Prenatal Exposure to Chemical Mixtures and Cognitive Flexibility among Adolescents
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Chemical Exposure Assessment
2.3. Cognitive Flexibility Assessment
2.4. Covariate Assessment
2.5. Statistical Analysis
3. Results
3.1. Study Population
3.2. Chemical Exposure Measures
3.3. Executive Function Measures
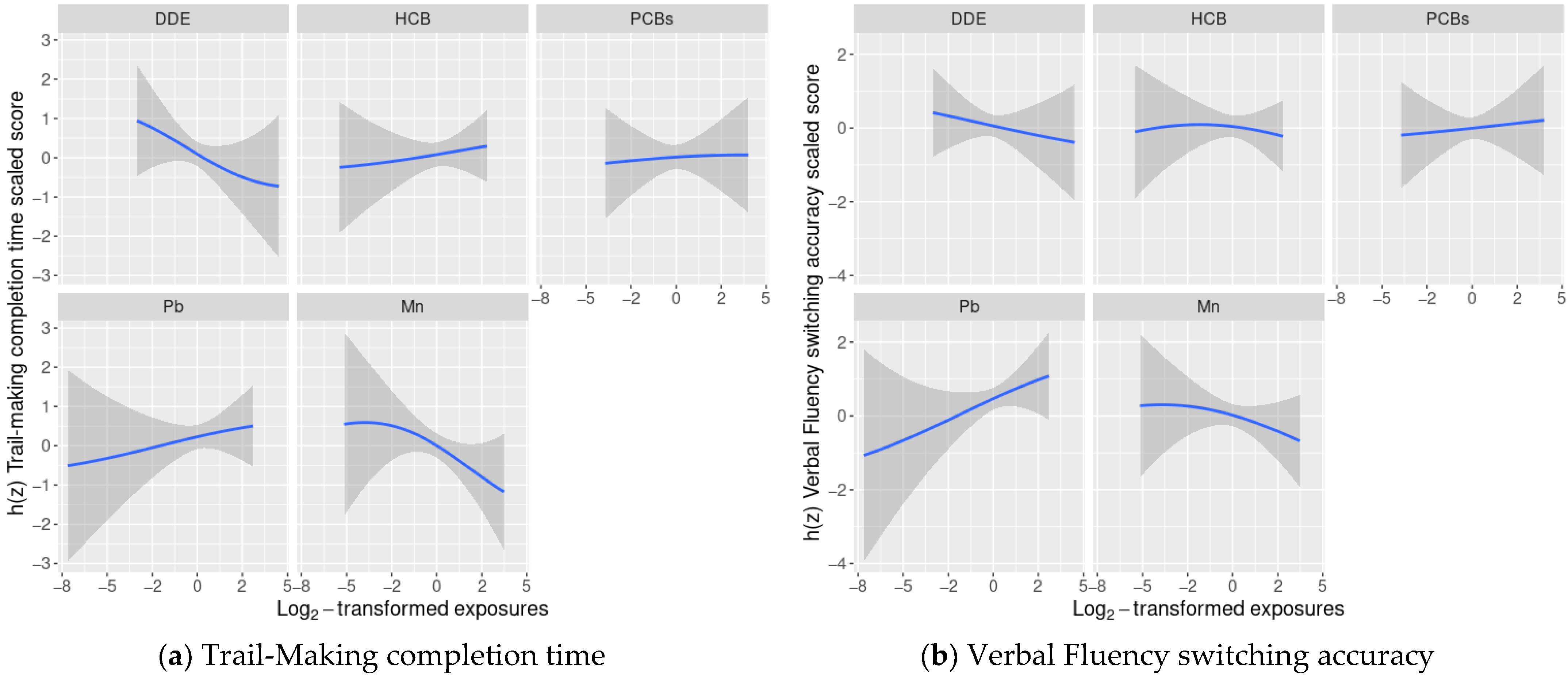
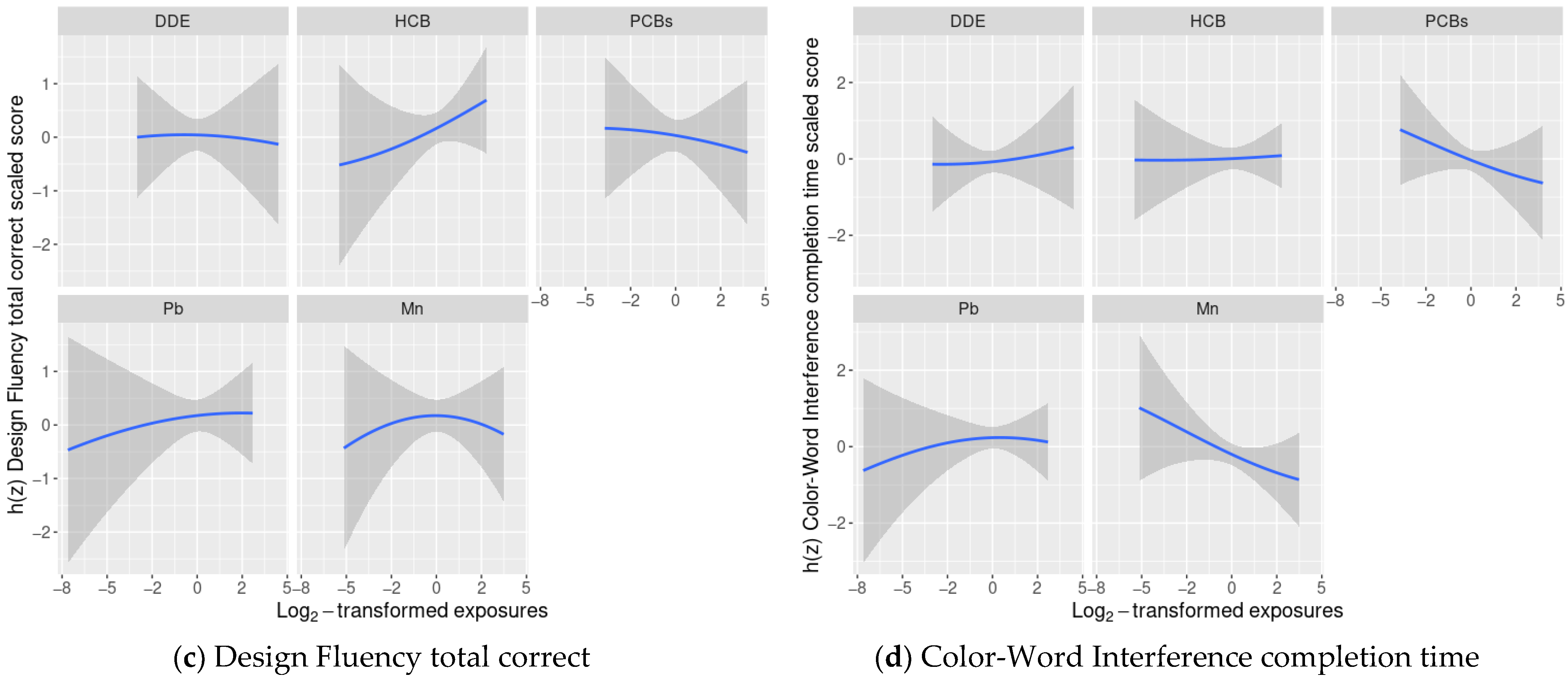
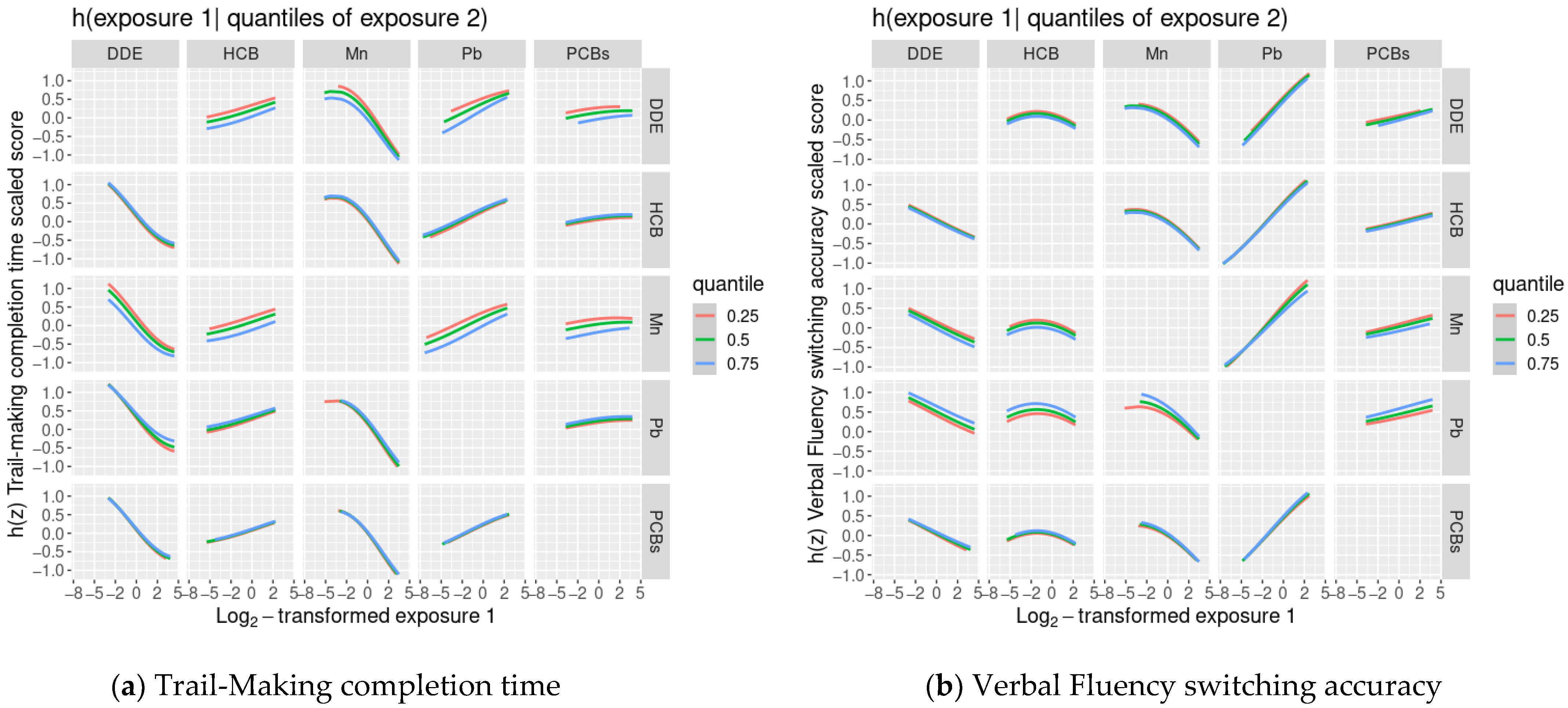
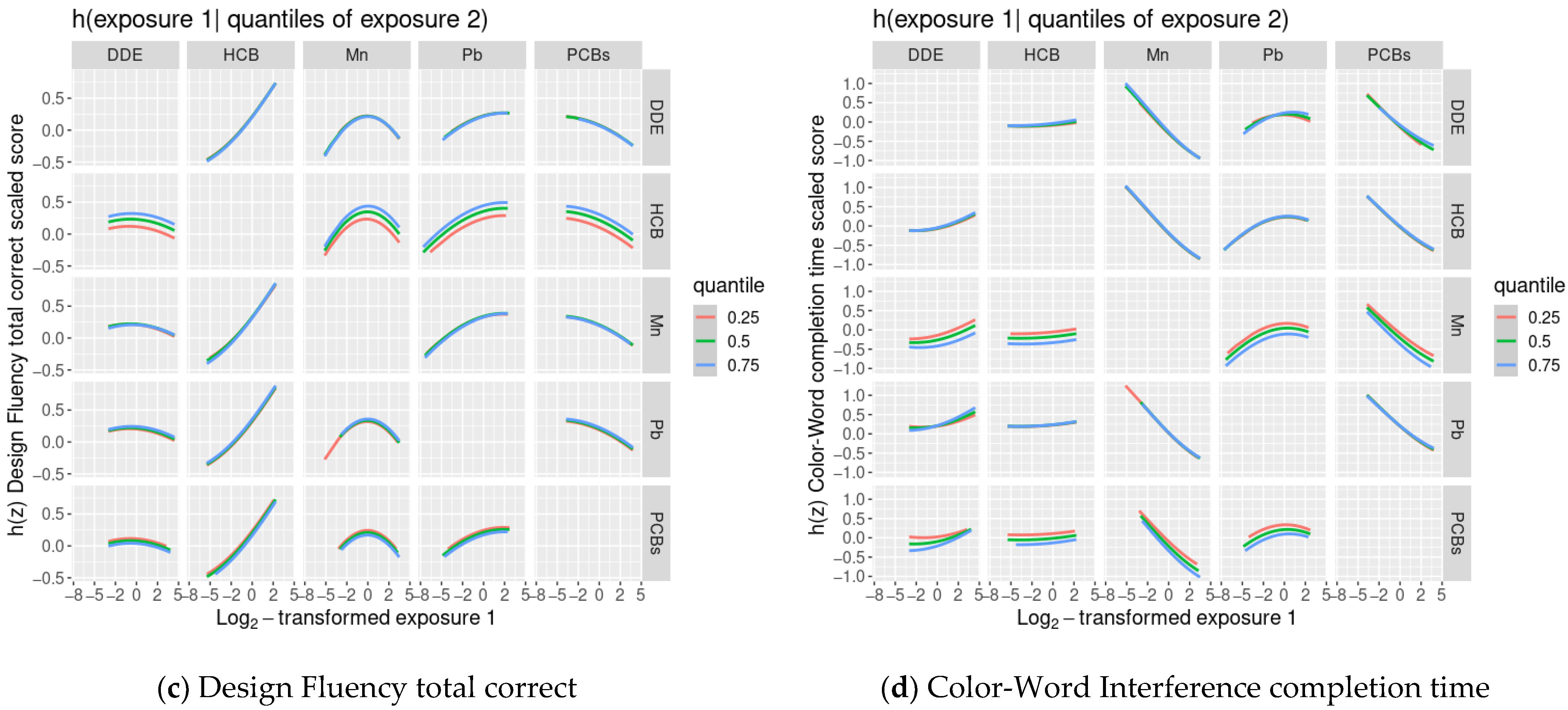
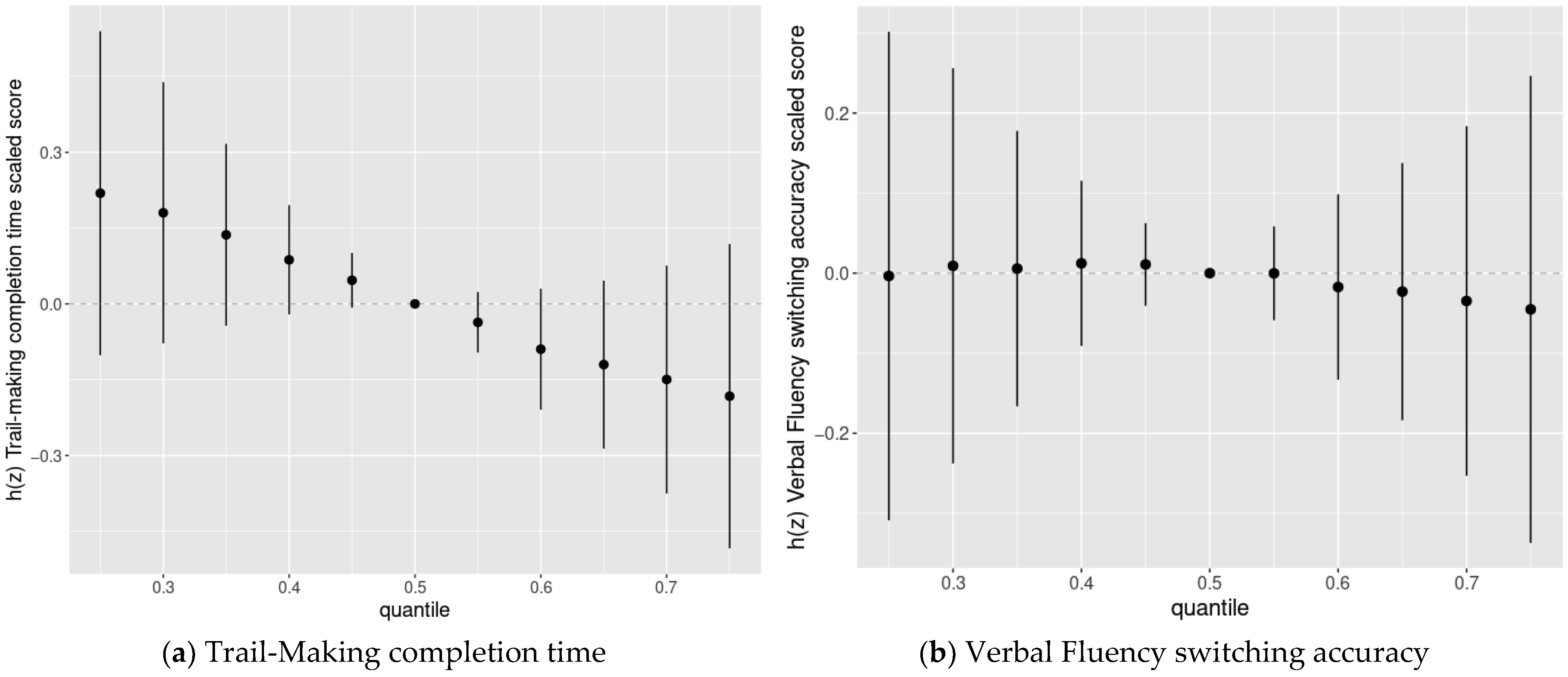
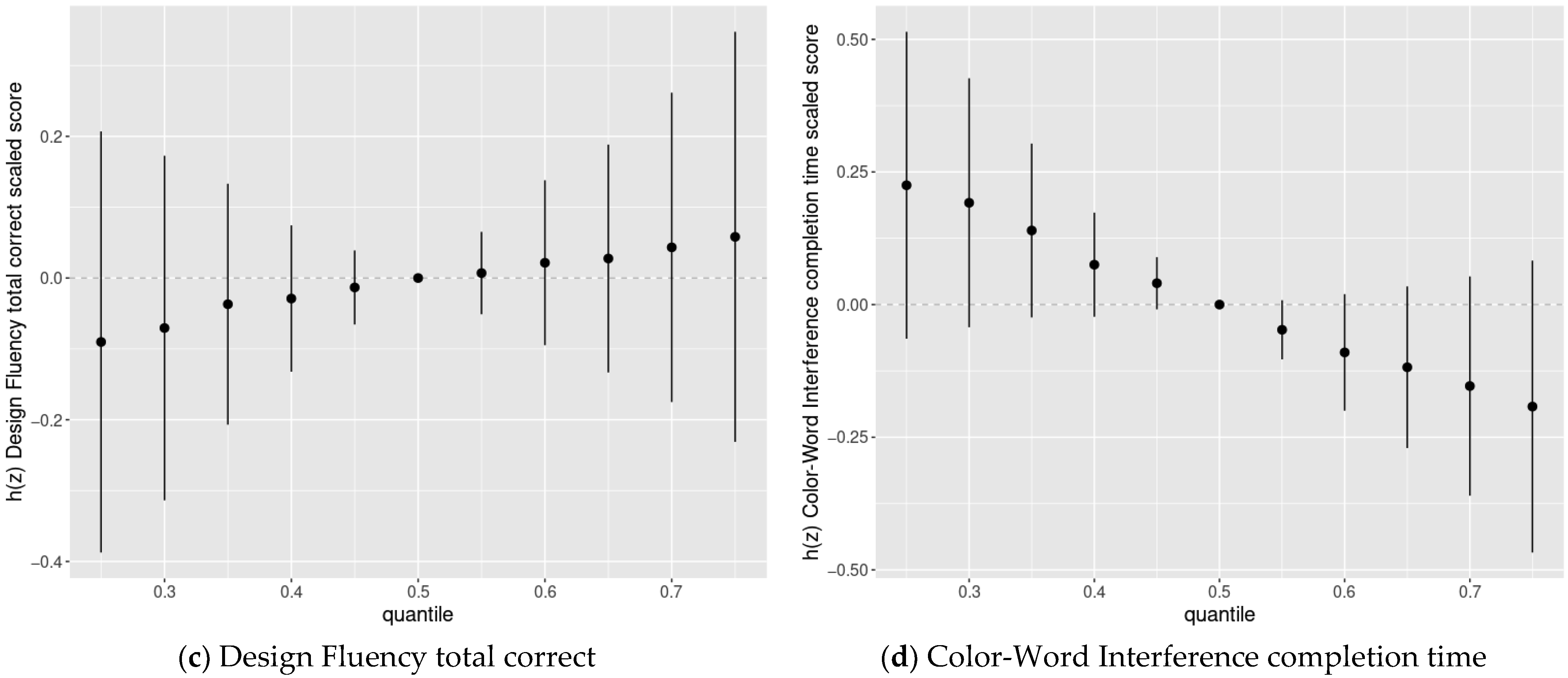
3.4. BKMR Analysis of Cognitive Flexibility Scaled Scores and Prenatal Chemical Mixture Exposures
3.5. Linear Regression Analyses of Cognitive Flexibility and Prenatal Chemical Mixture Exposures
3.6. Assessment of Effect Modification by Sex and PNSDI
3.7. Secondary Analyses: Negative Binomial and Logistic Regression Analyses of Cognitive Flexibility and Prenatal Chemical Mixture Exposures
3.8. Secondary Analyses: Seven-Chemical Mixture
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Diamond, A. Executive Functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [Green Version]
- Davidson, M.C.; Amso, D.; Anderson, L.C.; Diamond, A. Development of Cognitive Control and Executive Functions from 4 to 13 Years: Evidence from Manipulations of Memory, Inhibition, and Task Switching. Neuropsychologia 2006, 44, 2037–2078. [Google Scholar] [CrossRef] [Green Version]
- Gabrys, R.L.; Tabri, N.; Anisman, H.; Matheson, K. Cognitive Control and Flexibility in the Context of Stress and Depressive Symptoms: The Cognitive Control and Flexibility Questionnaire. Front. Psychol. 2018, 9, 2219. [Google Scholar] [CrossRef]
- Kercood, S.; Lineweaver, T.T.; Frank, C.C.; Fromm, E.D. Cognitive Flexibility and Its Relationship to Academic Achievement and Career Choice of College Students With and Without Attention Deficit Hyperactivity Disorder. J. Postsecond. Educ. Disabil. 2017, 30, 329. [Google Scholar]
- Roshani, F.; Piri, R.; Malek, A.; Michel, T.M.; Vafaee, M.S. Comparison of Cognitive Flexibility, Appropriate Risk-Taking and Reaction Time in Individuals with and without Adult ADHD. Psychiatry Res. 2020, 284, 112494. [Google Scholar] [CrossRef]
- Yu, Y.; Yu, Y.; Lin, Y. Anxiety and Depression Aggravate Impulsiveness: The Mediating and Moderating Role of Cognitive Flexibility. Psychol. Heal. Med. 2019, 25, 25–36. [Google Scholar] [CrossRef]
- Dickstein, D.P.; Nelson, E.E.; McClure, E.B.; Grimley, M.E.; Knopf, L.; Brotman, M.A.; Rich, B.A.; Pine, D.S.; Leibenluft, E. Cognitive Flexibility in Phenotypes of Pediatric Bipolar Disorder. J. Am. Acad. Child Adolesc. Psychiatry 2007, 46, 341–355. [Google Scholar] [CrossRef]
- Chamberlain, S.R.; Fineberg, N.A.; Blackwell, A.D.; Robbins, T.W.; Sahakian, B.J. Motor Inhibition and Cognitive Flexibility in Obsessive-Compulsive Disorder and Trichotillomania. Am. J. Psychiatry 2006, 163, 1282–1284. [Google Scholar] [CrossRef]
- Thoma, P.; Wiebel, B.; Daum, I. Response Inhibition and Cognitive Flexibility in Schizophrenia with and without Comorbid Substance Use Disorder. Schizophr. Res. 2007, 92, 168–180. [Google Scholar] [CrossRef]
- Grandjean, P.; Landrigan, P.J. Neurobehavioural Effects of Developmental Toxicity. Lancet Neurol. 2014, 13, 330–338. [Google Scholar] [CrossRef] [Green Version]
- EWG. Body Burden: The Pollution in Newborns. Available online: https://www.ewg.org/research/body-burden-pollution-newborns (accessed on 11 May 2020).
- U.S. Centers for Disease Control and Prevention (CDC). Fourth National Report on Human Exposure to Environmental Chemicals: Updated Tables, January 2019; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2019; Volume 1.
- Fruh, V.; Rifas-Shiman, S.L.; Amarasiriwardena, C.; Cardenas, A.; Bellinger, D.C.; Wise, L.A.; White, R.F.; Wright, R.O.; Oken, E.; Claus Henn, B. Prenatal Lead Exposure and Childhood Executive Function and Behavioral Difficulties in Project Viva. NeuroToxicology 2019, 75, 105–115. [Google Scholar] [CrossRef]
- Jacobson, J.L.; Jacobson, S.W. Prenatal Exposure to Polychlorinated Biphenyls and Attention at School Age. J. Pediatrics 2003, 143, 780–788. [Google Scholar] [CrossRef]
- Stewart, P.; Fitzgerald, S.; Reihman, J.; Gump, B.; Lonky, E.; Darvill, T.; Pagano, J.; Hauser, P. Prenatal PCB Exposure, the Corpus Callosum, and Response Inhibition. Environ. Health Perspect. 2003, 111, 1670–1677. [Google Scholar] [CrossRef] [Green Version]
- Stewart, P.; Reihman, J.; Gump, B.; Lonky, E.; Darvill, T.; Pagano, J. Response Inhibition at 8 and 9 1/2 Years of Age in Children Prenatally Exposed to PCBs. Neurotoxicol. Teratol. 2005, 27, 771–780. [Google Scholar] [CrossRef]
- Sagiv, S.K.; Thurston, S.W.; Bellinger, D.C.; Altshul, L.M.; Korrick, S.A. Neuropsychological Measures of Attention and Impulse Control among 8-Year-Old Children Exposed Prenatally to Organochlorines. Environ. Health Perspect. 2012, 120, 904–909. [Google Scholar] [CrossRef] [Green Version]
- Ribas-Fitó, N.; Torrent, M.; Carrizo, D.; Muñoz-Ortiz, L.; Júlvez, J.; Grimalt, J.O.; Sunyer, J. In Utero Exposure to Background Concentrations of DDT and Cognitive Functioning among Preschoolers. Am. J. Epidemiol. 2006, 164, 955–962. [Google Scholar] [CrossRef] [Green Version]
- Gaspar, F.W.; Harley, K.G.; Kogut, K.; Chevrier, J.; Mora, A.M.; Sjödin, A.; Eskenazi, B. Prenatal DDT and DDE Exposure and Child IQ in the CHAMACOS Cohort. Environ. Int. 2015, 85, 206–212. [Google Scholar] [CrossRef] [Green Version]
- Kyriklaki, A.; Vafeiadi, M.; Kampouri, M.; Koutra, K.; Roumeliotaki, T.; Chalkiadaki, G.; Anousaki, D.; Rantakokko, P.; Kiviranta, H.; Fthenou, E.; et al. Prenatal Exposure to Persistent Organic Pollutants in Association with Offspring Neuropsychological Development at 4 Years of Age: The Rhea Mother-Child Cohort, Crete, Greece. Environ. Int. 2016, 97, 204–211. [Google Scholar] [CrossRef]
- Forns, J.; Torrent, M.; Garcia-Esteban, R.; Grellier, J.; Gascon, M.; Julvez, J.; Guxens, M.; Grimalt, J.O.; Sunyer, J. Prenatal Exposure to Polychlorinated Biphenyls and Child Neuropsychological Development in 4-Year-Olds: An Analysis per Congener and Specific Cognitive Domain. Sci. Total Environ. 2012, 432, 338–343. [Google Scholar] [CrossRef]
- Ericson, J.E.; Crinella, F.M.; Clarke-Stewart, K.A.; Allhusen, V.D.; Chan, T.; Robertson, R.T. Prenatal Manganese Levels Linked to Childhood Behavioral Disinhibition. Neurotoxicol. Teratol. 2007, 29, 181–187. [Google Scholar] [CrossRef]
- Bauer, J.A.; Henn, B.C.; Austin, C.; Zoni, S.; Fedrighi, C.; Cagna, G.; Placidi, D.; White, R.F.; Yang, Q.; Coull, B.A.; et al. Manganese in Teeth and Neurobehavior: Sex-Specific Windows of Susceptibility HHS Public Access. Environ. Int. 2017, 108, 299–308. [Google Scholar] [CrossRef]
- Rosado, J.L.; Ronquillo, D.; Kordas, K.; Rojas, O.; Alatorre, J.; Lopez, P.; Garcia-Vargas, G.; del Carmen Caamaño, M.; Cebrián, M.E.; Stoltzfus, R.J. Arsenic Exposure and Cognitive Performance in Mexican Schoolchildren. Environ. Health Perspect. 2007, 115, 1371–1375. [Google Scholar] [CrossRef] [Green Version]
- Wasserman, G.A.; Liu, X.; LoIacono, N.J.; Kline, J.; Factor-Litvak, P.; Van Geen, A.; Mey, J.L.; Levy, D.; Abramson, R.; Schwartz, A.; et al. A Cross-Sectional Study of Well Water Arsenic and Child IQ in Maine Schoolchildren. Environ. Health A Glob. Access Sci. Source 2014, 13, 23. [Google Scholar] [CrossRef] [Green Version]
- Wasserman, G.A.; Liu, X.; Parvez, F.; Chen, Y.; Factor-Litvak, P.; LoIacono, N.J.; Levy, D.; Shahriar, H.; Uddin, M.N.; Islam, T.; et al. A Cross-Sectional Study of Water Arsenic Exposure and Intellectual Function in Adolescence in Araihazar, Bangladesh. Environ. Int. 2018, 118, 304–313. [Google Scholar] [CrossRef]
- Tsai, S.Y.; Chou, H.Y.; The, H.W.; Chen, C.M.; Chen, C.J. The Effects of Chronic Arsenic Exposure from Drinking Water on the Neurobehavioral Development in Adolescence. Neurotoxicology 2003, 24, 747–753. [Google Scholar] [CrossRef]
- Carpenter, D.O.; Arcaro, K.; Spink, D.C. Understanding the Human Health Effects of Chemical Mixtures. Environ. Health Perspect. 2002, 110, 25–42. [Google Scholar] [CrossRef] [Green Version]
- Boucher, O.; Burden, M.J.; Muckle, G.; Saint-Amour, D.; Ayotte, P.; Dewailly, É.; Nelson, C.A.; Jacobson, S.W.; Jacobson, J.L. Response Inhibition and Error Monitoring during a Visual Go/No-Go Task in Inuit Children Exposed to Lead, Polychlorinated Biphenyls, and Methylmercury. Environ. Health Perspect. 2012, 120, 608–615. [Google Scholar] [CrossRef] [Green Version]
- Yorifuji, T.; Debes, F.; Weihe, P.; Grandjean, P. Prenatal Exposure to Lead and Cognitive Deficit in 7- and 14-Year-Old Children in the Presence of Concomitant Exposure to Similar Molar Concentration of Methylmercury. Neurotoxicol. Teratol. 2011, 33, 205–211. [Google Scholar] [CrossRef] [Green Version]
- Wasserman, G.A.; Liu, X.; Parvez, F.; Factor-Litvak, P.; Ahsan, H.; Levy, D.; Kline, J.; van Geen, A.; Mey, J.; Slavkovich, V.; et al. Arsenic and Manganese Exposure and Children’s Intellectual Function. NeuroToxicology 2011, 32, 450–457. [Google Scholar] [CrossRef] [Green Version]
- Barg, G.; Daleiro, M.; Queirolo, E.I.; Ravenscroft, J.; Mañay, N.; Peregalli, F.; Kordas, K. Association of Low Lead Levels with Behavioral Problems and Executive Function Deficits in Schoolers from Montevideo, Uruguay. Int. J. Environ. Res. Public Health 2018, 15, 2735. [Google Scholar] [CrossRef] [Green Version]
- Behforooz, B.; Newman, J.; Gallo, M.V.; Schell, L.M. PCBs and Measures of Attention and Impulsivity on a Continuous Performance Task of Young Adults. Neurotoxicology Teratol. 2017, 64, 29–36. [Google Scholar] [CrossRef]
- Lucchini, R.G.; Guazzetti, S.; Renzetti, S.; Conversano, M.; Cagna, G.; Fedrighi, C.; Giorgino, A.; Peli, M.; Placidi, D.; Zoni, S.; et al. Neurocognitive Impact of Metal Exposure and Social Stressors among Schoolchildren in Taranto, Italy. Environ. Health A Glob. Access Sci. Source 2019, 18, 67. [Google Scholar] [CrossRef] [Green Version]
- Choi, A.L.; Levy, J.I.; Dockery, D.W.; Ryan, L.M.; Tolbert, P.E.; Altshul, L.M.; Korrick, S.A. Does Living near a Superfund Site Contribute to Higher Polychlorinated Biphenyl (PCB) Exposure? Environ. Health Perspect. 2006, 114, 1092–1098. [Google Scholar] [CrossRef] [Green Version]
- Korrick, S.A.; Altshul, L.M.; Tolbert, P.E.; Burse, V.W.; Needham, L.L.; Monson, R.R. Measurement of PCBs, DDE, and Hexachlorobenzene in Cord Blood from Infants Born in Towns Adjacent to a PCB-Contaminated Waste Site. J. Expo. Anal. Environ. Epidemiol. 2000, 10, 743–754. [Google Scholar] [CrossRef] [Green Version]
- Orenstein, S.T.C.; Thurston, S.W.; Bellinger, D.C.; Schwartz, J.D.; Amarasiriwardena, C.J.; Altshul, L.M.; Korrick, S.A. Prenatal Organochlorine and Methylmercury Exposure and Memory and Learning in School-Age Children in Communities near the New Bedford Harbor Superfund Site, Massachusetts. Environ. Health Perspect. 2014, 122, 1253–1259. [Google Scholar] [CrossRef] [Green Version]
- Myers, G.J.; Davidson, P.W. Prenatal Methylmercury Exposure and Children: Neurologic, Developmental, and Behavioral Research. Environ. Health Perspect. 1998, 106, 841–847. [Google Scholar]
- Amaral, A.F.S.; Porta, M.; Silverman, D.T.; Milne, R.L.; Kogevinas, M.; Rothman, N.; Cantor, K.P.; Jackson, B.P.; Pumarega, J.A.; López, T.; et al. Pancreatic Cancer Risk and Levels of Trace Elements. Gut 2012, 61, 1583–1588. [Google Scholar] [CrossRef] [Green Version]
- Delis, D.; Kaplan, E.; Kramer, J. Delis-Kaplan Executive Function System; Harcourt Assessment, Inc.: San Antonio, TX, USA, 2001. [Google Scholar]
- Caldwell, B.; Bradley, R. Home Observation for Measurement of the Environment; Dorsey: New York, NY, USA, 1985. [Google Scholar]
- Kaufman, A.; Kaufman, N. Kaufman Brief Intelligence Test; American Guidance Service: Circle Pines, MN, USA, 1990. [Google Scholar]
- Bobb, J.F.; Claus Henn, B.; Valeri, L.; Coull, B.A. Statistical Software for Analyzing the Health Effects of Multiple Concurrent Exposures via Bayesian Kernel Machine Regression. Environ. Health A Glob. Access Sci. Source 2018, 17, 67. [Google Scholar] [CrossRef] [Green Version]
- Bobb, J.F. Bkmr: Bayesian Kernel Machine Regression; R Package Version 0.2.0. 2017. Available online: https://CRAN.R-project.org/package=bkmr (accessed on 11 May 2020).
- Hernán, M.A.; Robins, J.M. How to Adjust for Selection Bias. In Causal Inference: What If; Chapman & Hall/CRC: Boca Raton, FL, USA, 2020; pp. 107–112. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Longnecker, M.P.; Wolff, M.S.; Gladen, B.C.; Brock, J.W.; Grandjean, P.; Jacobson, J.L.; Korrick, S.A.; Rogan, W.J.; Weisglas-Kuperus, N.; Hertz-Picciotto, I.; et al. Comparison of Polychlorinated Biphenyl Levels across Studies of Human Neurodevelopment. Environ. Health Perspect. 2003, 111, 65–70. [Google Scholar] [CrossRef] [Green Version]
- Ettinger, A.S.; Egan, K.B.; Homa, D.M.; Brown, M.J. Blood Lead Levels in U.S. Women of Childbearing Age. Environ. Health Perspect. 2020, 1976–2016. [Google Scholar] [CrossRef] [Green Version]
- Arbuckle, T.E.; Liang, C.L.; Morisset, A.S.; Fisher, M.; Weiler, H.; Cirtiu, C.M.; Legrand, M.; Davis, K.; Ettinger, A.S.; Fraser, W.D. Maternal and Fetal Exposure to Cadmium, Lead, Manganese and Mercury: The MIREC Study. Chemosphere 2016, 163, 270–282. [Google Scholar] [CrossRef] [Green Version]
- McDowell, M.A.; Dillon, C.F.; Osterloh, J.; Bolger, P.M.; Pellizzari, E.; Fernando, R.; de Oca, R.M.; Schober, S.E.; Sinks, T.; Jones, R.L.; et al. Hair Mercury Levels in U.S. Children and Women of Childbearing Age: Reference Range Data from NHANES 1999–2000. Environ. Health Perspect. 2004, 112, 1165–1171. [Google Scholar] [CrossRef]
- Ettinger, A.S.; Arbuckle, T.E.; Fisher, M.; Liang, C.L.; Davis, K.; Cirtiu, C.M.; Bélanger, P.; LeBlanc, A.; Fraser, W.D. Arsenic Levels among Pregnant Women and Newborns in Canada: Results from the Maternal-Infant Research on Environmental Chemicals (MIREC) Cohort. Environ. Res. 2017, 153, 8–16. [Google Scholar] [CrossRef] [Green Version]
- Bellinger, D.C. What Is an Adverse Effect? A Possible Resolution of Clinical and Epidemiological Perspectives on Neurobehavioral Toxicity. Environ. Res. 2004, 95, 394–405. [Google Scholar] [CrossRef]
- Sanders, A.P.; Claus Henn, B.; Wright, R.O. Perinatal and Childhood Exposure to Cadmium, Manganese, and Metal Mixtures and Effects on Cognition and Behavior: A Review of Recent Literature. Curr. Environ. Health Rep. 2015, 2, 284–294. [Google Scholar] [CrossRef] [Green Version]
- Agency for Toxic Substances & Disease Registry (ATSDR). Toxicological Profile for Manganese; U.S Department of Health and Human Services, Public Health Service: Atlanta, GA, USA, 2012.
- Oppenheimer, A.V.; Bellinger, D.C.; Coull, B.A.; Weisskopf, M.G.; Zemplenyi, M.; Korrick, S.A. Prenatal Exposure to Chemical Mixtures and Inhibition among Adolescents. Toxics 2021, 9, 311. [Google Scholar] [CrossRef]
- Oppenheimer, A.V.; Bellinger, D.C.; Coull, B.A.; Weisskopf, M.A.; Korrick, S.A. Prenatal Exposure to Chemical Mixtures and Working Memory among Adolescents. Environ Res. 2021. [Google Scholar] [CrossRef]
- Choi, A.L.; Cordier, S.; Weihe, P.; Grandjean, P. Negative Confounding in the Evaluation of Toxicity: The Case of Methylmercury in Fish and Seafood. Crit. Rev. Toxicol. 2008, 38, 877–893. [Google Scholar] [CrossRef] [Green Version]
- Wilschefski, S.; Baxter, M. Inductively Coupled Plasma Mass Spectrometry: Introduction to Analytical Aspects. Clin. Biochem. Rev. 2019, 40, 115–133. [Google Scholar] [CrossRef]
- Coetzee, D.J.; McGovern, P.M.; Rao, R.; Harnack, L.J.; Georgieff, M.K.; Stepanov, I. Measuring the Impact of Manganese Exposure on Children’s Neurodevelopment: Advances and Research Gaps in Biomarker-Based Approaches. Environ. Health A Glob. Access Sci. Source 2016, 15, 91. [Google Scholar] [CrossRef] [Green Version]
- Bauer, J.A.; Devick, K.L.; Bobb, J.F.; Coull, B.A.; Bellinger, D.; Benedetti, C.; Cagna, G.; Fedrighi, C.; Guazzetti, S.; Oppini, M.; et al. Associations of a Metal Mixture Measured in Multiple Biomarkers with IQ: Evidence from Italian Adolescents Living near Ferroalloy Industry. Environ. Health Perspect. 2020, 128, 097002. [Google Scholar] [CrossRef]
- Gunier, R.B.; Mora, A.M.; Smith, D.; Arora, M.; Austin, C.; Eskenazi, B.; Bradman, A. Biomarkers of Manganese Exposure in Pregnant Women and Children Living in an Agricultural Community in California. Environ. Sci. Technol. 2014, 48, 14695–14702. [Google Scholar] [CrossRef]
| Descriptive Characteristic | Main Analysis Group, n = 373 | Excluded Group, n = 415 | |||||
|---|---|---|---|---|---|---|---|
| Cognitive Flexibility Measures 2 | n (%) | Mean (SD) | Range | n (%) | Mean (SD) | Range | p-Value 3 |
| Trail-Making | |||||||
| Completion time scaled score | 373 | 9.6 (2.8) | 1–14 | 155 | 9.1 (2.8) | 1–14 | 0.1 |
| Total errors | 373 | 0.9 (1.1) | 0–5 | 154 | 1.0 (1.4) | 0–13 | 0.5 |
| Overall Trail-Making performance | |||||||
| Best performance | 113 (30.3) | 45 (10.8) | 0.9 | ||||
| Poor performance | 260 (69.7) | 109 (26.3) | |||||
| Missing | 0 | 261 (62.9) | |||||
| Verbal Fluency | |||||||
| Switching accuracy scaled score | 373 | 9.2 (2.8) | 2–17 | 155 | 9.0 (2.8) | 1–17 | 0.3 |
| Total errors | 373 | 0.8 (1.2) | 0–7 | 155 | 0.9 (1.3) | 0–7 | 0.4 |
| Design Fluency | |||||||
| Total correct scaled score | 373 | 9.9 (2.8) | 2–18 | 155 | 9.6 (2.6) | 2–17 | 0.3 |
| Total errors | 373 | 2.6 (3.1) | 0–22 | 155 | 2.6 (2.6) | 0–16 | 0.2 |
| Color-Word Interference | |||||||
| Completion time scaled score | 373 | 9.9 (2.6) | 1–15 | 154 | 9.8 (2.7) | 1–14 | 0.7 |
| Total errors | 373 | 2.6 (2.4) | 0–19 | 154 | 2.8 (2.4) | 0–11 | 0.4 |
| Overall Color-Word performance | |||||||
| Best performance | 83 (22.3) | 34 (8.2) | 1.0 | ||||
| Poor performance | 290 (77.7) | 120 (28.9) | |||||
| Missing | 0 | 261 (62.9) | |||||
| Exposure Measures 4 | |||||||
| Cord serum DDE (ng/g) | 373 | 0.6 (1.2) | 0.02–14.9 | 378 | 0.4 (0.4) | 0.0–4.2 | <0.01 * |
| Cord serum HCB (ng/g) | 373 | 0.03 (0.02) | 0.0–0.1 | 378 | 0.03 (0.05) | 0.0–0.7 | 0.1 |
| Cord serum ΣPCB4 (ng/g) | 373 | 0.3 (0.3) | 0.01–4.4 | 378 | 0.2 (0.2) | 0.01–1.9 | 0.05 |
| Cord blood Pb (μg/dL) | 373 | 1.4 (0.9) | 0.0–9.4 | 375 | 1.7 (1.7) | 0.0–17.4 | <0.01 * |
| Cord blood Mn (µg/dL) | 373 | 4.2 (1.6) | 0.7–14.6 | 335 | 4.3 (2.0) | 0.2–22.1 | 0.6 |
| Covariate Measures 5 | |||||||
| Child Characteristics | |||||||
| Race/Ethnicity | 0.09 | ||||||
| Non–Hispanic White | 263 (70.5) | 268 (64.6) | |||||
| Hispanic | 33 (8.8) | 56 (13.5) | |||||
| Other | 77 (20.6) | 89 (21.4) | |||||
| Missing | 0 | 2 (0.5) | |||||
| Sex | 0.05 | ||||||
| Male | 179 (48.0) | 229 (55.2) | |||||
| Female | 194 (52.0) | 186 (44.8) | |||||
| Age at Exam | 373 | 15.5 (0.6) | 14.4–17.8 | 155 | 15.7 (0.7) | 13.9–17.9 | <0.01 * |
| Home Score | 373 | 43.9 (6.3) | 21–56 | 118 | 42.7 (6.0) | 27–53 | 0.07 |
| Year of Birth | <0.01 * | ||||||
| 1993–1994 | 100 (26.8) | 159 (38.3) | |||||
| 1995–1996 | 153 (41.0) | 147 (35.4) | |||||
| 1997–1998 | 120 (32.2) | 109 (26.3) | |||||
| Maternal Characteristics | |||||||
| Marital status at birth | <0.01 * | ||||||
| Not married | 136 (36.5) | 195 (47.0) | |||||
| Married | 237 (63.5) | 165 (39.8) | |||||
| Missing | 0 | 55 (13.3) | |||||
| Maternal IQ | 373 | 99.4 (10.4) | 57–124 | 262 | 95.8 (10.2) | 72–126 | <0.01 * |
| Seafood during pregnancy (serv/day) | 373 | 0.5 (0.6) | 0–5.3 | 260 | 0.6 (0.7) | 0–6 | 0.6 |
| Smoking during pregnancy | 0.1 | ||||||
| No | 272 (72.9) | 210 (50.6) | |||||
| Yes | 101 (27.1) | 103 (24.8) | |||||
| Missing | 0 | 102 (24.6) | |||||
| Household Characteristics at Birth | |||||||
| Maternal education | <0.01 * | ||||||
| ≤High School | 190 (50.9) | 231 (55.7) | |||||
| >High School | 183 (49.1) | 127 (30.6) | |||||
| Missing | 0 | 57 (13.7) | |||||
| Paternal Education | <0.01 * | ||||||
| ≤High School | 246 (66.0) | 266 (64.1) | |||||
| >High School | 127 (34.0) | 81 (19.5) | |||||
| Missing | 0 | 68 (16.4) | |||||
| Annual Household Income | <0.01 * | ||||||
| <$20,000 | 115 (30.8) | 150 (36.1) | |||||
| ≥$20,000 | 258 (69.2) | 201 (48.4) | |||||
| Missing | 0 | 64 (15.4) | |||||
| Examination Characteristics | |||||||
| Examiner | 0.4 | ||||||
| 1 | 277 (74.3) | 121 (29.2) | |||||
| 2 | 96 (25.7) | 34 (8.2) | |||||
| Missing | 0 | 260 (62.7) | |||||
| Exposure | Trail-Making Completion Time Difference (95% CI) | Verbal Fluency Switching Accuracy Difference (95% CI) | Design Fluency Total Correct Difference (95% CI) | Color-Word Interference Completion Time Difference (95% CI) |
|---|---|---|---|---|
| Log2 DDE | −0.23 (−0.52, 0.06) | −0.10 (−0.40, 0.20) | −0.01 (−0.32, 0.30) | 0.06 (−0.22, 0.34) |
| Log2 HCB | 0.11 (−0.20, 0.42) | −0.06 (−0.38, 0.26) | 0.24 (−0.09, 0.58) | 0.05 (−0.26, 0.35) |
| Log2 ΣPCB4 | 0.02 (−0.30, 0.34) | 0.05 (−0.27, 0.38) | −0.09 (−0.44, 0.25) | −0.21 (−0.52, 0.10) |
| Log2 Pb | 0.14 (−0.16, 0.43) | 0.27 (−0.03, 0.57) | 0.05 (−0.27, 0.36) | 0.04 (−0.24, 0.33) |
| Log2 Mn | −0.60 (−1.16, −0.05) * | −0.28 (−0.85, 0.29) | −0.10 (−0.70, 0.50) | −0.53 (−1.08, 0.01) |
| Exposure | Trail-Making Completion Time | Verbal Fluency Switching Accuracy | ||||
| Difference (95% CI) | Difference (95% CI) | |||||
| Males | Females | p for Interaction | Males | Females | p for Interaction | |
| Log2 DDE | −0.31 (−0.73, 0.12) | −0.14 (−0.58, 0.30) | 0.6 | −0.24 (−0.65, 0.16) | 0.02 (−0.45, 0.50) | 0.3 |
| Log2 HCB | 0.09 (−0.37, 0.55) | 0.14 (−0.31, 0.59) | 0.6 | −0.22 (−0.67, 0.22) | −0.07 (−0.56, 0.42) | 0.9 |
| Log2 ΣPCB4 | 0.18 (−0.29, 0.65) | −0.16 (−0.63, 0.32) | 0.6 | 0.10 (−0.35, 0.55) | −0.04 (−0.55, 0.48) | 0.4 |
| Log2 Pb | 0.44 (−0.10, 0.97) | −0.01 (−0.37, 0.35) | 0.1 | 0.64 (0.12, 1.15) * | 0.09 (−0.30, 0.48) | 0.2 |
| Log2 Mn | −0.15 (−0.95, 0.66) | −0.80 (−1.59, 0.00) | 0.3 | −0.47 (−1.25, 0.31) | −0.05 (−0.91, 0.81) | 0.6 |
| Exposure | Design Fluency Total Correct Difference (95% CI) | Color-Word Interference Completion Time | ||||
| Difference (95% CI) | ||||||
| Males | Females | p for Interaction | Males | Females | p for Interaction | |
| Log2 DDE | 0.20 (−0.27, 0.67) | −0.10 (−0.57, 0.36) | 0.4 | 0.09 (−0.34, 0.52) | 0.10 (−0.31, 0.51) | 0.9 |
| Log2 HCB | −0.15 (−0.67, 0.36) | 0.73 (0.26, 1.20) * | 0.01 * | −0.02 (−0.49, 0.44) | 0.07 (−0.34, 0.49) | 0.6 |
| Log2 ΣPCB4 | −0.04 (−0.56, 0.48) | −0.29 (−0.79, 0.21) | 0.7 | −0.37 (−0.84, 0.10) | −0.08 (−0.53, 0.36) | 0.2 |
| Log2 Pb | 0.29 (−0.31, 0.88) | −0.10 (−0.47, 0.28) | 0.2 | 0.03 (−0.51, 0.57) | −0.03 (−0.36, 0.31) | 0.8 |
| Log2 Mn | −0.45 (−1.35, 0.44) | 0.40 (−0.43, 1.23) | 0.2 | −0.54 (−1.35, 0.27) | −0.31 (−1.05, 0.43) | 0.8 |
| Exposure | Trail-Making Completion Time | Verbal Fluency Switching Accuracy | ||||
| Difference (95% CI) | Difference (95% CI) | |||||
| PNSDI < 3 | PNSDI ≥ 3 | p for Interaction | PNSDI < 3 | PNSDI ≥ 3 | p for Interaction | |
| Log2 DDE | −0.13 (−0.46, 0.20) | −0.36 (−0.97, 0.25) | 0.3 | −0.12 (−0.48, 0.25) | −0.15 (−0.71, 0.41) | 0.9 |
| Log2 HCB | 0.23 (−0.14, 0.60) | −0.22 (−0.82, 0.38) | 0.1 | 0.06 (−0.35, 0.46) | −0.36 (−0.92, 0.19) | 0.2 |
| Log2 ΣPCB4 | −0.10 (−0.47, 0.26) | 0.42 (−0.25, 1.09) | 0.2 | 0.10 (−0.30, 0.50) | 0.16 (−0.46, 0.78) | 0.9 |
| Log2 Pb | −0.01 (−0.37, 0.35) | 0.09 (−0.49, 0.67) | 0.7 | 0.20 (−0.19, 0.60) | 0.26 (−0.27, 0.80) | 0.6 |
| Log2 Mn | −0.65 (−1.31, 0.02) | −0.37 (−1.44, 0.70) | 0.8 | −0.27 (−1.00, 0.45) | −0.22 (−1.21, 0.77) | 0.7 |
| Exposure | Design Fluency Total Correct Difference (95% CI) | Color-Word Interference Completion Time | ||||
| Difference (95% CI) | ||||||
| PNSDI < 3 | PNSDI ≥ 3 | p for interaction | PNSDI < 3 | PNSDI ≥ 3 | p for Interaction | |
| Log2 DDE | 0.05 (−0.35, 0.45) | −0.18 (−0.71, 0.35) | 0.4 | 0.20 (−0.12, 0.53) | −0.08 (−0.65, 0.49) | 0.2 |
| Log2 HCB | 0.59 (0.15, 1.04) * | −0.38 (−0.90, 0.14) | 0.01 * | 0.13 (−0.23, 0.50) | −0.12 (−0.68, 0.44) | 0.3 |
| Log2 ΣPCB4 | −0.29 (−0.73, 0.15) | 0.42 (−0.17, 1.00) | 0.1 | −0.35 (−0.71, 0.00) | 0.21 (−0.41, 0.84) | 0.2 |
| Log2 Pb | −0.07 (−0.51, 0.36) | 0.12 (−0.38, 0.62) | 0.5 | −0.18 (−0.54, 0.18) | 0.20 (−0.34, 0.74) | 0.3 |
| Log2 Mn | −0.12 (−0.92, 0.68) | −0.26 (−1.19, 0.67) | 0.8 | −0.50 (−1.16, 0.15) | −0.50 (−1.51, 0.50) | 0.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oppenheimer, A.V.; Bellinger, D.C.; Coull, B.A.; Weisskopf, M.G.; Korrick, S.A. Prenatal Exposure to Chemical Mixtures and Cognitive Flexibility among Adolescents. Toxics 2021, 9, 329. https://doi.org/10.3390/toxics9120329
Oppenheimer AV, Bellinger DC, Coull BA, Weisskopf MG, Korrick SA. Prenatal Exposure to Chemical Mixtures and Cognitive Flexibility among Adolescents. Toxics. 2021; 9(12):329. https://doi.org/10.3390/toxics9120329
Chicago/Turabian StyleOppenheimer, Anna V., David C. Bellinger, Brent A. Coull, Marc G. Weisskopf, and Susan A. Korrick. 2021. "Prenatal Exposure to Chemical Mixtures and Cognitive Flexibility among Adolescents" Toxics 9, no. 12: 329. https://doi.org/10.3390/toxics9120329
APA StyleOppenheimer, A. V., Bellinger, D. C., Coull, B. A., Weisskopf, M. G., & Korrick, S. A. (2021). Prenatal Exposure to Chemical Mixtures and Cognitive Flexibility among Adolescents. Toxics, 9(12), 329. https://doi.org/10.3390/toxics9120329






