Immunotoxicity and Transcriptome Analyses of Zebrafish (Danio rerio) Embryos Exposed to 6:2 FTSA
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Chemicals and Test Solutions
2.2. Adult Zebrafish Rearing and Embryo Collection
2.3. Experimental Design
2.4. Biochemical Index Detection
2.5. Immune-Related Protein Expression
2.6. Transcriptomic Analysis
2.7. Statistical Analysis
3. Results
3.1. Effect of 6:2 FTSA on Oxidative Stress
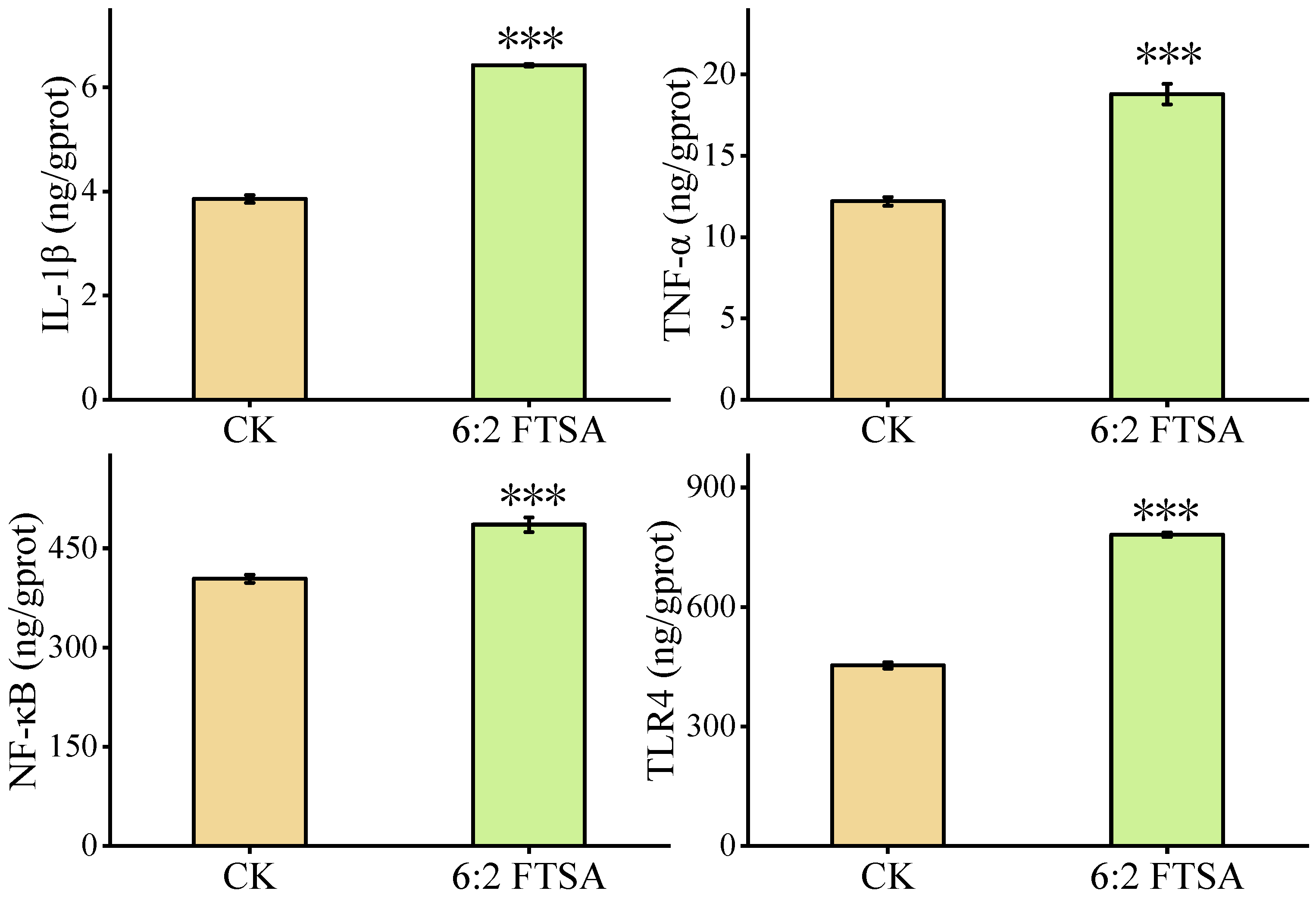
3.2. Effect of 6:2 FTSA on the Inflammatory Response
3.3. Effect of 6:2 FTSA on Immune-Related Proteins
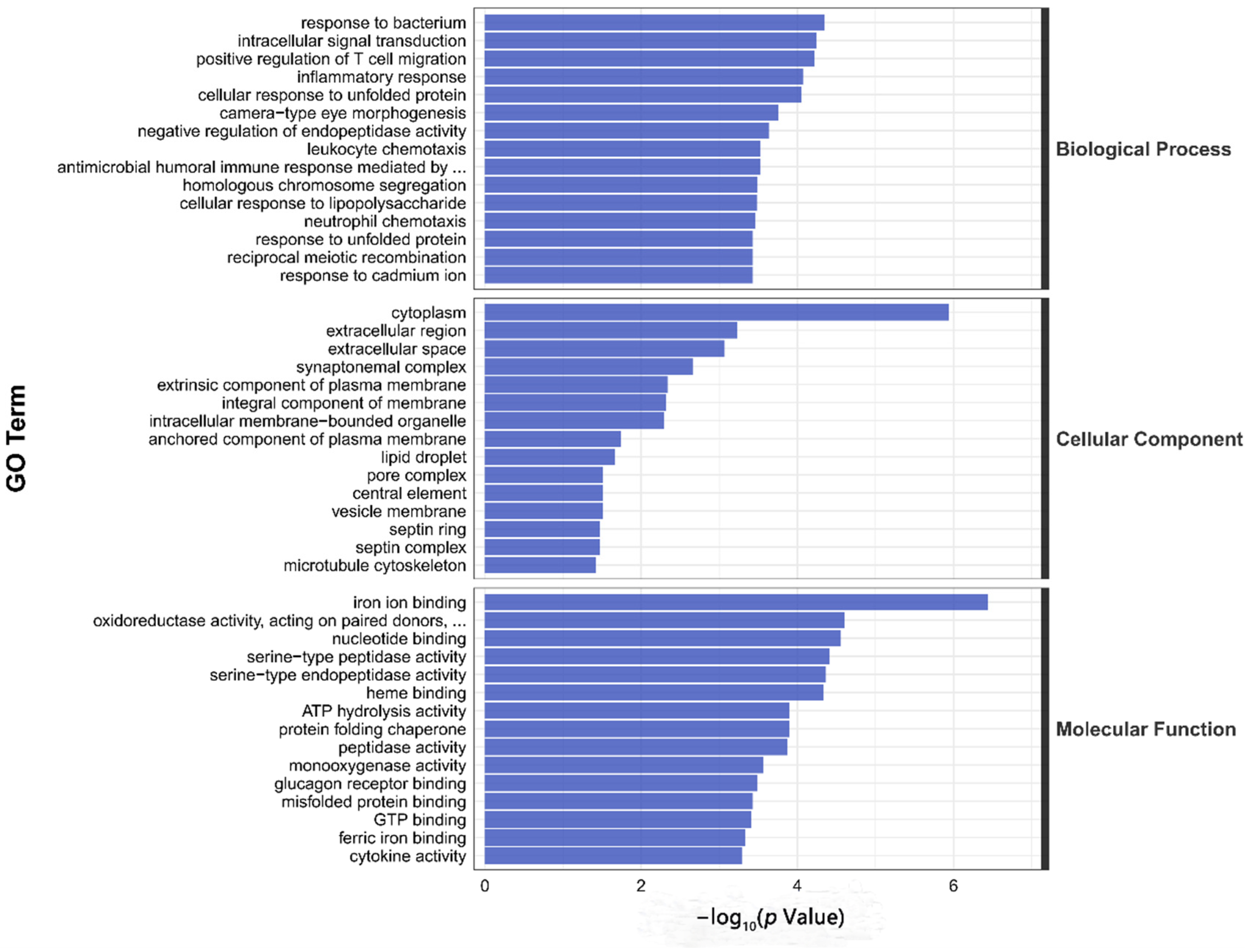
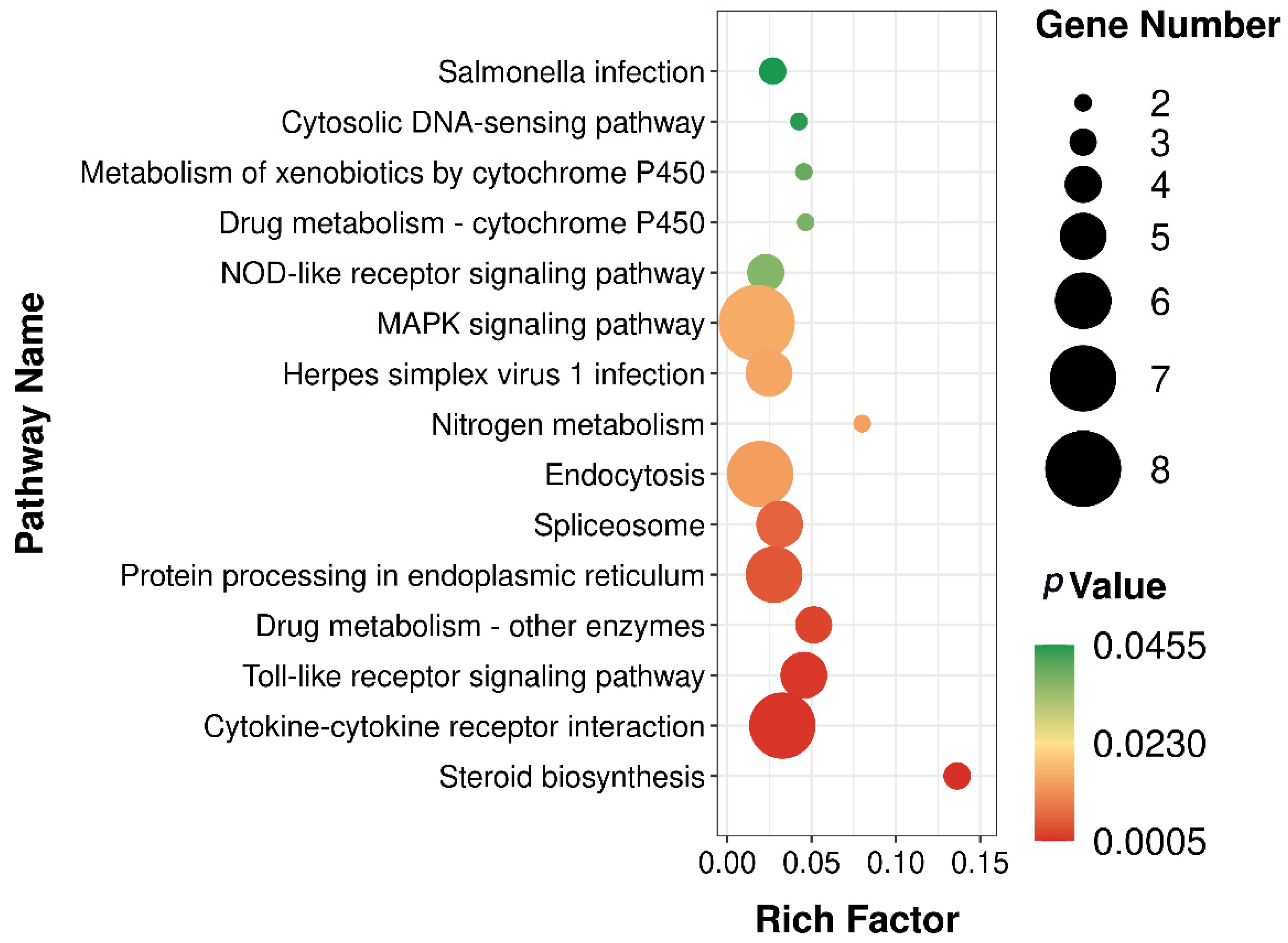
3.4. Transcriptome Sequencing Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chu, S.; Letcher, R.J. Linear and branched perfluorooctane sulfonate isomers in technical product and environmental samples by in-port derivatization-gas chromatography-mass spectrometry. Anal. Chem. 2009, 81, 4256–4262. [Google Scholar] [CrossRef] [PubMed]
- Falandysz, J.; Taniyasu, S.; Gulkowska, A.; Yamashita, N.; Schulte-Oehlmann, U. Is fish a major source of fluorinated surfactants and repellents in humans living on the Baltic Coast? Environ. Sci. Technol. 2006, 40, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Uwayezu, J.-N.; Yeung, L.W.; Bäckström, M. Sorption of PFOS isomers on goethite as a function of pH, dissolved organic matter (humic and fulvic acid) and sulfate. Chemosphere 2019, 233, 896–904. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Song, X.; Liu, Z.; Ding, X.; Chen, H.; Ding, D. Occurrence, source apportionment, plant bioaccumulation and human exposure of legacy and emerging per- and polyfluoroalkyl substances in soil and plant leaves near a landfill in China. Sci. Total. Environ. 2021, 776, 145731. [Google Scholar] [CrossRef] [PubMed]
- Rayne, S.; Forest, K. Perfluoroalkyl sulfonic and carboxylic acids: A critical review of physicochemical properties, levels and patterns in waters and wastewaters, and treatment methods. J. Environ. Sci. Health Part A 2009, 44, 1145–1199. [Google Scholar] [CrossRef]
- Jantzen, C.E.; Annunziato, K.M.; Cooper, K.R. Behavioral, morphometric, and gene expression effects in adult zebrafish (Danio rerio) embryonically exposed to PFOA, PFOS, and PFNA. Aquat. Toxicol. 2016, 180, 123–130. [Google Scholar] [CrossRef]
- Chen, J.; Wang, X.; Ge, X.; Wang, D.; Wang, T.; Zhang, L.; Tanguay, R.L.; Simonich, M.; Huang, C.; Dong, Q. Chronic perfluorooctanesulphonic acid (PFOS) exposure produces estrogenic effects in zebrafish. Environ. Pollut. 2016, 218, 702–708. [Google Scholar] [CrossRef]
- Schröter-Kermani, C.; Müller, J.; Jürling, H.; Conrad, A.; Schulte, C. Retrospective monitoring of perfluorocarboxylates and perfluorosulfonates in human plasma archived by the German Environmental Specimen Bank. Int. J. Hyg. Environ. Health 2013, 216, 633–640. [Google Scholar] [CrossRef]
- Louisse, J.; Rijkers, D.; Stoopen, G.; Janssen, A.; Staats, M.; Hoogenboom, R.; Kersten, S.; Peijnenburg, A. Perfluorooctanoic acid (PFOA), perfluorooctane sulfonic acid (PFOS), and perfluorononanoic acid (PFNA) increase triglyceride levels and decrease cholesterogenic gene expression in human HepaRG liver cells. Arch. Toxicol. 2020, 94, 3137–3155. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, K.; Li, W.; Chai, Y.; Zhu, J.; Chu, B.; Li, N.; Yan, J.; Zhang, S.; Yang, Y. Varied thyroid disrupting effects of perfluorooctanoic acid (PFOA) and its novel alternatives hexafluoropropylene-oxide-dimer-acid (GenX) and ammonium 4,8-dioxa-3H-perfluorononanoate (ADONA) in vitro. Environ. Int. 2021, 156, 106745. [Google Scholar] [CrossRef]
- Coperchini, F.; Croce, L.; Denegri, M.; Pignatti, P.; Agozzino, M.; Netti, G.S.; Imbriani, M.; Rotondi, M.; Chiovato, L. Adverse effects of in vitro GenX exposure on rat thyroid cell viability, DNA integrity and thyroid-related genes expression. Environ. Pollut. 2020, 264, 114778. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Lai, H.; Wang, Q.; Martínez, R.; Zhang, M.; Liu, Y.; Huang, J.; Deng, M.; Tu, W. Immunotoxicity of F 53B, an alternative to PFOS, on zebrafish (Danio rerio) at different early life stages. Sci. Total. Environ. 2021, 790, 148165. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.J.; Pyo, M.C.; Park, Y.; Kim, B.Y.; Lee, K.-W. Hexafluoropropylene oxide dimer acid (GenX) exposure induces apoptosis in HepG2 cells. Heliyon 2021, 7, e08272. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Huang, J.; Zhang, K.; Yu, G.; Deng, S.; Wang, B. Stability of 6:2 fluorotelomer sulfonate in advanced oxidation processes: Degradation kinetics and pathway. Environ. Sci. Pollut. Res. 2013, 21, 4634–4642. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-H.; Shi, Y.; Strynar, M.; Chu, K.-H. Desulfonation and defluorination of 6:2 fluorotelomer sulfonic acid (6:2 FTSA) by Rhodococcus jostii RHA1: Carbon and sulfur sources, enzymes, and pathways. J. Hazard. Mater. 2021, 423, 127052. [Google Scholar] [CrossRef] [PubMed]
- Sheng, N.; Zhou, X.; Zheng, F.; Pan, Y.; Guo, X.; Guo, Y.; Sun, Y.; Dai, J. Comparative hepatotoxicity of 6:2 fluorotelomer carboxylic acid and 6:2 fluorotelomer sulfonic acid, two fluorinated alternatives to long-chain perfluoroalkyl acids, on adult male mice. Arch. Toxicol. 2016, 91, 2909–2919. [Google Scholar] [CrossRef]
- Marchiandi, J.; Szabo, D.; Dagnino, S.; Green, M.P.; Clarke, B.O. Occurrence and fate of legacy and novel per- and polyfluoroalkyl substances (PFASs) in freshwater after an industrial fire of unknown chemical stockpiles. Environ. Pollut. 2021, 278, 116839. [Google Scholar] [CrossRef]
- Wang, S.; Ding, G.; Liu, Y.; Dou, Z.; Chen, H.; Ya, M.; Lin, X.; Li, Q.; Li, Y.; Wang, X. Legacy and emerging persistent organic pollutants in the marginal seas of China: Occurrence and phase partitioning. Sci. Total. Environ. 2022, 827, 154274. [Google Scholar] [CrossRef]
- Feng, X.; Ye, M.; Li, Y.; Zhou, J.; Sun, B.; Zhu, Y.; Zhu, L. Potential sources and sediment-pore water partitioning behaviors of emerging per/polyfluoroalkyl substances in the South Yellow Sea. J. Hazard. Mater. 2020, 389, 122124. [Google Scholar] [CrossRef]
- Åkerblom, S.; Negm, N.; Wu, P.; Bishop, K.; Ahrens, L. Variation and accumulation patterns of poly- and perfluoroalkyl substances (PFAS) in European perch (Perca fluviatilis) across a gradient of pristine Swedish lakes. Sci. Total Environ. 2017, 599-600, 1685–1692. [Google Scholar] [CrossRef]
- Munoz, G.; Desrosiers, M.; Vetter, L.; Duy, S.V.; Jarjour, J.; Liu, J.; Sauvé, S. Bioaccumulation of Zwitterionic Polyfluoroalkyl Substances in Earthworms Exposed to Aqueous Film-Forming Foam Impacted Soils. Environ. Sci. Technol. 2020, 54, 1687–1697. [Google Scholar] [CrossRef] [PubMed]
- Kaboré, H.A.; Goeury, K.; Desrosiers, M.; Duy, S.V.; Liu, J.; Cabana, G.; Munoz, G.; Sauvé, S. Novel and legacy per- and polyfluoroalkyl substances (PFAS) in freshwater sporting fish from background and firefighting foam impacted ecosystems in Eastern Canada. Sci. Total. Environ. 2021, 816, 151563. [Google Scholar] [CrossRef] [PubMed]
- Hoke, R.A.; Ferrell, B.D.; Ryan, T.; Sloman, T.L.; Green, J.W.; Nabb, D.L.; Mingoia, R.; Buck, R.C.; Korzeniowski, S.H. Aquatic hazard, bioaccumulation and screening risk assessment for 6:2 fluorotelomer sulfonate. Chemosphere 2015, 128, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Menger, F.; Pohl, J.; Ahrens, L.; Carlsson, G.; Örn, S. Behavioural effects and bioconcentration of per- and polyfluoroalkyl substances (PFASs) in zebrafish (Danio rerio) embryos. Chemosphere 2019, 245, 125573. [Google Scholar] [CrossRef] [PubMed]
- Martínez, R.; Navarro-Martín, L.; Luccarelli, C.; Codina, A.E.; Raldúa, D.; Barata, C.; Tauler, R.; Piña, B. Unravelling the mechanisms of PFOS toxicity by combining morphological and transcriptomic analyses in zebrafish embryos. Sci. Total. Environ. 2019, 674, 462–471. [Google Scholar] [CrossRef]
- Dang, Y.; Wang, F.; Liu, C. Real-time PCR array to study the effects of chemicals on the growth hormone/insulin-like growth factors (GH/IGFs) axis of zebrafish embryos/larvae. Chemosphere 2018, 207, 365–376. [Google Scholar] [CrossRef]
- Chen, J.; Tanguay, R.L.; Tal, T.L.; Gai, Z.; Ma, X.; Bai, C.; Tilton, S.C.; Jin, D.; Yang, D.; Huang, C.; et al. Early life perfluorooctanesulphonic acid (PFOS) exposure impairs zebrafish organogenesis. Aquat. Toxicol. 2014, 150, 124–132. [Google Scholar] [CrossRef]
- Guo, J.; Wu, P.; Cao, J.; Luo, Y.; Chen, J.; Wang, G.; Guo, W.; Wang, T.; He, X. The PFOS disturbed immunomodulatory functions via nuclear Factor-kappaB signaling in liver of zebrafish (Danio rerio). Fish Shellfish Immunol. 2019, 91, 87–98. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Liang, L.; Pan, Y.; Bin, L.; Liu, Y.; Huang, W.; Li, R.; Lai, K.P. Immunotoxicity mechanisms of perfluorinated compounds PFOA and PFOS. Chemosphere 2021, 291, 132892. [Google Scholar] [CrossRef]
- Huang, J.; Wang, Q.; Liu, S.; Lai, H.; Tu, W. Comparative chronic toxicities of PFOS and its novel alternatives on the immune system associated with intestinal microbiota dysbiosis in adult zebrafish. J. Hazard. Mater. 2022, 425, 127950. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Liu, K.; Li, J.; Chen, L.; Lin, K. Uptake and depuration kinetics of lead (Pb) and biomarker responses in the earthworm Eisenia fetida after simultaneous exposure to decabromodiphenyl ether (BDE209). Ecotoxicol. Environ. Saf. 2015, 113, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhang, Y.; Zhang, J.-Y.; Lin, H.; Chen, J.; Hong, H. The toxicity of 2,6-dichlorobenzoquinone on the early life stage of zebrafish: A survey on the endpoints at developmental toxicity, oxidative stress, genotoxicity and cytotoxicity. Environ. Pollut. 2018, 245, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Wu, Y.; Wang, Q.; Wan, J.; Deng, M.; Tu, W. Comparison of toxicokinetics and toxic effects of PFOS and its novel alternative OBS in zebrafish larvae. Chemosphere 2020, 265, 129116. [Google Scholar] [CrossRef]
- Wu, Y.; Huang, J.; Deng, M.; Jin, Y.; Yang, H.; Liu, Y.; Cao, Q.; Mennigen, J.A.; Tu, W. Acute exposure to environmentally relevant concentrations of Chinese PFOS alternative F-53B induces oxidative stress in early developing zebrafish. Chemosphere 2019, 235, 945–951. [Google Scholar] [CrossRef]
- Zhang, C.; Du, Z.; Wang, J.; Wang, J.; Zhou, T.; Li, B.; Zhu, L.; Li, W.; Hou, K. Exposed zebrafish (Danio rerio) to imidazolium-based ionic liquids with different anions and alkyl-chain lengths. Chemosphere 2018, 203, 381–386. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiao, Y.; Li, Z.; Tao, Y.; Yang, Y. Hazards of phthalates (PAEs) exposure: A review of aquatic animal toxicology studies. Sci. Total. Environ. 2021, 771, 145418. [Google Scholar] [CrossRef]
- Shao, Y.; Wang, J.; Du, Z.; Li, B.; Zhu, L.; Wang, J.; Zhang, S. Toxic effect of [Omim]BF 4 and [Omim]Br on antioxidant stress and oxidative damage in earthworms (Eisenia fetida). Environ. Toxicol. Pharmacol. 2018, 60, 37–44. [Google Scholar] [CrossRef]
- Zheng, J.-L.; Yuan, S.-S.; Wu, C.-W.; Li, W.-Y. Chronic waterborne zinc and cadmium exposures induced different responses towards oxidative stress in the liver of zebrafish. Aquat. Toxicol. 2016, 177, 261–268. [Google Scholar] [CrossRef]
- Wu, Y.; Deng, M.; Jin, Y.; Mu, X.; He, X.; Luu, N.-T.; Yang, C.; Tu, W. Uptake and elimination of emerging polyfluoroalkyl substance F-53B in zebrafish larvae: Response of oxidative stress biomarkers. Chemosphere 2018, 215, 182–188. [Google Scholar] [CrossRef]
- Wang, S.; Zhuang, C.; Du, J.; Wu, C.; You, H. The presence of MWCNTs reduces developmental toxicity of PFOS in early life stage of zebrafish. Environ. Pollut. 2017, 222, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Cai, J.; Wang, S.; You, H. Oxidative stress and apotosis to Zebrafish (Danio rerio) embryos exposed to perfluorooctane sulfonate (PFOS) and ZnO nanoparticles. Int. J. Occup. Med. Environ. Health 2017, 30, 213–229. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Wang, Q.; Liu, S.; Zhang, M.; Liu, Y.; Sun, L.; Wu, Y.; Tu, W. Crosstalk between histological alterations, oxidative stress and immune aberrations of the emerging PFOS alternative OBS in developing zebrafish. Sci. Total. Environ. 2021, 774, 145443. [Google Scholar] [CrossRef]
- Yang, H.; Lai, H.; Huang, J.; Sun, L.; Mennigen, J.A.; Wang, Q.; Liu, Y.; Jin, Y.; Tu, W. Polystyrene microplastics decrease F–53B bioaccumulation but induce inflammatory stress in larval zebrafish. Chemosphere 2020, 255, 127040. [Google Scholar] [CrossRef]
- Bian, K.; Harari, Y.; Zhong, M.; Lai, M.; Castro, G.; Weisbrodt, N.; Murad, F. Down-Regulation of Inducible Nitric-Oxide Synthase (NOS-2) During Parasite-Induced Gut Inflammation: A Path to Identify a Selective NOS-2 Inhibitor. Mol. Pharmacol. 2001, 59, 939–947. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Park, M.; Jeong, Y. Purification and characterization of lysozyme from filipino venus, Ruditapes philippinarum. Food Sci. Biotechnol. 2012, 21, 1463–1468. [Google Scholar] [CrossRef]
- Chen, M.; Guo, T.; He, K.; Zhu, L.; Jin, H.; Wang, Q.; Liu, M.; Yang, L. Biotransformation and bioconcentration of 6:2 and 8:2 polyfluoroalkyl phosphate diesters in common carp (prinus carpio): Underestimated ecological risks. Sci. Total. Environ. 2018, 656, 201–208. [Google Scholar] [CrossRef]
- Wang, L.; Zhou, Y.; Ma, X.; Sun, W.; Liu, H. Perfluorooctanoic acid-induced lipid metabolism disorder in SD rat liver and its effect on the expression of fatty acid metabolism-related proteins. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2022, 47, 18–25. [Google Scholar]
- Bonato, M.; Corrà, F.; Bellio, M.; Guidolin, L.; Tallandini, L.; Irato, P.; Santovito, G. PFAS Environmental Pollution and Antioxidant Responses: An Overview of the Impact on Human Field. Int. J. Environ. Res. Public Health 2020, 17, 8020. [Google Scholar] [CrossRef]
- Park, S.; Moon, N.R.; Kang, S.; Kim, D. Ferulic acid and vinpocetine intake improves memory function by enhancing insulin sensitivity and reducing neuroinflammation and oxidative stress in type 2 diabetic animals with induced Alzheimer’s disease. J. Funct. Foods 2022, 95, 105180. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Y.; Li, H.; Huna, R.; Tan, X.; Li, N.; Zhang, Y.; Jiao, X.; Liu, M. C5aR antagonist inhibits LPS-induced inflammation in human gingival fibroblasts via NF-kappaB and MAPK signaling pathways. J. Appl. Oral. Sci. 2023, 31, e20220404. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Shen, L.; Fang, W.; Zhang, X.; Zhong, Y. Perfluorooctanoic acid-induced immunotoxicity via NF-kappa B pathway in zebrafish (Danio rerio) kidney. Fish Shellfish. Immunol. 2021, 113, 9–19. [Google Scholar] [CrossRef]
- Zhang, H.; Fang, W.; Wang, D.; Gao, N.; Ding, Y.; Chen, C. The role of interleukin family in perfluorooctanoic acid (PFOA)-induced immunotoxicity. J. Hazard. Mater. 2014, 280, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Kutikhin, A.G.; E Yuzhalin, A. C-type lectin receptors and RIG-I-like receptors: New points on the oncogenomics map. Cancer Manag. Res. 2012, 4, 39–53. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cyktor, J.C.; Mellors, J.W. Toll-Like Receptor Agonists: Can They Exact a Toll on Human Immunodeficiency Virus Persistence? Clin. Infect. Dis. 2017, 64, 1696–1698. [Google Scholar] [CrossRef]
- Kent, A.; Blander, J.M. Nod-Like Receptors: Key Molecular Switches in the Conundrum of Cancer. Front. Immunol. 2014, 5, 185. [Google Scholar] [CrossRef]
- Jing, L.; Zheng, D.; Sun, X.; Shi, Z. DBDPE upregulates NOD-like receptor signaling to induce NLRP3 inflammasome-mediated HAECs pyroptosis. Environ. Pollut. 2023, 318, 120882. [Google Scholar] [CrossRef]
- Shaw, D.K.; McClure, E.E.; Wang, X.; Pedra, J.H.F. Deviant Behavior: Tick-Borne Pathogens and Inflammasome Signaling. Veter- Sci. 2016, 3, 27. [Google Scholar] [CrossRef]
- Ngo, V.N.; Young, R.M.; Schmitz, R.; Jhavar, S.; Xiao, W.; Lim, K.H.; Kohlhammer, H.; Xu, W.; Yang, Y.; Zhao, H.; et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 2011, 470, 115–119. [Google Scholar] [CrossRef]
- Chang, M.X.; Wang, Y.P.; Nie, P. Zebrafish peptidoglycan recognition protein SC (zfPGRP-SC) mediates multiple intracellular signaling pathways. Fish Shellfish Immunol. 2009, 26, 264–274. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Mehrpour, O.; Buhrmann, C.; Pourbagher-Shahri, A.M.; Shakibaei, M.; Samarghandian, S. Organophosphorus Compounds and MAPK Signaling Pathways. Int. J. Mol. Sci. 2020, 21, 4258. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Qiu, W.; Zhang, S.; Wang, J.; Yang, X.; Xu, B.; Magnuson, J.T.; Xu, E.G.; Wu, M.; Zheng, C. Poly- and Perfluoroalkyl Substances Induce Immunotoxicity via the TLR Pathway in Zebrafish: Links to Carbon Chain Length. Environ. Sci. Technol. 2023, 15, 6139–6149. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Zhou, B. The Role of Nrf2 and MAPK Pathways in PFOS-Induced Oxidative Stress in Zebrafish Embryos. Toxicol. Sci. 2010, 115, 391–400. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Ren, Z.; Chen, M. Immunotoxicity and Transcriptome Analyses of Zebrafish (Danio rerio) Embryos Exposed to 6:2 FTSA. Toxics 2023, 11, 459. https://doi.org/10.3390/toxics11050459
Zhang J, Ren Z, Chen M. Immunotoxicity and Transcriptome Analyses of Zebrafish (Danio rerio) Embryos Exposed to 6:2 FTSA. Toxics. 2023; 11(5):459. https://doi.org/10.3390/toxics11050459
Chicago/Turabian StyleZhang, Jing, Zongming Ren, and Meng Chen. 2023. "Immunotoxicity and Transcriptome Analyses of Zebrafish (Danio rerio) Embryos Exposed to 6:2 FTSA" Toxics 11, no. 5: 459. https://doi.org/10.3390/toxics11050459
APA StyleZhang, J., Ren, Z., & Chen, M. (2023). Immunotoxicity and Transcriptome Analyses of Zebrafish (Danio rerio) Embryos Exposed to 6:2 FTSA. Toxics, 11(5), 459. https://doi.org/10.3390/toxics11050459





