Mercury in Soil and Forage Plants from Artisanal and Small-Scale Gold Mining in the Bombana Area, Indonesia
Abstract
1. Introduction
2. Materials and Methods
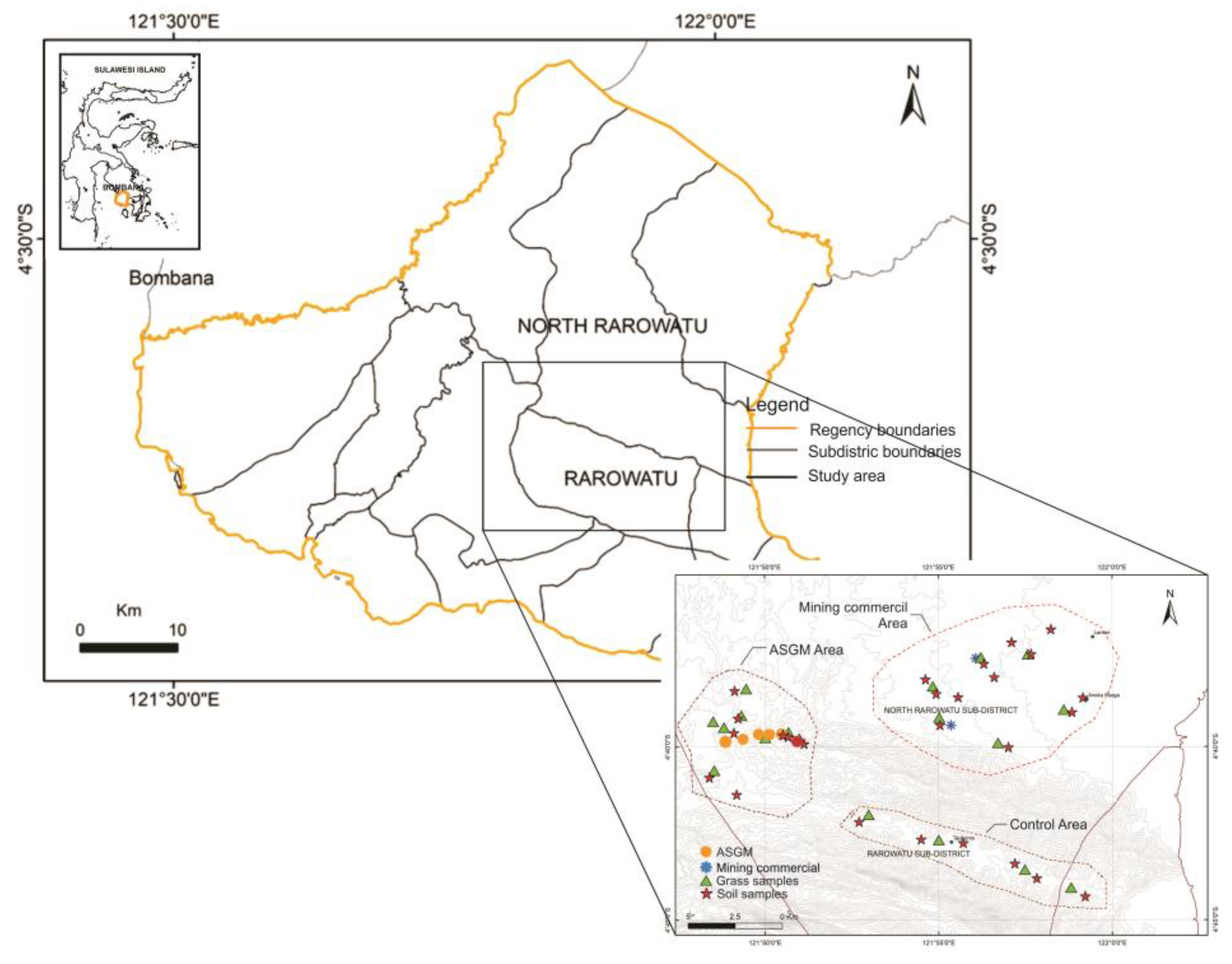
2.1. Study Area
2.2. Sample Collection
2.2.1. Soils
2.2.2. Forage Plants
2.3. Sample Preparation
2.3.1. Soil Sample
2.3.2. Forage-Plant Samples
2.4. Sample Analysis
2.5. Statistical Analysis
3. Results
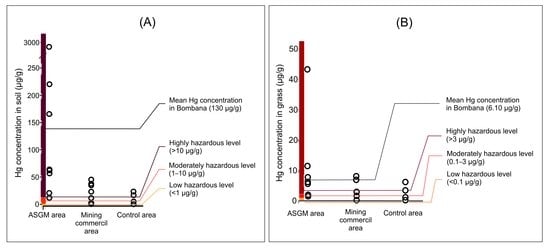
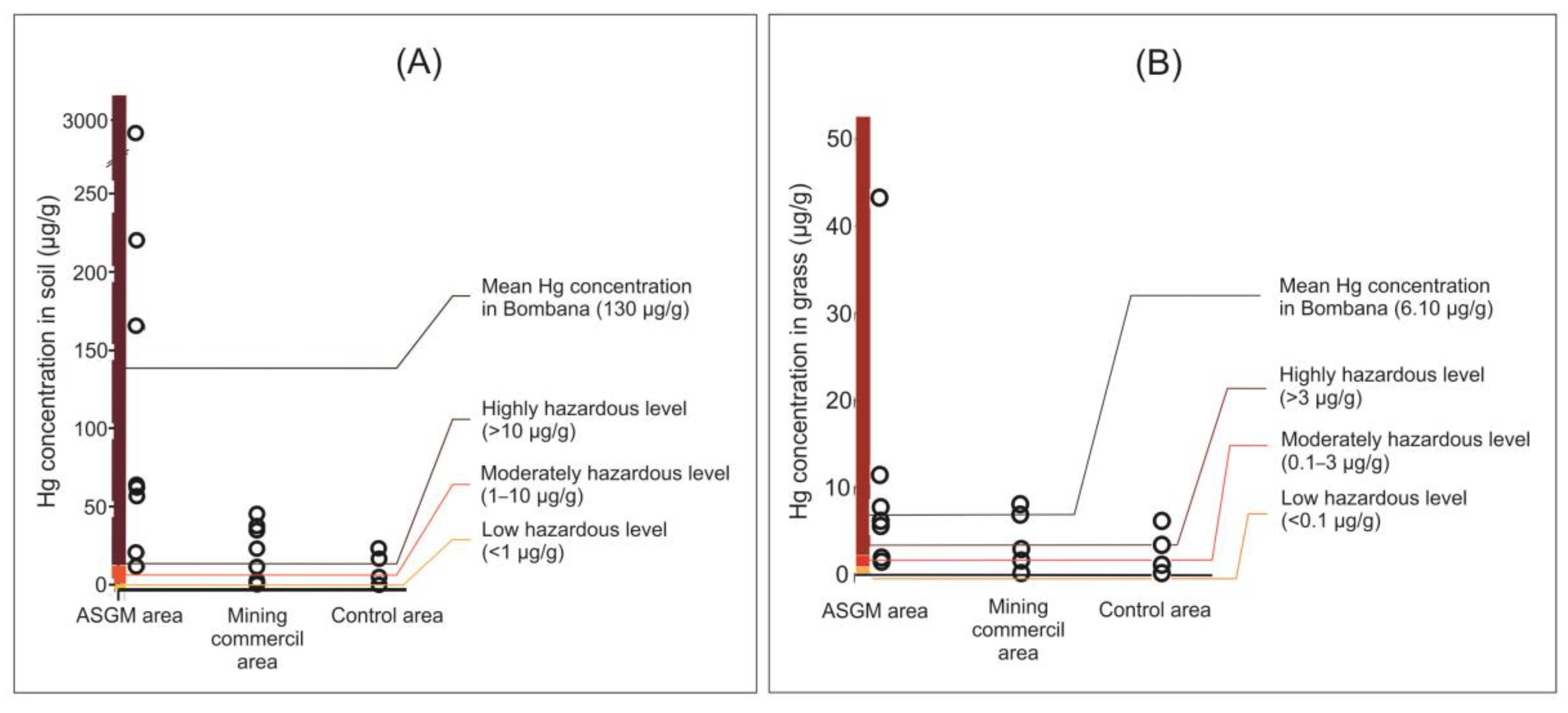
3.1. Hg Distribution in Soils
3.2. Forage Plants Hg Contamination
4. Discussion
4.1. Source of Hg Pollution in the Terrestrial Environment
4.2. Hg in Soils
4.3. Hg in Forage Plants
4.4. Effects of Hg-Polluted Soil on Forage-Plant Growth
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Veiga, M.; Morais, H. Backgrounder: Artisanal and Small–Scale Mining (ASM) in Developing Countries; CIRDI: Vancouver, BC, Canada, 2018; pp. 1–3. Available online: https://static1.squarespace.com/static/5bb24d3c9b8fe8421e87bbb6/t/5c2a832988251b499681df64/1546289962450/CIRDI-ASM-Backgrounder_2015Apr10.pdf (accessed on 24 December 2019).
- BaliFokus. Mercury Trade and Supply in Indonesia; Denpasar, BaliFokus Foundation: Denpasar, Indonesia, 2017. [Google Scholar]
- IPEN. A Toxics-Free Future. Available online: http://www.ipen.org/projects/international-mercury-treaty-enabling-activities-program-imeap (accessed on 11 June 2018).
- IIED. Global Trends in Artisanal and Small-Scale Mining (ASM): A Review of Key Numbers and Issues; IIED: London, UK, 2018; Available online: https://www.iisd.org/sites/default/files/publications/igf-asm-global-trends.pdf (accessed on 24 December 2019).
- US-EPA. Mercury Study Report to Congress; III: Fate; US-EPA: Washington, DC, USA, 1997; p. 376. [Google Scholar]
- Basri; Sakakibara, M.; Sera, K. Current Mercury Exposure from Artisanal and Small-Scale Gold Mining in Bombana, Southeast Sulawesi, Indonesia—Future Significant Health Risks. Toxics 2017, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Prasad, M.N.V.; Sajwan, K.S.; Naidu, R. Trace Elements in the Environment: Biogeochemistry, Biotechnology, and Bioremediation; CRC Press: Boca Raton, FL, USA, 2006; ISBN 9781420032048. [Google Scholar]
- Finkelman, R.B. Medical Geology; Elsevier Academic Press: California, USA, 2001; Volume 46, ISBN 0126363412. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1310961/ (accessed on 24 December 2019).
- Esdaile, L.J.; Chalker, J.M. The Mercury Problem in Artisanal and Small-Scale Gold Mining. Chemistry 2018, 24, 6905–6916. [Google Scholar] [CrossRef] [PubMed]
- Persaud, A.; Telmer, K. Developing Baseline Estimates of Mercury Use in Artisanal and Small-Scale Gold Mining Communities: A Practical Guide (Version 1.0); ASGM: Artisanal Gold Council: Victoria, TX, USA, 2015; Available online: https://www.unenvironment.org/resources/report/developing-baseline-estimates-mercury-use-artisanal-and-small-scale-gold-mining (accessed on 24 December 2019)ISBN 9780993945946.
- Li, R.; Wu, H.; Ding, J.; Fu, W.; Gan, L.; Li, Y. Mercury pollution in vegetables, grains and soils from areas surrounding coal-fired power plants. Sci. Rep. 2017, 7, 46545. [Google Scholar] [CrossRef] [PubMed]
- Gautam, P.K.; Gautam, R.K.; Banerjee, S.; Chattopadhyaya, M.C.; Pandey, J.D. Heavy metals in the environment: Fate, transport, toxicity and remediation technologies. Heavy Met. Sources Toxic Remediat. Technol. 2016, 101–130. [Google Scholar] [CrossRef]
- Petrus, A.K.; Rutner, C.; Liu, S.; Wang, Y.; Wiatrowski, H.A. Mercury reduction and methyl mercury degradation by the soil bacterium Xanthobacter autotrophicus Py2. Appl. Environ. Microbiol. 2015, 81, 7833–7838. [Google Scholar] [CrossRef]
- Sandrin, T.R.; Maier, R.M. Impact of metals on the biodegradation of organic pollutants. Environ. Health Perspect. 2003, 111, 1093–1101. [Google Scholar] [CrossRef]
- Jaishankar, M.; Tseten, T.; Anbalagan, N.; Mathew, B.B.; Beeregowda, K.N. Toxicity, mechanism and health effects of some heavy metals. Interdiscip. Toxicol. 2014, 7, 60–72. [Google Scholar] [CrossRef]
- Hindersah, R.; Risamasu, R.; Kalay, A.M.; Dewi, T.; Makatita, I. Mercury contamination in soil, tailing and plants on agricultural fields near closed gold mine in Buru Island, Maluku. J. Degrad. Min. Lands Manag. 2018, 5, 1027–1034. [Google Scholar] [CrossRef][Green Version]
- Mahmud, M.; Lihawa, F.; Saleh, Y.; Desei, F.; Banteng, B. Study of Mercury Concentration in Plants in Traditional Buladu Gold Mining; IOP Publishing: Bristol, UK, 2019; Volume 314, Available online: https://iopscience.iop.org/article/10.1088/1755-1315/314/1/012018 (accessed on 24 December 2019).
- Sari, M.M.; Inoue, T.; Matsumoto, Y.; Yokota, K.; Isrun, I. Assessing a Mercury Affected Area from Small-scale Gold Mining in Poboya, Central Sulawesi, Indonesia. Environ. Ecol. Res. 2016, 4, 223–230. [Google Scholar] [CrossRef][Green Version]
- Sakakibara, M.; Sera, K.; Kurniawan, I.A. Mercury Contamination of Cattle in Artisanal and Small-Scale Gold Mining in Bombana, Southeast Sulawesi, Indonesia. Geosciences 2017, 7, 133. [Google Scholar]
- US-EPA. Operating Procedure: Soil Sampling; US-EPA: Washington, DC, USA, 2014. Available online: https://www.epa.gov/sites/production/files/2015-06/documents/Soil-Sampling.pdf (accessed on 24 December 2019).
- TNAU. Soil Sampling Procedure. Available online: http://agritech.tnau.ac.in/agriculture/agri_soil_sampling.html (accessed on 24 August 2019).
- Boudreault, J.-P.; Dubé, J.-S.; Sona, M.; Hardy, É. Analysis of procedures for sampling contaminated soil using Gy’s Sampling Theory and Practice. Sci. Total Environ. 2012, 425, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Funk, V. The importance of herbaria. Plant Sci. Bull. 2003, 49, 94–95. [Google Scholar]
- O’Kelly, B.C. Oven-Drying Characteristics of Soils of Different Origins: Drying Technology; Talylor Francis: Abingdon, UK, 2007; Volume 23, Available online: https://www.tandfonline.com/doi/abs/10.1081/DRT-200059149 (accessed on 24 December 2019).
- Moritsuka, N.; Izawa, G.; Katsura, K.; Matsui, N. Simple method for measuring soil sand content by nylon mesh sieving. Soil Sci. Plant Nutr. 2015, 61, 501–505. [Google Scholar] [CrossRef][Green Version]
- Li, J.; Hitch, M. Ultra-fine grinding and mechanical activation of mine waste rock using a planetary mill for mineral carbonation. Int. J. Miner. Process. 2017, 158, 18–26. [Google Scholar] [CrossRef]
- Damas, K.; Kiapranis, R. Laboratory Manual for Collecting and Processing Plant Species Laboratory Manual for Collecting and Processing of Plant Samples; Papua New Guinea Forest Research Institute: Lae, Papua New Guinea, 2017; Available online: http://www.fao.org/fileadmin/templates/mountain_partnership/doc/Biodiversity_and_REDD_/Manuals/LABORATORY_MANUAL_FOR_COLLECTING_AND_PROCESSING_PLANT_SPECIES.pdf (accessed on 24 December 2019).
- Miodrag. Sample Preparation Techniques for Soil, Plant, and Animal Samples; Springer: Berlin/Heidelberg, Germany, 2016; Available online: https://www.springer.com/gp/book/9781493931842 (accessed on 24 December 2019).
- NIES. National Institute for Environmental Studies, Japan. Available online: http://www.nies.go.jp/index-e.html (accessed on 24 August 2019).
- Gonsior, B. Particle Induced X-Ray Emission (PIXE). Technol. Instrum. Anal. Chem. 1988, 8, 123–179. [Google Scholar]
- Sakakibara, M. Ratnawati Economic features of the artisanal and small-scale gold mining industry in Bombana, Southeast Sulawesi, Indonesia. IOP Conf. Ser. Earth Environ. Sci. 2017, 71, 12016. [Google Scholar]
- Driscoll, C.T.; Mason, R.P.; Chan, H.M.; Jacob, D.J.; Pirrone, N. Mercury as a global pollutant: Sources, pathways, and effects. Environ. Sci. Technol. 2013, 47, 4967–4983. [Google Scholar] [CrossRef]
- Wang, D.; Shi, X.; Wei, S. Accumulation and transformation of atmospheric mercury in soil. Sci. Total Environ. 2003, 304, 209–214. [Google Scholar] [CrossRef]
- European Commission (EC). Management of Tailings and Waste-Rock in Mining Activities; European Commission (EC): Brussels, Belgium, 2009; Available online: http://www.mercury.org.cn/kzjs/batbep/201109/P020121211404439274558.pdf. (accessed on 24 December 2019).
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Molecular. Clin. Environ. Toxicol. 2012, 101, 1–30. [Google Scholar]
- WHO. Mercury and Health. Available online: https://www.who.int/en/news-room/fact-sheets/detail/mercury-and-health (accessed on 28 January 2020).
- Petersen, G.; Munthe, J.; Pleijel, K.; Bloxam, R.; Kumar, A.V. A comprehensive Eulerian modeling framework for airborne mercury species: Development and testing of the Tropospheric Chemistry Module (TCM). Atmos. Environ. 1998, 32, 829–843. [Google Scholar] [CrossRef]
- Slemr, F.; Scheel, H.E. Trends in atmospheric mercury concentrations at the summit of the Wank mountain, Southern Germany. Atmos. Environ. 1998, 32, 845–853. [Google Scholar] [CrossRef]
- Vries, W. De Critical limits for cadmium, lead and mercury related to ecotoxicological effects on soil organisms, aquatic organisms, plants, animals and humans. In Proceedings of the Expert Meeting on Critical Limits for Heavy Metals and Methods for Their Application, Berlin, Germany, 2–4 December 2002; Available online: https://edepot.wur.nl/52354 (accessed on 24 December 2019).
- Manahan, S.E. Fundamentals of Environmental and Toxicological Chemistry; Taylor & Francis: Abingdon, UK, 2013; Available online: https://www.taylorfrancis.com/books/9780429097744 (accessed on 24 December 2019)ISBN 9781466553170.
- Tangahu, B.V.; Sheikh Abdullah, S.R.; Basri, H.; Idris, M.; Anuar, N.; Mukhlisin, M. A review on heavy metals (As, Pb, and Hg) uptake by plants through phytoremediation. Int. J. Chem. Eng. 2011, 2011, 939161. [Google Scholar] [CrossRef]
- Chibuike, G.U.; Obiora, S.C. Heavy metal polluted soils: Effect on plants and bioremediation methods. Appl. Environ. Soil Sci. 2014, 2014. [Google Scholar] [CrossRef]
- Azevedo, R.; Rodriguez, E. Phytotoxicity of Mercury in Plants: A Review. J. Bot. 2012, 2012, 848614. [Google Scholar] [CrossRef]
- Chandra Shekar, C.H.; Sammaiah, D.; Shasthree, T.; Jaganmohan Reddy, K. Effect of mercury on tomato growth and yield attributes. Int. J. Pharma Bio Sci. 2011, 2, 358–364. [Google Scholar]
- Du, X.; Zhu, Y.G.; Liu, W.J.; Zhao, X.S. Uptake of mercury (Hg) by seedlings of rice (Oryza sativa L.) grown in solution culture and interactions with arsenate uptake. Environ. Exp. Bot. 2005, 54, 1–7. [Google Scholar] [CrossRef]
- Kibria, M. Effects of mercury on some growth parameters of rice (Oryza sativa L.). Soil Environ. 2008, 27, 23–28. [Google Scholar]
| Hg Concentration (µg/g) | Sampling Group | ||
|---|---|---|---|
| Control Area (n = 6) | Mining Commercil Area (n = 12) | ASGM Area (n = 8) | |
| Minimum | 0 | 0 | 12.0 |
| Median | 2.40 | 2.40 | 63.0 |
| Mean ± SD | 7.40 ± 9.90 | 13.0 ± 17.0 | 390 ± 860 |
| Maximum | 23.0 | 45.0 | 2500 |
| Independent t-test (each contaminated area vs control area | (p = 0.30)** | (p = 0.47) ** | |
| One-Way ANOVA + Post hoc Tamhane’s | (p = 0.195) ** | ||
| Hg Concentration (µg/g) | Sampling Group | ||
|---|---|---|---|
| Control Area (n = 4) | Mining Commercil Area (n = 6) | ASGM Area (n = 8) | |
| Minimum | 0 | 0 | 1.50 |
| Median | 2.20 | 2.20 | 5.90 |
| Mean ± SD | 2.70 ± 2.80 | 3.20 ± 3.50 | 9.90 ± 14 |
| Maximum | 23.0 | 45.0 | 2500 |
| Independent t-test (each contaminant area vs control area | (p = 0.33)** | (p = 0.79) ** | |
| One-Way ANOVA + Posthoc Bonferroni | (p = 0.354) ** | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basri; Sakakibara, M.; Sera, K. Mercury in Soil and Forage Plants from Artisanal and Small-Scale Gold Mining in the Bombana Area, Indonesia. Toxics 2020, 8, 15. https://doi.org/10.3390/toxics8010015
Basri, Sakakibara M, Sera K. Mercury in Soil and Forage Plants from Artisanal and Small-Scale Gold Mining in the Bombana Area, Indonesia. Toxics. 2020; 8(1):15. https://doi.org/10.3390/toxics8010015
Chicago/Turabian StyleBasri, Masayuki Sakakibara, and Koichiro Sera. 2020. "Mercury in Soil and Forage Plants from Artisanal and Small-Scale Gold Mining in the Bombana Area, Indonesia" Toxics 8, no. 1: 15. https://doi.org/10.3390/toxics8010015
APA StyleBasri, Sakakibara, M., & Sera, K. (2020). Mercury in Soil and Forage Plants from Artisanal and Small-Scale Gold Mining in the Bombana Area, Indonesia. Toxics, 8(1), 15. https://doi.org/10.3390/toxics8010015