Nose-Only Exposure to Cherry- and Tobacco-Flavored E-Cigarettes Induced Lung Inflammation in Mice in a Sex-Dependent Manner
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Animals
2.3. E-Cigarette Device and Liquids
2.4. E-Cigarette Exposure
2.5. BALF Collection and Cell Counts
2.6. Protein Extraction
2.7. Pro-Inflammatory Cytokines/Chemokines
2.8. Immunoblot Assay
2.9. Proton Nuclear Magnetic Resonance Spectroscopy Chemical Assay
2.10. Statistical Analysis
3. Results
3.1. NMR Analysis of Flavored E-liquids for Flavoring Chemicals
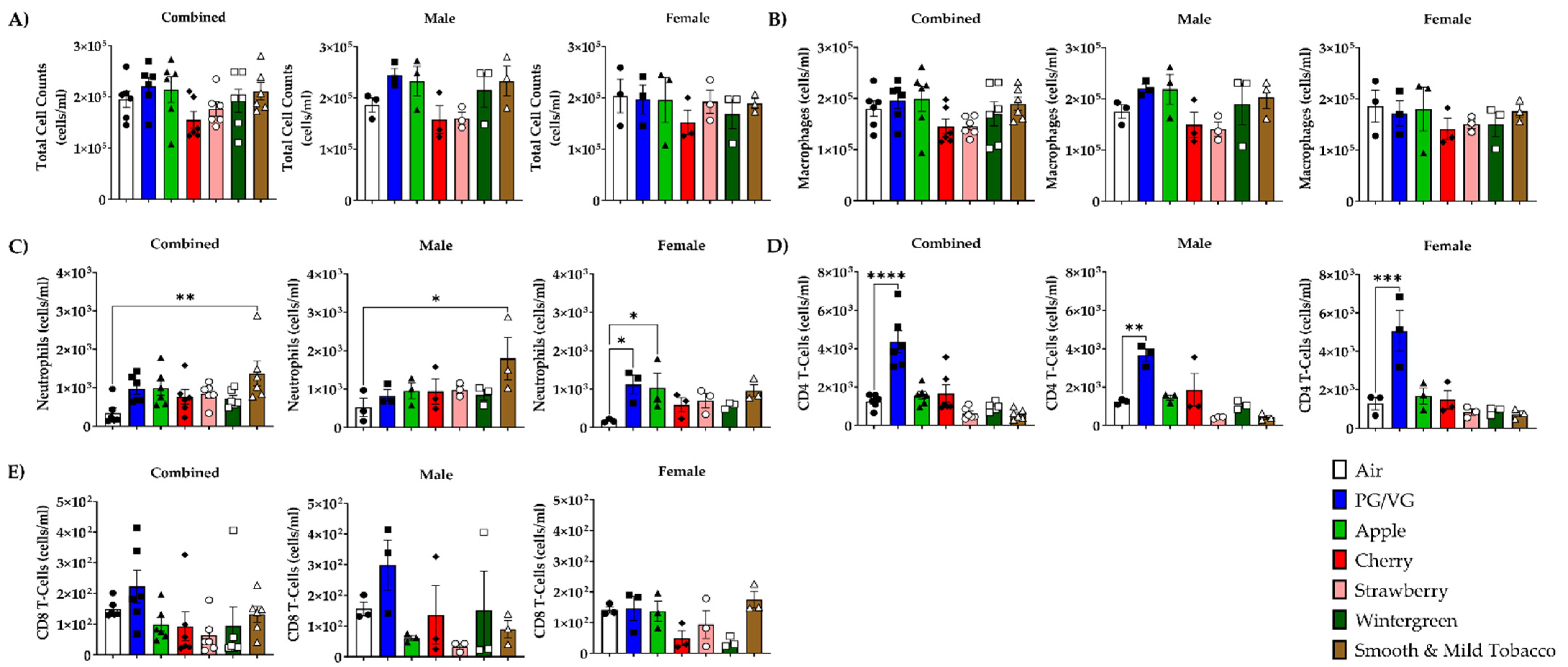
3.2. Alterations in Inflammatory Cell Influx in Lung by E-cigarette Flavors
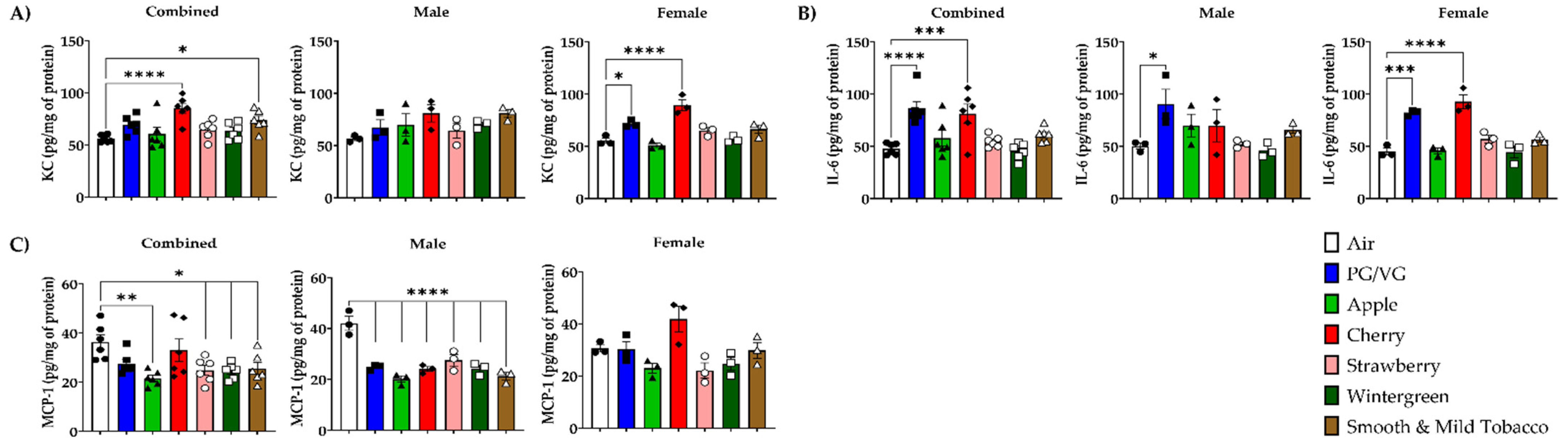
3.3. Alteration of Pro-Inflammatory Cytokines/Chemokines Levels in Lungs by E-cigarette Flavors
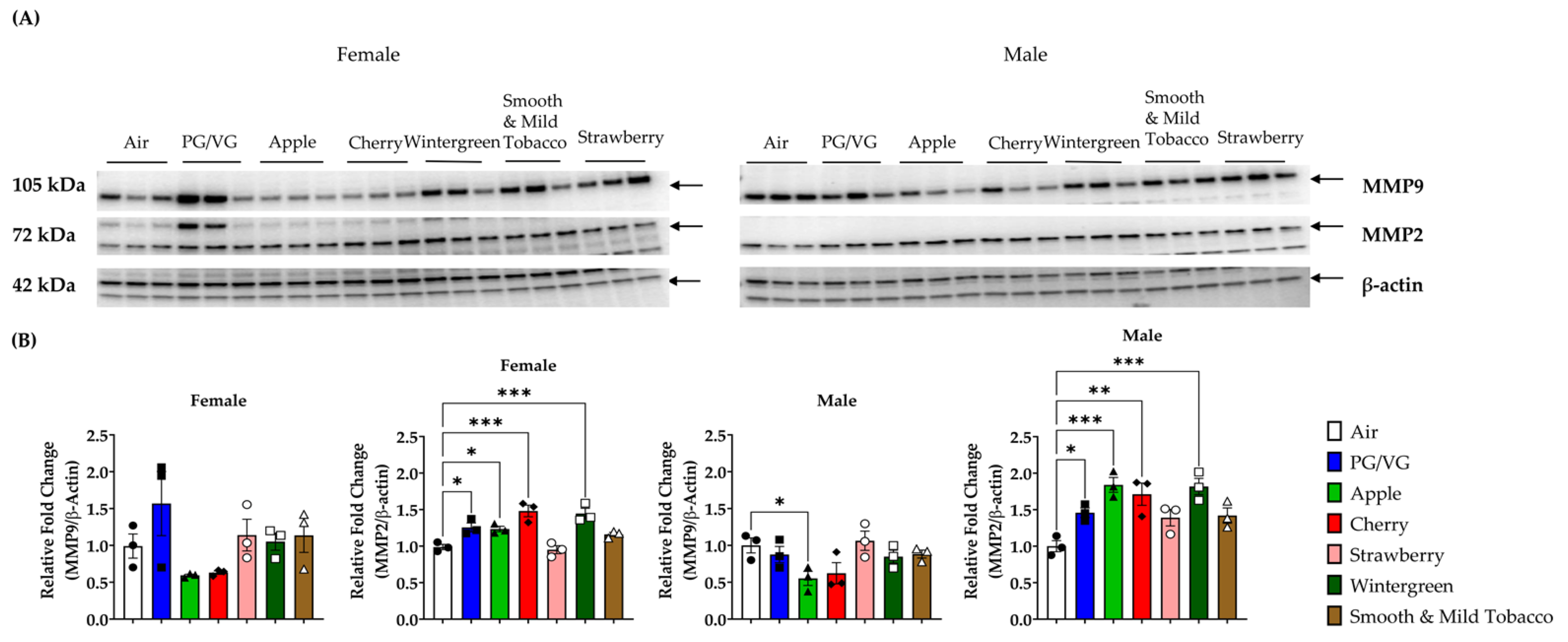
3.4. Alterations in Matrix Metalloproteinase Levels in Lungs by E-cigarette Flavors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Cao, Y.; Wu, D.; Ma, Y.; Ma, X.; Wang, S.; Li, F.; Li, M.; Zhang, T. Toxicity of electronic cigarettes: A general review of the origins, health hazards, and toxicity mechanisms. Sci. Total Environ. 2021, 772, 145475. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Muthumalage, T.; Rahman, I. Mechanisms of toxicity and biomarkers of flavoring and flavor enhancing chemicals in emerging tobacco and non-tobacco products. Toxicol. Lett. 2018, 288, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Cornelius, M.E.; Wang, T.W.; Jamal, A.; Loretan, C.G.; Neff, L.J. Tobacco Product Use Among Adults—United States, 2019. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1736–1742. [Google Scholar] [CrossRef] [PubMed]
- Villarroel, M.A.; Cha, A.E.; Vahratian, A. Electronic Cigarette Use among US Adults, 2018. NCHS Data Brief. 2020, 365, 1–8. [Google Scholar]
- Soneji, S.S.; Knutzen, K.E.; Villanti, A.C. Use of Flavored E-Cigarettes Among Adolescents, Young Adults, and Older Adults: Findings From the Population Assessment for Tobacco and Health Study. Public Health Rep. 2019, 134, 282–292. [Google Scholar] [CrossRef]
- Schneller, L.M.; Bansal-Travers, M.; Goniewicz, M.L.; McIntosh, S.; Ossip, D.; O’Connor, R.J. Use of Flavored E-Cigarettes and the Type of E-Cigarette Devices Used among Adults and Youth in the US-Results from Wave 3 of the Population Assessment of Tobacco and Health Study (2015–2016). Int. J. Environ. Res. Public Health 2019, 16, 2991. [Google Scholar] [CrossRef] [PubMed]
- Park-Lee, E.; Ren, C.; Sawdey, M.D.; Gentzke, A.S.; Cornelius, M.; Jamal, A.; Cullen, K.A. Notes from the Field: E-Cigarette Use Among Middle and High School Students—National Youth Tobacco Survey, United States, 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1387–1389. [Google Scholar] [CrossRef] [PubMed]
- Amrock, S.M.; Zakhar, J.; Zhou, S.; Weitzman, M. Perception of e-cigarette harm and its correlation with use among US adolescents. Nicotine Tob. Res. 2015, 17, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Feng, B.; Weaver, S.R.; Pechacek, T.F.; Slovic, P.; Eriksen, M.P. Changing Perceptions of Harm of e-Cigarette vs. Cigarette Use Among Adults in 2 US National Surveys From 2012 to 2017. JAMA Netw. Open 2019, 2, e191047. [Google Scholar] [CrossRef]
- Hua, M.; Omaiye, E.E.; Luo, W.; McWhirter, K.J.; Pankow, J.F.; Talbot, P. Identification of Cytotoxic Flavor Chemicals in Top-Selling Electronic Cigarette Refill Fluids. Sci. Rep. 2019, 9, 2782. [Google Scholar] [CrossRef]
- Gerloff, J.; Sundar, I.K.; Freter, R.; Sekera, E.R.; Friedman, A.E.; Robinson, R.; Pagano, T.; Rahman, I. Inflammatory Response and Barrier Dysfunction by Different e-Cigarette Flavoring Chemicals Identified by Gas Chromatography-Mass Spectrometry in e-Liquids and e-Vapors on Human Lung Epithelial Cells and Fibroblasts. Appl. Vitro Toxicol. 2017, 3, 28–40. [Google Scholar] [CrossRef]
- Muthumalage, T.; Prinz, M.; Ansah, K.O.; Gerloff, J.; Sundar, I.K.; Rahman, I. Inflammatory and Oxidative Responses Induced by Exposure to Commonly Used e-Cigarette Flavoring Chemicals and Flavored e-Liquids without Nicotine. Front. Physiol. 2017, 8, 1130. [Google Scholar] [CrossRef] [PubMed]
- Tierney, P.A.; Karpinski, C.D.; Brown, J.E.; Luo, W.; Pankow, J.F. Flavour chemicals in electronic cigarette fluids. Tob. Control 2016, 25, e10–e15. [Google Scholar] [CrossRef] [PubMed]
- Clapp, P.W.; Lavrich, K.S.; van Heusden, C.A.; Lazarowski, E.R.; Carson, J.L.; Jaspers, I. Cinnamaldehyde in flavored e-cigarette liquids temporarily suppresses bronchial epithelial cell ciliary motility by dysregulation of mitochondrial function. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 316, L470–L486. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Khan, N.A.; Muthumalage, T.; Lawyer, G.R.; McDonough, S.R.; Chuang, T.D.; Gong, M.; Sundar, I.K.; Rehan, V.K.; Rahman, I. Dysregulated repair and inflammatory responses by e-cigarette-derived inhaled nicotine and humectant propylene glycol in a sex-dependent manner in mouse lung. FASEB Bioadv. 2019, 1, 609–623. [Google Scholar] [CrossRef] [PubMed]
- Hariri, L.P.; Flashner, B.M.; Kanarek, D.J.; O’Donnell, W.J.; Soskis, A.; Ziehr, D.R.; Frank, A.; Nandy, S.; Berigei, S.R.; Sharma, A.; et al. E-Cigarette Use, Small Airway Fibrosis, and Constrictive Bronchiolitis. NEJM Evid. 2022, 1, EVIDoa2100051. [Google Scholar] [CrossRef]
- Osei, A.D.; Mirbolouk, M.; Orimoloye, O.A.; Dzaye, O.; Uddin, S.M.I.; Benjamin, E.J.; Hall, M.E.; DeFilippis, A.P.; Bhatnagar, A.; Biswal, S.S.; et al. Association Between E-Cigarette Use and Chronic Obstructive Pulmonary Disease by Smoking Status: Behavioral Risk Factor Surveillance System 2016 and 2017. Am. J. Prev. Med. 2020, 58, 336–342. [Google Scholar] [CrossRef]
- Bhatta, D.N.; Glantz, S.A. Association of E-Cigarette Use with Respiratory Disease among Adults: A Longitudinal Analysis. Am. J. Prev. Med. 2020, 58, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Dautzenberg, B.; Bricard, D. Real-Time Characterization of E-Cigarettes Use: The 1 Million Puffs Study. J. Addict. Res. Ther. 2015, 6, 4172. [Google Scholar] [CrossRef]
- Eshraghian, E.A.; Al-Delaimy, W.K. A review of constituents identified in e-cigarette liquids and aerosols. Tob. Prev. Cessat. 2021, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Krusemann, E.J.Z.; Pennings, J.L.A.; Cremers, J.; Bakker, F.; Boesveldt, S.; Talhout, R. GC-MS analysis of e-cigarette refill solutions: A comparison of flavoring composition between flavor categories. J. Pharm. Biomed. Anal. 2020, 188, 113364. [Google Scholar] [CrossRef] [PubMed]
- Khachatoorian, C.; McWhirter, K.J.; Luo, W.; Pankow, J.F.; Talbot, P. Tracing the movement of electronic cigarette flavor chemicals and nicotine from refill fluids to aerosol, lungs, exhale, and the environment. Chemosphere 2022, 286, 131494. [Google Scholar] [CrossRef]
- Allen, J.G.; Flanigan, S.S.; LeBlanc, M.; Vallarino, J.; MacNaughton, P.; Stewart, J.H.; Christiani, D.C. Flavoring Chemicals in E-Cigarettes: Diacetyl, 2,3-Pentanedione, and Acetoin in a Sample of 51 Products, Including fruit-, Candy-, and Cocktail-Flavored E-Cigarettes. Environ. Health Perspect. 2016, 124, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Andersen, A. Final report on the safety assessment of benzaldehyde. Int. J. Toxicol. 2006, 25 (Suppl. S1), 11–27. [Google Scholar] [CrossRef]
- Szafran, B.N.; Pinkston, R.; Perveen, Z.; Ross, M.K.; Morgan, T.; Paulsen, D.B.; Penn, A.L.; Kaplan, B.L.F.; Noel, A. Electronic-Cigarette Vehicles and Flavoring Affect Lung Function and Immune Responses in a Murine Model. Int. J. Mol. Sci. 2020, 21, 6022. [Google Scholar] [CrossRef]
- Sussan, T.E.; Gajghate, S.; Thimmulappa, R.K.; Ma, J.; Kim, J.H.; Sudini, K.; Consolini, N.; Cormier, S.A.; Lomnicki, S.; Hasan, F.; et al. xposure to electronic cigarettes impairs pulmonary anti-bacterial and anti-viral defenses in a mouse model. PLoS ONE 2015, 10, e0116861. [Google Scholar] [CrossRef] [PubMed]
- Larcombe, A.N.; Janka, M.A.; Mullins, B.J.; Berry, L.J.; Bredin, A.; Franklin, P.J. The effects of electronic cigarette aerosol exposure on inflammation and lung function in mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2017, 313, L67–L79. [Google Scholar] [CrossRef] [PubMed]
- Madison, M.C.; Landers, C.T.; Gu, B.H.; Chang, C.Y.; Tung, H.Y.; You, R.; Hong, M.J.; Baghaei, N.; Song, L.Z.; Porter, P.; et al. Electronic cigarettes disrupt lung lipid homeostasis and innate immunity independent of nicotine. J. Clin. Investig. 2019, 129, 4290–4304. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Sundar, I.; Li, D.; Lucas, J.; Muthumalage, T.; McDonough, S.; Rahman, I. E-cigarette-Induced Pulmonary Inflammation and Dysregulated Repair are Mediated by nAChR alpha7 Receptor: Role of nAChR alpha7 in ACE2 Covid-19 receptor regulation. Respir. Res. 2020, 21, 154. [Google Scholar] [CrossRef] [PubMed]
- Husari, A.; Shihadeh, A.; Talih, S.; Hashem, Y.; El Sabban, M.; Zaatari, G. Acute Exposure to Electronic and Combustible Cigarette Aerosols: Effects in an Animal Model and in Human Alveolar Cells. Nicotine Tob. Res. 2016, 18, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Glynos, C.; Bibli, S.I.; Katsaounou, P.; Pavlidou, A.; Magkou, C.; Karavana, V.; Topouzis, S.; Kalomenidis, I.; Zakynthinos, S.; Papapetropoulos, A. Comparison of the effects of e-cigarette vapor with cigarette smoke on lung function and inflammation in mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2018, 315, L662–L672. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Peng, G.; Meister, M.; Yao, H.; Yang, J.J.; Zou, M.H.; Liu, Z.R.; Ji, X. Electronic Cigarette Exposure Enhances Lung Inflammatory and Fibrotic Responses in COPD Mice. Front. Pharmacol. 2021, 12, 726586. [Google Scholar] [CrossRef]
- Garcia-Arcos, I.; Geraghty, P.; Baumlin, N.; Campos, M.; Dabo, A.J.; Jundi, B.; Cummins, N.; Eden, E.; Grosche, A.; Salathe, M.; et al. Chronic electronic cigarette exposure in mice induces features of COPD in a nicotine-dependent manner. Thorax 2016, 71, 1119–1129. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.H.; Pham, A.K.; Davis, B.; Smiley-Jewell, S.; Wang, L.; Kodavanti, U.P.; Takeuchi, M.; Tancredi, D.J.; Pinkerton, K.E. Sex and strain-based inflammatory response to repeated tobacco smoke exposure in spontaneously hypertensive and Wistar Kyoto rats. Inhal. Toxicol. 2016, 28, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Demedts, I.K.; Brusselle, G.G.; Bracke, K.R.; Vermaelen, K.Y.; Pauwels, R.A. Matrix metalloproteinases in asthma and COPD. Curr. Opin. Pharmacol. 2005, 5, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Higham, A.; Rattray, N.J.; Dewhurst, J.A.; Trivedi, D.K.; Fowler, S.J.; Goodacre, R.; Singh, D. Electronic cigarette exposure triggers neutrophil inflammatory responses. Respir. Res. 2016, 17, 56. [Google Scholar] [CrossRef]
- Scott, A.; Lugg, S.T.; Aldridge, K.; Lewis, K.E.; Bowden, A.; Mahida, R.Y.; Grudzinska, F.S.; Dosanjh, D.; Parekh, D.; Foronjy, R.; et al. Pro-inflammatory effects of e-cigarette vapour condensate on human alveolar macrophages. Thorax 2018, 73, 1161–1169. [Google Scholar] [CrossRef]
- Singh, K.P.; Lawyer, G.; Muthumalage, T.; Maremanda, K.P.; Khan, N.A.; McDonough, S.R.; Ye, D.; McIntosh, S.; Rahman, I. Systemic biomarkers in electronic cigarette users: Implications for noninvasive assessment of vaping-associated pulmonary injuries. ERJ Open Res. 2019, 5, 00182–2019. [Google Scholar] [CrossRef]
- Ghosh, A.; Coakley, R.D.; Ghio, A.J.; Muhlebach, M.S.; Esther, C.R., Jr.; Alexis, N.E.; Tarran, R. Chronic E-Cigarette Use Increases Neutrophil Elastase and Matrix Metalloprotease Levels in the Lung. Am. J. Respir. Crit. Care Med. 2019, 200, 1392–1401. [Google Scholar] [CrossRef]

| E-liquids | Flavoring Chemicals | Concentration | PG:VG |
|---|---|---|---|
| Apple | Hexyl Acetate | 0.43 ± 0.04 mg/mL | 46:54 |
| Ethyl Maltol | 0.30 ± 0.05 mg/mL | ||
| Cherry | Benzaldehyde | 0.12 ± 0.01 mg/mL | 51:49 |
| Strawberry | Ethyl Maltol | 0.32 ± 0.05 mg/mL | 50:50 |
| Maltol | 0.24 ± 0.04 mg/mL | ||
| Wintergreen | Methyl Salicylate | 9.70 ± 0.50 mg/mL | 49:51 |
| Smooth & Mild Tobacco | Maltol | 1.13 ± 0.02 mg/mL | 49:51 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lamb, T.; Muthumalage, T.; Meehan-Atrash, J.; Rahman, I. Nose-Only Exposure to Cherry- and Tobacco-Flavored E-Cigarettes Induced Lung Inflammation in Mice in a Sex-Dependent Manner. Toxics 2022, 10, 471. https://doi.org/10.3390/toxics10080471
Lamb T, Muthumalage T, Meehan-Atrash J, Rahman I. Nose-Only Exposure to Cherry- and Tobacco-Flavored E-Cigarettes Induced Lung Inflammation in Mice in a Sex-Dependent Manner. Toxics. 2022; 10(8):471. https://doi.org/10.3390/toxics10080471
Chicago/Turabian StyleLamb, Thomas, Thivanka Muthumalage, Jiries Meehan-Atrash, and Irfan Rahman. 2022. "Nose-Only Exposure to Cherry- and Tobacco-Flavored E-Cigarettes Induced Lung Inflammation in Mice in a Sex-Dependent Manner" Toxics 10, no. 8: 471. https://doi.org/10.3390/toxics10080471
APA StyleLamb, T., Muthumalage, T., Meehan-Atrash, J., & Rahman, I. (2022). Nose-Only Exposure to Cherry- and Tobacco-Flavored E-Cigarettes Induced Lung Inflammation in Mice in a Sex-Dependent Manner. Toxics, 10(8), 471. https://doi.org/10.3390/toxics10080471







