Some Unmodified Household Adsorbents for the Adsorption of Benzalkonium Chloride—A Kinetic and Thermodynamic Case Study for Commercially Available Paper
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Choice of Adsorbent
- Low price and easy availability (commercial sources);
- Good standardization (already ensured by the producer);
- An approximatelly zero value for the 262 nm absorbance of the adsorbent-water mixture, so that it does not interfere with the values provided by BAC;
- A high equilibrium adsorption yield coupled with a low total S-L contact time required until equilibrium.
3.2. Experimental Data and Their Processing Method
3.3. Process Kinetics
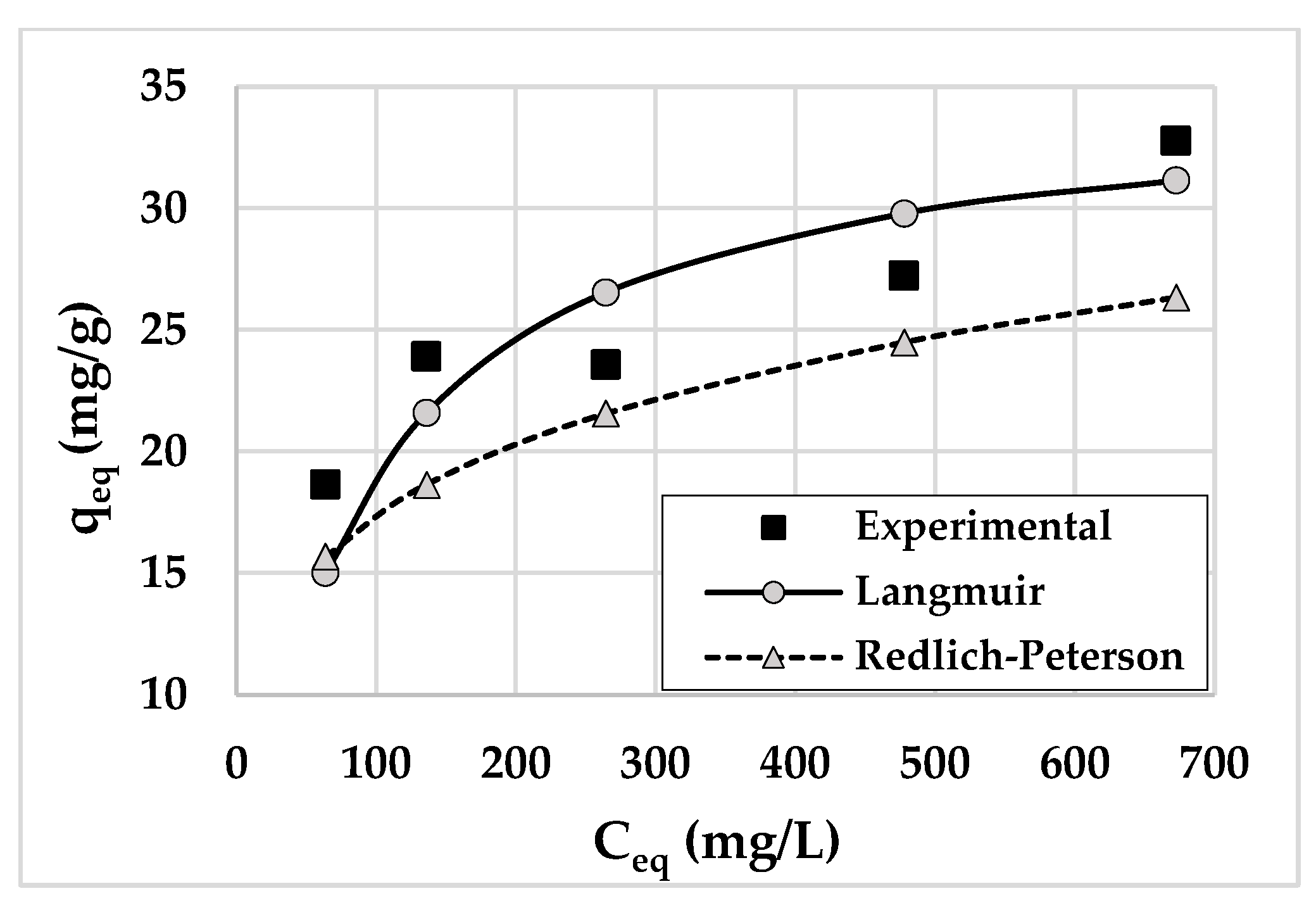
3.4. Adsorption Isotherms and Mechanism
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahuja, K.; Bayas, S. Benzalkonium Chloride Market Size by Type (Benzalkonium Chloride 80%, Benzalkonium Chloride 50%), by Application (Biocide [Water Treatment Chemical, Disinfectant, Preservative], Phase Transfer Catalyst), by End-Use (Food & Beverage, Cosmetics, Polymers & Coatings, Pharmaceuticals, Chemical Industry, Pulp and Paper, Leather & Textile, Oil & Gas), Industry Analysis Report, Regional Outlook, Growth Potential, Price Trend, Competitive Market Share & Forecast, 2021–2027. Global Market Insights; Report ID: GMI3233. 2021. Available online: https://www.gminsights.com/industry-analysis/benzalkonium-chloride-market (accessed on 30 October 2023).
- Zhang, C.; Tezel, U.; Li, K.; Liu, D.; Ren, R.; Du, J.; Pavlostathis, S.G. Evaluation and Modeling of Benzalkonium Chloride Inhibition and Biodegradation in Activated Sludge. Water Res. 2011, 45, 1238–1246. [Google Scholar] [CrossRef] [PubMed]
- Nan, H.; Wang, T.; Wang, W.L.; Wu, Q.Y.; Li, A.; Hu, H.Y. UV/Chlorine as an Advanced Oxidation Process for the Degradation of Benzalkonium Chloride: Synergistic Effect, Transformation Products and Toxicity Evaluation. Water Res. 2017, 114, 246–253. [Google Scholar]
- Abott, T.; Kor-Bicakci, G.; Islam, M.S.; Eskicioglu, C. A Review on the Fate of Legacy and Alternative Antimicrobials and Their Metabolites during Wastewater and Sludge Treatment. Int. J. Mol. Sci. 2020, 21, 9241. [Google Scholar] [CrossRef]
- Kim, S.; Ji, K.; Shin, H.; Park, S.; Kho, Y.; Park, K.; Kim, K.; Choi, K. Occurrences of Benzalkonium Chloride in Streams Near a Pharmaceutical Manufacturing Complex in Korea and Associated Ecological Risk. Chemosphere 2020, 256, 127084. [Google Scholar] [CrossRef]
- Barber, O.W.; Hartmann, E.M. Benzalkonium chloride: A Systematic Review of Its Environmental Entry through Wastewater Treatment, Potential Impact, and Mitigation Strategies. Crit. Rev. Environ. Sci. Technol. 2022, 52, 2691–2719. [Google Scholar] [CrossRef]
- Fuchsman, P.; Fetters, K.; O’Connor, M.; Henning, M.; Brown, L.; Mrdjen, I.; Stanton, K. Ecological Risk Analysis for Benzalkonium Chloride, Benzethonium Chloride, and Chloroxylenol in US Disinfecting and Sanitizing Products. Environ. Toxicol. Chem. 2022, 41, 3095–3115. [Google Scholar] [CrossRef]
- Sreevidya, V.S.; Lenz, K.A.; Svoboda, K.R.; Ma, H. Benzalkonium Chloride, Benzethonium Chloride, and Chloroxylenol—Three Replacement Antimicrobials are More Toxic than Triclosan and Triclocarban in Two Model Organisms. Environ. Pollut. 2018, 235, 814–824. [Google Scholar] [CrossRef] [PubMed]
- Ikisa, K.G.; Banatunde, B.B.; Hart, A.I. Acute Toxicity of Benzalkonium Chloride Mixture with Treated Produced Water to Juveniles of Freshwater Tilapia- Oreochromis niloticus. J. Appl. Sci. Environ. Manag. 2019, 23, 1169–1174. [Google Scholar] [CrossRef]
- Lavorgna, M.; Russo, C.; D’Abrosca, B.; Parrella, A.; Isidori, M. Toxicity and Genotoxicity of the Quaternary Ammonium Compound Benzalkonium Chloride (BAC) Using Daphnia magna and Ceriodaphnia dubia as Model Systems. Environ. Pollut. 2016, 210, 34–39. [Google Scholar] [CrossRef]
- Perez, P.; Fernandez, E.; Beiras, R. Toxicity of Benzalkonium Chloride on Monoalgal Cultures and Natural Assemblages of Marine Phytoplankton. Water Air Soil. Pollut. 2009, 201, 319–330. [Google Scholar] [CrossRef]
- Sousa, B.; Domingues, I.; Nunes, B. A Fish Perspective on SARS-CoV-2: Toxicity of Benzalkonium Chloride on Danio rerio. Environ. Toxicol. Pharmacol. 2023, 102, 104200. [Google Scholar] [CrossRef]
- Benzalkonium Chloride, Safety Data Sheet; Spectrum Chemical Mfg. Corp.: Gardena, CA, USA, 2022; p. 7.
- Benzalkonium Chloride 50%, Safety Data Sheet; Fisher Scientific Company: Fair Lawn, NJ, USA, 2021; pp. 2, 5–6.
- Benzalkonium Chloride 80%, Safety Data Sheet; Laboratoriumdiscounter: Ijmuiden, The Netherlands, 2023; pp. 2, 4–5.
- Tezel, U.; Giles, H.; deRussy, A.; Pavlostathis, S.G. Effect of Counter Ions and Natural Organic Matter on the Toxicity of Benzalkonium Chloride. Proc. Water Environ. Fed. 2009, 17, 621–631. [Google Scholar] [CrossRef]
- Sahoo, T.R.; Prelot, B. Adsorption Processes for the Removal of Contaminants from Wastewater: The Perspective Role of Nanomaterials and Nanotechnology. In Nanomaterials for the Detection and Removal of Wastewater Pollutants; Bonelli, B., Freyria, F.S., Rossetti, I., Sethi, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; Chapter 7; pp. 161–222. [Google Scholar]
- Krstić, V. Role of Zeolite Adsorbent in Water Treatment. In Handbook of Nanomaterials for Wastewater Treatment: Fundamentals and Scale Up Issues; Bhanvase, B., Sonawane, S., Pawade, V., Pandi, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; Chapter 14; pp. 417–481. [Google Scholar]
- Meez, E.; Rahdar, A.; Kyaz, G.Z. Sawdust for the Removal of Heavy Metals from Water: A Review. Molecules 2021, 26, 4318. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.M.; Carr, C.M. Biomass-derived Porous Carbonaceous Materials and Their Composites as Adsorbents for Cationic and Anionic Dyes: A Review. Chemosphere 2021, 265, 129087. [Google Scholar] [CrossRef] [PubMed]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and Its Removal from Aqueous Solution by Adsorption: A Review. Adv. Colloid. Interface Sci. 2014, 209, 172–174. [Google Scholar] [CrossRef] [PubMed]
- Sahmoune, M.N.; Yeddou, A.R. Potential of Sawdust Materials for the Removal of Dyes and Heavy Metals: Examination of Isotherms and Kinetics. Desalin. Water Treat. 2016, 57, 24019–24034. [Google Scholar] [CrossRef]
- Chikri, R.; Elhadiri, N.; Bechanaa, M.; El Maguana, Y. Efficiency of Sawdust as Low-Cost Adsorbent for Dyes Removal. J. Chem. 2020, 2020, 8813420. [Google Scholar] [CrossRef]
- Markovic, R.; Stevanovic, J.; Stevanovic, Z.; Bugarin, M.; Nedeljkovic, D.; Grujic, A.; Stajic-Trosic, J. Using the Low-Cost Waste Materials for Heavy Metals Removal from the Mine Wastewater. Mater. Trans. 2011, 52, 1849–1852. [Google Scholar] [CrossRef]
- Dorri, Y.; Kurien, B.T. Paper Adsorbents Remove Coomassie Blue from Gel Destain and Used Gel Stain in an Environment-Friendly Manner. Methods Mol. Biol. 2018, 1853, 259–268. [Google Scholar]
- Diaz-Linan, M.C.; Lucena, R.; Cardenas, S.; Lopez-Lorente, A.I. Unmodified Cellulose Filter Paper, a Sustainable and Affordable Sorbent for the Isolation of Biogenic Amines from Beer Samples. J. Chromatogr. A 2021, 1651, 462297. [Google Scholar] [CrossRef]
- Nabais, J.M.V.; Laguinhas, C.E.; Román, S. Biomass Novel Adsorbents for Phenol and Mercury Removal. Molecules 2022, 27, 7345. [Google Scholar] [CrossRef] [PubMed]
- Vancea, C.; Mladin, G.; Ciopec, M.; Negrea, A.; Duteanu, N.; Negrea, P.; Mosoarca, G.; Ianasi, C. Arsenic Removal Using Unconventional Material with Iron Content: Batch Adsorption and Column Study. Toxics 2023, 11, 849. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Sang, H.; Zheng, J.; Yang, S.; Gu, Z.; Wu, H.; Wei, Y. Preparation and Density Functional Theory Studies of Aluminosilicate-Based Ceramic Solidified Products for Sr Immobilization. Toxics 2023, 11, 850. [Google Scholar] [CrossRef] [PubMed]
- Tanada, M.; Miyoshi, T.; Nakamura, T.; Tanada, S. Adsorption Removal of Benzalkonium Chloride by Granular Activated Carbon for Medical Waste Water Treatment. Asia Pac. J. Public Health 1991, 5, 27–31. [Google Scholar] [CrossRef]
- Zanini, G.P.; Ovesen, R.G.; Hansen, H.C.B.; Strobel, B.W. Adsorption of the Disinfectant Benzalkonium Chloride on Montmorillonite. Synergistic Effect in Mixture of Molecules with Different Chain Lengths. J. Environ. Manag. 2013, 128, 100–105. [Google Scholar] [CrossRef]
- Ong, L.K.; Soetaredjo, F.E.; Kurniawan, A.; Ayucitra, A.; Liu, J.-C.; Ismadji, S. Investigation on the Montmorillonite Adsorption of Biocidal Compounds Incorporating Thermodynamical-Based Multicomponent Adsorption Isotherm. J. Chem. Eng. 2014, 241, 9–18. [Google Scholar] [CrossRef]
- Ilari, R.; Etcheverry, M.; Waiman, C.V.; Zanini, G.P. A Simple Cation Exchange Model to Assess the Competitive Adsorption between the Herbicide Paraquat and the Biocide Benzalkonium Chloride on Montmorillonite. Colloids Surf. A Physicochem. Eng. Asp. 2021, 611, 125797. [Google Scholar] [CrossRef]
- Paun, I.; Covaliu, C.I.; Pirvu, F.; Paraschiv, G.; Nita-Lazar, M. Benzalkonium Bromide Cationic Surfactant Removal from Wastewater Using Magnetite Nanomaterial. Rev. Chim. 2020, 71, 289–296. [Google Scholar] [CrossRef]
- Turku, I.; Sainio, T.; Paatero, E. Removal of Benzalkonium Chloride by Polymeric Adsorbents. In Proceedings of the Book of Abstracts European Congress of Chemical Engineering (ECCE-6), Copenhagen, Denmark, 16–20 September 2007. [Google Scholar]
- Kim, T.-K.; Jang, M.; Hwang, Y.S. Adsorption of Benzalkonium Chlorides onto Polyethylene Microplastics: Mechanism and Toxicity Evaluation. J. Hazard. Mater. 2022, 426, 128076. [Google Scholar] [CrossRef]
- Kim, T.-K.; Choe, W.-S.; Kim, T.; Chae, S.-H.; Hwang, J.S.; Zoh, K.-D. Adsorption of benzalkonium chlorides onto powdered activated carbon: Mechanisms and detoxification. Environ. Eng. Res. 2022, 27, 210496. [Google Scholar] [CrossRef]
- do Nascimento, D.S.; Etcheverry, M.; Orduz, E.A.; Waiman, C.V.; Zanini, G.P. Adsorption of cationic surfactant as a probe of the montmorillonite surface reactivity in the alginate hydrogel composites. RSC Adv. 2022, 12, 35469–35476. [Google Scholar] [CrossRef] [PubMed]
- Farias, T.; de Ménorval, L.C.; Zajac, J.; Rivera, A. Benzalkonium Chloride and Sulfamethoxazole Adsorption onto Natural Clinoptilolite: Effect of Time, Ionic Strength, pH and Temperature. J. Colloid. Interface Sci. 2011, 363, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Bin, T.; Kulshreshtha, A.K.; Al-Shakhshir, R.; Hem, S.L. Adsorption of Benzalkonium Chloride by Filter Membranes: Mechanisms and Effect of Formulation and Processing Parameters. Pharm. Dev. Technol. 1999, 4, 151–165. [Google Scholar] [CrossRef] [PubMed]
- Penshovska, V.; Atanaskova, E.; Cholakova, I.; Stojanovska, N.A.; Angelovska, V.; Mojsovska, M. Selectopn of Filter for Sterilization Using Process of Membrane Filtration Based on Potential Adsorption of Benzalkonium Chloride on Filter Membranes. In Proceedings of the 7th Conference on Pharmacy (Modern Trends in Pharmacy, Opportunities and Challenges), Ohrid, Macedonia, 5–9 October 2022. [Google Scholar]
- Šĉiban, M.; Klašnja, M.; Škrbić, B. Modified Softwood Sawdust as Adsorbent of Heavy Metal Ions from Water. J. Hazard. Mater. 2006, 136, 266–271. [Google Scholar] [CrossRef]
- Argun, M.E.; Dursun, S.; Ozdemir, C.; Karatas, M. Heavy Metal Adsorption by Modified Oak Sawdust: Thermodynamics and Kinetics. J. Hazard. Mater. 2007, 141, 77–85. [Google Scholar] [CrossRef]
- Ellison, S.L.R.; Barwick, V.J.; Farrant, T.J.D. Practical Statistics for the Analytical Scientist, 2nd ed.; RSC Publishing: Cambridge, UK, 2009; pp. 59–65, 92–108. [Google Scholar]
- Csavdari, A.A. Detailed Kinetics of a Diffusion Driven Adsorption Process. Stud. Univ. Babes-Bolyai Chem. 2023, 68, 179–189. [Google Scholar]
- Joglekar, A.M.; May, A.T. Product Excellence through Design of Experiments. CFW 1987, 32, 857–868. [Google Scholar]
- Rahman, M.M.; Muttakin, M.; Pal, A.; Shafiullah, A.Z.; Saha, B.B. A Statistical Approach to Determine Optimal Models for IUPAC-Classified Adsorption Isotherms. Energies 2019, 12, 4565. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Da’ana, D.A. Guidelines for the Use and Interpretation of Adsorption Isotherm Models: A Review. J. Hazard. Mater. 2020, 393, 122383. [Google Scholar] [CrossRef]
- Dada, A.O.; Olalekan, A.P.; Olatunya, A.M.; Dada, O. Langmuir, Freundlich, Temkin and Dubinin–Radushkevich Isotherms Studies of Equilibrium Sorption of Zn2+ Unto Phosphoric Acid Modified Rice Husk. IOSR-JAC 2012, 3, 38–45. [Google Scholar]
- Serafin, J.; Dziejarski, B. Application of Isotherm Models and Error Functions in Activated Carbon CO2 sorption processes. Microporous Mesoporous Mater. 2023, 354, 112513. [Google Scholar] [CrossRef]
- Lima, E.C.; Hosseini-Bandegharaei, A.; Moreno-Pirajan, J.C.; Anastopoulos, I. A Critical Review of the Estimation of the Thermodynamic Parameters on Adsorption Equilibria. Wrong Use of Equilibrium Constant in the Van’t Hoff Equation for Calculation of Thermodynamic Parameters of Adsorption. J. Mol. Liq. 2019, 273, 425–434. [Google Scholar] [CrossRef]
- Saha, P.; Chowdhury, S. Insight into Adsorption Thermodynamics. In Thermodynamics; Tadashi, M., Ed.; INTECH: Rijeka, Croatia, 2011; Chapter 16; pp. 349–364. [Google Scholar]

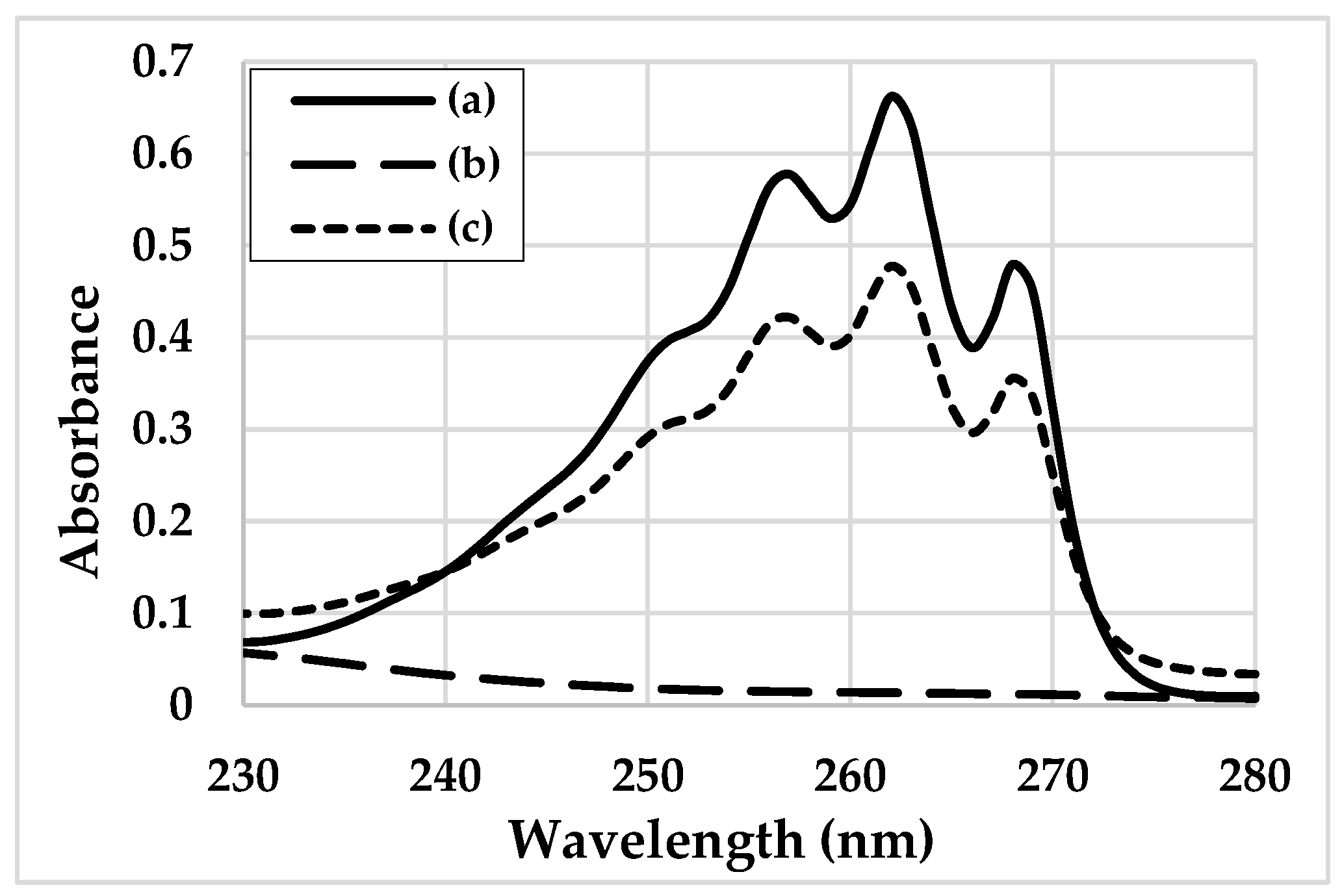


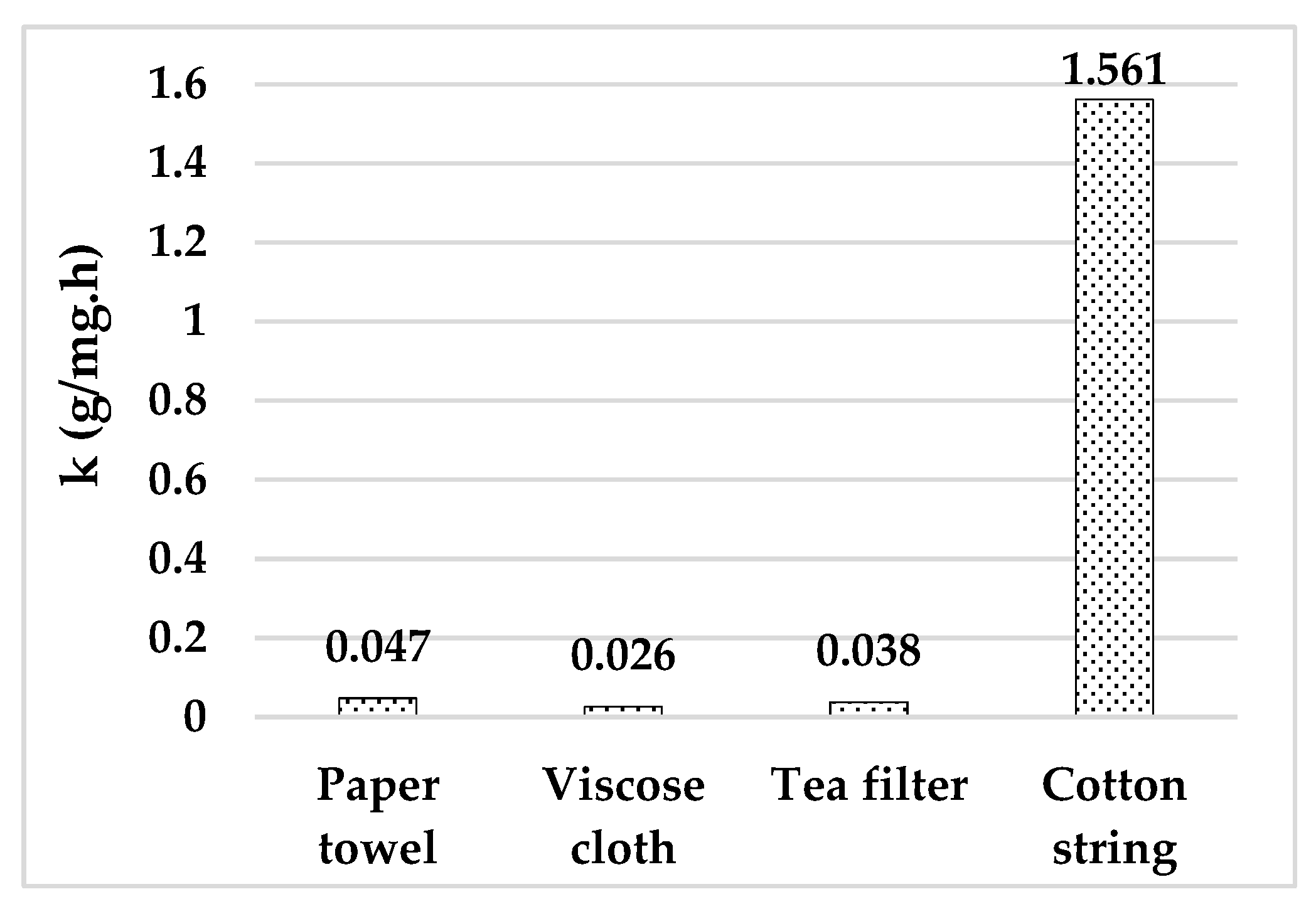

| Adsorbent | Price (EUR/kg) | Availability | Standardization Degree 1 | Absorbance at 262 nm |
|---|---|---|---|---|
| White household paper towel | 7.5 | Easy (retail) |
| 0.015 ± 0.012 |
| Light blue viscose household cloth | 30.2 | Easy (retail) |
| 0.018 ± 0.009 |
| White tea filter paper | 82.2 | Easy (retail) |
| 0.018 ± 0.008 |
| White cotton string | 16.7 | Easy (retail) |
| 0.003 ± 0.003 |
| Sawdust | 0.16 | Easy (household) |
| From 0.38 to 3.60 |
| T (K) | 100 × k (g/mg·h) | R2adjusted |
|---|---|---|
| 291 ± 0.1 | 4.74 ± 0.08 | 0.9932 |
| 298 ± 0.1 | 7.00 ± 0.23 | 0.9997 |
| 308 ± 0.1 | 10.44 ± 0.17 | 0.9971 |
| 318 ± 0.1 | 72.93 ± 1.68 | 0.9889 |
| Ceq (mg/L) | qeq—Experimental (mg/g) | qeq—Calculated (mg/g) | R2adjusted |
|---|---|---|---|
| 63.63 ± 4.03 | 18.64 ± 0.02 | 18.69 ± 0.04 | 0.9980 |
| 135.98 ± 2.09 | 23.90 ± 0.07 | 23.87 ± 0.02 | 0.9998 |
| 264.36 ± 2.11 | 23.56 ± 1.04 | 23.53 ± 1.01 | 0.9997 |
| 477.76 ± 9.13 | 27.22 ± 0.01 | 27.25 ± 0.01 | 0.9993 |
| 672.30 ± 11.17 | 32.77 ± 1.05 | 33.33 ± 0.07 | 0.9980 |
| Model 1,2 | Equation | Parameters 3 | RMSD | HYBRID | R2adjsuted |
|---|---|---|---|---|---|
| Langmuir 1 | qmax = 35.088 KL = 11.760 | 2.777 | 56.052 | 0.956 | |
| Freundlich 1 | KF = 7.900 n = 4.762 | 1.719 | 18.135 | 0.886 | |
| Dubinin–Radushkevich 1 | qs = 3.10−4 kad = 27.719 | 3.079 | 53.815 | 0.674 | |
| Temkin 1 | bT = 472.270 AT = 0.530 | 1.861 | 21.275 | 0.852 | |
| Hill 2 | qsH = 7191.3 kD = 744.214 nH = 0.168 | 2.268 | 41.914 | 0.865 | |
| Redlich–Peterson 2 | KR = 3673.7 aR = 464.983 g = 0.790 | 1.720 | 27.914 | 0.988 | |
| Toth 2 | KT = 7.492 aT = 0 t = 1.266 | 1.707 | 27.388 | 0.848 | |
| Radke–Prausnitz 2 | aRP = 3112.800 KRP = 7.493 ᵝR = 0.220 | 1.708 | 27.504 | 0.857 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bitay, E.; Csavdari, A. Some Unmodified Household Adsorbents for the Adsorption of Benzalkonium Chloride—A Kinetic and Thermodynamic Case Study for Commercially Available Paper. Toxics 2023, 11, 950. https://doi.org/10.3390/toxics11120950
Bitay E, Csavdari A. Some Unmodified Household Adsorbents for the Adsorption of Benzalkonium Chloride—A Kinetic and Thermodynamic Case Study for Commercially Available Paper. Toxics. 2023; 11(12):950. https://doi.org/10.3390/toxics11120950
Chicago/Turabian StyleBitay, Enikő, and Alexandra Csavdari. 2023. "Some Unmodified Household Adsorbents for the Adsorption of Benzalkonium Chloride—A Kinetic and Thermodynamic Case Study for Commercially Available Paper" Toxics 11, no. 12: 950. https://doi.org/10.3390/toxics11120950
APA StyleBitay, E., & Csavdari, A. (2023). Some Unmodified Household Adsorbents for the Adsorption of Benzalkonium Chloride—A Kinetic and Thermodynamic Case Study for Commercially Available Paper. Toxics, 11(12), 950. https://doi.org/10.3390/toxics11120950









