Fate of Pyriproxyfen in Soils and Plants
Abstract
1. Introduction
2. Fate in Soils
2.1. Photolysis
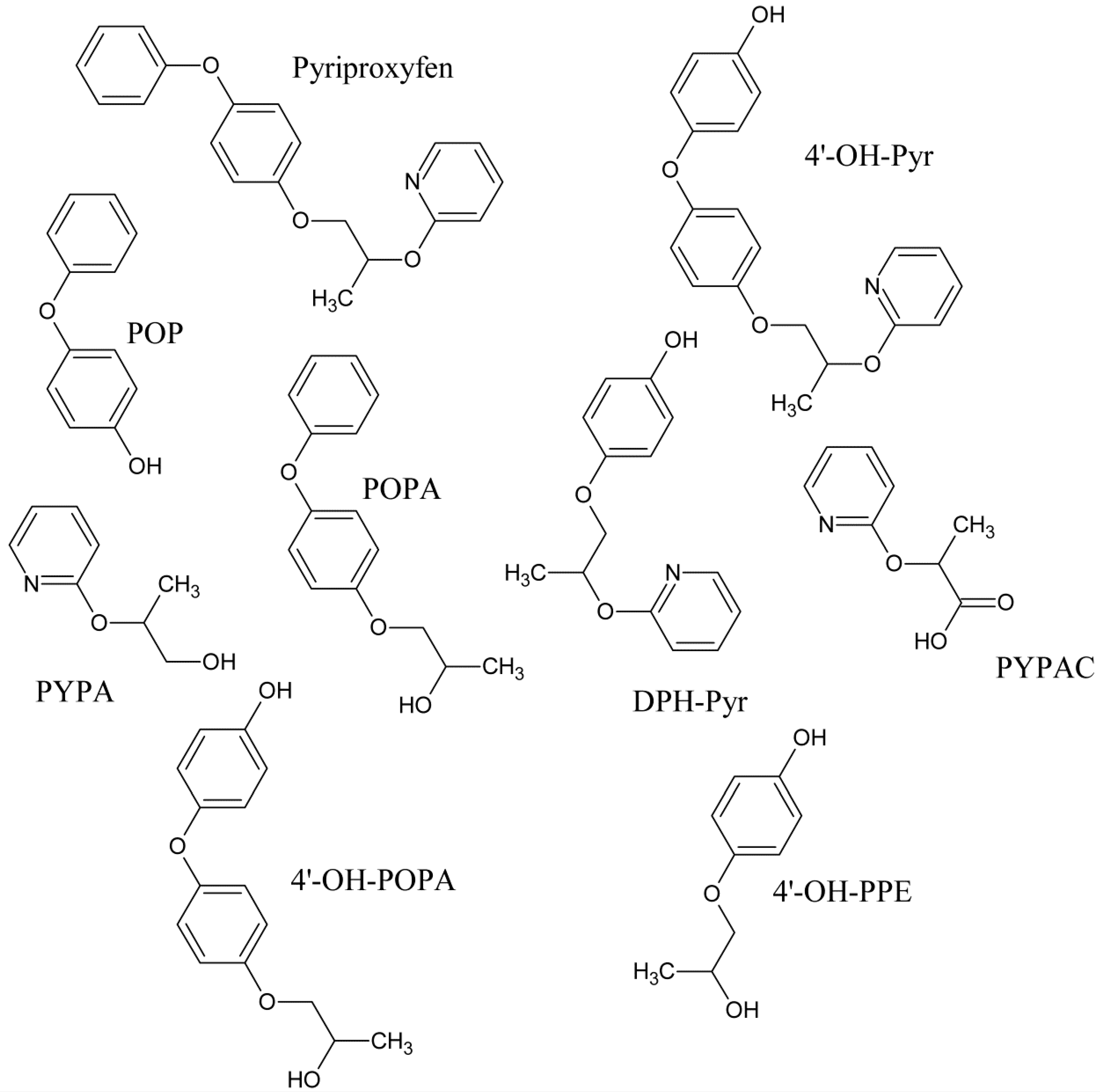
2.2. Metabolism
2.3. Mobility
3. Fate in Plants
3.1. Identification of Metabolites
3.2. Dissipation
3.3. Residues in Fruits and Vegetables
4. Concluding Remarks
Funding
Conflicts of Interest
References
- Doublet, J.; Mamy, L.; Barriuso, E. Delayed degradation in soil of foliar herbicides glyphosate and sulcotrione previously absorbed by plants: Consequences on herbicide fate and risk assessment. Chemosphere 2009, 77, 582–589. [Google Scholar] [CrossRef]
- Alekseeva, T.; Kolyagin, Y.; Sancelme, M.; Besse-Hoggan, P. Effect of soil properties on pure and formulated mesotrione adsorption onto vertisol (Limagne plane, Puy-de-Dôme, France). Chemosphere 2014, 111, 177–183. [Google Scholar] [CrossRef]
- Mamy, L.; Barriuso, E.; Gabrielle, B. Glyphosate fate in soils when arriving in plant residues. Chemosphere 2016, 154, 425–433. [Google Scholar] [CrossRef]
- Devillers, J. Endocrine Disruption Modeling; CRC Press: Boca Raton, FL, USA, 2009; ISBN 9781138111912. [Google Scholar]
- Saltzmann, K.A.; Saltzmann, K.D.; Neal, J.J.; Scharf, M.E.; Bennett, G.W. Effects of the juvenile hormone analog pyriproxyfen on German cockroach, Blattella germanica (L.), tergal gland development and production of tergal gland secretion proteins. Arch. Insect Biochem. Physiol. 2006, 63, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Biale, H.; Geden, C.J.; Chiel, E. Effects of pyriproxyfen on wild populations of the housefly, Musca domestica, and compatibility with its principal parasitoids. Pest Manag. Sci. 2017, 73, 2456–2464. [Google Scholar] [CrossRef] [PubMed]
- Ross, D.H.; Pennington, R.G.; Cruthers, L.R.; Slone, R.L. Efficacy of a permethrin and pyriproxyfen product for control of fleas, ticks and mosquitoes on dogs. Canine Pract. 1997, 22, 53–58. [Google Scholar]
- Stanneck, D.; Larsen, K.S.; Mencke, N. An evaluation of the effects of pyriproxyfen on eggs and adults of the cat flea, Ctenocephalides felis felis (Siphonaptera: Pulicidae). Irish Vet. J. 2002, 55, 383–387. [Google Scholar]
- Estrada, J.G.; Mulla, M.S. Evaluation of two new insect growth regulators against mosquitoes in the laboratory. J. Am. Mosq. Control. Assoc. 1986, 2, 57–60. [Google Scholar]
- Kawada, H.; Dohara, K.; Shinjo, G. Laboratory and field evaluation of an insect growth regulator, 4-phenoxyphenyl (RS)-2-(2-pyridyloxy)propyl ether, as a mosquito larvicide. Med. Èntomol. Zool. 1988, 39, 339–346. [Google Scholar] [CrossRef][Green Version]
- Yapabandara, A.M.G.M.; Curtis, C.F. Laboratory and field comparisons of pyriproxyfen, polystyrene beads and other larvicidal methods against malaria vectors in Sri Lanka. Acta Trop. 2002, 81, 211–223. [Google Scholar] [CrossRef]
- Alston, D.G.; Murray, M.; Reding, M.E. San Jose scale (Quadraspidiotus perniciosus). In Utah Pests Fact Sheet; ENT-153-06; Utah State University Extension and Utah Plant Pest Diagnostic Laboratory: Logan, UT, USA, 2011. [Google Scholar]
- Fisher, A., 2nd; Colman, C.; Hoffmann, C.; Fritz, B.; Rangel, J. The effects of the insect growth regulators methoxyfenozide and pyriproxyfen and the acaricide bifenazate on honey bee (hymenoptera: Apidae) forager survival. J. Econ. Èntomol. 2018, 111, 510–516. [Google Scholar] [CrossRef]
- Mahmoudvand, M.; Moharramipour, S.; Iranshahi, M. Effects of pyriproxyfen on life table indices of Plutella xylostella in multigenerations. Psyche A J. Èntomol. 2015. [Google Scholar] [CrossRef]
- Abbas, F.; Fares, A. Best management practices in citrus production. Tree Sci. Biotech. 2009, 3, 1–11. [Google Scholar]
- Franco, A.A.; Zanardi, O.; Jacob, C.; de Oliveira, M.B.R.; Yamamoto, P.T. Susceptibility of Euseius concordis (Mesostigmata: Phytoseiidae) to pesticides used in citrus production systems. Exp. Appl. Acarol. 2017, 73, 61–77. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, A.R.; Kontsedalov, S.; Denholm, I.; Ishaaya, I. Dynamics of insecticide resistance in Bemisia tabaci: A case study with the insect growth regulator pyriproxyfen. Pest Manag. Sci. 2002, 58, 1096–1100. [Google Scholar] [CrossRef] [PubMed]
- Gomes, I.N.; Vieira, K.I.C.; Gontijo, L.; Resende, H.C. Honeybee survival and flight capacity are compromised by insecticides used for controlling melon pests in Brazil. Ecotoxicology 2019, 29, 97–107. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Conclusion on the peer review of the pesticide risk assessment of the active substance pyriproxyfen. Appendix EFSA J. 2019, 17, 5732. Available online: https://doi.org/10.2903/j.efsa.2019.5732 (accessed on 3 March 2020).
- Sullivan, J.; Goh, K.S. Environmental fate and properties of pyriproxyfen. J. Pestic. Sci. 2008, 33, 339–350. [Google Scholar] [CrossRef]
- FAO. Joint Meeting Pesticide Residues, PYRIPROXYFEN. 1999. Available online: http://www.fao.org/ (accessed on 25 December 2019).
- Fenoll, J.; Ruiz, E.; Hellín, P.; Martínez, C.M.; Flores, P. Rate of loss of insecticides during soil solarization and soil biosolarization. J. Hazard. Mater. 2011, 185, 634–638. [Google Scholar] [CrossRef]
- Ulrich, E.M.; Morrison, C.N.; Goldsmith, M.R.; Foreman, W.T. Chiral pesticides: Identification, description, and environmental implications. Rev. Environ. Contam. Toxicol. 2012, 217, 1–74. [Google Scholar]
- Sekhon, B.S. Chiral pesticides. J. Pestic. Sci. 2009, 34, 1–12. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J.; Yao, Y. Enantioselective effects of chiral pesticides on their primary targets and secondary targets. Curr. Protein Pept. Sci. 2017, 18, 22–32. [Google Scholar] [CrossRef]
- de Albuquerque, N.C.P.; Carrão, D.B.; Habenschus, M.D.; de Oliveira, A.R.M. Metabolism studies of chiral pesticides: A critical review. J. Pharm. Biomed. Anal. 2018, 147, 89–109. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Wang, P.; Zhou, Z.; Liu, D. Enantioselective dissipation of pyriproxyfen in soils and sand. Chirality 2017, 29, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.S.; Yen, J.H.; Chen, W.C.; Wang, Y.S. Soil dissipation of juvenile hormone analog insecticide pyriproxyfen and its effect on the bacterial community. J. Environ. Sci. Heal. Part B 2012, 47, 13–21. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, L.; Wang, P.; Liu, D.; Zhou, Z. Enantioselective dissipation of pyriproxyfen in soil under fertilizers use. Ecotoxicol. Environ. Saf. 2019, 167, 404–411. [Google Scholar] [CrossRef]
- Liu, H.; Yi, X.; Bi, J.; Wang, P.; Liu, N.; Zhou, Z. The enantioselective environmental behavior and toxicological effects of pyriproxyfen in soil. J. Hazard. Mater. 2019, 365, 97–106. [Google Scholar] [CrossRef]
- Devillers, J.; Farret, R.; Girardin, P.; Rivière, J.L.; Soulas, G. Indicateurs Pour Evaluer les Risques Liés à l’Utilisation des Pesticides; Lavoisier: Paris, France, 2005; ISBN 2743007478. [Google Scholar]
- Pérez-Lucas, G.; Vela, N.; El Aatik, A.; Navarro, S. Environmental risk of groundwater pollution by pesticide leaching through the soil profile. In Pesticides—Use and Misuse and Their Impact in the Environment; Larramendy, M., Soloneski, S., Eds.; IntechOpen: London, UK, 2018; Available online: https://www.intechopen.com/ (accessed on 3 March 2020).
- Bintein, S.; Devillers, J. Evaluating the environmental fate of lindane in France. Chemosphere 1996, 32, 2427–2440. [Google Scholar] [CrossRef]
- Bintein, S.; Devillers, J. Evaluating the environmental fate of atrazine in France. Chemosphere 1996, 32, 2441–2456. [Google Scholar] [CrossRef]
- Bintein, S.; Devillers, J. QSAR for organic chemical sorption in soils and sediments. Chemosphere 1994, 28, 1171–1188. [Google Scholar] [CrossRef]
- Fenoll, J.; Ruiz, E.; Flores, P.; Hellín, P.; Navarro, S. Leaching potential of several insecticides and fungicides through disturbed clay-loam soil columns. Int. J. Environ. Anal. Chem. 2010, 90, 276–285. [Google Scholar] [CrossRef]
- Schaefer, C.H.; Dupras, E.F.; Mulligan, F.S., 3rd. Studies on the environmental persistence of S-31183 (pyriproxyfen): Adsorption onto organic matter and potential for leaching through soil. Ecotoxicol. Environ. Saf. 1991, 21, 207–214. [Google Scholar] [CrossRef]
- WHO. WHO Specifications and Evaluations for Public Health Pesticides: Pyriproxyfen, 4-Phenoxyphenyl (RS)-2-(2-Pyridyloxy)Propyl Ether; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Fukushima, M.; Fujisawa, T.; Katagi, T. Tomato metabolism and porphyrin-catalyzed oxidation of pyriproxyfen. J. Agric. Food Chem. 2005, 53, 5353–5358. [Google Scholar] [CrossRef] [PubMed]
- Sulaiman, A.A.; Kmel, A.; Al-Rajhi, D.H. Detection and elimination of certain insecticide residues from tomatoes and green peppers fruits. Alex. Sci. Exch. J. 2008, 29, 223–228. [Google Scholar]
- Fenoll, J.; Ruiz, E.; Hellin, P.; Lacasa, A.; Flores, P. Dissipation rates of insecticides and fungicides in peppers grown in greenhouse and under cold storage conditions. Food Chem. 2009, 113, 727–732. [Google Scholar] [CrossRef]
- Dong, M.; Wen, G.; Tang, H.; Wang, T.; Zhao, Z.; Song, W.; Wang, W.; Zhao, L. Dissipation and safety evaluation of novaluron, pyriproxyfen, thiacloprid and tolfenpyrad residues in the citrus-field ecosystem. Food Chem. 2018, 269, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Payá, P.; Oliva, J.; Cámara, M.A.; Barba, A. Dissipation of fenoxycarb and pyriproxyfen in fresh and canned peach. J. Environ. Sci. Heal. Part B 2007, 42, 767–773. [Google Scholar] [CrossRef] [PubMed]
- Payá, P.; Mulero, J.; Oliva, J.; Cámara, M.A.; Barba, A. Influence of the matrix in bioavailability of flufenoxuron, lufenuron, pyriproxyfen and fenoxycarb residues in grapes and wine. Food Chem. Toxicol. 2013, 60, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Du, P.; Wu, X.; Xu, J.; Dong, F.; Shi, Y.; Li, Y.; Liu, X.; Zheng, Y. Different residue behaviors of four pesticides in mushroom using two different application methods. Environ. Sci. Pollut. Res. 2018, 25, 8377–8387. [Google Scholar] [CrossRef]
- Du, P.; Wu, X.; He, H.; Zhang, Y.; Xu, J.; Dong, F.; Zheng, Y.; Liu, X. Evaluation of the safe use and dietary risk of beta-cypermethrin, pyriproxyfen, avermectin, diflubenzuron and chlorothalonil in button mushroom. Sci. Rep. 2017, 7, 8694. [Google Scholar] [CrossRef]
- Petersen, A.; Jensen, B.H.; Andersen, J.H.; Poulsen, M.E.; Christensen, T.; Nielsen, E. Pesticide Residues. Results from the Period 2004–2011; National Food Institute, Technical University of Denmark: Søborg, Denmark, 2013; ISBN 978-87-92763-78-5. [Google Scholar]
- Esturk, O.; Yakar, Y.; Ayhan, Z. Pesticide residue analysis in parsley, lettuce and spinach by LC-MS/MS. J. Food Sci. Technol. 2014, 51, 458–466. [Google Scholar] [CrossRef]
- Szpyrka, E.; Kurdziel, A.; Rupar, J.; Słowik-Borowiec, M. Pesticide residues in fruit and vegetable crops from the central and eastern region of Poland. Rocz. Panstw Zakl Hig. 2015, 66, 107–113. [Google Scholar] [PubMed]
- Szpyrka, E.; Kurdziel, A.; Matyaszek, A.; Podbielska, M.; Rupar, J.; Słowik-Borowiec, M. Evaluation of pesticide residues in fruits and vegetables from the region of south-eastern Poland. Food Control. 2015, 48, 137–142. [Google Scholar] [CrossRef]
- Ramadan, M.F.A.; Abdel-Hamid, M.M.A.; Altorgoman, M.M.F.; AlGaramah, H.A.; Alawi, M.; Shati, A.A.; Shweeta, H.A.; Awwad, N.S. Evaluation of pesticide residues in vegetables from the Asir Region, Saudi Arabia. Molecules 2020, 25, 205. [Google Scholar] [CrossRef]
- Abdallah, O.I.; Alamer, S.S.; Alrasheed, A.M. Monitoring pesticide residues in dates marketed in Al-Qassim, Saudi Arabia using a QuEChERS methodology and liquid chromatography-tandem mass spectrometry. Biomed. Chromatogr. 2018, 32, e4199. [Google Scholar] [CrossRef]
- Ahemad, M. Growth suppression of legumes in pyriproxyfen stressed soils: A comparative study. Emir. J. Food Agric. 2014, 26, 66–72. [Google Scholar] [CrossRef]
- Coskurn, Y.; Kilic, S.; Duran, R.E. The effects of the insecticide pyriproxyfen on germination, development and growth responses of maize seedlings. Fres. Environ. Bull. 2015, 24, 278–284. [Google Scholar]
- Ahemad, M.; Khan, M.S. Comparative toxicity of selected insecticides to pea plants and growth promotion in response to insecticide-tolerant and plant growth promoting Rhizobium leguminosarum. Crop. Prot. 2010, 29, 325–329. [Google Scholar] [CrossRef]
- Ahemad, M.; Khan, M.S. Assessment of plant growth promoting activities of rhizobacterium Pseudomonas putida under insecticide-stress. Microbiol. J. 2011, 1, 54–64. [Google Scholar]
- Meli, S.M.; Renda, A.; Nicelli, M.; Capri, E. Studies on pesticide spray drift in a Mediterranean citrus area. Agronomie 2003, 23, 667–672. [Google Scholar] [CrossRef]
- Cunha, J.P.; Chueca, P.; Garcerá, C.; Molto, E. Risk assessment of pesticide spray drift from citrus applications with air-blast sprayers in Spain. Crop. Prot. 2012, 42, 116–123. [Google Scholar] [CrossRef]
- Fogel, M.N.; Schneider, M.I.; Rimoldi, F.; Ladux, L.S.; Desneux, N.; Ronco, A.E. Toxicity assessment of four insecticides with different modes of action on pupae and adults of Eriopis connexa (Coleoptera: Coccinellidae), a relevant predator of the Neotropical Region. Environ. Sci. Pollut. Res. 2016, 23, 14918–14926. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Zamora, J.E.; Castillo, M.L.; Avilla, C. Side effects of different pesticides used in citrus on the adult stage of the parasitoid Aphytis melinus DeBach (Hymenoptera Aphelinidae) and its progeny. Span. J. Agric. Res. 2013, 11, 494–504. [Google Scholar] [CrossRef]
- Devillers, J.; Decourtye, A.; Budzinski, H.; Pham-Delègue, M.H.; Cluzeau, S.; Maurin, G. Comparative toxicity and hazards of pesticides to Apis and non-Apis bees: A chemometrical study. SAR QSAR Environ. Res. 2003, 14, 389–403. [Google Scholar] [CrossRef] [PubMed]
- Devillers, J.; Devillers, H.; Decourtye, A.; Fourrier, J.; Aupinel, P.; Fortini, D. Agent-based modeling of the long term effects of pyriproxyfen on honey bee population. In Silico Bees; Devillers, J., Ed.; CRC Press: Boca Raton, FL, USA, 2014; pp. 179–208. ISBN 9781138374706. [Google Scholar]
- Fourrier, J.; Deschamps, M.; Droin, L.; Alaux, C.; Fortini, D.; Beslay, D.; Le Conte, Y.; Devillers, J.; Aupinel, P.; Decourtye, A. Larval exposure to the juvenile hormone analog pyriproxyfen disrupts acceptance of and social behavior performance in adult honeybees. PLoS ONE 2015, 10, e0132985. [Google Scholar] [CrossRef]
| Medium | Sand % | Silt % | Clay % | OM 1 % | MO 2 % | pH | t1/2 (+)-Pyr days | t1/2 (-)-Pyr days | Rq. 3 |
|---|---|---|---|---|---|---|---|---|---|
| Sandy clay loam | 63.2 | 12 | 24.8 | 7.57 | 10 | 8.49 | 22.28 | 23.02 | |
| 63.2 | 12 | 24.8 | 7.57 | 25 | 8.49 | 16.7 | 16.12 | ||
| 63.2 | 12 | 24.8 | 7.57 | 25 | 8.49 | 14.23 | 13.40 | 1 | |
| 63.2 | 12 | 24.8 | 7.57 | 25 | 8.49 | 15.79 | 15.57 | 2 | |
| 63.2 | 12 | 24.8 | 7.57 | 50 | 8.49 | 25.29 | 25.20 | ||
| Clay loam | 37.2 | 24 | 38.8 | 36.76 | 10 | 7.77 | 15.89 | 15.71 | |
| 37.2 | 24 | 38.8 | 36.76 | 25 | 7.77 | 4.77 | 4.66 | ||
| 37.2 | 24 | 38.8 | 36.76 | 25 | 7.77 | 4.43 | 4.43 | 1 | |
| 37.2 | 24 | 38.8 | 36.76 | 25 | 7.77 | 7.80 | 7.67 | 2 | |
| 37.2 | 24 | 38.8 | 36.76 | 50 | 7.77 | 27.07 | 26.45 | ||
| Loam | 32 | 44 | 24 | 20.48 | 25 | 6.55 | 7.01 | 7.10 | |
| 32 | 44 | 24 | 20.48 | 25 | 6.55 | 6.48 | 6.42 | 1 | |
| 32 | 44 | 24 | 20.48 | 25 | 6.55 | 31.08 | 29.87 | 2 | |
| Sandy loam | 73.2 | 16 | 10.8 | 28.68 | 25 | 8.17 | 4.82 | 5.62 | |
| 73.2 | 16 | 10.8 | 28.68 | 25 | 8.17 | 4.38 | 4.98 | 1 | |
| 73.2 | 16 | 10.8 | 28.68 | 25 | 8.17 | 5.14 | 5.05 | 2 | |
| 73.2 | 16 | 10.8 | 28.68 | 25 | 8.17 | 11.99 | 14.68 | 3 | |
| Clay loam | 26 | 38 | 36 | 6.05 | 25 | 5.18 | 9.96 | 9.64 | |
| Sand | 100 | 0 | 0 | 6.02 | 25 | 8.33 | 25.86 | 40.29 |
| Soil Type | pH | OM 1 | SP1 2 | SP2 | SP3 | t1/2 (-)-Pyr | t1/2 (+)-Pyr | t1/2 (-)-4′-OH-Pyr | t1/2 (+)-4′-OH-Pyr |
|---|---|---|---|---|---|---|---|---|---|
| Loamy sand | 8.10 | 12.4 | 845 | 85 | 70 | 2.11 | 2.11 | 3.29 | 2.80 |
| Clay #1 | 8.05 | 47.9 | 281 | 130 | 589 | 8.39 | 6.14 | 5.11 | 6.56 |
| Sandy clay loam #1 | 8 | 17.8 | 475 | 247 | 278 | 4.49 | 3.67 | 7.38 | 3.90 |
| Clay #2 | 7.89 | 8.69 | 309 | 253 | 438 | 9.61 | 9.69 | 8.76 | 9.97 |
| Sandy clay loam #2 | 7.94 | 8.31 | 642 | 155 | 203 | 7.31 | 8.75 | 11.21 | 13.30 |
| Medium | Sand % | Silt % | Clay % | OM 1 % | pH | CEC 2 | Koc |
|---|---|---|---|---|---|---|---|
| Loam | 56 | 30 | 15 | 8.2 | 7.1 | 32 | 1.30 × 104 |
| Clay loam | 55 | 26 | 19 | 1.9 | 7 | 6.3 | 5.80 × 104 |
| 21 | 47 | 32 | 5 | 7 | 21 | 1.10 × 104 | |
| Sandy loam | 72 | 18 | 11 | 0.9 | 7.2 | 2.8 | 2.70 × 104 |
| 60 | 25 | 15 | 1.65 | 8 | 9.7 | 1.26 × 104 | |
| Silt loam | 29 | 58 | 13 | 1.1 | 7 | 13 | 2.69 × 104 |
| Silty clay loam | 7 | 53 | 49 | 1.4 | 7.8 | 27 | 3.42 × 104 |
| Sand | 97 | 1 | 2 | 0.3 | 5.4 | 1.1 | 1.16 × 104 |
| Medium | Sand % | Silt % | Clay % | OM 1 % | pH | CEC 2 | Koc 4′-OH-Pyr | Koc PYPAC |
|---|---|---|---|---|---|---|---|---|
| Sand | 92 | 3.6 | 4.4 | 0.22 | 6 | 0.82 | 4250 | 85 |
| Sandy loam | 75 | 18 | 7.2 | 0.96 | 6.9 | 6.6 | 3810 | 21 |
| Silt loam | 35 | 54 | 11 | 1.8 | 6.9 | 8.9 | 3060 | 32 |
| Clay loam | 33 | 28 | 39 | 2.1 | 7.9 | 15.8 | 920 | 9 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devillers, J. Fate of Pyriproxyfen in Soils and Plants. Toxics 2020, 8, 20. https://doi.org/10.3390/toxics8010020
Devillers J. Fate of Pyriproxyfen in Soils and Plants. Toxics. 2020; 8(1):20. https://doi.org/10.3390/toxics8010020
Chicago/Turabian StyleDevillers, James. 2020. "Fate of Pyriproxyfen in Soils and Plants" Toxics 8, no. 1: 20. https://doi.org/10.3390/toxics8010020
APA StyleDevillers, J. (2020). Fate of Pyriproxyfen in Soils and Plants. Toxics, 8(1), 20. https://doi.org/10.3390/toxics8010020




