Aristolochic Acids: Newly Identified Exposure Pathways of this Class of Environmental and Food-Borne Contaminants and its Potential Link to Chronic Kidney Diseases
Abstract
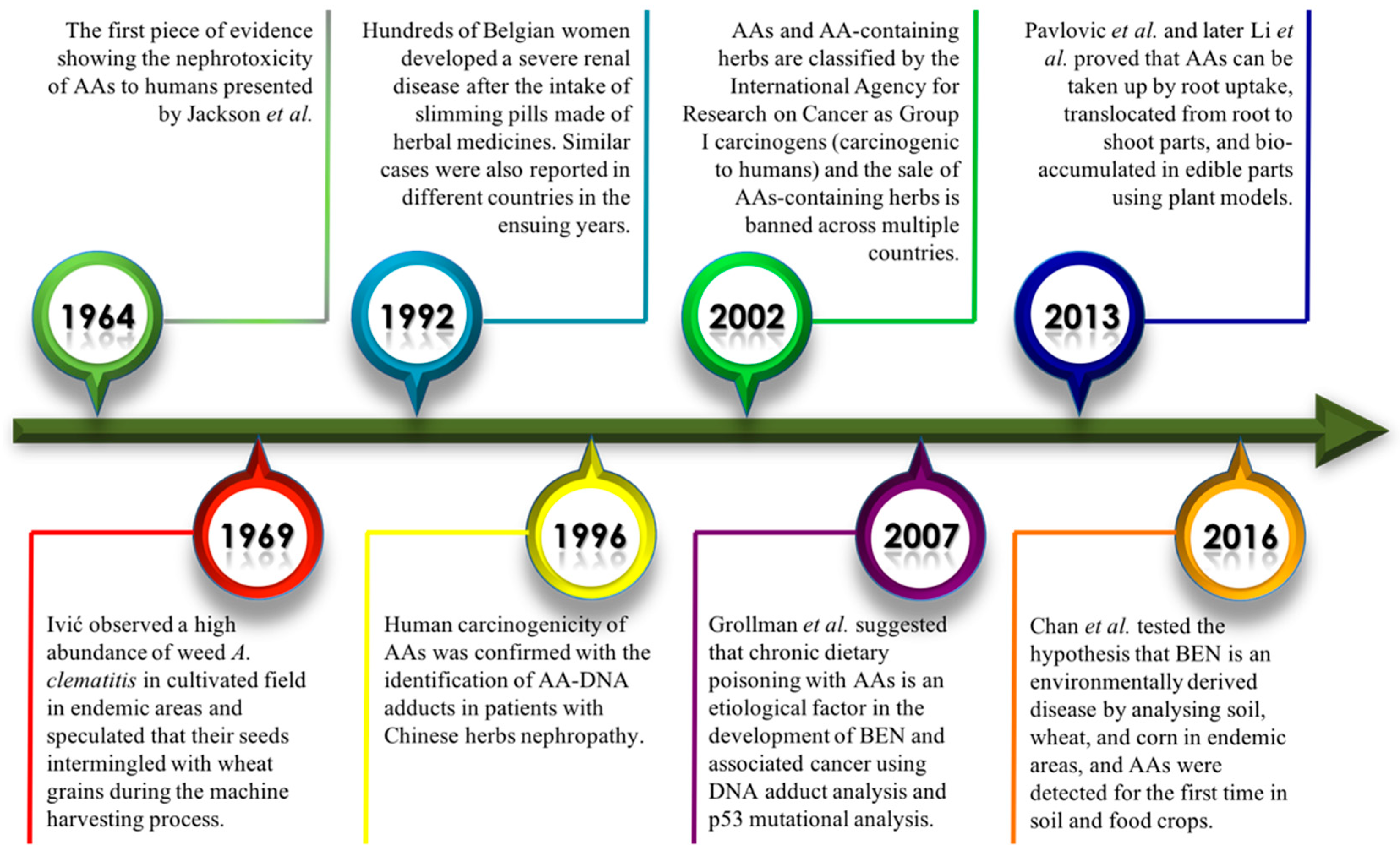
1. Introduction
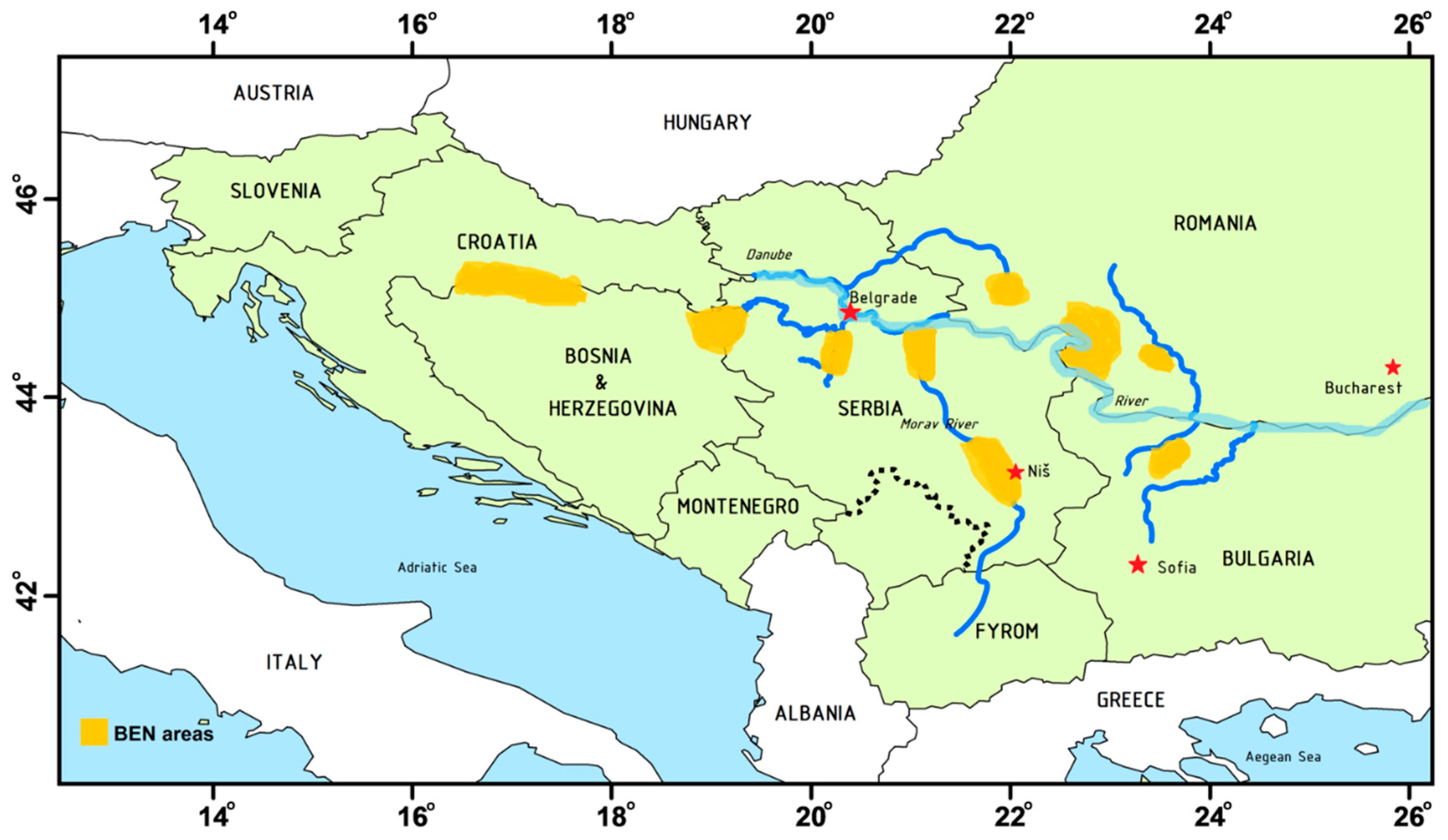
2. The Occurrence of Aristolochic Acids in the Environment
3. Uptake and Bioaccumulation in Food Crops
4. Risks to Human Health
5. Possible Metabolites in the Environment
6. Overview of Potential Biomarkers for AA intoxication
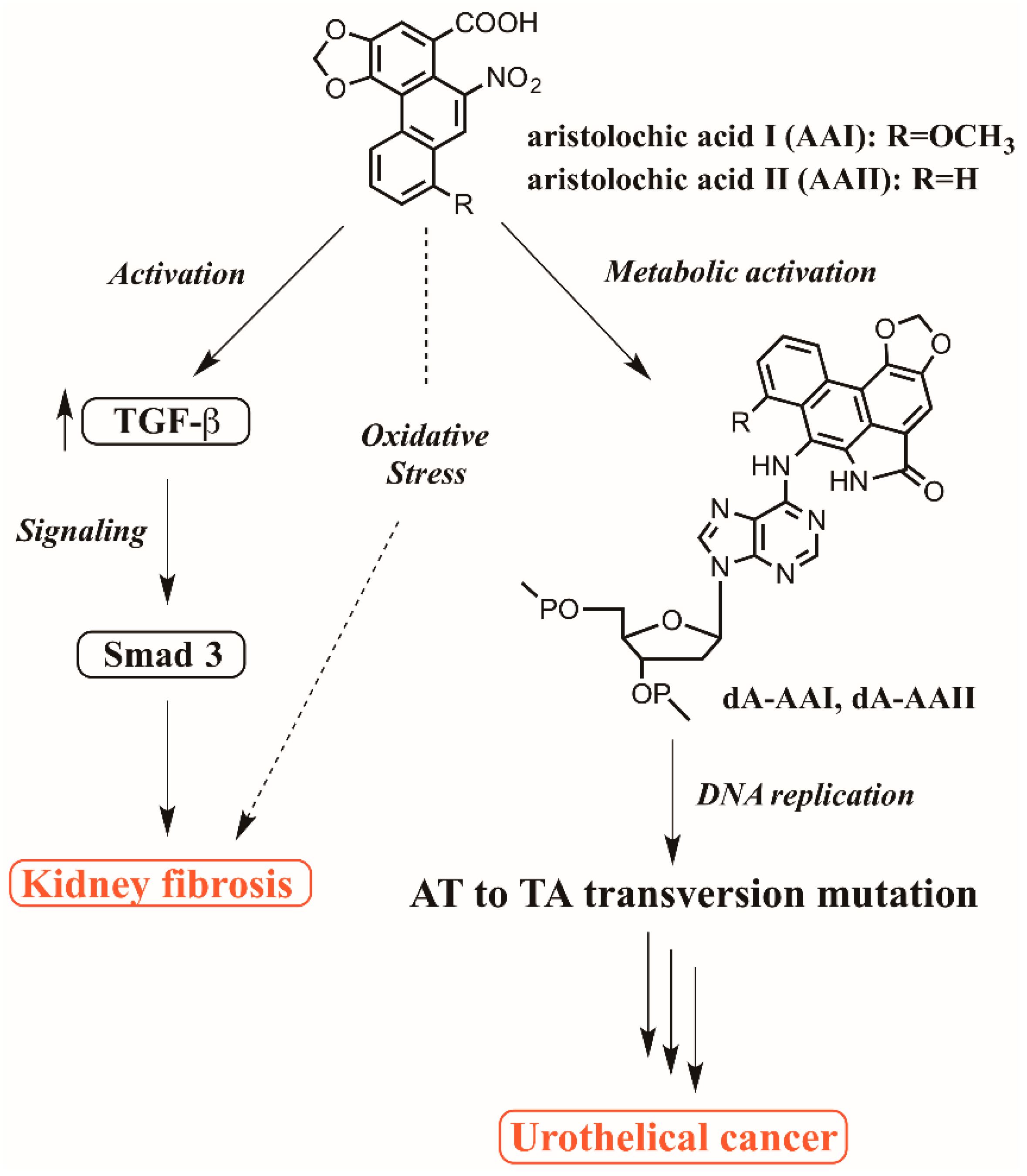
7. Postulated Mechanism for AA-associated Kidney Fibrosis
8. Challenges and Future Perspectives
Funding
Acknowledgments
Conflicts of Interest
References
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans; World Health Organization; International Agency for Research on Cancer. Some Traditional Herbal Medicines, Some Mycotoxins, Naphthalene and Styrene; World Health Organization: Lyon, France, 2002; pp. 1–556. [Google Scholar]
- Jackson, L.; Kofman, S.; Weiss, A.; Brodovsky, H. Aristolochic acid (NSC-50413): Phase I clinical study. Cancer Chemother. Rep. 1964, 42, 65. [Google Scholar]
- Mengs, U.; Lang, W.; Poch, J.A. The carcinogenic action of aristolochic acid in rats. Arch. Toxicol. 1982, 51, 107–119. [Google Scholar] [CrossRef]
- Mengs, U. Acute toxicity of aristolochic acid in rodents. Arch. Toxicol. 1987, 59, 328–331. [Google Scholar] [CrossRef]
- Schmeiser, H.H.; Bieler, C.A.; Wiessler, M.; van Ypersele de Strihou, C.; Cosyns, J.P. Detection of DNA adducts formed by aristolochic acid in renal tissue from patients with Chinese herbs nephropathy. Cancer Res. 1996, 56, 2025–2028. [Google Scholar] [PubMed]
- Cosyns, J.P.; Jadoul, M.; Squifflet, J.P.; De Plaen, J.F.; Ferluga, D.; Van Ypersele De Strihou, C. Chinese herbs nephropathy: A clue to balkan endemic nephropathy? Kidney Int. 1994, 45, 1680–1688. [Google Scholar] [CrossRef] [PubMed]
- Kessler, D.A. Cancer and herbs. N. Engl. J. Med. 2000, 342, 1742–1743. [Google Scholar] [CrossRef]
- Vanherweghem, J.-L.; Depierreux, M.F.; Tielemans, C.; Abramowicz, D.; Dratwa, M.; Jadoul, M.; Richard, C.; Vandervelde, D.; Verbeelen, D.; Vanhaelen-Fastre, R.; et al. Rapidly progressive interstitial renal fibrosis in young women: Association with slimming regimen including Chinese herbs. Lancet 1993, 341, 387–391. [Google Scholar] [CrossRef]
- Vanhaelen, M.; Vanhaelen-Fastre, R.; But, P.; Vanherweghem, J.L. Identification of aristolochic acid in Chinese herbs. Lancet 1994, 343, 174. [Google Scholar] [CrossRef]
- Nortier, J.L.; Martinez, M.C.; Schmeiser, H.H.; Arlt, V.M.; Bieler, C.A.; Petein, M.; Depierreux, M.F.; De Pauw, L.; Abramowicz, D.; Vereerstraeten, P.; et al. Urothelial carcinoma associated with the use of a Chinese herb (Aristolochia fangchi). N. Engl. J. Med. 2000, 342, 1686–1692. [Google Scholar] [CrossRef]
- Jadot, I.; Declèves, A.-E.; Nortier, J.; Caron, N. An integrated view of aristolochic acid nephropathy: Update of the literature. Int. J. Mol. Sci. 2017, 18, 297. [Google Scholar] [CrossRef] [PubMed]
- Tangtong, C. Environmental Processes Controlling the Fate and Transport of Aristolochic Acid in Agricultural Soil and Copper in Contaminated Lake Sediment; Michigan State University: East Lansing, MI, USA, 2014. [Google Scholar]
- Tatu, C.A.; Orem, W.H.; Finkelman, R.B.; Feder, G.L. The etiology of balkan endemic nephropathy: Still more questions than answers. Environ. Health Perspect. 1998, 106, 689–700. [Google Scholar] [CrossRef] [PubMed]
- Grollman, A.P.; Shibutani, S.; Moriya, M.; Miller, F.; Wu, L.; Moll, U.; Suzuki, N.; Fernandes, A.; Rosenquist, T.; Medverec, Z.; et al. Aristolochic acid and the etiology of endemic (balkan) nephropathy. Proc. Natl. Acad. Sci. USA 2007, 104, 12129–12134. [Google Scholar] [CrossRef] [PubMed]
- Ivić, M. Etiology of endemic nephropathy. Lijec. Vjesn. 1969, 91, 1273–1281. [Google Scholar]
- Chan, W.; Pavlović, N.M.; Li, W.; Chan, C.K.; Liu, J.; Deng, K.; Wang, Y.; Milosavljević, B.; Kostić, E.N. Quantitation of aristolochic acids in corn, wheat grain, and soil samples collected in serbia: Identifying a novel exposure pathway in the etiology of balkan endemic nephropathy. J. Agric. Food Chem. 2016, 64, 5928–5934. [Google Scholar] [CrossRef] [PubMed]
- Grollman, A.P. Aristolochic acid nephropathy: Harbinger of a global iatrogenic disease. Environ. Mol. Mutagen. 2013, 54, 1–7. [Google Scholar] [CrossRef]
- Ma, J.S. The geographical distribution and the system of Aristolochiaceae. Acta Phytotaxon. Sin. 1990, 28, 345–355. [Google Scholar]
- Pavlović, N.M.; Maksimović, V.; Maksimović, J.D.; Orem, W.H.; Tatu, C.A.; Lerch, H.E.; Bunnell, J.E.; Kostić, E.N.; Szilagyi, D.N.; Paunescu, V. Possible health impacts of naturally occurring uptake of aristolochic acids by maize and cucumber roots: Links to the etiology of endemic (balkan) nephropathy. Environ. Geochem. Health 2013, 35, 215–226. [Google Scholar] [CrossRef]
- Mantle, P.G.; Herman, D.; Tatu, C. Is aristolochic acid really the cause of the Balkan endemic nephropathy? J. Controversies Biomed. Res. 2016, 2, 9–20. [Google Scholar] [CrossRef][Green Version]
- Chan, C.-K.; Liu, Y.; Pavlović, N.M.; Chan, W. Etiology of Balkan endemic nephropathy: An update on aristolochic acids exposure mechanisms. Chem. Res. Toxicol. 2018, 31, 1109–1110. [Google Scholar] [CrossRef]
- Li, W.; Chan, C.-K.; Liu, Y.; Yao, J.; Mitić, B.; Kostić, E.N.; Milosavljević, B.S.; Davinić, I.; Orem, W.H.; Tatu, C.A.; et al. Aristolochic acids as persistent soil pollutants: Determination of risk for human exposure and nephropathy from plant uptake. J Agric Food Chem. 2018, 66, 11468–11476. [Google Scholar] [CrossRef]
- Pavlović, N.M. Balkan endemic nephropathy-current status and future perspectives. Clin. Kidney J. 2013, 6, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Cachet, X.; Langrand, J.; Bottai, C.; Dufat, H.; Locatelli-Jouans, C.; Nossin, E.; Boucaud-Maitre, D. Detection of aristolochic acids I and II in “chiniy-Trèf”, a traditional medicinal preparation containing caterpillars feeding on Aristolochia trilobata L. in Martinique, French West Indies. Toxicon 2016, 114, 28–30. [Google Scholar] [CrossRef]
- Peralta-Videa, J.R.; Lopez, M.L.; Narayan, M.; Saupe, G.; Gardea-Torresdey, J. The biochemistry of environmental heavy metal uptake by plants: Implications for the food chain. Int. J. Biochem. Cell Biol. 2009, 41, 1665–1677. [Google Scholar] [CrossRef]
- Behrendt, H.; Brüggemann, R. Modelling the fate of organic chemicals in the soil plant environment: Model study of root uptake of pesticides. Chemosphere 1993, 27, 2325–2332. [Google Scholar] [CrossRef]
- Wang, Y.; Chan, K.K.J.; Chan, W. Plant uptake and metabolism of nitrofuran antibiotics in spring onion grown in nitrofuran-contaminated soil. J. Agric. Food Chem. 2017, 65, 4255–4261. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Hu, Q.; Chan, W. Uptake and accumulation of nephrotoxic and carcinogenic aristolochic acids in food crops grown in Aristolochia clematitis-contaminated soil and water. J. Agric. Food Chem. 2016, 64, 107–112. [Google Scholar] [CrossRef]
- Gruia, A.T.; Oprean, C.; Ivan, A.; Cristea, M.; Draghia, L.; Damiescu, R.; Pavlovic, N.M.; Paunescu, V.; Tatu, C.A. Balkan endemic nephropathy and aristolochic acid I: An investigation into the role of soil and soil organic matter contamination, as a potential natural exposure pathway. Environ. Geochem. Health 2018, 40, 1437–1448. [Google Scholar] [CrossRef]
- De Broe, M.E. Chinese herbs nephropathy and balkan endemic nephropathy: Toward a single entity, aristolochic acid nephropathy. Kidney Int. 2012, 81, 513–515. [Google Scholar] [CrossRef]
- González-Curbelo, M.Á.; Hernández-Borges, J.; Borges-Miquel, T.M.; Rodríguez-Delgado, M.Á. Determination of organophosphorus pesticides and metabolites in cereal-based baby foods and wheat flour by means of ultrasound-assisted extraction and hollow-fiber liquid-phase microextraction prior to gas chromatography with nitrogen phosphorus detection. J. Chromatogr. A 2013, 1313, 166–174. [Google Scholar] [CrossRef]
- Li, W.; Chan, C.-K.; Wong, Y.L.; Chan, K.J.; Chan, H.W.; Chan, W. Cooking methods employing natural anti-oxidant food additives effectively reduced concentration of nephrotoxic and carcinogenic aristolochic acids in contaminated food grains. Food Chem. 2018, 264, 270–276. [Google Scholar] [CrossRef]
- Gifford, F.J.; Gifford, R.M.; Eddleston, M.; Dhaun, N. Endemic nephropathy around the world. Kidney Int. Rep. 2017, 2, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Priestap, H.A.; Velandia, A.E.; Johnson, J.V.; Barbieri, M.A. Secondary metabolite uptake by the Aristolochia-feeding papilionoid butterfly Battus polydamas. Biochem. Syst. Ecol. 2012, 40, 126–137. [Google Scholar] [CrossRef]
- Ahmad, S.A.; Hopkins, T.L. β-Glucosylation of plant phenolics by phenol β-glucosyltransferase in larval tissues of the tobacco hornworm, Manduca sexta (L. ) Insect Biochem. Mol. Biol. 1993, 23, 581–589. [Google Scholar] [CrossRef]
- Bieler, C.A.; Stiborova, M.; Wiessler, M.; Cosyns, J.-P.; van Ypersele de Strihou, C.; Schmeiser, H.H. 32P-post-labelling analysis of DNA adducts formed by aristolochic acid in tissues from patients with Chinese herbs nephropathy. Carcinogenesis 1997, 18, 1063–1067. [Google Scholar] [CrossRef]
- Rosenquist, T.A.; Grollman, A.P. Mutational signature of aristolochic acid: Clue to the recognition of a global disease. DNA Repair 2016, 44, 205–211. [Google Scholar] [CrossRef]
- Chen, M.; Gong, L.; Qi, X.; Xing, G.; Luan, Y.; Wu, Y.; Xiao, Y.; Yao, J.; Li, Y.; Xue, X.; et al. Inhibition of renal NQO1 activity by dicoumarol suppresses nitroreduction of aristolochic acid I and attenuates its nephrotoxicity. Toxicol. Sci. 2011, 122, 288–296. [Google Scholar] [CrossRef]
- Schmeiser, H.H.; Nortier, J.L.; Singh, R.; Gamboa da Costa, G.; Sennesael, J.; Cassuto-Viguier, E.; Ambrosetti, D.; Rorive, S.; Pozdzik, A.; Phillips, D.H.; et al. Exceptionally long-term persistence of DNA adducts formed by carcinogenic aristolochic acid I in renal tissue from patients with aristolochic acid nephropathy. Int. J. Cancer 2014, 135, 562–567. [Google Scholar] [CrossRef]
- Schmeiser, H.H.; Stiborova, M.; Arlt, V.M. Chemical and molecular basis of the carcinogenicity of Aristolochia plants. Curr. Opin. Drug Discov. Dev. 2009, 12, 141–148. [Google Scholar]
- Stiborova, M.; Arlt, V.M.; Schmeiser, H.H. DNA adducts formed by aristolochic acid are unique biomarkers of exposure and explain the initiation phase of upper urothelial cancer. Int. J. Mol. Sci. 2017, 18, 2144. [Google Scholar] [CrossRef]
- Fernando, R.C.; Schmeiser, H.H.; Scherf, H.R.; Wiessler, M. Formation and persistence of specific purine DNA adducts by 32P-postlabelling in target and non-target organs of rats treated with aristolochic acid I. IARC Sci. Publ. 1993, 124, 167–171. [Google Scholar]
- Liu, Y.; Chan, C.-K.; Jin, L.; Wong, S.-K.; Chan, W. Quantitation of DNA adducts in target and nontarget organs of aristolochic acid I-exposed rats: Correlating DNA adduct levels with organotropic activities. Chem. Res. Toxicol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.; Dickman, K.G.; Moriya, M.; Zavadil, J.; Sidorenko, V.S.; Edwards, K.L.; Gnatenko, D.V.; Wu, L.; Turesky, R.J.; Wu, X.R.; et al. Aristolochic acid-associated urothelial cancer in Taiwan. Proc. Natl. Acad. Sci. USA 2012, 109, 8241–8246. [Google Scholar] [CrossRef]
- Bakkar, A.A.; Wallerand, H.; Radvanyi, F.; Lahaye, J.B.; Pissard, S.; Lecerf, L.; Kouyoumdjian, J.C.; Abbou, C.C.; Pairon, J.C.; Jaurand, M.C.; et al. FGFR3 and TP53 gene mutations define two distinct pathways in urothelial cell carcinoma of the bladder. Cancer Res. 2003, 63, 108–8112. [Google Scholar]
- McCormick, F. Ras-related proteins in signal transduction and growth control. Mol. Reprod. Dev. 1995, 42, 500–506. [Google Scholar] [CrossRef]
- Li, Y.C.; Tsai, S.H.; Chen, S.M.; Chang, Y.M.; Huang, T.C.; Huang, Y.P.; Chang, C.T.; Lee, J.A. Aristolochic acid-induced accumulation of methylglyoxal and Nε-(carboxymethyl)lysine: An important and novel pathway in the pathogenic mechanism for aristolochic acid nephropathy. Biochem. Biophys. Res. Commun. 2012, 423, 832–837. [Google Scholar] [CrossRef] [PubMed]
- Thornalley, P.J. Glyoxalase I-structure, function and a critical role in the enzymatic defence against glycation. Biochem. Soc. Trans. 2003, 31, 1343–1348. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shen, H.M.; Ong, C.N. Role of intracellular thiol depletion, mitochondrial dysfunction and reactive oxygen species in Salvia Miltiorrhiza-induced apoptosis in human hepatoma HepG2 cells. Life Sci. 2001, 69, 1833–1850. [Google Scholar] [CrossRef]
- Priestap, H.A.; Barbieri, M.A. Conversion of aristolochic acid I into aristolic acid by reaction with cysteine and glutathione: Biological implications. J. Nat. Prod. 2013, 76, 965–968. [Google Scholar] [CrossRef]
- Lill, R.; Mühlenhoff, U. Iron–sulfur-protein biogenesis in eukaryotes. Trends. Biochem. Sci. 2005, 30, 133–141. [Google Scholar] [CrossRef]
- Baker, D.H.; Czarnecki-Maulden, G.L. Pharmacologic role of cysteine in ameliorating or exacerbating mineral toxicities. J. Nutr. 1987, 117, 1003–1010. [Google Scholar] [CrossRef]
- Pompella, A.; Visvikis, A.; Paolicchi, A.; De Tata, V.; Casini, A.F. The changing faces of glutathione, a cellular protagonist. Biochem. Pharmacol. 2003, 66, 1499–1503. [Google Scholar] [CrossRef]
- Yang, L.; Li, X.; Wang, H. Possible mechanisms explaining the tendency towards interstitial fibrosis in aristolochic acid-induced acute tubular necrosis. Nephrol. Dial. Transplant. 2007, 22, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Lebeau, C.; Arlt, V.M.; Schmeiser, H.H.; Boom, A.; Verroust, P.J.; Devuyst, O.; Beauwens, R. Aristolochic acid impedes endocytosis and induces DNA adducts in proximal tubule cells. Kidney Int. 2001, 6, 1332–1342. [Google Scholar] [CrossRef] [PubMed]
- Lebeau, C.; Debelle, F.D.; Arlt, V.M. Early proximal tubule injury in experimental aristolochic acid nephropathy: Functional and histological studies. Nephrol. Dial. Transplant. 2005, 20, 2321–2332. [Google Scholar] [CrossRef]
- Toback, F.G. Regeneration after acute tubular necrosis. Kidney Int. 1996, 41, 226. [Google Scholar] [CrossRef][Green Version]
- Pozdzik, A.A.; Salmon, I.J.; Debelle, F.D.; Decaestecker, C.; van den Branden, C.; Verbeelen, D.; Deschodt-Lanckman, M.M.; Vanherweghem, J.-L.; Nortier, J.L. Aristolochic acid induces proximal tubule apoptosis and epithelial to mesenchymal transformation. Kidney Int. 2008, 73, 595–607. [Google Scholar] [CrossRef]
- Pozdzik, A.A.; Salmon, I.J.; Husson, C.P.; Decaestecker, C.; Rogier, E.; Bourgeade, M.-F.; Deschodt-Lanckman, M.M.; Vanherweghem, J.-L.; Nortier, J.L. Patterns of interstitial inflammation during the evolution of renal injury in experimental aristolochic acid nephropathy. Nephrol. Dial. Transplant. 2008, 23, 2480–2491. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Z.; Wang, D.; Wang, Y.; Li, Y.; Wu, G. TGF-β1/Smads signaling stimulates renal interstitial fibrosis in experimental AAN. J. Recept. Signal Transduct. Res. 2009, 29, 280–285. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Z.; Shen, H.; Lu, Y.; Li, H.; Ren, X.; Wu, G. TGF-β1/Smad7 Signaling Stimulates Renal Tubulointerstitial Fibrosis Induced by AAI. J. Recept. Signal Transduct. 2008, 28, 413–428. [Google Scholar] [CrossRef]
- Zhou, L.; Fu, P.; Huang, X.R.; Liu, F.; Chung, A.C.K.; Lai, K.N.; Lan, H.Y. Mechanism of chronic aristolochic acid nephropathy: Role of Smad3. Am. J. Physiol. Ren. Physiol. 2010, 298, F1006–F1017. [Google Scholar] [CrossRef]
- Dai, X.-Y.; Zhou, L.; Huang, X.-R.; Fu, P.; Lan, H.-Y. Smad7 protects against chronic aristolochic acid nephropathy in mice. Oncotarget 2015, 6, 11930–11944. [Google Scholar] [CrossRef] [PubMed]
- Chung, A.C.K.; Dong, Y.; Yang, W.; Zhong, X.; Li, R.; Lan, H.Y. Smad7 suppresses renal fibrosis via altering expression of TGF-β/Smad3-regulated microRNAs. Mol. Ther. 2013, 21, 388–398. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.C.; Wang, W.; Huang, X.R.; Fu, P.; Chen, T.H.; Sheikh-Hamad, D.; Lan, H.Y. Ultrasound-microbubble-mediated gene transfer of inducible Smad7 blocks transforming growth factor-beta signaling and fibrosis in rat remnant kidney. Am. J. Pathol. 2005, 166, 761–771. [Google Scholar] [CrossRef]
- Wang, Z.; Zhao, J.; Zhang, J.; Wei, J.; Zhang, J.; Huang, Y. Protective effect of BMP-7 against aristolochic acid-induced renal tubular epithelial cell injury. Toxicol. Lett. 2010, 198, 348–357. [Google Scholar] [CrossRef]
- Li, Y.; Wang, Z.; Wang, S.; Zhao, J.; Zhang, J.; Huang, Y. Gremlin-mediated decrease in bone morphogenetic protein signaling promotes aristolochic acid-induced epithelial-to-mesenchymal transition (EMT) in HK-2 cells. Toxicology 2012, 297, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Ban, T.H.; Min, J.-W.; Seo, C.; Kim, D.R.; Lee, Y.H.; Chung, B.H.; Jeong, K.-H.; Lee, J.W.; Kim, B.S.; Lee, S.-H.; et al. Update of aristolochic acid nephropathy in Korea. Korean J. Intern. Med. 2018, 33, 961–969. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan, C.-K.; Liu, Y.; Pavlović, N.M.; Chan, W. Aristolochic Acids: Newly Identified Exposure Pathways of this Class of Environmental and Food-Borne Contaminants and its Potential Link to Chronic Kidney Diseases. Toxics 2019, 7, 14. https://doi.org/10.3390/toxics7010014
Chan C-K, Liu Y, Pavlović NM, Chan W. Aristolochic Acids: Newly Identified Exposure Pathways of this Class of Environmental and Food-Borne Contaminants and its Potential Link to Chronic Kidney Diseases. Toxics. 2019; 7(1):14. https://doi.org/10.3390/toxics7010014
Chicago/Turabian StyleChan, Chi-Kong, Yushuo Liu, Nikola M. Pavlović, and Wan Chan. 2019. "Aristolochic Acids: Newly Identified Exposure Pathways of this Class of Environmental and Food-Borne Contaminants and its Potential Link to Chronic Kidney Diseases" Toxics 7, no. 1: 14. https://doi.org/10.3390/toxics7010014
APA StyleChan, C.-K., Liu, Y., Pavlović, N. M., & Chan, W. (2019). Aristolochic Acids: Newly Identified Exposure Pathways of this Class of Environmental and Food-Borne Contaminants and its Potential Link to Chronic Kidney Diseases. Toxics, 7(1), 14. https://doi.org/10.3390/toxics7010014




