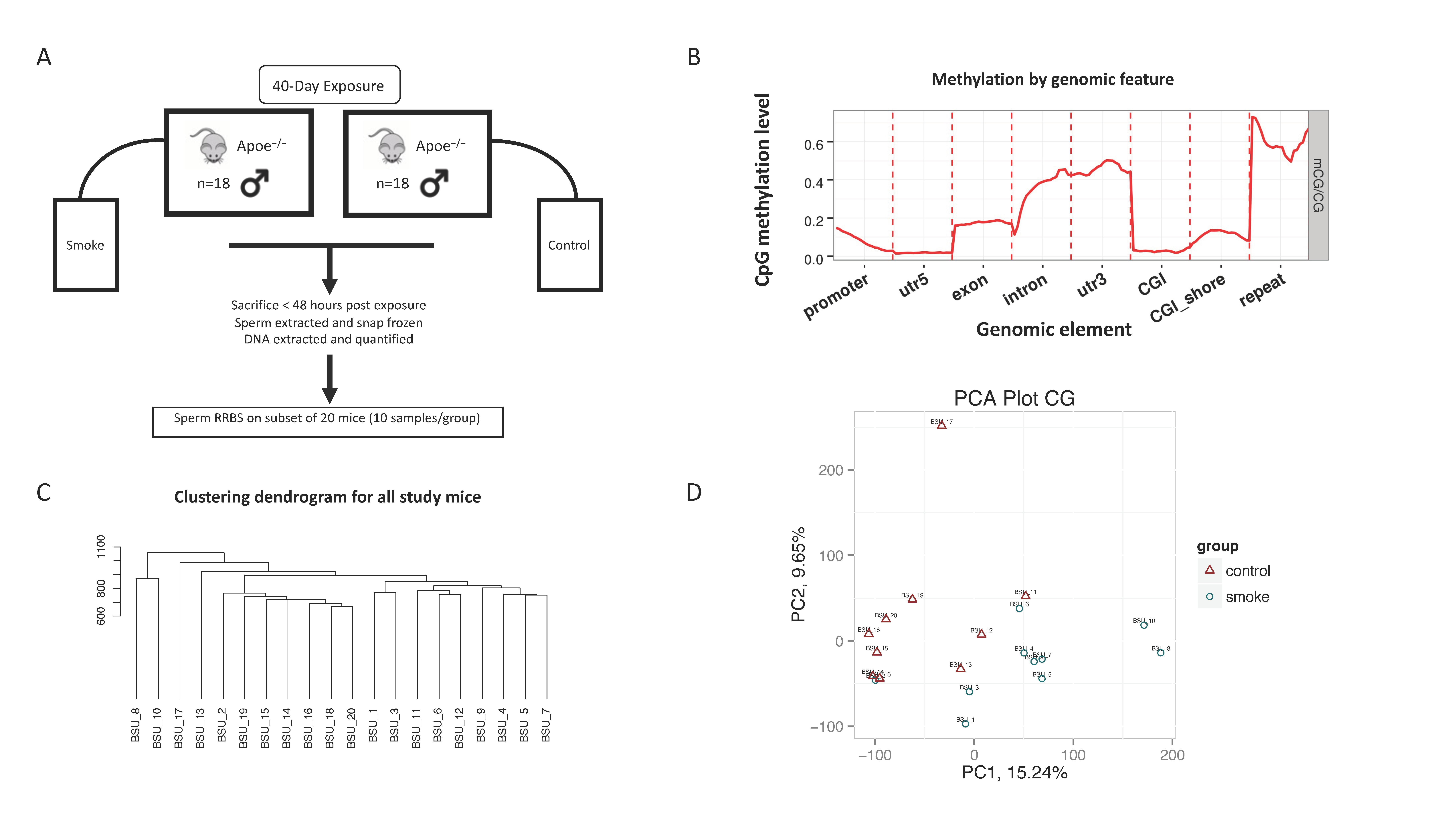
2.1.2. Experimental Design
2.1.3. Douglas Fir Smoke Exposure
2.2. Molecular Analyses
2.2.1. Sperm Collection and DNA Extraction
2.2.2. RRBS Library Construction
2.2.3. Bioinformatics Analyses
2.2.4. Gene Ontology
3. Results
3.1. Overview of DNA Methylation Characteristics
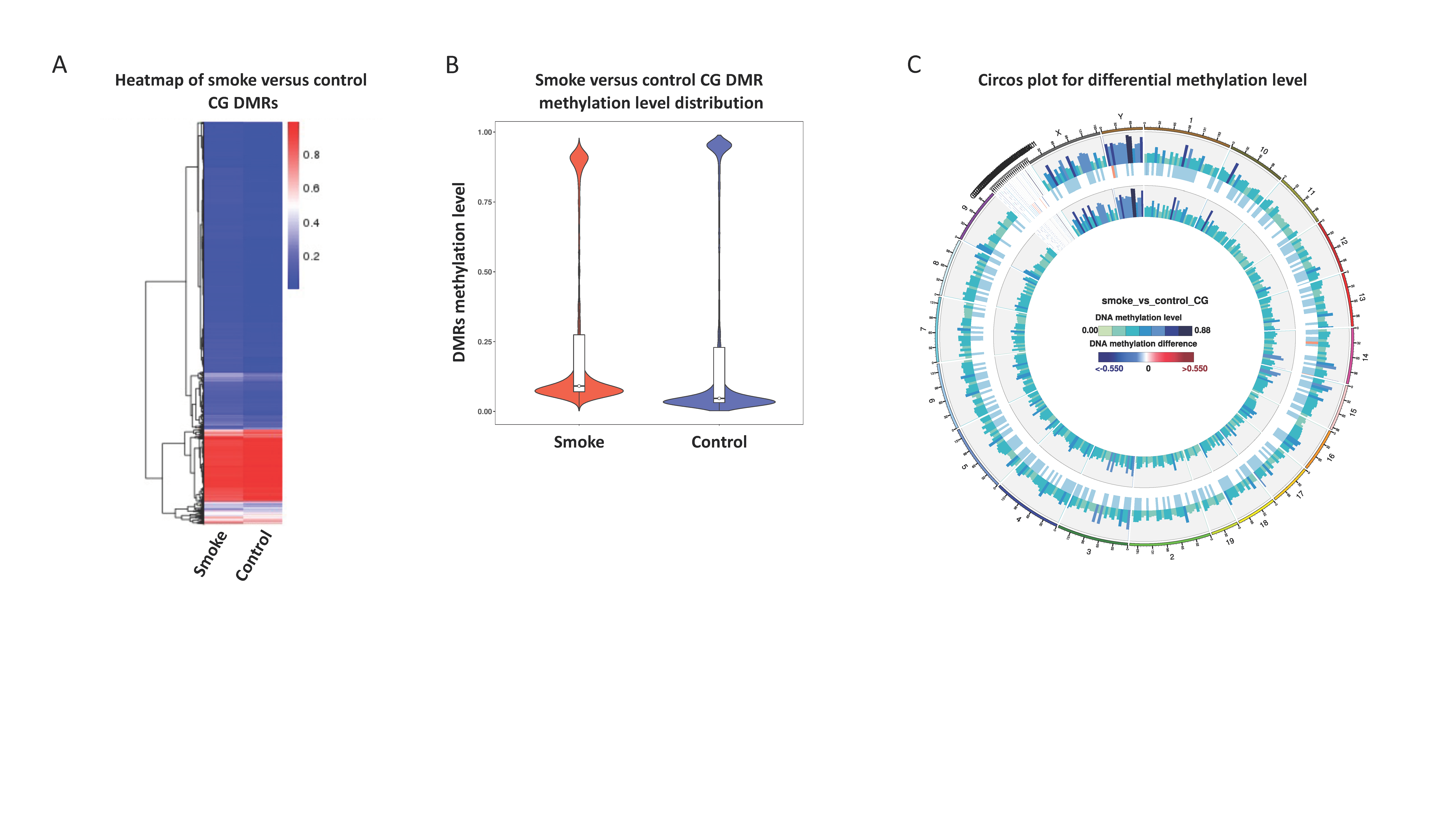
3.2. Simulated Wildfire Smoke-Induced Changes in Sperm DNA Methylation
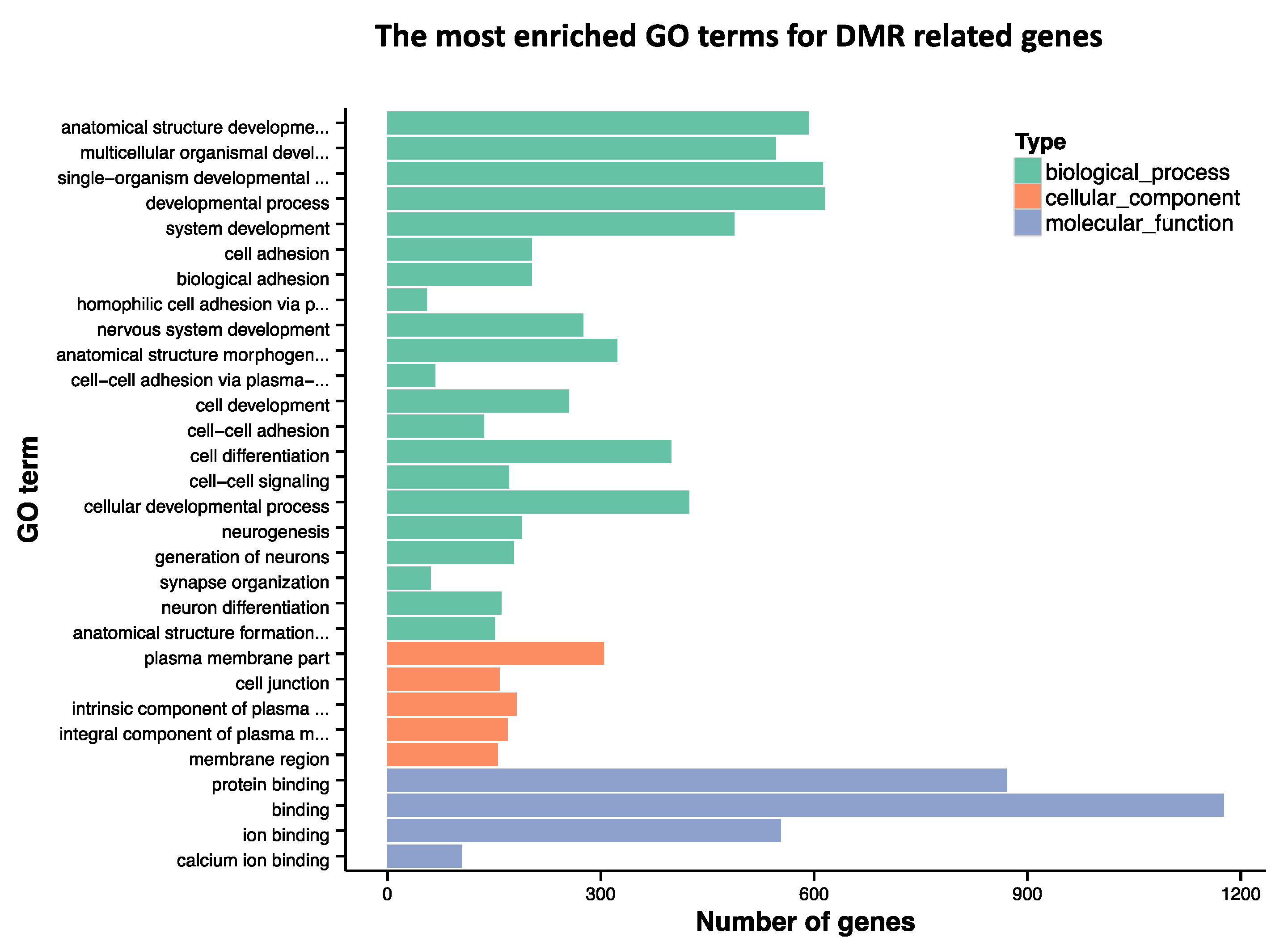
3.3. GO Term Analysis for DMR-Annotated Genes
3.4. GO Term Analysis for DMRs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adetona, O.; Reinhardt, T.E.; Domitrovich, J.; Broyles, G.; Adetona, A.M.; Kleinman, M.T.; Ottmar, R.D.; Naeher, L.P. Review of the health effects of wildland fire smoke on wildland firefighters and the public. Inhal. Toxicol. 2016, 28, 95–139. [Google Scholar] [CrossRef]
- McClure, C.D.; Jaffe, D.A. US particulate matter air quality improves except in wildfire-prone areas. Proc. Natl. Acad. Sci. USA 2018, 115, 7901–7906. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.C.; Mickley, L.J.; Sulprizio, M.P.; Dominici, F.; Yue, X.; Ebisu, K.; Anderson, G.B.; Khan, R.F.A.; Bravo, M.A.; Bell, M.L. Particulate air pollution from wildfires in the Western US under climate change. Clim. Chang. 2016, 138, 655–666. [Google Scholar] [CrossRef] [Green Version]
- Reid, J.S.; Koppmann, R.; Eck, T.F.; Eleuterio, D.P. A review of biomass burning emissions part II: Intensive physical properties of biomass burning particles. Atmos. Chem. Phys. 2005, 5, 799–825. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.C.; Pereira, G.; Uhl, S.A.; Bravo, M.A.; Bell, M.L. A systematic review of the physical health impacts from non-occupational exposure to wildfire smoke. Environ. Res. 2015, 136, 120–132. [Google Scholar] [CrossRef] [Green Version]
- Oudin, A.; Segersson, D.; Adolfsson, R.; Forsberg, B. Association between air pollution from residential wood burning and dementia incidence in a longitudinal study in Northern Sweden. PLoS ONE 2018, 13, e0198283. [Google Scholar] [CrossRef] [Green Version]
- Schuller, A.; Montrose, L. Influence of Woodsmoke Exposure on Molecular Mechanisms Underlying Alzheimer’s Disease: Existing Literature and Gaps in Our Understanding. Genet. Epigenet. 2020, 13. [Google Scholar] [CrossRef]
- Willson, B.E.; Pinkerton, K.E.; Lasley, B.; Gee, N. Effect of wildfire smoke on pregnancy outcomes in the non-human primate. Fertil. Steril. 2019, 112, e13. [Google Scholar] [CrossRef]
- Aguilera, R.; Corringham, T.; Gershunov, A.; Benmarhnia, T. Wildfire smoke impacts respiratory health more than fine particles from other sources: Observational evidence from Southern California. Nat. Commun. 2021, 12, 1493. [Google Scholar] [CrossRef] [PubMed]
- Ford, B.; Martin, M.V.; Zelasky, S.E.; Fischer, E.V.; Anenberg, S.C.; Heald, C.L.; Pierce, J.R. Future Fire Impacts on Smoke Concentrations, Visibility, and Health in the Contiguous United States. GeoHealth 2018, 2, 229–247. [Google Scholar] [CrossRef] [Green Version]
- Heßelbach, K.; Kim, G.-J.; Flemming, S.; Häupl, T.; Bonin, M.; Dornhof, R.; Günther, S.; Merfort, I.; Humar, M. Disease relevant modifications of the methylome and transcriptome by particulate matter (PM2.5) from biomass combustion. Epigenetics 2017, 12, 779–792. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Law, J.A.; Jacobsen, S.E. Establishing, maintaining and modifying DNA methylation patterns in plants and animals. Nat. Rev. Genet. 2010, 11, 204–220. [Google Scholar] [CrossRef]
- Tang, Q.; Pan, F.; Yang, J.; Fu, Z.; Lu, Y.; Wu, X.; Han, X.; Chen, M.; Lu, C.; Xia, Y.; et al. Idiopathic male infertility is strongly associated with aberrant DNA methylation of imprinted loci in sperm: A case-control study. Clin. Epigenet. 2018, 10, 134. [Google Scholar] [CrossRef]
- Sosedova, L.M.; Vokina, V.A.; Novikov, M.A.; Rukavishnikov, V.S.; Andreeva, E.S.; Zhurba, O.M.; Alekseenko, A.N. Paternal Biomass Smoke Exposure in Rats Produces Behavioral and Cognitive Alterations in the Offspring. Toxics 2020, 9, 3. [Google Scholar] [CrossRef]
- Jang, H.S.; Shin, W.J.; Lee, J.E.; Do, J.T. CpG and Non-CpG Methylation in Epigenetic Gene Regulation and Brain Function. Genes 2017, 8, 148. [Google Scholar] [CrossRef] [Green Version]
- Sasso, G.L.; Schlage, W.K.; Boué, S.; Veljkovic, E.; Peitsch, M.C.; Hoeng, J. The Apoe(-/-) mouse model: A suitable model to study cardiovascular and respiratory diseases in the context of cigarette smoke exposure and harm reduction. J. Transl. Med. 2016, 14, 146. [Google Scholar] [CrossRef] [Green Version]
- Garg, P.; Roche, T.; Eden, M.; Matz, J.; Oakes, J.M.; Bellini, C.; Gollner, M.J. Effect of moisture content and fuel type on emissions from vegetation using a steady state combustion apparatus. Int. J. Wildland Fire 2021. submitted. [Google Scholar]
- Meissner, A.; Mikkelsen, T.S.; Gu, H.; Wernig, M.; Hanna, J.; Sivachenko, A.; Zhang, X.; Bernstein, B.E.; Nusbaum, C.; Jaffe, D.B.; et al. Genome-scale DNA methylation maps of pluripotent and differentiated cells. Nature 2008, 454, 766–770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krueger, F.; Andrews, S.R. Bismark: A flexible aligner and methylation caller for Bisulfite-Seq applications. Bioinformatics 2011, 27, 1571–1572. [Google Scholar] [CrossRef]
- Gifford, C.A.; Ziller, M.J.; Gu, H.; Trapnell, C.; Donaghey, J.; Tsankov, A.; Shalek, A.K.; Kelley, D.R.; Shishkin, A.A.; Issner, R.; et al. Transcriptional and epigenetic dynamics during specification of human embryonic stem cells. Cell 2013, 153, 1149–1163. [Google Scholar] [CrossRef] [Green Version]
- Habibi, E.; Brinkman, A.B.; Arand, J.; Kroeze, L.I.; Kerstens, H.H.D.; Matarese, F.; Lepikhov, K.; Gut, M.; Brun-Heath, I.; Hubner, N.C.; et al. Whole-genome bisulfite sequencing of two distinct interconvertible DNA methylomes of mouse embryonic stem cells. Cell Stem Cell 2013, 13, 360–369. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.; Xu, T.; Feng, H.; Chen, L.; Li, B.; Yao, B.; Qin, Z.; Jin, P.; Conneely, K.N. Detection of differentially methylated regions from whole-genome bisulfite sequencing data without replicates. Nucleic Acids Res. 2015, 43, e141. [Google Scholar] [CrossRef] [Green Version]
- Gene Ontology Overview. Gene Ontology Resource. Available online: http://geneontology.org/docs/ontology-documentation/ (accessed on 19 July 2021).
- McLean, C.Y.; Bristor, D.; Hiller, M.; Clarke, S.L.; Schaar, B.T.; Lowe, C.B.; Wenger, A.M.; Bejerano, G. GREAT improves functional interpretation of cis -regulatory regions. Nat. Biotechnol. 2010, 28, 495–501. [Google Scholar] [CrossRef] [Green Version]
- Jarmasz, J.S.; Stirton, H.; Davie, J.R.; del Bigio, M.R. DNA methylation and histone post-translational modification stability in post-mortem brain tissue. Clin. Epigenet. 2019, 11, 5. [Google Scholar] [CrossRef] [Green Version]
- Jin, B.; Li, Y.; Robertson, K.D. DNA Methylation. Genes Cancer 2011, 2, 607–617. [Google Scholar] [CrossRef] [Green Version]
- Jaenisch, R.; Bird, A. Epigenetic regulation of gene expression: How the genome integrates intrinsic and environmental signals. Nat. Genet. 2003, 33, 245–254. [Google Scholar] [CrossRef]
- Monk, M.; Salpekar, A. Expression of imprinted genes in human preimplantation development. Mol. Cell Endocrinol. 2001, 183 (Suppl. 1), S35–S40. [Google Scholar] [CrossRef]
- Egger, G.; Liang, G.; Aparicio, A.; Jones, P.A. Epigenetics in human disease and prospects for epigenetic therapy. Nature 2004, 429, 457–463. [Google Scholar] [CrossRef]
- Schrott, R.; Rajavel, M.; Acharya, K.; Huang, Z.; Acharya, C.; Hawkey, A.; Pippen, E.; Lyerly, K.; Levin, E.D.; Murphy, S.K. Sperm DNA methylation altered by THC and nicotine: Vulnerability of neurodevelopmental genes with bivalent chromatin. Sci. Rep. 2020, 10, 16022. [Google Scholar] [CrossRef]
- Milton, L.A.; White, A.R. The potential impact of bushfire smoke on brain health. Neurochem. Int. 2020, 139, 104796. [Google Scholar] [CrossRef]
- Kim, Y.H.; Warren, S.H.; Krantz, Q.T.; King, C.; Jaskot, R.; Preston, W.T.; George, B.J.; Hays, M.D.; Landis, M.S.; Higuchi, M.; et al. Mutagenicity and Lung Toxicity of Smoldering vs. Flaming Emissions from Various Biomass Fuels: Implications for Health Effects from Wildland Fires. Environ. Health Perspect 2018, 126, 017011. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, T.G.; James, E.R.; Alonso, D.F.; Hoidal, J.R.; Murphy, P.J.; Hotaling, J.M.; Cairns, B.R.; Carrell, D.T.; Aston, K.I. Cigarette smoking significantly alters sperm DNA methylation patterns. Andrology 2017, 5, 1089–1099. [Google Scholar] [CrossRef]
- Alkhaled, Y.; Laqqan, M.; Tierling, S.; Porto, C.L.; Amor, H.; Hammadeh, M.E. Impact of cigarette-smoking on sperm DNA methylation and its effect on sperm parameters. Andrologia 2018, 50, e12950. [Google Scholar] [CrossRef]
- Murphy, S.K.; Itchon-Ramos, N.; Visco, Z.; Huang, Z.; Grenier, C.; Schrott, R.; Acharya, K.; Boudreau, M.-H.; Price, T.M.; Rabourn, D.J.; et al. Cannabinoid exposure and altered DNA methylation in rat and human sperm. Epigenetics 2018, 13, 1208–1221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jenkins, T.G.; Aston, K.I.; Pflueger, C.; Cairns, B.R.; Carrell, D.T. Age-associated sperm DNA methylation alterations: Possible implications in offspring disease susceptibility. PLoS Genet. 2014, 10, e1004458. [Google Scholar] [CrossRef]
- Yauk, C.; Polyzos, A.; Rowan-Carroll, A.; Somers, C.M.; Godschalk, R.W.; Van Schooten, F.J.; Berndt, M.L.; Pogribny, I.P.; Koturbash, I.; Williams, A.; et al. Germ-line mutations, DNA damage, and global hypermethylation in mice exposed to particulate air pollution in an urban/industrial location. Proc. Natl. Acad. Sci. USA 2008, 105, 605–610. [Google Scholar] [CrossRef] [Green Version]
- Montjean, D.; Zini, A.; Ravel, C.; Belloc, S.; Dalleac, A.; Copin, H.; Boyer, P.; McElreavey, K.; Benkhalifa, M. Sperm global DNA methylation level: Association with semen parameters and genome integrity. Andrology 2015, 3, 235–240. [Google Scholar] [CrossRef]
- Garrido, N.; Cruz, F.; Egea, R.R.; Simon, C.; Sadler-Riggleman, I.; Beck, D.; Nilsson, E.; Maamar, M.B.; Skinner, M.K. Sperm DNA methylation epimutation biomarker for paternal offspring autism susceptibility. Clin. Epigenet. 2021, 13, 6. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, J.; Duan, J.; Gao, X.; Zhu, W.; Lu, X.; Yang, L.; Zhang, J.; Li, G.; Ci, W.; et al. Programming and Inheritance of Parental DNA Methylomes in Mammals. Cell 2014, 157, 979–991. [Google Scholar] [CrossRef] [Green Version]
- Hughes, V. Epigenetics: The sins of the father. Nature 2014, 507, 22–24. [Google Scholar] [CrossRef] [PubMed]
- Skinner, M.K. Environmental epigenetic transgenerational inheritance and somatic epigenetic mitotic stability. Epigenetics 2011, 6, 838–842. [Google Scholar] [CrossRef] [Green Version]
- Quadrana, L.; Colot, V. Plant Transgenerational Epigenetics. Annu. Rev. Genet. 2016, 50, 467–491. [Google Scholar] [CrossRef]
- Northstone, K.; Golding, J.; Smith, G.D.; Miller, L.L.; Pembrey, M. Prepubertal start of father’s smoking and increased body fat in his sons: Further characterisation of paternal transgenerational responses. Eur. J. Hum. Genet. 2014, 22, 1382–1386. [Google Scholar] [CrossRef] [Green Version]
- Anway, M.D.; Cupp, A.S.; Uzumcu, M.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005, 308, 1466–1469. [Google Scholar] [CrossRef] [Green Version]
- Skinner, M.K.; Maamar, M.B.; Sadler-Riggleman, I.; Beck, D.; Nilsson, E.; McBirney, M.; Klukovich, R.; Xie, Y.; Tang, C.; Yan, W. Alterations in sperm DNA methylation, non-coding RNA and histone retention associate with DDT-induced epigenetic transgenerational inheritance of disease. Epigenet. Chromatin 2018, 11, 8. [Google Scholar] [CrossRef] [Green Version]
- Iqbal, K.; Tran, D.A.; Li, A.X.; Warden, C.; Bai, A.Y.; Singh, P.; Wu, X.; Pfeifer, G.P.; Szabó, P.E. Deleterious effects of endocrine disruptors are corrected in the mammalian germline by epigenome reprogramming. Genome Biol. 2015, 16, 59. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schuller, A.; Bellini, C.; Jenkins, T.G.; Eden, M.; Matz, J.; Oakes, J.; Montrose, L. Simulated Wildfire Smoke Significantly Alters Sperm DNA Methylation Patterns in a Murine Model. Toxics 2021, 9, 199. https://doi.org/10.3390/toxics9090199
Schuller A, Bellini C, Jenkins TG, Eden M, Matz J, Oakes J, Montrose L. Simulated Wildfire Smoke Significantly Alters Sperm DNA Methylation Patterns in a Murine Model. Toxics. 2021; 9(9):199. https://doi.org/10.3390/toxics9090199
Chicago/Turabian StyleSchuller, Adam, Chiara Bellini, Timothy G. Jenkins, Matthew Eden, Jacqueline Matz, Jessica Oakes, and Luke Montrose. 2021. "Simulated Wildfire Smoke Significantly Alters Sperm DNA Methylation Patterns in a Murine Model" Toxics 9, no. 9: 199. https://doi.org/10.3390/toxics9090199
APA StyleSchuller, A., Bellini, C., Jenkins, T. G., Eden, M., Matz, J., Oakes, J., & Montrose, L. (2021). Simulated Wildfire Smoke Significantly Alters Sperm DNA Methylation Patterns in a Murine Model. Toxics, 9(9), 199. https://doi.org/10.3390/toxics9090199






