Dichlorodiphenyltrichloroethane and the Adrenal Gland: From Toxicity to Endocrine Disruption
Abstract
1. Endocrine Disruptors
2. Dichlorodiphenyltrichloroethane (DDT)
3. History of Investigations on Affection of the Adrenal Glands by Subtoxic and Toxic Doses of DDT
4. Studies on the Effects of Low Doses of DDT on the Adrenal Glands
4.1. Disorders of Adrenal Gland Secretion Induced by Low Doses of DDT
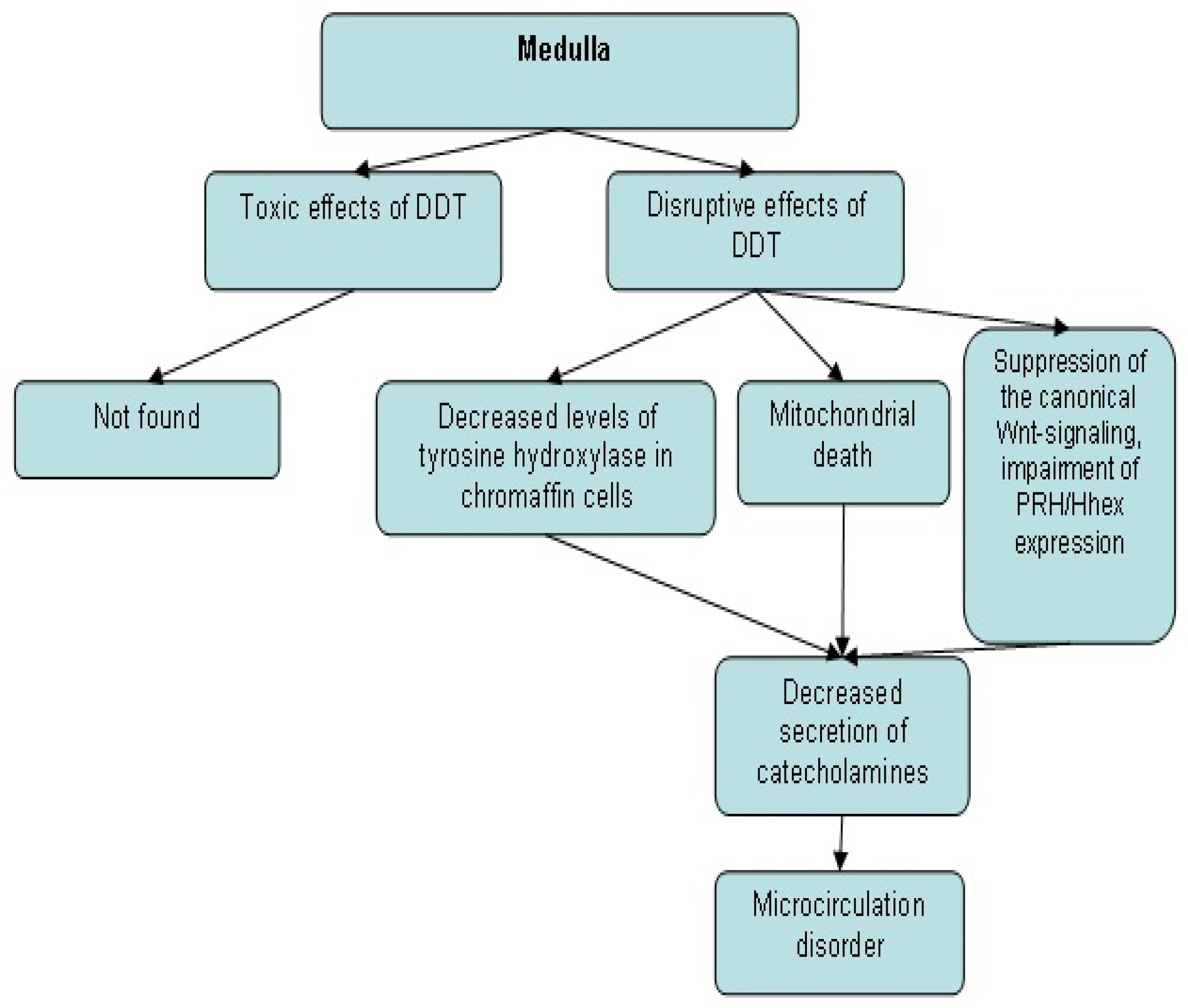
4.1.1. Disruption of Adrenal Medulla Hormone Secretion by Low-Dose Exposure to DDT
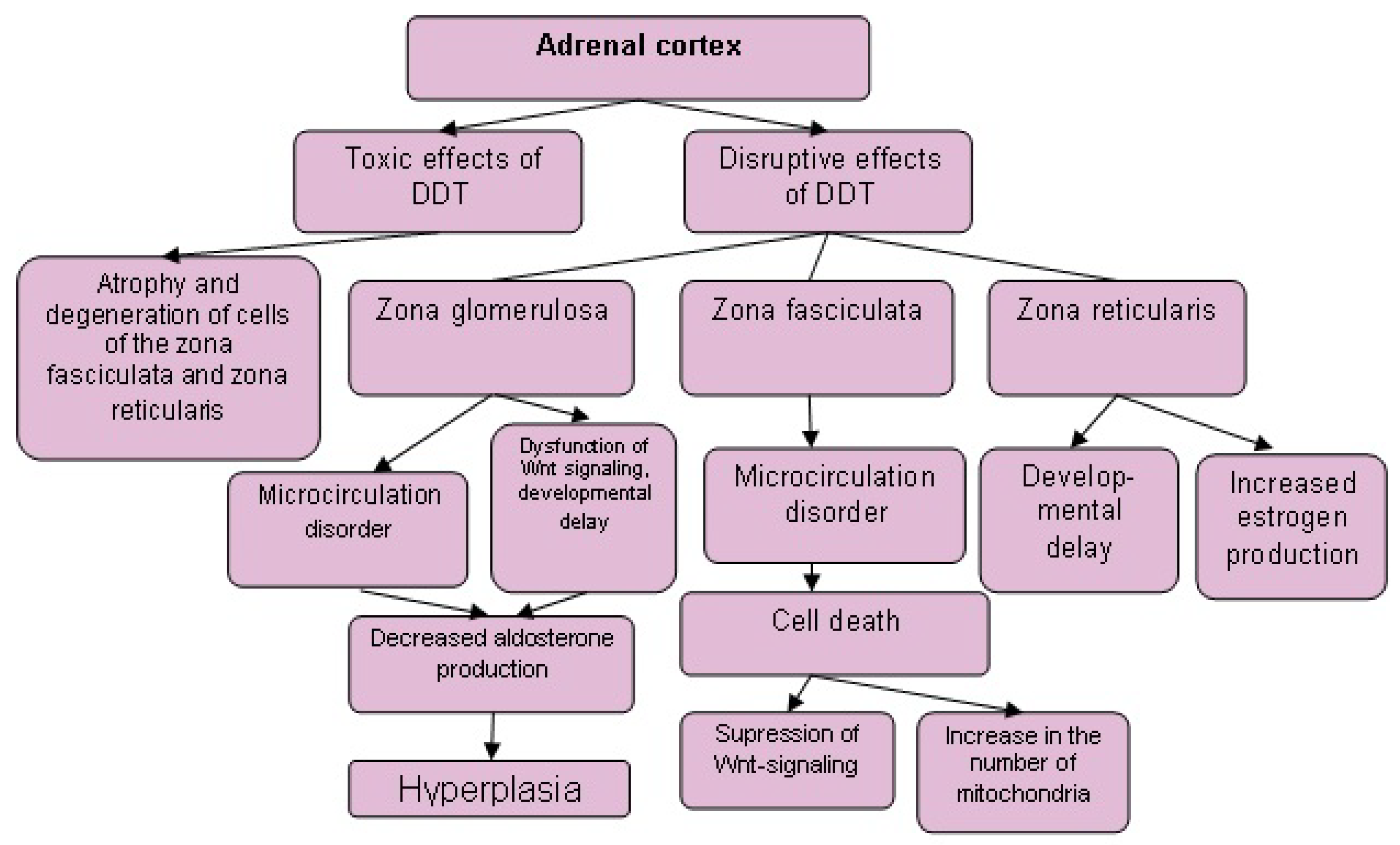
4.1.2. Disruption of Adrenal Cortex Hormone Secretion by Low-Dose Exposure to DDT
4.2. Changes in the Fine Structure of Adrenal Cells after Low-Dose Exposure to DDT
4.2.1. Ultrastructural Changes of Chromaffin Cells
4.2.2. Ultrastructural Changes of Zona Glomerulosa Cells
4.2.3. Ultrastructural Changes of Zona Fasciculata Cells
4.2.4. Ultrastructural Changes of Zona Reticularis Cells
5. Dysmorphogenetic Disorders in the Adrenal Glands Induced by Low-Dose Exposure to DDT
6. Comparison of the Effects of Exposure to Toxic and Disruptive Doses of DDT
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Zoeller, R.T.; Brown, T.R.; Doan, L.L.; Gore, A.C.; Skakkebaek, N.E.; Soto, A.M.; Woodruff, T.J.; Vom Saal, F.S.V. Endocrine-disrupting chemicals and public health protection: A statement of principles from The Endocrine Society. Endocrinology 2012, 153, 4097–4110. [Google Scholar] [CrossRef] [PubMed]
- Guillette, L.J., Jr. Endocrine disrupting contaminants—Beyond the dogma. Environ. Health Perspect. 2006, 114 (Suppl. 1), 9–12. [Google Scholar] [CrossRef] [PubMed]
- Vasseghian, Y.; Hosseinzadeh, S.; Khataee, A.; Dragoi, E.-N. The concentration of persistent organic pollutants in water resources: A global systematic review, meta-analysis and probabilistic risk assessment. Sci. Total. Environ. 2021, 796, 149000. [Google Scholar] [CrossRef] [PubMed]
- Colborn, T.; Saal, F.S.V.; Soto, A.M. Developmental effects of endocrine-disrupting chemicals in wildlife and humans. Environ. Health Perspect. 1993, 101, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.A.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. EDC-2: The Endocrine Society’s second scientific statement on endocrine-disrupting chemicals. Endocr. Rev. 2015, 36, E1–E150. [Google Scholar] [CrossRef]
- Kodavanti, P.R.S. Neurotoxicity of persistent organic pollutants: Possible mode(s) of action and further considerations. Dose Response 2005, 3, 273–305. [Google Scholar] [CrossRef] [PubMed]
- Mullerova, D.; Kopecky, J.; Matejkova, D.; Muller, L.; Rosmus, J.; Racek, J.; Sefrna, F.; Opatrna, S.; Kuda, O.; Matejovic, M. Negative association between plasma levels of adiponectin and polychlorinated biphenyl 153 in obese women under non-energy-restrictive regime. Int. J. Obes. 2008, 32, 1875–1878. [Google Scholar] [CrossRef]
- Dickerson, S.M.; Cunningham, S.L.; Patisaul, H.B.; Woller, M.J.; Gore, A.C. Endocrine disruption of brain sexual differentiation by developmental PCB exposure. Endocrinology 2011, 152, 581–594. [Google Scholar] [CrossRef]
- Castellani, F.; Manzoli, L.; Martellucci, C.A.; Flacco, M.E.; Astolfi, M.L.; Fabiani, L.; Mastrantonio, R.; Avino, P.; Protano, C.; Vitali, M. Levels of polychlorinated dibenzo-p-dioxins/furans and polychlorinated biphenyls in free-range hen eggs in central italy and estimated human dietary exposure. J. Food Prot. 2021, 84, 1455–1462. [Google Scholar] [CrossRef]
- Palioura, E.; Kandaraki, E.; Diamanti-Kandarakis, E. Endocrine disruptors and polycystic ovary syndrome: A focus on Bisphenol A and its potential pathophysiological aspects. Horm. Mol. Biol. Clin. Investig. 2014, 17, 137–144. [Google Scholar] [CrossRef]
- Molina, A.M.; Abril, N.; Lora, A.J.; Huertas-Abril, P.V.; Ayala, N.; Blanco, C.; Moyano, M. Proteomic profile of the effects of low-dose bisphenol A on zebrafish ovaries. Food Chem. Toxicol. 2021, 156, 112435. [Google Scholar] [CrossRef]
- Lema, S.C.; Dickey, J.T.; Schultz, I.R.; Swanson, P. Dietary exposure to 2,2′,4,4′-tetrabromodiphenyl ether (PBDE-47) alters thyroid status and thyroid hormone-regulated gene transcription in the pituitary and brain. Environ. Health Perspect. 2008, 116, 1694–1699. [Google Scholar] [CrossRef]
- Szabo, D.T.; Richardson, V.M.; Ross, D.G.; Diliberto, J.J.; Kodavanti, P.R.S.; Birnbaum, L.S. Effects of perinatal PBDE exposure on hepatic phase I, phase II, phase III, and deiodinase 1 gene expression involved in thyroid hormone metabolism in male rat pups. Toxicol. Sci. 2009, 107, 27–39. [Google Scholar] [CrossRef]
- Costa, L.G.; de Laat, R.; Tagliaferri, S.; Pellacani, C. A mechanistic view of polybrominated diphenyl ether (PBDE) developmental neurotoxicity. Toxicol. Lett. 2014, 230, 282–294. [Google Scholar] [CrossRef] [PubMed]
- Fisher, J.S. Environmental anti-androgens and male reproductive health: Focus on phthalates and testicular dysgenesis syndrome. Reproduction 2004, 127, 305–315. [Google Scholar] [CrossRef]
- Kimura, E.; Kohda, M.; Maekawa, F.; Fujii-Kuriyama, Y.; Tohyama, C. Neurons expressing the aryl hydrocarbon receptor in the locus coeruleus and island of Calleja major are novel targets of dioxin in the mouse brain. Histochem. Cell Biol. 2021, 156, 147–163. [Google Scholar] [CrossRef]
- Doğan, M.F.; Türkmen, N.B.; Taşlıdere, A.; Şahin, Y.; Çiftçi, O. The protective effects of capsaicin on oxidative damage-induced by 2,3,7,8-tetrachlorodibenzo-p-dioxin in rats. Drug Chem. Toxicol. 2021, 1, 8. [Google Scholar] [CrossRef]
- Yaglova, N.V.; Yaglov, V.V. Endocrine disruptors are a novel direction of endocrinologic scientific investigation. Ann. Russ. Acad. Med. Sci. USA 2012, 67, 56–61. [Google Scholar] [CrossRef][Green Version]
- Hamid, N.; Junaid, M.; Pei, D.-S. Combined toxicity of endocrine-disrupting chemicals: A review. Ecotoxicol. Environ. Saf. 2021, 215, 112136. [Google Scholar] [CrossRef]
- Street, M.E.; Angelini, S.; Bernasconi, S.; Burgio, E.; Cassio, A.; Catellani, C.; Cirillo, F.; Deodati, A.; Fabbrizi, E.; Fanos, V.; et al. Current knowledge on endocrine disrupting chemicals (EDCs) from animal biology to humans, from pregnancy to adulthood: Highlights from a national Italian meeting. Int. J. Mol. Sci. 2018, 19, 1647. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Possible Developmental Early Effects of Endocrine Disruptors on Child Health; WHO: Geneva, Switzerland, 2012; p. 93. [Google Scholar]
- Özen, S.; Darcan, Ş. Effects of environmental endocrine disruptors on pubertal development. J. Clin. Res. Pediatr. Endocrinol. 2011, 3, 1–6. [Google Scholar] [CrossRef]
- De Roos, A.J.; Schinasi, L.H.; Miligi, L.; Cerhan, J.R.; Bhatti, P.; Mannetje, A.; Baris, D.; Benavente, Y.; Benke, G.; Clavel, J.; et al. occupational insecticide exposure and risk of non-Hodgkin lymphoma: A pooled case-control study from the InterLymph Consortium. Int. J. Cancer 2021, 149, 1768–1786. [Google Scholar] [CrossRef]
- DDT and Its Derivatives: Environmental Aspects; United Nations Environment Programme: Nairobi, Kenya; The International Labour Organisation: Geneva, Switzerland; The World Health Organization: Geneva, Switzerland, 1973.
- Longnecker, M.P. Invited commentary: Why DDT matters now. Am. J. Epidemiology 2005, 162, 726–728. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Chevrier, J.; Rosas, L.G.; Anderson, H.A.; Bornman, M.S.; Bouwman, H.; Chen, A.; Cohn, B.A.; de Jager, C.; Henshel, D.S.; et al. The Pine River statement: Human health consequences of DDT use. Environ. Health Perspect. 2009, 117, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Grimsrud, T.K.; Langseth, H.; Barr, D.B.; Bassig, B.A.; Blair, A.; Cantor, K.P.; Gammon, M.D.; Lan, Q.; Rothman, N.; et al. Prediagnostic serum concentrations of organochlorine pesticides and non-Hodgkin lymphoma: A nested case-control study in the Norwegian Janus Serum Bank Cohort. Environ. Res. 2020, 187, 109515. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.M.; Bharat, G.K.; Chakraborty, P.; Martiník, J.; Audy, O.; Kukučka, P.; Přibylová, P.; Kukreti, P.K.; Sharma, A.; Kalina, J.; et al. A comprehensive assessment of endocrine-disrupting chemicals in an Indian food basket: Levels, dietary intakes, and comparison with European data. Environ. Pollut. 2021, 288, 117750. [Google Scholar] [CrossRef]
- Anand, N.; Chakraborty, P.; Ray, S. Human exposure to organochlorine, pyrethroid and neonicotinoid pesticides: Comparison between urban and semi-urban regions of India. Environ. Pollut. 2021, 270, 116156. [Google Scholar] [CrossRef]
- He, Y.; Guo, C.; Lv, J.; Deng, Y.; Xu, J. Occurrence, sources, and ecological risks of three classes of insecticides in sediments of the Liaohe River basin, China. Environ. Sci. Pollut. Res. 2021, 1, 10. [Google Scholar] [CrossRef]
- Negatu, B.; Dugassa, S.; Mekonnen, Y. Environmental and health risks of pesticide use in Ethiopia. J. Health Pollut. 2021, 11, 210601. [Google Scholar] [CrossRef]
- Bachman, M.J.; Keller, J.M.; West, K.L.; Jensen, B.A. Persistent organic pollutant concentrations in blubber of 16 species of cetaceans stranded in the Pacific Islands from 1997 through 2011. Sci. Total. Environ. 2014, 488–489, 115–123. [Google Scholar] [CrossRef]
- Bouwman, H.; Govender, D.; Underhill, L.; Polder, A. Chlorinated, brominated and fluorinated organic pollutants in African Penguin eggs: 30 years since the previous assessment. Chemosphere 2015, 126, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gałgowska, M.; Pietrzak-Fiećko, R. The level of selected organochlorine compounds residues in popular edible mushrooms from north-eastern Poland. Food Chem. 2021, 353, 129441. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Kong, D.; Li, L.; Zou, C.; Chen, F.; Li, M.; Cao, T.; Yu, C.; Song, J.; Jia, W.; et al. Distribution and sources of DDT and its metabolites in porewater and sediment from a typical tropical bay in the South China Sea. Environ. Pollut. 2020, 267, 115492. [Google Scholar] [CrossRef] [PubMed]
- Achour, A.; Derouiche, A.; Barhoumi, B.; Kort, B.; Cherif, D.; Bouabdallah, S.; Sakly, M.; Ben Rhouma, K.; Touil, S.; Driss, M.R.; et al. Organochlorine pesticides and polychlorinated biphenyls in human adipose tissue from northern Tunisia: Current extent of contamination and contributions of socio-demographic characteristics and dietary habits. Environ. Res. 2017, 156, 635–643. [Google Scholar] [CrossRef]
- Jugan, J.; Lind, P.M.; Salihovic, S.; Stubleski, J.; Kärrman, A.; Lind, L.; La Merrill, M.A. The associations between p,p’-DDE levels and plasma levels of lipoproteins and their subclasses in an elderly population determined by analysis of lipoprotein content. Lipids Health Dis. 2020, 19, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Oyinloye, J.; Oyekunle, J.; Ogunfowokan, A.; Msagati, T.; Adekunle, A.; Nety, S. Human health risk assessments of organochlorine pesticides in some food crops from Esa-Oke farm settlement, Osun State, Nigeria. Heliyon 2021, 7, e07470. [Google Scholar] [CrossRef]
- World Health Organization. Pesticide Residues in Food—2016 Evaluations. Part II–Toxicological; WHO and FAO: Geneva, Italy, 2017. [Google Scholar]
- Liang, C.-P.; Sack, C.; McGrath, S.; Cao, Y.; Thompson, C.J.; Robin, L.P. US Food and Drug Administration regulatory pesticide residue monitoring of human foods: 2009–2017. Food Addit. Contam. Part A 2021, 38, 1520–1538. [Google Scholar] [CrossRef]
- Cueto, C.; Brown, J.H.U.; Richardson, A.P., Jr. Biological studies on an adrenocorticolytic agent and the isolation of the active components. Endocrinology 1958, 62, 334–339. [Google Scholar] [CrossRef]
- Copeland, M.; Cranmer, M. Effects of o,p’-DDT on the adrenal gland and hepatic microsomal enzyme system in the beagle dog. Toxicol. Appl. Pharmacol. 1974, 27, 1–10. [Google Scholar] [CrossRef]
- Jukes, T.H. Effects of DDE. Nature 1995, 376, 545. [Google Scholar] [CrossRef]
- Lehman, J.W.; Peterle, T.J.; Mills, C.M. Effects of DDT on bobwhite quail (Colinus virginianus) adrenal gland. Bull. Environ. Contam. Toxicol. 1974, 11, 407–414. [Google Scholar] [CrossRef]
- Asp, V.; Ullerås, E.; Lindström, V.; Bergström, U.; Oskarsson, A.; Brandt, I. Biphasic hormonal responses to the adrenocorticolytic DDT metabolite 3-methylsulfonyl-DDE in human cells. Toxicol. Appl. Pharmacol. 2010, 242, 281–289. [Google Scholar] [CrossRef]
- Benecke, R.; Keller, E.; Vetter, B.; De Zeeuw, R.A. Plasma level monitoring of mitotane (o,p’-DDD) and its metabolite (o,p’-DDE) during long-term treatment of Cushing’s disease with low doses. Eur. J. Clin. Pharmacol. 1991, 41, 259–261. [Google Scholar] [CrossRef] [PubMed]
- Kasperlik-Zaluska, A. Clinical results of the use of mitotane for adrenocortical carcinoma. Braz. J. Med. Biol. Res. 2000, 33, 1191–1196. [Google Scholar] [CrossRef] [PubMed]
- Asp, V.; Lindström, V.; Olsson, J.A.; Bergström, U.; Brandt, I. Cytotoxicity and decreased corticosterone production in adrenocortical Y-1 cells by 3-methylsulfonyl-DDE and structurally related molecules. Arch. Toxicol. 2008, 83, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Lindhe, Ö.; Skogseid, B.; Brandt, I. Cytochrome P450-catalyzed binding of 3-methylsulfonyl-DDE and o,p’-DDD in human adrenal zona fasciculata/reticularis. J. Clin. Endocrinol. Metab. 2002, 87, 1319–1326. [Google Scholar] [CrossRef]
- Hartman, D. Neurophysiological Toxicology: Identification and Assesment of Human Neurotoxic Syndromes, 2nd ed.; Springer Science & Business Media: New York, NY, USA, 1995; p. 266. [Google Scholar]
- Ascherio, A.; Chen, H.; Weisskopf, M.G.; O’Reilly, E.; ScD, M.L.M.; Calle, E.E.; Schwarzschild, M.A.; Thun, M.J. Pesticide exposure and risk for Parkinson’s disease. Ann. Neurol. 2006, 60, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Hatcher, J.M.; DeLea, K.C.; Richardson, J.R.; Pennell, K.D.; Miller, G.W. Disruption of dopamine transport by DDT and its metabolites. Neurotoxicology 2008, 29, 682–690. [Google Scholar] [CrossRef]
- Burlington, H.; Lindeman, V.F. Effect of DDT on testes and secondary sex characters of white leghorn cockerels. Exp. Biol. Med. 1950, 74, 48–51. [Google Scholar] [CrossRef]
- Louis, G.M.B.; Smarr, M.M.; Sun, L.; Chen, Z.; Honda, M.; Wang, W.; Karthikraj, R.; Weck, J.; Kannan, K. Endocrine disrupting chemicals in seminal plasma and couple fecundity. Environ. Res. 2018, 163, 64–70. [Google Scholar] [CrossRef]
- Rhouma, K.B.; Tébourbi, O.; Krichah, R.; Sakly, M. Reproductive toxicity of DDT in adult male rats. Hum. Exp. Toxicol. 2001, 20, 393–397. [Google Scholar] [CrossRef]
- Anand, M.; Taneja, A. Organochlorine pesticides residue in placenta and their influence on anthropometric measures of infants. Environ. Res. 2020, 182, 109106. [Google Scholar] [CrossRef]
- Yin, S.; Zhang, J.; Guo, F.; Poma, G.; Covaci, A.; Liu, W. Transplacental transfer mechanism of organochlorine pesticides: An in vitro transcellular transport study. Environ. Int. 2020, 135, 105402. [Google Scholar] [CrossRef]
- Vizcaino, E.; Grimalt, J.; Somoano, A.F.; Tardon, A. Transport of persistent organic pollutants across the human placenta. Environ. Int. 2014, 65, 107–115. [Google Scholar] [CrossRef]
- Zhu, Z.; Cao, F.; Li, X. Epigenetic programming and fetal metabolic programming. Front. Endocrinol. 2019, 10, 764. [Google Scholar] [CrossRef]
- El-Saeid, M.H.; Hassanin, A.S.; Bazeyad, A.Y. Levels of pesticide residues in breast milk and the associated risk assessment. Saudi J. Biol. Sci. 2021, 28, 3741–3744. [Google Scholar] [CrossRef]
- Witczak, A.; Pohoryło, A.; Abdel-Gawad, H. Endocrine-disrupting organochlorine pesticides in human breast milk: Changes during lactation. Nutrients 2021, 13, 229. [Google Scholar] [CrossRef]
- Zhang, X.; Cheng, C.; Zhang, G.; Xiao, M.; Li, L.; Wu, S.; Lu, X. Co-exposure to BPA and DEHP enhances susceptibility of mammary tumors via up-regulating Esr1/HDAC6 pathway in female rats. Ecotoxicol. Environ. Saf. 2021, 221, 112453. [Google Scholar] [CrossRef] [PubMed]
- Fang, G.-Z.; Huang, G.-Y.; Ying, G.-G.; Qiu, S.-Q.; Shi, W.-J.; Xie, L.; Yang, Y.-Y.; Ma, D.-D. Endocrine disrupting effects of binary mixtures of 17β-estradiol and testosterone in adult female western mosquitofish (Gambusia affinis). Ecotoxicol. Environ. Saf. 2021, 208, 111566. [Google Scholar] [CrossRef] [PubMed]
- Yaglova, N.; Tsomartova, D.; Yaglov, V. Effect of prenatal and postnatal exposure to low doses of DDT on catecholamine secretion in rats in different period of ontogeny. Bull. Exp. Biol. Med. 2017, 163, 422–424. [Google Scholar] [CrossRef] [PubMed]
- Dronjak, S.; Gavrilovic, L. Effects of stress on catecholamine stores in central and peripheral tissues of long-term socially isolated rats. Braz. J. Med. Biol. Res. 2006, 39, 785–790. [Google Scholar] [CrossRef]
- Timokhina, E.; Nazimova, S.; Tsomartova, D.; Yaglova, N.; Obernikhin, S.; Yaglov, V. Morphological and cytophysiological changes in the adult rat adrenal medulla after prenatal and postnatal exposure to endocrine-disrupting DDT. Sovrem. Teh. Med. 2020, 12, 50. [Google Scholar] [CrossRef]
- Yaglova, N.V.; Obernikhin, S.S.; Yaglov, V.V.; Tsomartova, D.A.; Nazimova, S.V.; Timokhina, E.P. Ultrastructural mechanisms of impaired aldosterone synthesis in rats exposed to DDT during prenatal and postnatal development. Bull. Exp. Biol. Med. 2020, 170, 101–105. [Google Scholar] [CrossRef]
- Yaglova, N.; Tsomartova, D.; Yaglov, V. Differences in production of adrenal steroid hormones in pubertal rats exposed to low doses of the endocrine disruptor DDT during prenatal and postnatal development. Biochem. Suppl. Ser. B Biomed. Chem. 2018, 12, 80–86. [Google Scholar] [CrossRef]
- Brock, B.J.; Waterman, M.R. Biochemical differences between rat and human cytochrome P450c17 support the different steroidogenic needs of these two species. Biochemistry 1999, 38, 1598–1606. [Google Scholar] [CrossRef]
- Bruder, E.D.; Nagler, A.K.; Raff, H. Oxygen-dependence of ACTH-stimulated aldosterone and corticosterone synthesis in the rat adrenal cortex: Developmental aspects. J. Endocrinol. 2002, 172, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Tezuka, Y.; Ishii, K.; Zhao, L.; Yamazaki, Y.; Morimoto, R.; Sasano, H.; Udager, A.M.; Satoh, F.; Turcu, A.F. ACTH stimulation maximizes the accuracy of peripheral steroid profiling in primary aldosteronism subtyping. J. Clin. Endocrinol. Metab. 2021, 420. [Google Scholar] [CrossRef]
- Engels, M.; Pijnenburg-Kleizen, K.J.; Utari, A.; Faradz, S.M.H.; Oude-Alink, S.; Van Herwaarden, A.; Span, P.N.; Sweep, F.C.; Van Der Grinten, H.L.C. Glucocorticoid activity of adrenal steroid precursors in untreated patients with congenital adrenal hyperplasia. J. Clin. Endocrinol. Metab. 2019, 104, 5065–5072. [Google Scholar] [CrossRef] [PubMed]
- Yaglova, N.; Tsomartova, D.; Obernikhin, S.; Yaglov, V.; Nazimova, S.; Tsomartova, E.; Chereshneva, E.; Ivanova, M.; Lomanovskaya, T. Differential disrupting effects of prolonged low-dose exposure to dichlorodiphenyltrichloroethane on androgen and estrogen production in males. Int. J. Mol. Sci. 2021, 22, 3155. [Google Scholar] [CrossRef] [PubMed]
- García-Sancho, J.; De Diego, A.M.G.; Garcia, A.G. Mitochondria and chromaffin cell function. Pflugers Arch. 2012, 464, 33–41. [Google Scholar] [CrossRef]
- Villanueva, J.; Viniegra, S.; Gimenez-Molina, Y.; García-Martinez, V.; Expósito-Romero, G.; del Mar Frances, M.; García-Sancho, J.; Gutiérrez, L.M. The position of mitochondria and ER in relation to that of the secretory sites in chromaffin cells. J. Cell Sci. 2014, 127, 5105–5114. [Google Scholar] [CrossRef] [PubMed]
- Yaglova, N.; Tsomartova, D.; Yaglov, V.; Obernikhin, S.; Nazimova, S. Altered secretion of adrenomedullary a-cells in rats after developmental exposure to low-dose dichlorodiphenyltrichloroethane. Clin. Exp. Morphol. 2018, 27, 30–34. [Google Scholar] [CrossRef]
- Nunnari, J.; Suomalainen, A. Mitochondria: In sickness and in health. Cell 2012, 148, 1145–1159. [Google Scholar] [CrossRef] [PubMed]
- Pignatelli, D.; Pinto, P.; Magalhães, M. The development of the adrenal gland zona glomerulosa in the rat. A morphological, immunohistochemical and biochemical study. Mol. Cell. Endocrinol. 1998, 140, 163–168. [Google Scholar] [CrossRef]
- Yaglova, N.; Obernikhin, S.; Yaglov, V.; Timokhina, E.; Nazimova, S.; Tsomartova, D. Age-dependent changes of mitochondrial structure regulate steroidogenic activity of rat adrenal cortical cells. Clin. Exp. Morphol. 2020, 9, 64–70. [Google Scholar] [CrossRef]
- Yaglova, N.V.; Obernikhin, S.S.; Yaglov, V.V.; Nazimova, S.V.; Timokhina, E.P. Cellular mechanisms of impaired mineralocorticoid and glucocorticoid production by endocrine disruptor DDT. J. Belarusian State Univ. Biol. 2021, 1, 12–19. [Google Scholar] [CrossRef]
- Yaglova, N.V.; Tsomartova, D.; Obernikhin, S.; Nazimova, S.; Yaglov, V. Changes in secretory activity of adrenal zona fasciculata cells in pubertal rats exposed to low doses of DDT in different periods of ontogeny. Bull. Exp. Biol. Med. 2018, 166, 283–286. [Google Scholar] [CrossRef]
- Tsomartova, D.A.; Yaglova, N.V.; Yaglov, V.V.; Obernikhin, S.S.; Nazimova, S.V.; Sledneva, Y.P. Morphological changes of the reticular zone of the cortex of adrenal glands and of the secretory activity of its corticosterocytes in the pubertal period in rats developing at the influence of the endocrinal disruptor dichlorodiphenyltrichloroethane. Morphol. Newsl. 2018, 26, 22–25. [Google Scholar] [CrossRef]
- Timokhina, E.P.; Yaglov, V.V.; Obernikhin, S.S.; Yaglova, N.V.; Nazimova, S.V.; Tsomartova, D.A. Role of reorganization of mitochondria structure in age-dependent changes of adrenal cortex function. J. Belarusian State Univ. Biol. 2020, 3, 13–18. [Google Scholar] [CrossRef]
- Val, P.; Swain, A. Gene dosage effects and transcriptional regulation of early mammalian adrenal cortex development. Mol. Cell. Endocrinol. 2010, 323, 105–114. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Katoh, M.; Katoh, M. Wnt signaling pathway and stem cell signaling network. Clin. Cancer Res. 2007, 13, 4042–4045. [Google Scholar] [CrossRef]
- Roop, D.; Toftgard, R. Hedgehog in Wnterland. Nat. Genet. 2008, 40, 1040–1041. [Google Scholar] [CrossRef]
- Alvarez-Medina, R.; Cayuso, J.; Okubo, T.; Takada, S.; Martí, E. Wnt canonical pathway restricts graded Shh/Gli patterning activity through the regulation of Gli3 expression. Development 2008, 135, 237–247. [Google Scholar] [CrossRef]
- Ramakrishnan, A.-B.; Cadigan, K.M. Wnt target genes and where to find them. F1000Research 2017, 6, 746. [Google Scholar] [CrossRef] [PubMed]
- Voloshanenko, O.; Schwartz, U.; Kranz, D.; Rauscher, B.; Linnebacher, M.; Augustin, I.; Boutros, M. β-catenin-independent regulation of Wnt target genes by RoR2 and ATF2/ATF4 in colon cancer cells. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Baum, B.; Georgiou, M. Dynamics of adherens junctions in epithelial establishment, maintenance, and remodeling. J. Cell Biol. 2011, 192, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Brembeck, F.H.; Schwarz-Romond, T.; Bakkers, J.; Wilhelm, S.; Hammerschmidt, M.; Birchmeier, W. Essential role of BCL9-2 in the switch between beta-catenin’s adhesive and transcriptional functions. Genes Dev. 2004, 18, 2225–2230. [Google Scholar] [CrossRef]
- Brembeck, F.H.; Rosário, M.; Birchmeier, W. Balancing cell adhesion and Wnt signaling, the key role of beta-catenin. Curr. Opin. Genet. Dev. 2006, 16, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Gumbiner, B.; Stevenson, B.; Grimaldi, A. The role of the cell adhesion molecule uvomorulin in the formation and maintenance of the epithelial junctional complex. J. Cell Biol. 1988, 107, 1575–1587. [Google Scholar] [CrossRef]
- Yaglova, N.V.; Tsomartova, D.A.; Obernikhin, S.S.; Nazimova, S.V.; Ivanova, M.Y.; Chereshneva, E.V.; Yaglov, V.V.; Lomanovskaya, T.A. Transcription factors β-catenin and Hex in postnatal development of the rat adrenal cortex: Implication in proliferation control. Heliyon 2021, 7, e05932. [Google Scholar] [CrossRef]
- Berthon, A.; Sahut-Barnola, I.; Lambert-Langlais, S.; De Joussineau, C.; Damon-Soubeyrand, C.; Louiset, E.; Taketo, M.M.; Tissier, F.; Bertherat, J.; Lefrançois-Martinez, A.-M.; et al. Constitutive beta-catenin activation induces adrenal hyperplasia and promotes adrenal cancer development. Hum. Mol. Genet. 2010, 19, 1561–1576. [Google Scholar] [CrossRef] [PubMed]
- Berthon, A.; Drelon, C.; Ragazzon, B.; Boulkroun, S.; Tissier, F.; Amar, L.; Samson-Couterie, B.; Zennaro, M.-C.; Plouin, P.-F.; Skah, S.; et al. Wnt/β-catenin signalling is activated in aldosterone-producing adenomas and controls aldosterone production. Hum. Mol. Genet. 2014, 23, 889–905. [Google Scholar] [CrossRef] [PubMed]
- Walczak, E.M.; Kuick, R.; Finco, I.; Bohin, N.; Hrycaj, S.M.; Wellik, D.M.; Hammer, G.D. Wnt signaling inhibits adrenal steroidogenesis by cell-autonomous and non-cell-autonomous mechanisms. Mol. Endocrinol. 2014, 28, 1471–1486. [Google Scholar] [CrossRef] [PubMed]
- Yaglova, N.; Obernikhin, S.; Nazimova, S.; Yaglov, V. Developmental exposure to endocrine disruptor dichlorodiphenyltrichloroethane alters transcriptional regulation of postnatal morphogenesis of adrenal zona fasciculata. Saudi J. Biol. Sci. 2020, 27, 3655–3659. [Google Scholar] [CrossRef]
- Yaglova, N.V.; Obernikhin, S.S.; Nazimova, S.V.; Yaglov, V.V.; Kosmachevskaya, O.V.; Topunov, A.F. Changes in transcriptional regulation of postnatal morphogenesis of the adrenal zona fasciculata caused by endocrine disruptor dichlorodiphenyltrichloroethane. Bull. Exp. Biol. Med. 2020, 168, 797–801. [Google Scholar] [CrossRef]
- Tsomartova, D.; Yaglova, N.; Yaglov, V. Changes in canonical β-catenin/Wnt signaling activation in the adrenal cortex of rats exposed to endocrine disruptor dichlorodiphenyltrichloroethane (DDT) during prenatal and postnatal ontogeny. Bull. Exp. Biol. Med. 2018, 164, 493–496. [Google Scholar] [CrossRef]
- Yaglova, N.V.; Tsomartova, D.A.; Obernikhin, S.; Nazimova, S.V. The role of the canonical Wnt-signaling pathway in morphogenesis and regeneration of the adrenal cortex in rats exposed to the endocrine disruptor dichlorodiphenyltrichloroethane during prenatal and postnatal development. Biol. Bull. 2019, 46, 74–81. [Google Scholar] [CrossRef]
- Tsomartova, D.A.; Yaglova, N.V.; Obernikhin, S.S.; Nazimova, S.V.; Yaglov, V.V. Secretion of adrenal zona glomerulosa cells in rats exposed to low doses of dichlorodiphenyltrichloroethane during prenatal and postnatal development. Russ. Open Med. J. 2018, 7, e0302. [Google Scholar] [CrossRef]
- Yaglova, N.V.; Obernikhin, S.S.; Yaglov, V.V.; Nazimova, S.V.; Timokhina, E.P.; Tsomartova, D.A. Low-dose exposure to endocrine disruptor dichlorodiphenyltrichloroethane (DDT) affects transcriptional regulation of adrenal zona reticularis in male rats. Bull. Exp. Biol. Med. 2021, 170, 682–685. [Google Scholar] [CrossRef]
- Yaglova, N.; Obernikhin, S.; Tsomartova, D.; Nazimova, S.; Yaglov, V.; Tsomartova, E.; Chereshneva, E.; Ivanova, M.; Lomanovskaya, T. Impaired morphogenesis and function of rat adrenal zona glomerulosa by developmental low-dose exposure to DDT is associated with altered Oct4 expression. Int. J. Mol. Sci. 2021, 22, 6324. [Google Scholar] [CrossRef]
- Jönsson, J.; Rodriguez-Martinez, H.; Brandt, I. Transplacental toxicity of 3-methylsulphonyl-DDE in the developing adrenal cortex in mice. Reprod. Toxicol. 1995, 9, 257–264. [Google Scholar] [CrossRef]
- Villa, R.; Orlandi, L.; Berruti, A.; Dogliotti, L.; Zaffaroni, N. Modulation of cytotoxic drug activity by mitotane and lonidamine in human adrenocortical carcinoma cells. Int. J. Oncol. 1999, 14, 133–138. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Timokhina, E.P.; Yaglov, V.V.; Nazimova, S.V. Dichlorodiphenyltrichloroethane and the Adrenal Gland: From Toxicity to Endocrine Disruption. Toxics 2021, 9, 243. https://doi.org/10.3390/toxics9100243
Timokhina EP, Yaglov VV, Nazimova SV. Dichlorodiphenyltrichloroethane and the Adrenal Gland: From Toxicity to Endocrine Disruption. Toxics. 2021; 9(10):243. https://doi.org/10.3390/toxics9100243
Chicago/Turabian StyleTimokhina, Ekaterina P., Valentin V. Yaglov, and Svetlana V. Nazimova. 2021. "Dichlorodiphenyltrichloroethane and the Adrenal Gland: From Toxicity to Endocrine Disruption" Toxics 9, no. 10: 243. https://doi.org/10.3390/toxics9100243
APA StyleTimokhina, E. P., Yaglov, V. V., & Nazimova, S. V. (2021). Dichlorodiphenyltrichloroethane and the Adrenal Gland: From Toxicity to Endocrine Disruption. Toxics, 9(10), 243. https://doi.org/10.3390/toxics9100243





