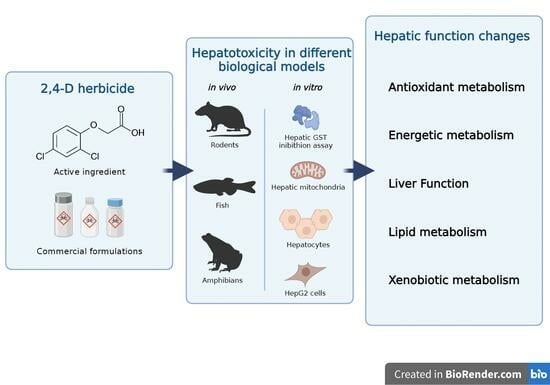
2,4-D Herbicide-Induced Hepatotoxicity: Unveiling Disrupted Liver Functions and Associated Biomarkers
Abstract
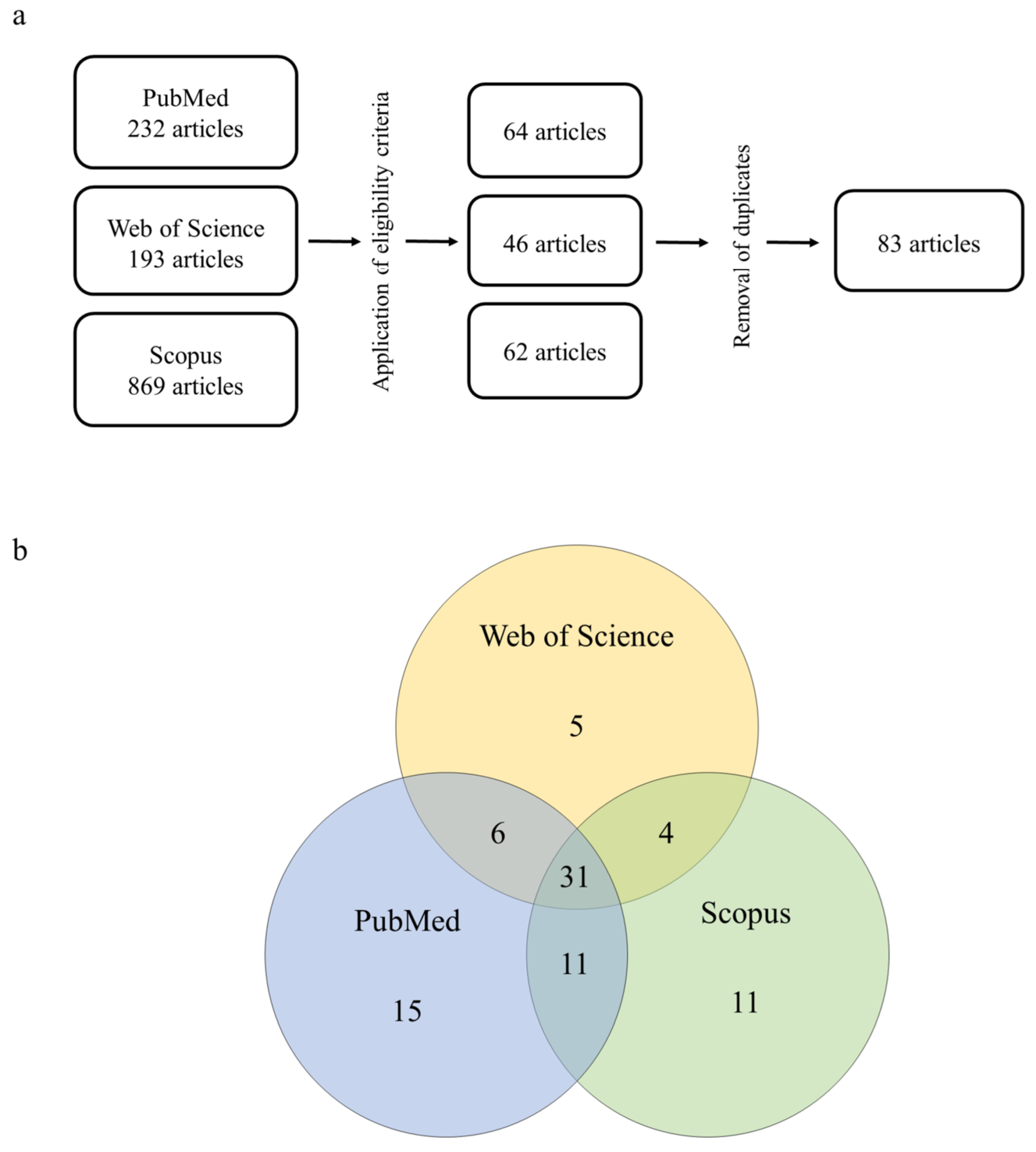
1. Introduction
2. Materials and Methods
- (i)
- Inclusion criteria: articles written and published in English; original and experimental articles; articles that used pure 2,4-D or commercial formulations containing it as the active ingredient; articles that used vertebrates or derivatives (e.g., cells, organelles, enzymes) as biological models; and articles that used biological samples derived from hepatic tissue.
- (ii)
- Exclusion criteria: articles that were not written and published in English; articles that studied other chemical compounds but not 2,4-D; articles that did not use vertebrates or derivatives as biological models; review articles; clinical cases, efficacy studies, protocols, technical reports, and studies that did not meet the research aims.
Overview
3. Results and Discussion
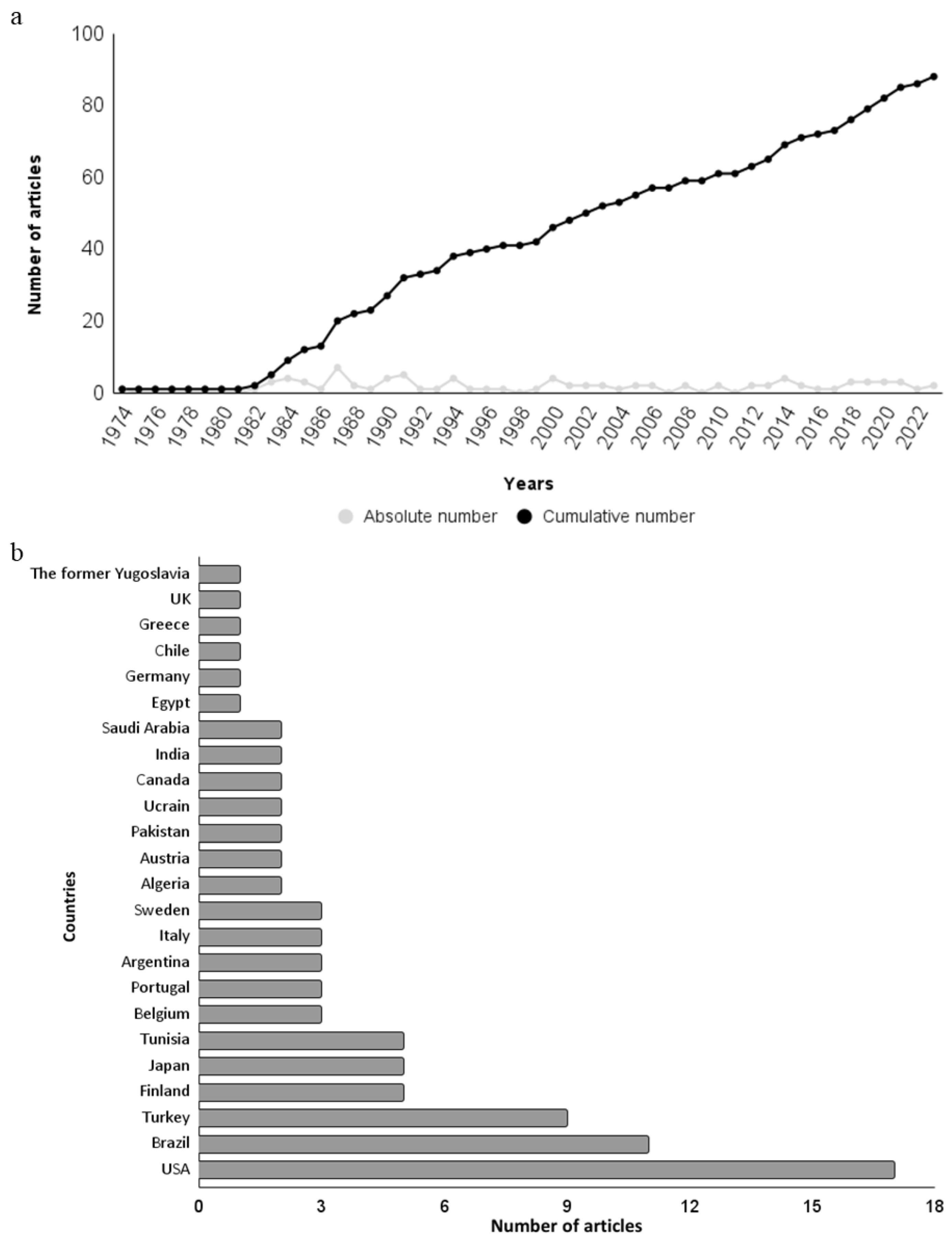
3.1. Historical Review and Geographical Distribution
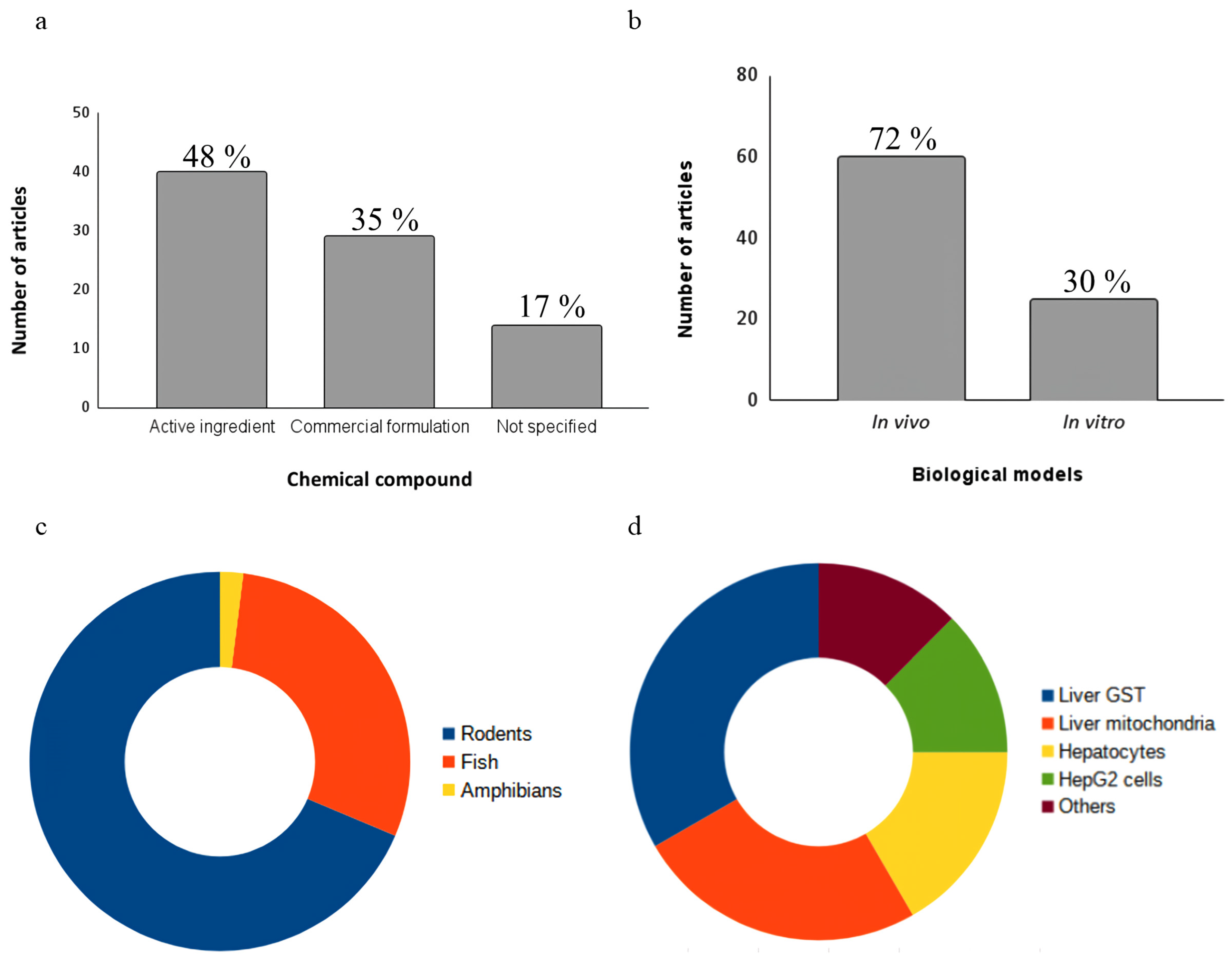
3.2. Chemical Compounds
3.3. Biological Models
3.4. Morphological Markers
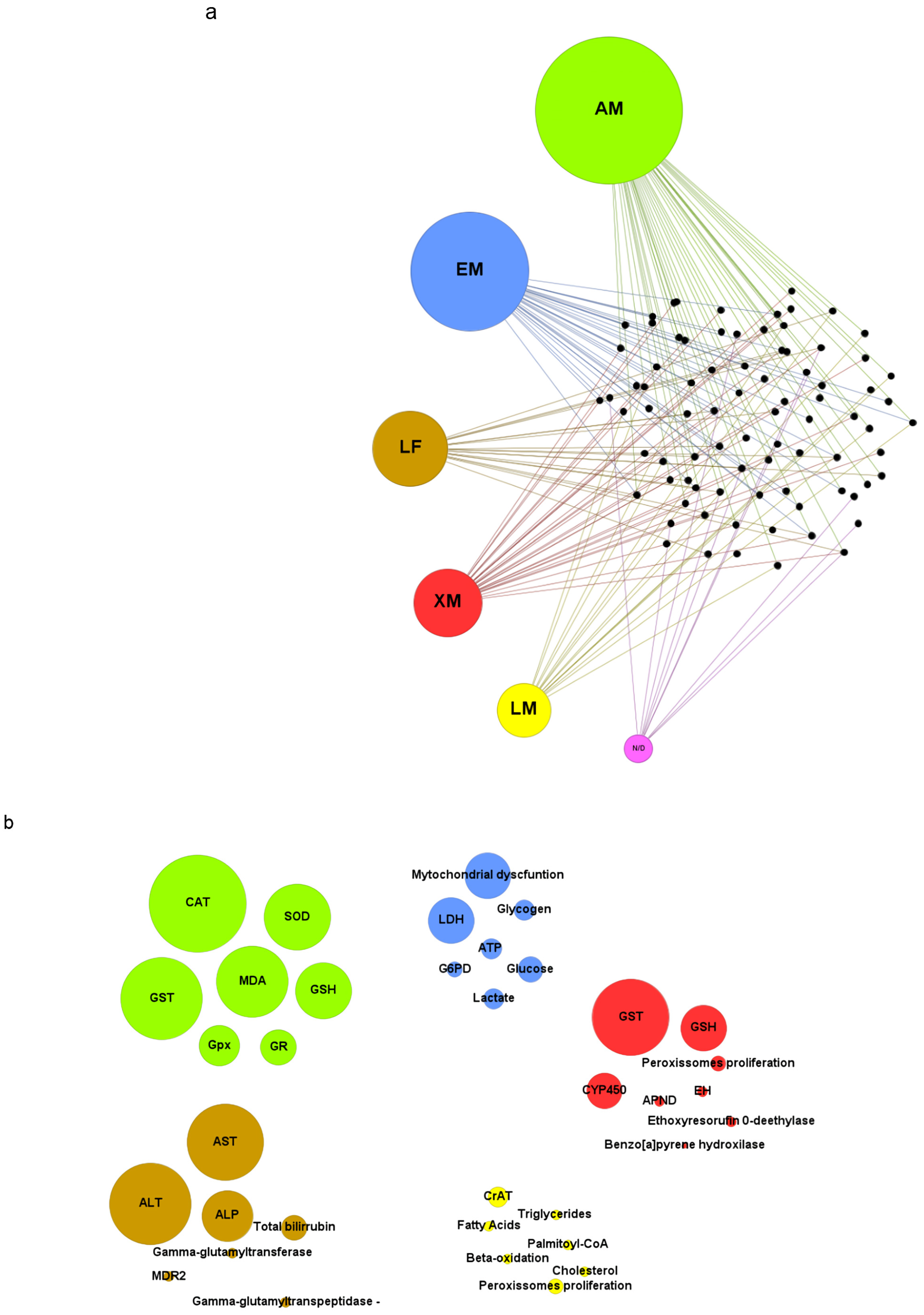
3.5. Toxicity Biomarkers
3.5.1. Antioxidant Metabolism
3.5.2. Energetic Metabolism
3.5.3. Lipid Metabolism
3.5.4. Liver Function
3.5.5. Xenobiotic Metabolism
3.6. Hepatoprotective Assessments
3.7. Pesticide Mixtures Containing 2,4-D
3.8. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, K.; Wu, J.-Q.; Jiang, L.-L.; Shen, L.-Z.; Li, J.-Y.; He, Z.-H.; Wei, P.; Lv, Z.; He, M.-F. Developmental toxicity of 2,4-dichlorophenoxyacetic acid in zebrafish embryos. Chemosphere 2017, 171, 40–48. [Google Scholar] [CrossRef]
- Tomlin, C. The Pesticide Manual: A World Compendium; British Crop Protection Council: Alton, UK, 2009. [Google Scholar]
- Islam, F.; Wang, J.; Farooq, M.A.; Khan, M.S.S.; Xu, L.; Zhu, J.; Zhao, M.; Muños, S.; Li, Q.X.; Zhou, W. Potential impact of the herbicide 2,4-dichlorophenoxyacetic acid on human and ecosystems. Environ. Int. 2018, 111, 332–351. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.X.; Vieira, L.; Souza, J.A.C.R.; Silva, M.G.F.; Muniz, M.S.; Souza, T.; Queiroga, F.R.; Machado, M.R.F.; da Silva, P.M.; Farias, D. Exposure to 2,4-D herbicide induces hepatotoxicity in zebrafish larvae. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2021, 248, 109110. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, A.P.; Morais, E.R.; Oliveira, E.C.; Ghisi, N. de C. Does exposure to environmental 2,4-dichlorophenoxyacetic acid concentrations increase mortality rate in animals? A meta-analytic review. Environ. Pollut. 2022, 303, 119179. [Google Scholar] [CrossRef] [PubMed]
- Freitas, L.; Valadares, L.d.A.; Camozzi, M.; de Oliveira, P.; Ferreira Machado, M.; Lima, F. Animal models in the neurotoxicology of 2,4-D. Hum. Exp. Toxicol. 2019, 38, 1178–1182. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, K. Auxin herbicides: Current status of mechanism and mode of action. Pest Manag. Sci. 2010, 66, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Song, Y. Insight into the mode of action of 2,4-dichlorophenoxyacetic acid (2,4-D) as an herbicide. J. Integr. Plant Biol. 2014, 56, 106–113. [Google Scholar] [CrossRef]
- Akbulut, C.; Yön Ertuğ, N.D. Histopathological Evaluation of Zebrafish (Danio rerio) Intestinal Tissue After Imidacloprid Exposure. Acta Aquat. Turc. 2019, 16, 360–365. [Google Scholar] [CrossRef]
- ANVISA. Monografia D7—2,4-D [Internet]; Agência Nacional de Vigilância Sanitária: Porto Alegre, Brazil, 2018. Available online: http://portal.anvisa.gov.br/Freitasetal.118registros-e-autorizacoes/agrotoxicos (accessed on 18 December 2023).
- Magnoli, K.; Carranza, C.S.; Aluffi, M.E.; Magnoli, C.E.; Barberis, C.L. Herbicides based on 2,4-D: Its behavior in agricultural environments and microbial biodegradation aspects. A review. Environ. Sci. Pollut. Res. 2020, 27, 38501–38512. [Google Scholar] [CrossRef]
- United State Department of Agriculture (USDA). Foreign agricultural service. In Circular Series; World Agricultural Production: Washington, DC, USA, 2017. [Google Scholar]
- Agency for Toxic Substances and Disease Registry. Toxicological Profile for 2,4-Dichlorophenoxyacetic acid [Internet]. 2020. Available online: https://www.atsdr.cdc.gov/ToxProfiles/tp.asp?id=1481&tid=288 (accessed on 18 December 2023).
- Freisthler, M.S.; Robbins, C.R.; Benbrook, C.M.; Young, H.A.; Haas, D.M.; Winchester, P.D.; Perry, M.J. Association between increasing agricultural use of 2,4-D and population biomarkers of exposure: Findings from the National Health and Nutrition Examination Survey, 2001–2014. Environ. Health 2022, 21, 23. [Google Scholar] [CrossRef]
- Peterson, M.A.; McMaster, S.A.; Riechers, D.E.; Skelton, J.; Stahlman, P.W. 2,4-D Past, Present, and Future: A Review. Weed Technol. 2016, 30, 303–345. [Google Scholar] [CrossRef]
- Kennepohl, E.; Munro, I. Phenoxy herbicides (2,4-D). In Handbook of Pesticide Toxicology, 2nd ed.; Krieger, R., Ed.; Academic Press: San Diego, CA, USA, 2001; pp. 1623–1638. [Google Scholar]
- IBAMA. Pesticide Commercialization Reports. [Internet]; Ministry of the Environment: Brasilia, Brazil, 2022. Available online: https://www.gov.br/ibama/en-us/topics/chemicals-and-biology/pesticides/pesticide-commercialization-reports (accessed on 15 February 2023).
- Casimero, M.; Abit, M.J.; Ramirez, A.H.; Dimaano, N.G.; Mendoza, J. Herbicide use history and weed management in Southeast Asia. Adv. Weed Sci. 2022, 40, e020220054. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Li, H.; Tao, F.; Li, S.; Tian, Z.; Xie, H. Formation and contamination of PCDD/Fs, PCBs, PeCBz, HxCBz and polychlorophenols in the production of 2,4-D products. Chemosphere 2013, 92, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Dehnert, G.K.; Karasov, W.H.; Wolman, M.A. 2,4-Dichlorophenoxyacetic acid containing herbicide impairs essential visually guided behaviors of larval fish. Aquat. Toxicol. 2019, 209, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Environmental Protection Agency (EPA). Reregistration Eligibility Decision for 2,4-D; Environmental Protection Agency (EPA): Washington, DC, USA, 2005. [Google Scholar]
- Gaaied, S.; Oliveira, M.; Le Bihanic, F.; Cachot, J.; Banni, M. Gene expression patterns and related enzymatic activities of detoxification and oxidative stress systems in zebrafish larvae exposed to the 2,4-dichlorophenoxyacetic acid herbicide. Chemosphere 2019, 224, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Nault, M.E.; Netherland, M.D.; Mikulyuk, A.; Skogerboe, J.G.; Asplund, T.; Hauxwell, J.; Toshner, P. Efficacy, selectivity, and herbicide concentrations following a whole-lake 2,4-D application targeting Eurasian watermilfoil in two adjacent northern Wisconsin lakes. Lake Reserv. Manag. 2014, 30, 1–10. [Google Scholar] [CrossRef]
- Atamaniuk, T.M.; Kubrak, O.I.; Storey, K.B.; Lushchak, V.I. Oxidative stress as a mechanism for toxicity of 2,4-dichlorophenoxyacetic acid (2,4-D): Studies with goldfish gills. Ecotoxicology 2013, 22, 1498–1508. [Google Scholar] [CrossRef]
- Ensminger, M.P.; Budd, R.; Kelley, K.C.; Goh, K.S. Pesticide occurrence and aquatic benchmark exceedances in urban surface waters and sediments in three urban areas of California, USA, 2008–2011. Environ. Monit. Assess. 2013, 185, 3697–3710. [Google Scholar] [CrossRef]
- Rodil, R.; Quintana, J.B.; Concha-Graña, E.; López-Mahía, P.; Muniategui-Lorenzo, S.; Prada-Rodríguez, D. Emerging pollutants in sewage, surface and drinking water in Galicia (NW Spain). Chemosphere 2012, 86, 1040–1049. [Google Scholar] [CrossRef]
- Meftaul, I.M.; Venkateswarlu, K.; Dharmarajan, R.; Annamalai, P.; Megharaj, M. Movement and Fate of 2,4-D in Urban Soils: A Potential Environmental Health Concern. ACS Omega 2020, 5, 13287–13295. [Google Scholar] [CrossRef]
- Yamini, Y.; Saleh, A. Ultrasound-assisted emulsification microextraction combined with injection-port derivatization for the determination of some chlorophenoxyacetic acids in water samples: Sample Preparation. J. Sep. Sci. 2013, 36, 2330–2338. [Google Scholar] [CrossRef] [PubMed]
- Tsaboula, A.; Papadakis, E.-N.; Vryzas, Z.; Kotopoulou, A.; Kintzikoglou, K.; Papadopoulou-Mourkidou, E. Environmental and human risk hierarchy of pesticides: A prioritization method, based on monitoring, hazard assessment and environmental fate. Environ. Int. 2016, 91, 78–93. [Google Scholar] [CrossRef] [PubMed]
- SISAGUA. Detection and Concentration of Pesticides from 2014 to 2017 in Human Drinking Water. [Internet]; Ministry of Health: Brasilia, Brazil, 2018. Available online: http://www.vigilanciasanitaria.sc.gov.br/index.php/saude-ambiental/sisagua (accessed on 10 April 2023).
- Zuanazzi, N.R.; Ghisi, N.D.C.; Oliveira, E.C. Analysis of global trends and gaps for studies about 2,4-D herbicide toxicity: A scientometric review. Chemosphere 2020, 241, 125016. [Google Scholar] [CrossRef] [PubMed]
- Tichati, L.; Trea, F.; Ouali, K. Potential Role of Selenium Against Hepatotoxicity Induced by 2,4-Dichlorophenoxyacetic Acid in Albino Wistar Rats. Biol. Trace Elem. Res. 2020, 194, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Tichati, L.; Trea, F.; Ouali, K. The antioxidant study proprieties of Thymus munbyanus aqueous extract and its beneficial effect on 2, 4-Dichlorophenoxyacetic acid -induced hepatic oxidative stress in albino Wistar rats. Toxicol. Mech. Methods 2021, 31, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Troudi, A.; Ben Amara, I.; Samet, A.M.; Zeghal, N. Oxidative stress induced by 2,4-phenoxyacetic acid in liver of female rats and their progeny: Biochemical and histopathological studies. Environ. Toxicol. 2012, 27, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Elufioye, T.O.; Habtemariam, S. Hepatoprotective effects of rosmarinic acid: Insight into its mechanisms of action. Biomed. Pharmacother. 2019, 112, 108600. [Google Scholar] [CrossRef] [PubMed]
- Vainio, H.; Nickels, J.; Linnainmaa, K. Phenoxy acid herbicides cause peroxisome proliferation in Chinese hamsters. Scand. J. Work Environ. Health 1982, 8, 70–73. [Google Scholar] [CrossRef]
- Vainio, H.; Linnainmaa, K.; Kähönen, M.; Nickels, J.; Hietanen, E.; Marniemi, J.; Peltonen, P. Hypolipidemia and peroxisome proliferation induced by phenoxyacetic acid herbicides in rats. Biochem. Pharmacol. 1983, 32, 2775–2779. [Google Scholar] [CrossRef]
- Hietanen, E.; Linnainmaa, K.; Vainio, H. Effects of Phenoxyherbicides and Glyphosate on the Hepatic and Intestinal Biotransformation Activities in the Rat. Acta Pharmacol. Toxicol. 1983, 53, 103–112. [Google Scholar] [CrossRef]
- Kawashima, Y.; Katoh, H.; Nakajima, S.; Kozuka, H.; Uchiyama, M. Effects of 2,4-dichlorophenoxyacetic acid and 2,4,5-trichlorophenoxyacetic acid on peroxisomal enzymes in rat liver. Biochem. Pharmacol. 1984, 33, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Y Kawashima; N Hanioka; H Kozuka Induction of microsomal stearoyl-CoA desaturase by the administration of various phenoxyacetic acid derivatives. J. Pharmacobiodyn. 1984, 7, 286–293. [CrossRef] [PubMed]
- Katoh, H.; Nakajima, S.; Kawashima, Y.; Kozuka, H.; Uchiyama, M. Induction of rat hepatic long-chain acyl-CoA hydrolases by various peroxisome proliferators. Biochem. Pharmacol. 1984, 33, 1081–1085. [Google Scholar] [CrossRef] [PubMed]
- Hietanen, E.; Ahotupa, M.; Heinonen, T.; Hamalainen, H.; Kunnas, T.; Linnainmaa, K.; Mantyla, E.; Vainio, H. Enhanced peroxisomal-oxidation of fatty acids and glutathione metabolism in rats exposed to phenoxy-acetic acid. Toxicology 1985, 34, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, B.; Meijer, J.; DePIERRE, J.W. Examination of the structural requirements for proliferation of peroxisomes and mitochondria in mouse liver by hypolipidemic agents, with special emphasis on structural analogues of 2-ethylhexanoic acid. Eur. J. Biochem. 1987, 163, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Gorzinski, S. Acute, pharmacokinetic, and subchronic toxicological studies of 2,4-dichlorophenoxyacetic acid*1, *2. Fundam. Appl. Toxicol. 1987, 9, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Lundgren, B.; Meijer, J.; DePierre, J.W. Induction of cytosolic and microsomal epoxide hydrolases and proliferation of peroxisomes and mitochondria in mouse liver after dietary exposure to p-chlorophenoxyacetic acid, 2,4-dichlorophenoxyacetic acid and 2,4,5-trichlorophenoxyacetic acid. Biochem. Pharmacol. 1987, 36, 815–821. [Google Scholar] [CrossRef]
- Mustonen, R.; Elovaara, E.; Zitting, A.; Linnainmaa, K.; Vainio, H. Effects of commercial chlorophenolate, 2,3,7,8-TCDD, and pure phenoxyacetic acids on hepatic peroxisome proliferation, xenobiotic metabolism and sister chromatid exchange in the rat. Arch. Toxicol. 1989, 63, 203–208. [Google Scholar] [CrossRef]
- Abdellatif, A.G.; Préat, V.; Vamecq, J.; Nilsson, R.; Roberfroid, M. Peroxisome proliferation and modulation of rat liver carcinogenesis by 2,4-dichlorophenoxyacetic acid, 2,4,5-trichlorophenoxyacetic acid, perfluorooctanoic acid and nafenopin. Carcinogenesis 1990, 11, 1899–1902. [Google Scholar] [CrossRef]
- Kuntz, D.J.; Rao, N.G.S.; Berg, I.E.; Khattree, R.; Chaturvedi, A.K. Toxicity of mixtures of parathion, toxaphene and/or 2,4-D in mice. J. Appl. Toxicol. 1990, 10, 257–266. [Google Scholar] [CrossRef]
- Kozuka, H.; Yamada, J.; Horie, S.; Watanabe, T. Characteristics of induction of peroxisomal fatty acid oxidation-related enzymes in rat liver by drugs relationships between structure and inducing activity. Biochem. Pharmacol. 1991, 41, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Inomata, N.; Yoshida, H.; Aoki, Y.; Tsunoda, M.; Yamamoto, M. Effects of MCPA and other phenoxyacid compounds on hepatic xenobiotic metabolism in rats. Tohoku J. Exp. Med. 1991, 165, 171–182. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chaturvedi, A.K.; Kuntz, D.J.; Rao, N.G.S. Metabolic aspects of the toxicology of mixtures of parathion, toxaphene and/or 2,4-D in mice. J. Appl. Toxicol. 1991, 11, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Knopp, D.; Schiller, F. Oral and dermal application of 2,4-dichlorophenoxyacetic acid sodium and dimethylamine salts to male rats: Investigations on absorption and excretion as well as induction of hepatic mixed-function oxidase activities. Arch. Toxicol. 1992, 66, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Paulino, C.A.; Guerra, J.L.; Oliveira, G.H.; Palermo-Neto, J. Acute, subchronic and chronic 2,4-dichlorophenoxyacetic acid (2,4-D) intoxication in rats. Vet. Hum. Toxicol. 1996, 38, 348–352. [Google Scholar]
- Miranda, S.; Vollrath, V.; Wielandt, A.M.; Loyola, G.; Bronfman, M.; Chianale, J. Overexpression of mdr2 gene by peroxisome proliferations in the mouse liver. J. Hepatol. 1997, 26, 1331–1339. [Google Scholar] [CrossRef] [PubMed]
- Badawi, A.F.; Cavalieri, E.L.; Rogan, E.G. Effect of chlorinated hydrocarbons on expression of cytochrome P450 1A1, 1A2 and 1B1 and 2- and 4-hydroxylation of 17β-estradiol in female Sprague–Dawley rats. Carcinogenesis 2000, 21, 1593–1599. [Google Scholar] [CrossRef]
- Di Paolo, O.; de Duffard, A.M.E.; Duffard, R. In vivo and in vitro binding of 2,4-dichlorophenoxyacetic acid to a rat liver mitochondrial protein. Chem. Biol. Interact. 2001, 137, 229–241. [Google Scholar] [CrossRef]
- Ozaki, K.; Mahler, J.F.; Haseman, J.K.; Moomaw, C.R.; Nicolette, M.L.; Nyska, A. Unique Renal Tubule Changes Induced in Rats and Mice by the Peroxisome Proliferators 2,4-Dichlorophenoxyacetic Acid (2,4-D) and WY-14643. Toxicol. Pathol. 2001, 29, 440–450. [Google Scholar] [CrossRef]
- Ge, R.; Tao, L.; Kramer, P.M.; Cunningham, M.L.; Pereira, M.A. Effect of peroxisome proliferators on the methylation and protein level of the c-myc protooncogene in B6C3F1 mice liver. J. Biochem. Mol. Toxicol. 2002, 16, 41–47. [Google Scholar] [CrossRef]
- Yilmaz, H.R.; Yuksel, E. Effect of 2,4-dichlorophenoxyacetic acid on the activities of some metabolic enzymes for generating pyridine nucleotide pool of cells from mouse liver. Toxicol. Ind. Health 2005, 21, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Celik, I.; Tuluce, Y.; Isik, I. Influence of subacute treatment of some plant growth regulators on serum marker enzymes and erythrocyte and tissue antioxidant defense and lipid peroxidation in rats. J. Biochem. Mol. Toxicol. 2006, 20, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Aydýn, H.; Baran, A.; Demirel, G.; Yýldýrým, M. Effects of 2,4-Dichlorophenoxyacetic acid (2,4-D) treatment on the epididymal spermatozoa, blood serum transaminases and its accumulation in liver of rats. Med. Weter. 2006, 62, 1337–1464. [Google Scholar]
- Nakbi, A.; Tayeb, W.; Grissa, A.; Issaoui, M.; Dabbou, S.; Chargui, I.; Ellouz, M.; Miled, A.; Hammami, M. Effects of olive oil and its fractions on oxidative stress and the liver’s fatty acid composition in 2,4-Dichlorophenoxyacetic acid-treated rats. Nutr. Metab. 2010, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Tayeb, W.; Nakbi, A.; Trabelsi, M.; Attia, N.; Miled, A.; Hammami, M. Hepatotoxicity induced by sub-acute exposure of rats to 2,4-Dichlorophenoxyacetic acid based herbicide “Désormone lourd”. J. Hazard. Mater. 2010, 180, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Nakbi, A.; Tayeb, W.; Dabbou, S.; Chargui, I.; Issaoui, M.; Zakhama, A.; Miled, A.; Hammami, M. Hypolipidimic and antioxidant activities of virgin olive oil and its fractions in 2,4-diclorophenoxyacetic acid–treated rats. Nutrition 2012, 28, 81–91. [Google Scholar] [CrossRef]
- Tayeb, W.; Nakbi, A.; Cheraief, I.; Miled, A.; Hammami, M. Alteration of lipid status and lipid metabolism, induction of oxidative stress and lipid peroxidation by 2,4-dichlorophenoxyacetic herbicide in rat liver. Toxicol. Mech. Methods 2013, 23, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Kalipci, E.; Ozdemir, C.; Oztas, H. Assessing eco-toxicological effects of industrial 2,4-D acid iso-octylester herbicide on rat pancreas and liver. Biotech. Histochem. 2013, 88, 202–207. [Google Scholar] [CrossRef]
- Mazhar, F.M.; Moawad, K.M.; El-Dakdoky, M.H.; Amer, A.S. Fetotoxicity of 2,4-dichlorophenoxyacetic acid in rats and the protective role of vitamin E. Toxicol. Ind. Health 2014, 30, 480–488. [Google Scholar] [CrossRef]
- Al-Baroudi, D.A.; Arafat, R.; El-kholy, T. Hepatoprotective effect of chamomile capitula extract against 2,4-dichlorophenoxyacetic acid-induced hepatotoxicity in rats. Life Sci. J. 2014, 11, 34–40. [Google Scholar]
- Dakhakhni, T.H.; Raouf, G.A.; Qusti, S.Y. Evaluation of the toxic effect of the herbicide 2, 4-D on rat hepatocytes: An FT-IR spectroscopic study. Eur. Biophys. J. 2016, 45, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Satapathy, A.; Rao, M. Protective effect of Curcumin on 2,4-Dichlorophenoxy acetic acid exerted Hepatotoxicity in Mice. Res. J. Pharm. Technol. 2018, 11, 637–642. [Google Scholar] [CrossRef]
- Shafeeq, S.; Mahboob, T. Magnesium supplementation ameliorates toxic effects of 2,4-dichlorophenoxyacetic acid in rat model. Hum. Exp. Toxicol. 2020, 39, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Bonfim, D.J.P.; Magalhães, L.R.; Chagas, P.H.N.; Serra, F.D.M.; Benatti, L.A.T.; Nai, G.A. Hepatic, renal, and pancreatic damage associated with chronic exposure to oral and inhaled 2,4-dichlorophenoxy acetic acid (2,4-d): An environmental exposure model in rats. Comp. Clin. Pathol. 2020, 29, 1001–1010. [Google Scholar] [CrossRef]
- Bueno Franco Salla, G.; Bracht, L.; Valderrama Parizotto, A.; Comar, J.F.; Peralta, R.M.; Bracht, F.; Bracht, A. Kinetics of the metabolic effects, distribution spaces and lipid-bilayer affinities of the organo-chlorinated herbicides 2,4-D and picloram in the liver. Toxicol. Lett. 2019, 313, 137–149. [Google Scholar] [CrossRef]
- Shafeeq, S.; Mahboob, T. 2,4-Dichlorophenoxyacetic acid induced hepatic and renal toxicological perturbations in rat model: Attenuation by selenium supplementation. Toxicol. Ind. Health 2021, 37, 152–163. [Google Scholar] [CrossRef]
- Ince, S.; Demirel, H.H.; Zemheri-Navruz, F.; Arslan-Acaroz, D.; Kucukkurt, I.; Acaroz, U.; Tureyen, A.; Demirkapi, E.N. Synergistic toxicity of ethanol and 2,4-dichlorophenoxyacetic acid enhances oxidant status, DNA damage, inflammation, and apoptosis in rats. Environ. Sci. Pollut. Res. 2022, 30, 10710–10723. [Google Scholar] [CrossRef]
- Nechalioti, P.-M.; Karampatzakis, T.; Mesnage, R.; Antoniou, M.N.; Ibragim, M.; Tsatsakis, A.; Docea, A.O.; Nepka, C.; Kouretas, D. Evaluation of perinatal exposure of glyphosate and its mixture with 2,4-D and dicamba οn liver redox status in Wistar rats. Environ. Res. 2023, 228, 115906. [Google Scholar] [CrossRef]
- Gallagher, E.; Digiulio, R. Effects of 2,4-dichlorophenoxyacetic acid and picloram on biotransformation, peroxisomal and serum enzyme activities in channel catfish (Ictalurus punctatus). Toxicol. Lett. 1991, 57, 65–72. [Google Scholar] [CrossRef]
- Neskovid, N.; Karan, V.; Elezovic, I.; Poleksic, V.; Budimir, M. Toxic effects of 2,4-D herbicide on fish. J. Environ. Sci. Health Part B 1994, 29, 265–279. [Google Scholar] [CrossRef]
- Oruç, E.Ö.; Üner, N. Combined effects of 2,4-D and azinphosmethyl on antioxidant enzymes and lipid peroxidation in liver of Oreochromis niloticus. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 2000, 127, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Ackers, J.T.; Johnston, M.F.; Haasch, M.L. Immunodetection of hepatic peroxisomal PMP70 as an indicator of peroxisomal proliferation in the mummichog, Fundulus heteroclitus. Mar. Environ. Res. 2000, 50, 361–365. [Google Scholar] [CrossRef] [PubMed]
- Özcan Oruç, E.; Üner, N. Marker enzyme assesment in the liver of Cyprinus carpio (L.) exposed to 2,4-D and azinphosmethyl: Marker Enzymes in Cyprinus carpio. J. Biochem. Mol. Toxicol. 2002, 16, 182–188. [Google Scholar] [CrossRef] [PubMed]
- da Fonseca, M.B.; Glusczak, L.; Silveira Moraes, B.; de Menezes, C.C.; Pretto, A.; Tierno, M.A.; Zanella, R.; Gonçalves, F.F.; Lúcia Loro, V. The 2,4-D herbicide effects on acetylcholinesterase activity and metabolic parameters of piava freshwater fish (Leporinus obtusidens). Ecotoxicol. Environ. Saf. 2008, 69, 416–420. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, R.; Loro, V.L.; Spanevello, R.; Silveira, F.A.; Luz, L.; Miron, D.S.; Fonseca, M.B.; Moraes, B.S.; Clasen, B. Metabolic and histological parameters of silver catfish (Rhamdia quelen) exposed to commercial formulation of 2,4-dichlorophenoxiacetic acid (2,4-D) herbicide. Pestic. Biochem. Physiol. 2008, 92, 133–137. [Google Scholar] [CrossRef]
- Matviishyn, T.M.; Kubrak, O.I.; Husak, V.V.; Storey, K.B.; Lushchak, V.I. Tissue-specific induction of oxidative stress in goldfish by 2,4-dichlorophenoxyacetic acid: Mild in brain and moderate in liver and kidney. Environ. Toxicol. Pharmacol. 2014, 37, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Vigário, A.F.; Sabóia-Morais, S.M.T. Effects of the 2,4-D herbicide on gills epithelia and liver of the fish Poecilia vivipara. Pesqui. Veterinária Bras. 2014, 34, 523–528. [Google Scholar] [CrossRef]
- Menezes, C.; Fonseca, M.B.; Leitemperger, J.; Pretto, A.; Moraes, B.S.; Murussi, C.R.; Baldisserotto, B.; Loro, V.L. Commercial formulation containing 2,4-D affects biochemical parameters and morphological indices of silver catfish exposed for 90 days. Fish Physiol. Biochem. 2015, 41, 323–330. [Google Scholar] [CrossRef]
- Yakovenko, B.V.; Tretyak, O.P.; Mekhed, O.B.; Iskevych, O.V. Effect of herbicides and surfactants on enzymes of energy metabolism in European carp. Ukr. J. Ecol. 2018, 8, 948–952. [Google Scholar] [CrossRef]
- Kaya, İ.; Yılmaz, M.; Kaya, M.M.; Kükürt, B.; Karapehlivan, M. The Effects of Carbaryl and 2,4-Dichlorophenoxyacetic Acid on Oxidative Stress Index in Capoeta capoeta (Guldensteadt 1773). Pak. J. Zool. 2018, 51, 189–193. Available online: http://researcherslinks.com/current-issues/The-Effects-of-Carbaryl-and-Dichlorophenoxyacetic/20/1/1847/html (accessed on 14 July 2023). [CrossRef]
- Zaffaroni, N.P.; Zavanella, T.; Cattaneo, A.; Arias, E. The toxicity of 2,4-dichlorophenoxyacetic acid to the adult crested newt. Environ. Res. 1986, 41, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Van Meter, R.J.; Glinski, D.A.; Purucker, S.T.; Henderson, W.M. Influence of exposure to pesticide mixtures on the metabolomic profile in post-metamorphic green frogs (Lithobates clamitans). Sci. Total Environ. 2018, 624, 1348–1359. [Google Scholar] [CrossRef] [PubMed]
- Curi, L.M.; Peltzer, P.M.; Sandoval, M.T.; Lajmanovich, R.C. Acute Toxicity and Sublethal Effects Caused by a Commercial Herbicide Formulated with 2,4-D on Physalaemus albonotatus Tadpoles. Water. Air. Soil Pollut. 2019, 230, 22. [Google Scholar] [CrossRef]
- Dierickx, P.J. Interaction of chlorophenoxyalkyl acid herbicides with rat-liver glutathione S-transferases. Food Chem. Toxicol. 1983, 21, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Vessey, D.A.; Boyer, T.D. Differential activation and inhibition of different forms of rat liver glutathione S-transferase by the herbicides 2,4-dichlorophenoxyacetate (2,4-D) and 2,4,5-trichlorophenoxyacetate (2,4,5-T). Toxicol. Appl. Pharmacol. 1984, 73, 492–499. [Google Scholar] [CrossRef]
- Dierickx, P.J. Hepatic glutathione S-transferases in rainbow trout and their interaction with 2,4-dichlorophenoxyacetic acid and 1,4-benzoquinone. Comp. Biochem. Physiol. Part C Comp. Pharmacol. 1985, 82, 495–500. [Google Scholar] [CrossRef]
- Singh, S. Inhibition of human glutathione S-transferases by 2,4-dichlorophenoxyacetate (2,4-D) and 2,4,5-trichlorophenoxyacetate (2,4,5-T). Toxicol. Appl. Pharmacol. 1985, 81, 328–336. [Google Scholar] [CrossRef]
- Elia, A.C.; Mantilacci, L.; Natali, M.; Principato, G. Association of glutathione peroxidase activity with an acidic glutathione S-transferase in carp liver. Ital. J. Zool. 2000, 67, 39–43. [Google Scholar] [CrossRef]
- Dierickx, P.J. Interaction of 1,4-benzoquinone and 2,4-dichlorophenoxyacetic acid with microsomal glutathione transferase from rat liver. Arch. Int. Physiol. Biochim. 1988, 96, 1–5. [Google Scholar] [CrossRef]
- Özaslan, M.S.; Demir, Y.; Aksoy, M.; Küfrevioğlu, Ö.I.; Beydemir, Ş. Inhibition effects of pesticides on glutathione- S -transferase enzyme activity of Van Lake fish liver. J. Biochem. Mol. Toxicol. 2018, 32, e22196. [Google Scholar] [CrossRef]
- Dixon, A.; Osterloh, J.; Becker, C. Inhibition of Palmitoyl Co-enzyme A Hydrolase in Mitochondria and Microsomes by Pharmaceutical Organic Anions. J. Pharm. Sci. 1990, 79, 103–105. [Google Scholar] [CrossRef]
- Zychlinski, L.; Zolnierowicz, S. Comparison of uncoupling activities of chlorophenoxy herbicides in rat liver mitochondria. Toxicol. Lett. 1990, 52, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Palmeira, C.M.; Moreno, A.J.; Madeira, V.M. Interactions of herbicides 2,4-D and dinoseb with liver mitochondrial bioenergetics. Toxicol. Appl. Pharmacol. 1994, 127, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L.F.; Campello, A.P.; Silveira, O. Effect of tordon 2,4-D 64/240 triethanolamine BR on the energy metabolism of rat liver mitochondria. J. Appl. Toxicol. 1994, 14, 21–26. [Google Scholar] [CrossRef]
- Oakes, D.J.; Pollak, J.K. Effects of a herbicide formulation, Tordon 75D®, and its individual components on the oxidative functions of mitochondria. Toxicology 1999, 136, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Palmeira, C.M.; Moreno, A.J.; Madeira, V.M.C. Metabolic alterations in hepatocytes promoted by the herbicides paraquat, dinoseb and 2,4-D. Arch. Toxicol. 1994, 68, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Palmeira, C.M.; Moreno, A.J.; Madeira, V.M.C. Thiols metabolism is altered by the herbicides paraquat, dinoseb and 2,4-D: A study in isolated hepatocytes. Toxicol. Lett. 1995, 81, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Grillo, M.P.; Benet, L.Z. In vitro studies on the chemical reactivity of 2,4-dichlorophenoxyacetyl-S-acyl-CoA thioester. Toxicol. Appl. Pharmacol. 2003, 187, 101–109. [Google Scholar] [CrossRef]
- Salvo, L.M.; Malucelli, M.I.C.; da Silva, J.R.M.C.; Alberton, G.C.; Silva De Assis, H.C. Toxicity assessment of 2,4-D and MCPA herbicides in primary culture of fish hepatic cells. J. Environ. Sci. Health Part B 2015, 50, 449–455. [Google Scholar] [CrossRef]
- Tuschl, H.; Schwab, C. Cytotoxic effects of the herbicide 2,4-dichlorophenoxyacetic acid in HepG2 cells. Food Chem. Toxicol. 2003, 41, 385–393. [Google Scholar] [CrossRef]
- Tuschl, H.; Schwab, C.E. Flow cytometric methods used as screening tests for basal toxicity of chemicals. Toxicol. In Vitro 2004, 18, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Bharadwaj, L.; Dhami, K.; Schneberger, D.; Stevens, M.; Renaud, C.; Ali, A. Altered gene expression in human hepatoma HepG2 cells exposed to low-level 2,4-dichlorophenoxyacetic acid and potassium nitrate. Toxicol. In Vitro 2005, 19, 603–619. [Google Scholar] [CrossRef] [PubMed]
- Barrón Cuenca, J.; De Oliveira Galvão, M.F.; Ünlü Endirlik, B.; Tirado, N.; Dreij, K. In vitro cytotoxicity and genotoxicity of single and combined pesticides used by Bolivian farmers. Environ. Mol. Mutagen. 2022, 63, 4–17. [Google Scholar] [CrossRef] [PubMed]
- Olson, R.J.; Trumble, T.E.; Gamble, W. Alterations in cholesterol and fatty acid biosynthesis in rat liver homogenates by aryloxy acids. Biochem. J. 1974, 142, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Santagostino, A.; Leone, M.P.; Maci, R.; Casale, A.; Marabini, L. Effects of Phenoxyacetic Acid Herbicides on Chicken Embryo Liver Drug Metabolizing Enzymes. Pharmacol. Toxicol. 1991, 68, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Evangelista de Duffard, A.; Fabra de Peretti, A.; Castro de Cantarini, S.; Duffard, R. Effects of 2,4-dichlorophenoxyacetic acid butyl ester on chick liver. Arch. Environ. Contam. Toxicol. 1993, 25, 204–211. Available online: http://link.springer.com/10.1007/BF00212131 (accessed on 14 July 2023). [CrossRef]
- Adeva-Andany, M.M.; Pérez-Felpete, N.; Fernández-Fernández, C.; Donapetry-García, C.; Pazos-García, C. Liver glucose metabolism in humans. Biosci. Rep. 2016, 36, e00416. [Google Scholar] [CrossRef]
- Almazroo, O.A.; Miah, M.K.; Venkataramanan, R. Drug Metabolism in the Liver. Clin. Liver Dis. 2017, 21, 1–20. [Google Scholar] [CrossRef]
- Ore, A.; Akinloye, O. Oxidative Stress and Antioxidant Biomarkers in Clinical and Experimental Models of Non-Alcoholic Fatty Liver Disease. Medicina 2019, 55, 26. [Google Scholar] [CrossRef]
- McGill, M.R. The Past and Present of Serum Aminotransferases and the Future of Liver Injury Biomarkers. EXCLI J 15Doc817 ISSN 1611-2156 [Internet]. 2016. Available online: https://www.excli.de/vol15/McGill_15122016_proof.pdf (accessed on 18 December 2023).
- Nagy, K.; Duca, R.C.; Lovas, S.; Creta, M.; Scheepers, P.T.J.; Godderis, L.; Ádám, B. Systematic review of comparative studies assessing the toxicity of pesticide active ingredients and their product formulations. Environ. Res. 2020, 181, 108926. [Google Scholar] [CrossRef]
- Mesnage, R.; Antoniou, M.N. Ignoring Adjuvant Toxicity Falsifies the Safety Profile of Commercial Pesticides. Front. Public Health 2018, 5, 361. [Google Scholar] [CrossRef] [PubMed]
- Bambino, K.; Morrison, J.; Chu, J. Hepatotoxicity in Zebrafish Larvae. In Developmental Toxicology [Internet]; Hansen, J.M., Winn, L.M., Eds.; Methods in Molecular Biology; Springer: New York, NY, USA, 2019; Volume 1965, pp. 129–138. Available online: http://link.springer.com/10.1007/978-1-4939-9182-2_9 (accessed on 24 November 2023)ISBN 978-1-4939-9181-5.
- Ingber, D.E. Human organs-on-chips for disease modelling, drug development and personalized medicine. Nat. Rev. Genet. 2022, 23, 467–491. [Google Scholar] [CrossRef] [PubMed]
- Lieschke, G.J.; Currie, P.D. Animal models of human disease: Zebrafish swim into view. Nat. Rev. Genet. 2007, 8, 353–367. [Google Scholar] [CrossRef] [PubMed]
- Ruiz de Arcaute, C.; Soloneski, S.; Larramendy, M.L. Toxic and genotoxic effects of the 2,4-dichlorophenoxyacetic acid (2,4-D)-based herbicide on the Neotropical fish Cnesterodon decemmaculatus. Ecotoxicol. Environ. Saf. 2016, 128, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, B.J.; Pack, M. Zebrafish Models of Human Liver Development and Disease. In Comprehensive Physiology [Internet], 1st ed.; Terjung, R., Ed.; Wiley: Hoboken, NJ, USA, 2013; pp. 1213–1230. Available online: https://onlinelibrary.wiley.com/doi/10.1002/cphy.c120021 (accessed on 17 June 2022).
- Trefts, E.; Gannon, M.; Wasserman, D.H. The liver. Curr. Biol. 2017, 27, R1147–R1151. [Google Scholar] [CrossRef] [PubMed]
- Malhi, H.; Guicciardi, M.E.; Gores, G.J. Hepatocyte Death: A Clear and Present Danger. Physiol. Rev. 2010, 90, 1165–1194. [Google Scholar] [CrossRef] [PubMed]
- Rui, L. Energy Metabolism in the Liver. In Comprehensive Physiology [Internet], 1st ed.; Terjung, R., Ed.; Wiley: Hoboken, NJ, USA, 2014; pp. 177–197. Available online: https://onlinelibrary.wiley.com/doi/10.1002/cphy.c130024 (accessed on 20 December 2023).
- Chen, Z.; Tian, R.; She, Z.; Cai, J.; Li, H. Role of oxidative stress in the pathogenesis of nonalcoholic fatty liver disease. Free Radic. Biol. Med. 2020, 152, 116–141. [Google Scholar] [CrossRef]
- Cichoż-Lach, H. Oxidative stress as a crucial factor in liver diseases. World J. Gastroenterol. 2014, 20, 8082. [Google Scholar] [CrossRef]
- Han, K.-H. Relationships among alcoholic liver disease, antioxidants, and antioxidant enzymes. World J. Gastroenterol. 2016, 22, 37. [Google Scholar] [CrossRef]
- Massarsky, A.; Kozal, J.S.; Di Giulio, R.T. Glutathione and zebrafish: Old assays to address a current issue. Chemosphere 2017, 168, 707–715. [Google Scholar] [CrossRef]
- Ramanathan, R.; Ali, A.H.; Ibdah, J.A. Mitochondrial Dysfunction Plays Central Role in Nonalcoholic Fatty Liver Disease. Int. J. Mol. Sci. 2022, 23, 7280. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.; Sahdeo, S.; Gray, J.A.; Morriseau, C.; Hammock, B.D.; Cortopassi, G. A high-throughput screen for mitochondrial function reveals known and novel mitochondrial toxicants in a library of environmental agents. Mitochondrion 2016, 31, 79–83. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Igbinosa, E.O.; Odjadjare, E.E.; Chigor, V.N.; Igbinosa, I.H.; Emoghene, A.O.; Ekhaise, F.O.; Igiehon, N.O.; Idemudia, O.G. Toxicological Profile of Chlorophenols and Their Derivatives in the Environment: The Public Health Perspective. Sci. World J. 2013, 2013, 460215. [Google Scholar] [CrossRef] [PubMed]
- Shannon, R.D.; Boardman, G.D.; Dietrich, A.M.; Bevan, D.R. Mitochondrial response to chlorophenols as a short-term toxicity assay. Environ. Toxicol. Chem. 1991, 10, 57–66. [Google Scholar] [CrossRef]
- Mansouri, A.; Gattolliat, C.-H.; Asselah, T. Mitochondrial Dysfunction and Signaling in Chronic Liver Diseases. Gastroenterology 2018, 155, 629–647. [Google Scholar] [CrossRef] [PubMed]
- Paradies, G. Oxidative stress, cardiolipin and mitochondrial dysfunction in nonalcoholic fatty liver disease. World J. Gastroenterol. 2014, 20, 14205. [Google Scholar] [CrossRef] [PubMed]
- Arya, D.S.; Arora, S.; Malik, S.; Nepal, S.; Kumari, S.; Ojha, S. Effect of Piper betle on cardiac function, marker enzymes, and oxidative stress in isoproterenol-induced cardiotoxicity in rats. Toxicol. Mech. Methods 2010, 20, 564–571. [Google Scholar] [CrossRef]
- Maharajan, K.; Muthulakshmi, S.; Nataraj, B.; Ramesh, M.; Kadirvelu, K. Toxicity assessment of pyriproxyfen in vertebrate model zebrafish embryos (Danio rerio): A multi biomarker study. Aquat. Toxicol. 2018, 196, 132–145. [Google Scholar] [CrossRef]
- Song, M.J.; Park, C.; Kim, H.; Han, S.; Lee, S.H.; Lee, D.H.; Chung, J.H. Carnitine acetyltransferase deficiency mediates mitochondrial dysfunction-induced cellular senescence in dermal fibroblasts. Aging Cell 2023, 22, e14000. [Google Scholar] [CrossRef]
- Okumoto, K.; Tamura, S.; Honsho, M.; Fujiki, Y. Peroxisome: Metabolic Functions and Biogenesis. In Peroxisome Biology: Experimental Models, Peroxisomal Disorders and Neurological Diseases [Internet]; Lizard, G., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 3–17. [Google Scholar] [CrossRef]
- Fransen, M.; Lismont, C.; Walton, P. The Peroxisome-Mitochondria Connection: How and Why? Int. J. Mol. Sci. 2017, 18, 1126. [Google Scholar] [CrossRef]
- Kleiboeker, B.; Lodhi, I.J. Peroxisomal regulation of energy homeostasis: Effect on obesity and related metabolic disorders. Mol. Metab. 2022, 65, 101577. [Google Scholar] [CrossRef] [PubMed]
- Giannini, E.G. Liver enzyme alteration: A guide for clinicians. Can. Med. Assoc. J. 2005, 172, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Sookoian, S.; Pirola, C.J. Liver enzymes, metabolomics and genome-wide association studies: From systems biology to the personalized medicine. World J. Gastroenterol. 2015, 21, 711. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, M.d.S.; Iano, F.G.; Rocia, V.; Yanai, M.M.; Leite, A.d.L.; Furlani, T.A.; Buzalaf, M.A.R.; de Oliveira, R.C. Alkaline phosphatase activity in plasma and liver of rats submitted to chronic exposure to fluoride. Braz. Arch. Biol. Technol. 2011, 54, 1187–1192. [Google Scholar] [CrossRef]
- Veith, A.; Moorthy, B. Role of cytochrome P450s in the generation and metabolism of reactive oxygen species. Curr. Opin. Toxicol. 2018, 7, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Warner, J.; Hardesty, J.; Zirnheld, K.; McClain, C.; Warner, D.; Kirpich, I. Soluble Epoxide Hydrolase Inhibition in Liver Diseases: A Review of Current Research and Knowledge Gaps. Biology 2020, 9, 124. [Google Scholar] [CrossRef]
- Monticelli Barizon, R.R.; Kummrow, F.; Fernandes De Albuquerque, A.; Assalin, M.R.; Rosa, M.A.; Cassoli De Souza Dutra, D.R.; Almeida Pazianotto, R.A. Surface water contamination from pesticide mixtures and risks to aquatic life in a high-input agricultural region of Brazil. Chemosphere 2022, 308, 136400. [Google Scholar] [CrossRef]
- Mansano, A.S.; Moreira, R.A.; Dornfeld, H.C.; Freitas, E.C.; Vieira, E.M.; Daam, M.A.; Rocha, O.; Seleghim, M.H.R. Individual and mixture toxicity of carbofuran and diuron to the protozoan Paramecium caudatum and the cladoceran Ceriodaphnia silvestrii. Ecotoxicol. Environ. Saf. 2020, 201, 110829. [Google Scholar] [CrossRef]
- Aparecida, M.; Campos Ventura-Camargo, B.D.; Miyuki, M. Toxicity of Herbicides: Impact on Aquatic and Soil Biota and Human Health. In Herbicides—Current Research and Case Studies in Use [Internet]; Price, A., Ed.; InTech: London, UK, 2013; Available online: http://www.intechopen.com/books/herbicides-current-research-and-case-studies-in-use/toxicity-of-herbicides-impact-on-aquatic-and-soil-biota-and-human-health (accessed on 18 July 2023).
- Lushchak, V.I.; Matviishyn, T.M.; Husak, V.V.; Storey, J.M.; Storey, K.B. Pesticide Toxicity: A Mechanistic Approach. EXCLI J 17Doc1101 ISSN 1611-2156 [Internet]. 2018. Available online: https://www.excli.de/vol17/Lushchak_08112018_proof.pdf (accessed on 18 July 2023).
- Zhang, C.; Wang, N.; Xu, Y.; Tan, H.-Y.; Li, S.; Feng, Y. Molecular Mechanisms Involved in Oxidative Stress-Associated Liver Injury Induced by Chinese Herbal Medicine: An Experimental Evidence-Based Literature Review and Network Pharmacology Study. Int. J. Mol. Sci. 2018, 19, 2745. [Google Scholar] [CrossRef]
- Zhao, S.; Iyengar, R. Systems Pharmacology: Network Analysis to Identify Multiscale Mechanisms of Drug Action. Annu. Rev. Pharmacol. Toxicol. 2012, 52, 505–521. [Google Scholar] [CrossRef]
- Cotterill, J.V.; Palazzolo, L.; Ridgway, C.; Price, N.; Rorije, E.; Moretto, A.; Peijnenburg, A.; Eberini, I. Predicting estrogen receptor binding of chemicals using a suite of in silico methods—Complementary approaches of (Q)SAR, molecular docking and molecular dynamics. Toxicol. Appl. Pharmacol. 2019, 378, 114630. [Google Scholar] [CrossRef] [PubMed]
- Liebsch, M.; Grune, B.; Seiler, A.; Butzke, D.; Oelgeschläger, M.; Pirow, R.; Adler, S.; Riebeling, C.; Luch, A. Alternatives to animal testing: Current status and future perspectives. Arch. Toxicol. 2011, 85, 841–858. [Google Scholar] [CrossRef] [PubMed]

| Biological Model | Exposure Compounds | Exposure Conditions | Cellular and Tissues Damage | Impaired Biochemical Markers | References |
|---|---|---|---|---|---|
| Chinese Hamsters | Commercial formulation (550 g/L) | AR: oral gavage T: 9 days C: 100 mg/kg of body weight | NA | ND: Peroxissomes plorifaration | Vainio et al. (1982) [36] |
| Rattus novergicus | Commercial formulation (550 mg/kg) | AR: oral gavage T: 2 weeks C: 100–200 mg/kg of body weight | NA | LM: peroxissome proliferation, CrAT, protein lipases AM: CAT | Vainio et al. (1983) [37] |
| Rattus novergicus | Commercial formulation (550 g/L) | AR.: intragastrically gavage T: 2 weeks C: 100, 150 and 200 mg/kg of body weight | NA | XM: EH, UDPglucuronosyltransferase, GST AM: GST | Hietanen et al. (1983) [38] |
| Rattus novergicus | Active ingredient | AR: feeding T: 14 h C: 0.25% w/w | NA | LM: CrAT, palmitoyl-CoA, triglycerides AM: CAT | Kawashima et al. (1984) [39] |
| Rattus novergicus | not specified | AR.: feeding T: 14 days C: 0.5% of diet | NA | LM: stearoyl-CoA | Kawashima et al. (1984) [40] |
| Rattus novergicus | Active ingredient | AR: feeding and subcutaneously T: 1 or 2 weeks C: 0.25% of diet or 0.93 mmole or 1.86 mmole per kg of body weight | NA | LM: acyl-CoA hydrolase II; β oxidation | Katoh et al. (1984) [41] |
| Rattus novergicus | Commercial formulation (550 g/L) | AR: intragastrically ET: 14 days CT: 1 mmol/kg of body weight | NA | LM: peroxissome proliferation, β-oxidation AM: GR | Hietanen et al. (1985) [42] |
| Mus musculus | not specified | AR: diet T: 4 days C: --- | Increase liver somatic index | LM: palmitoil-CoA, CrAT EM: cytochrome oxidase | Lundgren et al. (1987) [43] |
| Rattus novergicus | Active ingredient | AR: gavage and feeding T: single dose and 13 days C: 553 mg/kg and 1090 mg/kg (single dose); 0, 15, 60, 100, or 150 mg/kg/day (13 days) | Dose levels of 100 or 150 mg/kg/day produced minimal swelling and increased staining homogeneity in the liver cells and were associated with a slight elevation in liver weight. | LF: ALT, ALP EM: glucose | Gorzinskj et al. (1987) [44] |
| Mus musculus | not specified | AR: feeding T: 4 days C: 100 mg/kg/bw | NA | XM: EH, CYP450, GST, peroxissome proliferation AM: GST | Lundgren et al. (1987) [45] |
| Rattus novergicus | Active ingredient (>99%) | AR.: intragastrically gavage T: 2 weeks C: 100 mg/kg of body weight | NA | XM: peroxisome proliferation, CYP450, UDP-glucunorosyl transferase, NADPH diaphorase | Mustonen et al. (1989) [46] |
| Rattus novergicus | Active ingredient | AR.: feeding T: 7 months C: 0.05% of diet | NA | LM: peroxissome proliferation, acyl Coa oxidase, dicarboxylyl CoA oxidase | Abdellatif et al.. (1990) [47] |
| Mus musculus | Active ingredient (97–99%) | AR.: oral intubathion T: 14 days exposure + 7 days recovery C: 50 mg/kg | Increase liver/body weight ratio | LF: ALT | Kuntz et al. (1990) [48] |
| Rattus novergicus | Active ingredient | AR: feeding T: 6 days C: 1.680 ppm | NA | LM: CrAT; carnitine palmitoyltransferase fatty acyl-CoA dehydrogenase cyanide-insensitive fatty acyl-CoA, peroxissome proliferation AM: CAT | Kozuka (1991) [49] |
| Rattus novergicus | not specified | AR.: oral T: 2 weeks C: 200 mg/kg/day | NA | XM: NADPH cytocrome C reductase, aniline hydroxylase, Cytocrome B, NADPH ferricicyanide reductase, aminopyrine N-demethylase | N Inomata et al. (1991) [50] |
| Mus musculus | Active ingredient (>97%) | AR: oral intubation T: 7 days C: 50 mg/kg of body weight | NA | XM: amidopyrine N-demethylas, Benzo [a]pyrene hydroxilase | Chaturvedi et al. (1991) [51] |
| Rattus novergicus | Commercial formulation | AR: oral and middorsal skin applications T: single dose C: 1.9 and 2.6 mg/kg of body weight | NA | XM: CYP450, ethylmorphine N-demethylase, ethoxyresorufin O-deethylase | Knopp and Schiller (1992) [52] |
| Rattus novergicus | not specified | AR: oral T: single dose; 30 days and 180 days C: 600 mg/kg (single dose) and 200 ppm (30 and 180 days) | NA | LF: AST, ALT, ALP EM: LDH, amylase, glucose ND: creatinine | Paulino et al. (1996) [53] |
| Mus musculus | Active ingredient | AR.: feeding T: 4 days C: 0.125% of diet | NA | LF: mdr2 gene | Miranda et al. (1997) [54] |
| Rattus novergicus | Active ingredient | AR: oral gavage T: single dose C: 375 mg/L | NA | XM: CYP1A1, CYP1A2, CYP1B1 | Badawi et al. (2000) [55] |
| Rattus novergicus | Active ingredient (>98%) | AR: injections T: 30 days C: 70 mg/kg of body weight | NA | EM: mitochondrial dysfunction | Di Paolo et al. (2001) [56] |
| Rattus novergicus; Mus musculus and Syrian hamsters | Active ingredient | AR: feeding T: 3 months C: 0, 12, 28, 83, 250, 700, and 1680 ppm (M. musculus); 0, 17, 83, 250, 750, 1250, and 2500 ppm (R. novergicus); 0, 12, 100, 500, 1000, and 5000 ppm (Syrian hamsters) | Increase of mice liver weith | XM: CYP450; peroxissome proliferation AM: CAT | Ozaki et al. (2001) [57] |
| Mus musculus | Active ingredient | AR: feeding T: 6 days C: 1.680 ppm | NA | ND: c-myc gene | Ge et al. (2002) [58] |
| Mus musculus | Active ingredient | AR: Intraperitoneally T: 55 days C: 3.8 mg/kg bw | NA | EM: LDH, MDH | Yilmaz and Yuksel (2005) [59] |
| Rattus novergicus | not specified | AR: drink water T: 25 days C: 50 and 100 ppm | NA | AM: SOD, GSH, GR, MDA EM: LDH, creatine kinase LF: AST XM: GSH | Celik et al. (2006) [60] |
| Rattus novergicus | Active ingredient | AR: Feed and drink water T: 30 days C: 25 ppm and 50 ppm (water) and 50 ppm and 100 ppm (food) | No hepatic damage was observed, but the level of 2,4-D in the liver was found to be significantly higher in both the feed and water groups compared to the control group. | NA | Aydin et al. (2006) [61] |
| Rattus novergicus | Active ingredient | AR: drink water T: 21 days C: 600 ppm or 126 mg/kg | Vascular congestion, cytoplasmic vacuolization, and mononuclear cells’ infiltration | AM: SOD, CAT, GPx, MDA LF: AST, ALT, ALP, γ-glutamyl transpeptidase EM: LDH | Troudi et al. (2012) [34] |
| Rattus novergicus | Commercial formulation (600 g/L) | AR: oral gavage T: 4 weeks C: 5 mg/kg/bw | NA | AM: SOD, CAT, GPx, GR, MDA LF: AST, ALT, ALP, γ-GGT, total bilirubin. LM: change in fatty acid composition | Nakbi. et al. (2010) [62] |
| Rattus novergicus | Commercial formulation (600 g/L) | AR: oral gavage T: 4 weeks C: 15, 75 and 150 mg/kg of body weight | Body weight decreased and the liver weight increased significantly. 2,4-D induced hepatic cord disruption, focal necrosis, vessel dilation and pycnotic nucleus. | LF: AST, ALT, ALP, γ-GGT AM: CAT, GR | Tayeb et al. (2010) [63] |
| Rattus novergicus | Commercial formulation (600 g/L) | AR: oral gavage T: 4 weeks C: 5 mg/kg of body weight/day | Vascular congestion and wide sinusoidal spaces and a necrotic | AM: SOD, CAT, GPx, MDA LF: AST, ALT LM: low-density lipoprotein, cholesterol | Nakbi et al. (2012) [64] |
| Rattus novergicus | Commercial formulation (600 g/L) | AR: oral gavage T: 28 days C: 15, 75 and 150 mg/kg/bw/day | NA | AM: SOD, CAT, GPx, GR, MDA LM: change in fatty acid composition | Tayeb et al. (2013) [65] |
| Rattus novergicus | not specified | AR: feeding T: 16 weeks C: 200 mg/kg/day | 2,4-D acid iso-octylester caused the formation of atypical cell foci (ACF) in the pancreata and livers of rats. | NA | Kalipici et al. (2013) [66] |
| Rattus novergicus | Active ingredient (≥90%) | AR: oral gavage T: 19 days C: 100 mg/kg of body weight | NA | AM: CAT, MDA, total antioxidant capacity | Mazhar. et al. (2014) [67] |
| Rattus novergicus | Commercial formulation | AR: oral T: 28 days C: 75 or 150 mg/kg of body weight | 2,4-D increased liver weight and induced nuclear changes in liver cells, including alterations in size and shape, irregularity, and slight distention of nuclear envelope, hepatic nuclei exhibited varying degrees of pyknosis, disaggregation and apoptosis. | LF: AST, ALT, ALP, total bilirubin AM: GR, SOD EM: LDH | Al-Baroudi et al. (2014) [68] |
| Rattus novergicus | Commercial formulation | AR: oral gavage T: 24 h (single dose) C: 639 mg/kg of body weight | NA | AM: hydroperoxyl and carbonyl lipids EM: glycogen | Dakhakhni et al. (2016) [69] |
| Mus musculus | Active ingredient | AR: oral T: 45 days C: 30, 60, 90 mg/kg/day | Vascular and hepatocellular lesions with necrotic changes and focal areas of necrosis in the liver. | AM: GSH, SOD, CAT, GPx, GR, GST and total –SH EM: ATP and SDH XM: GSH and GST | Satapathy and Rao (2018) [70] |
| Rattus novergicus | Active ingredient | AR: oral gavage T: 4 weeks C: 150 mg/kg/day | NA | AM: SOD, CAT, GSH, MDA LF: AST, ALT XM: GSH ND: Urea and creatinine | Shafeeq and Mahboob (2020) [71] |
| Rattus novergicus | Commercial formulation (806 g/L) | AR: inhalation and feed T: 6 months C: 3.71/6.19 and 9.28×10−3 g a.i./ha | The groups exposed to oral 2,4-D had a higher incidence of steatosis, and those exposed to high doses had increased liver inflammation. | LF: ALT | Bonfim et al. (2020) [72] |
| Rattus novergicus | Commercial formulation (600 g/L) | AR: oral gavage T: 4 weeks C: 5 mg/kg/bw/day | Rat livers shown perivascular inflammatory infiltration around the vessel, sinusoidal dilatation and vacuolization of hepatocytes. | AM: SOD, CAT, GSH, GPx, GST, MDA LF: AST, ALT, ALP, total bilirubin EM: LDH XM: GST, GSH | Tichati et al. (2020) [32] |
| Rattus novergicus | Active ingredient (>98%) | AR: cannulation of portal and cava veins liver T: 20 min. C: 10–400 µM | Membrane lipid bilayer deformity | EM: NADH, NAD+, lactate, glycolisis, gluconeogenesis | Salla et al. (2019) [73] |
| Rattus novergicus | Active ingredient | AR: oral gavage T: 4 weeks C: 150 mg/kg/day | NA | AM: SOD, CAT, GSH, MDA LF: AST, ALT, ALP XM: GSH ND: urea and creatinine | Shafeeq and Mahboob (2021) [74] |
| Rattus novergicus | Commercial formulation (600 g/L) | AR: oral gavage T: 30 days C: 5 mg/kg/bw | 2,4-D increases relative and absolute liver weights. Furthermore, 2,4-D induces severe infiltration of mononuclear inflammatory cells with vacuolar degeneration around a dilated central lobular vein, congestion of the hepatic sinusoids, and degenerative hepatocytes with largely vacuolated cytoplasm and a large number of lipid droplets. | AM: SOD, CAT, GPx, GST, MDA, carbonyl proteins LF: AST, ALT, ALP, γ-GGT EM: LDH XM: GST, GSH | Tichati et al. (2021) [33] |
| Rattus novergicus | Commercial formulation (480 g/L) | AR: oral T: 60 days C: 5 mg/kg of body weight | In the liver tissue of rats, focal areas of mononuclear cell infiltration in the pericentral and periacinal region, sinusoidal dilatation, and hyperemia in the vessels and areas of pyknosis and parenchymal degeneration in the nuclei of hepatocytes were determined. | LF: AST, ALT, ALP AM: SOD, GSH, CAT, MDA XM: GSH ND: NF-κB, COX-2, TNF-α, MCP-I, TGFβI, and CYP2E P53, Bax/Bcl-2, caspase-3, caspase-8, caspase-9, and PARP | Sinan Ince et al. (2022) [75] |
| Rattus novergicus | 2,4-D, gliphosate and dicamba (not specified) | AR: drink water T: 90 days C: gliphosate (0.5 mg/kg bw/day) + 2,4-D (0.3 mg/kg bw/day) + dicamba (0.02 mg/kg bw/day) | NA | AM: GSH and MDA | Nechalioti et al. (2023) [76] |
| Ictalurus punctatus | Active ingredient 2,4-D (>99%) Picloram (>99%) | AR: water expossure T: 10 days C: 22.5, 7.5, and 2.25 mg/L | NA | XM: ethoxyresorufin 0-deethylase | Gallagher and Digiulio (1991) [77] |
| Cyprinus carpio | Active ingredient (>98%) | AR: water exposure T: 96 h and 14 days C: 310, 295 and 270 mg/L (96 h) 150, 200, and 250 mg/L (14 days) | Hepatocycites shown slight vacuolar degeneration and pycnotic nuclei (some of them displaced). | LF: AST, ALT | Neskovic et al. (1994) [78] |
| Oreochromis niloticus | Commercial formulation (500 g/L) | AR: water exposure T: 96 h C: 27 ppm | NA | AM: SOD, GPx, GR EM: glucose-6-phosphate dehydrogenase | Oruç. and Uner (2000) [79] |
| Fundulus heteroclitus | not specified | AR: water expossure T: 21 days C: 0.04, 0.41, and 4.1 µM | NA | ND: peroxissome proliferation | Ackers et al. (2000) [80] |
| Cyprinus carpio | Commercial formulation (500 g/L) | AR: water exposure T: 96 h C: 87 ppm | NA | AM: GST, SOD EM: G6PD XM: GST | Oruç and Uner (2002) [81] |
| Leporinus obtusidens | Commercial formulation (868 g/L) | AR: water exposure T: 96 h C: 1 and 10 mg/L | NA | EM: glycogen, lactate, glucose | Fonseca et al. (2008) [82] |
| Rhamdia quelen | Commercial formulation (720 g/L) | AR: water exposure T: 96 h C: 0, 400, 600 and 700 mg/L | Hepatocyte vacualization and changes in its arrangement cords. | EM: glycogen, lactate, glucose | Cattaneo et al. (2008) [83] |
| Carassius auratus | Active ingredient | AR: water exposure T: 90 h C: 1, 10 and 100 mg/L | NA | AM: carbonyl proteins, lipid peroxidases LM: lipid peroxidases | Matviishyn et al. (2014) [84] |
| Poecilia vivipara | Commercial formulation (868 g/L) | AR: water exposure T: 48 h C: 10, 20 and 40 μL | Swollen nuclei and cytoplasmic vacuolization. Finally, the 40 μL/L group presented blood vessel alterations indicating vasodilatation, hepatocytes with swollen nuclei, Ito cells, and micronuclei. | NA | Vigário and Sabóia-Morais (2014) [85] |
| Rhamdia quelen | Commercial formulation (720 g/L) | AR: water exposure T: 90 days C: 0.5 and 2 mg/L | NA | AM: CAT, MDA EM: glycogen; lactate, glucose | Menezes. et al. (2015) [86] |
| Cyprinus carpio L | not specified | A.R.: water exposure T: --- C: 0.2 mg/dm3 | NA | EM: ICDH, LDH, G6PD | Yakovenko et al. (2018) [87] |
| Capoeta capoeta | not specified | AR: water expossure T: 7 days C: 10 and 20 mg/L | NA | AM: plasma oxidative status index LF: AST | Kaya et al. (2018) [88] |
| Danio rerio | Active ingredient (>97%) | AR: water exposure T: 48 h C: 2.5, 5 and 10 mg/L | Hepatocytes had heterogeneous eosinophilic, cytosol vacuolization and cell nucleus were eccentric. Loss of cell boundaries and liver with necrotic appearance. Release of cytosolic content among adjacent cells. | LF: AST, ALT, ALP AM: CAT, GST XM: GST EM: LDH | Martins et al. (2021) [4] |
| Triturus cristatus carnifex | Commercial formulation (37% of 2,4-D as iso-octylic ester) | AR: water exposure T: 3 months C: 25, 50, 75, 100, 125, and 150 ppm | Vacuolar degeneration of liver parenchyma and necrosis of kidney tubules. | NA | Zaffaroni et al. (1986) [89] |
| Lithobates clamitans | Active ingredient (>98%) | AR: soil exposition T: 2 days C: 14.3 µg/cm2 | NA | NA | Van Meter et al. (2018) [90] |
| Physalaemus albonotatus | Commercial formulation) (48.5% w/v of active ingredient) | AR: water exposure T: 96 h (acute) and 49 days (chronic) C: 350, 700, 1400, and 2400 mg/L(acute); 43.7, 87.5, 175 or 262.5 mg/L (chronic) | The liver of treated tadpoles showed enlargement of hepatic sinusoids, hypervascularization, dilation of blood vessels, and vacuolization of hepatocytes | NA | Curi et al. (2019) [91] |
| Biological Model | Exposure Compounds | Exposure Conditions | Impaired Biochemical Markers | References |
|---|---|---|---|---|
| Liver GST of Rattus novergicus | Active ingredient | AR.: enzyme kinetics T: --- C:--- | AM: GST XM: GST | Dierickx (1983) [92] |
| Liver GST of Rattus novergicus | Active ingredient (>99%) | AR: enzyme kinetics T: --- C: 2–12 mM | XM: GST AM: GST | Vessey and Boyer (1984) [93] |
| Liver GST of Salmo gairdneri | Active ingredient | AR: enzyme kinetics T: --- C: 2 mM | AM: GST XM: GST | Dierick (1985) [94] |
| Liver GST of Homo sapiens (autopsy) | Active ingredient (>97%) | AR: --- T: --- C: --- | AM: GST XM: GST | Singh (1985) [95] |
| Liver GST of Cyprinus carpio | not specified | AR: cell culture T: --- C: --- | AM: GST XM: GST | Elia et al. (2000) [96] |
| Liver GST of Rattus novergicus | not specified | AR: --- T: --- C: --- | AM: GST XM: GST | Dierickx (1988) [97] |
| Liver GST of Chalcalburnus tarichii Pallas | Active ingredient | AR.: --- T: --- C: 0.6, 0.23 and 0.57 mM | AM: GST XM: GST | Özaslan et al. (2018) [98] |
| Liver mitochondria of Rattus novergicus | Active ingredient | AR.: cell culture T: --- C: 0, 0.2, 0.5, 1.0, and 2 mM. | LM: palmitoyl CoA hydrolase, fatty acyl CoA EM: mitochondrial dysfunction | Dixon et al. (1990) [99] |
| Liver mitochondria of Rattus novergicus | not specified | AR: cell culture T: --- C: 0.1–4.0 mM | EM: mitochondrial dysfunction | Zychlinski and Zolnierowicz (1990) [100] |
| Liver mitochondria of Rattus novergicus | Active ingredient | AR: cell culture T: --- C: 100, 200, 300, 400, 500, 600, 700 and 800 µM | EM: SDH, cytochrome c reductase, mitochondrial dysfunction | Palmeira et al. (1994) [101] |
| Liver mitochondria of Rattus novergicus | Commercial formulation (2,4-D 1.08 M + Picloram 0.265 M) | AR: cell culture T: --- C: 66.2 nmol picloram + 270 nmol 2,4-D mg−1 protein | EM: NADH oxidase, NADH cytochrome c reductase, ATP, mitochondrial dysfunction | Pereira et al. (1994) [102] |
| Liver mitochondria of Rattus novergicus | Commercial formulation. Tordon (2,4-D 300 g/L + picloram 75 g/L) | AR.: cell culture T: --- C: --- | EM: mitochondrial dysfunction | Oakes and Pollak (1999) [103] |
| Liver Rattus novergicus mitochondria | Active ingredient (>98%) | AR: injections T: 30 days C: 70 mg/kg of body weight | EM: mitochondrial dysfunction | Di Paolo et al. (2001) [56] |
| Hepatocytes of Rattus novergicus | Active ingredient | AR: cell culture T: --- C: 1–10 mM | EM: LDH, ATP, ADP, AMP, NADH, NAD+ AM: GSH, GSSG XM: GSH, GSSG | Palmeira et al. (1994) [104] |
| Hepatocytes of Rattus novergicus | Active ingredient (>98%) | AR: cell culture T: 200 min C: 1, 5 and 10 mM | AM: MDA, proteins thiol, GSH XM: GSH | Palmeira et al. (1995) [105] |
| Hepatocytes of Rattus novergicus | Active ingredient | AR: cell culture ET: 3 months C.T: 1 mM | NA | Li et al. (2003) [106] |
| Hepatocytes of Metynnis roosevelti | Active ingredient | AR: cell culture T: --- C: 0.275, 2.75 and 27.5 mg/L | EM: mitochondrial dysfunction | Salvo et al. (2015) [107] |
| HepG2 cells | Active ingredient | AR: cell culture T: 48 h C: 4, 8 and 16 mM | EM: mitochondrial dysfunction ND: Cell cicle alterations, apoptose, DNA damage | Tuschl and Schwab (2003) [108] |
| HepG2 cells | Active ingredient | AR: cell culture T: 48 h C: 8, 14 and 16 mM | ND: Cell cycle alterations, apoptosis, DNA damage | Tuschl and Schwab (2004) [109] |
| HepG2 cells | Commercial formulation | AR: cell culture T: --- C: 0.1 nM to 4 mM | ND: Genes involved in stress response, cell cycle control, immunological and DNA repair genes. (FTH1, FTL, PCNA, DCLRE1C, TCLK1, JM11, VEGF, USP19, DDB2, IL1RL1, PTGER3 and GTF2A.) | Bharadwaj et al. (2005) [110] |
| HepG2 cell | Active ingredient (>90%) | AR.: cell culture T: --- C: 0.001–0.1 mM | NA | Barrón Cuenca et al. (2022) [111] |
| Liver homogenates of Rattus novergicus | Active ingredient | AR: cell culture T: --- C: --- | LM: cholesterol | Olson et al. (1974) [112] |
| Chicken embryo | Commercial formulation (37%) | AR.: injected into the air cell of the eggs T: 19 days C: 1, 2 and 4 mg/egg | XM: ethoxycoumarin O-deethylase, GST AM: GST | Santagostino et al. (1991) [113] |
| Chicken Liver | Commercial formulation (31.6% w/v) | AR: fertilized eggs were externally treated T: 21 days C: 3.1 mg | EM: G6Pase LM: total lipids AM: CAT | Duffard et al. (1993) [114] |
| Biological Model | Hepatoprotective Agent | Concentrations and Time of Exposure | Hepatoprotective Effects | References |
|---|---|---|---|---|
| Rattus novergicus | Extra virgin olive oil (EVOO) and its hydrophilic fraction (OOHF) | C: 2,4-D (5 mg/kg body weight) + EVOO (300 μL/day) or OOHF (1 mL/day) T: 4 weeks | EVOO and OOHF supplementation induced a significant increase in the antioxidant enzyme activities (SOD, CAT, GPx and GR) and liver markers (AST, ALT and total bilirubin) and a decrease in the conjugated diene (CD) and thiobarbituric acid-reactive substance (TBAR) levels in the liver. | Nakbi, A. et al. (2010) [62] |
| Rattus novergicus | Extra virgin olive oil (EVOO) and its hydrophilic fraction (OOHF) | C: 2,4-D (5 mg/kg body weight) + EVOO (300 μL/day) or OOHF (1 mL/day) T: 4 weeks | EVOO and OOHF supplementation induced a significant increase in the antioxidant enzyme activities (SOD, CAT, GPx) and liver markers (AST, ALT and total bilirubin), and decreased MDA levels in the liver. | Nakbi, A. et al. (2012) [64] |
| Rattus novergicus | Chamomile capitula extract | C: 2,4-D (75 or 150 mg/kg body weight) + Chamomile capitula extract—(500 mg/kg body weight) T: 28 days | Chamomile capitula extract presented antioxidant effects, improving the levels of SOD and GR. The levels of hepatic enzymes AST, ALT, ALP, and LDH decreased, as well as levels of total bilirubin. Additionally, the degenerative damages in the hepatic tissue caused by 2,4-D were also alleviated. | Al-Baroudi et al. (2014) [68] |
| Mus musculus | Curcumin | C: 2,4-D (30, 60, 90 mg/kg/day) + Curcumin (10 mg/kg/day) T: 45 days | Curcumin supplementation exhibited antioxidant effects, mainly normalizing the levels of GSH, GR, and lipid peroxidation. Furthermore, curcumin supplementation reduced hepatic tissue damage. | Satapathy and Rao (2018) [70] |
| Rattus novergicus | Magnesium (Mg) | C: 2,4-D (150 mg/kg body weight/day) + Mg supplement (50 mg/kg body weight/day) T: 4 weeks | Mg supplementation exhibited its antioxidant properties by significantly improving urea, creatinine SOD, MDA, CAT, GSH and MDA levels and antioxidant enzyme activities. Hepatic markers were also improved: AST, ALT and ALP and absolute liver weight. | Shafeeq and Mahboob (2020) [71] |
| Rattus novergicus | Selenium (Se) | C: 2,4-D (5 mg/kg body weight/day) + Se supplement (1 mg/kg body weight/day) T: 4 weeks | Se supplementation in 2,4-D-treated rats elicited a reduction in the toxic effects of the pesticide by improving the studied parameters (absolute liver weight, total bilirubin, AST, ALP, LDH, MDA and carbonyl proteins), which was confirmed by the histological study of the liver. | Tichati, L. et al. (2020) [32] |
| Rattus novergicus | Selenium (Se) | C: 2,4-D (150 mg/kg body weight/day) + Se supplement (1 mg/kg body weight/day) T: 4 weeks | Se supplementation exhibited its antioxidant properties by significantly improving urea, creatinine, ALP, AST, and ALT, and MDA levels and antioxidant enzyme activities. Hepatic and renal toxicities were attenuated via Se supplementation. | Shafeeq and Mahboob (2021) [74] |
| Rattus novergicus | Thymus munbyanus extract (AETM) | C: 2,4-D (5 mg/kg body weight) + AETM (10 mL/kg body weight) T: 30 days | AETM supplementation showed a marked enhancement in the above altered hepatic functional and antioxidant parameters (CAT, GST, total bilirubin, AST, ALP, MDA, carbonyl proteins) and liver histopathology. | Tichati, L. et al. (2021) [33] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, R.X.; Carvalho, M.; Maia, M.E.; Flor, B.; Souza, T.; Rocha, T.L.; Félix, L.M.; Farias, D. 2,4-D Herbicide-Induced Hepatotoxicity: Unveiling Disrupted Liver Functions and Associated Biomarkers. Toxics 2024, 12, 35. https://doi.org/10.3390/toxics12010035
Martins RX, Carvalho M, Maia ME, Flor B, Souza T, Rocha TL, Félix LM, Farias D. 2,4-D Herbicide-Induced Hepatotoxicity: Unveiling Disrupted Liver Functions and Associated Biomarkers. Toxics. 2024; 12(1):35. https://doi.org/10.3390/toxics12010035
Chicago/Turabian StyleMartins, Rafael Xavier, Matheus Carvalho, Maria Eduarda Maia, Bruno Flor, Terezinha Souza, Thiago Lopes Rocha, Luís M. Félix, and Davi Farias. 2024. "2,4-D Herbicide-Induced Hepatotoxicity: Unveiling Disrupted Liver Functions and Associated Biomarkers" Toxics 12, no. 1: 35. https://doi.org/10.3390/toxics12010035
APA StyleMartins, R. X., Carvalho, M., Maia, M. E., Flor, B., Souza, T., Rocha, T. L., Félix, L. M., & Farias, D. (2024). 2,4-D Herbicide-Induced Hepatotoxicity: Unveiling Disrupted Liver Functions and Associated Biomarkers. Toxics, 12(1), 35. https://doi.org/10.3390/toxics12010035