A Review of Common Cyanotoxins and Their Effects on Fish
Abstract
1. Introduction
2. Spatial Distribution of Common Cyanotoxins in Water Bodies
3. Factors Controlling Cyanotoxin Production
4. Bioaccumulation of Cyanotoxins in Fish
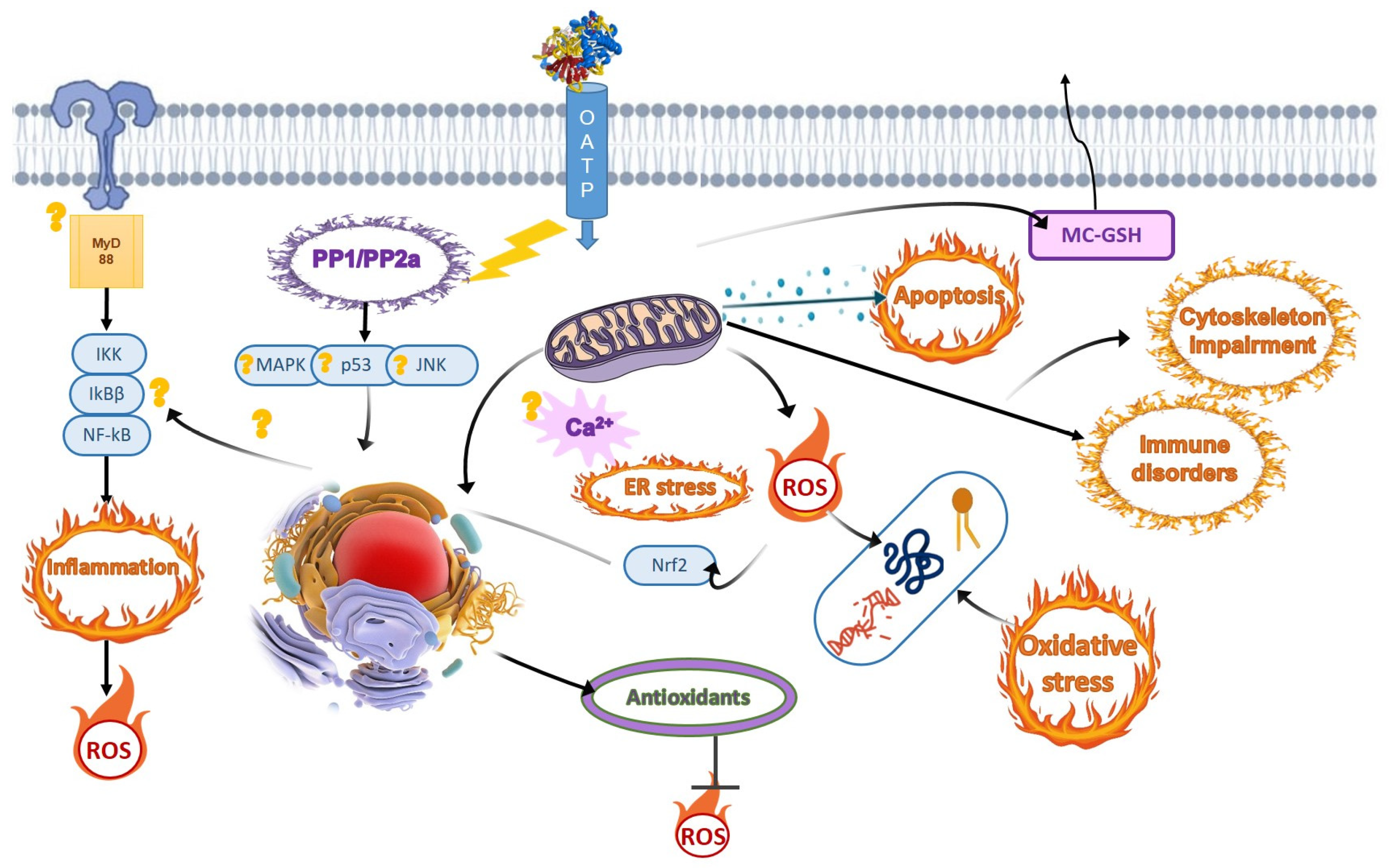
5. Oxidative Stress Is the Dominant Downstream Adverse Effect of Cyanotoxins
6. Immunomodulatory and Inflammatory Effects of Cyanotoxins
7. Endocrine Disruption in Fish Induced by Cyanotoxins
8. Cyanotoxin Exposure Triggers Mitochondrial Damage and Endoplasmic Reticulum Stress in Fish
9. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Paerl, H.W. Mitigating Toxic Planktonic Cyanobacterial Blooms in Aquatic Ecosystems Facing Increasing Anthropogenic and Climatic Pressures. Toxins 2018, 10, 76. [Google Scholar] [CrossRef]
- Vilar, M.; Ferrão-Filho, A. (Eco)Toxicology of Cyanobacteria and Cyanotoxins: From Environmental Dynamics to Adverse Effects. Toxics 2022, 10, 648. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Jones, M.R.; Pinto, E.; Torres, M.A.; Dörr, F.; Mazur-Marzec, H.; Szubert, K.; Tartaglione, L.; Dell’Aversano, C.; Miles, C.O.; Beach, D.G.; et al. CyanoMetDB, a comprehensive public database of secondary metabolites from cyanobacteria. Water Res. 2021, 196, 117017. [Google Scholar] [CrossRef] [PubMed]
- Janssen, E.M. Cyanobacterial peptides beyond microcystins—A review on co-occurrence, toxicity, and challenges for risk assessment. Water Res. 2019, 151, 488–499. [Google Scholar] [CrossRef]
- Rastogi, R.P.; Madamwar, D.; Incharoensakdi, A. Bloom Dynamics of Cyanobacteria and Their Toxins: Environmental Health Impacts and Mitigation Strategies. Front. Microbiol. 2015, 6, 1254. [Google Scholar] [CrossRef]
- Onyango, D.M.; Orina, P.S.; Ramkat, R.C.; Kowenje, C.; Githukia, C.M.; Lusweti, D.; Lung’ayia, H.B.O. Review of current state of knowledge of microcystin and its impacts on fish in Lake Victoria. Lakes Reserv. 2020, 25, 350–361. [Google Scholar] [CrossRef]
- Faassen, E.J.; Lurling, M. Occurrence of the microcystins MC-LW and MC-LF in Dutch surface waters and their contribution to total microcystin toxicity. Mar. Drugs 2013, 11, 2643–2654. [Google Scholar] [CrossRef]
- Bi, X.; Dai, W.; Wang, X.; Dong, S.; Zhang, S.; Zhang, D.; Wu, M. Microcystins distribution, bioaccumulation, and Microcystis genotype succession in a fish culture pond. Sci. Total Environ. 2019, 688, 380–388. [Google Scholar] [CrossRef]
- Malbrouck, C.; Trausch, G.; Devos, P.; Kestemont, P. Effect of microcystin-LR on protein phosphatase activity in fed and fasted juvenile goldfish Carassius auratus. Toxicon 2004, 43, 295–301. [Google Scholar] [CrossRef]
- Pearson, L.; Mihali, T.; Moffitt, M.; Kellmann, R.; Neilan, B. On the chemistry, toxicology and genetics of the cyanobacterial toxins, microcystin, nodularin, saxitoxin and cylindrospermopsin. Mar. Drugs. 2010, 8, 1650–1680. [Google Scholar] [CrossRef] [PubMed]
- Svirčev, Z.; Lalić, D.; Bojadžija Savić, G.; Tokodi, N.; Drobac Backović, D.; Chen, L.; Meriluoto, J.; Codd, G.A. Global geographical and historical overview of cyanotoxin distribution and cyanobacterial poisonings. Arch. Toxicol. 2019, 93, 2429–2481. [Google Scholar] [CrossRef]
- Liyanage, H.M.; Arachchi, D.N.; Abeysekara, T.; Guneratne, L. Toxicology of freshwater cyanobacteria. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2016, 34, 137–168. [Google Scholar] [CrossRef] [PubMed]
- Molica, R.J.R.; Oliveira, E.J.A.; Carvalho, P.V.V.C.; Costa, A.N.S.F.; Cunha, M.C.C.; Melo, G.L.; Azevedo, S.M.F.O. Occurrence of saxitoxins and an anatoxin-a(s)-like anticholinesterase in a Brazilian drinking water supply. Harmful Algae 2005, 4, 743–753. [Google Scholar] [CrossRef]
- Cirés, S.; Ballot, A. A review of the phylogeny, ecology and toxin production of bloom-forming Aphanizomenon spp. and related species within the Nostocales (cyanobacteria). Harmful Algae 2016, 54, 21–43. [Google Scholar] [CrossRef]
- Fiore, M.F.; de Lima, S.T.; Carmichael, W.W.; McKinnie, S.M.K.; Chekan, J.R.; Moore, B.S. Guanitoxin, Re-Naming a Cyanobacterial Organophosphate Toxin. Harmful Algae 2020, 92, 101737. [Google Scholar] [CrossRef]
- Durán-Riveroll, L.; Cembella, A. Guanidinium Toxins and Their Interactions with Voltage-Gated Sodium Ion Channels. Mar. Drugs 2017, 15, 303. [Google Scholar] [CrossRef]
- Rzymski, P.; Poniedziałek, B. In search of environmental role of cylindrospermopsin: A review on global distribution and ecology of its producers. Water Res. 2014, 66, 320–337. [Google Scholar] [CrossRef]
- Dörr, F.A.; Pinto, E.; Soares, R.M.; Azevedo, S.M.F.O. Microcystins in South American Aquatic Ecosystems: Occurrence, Toxicity and Toxicological Assays. Toxicon 2010, 56, 1247–1256. [Google Scholar] [CrossRef]
- Meriluoto, J.; Blaha, L.; Bojadzija, G.; Bormans, M.; Brient, L.; Codd, G.A.; Drobac, D.; Faassen, E.J.; Fastner, J.; Hiskia, A.; et al. Toxic cyanobacteria and cyanotoxins in European waters—Recent progress achieved through the CYANOCOST Action and challenges for further research. Adv. Oceanogr. Limnol. 2017, 8, 144–161. 18p. [Google Scholar] [CrossRef]
- Kurmayer, R.; Christiansen, G.; Fastner, J.; Borner, T. Abundance of active and inactive microcystin genotypes in populations of the toxic cyanobacterium Planktothrix spp. Environ. Microbiol. 2004, 6, 831–841. [Google Scholar] [CrossRef]
- Rantala, A.; Rajaniemi-Wacklin, P.; Lyra, C.; Lepistö, L.; Rintala, J.; Mankiewicz-Boczek, J.; Sivonen, K. Detection of microcystin-producing cyanobacteria in Finnish lakes with genus-specific microcystin synthetase gene E (mcyE) PCR and associations with environmental factors. Appl Environ Microbiol. 2006, 72, 6101–6110. [Google Scholar] [CrossRef]
- Lopes, V.R.; Ramos, V.; Martins, A.; Sousa, M.; Welker, M.; Antunes, A.; Vasconcelos, V.M. Phylogenetic, chemical and morphological diversity of cyanobacteria from Portuguese temperate estuaries. Mar. Environ. Res. 2012, 73, 7–16. [Google Scholar] [CrossRef]
- Da S Ferrão-Filho, A.; Soares, M.C.S.; de Freitas Magalhães, V.; Azevedo, S.M.F.O. Biomonitoring of Cyanotoxins in Two Tropical Reservoirs by Cladoceran Toxicity Bioassays. Ecotoxicol. Environ. Saf. 2009, 72, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Horyn, O.; Budzyńska, A.; Jurczak, T.; Kokociński, M.; Niedzielski, P.; Klimaszyk, P.; Falfushynska, H. A report of Cylindrospermopsis raciborskii and other cyanobacteria in the water reservoirs of power plants in Ukraine. Environ. Sci. Pollut. Res. Int. 2018, 25, 15245–15252. [Google Scholar] [CrossRef]
- Mohamed, Z.A.; Al Shehri, A.M. Microcystin-producing blooms of Anabaenopsis arnoldi in a potable mountain lake in Saudi Arabia. FEMS Microbiol. Ecol. 2009, 69, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Ge, S.; Qiao, X.; Zhao, X.; Li, X.; Liu, Y. Microcystin in source water: Pollution characteristics and human health risk assessment. RSC Adv. 2021, 11, 6415–6422. [Google Scholar] [CrossRef] [PubMed]
- Laureano-Rosario, A.E.; McFarland, M.; Bradshaw, D.J., 2nd; Metz, J.; Brewton, R.A.; Pitts, T.; Perricone, C.; Schreiber, S.; Stockley, N.; Wang, G.; et al. Dynamics of microcystins and saxitoxin in the Indian River Lagoon, Florida. Harmful Algae. 2021, 103, 102012. [Google Scholar] [CrossRef]
- Kotak, B.G.; Zurawell, R.W. Cyanobacterial toxins in Canadian freshwaters: A review. Lake Reserv. Manag. 2007, 23, 109–122. [Google Scholar] [CrossRef]
- Ballot, A.; Sandvik, M.; Rundberget, T.; Owen Miles, C. Diversity of cyanobacteria and cyanotoxins in Hartbeespoort Dam, South Africa. Mar. Freshwater Res. 2014, 65, 175–189. [Google Scholar] [CrossRef]
- Koker, L.; Aakcaalan, R.; Oguz, A.; Gaygusuz, O.; Gurevin, C.; Akat Kose, C.; Gucver, S.; Karaaslan, Y.; Erturk, A.; Albay, M.; et al. Distribution of toxic cyanobacteria and cyanotoxins in Turkish waterbodies. J. Environ. Prot. Ecol. 2017, 18, 425–432. [Google Scholar]
- Wang, J.; Zhang, S.; Mu, X.; Hu, X.; Ma, Y. Research Characteristics on Cyanotoxins in Inland Water: Insights from Bibliometrics. Water. 2022, 14, 667. [Google Scholar] [CrossRef]
- Xiao, M.; Li, M.; Reynolds, C.S. Colony formation in the cyanobacterium Microcystis. Biol. Rev. Camb. Philos. Soc. 2018, 93, 1399–1420. [Google Scholar] [CrossRef]
- Huo, D.; Gan, N.; Geng, R.; Cao, Q.; Song, L.; Yu, G.; Li, R. Cyanobacterial blooms in China: Diversity, distribution, and cyanotoxins. Harmful Algae. 2021, 109, 102106. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Chen, W.; Peng, L.; Wan, N.; Gan, N.; Zhang, X. Distribution and bioaccumulation of microcystins in water columns: A systematic investigation into the environmental fate and the risks associated with microcystins in Meiliang Bay, Lake Taihu. Water Res. 2007, 41, 2853–2864. [Google Scholar] [CrossRef]
- Qin, B.; Zhu, G.; Gao, G.; Zhang, Y.; Li, W.; Paerl, H.W.; Carmichael, W.W. A drinking water crisis in Lake Taihu, China: Linkage to climatic variability and lake management. Environ. Manag. 2010, 45, 105–112. [Google Scholar] [CrossRef]
- Sakai, H.; Hao, A.; Iseri, Y.; Wang, S.; Kuba, T.; Zhang, Z.; Katayama, H. Occurrence and Distribution of Microcystins in Lake Taihu, China. Sci. World J. 2013, 2013, 838176. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Environmental Studies, 2011. Microbial Culture Collection at National Institute for Environmental Studies. Available online: http://mcc.nies.go.jp/ (accessed on 21 December 2022).
- Hodoki, Y.; Ohbayashi, K.; Kobayashi, Y.; Okuda, N.; Nakano, S.-i. Detection and identification of potentially toxic cyanobacteria: Ubiquitous distribution of Microcystis aeruginosa and Cuspidothrix issatschenkoi in Japanese lakes. Harmful Algae 2012, 16, 49–57. [Google Scholar] [CrossRef]
- Watanabe, M. Phytoplankton studies of Lake Kasumigaura (2) On some rare or interesting algae. Bull. Natl. Sci. Mus. Tokyo Ser. B 1985, 11, 137–142. [Google Scholar]
- Watanabe, M. Studies on the planktonic blue-green algae 3. Some Aphanizomenon species in Hokkaido, northern Japan. Bull. Natl. Sci. Mus. Tokyo Ser. B 1991, 17, 141–150. [Google Scholar]
- Willén, E.; Ahlgren, G.; Tilahun, G.; Spoof, L.; Neffling, M.-R.; Meriluoto, J. Cyanotoxin production in seven Ethiopian Rift Valley lakes. Inland Waters 2011, 1, 81–91. [Google Scholar] [CrossRef]
- Oberholster, P.J.; Myburgh, J.G.; Govender, D.; Bengis, R.; Botha, A.M. Identification of toxigenic Microcystis strains after incidents of wild animal mortalities in the Kruger National Park, South Africa. Ecotoxicol. Environ. Saf. 2009, 72, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xu, C.; Liu, Y.; Jeppesen, E.; Svenning, J.C.; Wu, J.; Zhang, W.; Zhou, T.; Wang, P.; Nangombe, S.; et al. From unusual suspect to serial killer: Cyanotoxins boosted by climate change may jeopardize megafauna. Innovation 2021, 2, 100092. [Google Scholar] [CrossRef] [PubMed]
- Preussel, K.; Stuken, A.; Wiedner, C.; Chorus, I.; Fastner, J. First report on cylindrospermopsin producing Aphanizomenon flos-aquae (Cyanobacteria) isolated from two German lakes. Toxicon 2006, 47, 156–162. [Google Scholar] [CrossRef]
- Fastner, J.; Rücker, J.; Stüken, A.; Preussel, K.; Nixdorf, B.; Chorus, I.; Köhler, A.; Wiedner, C. Occurrence of the cyanobacterial toxin cylindrospermopsin in northeast Germany. Environ. Toxicol. 2007, 22, 26–32. [Google Scholar] [CrossRef]
- Bláhová, L.; Babica, P.; Adamovský, O.; Kohoutek, J.; Maršálek, B.; Bláha, L. Analyses of cyanobacterial toxins (microcystins, cylindrospermopsin) in the reservoirs of the Czech Republic and evaluation of health risks. Environ. Chem. Lett. 2008, 6, 223–227. [Google Scholar] [CrossRef]
- Brient, L.; Lengronne, M.; Bormans, M.; Fastner, J. First occurrence of cylindrospermopsin in freshwater in France. Environ. Toxicol. 2009, 24, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Messineo, V.; Melchiorre, S.; Di Corcia, A.; Gallo, P.; Bruno, M. Seasonal succession of Cylindrospermopsis raciborskii and Aphanizomenon ovalisporum blooms with cylindrospermopsin occurrence in the volcanic Lake Albano, Central Italy. Environ. Toxicol. 2010, 25, 18–27. [Google Scholar]
- Cirés, S.; Wörmer, L.; Ballot, A.; Agha, R.; Wiedner, C.; Velázquez, D.; Casero, M.C.; Quesada, A. Phylogeography of cylindrospermopsin and paralytic shellfish toxin-producing Nostocales cyanobacteria from Mediterranean Europe (Spain). Appl. Environ. Microbiol. 2014, 80, 1359–1370. [Google Scholar] [CrossRef] [PubMed]
- Scarlett, K.R.; Kim, S.; Lovin, L.M.; Chatterjee, S.; Scott, J.T.; Brooks, B.W. Global scanning of cylindrospermopsin: Critical review and analysis of aquatic occurrence, bioaccumulation, toxicity and health hazards. Sci. Total Environ. 2020, 738, 139807. [Google Scholar] [CrossRef]
- Messineo, V.; Bogialli, S.; Melchiorre, S.; Sechi, N.; Luglie, A.; Casiddu, P.; Mariani, M.A.; Padedda, B.M.; Di Corcia, A.; Mazza, R.; et al. Cyanobacterial toxins in Italian freshwaters. Limnologica 2009, 39, 95–106. [Google Scholar] [CrossRef]
- Hercog, K.; Maisanaba, S.; Filipič, M.; Jos, Á.; Cameán, A.M.; Žegura, B. Genotoxic potential of the binary mixture of cyanotoxins microcystin-LR and cylindrospermopsin. Chemosphere 2017, 189, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Falfushynska, H.; Horyn, O.; Osypenko, I.; Rzymski, P.; Wejnerowski, Ł.; Dziuba, M.K.; Sokolova, I.M. Multibiomarker-based assessment of toxicity of central European strains of filamentous cyanobacteria Aphanizomenon gracile and Raphidiopsis raciborskii to zebrafish Danio rerio. Water Res. 2021, 194, 116923. [Google Scholar] [CrossRef] [PubMed]
- Jia, N.; Wang, Y.; Guan, Y.; Chen, Y.; Li, R.; Yu, G. Occurrence of Raphidiopsis raciborskii blooms in cool waters: Synergistic effects of nitrogen availability and ecotypes with adaptation to low temperature. Environ. Pollut. 2021, 270, 116070. [Google Scholar] [CrossRef]
- Bittencourt-Oliveira, M.d.C.; Piccin-Santos, V.; Moura, A.N.; Aragão-Tavares, N.K.C.; Cordeiro-Araújo, M.K. Cyanobacteria, Microcystins and Cylindrospermopsin in Public Drinking Supply Reservoirs of Brazil. An. Acad. Bras. Cienc. 2014, 86, 297–310. [Google Scholar] [CrossRef]
- Hoff-Risseti, C.; Dörr, F.A.; Schaker, P.D.C.; Pinto, E.; Werner, V.R.; Fiore, M.F. Cylindrospermopsin and Saxitoxin Synthetase Genes in Cylindrospermopsis Raciborskii Strains from Brazilian Freshwater. PLoS ONE 2013, 8, e74238. [Google Scholar] [CrossRef]
- Rzymski, P.; Poniedziałek, B. Blue-green algae blooms: Environmental and health consequences. In Eutrophication. Causes, Economic Implications and Future Challenges; Lambert, A., Roux, C., Eds.; Nova Science Publishers: New York, NY, USA, 2014; pp. 155–181. [Google Scholar]
- Mantzouki, E.; Lürling, M.; Fastner, J.; De Senerpont Domis, L.; Wilk-Woźniak, E.; Koreivienė, J.; Seelen, L.; Teurlincx, S.; Verstijnen, Y.; Krztoń, W.; et al. Temperature Effects Explain Continental Scale Distribution of Cyanobacterial Toxins. Toxins 2018, 10, 156. [Google Scholar] [CrossRef]
- Scherer, P.I.; Raeder, U.; Geist, J.; Zwirglmaier, K. Influence of temperature, mixing, and addition of microcystin-LR on microcystin gene expression in Microcystis aeruginosa. Microbiol. Open 2016, 6, e00393. [Google Scholar] [CrossRef]
- Wejnerowski, Ł.; Rzymski, P.; Kokociński, M.; Meriluoto, J. The Structure and Toxicity of Winter Cyanobacterial Bloom in a Eutrophic Lake of the Temperate Zone. Ecotoxicology 2018, 27, 752–760. [Google Scholar] [CrossRef]
- Briand, E.; Yepremian, C.; Humbert, J.F.; Quiblier, C. Competition between microcystin- and non-microcystin producing Planktothrix agardhii (cyanobacteria) strains under different environmental conditions. Environ. Microbiol. 2008, 10, 3337–3348. [Google Scholar] [CrossRef]
- Walls, J.T.; Wyatt, K.H.; Doll, J.C.; Rubenstein, E.M.; Rober, A.R. Hot and toxic: Temperature regulates microcystin release from cyanobacteria. Sci. Total Environ. 2018, 610–611, 786–795. [Google Scholar] [CrossRef]
- Harke, M.J.; Gobler, C.J. Daily transcriptome changes reveal the role of nitrogen in controlling microcystin synthesis and nutrient transport in the toxic cyanobacterium, Microcystis aeruginosa. BMC Genom. 2015, 16, 1068. [Google Scholar] [CrossRef]
- Gobler, C.J.; Burkholder, J.M.; Davis, T.W.; Harke, M.J.; Johengen, T.; Stow, C.A.; Van de Waal, D.B. The dual role of nitrogen supply in controlling the growth and toxicity of cyanobacterial blooms. Harmful Algae. 2016, 54, 87–97. [Google Scholar] [CrossRef]
- Savadova-Ratkus, K.; Mazur-Marzec, H.; Karosienė, J.; Kasperovičienė, J.; Paškauskas, R.; Vitonytė, I.; Koreivienė, J. Interplay of Nutrients, Temperature, and Competition of Native and Alien Cyanobacteria Species Growth and Cyanotoxin Production in Temperate Lakes. Toxins 2021, 13, 23. [Google Scholar] [CrossRef]
- Posch, T.; Köster, O.; Salcher, M.M.; Pernthaler, J. Harmful filamentouscyanobacteria favoured by reduced water turnover with lake warming. Nat. Clim. Chang. 2012, 2, 809–813. [Google Scholar] [CrossRef]
- Davis, T.W.; Berry, D.L.; Boyer, G.L.; Gobler, C.J. The effects of temperature and nutrients on the growth and dynamics of toxic and non-toxic strains of Microcystis during cyanobacteria blooms. Harmful Algae 2009, 8, 715–725. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Codd, G.A. Co-Occurrence of Cyanobacteria and Cyanotoxins with Other Environmental Health Hazards: Impacts and Implications. Toxins 2020, 12, 629. [Google Scholar] [CrossRef] [PubMed]
- Ceballos–Laita, L.; Calvo–Begueria, L.; Lahoz, J.; Bes, M.-T.; Fillat, M.F.; Peleato, M.-L. γ–lindane increases microcystin synthesis in microcystis aeruginosa pcc7806. Mar. Drugs 2015, 13, 5666–5680. [Google Scholar] [CrossRef]
- Wu, L.; Qiu, Z.; Zhou, Y.; Du, Y.; Liu, C.; Ye, J.; Hu, X. Physiological effects of the herbicide glyphosate on the cyanobacterium microcystis aeruginosa. Aquat. Toxicol. 2016, 178, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Song, Q.; Wang, C.; Zhou, C.; Lu, C.; Zhao, M. Effects of glufosinate on the growth of and microcystin production by Microcystis aeruginosa at environmentally relevant concentrations. Sci. Total Environ. 2017, 575, 513–518. [Google Scholar] [CrossRef]
- Cerbin, S.; Kraak, M.H.S.; de Voogt, P.; Visser, P.M.; van Donk, E. Combined and single effects of pesticide carbaryl and toxic Microcystis aeruginosa on the life history of Daphnia pulicaria. Hydrobiologia 2010, 643, 129–138. [Google Scholar] [CrossRef]
- Facey, J.A.; Apte, S.C.; Mitrovic, S.M. A review of the effect of trace metals on freshwater cyanobacterial growth and toxin production. Toxins 2019, 11, 643. [Google Scholar] [CrossRef]
- Downs, T.M.; Schallenberg, M.; Burns, C.W. Responses of Lake Phytoplankton to Micronutrient Enrichment: A Study in Two New Zealand Lakes and an Analysis of Published Data. Aquat. Sci. 2008, 70, 347–360. [Google Scholar] [CrossRef]
- The State of World Fisheries and Aquaculture. 2020. Available online: https://www.fao.org/documents/card/en/c/ca9229en (accessed on 22 January 2023).
- Chia, M.A.; Abdulwahab, R.; Ameh, I.; Balogun, J.K.; Auta, J. Farmed Tilapia as an Exposure Route to Microcystins in Zaria-Nigeria: A Seasonal Investigation. Environ. Pollut. 2021, 271, 116366. [Google Scholar] [CrossRef] [PubMed]
- Jing, M.; Lin, D.; Lin, J.; Li, Q.; Yan, H.; Feng, X. Mercury, Microcystins and Omega-3 Polyunsaturated Fatty Acids in Farmed Fish in Eutrophic Reservoir: Risk and Benefit Assessment. Environ. Pollut. 2021, 270, 116047. [Google Scholar] [CrossRef]
- Ferrão-Filho, A.D.S.; Kozlowsky-Suzuki, B. Cyanotoxins: Bioaccumulation and effects on aquatic animals. Mar. Drugs 2011, 9, 2729–2772. [Google Scholar] [CrossRef]
- Ibelings, B.W.; Bruning, K.; Jonge, J.; Wolfstein, K.; Dionisio, L.M.; Postma, J.; Burger, T. Distribuition of microcystins in a lake foodweb: No evidence for biomagnification. Microb. Ecol. 2005, 49, 87–500. [Google Scholar] [CrossRef]
- Zhang, D.; Xie, P.; Liu, Y.; Qiu, T. Transfer, distribution and bioaccumulation of microcystins in the aquatic food web in Lake Taihu, China, with potential risks to human health. Sci. Total Environ. 2009, 407, 2191–2199. [Google Scholar] [CrossRef] [PubMed]
- Dyble, J.; Gossiaux, D.; Landrum, P.; Kashian, D.R.; Pothoven, S. A kinetic study of accumulation and elimination of microcystin-LR in yellow perch (Perca flavescens) tissue and implications for human fish consumption. Mar. Drugs. 2011, 9, 2553–2571. [Google Scholar] [CrossRef] [PubMed]
- Shahmohamadloo, R.S.; Ortiz Almirall, X.; Simmons, D.B.D.; Poirier, D.G.; Bhavsar, S.P.; Sibley, P.K. Fish tissue accumulation and proteomic response to microcystins is species-dependent. Chemosphere 2022, 287 Pt 1, 132028. [Google Scholar] [CrossRef]
- Greer, B.; Meneely, J.P.; Elliott, C.T. Uptake and accumulation of Microcystin-LR based on exposure through drinking water: An animal model assessing the human health risk. Sci. Rep. 2018, 8, 4913. [Google Scholar] [CrossRef]
- Cazenave, J.; Wunderlin, D.A.; de Los Angeles Bistoni, M.; Amé, M.V.; Krause, E.; Pflugmacher, S.; Wiegand, C. Uptake, tissue distribution and accumulation of microcystin-RR in Corydoras paleatus, Jenynsia multidentata and Odontesthes bonariensis. A field and laboratory study. Aquat. Toxicol. 2005, 75, 178–190. [Google Scholar] [CrossRef] [PubMed]
- Ernst, B.; Hoeger, S.J.; O’Brien, E.; Dietrich, D.R. Oral toxicity of the microcystin containing cyanobacterium Planktothrix rubescens in European whitefish (Coregonus lavaretus). Aquat. Toxicol. 2006, 79, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Papadimitriou, T.; Kagalou, I.; Stalikas, C.; Pilidis, G.; Leonardos, I.D. Assessment of microcystins distribution and biomagnification in tissues of the aquatic food web compartments from a shallow lake and potential risks for public health. Ecotoxicology 2012, 21, 1155–1166. [Google Scholar] [CrossRef]
- Fischer, W.J.; Altheimer, S.; Cattori, V.; Meier, P.J.; Dietrich, D.R.; Hagenbuch, B. Organic anion transporting polypeptides expressed in liver and brain mediate uptake of microcystin. Toxicol. Appl. Pharmacol. 2005, 203, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Feurstein, D.; Holst, K.; Fischer, A.; Dietrich, D.R. Oatp-associated uptake and toxicity of microcystins in primary murine whole brain cells. Toxicol. Appl. Pharmacol. 2009, 234, 247–255. [Google Scholar] [CrossRef]
- Lu, H.; Choudhuri, S.; Ogura, K.; Csanaky, I.L.; Lei, X.; Cheng, X.; Song, P.Z.; Klaassen, C.D. Characterization of organic anion transporting polypeptide 1b2-null mice: Essential role in hepatic uptake/toxicity of phalloidin and microcystin-LR. Toxicol. Sci. 2008, 103, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Steiner, K.; Hagenbuch, B.; Dietrich, D.R. Molecular cloning and functional characterization of a rainbow trout liver Oatp. Toxicol. App. Pharmacol. 2014, 280, 534–542. [Google Scholar] [CrossRef]
- Steiner, K.; Zimmermann, L.; Hagenbuch, B.; Dietrich, D. Zebrafish Oatp-mediated transport of microcystin congeners. Arch. Toxicol. 2016, 90, 1129–1139. [Google Scholar] [CrossRef]
- Saker, M.L.; Eaglesham, G.K. The accumulation of cylindrospermopsin from the cyanobacterium Cylindrospermopsis raciborskii in tissues of the Redclaw crayfish Cherax quadricarinatus. Toxicon 1999, 37, 1065–1077. [Google Scholar] [CrossRef]
- Robinson, N.A.; Pace, J.G.; Matson, C.F.; Miura, G.A.; Lawrence, W.B. Tissue distribution, excretion and hepatic biotransformation of microcystin-LR in mice. J. Pharmacol. Exp. Ther. 1991, 256, 176–182. [Google Scholar]
- Metcalf, J.S.; Beattie, K.A.; Pflugmacher, S.; Codd, G.A. Immuno-crossreactivity and toxicity assessment of conjugation products of the cyanobacterial toxin, microcystin-LR. FEMS Microbiol. Lett. 2000, 189, 155–158. [Google Scholar] [CrossRef]
- Amado, L.L.; Monserrat, J.M. Oxidative stress generation by microcystins in aquatic animals: Why and how. Environ. Int. 2010, 36, 226–235. [Google Scholar] [CrossRef]
- Martins, N.D.; Yunes, J.S.; Monteiro, D.A.; Rantin, F.T.; Kalinin, A.L. Microcystin-LR Leads to Oxidative Damage and Alterations in Antioxidant Defense System in Liver and Gills of Brycon Amazonicus (SPIX & AGASSIZ, 1829). Toxicon 2017, 139, 109–116. [Google Scholar]
- Li, X.; Liu, Y.; Song, L.; Liu, J. Responses of antioxidant systems in the hepatocytes of common carp (Cyprinus carpio L.) to the toxicity of microcystin-LR. Toxicon 2003, 42, 85–89. [Google Scholar] [CrossRef]
- Jiang, J.; Gu, X.; Song, R.; Zhang, Q.; Geng, J.; Wang, X.; Yang, L. Time-dependent oxidative stress and histopathological changes in Cyprinus carpio L. exposed to microcystin-LR. Ecotoxicology 2011, 20, 1000–1009. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Shan, Z.; Xu, W.; Wang, X.; Zhou, J.; Kong, D.; Xu, J. Microcystin-LR induced reactive oxygen species mediate cytoskeletal disruption and apoptosis of hepatocytes in Cyprinus carpio L. PLoS ONE 2013, 8, e84768. [Google Scholar] [CrossRef]
- Wu, Y.-L.; He, Y.; Shi, J.-J.; Zheng, T.-X.; Lin, X.-J.; Lin, X. Microcystin-LR Promotes Necroptosis in Primary Mouse Hepatocytes by Overproducing Reactive Oxygen Species. Toxicol. Appl. Pharmacol. 2019, 377, 114626. [Google Scholar] [CrossRef] [PubMed]
- Falfushynska, H.; Horyn, O.; Brzozowska, A.; Fedoruk, O.; Buyak, B.; Poznansky, D.; Poniedziałek, B.; Kokociński, M.; Rzymski, P. Is the presence of Central European strains of Raphidiopsis (Cylindrospermopsis) raciborskii a threat to a freshwater fish? An in vitro toxicological study in common carp cells. Aquat. Toxicol. 2019, 206, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol. 2006, 141, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Prieto, A.I.; Jos, A.; Pichardo, S.; Moreno, I.; Cameán, A.M. Differential oxidative stress responses to microcystins LR and RR in intraperitoneally exposed tilapia fish (Oreochromis sp.). Aquat. Toxicol. 2006, 77, 314–321. [Google Scholar] [CrossRef]
- Evans, D.M.; Hughes, J.; Jones, L.F.; Murphy, P.J.; Falfushynska, H.; Horyn, O.; Sokolova, I.M.; Christensen, J.; Coles, S.J.; Rzymski, P. Elucidating cylindrospermopsin toxicity via synthetic analogues: An in vitro approach. Chemosphere 2019, 234, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Atencio, L.; Moreno, I.; Jos, A.; Pichardo, S.; Moyano, R.; Blanco, A.; Cameán, A.M. Dose-dependent antioxidant responses and pathological changes in tenca (Tinca tinca) after acute oral exposure to Microcystis under laboratory conditions. Toxicon 2008, 52, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, D.; Fan, Z.; Li, J.; Gao, L.; Wang, Y.; Wang, L. Microcystin-LR induces ferroptosis in intestine of common carp (Cyprinus carpio). Ecotoxicol. Environ. Saf. 2021, 223, 112610. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Yang, W.; Chen, Y.; Yang, Z. Effect of purified microcystin on oxidative stress of silver carp Hypophthalmichthys molitrix larvae under different ammonia concentrations. Biochem. Syst. Ecol. 2011, 39, 536–543. [Google Scholar] [CrossRef]
- Li, L.; Xie, P.; Guo, L.G. Antioxidant response in liver of the phytoplanktivorous bighead carp (Aristichthys nobilis) intraperitoneally-injected with extracted microcystins. Fish Physiol. Biochem. 2010, 36, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Paskerová, H.; Hilscherová, K.; Bláha, L. Oxidative stress and detoxification biomarker responses in aquatic freshwater vertebrates exposed to microcystins and cyanobacterial biomass. Environ. Sci. Pollut. Res. 2012, 19, 2024–2037. [Google Scholar] [CrossRef]
- Wang, L.; Liang, X.F.; Liao, W.Q.; Lei, L.M.; Han, B.P. Structural and functional characterization of microcystin detoxification-related liver genes in a phytoplanktivorous fish, Nile tilapia (Oreochromis niloticus). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2006, 144, 216–227. [Google Scholar] [CrossRef]
- Fu, J.; Xie, P. The acute effects of microcystin LR on the transcription of nine glutathione S-transferase genes in common carp Cyprinus carpio L. Aquat. Toxicol. 2006, 80, 261–266. [Google Scholar] [CrossRef]
- Wiegand, C.; Pflugmacher, S.; Oberemm, A.; Meems, N.; Beattie, K.A.; Steinberg, C.E.W.; Codd, G.A. Uptake and effects of microcystin-LR on detoxication enzymes of early life stages of the zebra fish (Danio rerio). Environ. Toxicol. 1999, 14, 89–95. [Google Scholar] [CrossRef]
- Malbrouck, C.; Trausch, G.; Devos, P.; Kestemont, P. Hepatic accumulation and effects of microcystin-LR on juvenile goldfish Carassius auratus L. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2003, 135, 39–48. [Google Scholar] [CrossRef]
- Cazenave, J.; Bistoni Mde, L.; Pesce, S.F.; Wunderlin, D.A. Differential detoxification and antioxidant response in diverse organs of Corydoras paleatus experimentally exposed to microcystin-RR. Aquat. Toxicol. 2006, 76, 1–12. [Google Scholar] [CrossRef]
- Cazenave, J.; Bistoni, M.d.l.A.; Zwirnmann, E.; Wunderlin, D.A.; Wiegand, C. Attenuating effects of natural organic matter on microcystin toxicity in zebra fish (Danio rerio) embryos—Benefits and costs of microcystin detoxication. Environ. Toxicol. 2006, 21, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, S.; Welker, M.; Vasconcelos, V.M. Changes in the GST activity of the mussel Mytilus galloprovincialis during exposure and depuration of microcystins. J. Exp. Zool. A Ecol. Genet. Physiol. 2009, 311, 226–230. [Google Scholar] [CrossRef]
- Kondo, F.; Ikai, Y.; Oka, H.; Okumura, M.; Ishikawa, N.; Harada, K.; Matsuura, K.; Murata, H.; Suzuki, M. Formation, characterization, and toxicity of the glutathione and cysteine conjugates of toxic heptapeptide microcystins. Chem. Res. Toxicol. 1992, 5, 591–596. [Google Scholar] [CrossRef]
- He, X.Q.; Chen, M.G.; Lin, G.X.; Ma, Q. Arsenic induces NAD(P)H-quinone oxidoreductase I by disrupting the Nrf2-Keap1-Cul3 complex and recruiting Nrf2-Maf to the antioxidant response element enhancer. J. Biol. Chem. 2006, 281, 23620–23631. [Google Scholar] [CrossRef]
- Kansanen, E.; Kuosmanen, S.M.; Leinonen, H.; Levonen, A. Redox Biology the Keap1-Nrf2 pathway: Mechanisms of activation and dysregulation in cancer. Redox. Biol. 2013, 1, 45–49. [Google Scholar] [CrossRef]
- Gan, N.; Sun, X.; Song, L. Activation of Nrf2 by microcystin-LR provides advantages for liver cancer cell growth. Chem. Res. Toxicol. 2010, 23, 1477–1484. [Google Scholar] [CrossRef]
- Lundqvist, J.; Pekar, H.; Oskarsson, A. Microcystins activate nuclear factor erythroid 2-related factor 2 (Nrf2) in human liver cells in vitro—Implications for an oxidative stress induction by microcystins. Toxicon 2017, 126, 47–50. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.L.; Liu, W.X.; Wen, C.G.; Qian, G.M.; Hu, B.Q.; Jian, S.Q.; Yang, G.; Dong, J. Effect of microcystin on the expression of Nrf2 and its downstream antioxidant genes from Cristaria plicata. Aquat. Toxicol. 2020, 225, 105526. [Google Scholar] [CrossRef] [PubMed]
- Nakano, M.; Nakano, Y.; Sato-Taki, T.; Mori, N.; Kojima, M.; Ohtake, A.; Shirai, M. Toxicity of Microcystis aeruginosa K-139 strain. Microbiol. Immunol. 1989, 33, 787–792. [Google Scholar] [CrossRef]
- Pahan, K.; Sheikh, F.G.; Namboodiri, A.M.; Singh, I. Inhibitors of protein phosphatase 1 and 2A differentially regulate the expression of inducible nitric-oxide synthase in rat astrocytes and macrophages. J. Biol. Chem. 1998, 273, 12219–12226. [Google Scholar] [CrossRef]
- Shaw, G.R.; Seawright, A.A.; Moore, M.R.; Lam, P.K.S. Cylindrospermopsin, a cyanobacterial alkaloid: Evaluation of its toxicological activity. Ther. Drug Monit. 2000, 22, 89–92. [Google Scholar] [CrossRef]
- Qiao, Q.; Liang, H.; Zhang, X. Effect of cyanobacteria on immune function of crucian carp (Carassius auratus) via chronic exposure in diet. Chemosphere 2013, 90, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Song, T.; Wang, L.; Jiang, L.; Zhou, Q.; Wang, W.; Liu, L.; Yang, P.; Zhang, X. Effects of dietary toxic cyanobacteria and ammonia exposure on immune function of blunt snout bream (Megalabrama amblycephala). Fish Shellfish Immun. 2018, 78, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Shu, Y.; Jiang, H.; Yuen, C.N.T.; Wang, W.; He, J.; Zhang, H.; Liu, G.; Wei, L.; Chen, L.; Wu, H. Microcystin-leucine arginine induces skin barrier damage and reduces resistance to pathogenic bacteria in Lithobates catesbeianus tadpoles. Ecotoxicol. Environ. Saf. 2022, 238, 113584. [Google Scholar] [CrossRef]
- Sierosławska, A.; Rymuszka, A.; Bownik, A.; Skowroński, T. The influence of microcystin-LR on fish phagocytic cells. Hum. Exp. Toxicol. 2007, 26, 603–607. [Google Scholar] [CrossRef]
- Rymuszka, A. Microcystin-LR induces cytotoxicity and affects carp immune cells by impairment of their phagocytosis and the organization of the cytoskeleton. J. Appl. Toxicol. 2013, 33, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Palíková, M.; Navrátil, S.; Krejãí, R.; Tûrba, F.; Tichý, F.; Kubala, L.; Marsálek, B.; Bláha, L. Outcomes of Repeated Exposure of the Carp (Cyprinus carpio L.) to Cyanobacteria Extract. Acta Vet. 2004, 73, 259–265. [Google Scholar] [CrossRef]
- Park, H.; Ishihara, D.; Cox, D. Regulation of tyrosine phosphorylation in macrophage phagocytosis and chemotaxis. Arch. Biochem. Biophys. 2011, 510, 101–111. [Google Scholar] [CrossRef]
- Chung, E.Y.; Kim, S.J.; Ma, X.J. Regulation of cytokine production during phagocytosis of apoptotic cells. Cell Res. 2006, 16, 154–161. [Google Scholar] [CrossRef]
- Kujbida, P.; Hatanaka, E.; Campa, A.; Colepicolo, P.; Pinto, E. Effects of microcystins on human polymorphonuclear leukocytes. Biochem. Biophys. Res. Commun. 2006, 341, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Shi, Q.; Wei, W.; Xu, F.; Nie, F.; Yang, H. Effects of microcystin-LR on the immune dysfunction and ultrastructure of hepatopancreas in giant freshwater prawn Macrobrachium rosenbergii. Fish Shellfish Immunol. 2019, 89, 586–594. [Google Scholar] [CrossRef]
- Rymuszka, A.; Adaszek, Ł. Pro- and anti-inflammatory cytokine expression in carp blood and head kidney leukocytes exposed to cyanotoxin stress--an in vitro study. Fish Shellfish Immunol. 2012, 33, 382–388. [Google Scholar] [CrossRef]
- Lin, W.; Guo, H.; Wang, L.; Zhang, D.; Wu, X.; Li, L.; Qiu, Y.; Yang, L.; Li, D.; Tang, R. Parental Transfer of Microcystin-LR-Induced Innate Immune Dysfunction of Zebrafish: A Cross-Generational Study. Environ. Sci. Technol. 2020, 54, 1014–1023. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, Y.; Wang, J.; Wang, L.; Xiang, Z.; Li, D.; Han, X. Microcystin-Leucine Arginine Causes Cytotoxic Effects in Sertoli Cells Resulting in Reproductive Dysfunction in Male Mice. Sci. Rep. 2016, 6, 39238. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Huang, F.; Massey, I.Y.; Wen, C.; Zheng, S.; Xu, S.; Yang, F. Effects of Microcystin-LR on the microstructure and inflammation-related factors of jejunum in mice. Toxins 2019, 11, 482. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Sun, B.; Chang, M.; Liu, Y.; Nie, P. Effects of cyanobacterial toxin microcystin-LR on the transcription levels of immune-related genes in grass carp Ctenopharyngodon idella. Environ. Biol. Fish. 2009, 85, 231–238. [Google Scholar] [CrossRef]
- Liu, G.; Ke, M.; Fan, X.; Zhang, M.; Zhu, Y.; Lu, T.; Sun, L.; Qian, H. Reproductive and endocrine-disrupting toxicity of Microcystis aeruginosa in female zebrafish. Chemosphere 2018, 192, 289–296. [Google Scholar] [CrossRef]
- Liu, W.; Chen, C.; Chen, L.; Wang, L.; Li, J.; Chen, Y.; Jin, J.; Kawan, A.; Zhang, X. Sex-dependent effects of microcystin-LR on hypothalamic-pituitary-gonad axis and gametogenesis of adult zebrafish. Sci. Rep. 2016, 6, 22819. [Google Scholar] [CrossRef]
- Hou, J.; Su, Y.; Lin, W.; Guo, H.; Li, L.; Anderson, D.M.; Li, D.; Tang, R.; Chi, W.; Zhang, X. Estrogenic potency of MC-LR is induced via stimulating steroidogenesis: In vitro and in vivo evidence. Environ. Pollut. 2018, 240, 615–622. [Google Scholar] [CrossRef]
- Wang, L.; Lin, W.; Zha, Q.; Guo, H.; Zhang, D.; Yang, L.; Li, L.; Li, D.; Tang, R. Persistent Exposure to Environmental Levels of Microcystin-LR Disturbs Cortisol Production via Hypothalamic-Pituitary-Interrenal (HPI) Axis and Subsequently Liver Glucose Metabolism in Adult Male Zebrafish (Danio rerio). Toxins 2020, 12, 282. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ying, F.; Chen, Y.; Han, X. Microcystin (-LR) affects hormones level of male mice by damaging hypothalamic-pituitary system. Toxicon 2012, 59, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.-J.; Chien, M.-S.; Wu, F.-J.; Chou, H.-N.; Lee, S.-J. Inhibition of Embryonic Development by Microcystin-LR in Zebrafish, Danio Rerio. Toxicon 2005, 45, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Sun, H.; Xie, P.; Wang, J.; Zhang, G.; Chen, N.; Yan, W.; Li, G. The Role of Apoptosis in MCLR-Induced Developmental Toxicity in Zebrafish Embryos. Aquat. Toxicol. 2014, 149, 25–32. [Google Scholar] [CrossRef]
- Zhan, C.; Zhang, F.; Liu, W.; Zhang, X. Microcystin-LR Promotes Zebrafish (Danio Rerio) Oocyte (in Vivo) Maturation by Activating ERK1/2-MPF Signaling Pathways, and CAMP Is Involved in This Process. Environ. Pollut. 2020, 259, 113843. [Google Scholar] [CrossRef]
- Sumpter, J.P.; Jobling, S. Vitellogenesis as a biomarker for estrogenic contamination of the aquatic environment. Environ. Health Perspect. 1995, 103 (Suppl. 7), 173–178. [Google Scholar] [CrossRef]
- Rogers, E.D.; Henry, T.B.; Twiner, M.J.; Gouffon, J.S.; McPherson, J.T.; Boyer, G.L.; Sayler, G.S.; Wilhelm, S.W. Global gene expression profiling in larval zebrafish exposed to microcystin-LR and microcystis reveals endocrine disrupting effects of Cyanobacteria. Environ. Sci. Technol. 2011, 45, 1962–1969. [Google Scholar] [CrossRef]
- Qiao, Q.; Liu, W.; Wu, K.; Song, T.; Hu, J.; Huang, X.; Wen, J.; Chen, L.; Zhang, X. Female zebrafish (Danio rerio) are more vulnerable than males to microcystin-LR exposure, without exhibiting estrogenic effects. Aquat. Toxicol. 2013, 142–143, 272–282. [Google Scholar] [CrossRef]
- Oziol, L.; Bouaïcha, N. First evidence of estrogenic potential of the cyanobacterial heptotoxins the nodularin-R and the microcystin-LR in cultured mammalian cells. J. Hazard. Mater. 2010, 174, 610–615. [Google Scholar] [CrossRef]
- Sotton, B.; Paris, A.; Le Manach, S.; Blond, A.; Lacroix, G.; Millot, A.; Duval, C.; Huet, H.; Qiao, Q.; Labrut, S.; et al. Metabolic changes in Medaka fish induced by cyanobacterial exposures in mesocosms: An integrative approach combining proteomic and metabolomic analyses. Sci. Rep. 2017, 7, 4051. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Q.; Le Manach, S.; Huet, H.; Duvernois-Berthet, E.; Chaouch, S.; Duval, C.; Sotton, B.; Ponger, L.; Marie, A.; Mathéron, L.; et al. An integrated omic analysis of hepatic alteration in medaka fish chronically exposed to cyanotoxins with possible mechanisms of reproductive toxicity. Environ. Pollut. 2016, 219, 119–131. [Google Scholar] [CrossRef]
- Liu, J.; Hernández, S.E.; Swift, S.; Singhal, N. Estrogenic activity of cylindrospermopsin and anatoxin-a and their oxidative products by FeIII-B*/H2O2. Water Res. 2018, 132, 309–319. [Google Scholar] [CrossRef]
- Gutendorf, B.; Westendorf, J. Comparison of an array of in vitro assays for the assessment of the estrogenic potential of natural and synthetic estrogens, phytoestrogens and xenoestrogens. Toxicology 2001, 166, 79–89. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, C.; Li, Y.; Imam, M.U.; Huang, H.; Liu, H.; Xin, Y.; Zhang, H. Novel Role of ER Stress and Autophagy in Microcystin-LR Induced Apoptosis in Chinese Hamster Ovary Cells. Front. Physiol. 2016, 7, 527. [Google Scholar] [CrossRef] [PubMed]
- Moraes, A.C.N.; Fallah, H.P.; de Magalhães, V.F.; Habibi, H.R. Cylindrospermopsin induces oocyte maturation and disrupts gene expression in zebrafish ovarian follicles. Environ. Toxicol. Pharmacol. 2022, 94, 103915. [Google Scholar] [CrossRef]
- Moraes, A.C.N.; Fallah, H.P.; Magalhães, V.F.; Habibi, H.R. Cylindrospermopsin directly disrupts spermatogenesis in isolated male zebrafish testis. Gen. Comp. Endocrinol. 2021, 313, 113891. [Google Scholar] [CrossRef]
- Chen, C.; Liu, W.; Wang, L.; Li, J.; Chen, Y.; Jin, J.; Atufa, K.; Zhang, X. Pathological damage and immunomodulatory effects of zebrafish exposed to microcystin-LR. Toxicon 2016, 118, 13–20. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, J.; Chen, Y.; Zhu, Y. Microcystin-RR induces apoptosis in fish lymphocytes by generating reactive oxygen species and causing mitochondrial damage. Fish Physiol. Biochem. 2008, 34, 307–312. [Google Scholar] [CrossRef]
- Ding, W.X.; Shen, H.M.; Shen, Y.; Zhu, H.G.; Ong, C.N. Microcystic cyanobacteria causes mitochondrial membrane potential alteration and reactive oxygen species formation in primary cultured rat hepatocytes. Environ. Health. Perspect. 1998, 106, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Ding, W.X.; Nam Ong, C. Role of oxidative stress and mitochondrial changes in cyanobacteria-induced apoptosis and hepatotoxicity. FEMS Microbiol. Lett. 2003, 220, 1–7. [Google Scholar] [CrossRef]
- Fladmark, K.E.; Brustugun, O.T.; Mellgren, G.; Krakstad, C.; Boe, R.; Vintermyr, O.K.; Schulman, H.; Doskeland, S.O. Ca2+/calmodulin-dependent protein kinase II is required for microcystin-induced apoptosis. J. Biol. Chem. 2002, 277, 2804–2811. [Google Scholar] [CrossRef] [PubMed]
- Zhan, C.; Liu, W.; Zhang, F.; Zhang, X. Microcystin-LR triggers different endoplasmic reticulum stress pathways in the liver, ovary, and offspring of zebrafish (Danio rerio). J. Hazard. Mater. 2020, 386, 121939. [Google Scholar] [CrossRef] [PubMed]
- Hetz, C.; Bernasconi, P.; Fisher, J.; Lee, A.H.; Bassik, M.C.; Antonsson, B.; Brandt, G.S.; Iwakoshi, N.N.; Schinzel, A.; Glimcher, L.H.; et al. Proapoptotic BAX and BAK modulate the unfolded protein response by a direct interaction with IRE1alpha. Science 2006, 312, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, H.; Huang, H.; Yuan, L.; Liu, C.; Wang, Y.; Cheng, X.; Zhuang, D.; Xu, M.; Chen, X.; et al. p53-Dependent pathway and the opening of mPTP mediate the apoptosis of co-cultured Sertoli-germ cells induced by microcystin-LR. Environ. Toxicol. 2019, 34, 1074–1084. [Google Scholar] [CrossRef]
- Riedl, S.; Shi, Y. Molecular mechanisms of caspase regulation during apoptosis. Nat. Rev. Mol. Cell Biol. 2004, 5, 897–907. [Google Scholar] [CrossRef]
- Tamura, R.E.; de Vasconcellos, J.F.; Sarkar, D.; Libermann, T.A.; Fisher, P.B.; Zerbini, L.F. GADD45 proteins: Central players in tumorigenesis. Curr. Mol. Med. 2012, 12, 634–651. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Shen, L.; Ye, X.; Zhou, D.; He, Y.; Li, Y.; Ding, Y.; Zhu, W.; Ding, J.; Zhang, H. Neurotoxic Anatoxin-a Can Also Exert Immunotoxicity by the Induction of Apoptosis on Carassius auratus Lymphocytes in vitro When Exposed to Environmentally Relevant Concentrations. Front. Physiol. 2020, 11, 316. [Google Scholar] [CrossRef]
- Leiers, T.; Bihlmayer, A.; Ammon, H.P.; Wahl, M.A. [Ca(2+)](i)- and insulin-stimulating effect of the non-membranepermeable phosphatase-inhibitor microcystin-LR in intact insulin-secreting cells (RINm5F). Br. J. Pharmacol. 2000, 130, 1406–1410. [Google Scholar] [CrossRef]
- Bury, N.; Flik, G.; Eddy, F.; Codd, G. The effects of cyanobacteria and the cyanobacterial toxin microcystin-LR on Ca2+ transport and Na+/K+-ATPase in tilapia gills. J. Exp. Biol. 1996, 199 Pt 6, 1319–1326. [Google Scholar] [CrossRef]
- Nakamura, K.; Bossy-Wetzel, E.; Burns, K.; Fadel, M.P.; Lozyk, M.; Goping, I.S.; Opas, M.; Bleackley, R.C.; Green, D.R.; Michalak, M. Changes in endoplasmic reticulum luminal environment affect cell sensitivity to apoptosis. J. Cell Biol. 2000, 150, 731–740. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Falfushynska, H.; Kasianchuk, N.; Siemens, E.; Henao, E.; Rzymski, P. A Review of Common Cyanotoxins and Their Effects on Fish. Toxics 2023, 11, 118. https://doi.org/10.3390/toxics11020118
Falfushynska H, Kasianchuk N, Siemens E, Henao E, Rzymski P. A Review of Common Cyanotoxins and Their Effects on Fish. Toxics. 2023; 11(2):118. https://doi.org/10.3390/toxics11020118
Chicago/Turabian StyleFalfushynska, Halina, Nadiia Kasianchuk, Eduard Siemens, Eliana Henao, and Piotr Rzymski. 2023. "A Review of Common Cyanotoxins and Their Effects on Fish" Toxics 11, no. 2: 118. https://doi.org/10.3390/toxics11020118
APA StyleFalfushynska, H., Kasianchuk, N., Siemens, E., Henao, E., & Rzymski, P. (2023). A Review of Common Cyanotoxins and Their Effects on Fish. Toxics, 11(2), 118. https://doi.org/10.3390/toxics11020118








