Temporal Persistence of Bromadiolone in Decomposing Bodies of Common Kestrel (Falco tinnunculus)
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Set-Up
2.2. Sample Acquisition
2.3. Chemicals and Reagents
2.4. Sample Preparation and Chemical Analysis
2.5. Instruments and Conditions
2.6. Statistics
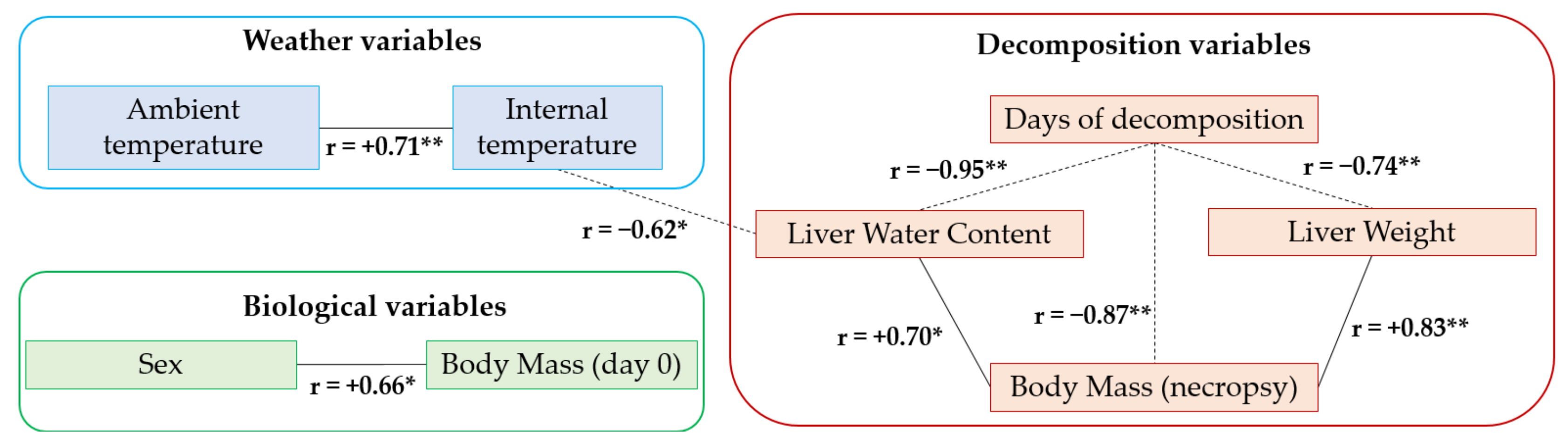
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Berny, P.; Buronfosse, T.; Lorgue, G. Anticoagulant Poisoning in Animals: A Simple New High-Performance Thin-Layer Chromatographic (HPTLC) Method for the Simultaneous Determination of Eight Anticoagulant Rodenticides in Liver Samples. J. Anal. Toxicol. 1995, 19, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Hosea, R.C. Exposure of non-target wildlife to anticoagulant rodenticides in California. Proc. Vertebr. Pest Conf. 2000, 19. [Google Scholar] [CrossRef]
- Winters, A.M.; Rumbeiha, W.K.; Winterstein, S.R.; Fine, A.E.; Munkhtsog, B.; Hickling, G.J. Residues in Brandt’s voles (Microtus brandti) exposed to bromadiolone-impregnated baits in Mongolia. Ecotoxicol. Environ. Saf. 2010, 73, 1071–1077. [Google Scholar] [CrossRef]
- Ruiz-Suárez, N.; Henríquez-Hernández, L.A.; Valerón, P.F.; Boada, L.D.; Zumbado, M.; Camacho, M.; Almeida-González, M.; Luzardo, O.P. Assessment of anticoagulant rodenticide exposure in six raptor species from the Canary Islands (Spain). Sci. Total Environ. 2014, 485–486, 371–376. [Google Scholar] [CrossRef]
- van den Brink, N.W.; Elliott, J.E.; Shore, R.F.; Rattner, B.A. Anticoagulant Rodenticides and Wildlife: Concluding Remarks; Springer: Berlin/Heidelberg, Germany, 2018; pp. 379–386. ISBN 9783319643755. [Google Scholar]
- Furie, B.; Bouchard, B.A.; Furie, B.C. Vitamin K-dependent biosynthesis of gamma-carboxyglutamic acid. Blood 1999, 93, 1798–1808. [Google Scholar]
- Crowell, M.D.; Broome, K.G.; Eason, C.T.; Fairweather, A.A.C.; Ogilvie, S.; Murphy, E.C. How long do vertebrate pesticides persist in living mammals? Priorities for research. DOC Res. Dev. 2013, 337, 1–18. [Google Scholar]
- Rattner, B.A.; Mastrota, F.N. Anticoagulant rodenticide toxicity to non-target wildlife under controlled exposure conditions. In Anticoagulant rodenticides and wildlife; Springer: Berlin/Heidelberg, Germany, 2018; pp. 45–86. [Google Scholar]
- Berny, P. Pesticides and the intoxication of wild animals. J. Vet. Pharmacol. Ther. 2007, 30, 93–100. [Google Scholar] [CrossRef]
- Mateo-Tomás, P.; Olea, P.P.; Sánchez-Barbudo, I.S.; Mateo, R. Alleviating human-wildlife conflicts: Identifying the causes and mapping the risk of illegal poisoning of wild fauna. J. Appl. Ecol. 2012, 49, 376–385. [Google Scholar] [CrossRef]
- Ruiz-Suárez, N.; Boada, L.D.; Henríquez-Hernández, L.A.; González-Moreo, F.; Suárez-Pérez, A.; Camacho, M.; Zumbado, M.; Almeida-González, M.; del Mar Travieso-Aja, M.; Luzardo, O.P. Continued implication of the banned pesticides carbofuran and aldicarb in the poisoning of domestic and wild animals of the Canary Islands (Spain). Sci. Total Environ. 2015, 505, 1093–1099. [Google Scholar] [CrossRef]
- Directive 2009/147/EC of the European Parliament and of the council of 30 November 2009 on the conservation of wild bird. Off. J. Eur. Union 2010, L 20/7. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32009L0147&from=EN (accessed on 12 January 2020).
- Council Directive 92/43/EC of 21 May 1992 on the conservation of natural habitats and of wild fauna and flora. Off. J. Eur. Union 1992, L 206/7-50. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:31992L0043&from=EN (accessed on 12 January 2020).
- Gabriel, M.W.; Woods, L.W.; Poppenga, R.; Sweitzer, R.A.; Thompson, C.; Matthews, S.M.; Higley, J.M.; Keller, S.M.; Purcell, K.; Barrett, R.H.; et al. Anticoagulant Rodenticides on our Public and Community Lands: Spatial Distribution of Exposure and Poisoning of a Rare Forest Carnivore. PLoS ONE 2012, 7, e40163. [Google Scholar] [CrossRef]
- Proulx, G.; Rodtka, D. Predator bounties in Western Canada cause animal suffering and compromise wildlife conservation efforts. Animals 2015, 5, 1034–1046. [Google Scholar] [CrossRef]
- Ogada, D.L. The power of poison: Pesticide poisoning of Africa’s wildlife. Ann. N. Y. Acad. Sci. 2014, 1322, 1–20. [Google Scholar] [CrossRef]
- Guitart, R.; Croubels, S.; Caloni, F.; Sachana, M.; Davanzo, F.; Vandenbroucke, V.; Berny, P. Animal poisoning in Europe Part 1: Farm livestock and poultry. Vet. J. 2010, 183, 249–254. [Google Scholar] [CrossRef]
- Albert, C.A.; Wilson, L.K.; Elliott, J.E. Anticoagulant Rodenticides in Three Owl Species from Western Canada, 1988-2003. Arch. Environ. Contam. Toxicol. 2010, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Barbudo, I.S.; Camarero, P.R.; Mateo, R. Primary and secondary poisoning by anticoagulant rodenticides of non-target animals in Spain. Sci. Total Environ. 2012, 420, 280–288. [Google Scholar] [CrossRef]
- López-Perea, J.J.; Camarero, P.R.; Molina-López, R.A.; Parpal, L.; Obón, E.; Solá, J.; Mateo, R. Interspecific and geographical differences in anticoagulant rodenticide residues of predatory wildlife from the Mediterranean region of Spain. Sci. Total Environ. 2015, 511, 259–267. [Google Scholar] [CrossRef]
- Mcfarland, S.E.; Mischke, R.H.; Hopster-Iversen, C.; Von Krueger, X.; Ammer, H.; Potschka, H.; Stürer, A.; Begemann, K.; Desel, H.; Greiner, M. Systematic account of animal poisonings in Germany, 2012–2015. Vet. Rec. 2017, 180, 327. [Google Scholar] [CrossRef]
- Poché, R.M. Rodent tissue residue and secondary hazard studies with bromadiolone. EPPO Bull. 1988, 18, 323–330. [Google Scholar] [CrossRef]
- Fournier-Chambrillon, C.; Berny, P.J.; Coiffier, O.; Barbedienne, P.; Dassé, B.; Delas, G.; Hubert, G.; Mazet, A.; Pouzenc, P.; Rosoux, R.; et al. Evidence of secondary poisoning of free-ranging riparian mustelids by anticoagulant rodenticides in France: Implications for conservation of European mink (Mustela lutreola). J. Wildl. Dis. 2004, 40, 688–695. [Google Scholar] [CrossRef]
- Regulation (EU) No 528/2012 of the European Parliament and of the Council of 22 May 2012 concerning the making available on the market and use of biocidal products. Off. J. Eur. Union 2012, 1–123. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2012:167:0001:0123:EN:PDF (accessed on 1 January 2020).
- Jacob, J.; Buckle, A. Use of anticoagulant rodenticides in different applications around the world. In Anticoagulant Rodenticides and Wildlife; Springer: Berlin/Heidelberg, Germany, 2018; pp. 11–43. [Google Scholar]
- Regulation (EC) No 1107/2009 of the European Parliament and of the Council of 21 October 2009 concerning the placing of plant protection products on the market and repealing Council Directives 79/117/EEC and 91/414/EEC. Off. J. Eur. Union 2009, 309, 1–50. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2009:309:0001:0050:en:PDF (accessed on 13 January 2020).
- Slankard, K.G.; Gaskill, C.L.; Cassone, L.M.; Rhoden, C.M. Changes in Detected Anticoagulant Rodenticide Exposure in Barn Owls (Tyto alba) in Kentucky, USA, in 2012–16. J. Wildl. Dis. 2019, 55, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Lohr, M.T.; Davis, R.A. Anticoagulant rodenticide use, non-target impacts and regulation: A case study from Australia. Sci. Total Environ. 2018, 634, 1372–1384. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Avian, D.A.B.V.P. Anticoagulant Rodenticide Exposure and Toxicosis in Four Species of Birds of Prey Presented to a Wildlife Clinic in Massachusetts, 2006–2010. J. Zoo Wildl. Med. 2011, 42, 88–97. [Google Scholar] [CrossRef]
- Christensen, T.K.; Lassen, P.; Elmeros, M. High exposure rates of anticoagulant rodenticides in predatory bird species in intensively managed landscapes in Denmark. Arch. Environ. Contam. Toxicol. 2012, 63, 437–444. [Google Scholar] [CrossRef]
- Langford, K.H.; Reid, M.; Thomas, K.V. The occurrence of second generation anticoagulant rodenticides in non-target raptor species in Norway. Sci. Total Environ. 2013, 450–451, 205–208. [Google Scholar] [CrossRef]
- Hughes, J.; Sharp, E.; Taylor, M.J.; Melton, L.; Hartley, G. Monitoring agricultural rodenticide use and secondary exposure of raptors in Scotland. Ecotoxicology 2013, 22, 974–984. [Google Scholar] [CrossRef]
- Stansley, W.; Cummings, M.; Vudathala, D.; Murphy, L.A. Anticoagulant Rodenticides in Red-Tailed Hawks, Buteo jamaicensis, and Great Horned Owls, Bubo virginianus, from New Jersey, USA, 2008–2010. Bull. Environ. Contam. Toxicol. 2014, 92, 6–9. [Google Scholar] [CrossRef]
- Lambert, O.; Pouliquen, H.; Larhantec, M.; Thorin, C.; L’Hostis, M. Exposure of raptors and waterbirds to anticoagulant rodenticides (difenacoum, bromadiolone, coumatetralyl, coumafen, brodifacoum): Epidemiological survey in Loire Atlantique (France). Bull. Environ. Contam. Toxicol. 2007, 79, 91–94. [Google Scholar] [CrossRef] [PubMed]
- Berny, P.; Gaillet, J.R. Acute poisoning of Red Kites (Milvus milvus) in France: Data from the SAGIR network. J. Wildl. Dis. 2008, 44, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.A.; Turk, A.; Long, S.M.; Wienburg, C.L.; Best, J.; Shore, R.F. Second generation anticoagulant rodenticides in tawny owls (Strix aluco) from Great Britain. Sci. Total Environ. 2008, 392, 93–98. [Google Scholar] [CrossRef]
- Hong, S.Y.; Morrissey, C.; Lin, H.S.; Lin, K.S.; Lin, W.L.; Yao, C.T.; Lin, T.E.; Chan, F.T.; Sun, Y.H. Frequent detection of anticoagulant rodenticides in raptors sampled in Taiwan reflects government rodent control policy. Sci. Total Environ. 2019, 691, 1051–1058. [Google Scholar] [CrossRef]
- García-Fernández, A.J. Avian ecotoxicology. In Encyclopedia of Toxicology, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 289–294. [Google Scholar]
- United States Environmental Protection Agency. Pesticide Fact Sheet. Name of Chemical: Difenacoum. Reason for Issuance: New Rodenticide. Off. Prev. Pestic. Toxic Subst. 2007, 7501C. Available online: https://www3.epa.gov/pesticides/chem_search/reg_actions/registration/fs_PC-011901_01-Sep-07.pdf (accessed on 23 January 2020).
- Brown, P.M.; Turnbull, G.; Charman, S.; Charlton, A.J.; Jones, A. Analytical Methods Used in the United Kingdom Wildlife Incident Investigation Scheme for the Detection ofAnimal Poisoning by Pesticides. J. AOAC Int. 2005, 88, 204–220. [Google Scholar] [CrossRef]
- Berny, P.; Buronfosse, T.; Buronfosse, F.; Lamarque, F.; Lorgue, G. Field evidences of secondary poisoning of foxes (Vulpes vulpes) and buzzards (Buteo buteo) by bromadiolone, a 4-year survey. Chemosphere 1997, 35, 1817–1829. [Google Scholar] [CrossRef]
- Martínez-López, E.; Romero, D.; Navas, I.; Gerique, C.; Jiménez, P.; García-Fernández, A.J. Detection of strychnine by gas chromatography-mass spectrometry in the carcass of a Bonelli’s eagle (Hieraaetus fasciatus). Vet. Rec. 2006, 159, 182–184. [Google Scholar]
- Valverde, I.; Espín, S.; Navas, I.; María-Mojica, P.; Gil, J.M.; García-Fernández, A.J. Protocol to classify the stages of carcass decomposition and estimate the time of death in small-size raptors. Eur. J. Wildl. Res. 2020, 66, 1–13. [Google Scholar] [CrossRef]
- Thomas, P.J.; Mineau, P.; Shore, R.F.; Champoux, L.; Martin, P.A.; Wilson, L.K.; Fitzgerald, G.; Elliott, J.E. Second generation anticoagulant rodenticides in predatory birds: Probabilistic characterisation of toxic liver concentrations and implications for predatory bird populations in Canada. Environ. Int. 2011, 37, 914–920. [Google Scholar] [CrossRef]
- Coeurdassier, M.; Poirson, C.; Paul, J.P.; Rieffel, D.; Michelat, D.; Reymond, D.; Legay, P.; Giraudoux, P.; Scheifler, R. The diet of migrant Red Kites Milvus milvus during a Water Vole Arvicola terrestris outbreak in eastern France and the associated risk of secondary poisoning by the rodenticide bromadiolone. Ibis 2012, 154, 136–146. [Google Scholar] [CrossRef]
- Stone, W.B.; Okoniewski, J.C.; Stedelin, J.R. Poisoning of Wildlife with Anticoagulant Rodenticides in New York. J. Wildl. Rehabil. 1999, 23, 13–17. [Google Scholar] [CrossRef]
- Giraudoux, P.; Tremollières, C.; Barbier, B.; Defaut, R.; Rieffel, D.; Bernard, N.; Lucot, É.; Berny, P. Persistence of bromadiolone anticoagulant rodenticide in Arvicola terrestris populations after field control. Environ. Res. 2006, 102, 291–298. [Google Scholar] [CrossRef]
- Suárez, O.V.; Cueto, G.R. Comparison of efficacy of second-generation anticoagulant rodenticides: Effect of active ingredients, type of formulation and commercial suppliers. Cogent Food Agric. 2018, 4, 1–9. [Google Scholar] [CrossRef]
- Murray, M. Ante-mortem and Post-mortem Signs of Anticoagulant Rodenticide Toxicosis in Birds of Prey. In Anticoagulant Rodenticides and Wildlife: Concluding Remarks; Springer: Berlin, Germany, 2018; Volume 5, pp. 109–134. [Google Scholar]
- Gómez-Ramírez, P.; Martínez-López, E.; Navas, I.; María-Mojica, P.; García-Fernández, A.J. A modification of QuEChERS method to analyse anticoagulant rodenticides using small blood samples. Rev. Toxicol. 2012, 29, 10–14. [Google Scholar]
- López-Perea, J.J.; Mateo, R. Secondary exposure to anticoagulant rodenticides and effects on predators. In Anticoagulant Rodenticides and Wildlife; Springer: Berlin, Germany, 2018; Volume 5, pp. 159–193. [Google Scholar]
- Wildlife Incident Investigation Scheme. Investigations of suspected incidents in United Kingdom. A report of the Environmental Panel of the Advisory Committee on Pesticides 2007; Department of Environmental Food and Rural Affairs: London, UK, 2007.
- Yarema, M.C.; Becker, C.E. Key concepts in postmortem drug redistribution. Clin. Toxicol. 2005, 43, 235–241. [Google Scholar] [CrossRef]
- Pélissier-Alicot, A.L.; Gaulier, J.M.; Champsaur, P.; Marquet, P. Mechanisms Underlying Postmortem Redistribution of Drugs: A Review. J. Anal. Toxicol. 2003, 27, 533–544. [Google Scholar] [CrossRef]
- Kennedy, M. Interpreting postmortem drug analysis and redistribution in determining cause of death: A review. Pathol. Lab. Med. Int. 2015, 7, 55–62. [Google Scholar] [CrossRef]
- Oates, D.W. Nebraska Game and Parks Commission. A Guide to Time of Death in Selected Wildlife Species; Nebraska Game and Parks Commission: Lincoln, NE, USA, 1984. [Google Scholar]
- Brooks, J.W. Postmortem Changes in Animal Carcasses and Estimation of the Postmortem Interval. Vet. Pathol. 2016, 53, 929–940. [Google Scholar] [CrossRef]
- Viero, A.; Montisci, M.; Pelletti, G.; Vanin, S. Crime scene and body alterations caused by arthropods: implications in death investigation. Int. J. Legal Med. 2019, 133, 307–316. [Google Scholar] [CrossRef]


| Days after Death | Internal Temperature (°C) 1 | Ambient Temperature (°C) 1 | Relative Humidity (%) 1 |
|---|---|---|---|
| Day 0 | NM | NM | NM |
| Day 1 | 32.87 | 30.17 | 58.31 |
| Day 3 | 32.74 | 33.17 | 44.63 |
| Day 4 | 28.67 | 27.97 | 60.33 |
| Day 7 | 32.81 | 30.10 | 50.65 |
| Day 15 | 33.90 | 30.09 | NM |
| ID | Sex | Group | Blood | Liver | ||||
|---|---|---|---|---|---|---|---|---|
| Concentration before Dosing (ng/g, w.w.) | Concentration before Euthanasia (ng/g, w.w.) | Decomposition Day | Liver Weight (g) | Concentration (ng/g, w.w.) | Concentration (ng/g, d.w.) | |||
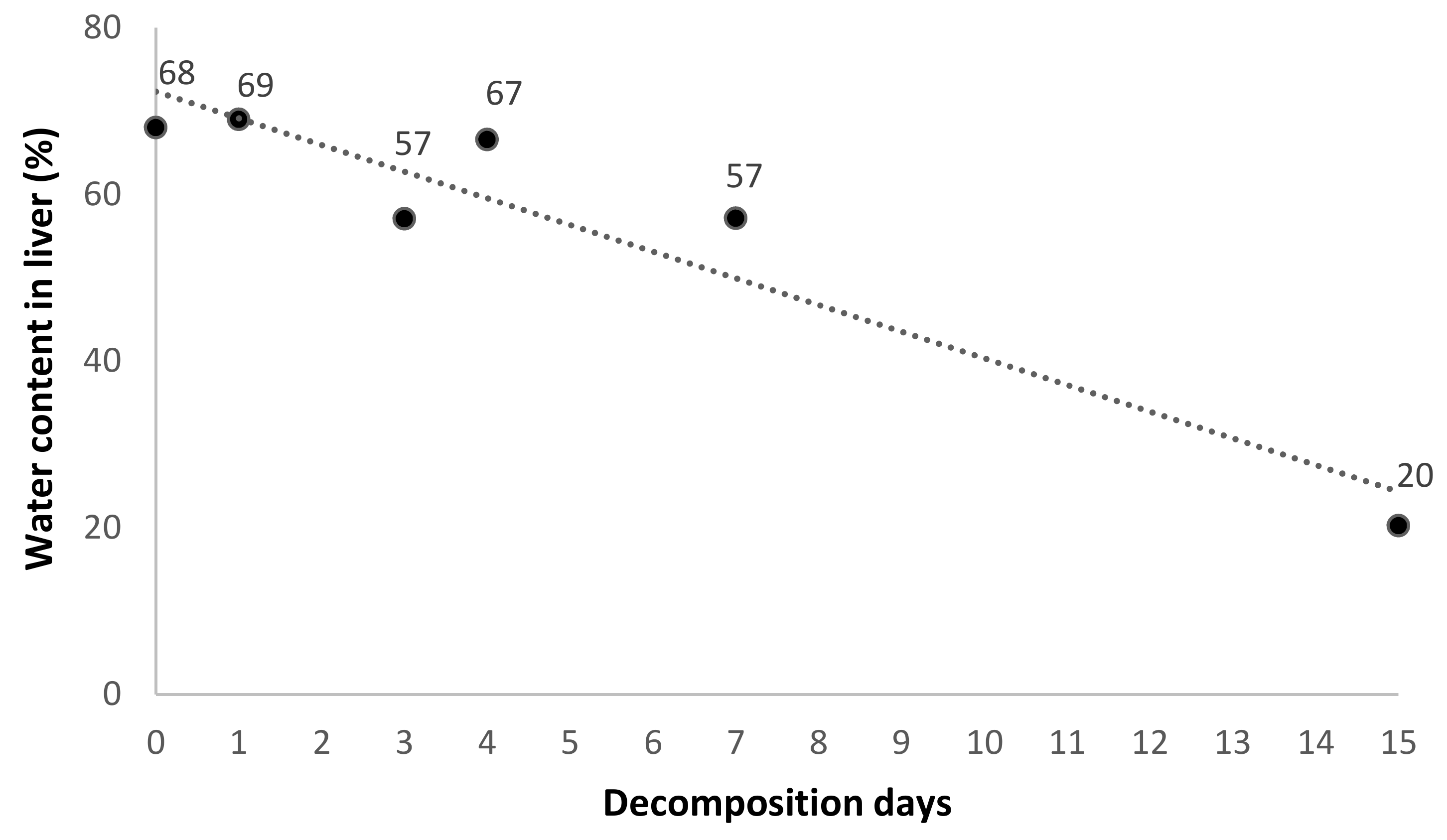
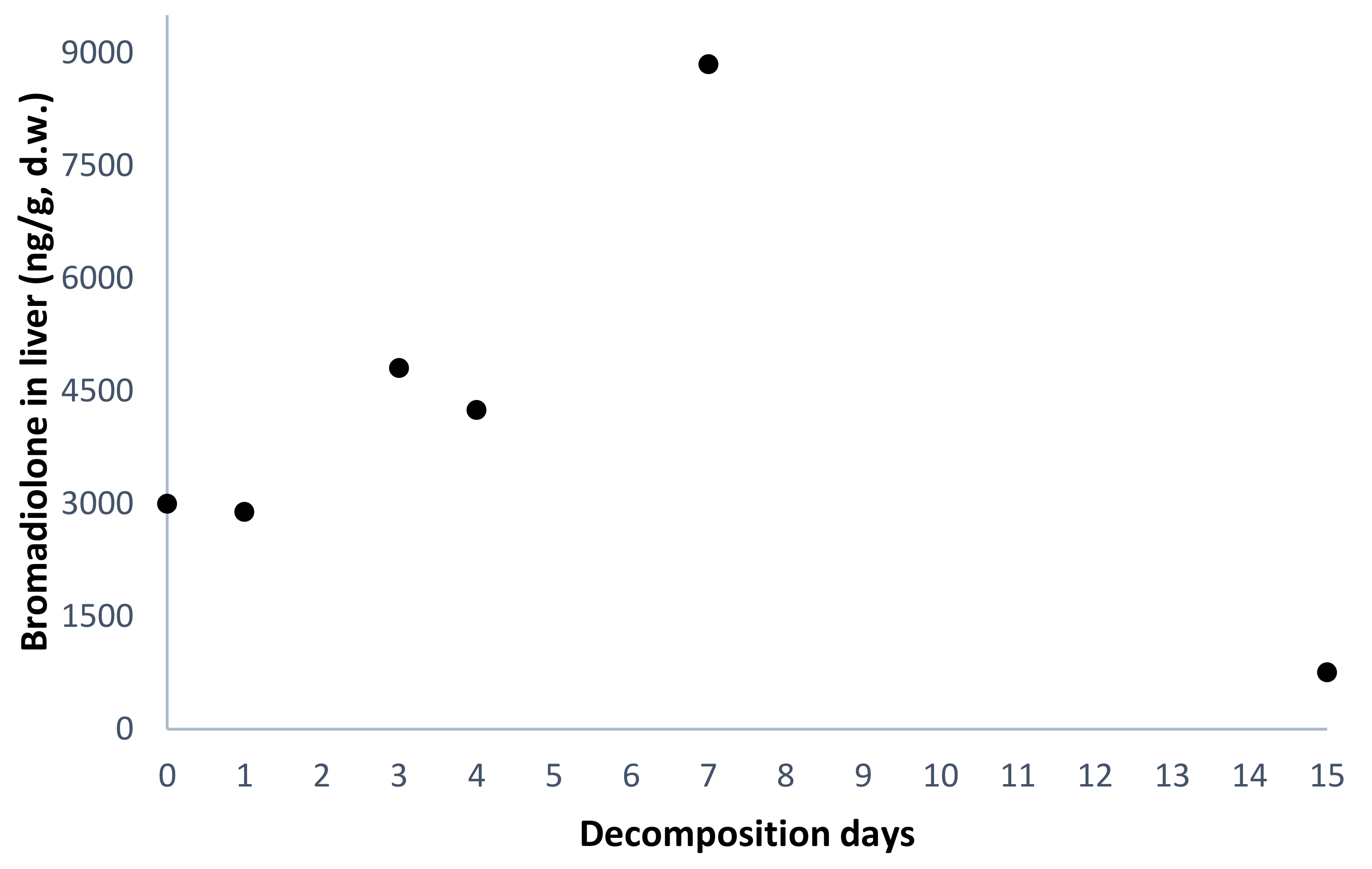
| #1 | F | Bromadiolone-dose group | nd | 47 | 0 | 5.0 | 960 | 3000 |
| #3 | M | nd | 57 | 1 | 4.1 | 896 | 2891 | |
| #5 | F | nd | 45 | 3 | 2.2 | 2062 | 4804 | |
| #7 | M | 4 | 135 | 4 | 2.2 | 1419 | 4245 | |
| #9 | M | nd | 76 | 7 | 1.0 | 3794 | 8848 | |
| #11 | M | nd | 60 | 15 | 1.0 | 603 | 756 | |
| #2 | M | Control group | nd | NA | 0 | 6.0 | 65 | 204 |
| #4 | M | nd | NA | 1 | 5.2 | 12 | 38 | |
| #6 | M | nd | NA | 3 | 2.8 | 36 | 84 | |
| #8 | M | nd | NA | 4 | 0.5 | nd | nd | |
| #10 | F | nd | NA | 7 | 0.5 | nd | nd | |
| #12 | F | nd | NA | 15 | 0.7 | 13 | 16 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valverde, I.; Espín, S.; Gómez-Ramírez, P.; Navas, I.; Sánchez-Virosta, P.; Torres-Chaparro, M.Y.; Jiménez, P.; María-Mojica, P.; García-Fernández, A.J. Temporal Persistence of Bromadiolone in Decomposing Bodies of Common Kestrel (Falco tinnunculus). Toxics 2020, 8, 98. https://doi.org/10.3390/toxics8040098
Valverde I, Espín S, Gómez-Ramírez P, Navas I, Sánchez-Virosta P, Torres-Chaparro MY, Jiménez P, María-Mojica P, García-Fernández AJ. Temporal Persistence of Bromadiolone in Decomposing Bodies of Common Kestrel (Falco tinnunculus). Toxics. 2020; 8(4):98. https://doi.org/10.3390/toxics8040098
Chicago/Turabian StyleValverde, Irene, Silvia Espín, Pilar Gómez-Ramírez, Isabel Navas, Pablo Sánchez-Virosta, María Y. Torres-Chaparro, Pedro Jiménez, Pedro María-Mojica, and Antonio J. García-Fernández. 2020. "Temporal Persistence of Bromadiolone in Decomposing Bodies of Common Kestrel (Falco tinnunculus)" Toxics 8, no. 4: 98. https://doi.org/10.3390/toxics8040098
APA StyleValverde, I., Espín, S., Gómez-Ramírez, P., Navas, I., Sánchez-Virosta, P., Torres-Chaparro, M. Y., Jiménez, P., María-Mojica, P., & García-Fernández, A. J. (2020). Temporal Persistence of Bromadiolone in Decomposing Bodies of Common Kestrel (Falco tinnunculus). Toxics, 8(4), 98. https://doi.org/10.3390/toxics8040098








