Neurotoxicity of Polycyclic Aromatic Hydrocarbons: A Systematic Mapping and Review of Neuropathological Mechanisms
Abstract
1. Introduction
2. Methodology
2.1. Acquisition of Data
2.2. Data Analysis
2.3. Country Collaboration Mapping of PAH Neurotoxicity Research Landscape
3. Results
3.1. Annual Research Outputs
3.2. Main Information
3.3. Analysis of Most Productive Authors
3.4. Country Collaboration
3.5. Thematic Areas
3.6. Corresponding Authors
3.7. Country Analysis
3.8. Journal Analysis
4. Discussion
4.1. Global Research Output Related to PAH Neurotoxicity
4.2. Neuropathological Mechanisms of PAHs
4.2.1. Effect of PAHs on Antioxidant Defence System
4.2.2. PAHs and Induction of Apoptosis
4.2.3. Effect of PAHs on Acetylcholinesterase Activity
4.2.4. PAHs and Developmental Neurotoxicity
4.2.5. Effect of PAHs on Learning, Behaviour and Memory Function
4.2.6. PAHs and Neurodegeneration
4.3. Therapeutic Strategies against PAH-Induced Neurotoxicity
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abdel-Shafy, H.I.; Mansour, M.S.M. A review on polycyclic aromatic hydrocarbons: Source, environmental impact, effect on human health and remediation. Egypt. J. Pet. 2016, 25, 107–123. [Google Scholar] [CrossRef]
- Lawal, A.T. Polycyclic aromatic hydrocarbons. A review. Cogent Environ. Sci. 2017, 3, 1339841. [Google Scholar] [CrossRef]
- Edokpayi, J.N.; Odiyo, J.O.; Popoola, O.E.; Msagati, T.A. Determination and Distribution of Polycyclic Aromatic Hydrocarbons in Rivers, Sediments and Wastewater Effluents in Vhembe District, South Africa. Int. J. Environ. Res. Public Health 2016, 13, 387. [Google Scholar] [CrossRef]
- Dong, C.D.; Chen, C.F.; Chen, C.W. Determination of polycyclic aromatic hydrocarbons in industrial harbor sediments by GC-MS. Int. J. Environ. Res. Public Health 2012, 9, 2175–2188. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.F.D.; Azevedo, D.D.A.; Aquino Neto, F.R.D. Distribution of polycyclic aromatic hydrocarbons in surface sediments and waters from Guanabara Bay, Rio de Janeiro, Brazil. J. Braz. Chem. Soc. 2007, 18, 628–637. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, S.; Jia, Z.; Ge, L.; Mei, L.; Sui, X.; Wang, X.; Li, B.; Wang, J.; Wu, S. Influence of Industrialization and Environmental Protection on Environmental Pollution: A Case Study of Taihu Lake, China. Int. J. Environ. Res. Public Health 2018, 15, 2628. [Google Scholar] [CrossRef]
- Balmer, J.E.; Hung, H.; Yu, Y.; Letcher, R.J.; Muir, D.C.G. Sources and environmental fate of pyrogenic polycyclic aromatic hydrocarbons (PAHs) in the Arctic. Emerg. Contam. 2019, 5, 128–142. [Google Scholar] [CrossRef]
- Srogi, K. Monitoring of environmental exposure to polycyclic aromatic hydrocarbons: A review. Environ. Chem. Lett. 2007, 5, 169–195. [Google Scholar] [CrossRef]
- Ramesh, A.; Harris, K.J.; Archibong, A.E. Chapter 40: Reproductive Toxicity of Polycyclic Aromatic Hydrocarbons. In Reproductive and Developmental Toxicology, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Rengarajan, T.; Rajendran, P.; Nandakumar, N.; Lokeshkumar, B.; Rajendran, P.; Nishigaki, I. Exposure to polycyclic aromatic hydrocarbons with special focus on cancer. Asian Pac. J. Trop. Biomed. 2015, 5, 182–189. [Google Scholar] [CrossRef]
- Ramos De Rainho, C.; Machado Corrêa, S.; Luiz Mazzei, J.; Alessandra Fortes Aiub, C.; Felzenszwalb, I. Genotoxicity of Polycyclic Aromatic Hydrocarbons and Nitro-Derived in Respirable Airborne Particulate Matter Collected from Urban Areas of Rio de Janeiro (Brazil). BioMed. Res. Int. 2013, 2013, 765352. [Google Scholar] [CrossRef]
- Silkworth, J.B.; Lipinskas, T.; Stoner, C.R. Immunosuppressive potential of several polycyclic aromatic hydrocarbons (PAHs) found at a Superfund site: New model used to evaluate additive interactions between benzo[a]pyrene and TCDD. Toxicology 1995, 105, 375–386. [Google Scholar] [CrossRef]
- O’Brien, J.L.; Langlois, P.H.; Lawson, C.C.; Scheuerle, A.; Rocheleau, C.M.; Waters, M.A.; Symanski, E.; Romitti, P.A.; Agopian, A.J.; Lupo, P.J. Maternal occupational exposure to polycyclic aromatic hydrocarbons and craniosynostosis among offspring in the national birth defects prevention study. Birth Defects Res. Part A Clin. Mol. Teratol. 2016, 106, 55–60. [Google Scholar] [CrossRef]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef] [PubMed]
- Goulaouic, S.; Foucaud, L.; Bennasroune, A.; Laval-Gilly, P.; Falla, J. Effect of Polycyclic Aromatic Hydrocarbons and Carbon Black Particles on Pro-Inflammatory Cytokine Secretion: Impact of PAH Coating Onto Particles. J. Immunotoxicol. 2008, 5, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Hong, S.; Liu, X.; Kim, C.; Jung, D.; Yim, U.H.; Shim, W.J.; Khim, J.S.; Giesy, J.P.; Choi, K. Endocrine disrupting potential of PAHs and their alkylated analogues associated with oil spills. Environ. Sci. Processes Impacts 2017, 19, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Archibong, A.; Ramesh, A.; Niaz, M.; Brooks, C.; Roberson, S.; Lunstra, D. Effects of Benzo(a)pyrene on Intra-testicular Function in F-344 Rats. Int. J. Environ. Res. Public Health 2008, 5, 32–40. [Google Scholar] [CrossRef]
- Choi, H.; Harrison, R.; Komulainen, H.; Saborit, J.M.D. Polycyclic aromatic hydrocarbons. In WHO Guidelines for Indoor Air Quality: Selected Pollutants; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Tian, F.J.; Li, W.X.; Lyu, Y.; Zhang, P.; Mu, J.B.; Pei, Q.L.; Zheng, J.P. Heat-shock protein 70 (HSP70) polymorphisms affect the risk of coke-oven emission-induced neurobehavioral damage. Neurotoxicology 2020, 76, 174–182. [Google Scholar] [CrossRef]
- McCallister, M.M.; Maguire, M.; Ramesh, A.; Aimin, Q.; Liu, S.; Khoshbouei, H.; Aschner, M.; Ebner, F.F.; Hood, D.B. Prenatal exposure to benzo(a)pyrene impairs later-life cortical neuronal function. NeuroToxicology 2008, 29, 846–854. [Google Scholar] [CrossRef]
- Peiffer, J.; Grova, N.; Hidalgo, S.; Salquebre, G.; Rychen, G.; Bisson, J.F.; Appenzeller, B.M.R.; Schroeder, H. Behavioral toxicity and physiological changes from repeated exposure to fluorene administered orally or intraperitoneally to adult male Wistar rats: A dose-response study. Neurotoxicology 2016, 53, 321–333. [Google Scholar] [CrossRef]
- Das, L.; Patel, B.; Patri, M. Adolescence benzo[a]pyrene treatment induces learning and memory impairment and anxiolytic like behavioral response altering neuronal morphology of hippocampus in adult male Wistar rats. Toxicol. Rep. 2019, 6, 1104–1113. [Google Scholar] [CrossRef]
- Peterson, B.S.; Rauh, V.A.; Bansal, R.; Hao, X.; Toth, Z.; Nati, G.; Walsh, K.; Miller, R.L.; Arias, F.; Semanek, D.; et al. Effects of Prenatal Exposure to Air Pollutants (Polycyclic Aromatic Hydrocarbons) on the Development of Brain White Matter, Cognition, and Behavior in Later Childhood. JAMA Psychiatry 2015, 72, 531. [Google Scholar] [CrossRef] [PubMed]
- Mortamais, M.; Pujol, J.; van Drooge, B.L.; Macia, D.; Martinez-Vilavella, G.; Reynes, C.; Sabatier, R.; Rivas, I.; Grimalt, J.; Forns, J.; et al. Effect of exposure to polycyclic aromatic hydrocarbons on basal ganglia and attention-deficit hyperactivity disorder symptoms in primary school children. Environ. Int. 2017, 105, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Jedrychowski, W.A.; Perera, F.P.; Camann, D.; Spengler, J.; Butscher, M.; Mroz, E.; Majewska, R.; Flak, E.; Jacek, R.; Sowa, A. Prenatal exposure to polycyclic aromatic hydrocarbons and cognitive dysfunction in children. Environ. Sci. Pollut. Res. 2015, 22, 3631–3639. [Google Scholar] [CrossRef] [PubMed]
- Perera, F.P.; Chang, H.-W.; Tang, D.; Roen, E.L.; Herbstman, J.; Margolis, A.; Huang, T.-J.; Miller, R.L.; Wang, S.; Rauh, V. Early-Life Exposure to Polycyclic Aromatic Hydrocarbons and ADHD Behavior Problems. PLoS ONE 2014, 9, e111670. [Google Scholar] [CrossRef] [PubMed]
- Perera, F.P.; Rauh, V.; Whyatt, R.M.; Tang, D.; Tsai, W.Y.; Bernert, J.T.; Tu, Y.H.; Andrews, H.; Barr, D.B.; Camann, D.E.; et al. A summary of recent findings on birth outcomes and developmental effects of prenatal ETS, PAH, and pesticide exposures. Neurotoxicology 2005, 26, 573–587. [Google Scholar] [CrossRef]
- Perera, F.P.; Rauh, V.; Whyatt, R.M.; Tsai, W.Y.; Tang, D.; Diaz, D.; Hoepner, L.; Barr, D.; Tu, Y.H.; Camann, D.; et al. Effect of prenatal exposure to airborne polycyclic aromatic hydrocarbons on neurodevelopment in the first 3 years of life among inner-city children. Environ. Health Perspect. 2006, 114, 1287–1292. [Google Scholar] [CrossRef]
- Wang, B.L.; Pang, S.T.; Zhang, X.L.; Li, X.L.; Sun, Y.G.; Lu, X.M.; Zhang, Q.; Zhang, Z.D. Levels and neurodevelopmental effects of polycyclic aromatic hydrocarbons in settled house dust of urban dwellings on preschool–aged children in Nanjing, China. Atmos. Pollut. Res. 2014, 5, 292–302. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, Y.; Zhang, Q.; Cui, J.; Rahaman, A.; Huang, X.-R.; Su, Y.; Gao, R.; Wang, B.; Baloch, Z. Effect of Benzo[a]pyrene-DNA Adduct in Cord Blood on the Neurodevelopment of 12-Month-Old Infants in Qingdao City. Neuropsychiatr. Dis. Treat. 2019, 15, 3351–3357. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Reprint—Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Phys. Ther. 2009, 89, 873–880. [Google Scholar] [CrossRef]
- Ruiz-Rosero, J.; Ramirez-Gonzalez, G.; Viveros-Delgado, J. Software survey: ScientoPy, a scientometric tool for topics trend analysis in scientific publications. Scientometrics 2019, 121, 1165–1188. [Google Scholar] [CrossRef]
- Aria, M.; Cuccurullo, C. Bibliometrix: An R-tool for comprehensive science mapping analysis. J. Informetr. 2017, 11, 959–975. [Google Scholar] [CrossRef]
- Ekundayo, T.C.; Olasehinde, T.A.; Okaiyeto, K.; Okoh, A.I. Microbial Pathogenesis and Pathophysiology of Alzheimer’s Disease: A Systematic Assessment of Microorganisms’ Implications in the Neurodegenerative Disease. Front. Neurosci. 2021, 15, 648484. [Google Scholar] [CrossRef] [PubMed]
- Jaccard, P. Distribution of alpine flora in the basin of Dranses and in some neighboring regions. Bull. Soc. Vaud. Sci. Nat. 1901, 37, 241–272. [Google Scholar]
- Mao, G.; Zhang, N. Analysis of Average Shortest-Path Length of Scale-Free Network. J. Appl. Math. 2013, 2013, 865643. [Google Scholar] [CrossRef]
- Avio, C.G.; Gorbi, S.; Milan, M.; Benedetti, M.; Fattorini, D.; d’Errico, G.; Pauletto, M.; Bargelloni, L.; Regoli, F. Pollutants bioavailability and toxicological risk from microplastics to marine mussels. Environ. Pollut. 2015, 198, 211–222. [Google Scholar] [CrossRef]
- Allen, J.L.; Oberdorster, G.; Morris-Schaffer, K.; Wong, C.; Klocke, C.; Sobolewski, M.; Conrad, K.; Mayer-Proschel, M.; Cory-Slechta, D.A. Developmental neurotoxicity of inhaled ambient ultrafine particle air pollution: Parallels with neuropathological and behavioral features of autism and other neurodevelopmental disorders. NeuroToxicology 2017, 59, 140–154. [Google Scholar] [CrossRef]
- Saunders, C.R.; Das, S.K.; Ramesh, A.; Shockley, D.C.; Mukherjee, S. Benzo(a)pyrene-induced acute neurotoxicity in the F-344 rat: Role of oxidative stress. J. Appl. Toxicol. 2006, 26, 427–438. [Google Scholar] [CrossRef]
- Jayasekara, S.; Sharma, R.P.; Drown, D.B. Effects of benzo[a]pyrene on steady-state levels of biogenic amines and metabolizing enzymes in mouse brain regions. Ecotoxicol. Environ. Saf. 1992, 24, 1–12. [Google Scholar] [CrossRef]
- Andersson, H.; Lindqvist, E.; Westerholm, R.; Grägg, K.; Almén, J.; Olson, L. Neurotoxic Effects of Fractionated Diesel Exhausts Following Microinjections in Rat Hippocampus and Striatum. Environ. Res. 1998, 76, 41–51. [Google Scholar] [CrossRef]
- Slotkin, T.A.; Seidler, F.J. Benzo[a]pyrene impairs neurodifferentiation in PC12 cells. Brain Res. Bull. 2009, 80, 17–21. [Google Scholar] [CrossRef][Green Version]
- Bouayed, J.; Desor, F.; Rammal, H.; Kiemer, A.K.; Tybl, E.; Schroeder, H.; Rychen, G.; Soulimani, R. Effects of lactational exposure to benzo[α]pyrene (B[α]P) on postnatal neurodevelopment, neuronal receptor gene expression and behaviour in mice. Toxicology 2009, 259, 97–106. [Google Scholar] [CrossRef]
- Slotkin, T.A.; Skavicus, S.; Ko, A.; Levin, E.D.; Seidler, F.J. The Developmental Neurotoxicity of Tobacco Smoke Can Be Mimicked by a Combination of Nicotine and Benzo[a]Pyrene: Effects on Cholinergic and Serotonergic Systems. Toxicol. Sci. 2019, 167, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Jinzhu, Y.; Qinli, Z.; Jin, Y.; Pan, K.; Jianjun, H.; Qiao, N. Aluminum and benzo[a]pyrene co-operate to induce neuronal apoptosis in vitro. J. Toxicol. Sci. 2015, 40, 365–373. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mehri, S.; Barangi, S.; Zamiri, E.; Karimi, G. The protective effect of melatonin on benzo(a)pyrene-induced brain injury: Role of apoptosis and autophagy pathways. Naunyn Schmiedebergs Arch. Pharmacol. 2020, 393, 2241–2251. [Google Scholar] [CrossRef]
- Mohanty, R.; Das, S.K.; Patri, M. Modulation of Benzo[a]Pyrene Induced Anxiolytic-Like Behavior by Retinoic Acid in Zebrafish: Involvement of Oxidative Stress and Antioxidant Defense System. Neurotox. Res. 2017, 31, 493–504. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Wu, C.-Y.; Hu, C.-H.; Pai, T.-W.; Chen, Y.-R.; Wang, W.-D. Integrated Hypoxia Signaling and Oxidative Stress in Developmental Neurotoxicity of Benzo[a]Pyrene in Zebrafish Embryos. Antioxidants 2020, 9, 731. [Google Scholar] [CrossRef] [PubMed]
- Das, S.K.; Patri, M. Neuropeptide Y expression confers benzo[a]pyrene induced anxiolytic like behavioral response during early adolescence period of male Wistar rats. Neuropeptides 2017, 61, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Tang, Y.; Duan, L.; Cheng, S.; Luo, L.; Cao, X.; Tu, B. Adverse effect of sub-chronic exposure to benzo(a)pyrene and protective effect of butylated hydroxyanisole on learning and memory ability in male Sprague-Dawley rat. J. Toxicol. Sci. 2014, 39, 739–748. [Google Scholar] [CrossRef]
- Petrovic, S.; Arsic, A.; Ristic-Medic, D.; Cvetkovic, Z.; Vucic, V. Lipid Peroxidation and Antioxidant Supplementation in Neurodegenerative Diseases: A Review of Human Studies. Antioxidants 2020, 9, 1128. [Google Scholar] [CrossRef]
- Patel, B.; Das, S.K.; Das, S.; Das, L.; Patri, M. Neonatal exposure to benzo[a]pyrene induces oxidative stress causing altered hippocampal cytomorphometry and behavior during early adolescence period of male Wistar rats. Int. J. Dev. Neurosci. 2016, 50, 7–15. [Google Scholar] [CrossRef]
- Gao, D.; Wu, M.; Wang, C.; Wang, Y.; Zuo, Z. Chronic exposure to low benzo[a]pyrene level causes neurodegenerative disease-like syndromes in zebrafish (Danio rerio). Aquat. Toxicol. 2015, 167, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Cheng, S.; He, J.; Liu, X.; Tang, Y.; Yuan, H.; He, L.; Lu, T.; Tu, B.; Wang, Y. Effects of subchronic exposure to benzo[a]pyrene (B[a]P) on learning and memory, and neurotransmitters in male Sprague-Dawley rat. Neurotoxicology 2011, 32, 188–198. [Google Scholar] [CrossRef]
- Gauthier, P.T.; Norwood, W.P.; Prepas, E.E.; Pyle, G.G. Behavioural alterations from exposure to Cu, phenanthrene, and Cu-phenanthrene mixtures: Linking behaviour to acute toxic mechanisms in the aquatic amphipod, Hyalella azteca. Aquat. Toxicol. 2016, 170, 377–383. [Google Scholar] [CrossRef]
- Saunders, C.R.; Shockley, D.C.; Knuckles, M.E. Fluoranthene-Induced Neurobehavioral Toxicity in F-344 Rats. Int. J. Toxicol. 2003, 22, 263–276. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Wang, C.; Li, B.; Wu, M.; Geng, H.; Chen, Y.; Zuo, Z. Exposure of Sebastiscus marmoratus embryos to pyrene results in neurodevelopmental defects and disturbs related mechanisms. Aquat. Toxicol. 2012, 116–117, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Redza-Dutordoir, M.; Averill-Bates, D.A. Activation of apoptosis signalling pathways by reactive oxygen species. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2016, 1863, 2977–2992. [Google Scholar] [CrossRef] [PubMed]
- Kannan, K.; Jain, S.K. Oxidative stress and apoptosis. Pathophysiology 2000, 7, 153–163. [Google Scholar] [CrossRef]
- Nie, J.S.; Zhang, H.M.; Zhao, J.; Liu, H.J.; Niu, Q. Involvement of mitochondrial pathway in benzo[a]pyrene-induced neuron apoptosis. Hum. Exp. Toxicol. 2014, 33, 240–250. [Google Scholar] [CrossRef]
- He, J.; Ji, X.; Li, Y.; Xue, X.; Feng, G.; Zhang, H.; Wang, H.; Gao, M. Subchronic exposure of benzo(a)pyrene interferes with the expression of Bcl-2, Ki-67, C-myc and p53, Bax, Caspase-3 in sub-regions of cerebral cortex and hippocampus. Exp. Toxicol. Pathol. 2016, 68, 149–156. [Google Scholar] [CrossRef]
- Lionetto, M.G.; Caricato, R.; Calisi, A.; Giordano, M.E.; Schettino, T. Acetylcholinesterase as a biomarker in environmental and occupational medicine: New insights and future perspectives. Biomed. Res. Int. 2013, 2013, 321213. [Google Scholar] [CrossRef]
- Hauser-Davis, R.A.; Lopes, R.M.; Ziolli, R.L. Inihibition of mullet (M. liza) brain acetylcholinesterase activity by in vitro polycyclic aromatic hydrocarbon exposure. Mar. Pollut. Bull. 2019, 140, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.-J.; Fang, H.-W. Polycyclic Aromatic Hydrocarbons Inhibit the Activity of Acetylcholinesterase Purified from Electric Eel. Biochem. Biophys. Res. Commun. 1997, 238, 367–369. [Google Scholar] [CrossRef] [PubMed]
- Maisano, M.; Cappello, T.; Natalotto, A.; Vitale, V.; Parrino, V.; Giannetto, A.; Oliva, S.; Mancini, G.; Cappello, S.; Mauceri, A.; et al. Effects of petrochemical contamination on caged marine mussels using a multi-biomarker approach: Histological changes, neurotoxicity and hypoxic stress. Mar. Environ. Res. 2017, 128, 114–123. [Google Scholar] [CrossRef]
- Kais, B.; Stengel, D.; Batel, A.; Braunbeck, T. Acetylcholinesterase in zebrafish embryos as a tool to identify neurotoxic effects in sediments. Environ. Sci. Pollut. Res. Int. 2015, 22, 16329–16339. [Google Scholar] [CrossRef]
- Aguilar, L.; Dzul-Caamal, R.; Rendón-Von Osten, J.; Da Cruz, A.L. Effects of Polycyclic Aromatic Hydrocarbons inGambusia yucatana, an Endemic Fish from Yucatán Peninsula, Mexico. Polycycl. Aromat. Compd. 2022, 42, 907–924. [Google Scholar] [CrossRef]
- Chen, C.; Tang, Y.; Jiang, X.; Qi, Y.; Cheng, S.; Qiu, C.; Peng, B.; Tu, B. Early Postnatal Benzo(a)pyrene Exposure in Sprague-Dawley Rats Causes Persistent Neurobehavioral Impairments that Emerge Postnatally and Continue into Adolescence and Adulthood. Toxicol. Sci. 2012, 125, 248–261. [Google Scholar] [CrossRef]
- Lee, J.; Kalia, V.; Perera, F.; Herbstman, J.; Li, T.; Nie, J.; Qu, L.R.; Yu, J.; Tang, D. Prenatal airborne polycyclic aromatic hydrocarbon exposure, LINE1 methylation and child development in a Chinese cohort. Environ. Int. 2017, 99, 315–320. [Google Scholar] [CrossRef]
- Perera, F.; Phillips, D.H.; Wang, Y.; Roen, E.; Herbstman, J.; Rauh, V.; Wang, S.; Tang, D. Prenatal exposure to polycyclic aromatic hydrocarbons/aromatics, BDNF and child development. Environ. Res. 2015, 142, 602–608. [Google Scholar] [CrossRef]
- Paulsen, M.; Ferguson-Smith, A.C. DNA methylation in genomic imprinting, development, and disease. J. Pathol. 2001, 195, 97–110. [Google Scholar] [CrossRef]
- Cohen-Cory, S.; Kidane, A.H.; Shirkey, N.J.; Marshak, S. Brain-derived neurotrophic factor and the development of structural neuronal connectivity. Dev. Neurobiol. 2010, 70, 271–288. [Google Scholar] [CrossRef]
- Lu, Y.; Christian, K.; Lu, B. BDNF: A key regulator for protein synthesis-dependent LTP and long-term memory? Neurobiol. Learn. Mem. 2008, 89, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Herbstman, J.B.; Tang, D.; Zhu, D.; Qu, L.; Sjödin, A.; Li, Z.; Camann, D.; Perera, F.P. Prenatal Exposure to Polycyclic Aromatic Hydrocarbons, Benzo[a]pyrene–DNA Adducts, and Genomic DNA Methylation in Cord Blood. Environ. Health Perspect. 2012, 120, 733–738. [Google Scholar] [CrossRef] [PubMed]
- Breton, C.V.; Byun, H.-M.; Wenten, M.; Pan, F.; Yang, A.; Gilliland, F.D. Prenatal Tobacco Smoke Exposure Affects Global and Gene-specific DNA Methylation. Am. J. Respir. Crit. Care Med. 2009, 180, 462–467. [Google Scholar] [CrossRef]
- Cho, J.; Sohn, J.; Noh, J.; Jang, H.; Kim, W.; Cho, S.K.; Seo, H.; Seo, G.; Lee, S.K.; Noh, Y.; et al. Association between exposure to polycyclic aromatic hydrocarbons and brain cortical thinning: The Environmental Pollution-Induced Neurological EFfects (EPINEF) study. Sci. Total. Environ. 2020, 737, 140097. [Google Scholar] [CrossRef]
- Chepelev, N.L.; Long, A.S.; Bowers, W.J.; Gagné, R.; Williams, A.; Kuo, B.; Phillips, D.H.; Arlt, V.M.; White, P.A.; Yauk, C.L. Transcriptional profiling of the mouse hippocampus supports an NMDAR-mediated neurotoxic mode of action for benzo[ a ]pyrene. Environ. Mol. Mutagenesis 2016, 57, 350–363. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.A.; Chen, F.H.; Huang, Z.Y.; Zhang, X.C. Elaidic acid enhanced the simultaneous neurotoxicity attributable to the cerebral pathological lesion resulted from oxidative damages induced by acrylamide and benzo(a)pyrene. Toxicol. Ind. Health 2011, 27, 661–672. [Google Scholar] [CrossRef]
- Crepeaux, G.; Bouillaud-Kremarik, P.; Sikhayeva, N.; Rychen, G.; Soulimani, R.; Schroeder, H. Late effects of a perinatal exposure to a 16 PAH mixture: Increase of anxiety-related behaviours and decrease of regional brain metabolism in adult male rats. Toxicol. Lett. 2012, 211, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Sas, K.; Robotka, H.; Toldi, J.; Vécsei, L. Mitochondria, metabolic disturbances, oxidative stress and the kynurenine system, with focus on neurodegenerative disorders. J. Neurol. Sci. 2007, 257, 221–239. [Google Scholar] [CrossRef]
- Crépeaux, G.; Bouillaud-Kremarik, P.; Sikhayeva, N.; Rychen, G.; Soulimani, R.; Schroeder, H. Exclusive prenatal exposure to a 16 PAH mixture does not impact anxiety-related behaviours and regional brain metabolism in adult male rats: A role for the period of exposure in the modulation of PAH neurotoxicity. Toxicol. Lett. 2013, 221, 40–46. [Google Scholar] [CrossRef]
- Sugahara, Y.; Kawaguchi, M.; Itoyama, T.; Kurokawa, D.; Tosa, Y.; Kitamura, S.; Handoh, I.C.; Nakayama, K.; Murakami, Y. Pyrene induces a reduction in midbrain size and abnormal swimming behavior in early-hatched pufferfish larvae. Mar. Pollut. Bull. 2014, 85, 479–486. [Google Scholar] [CrossRef]
- Geier, M.C.; James Minick, D.; Truong, L.; Tilton, S.; Pande, P.; Anderson, K.A.; Teeguardan, J.; Tanguay, R.L. Systematic developmental neurotoxicity assessment of a representative PAH Superfund mixture using zebrafish. Toxicol. Appl. Pharmacol. 2018, 354, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Vignet, C.; Le Menach, K.; Lyphout, L.; Guionnet, T.; Frère, L.; Leguay, D.; Budzinski, H.; Cousin, X.; Bégout, M.-L. Chronic dietary exposure to pyrolytic and petrogenic mixtures of PAHs causes physiological disruption in zebrafish—part II: Behavior. Environ. Sci. Pollut. Res. 2014, 21, 13818–13832. [Google Scholar] [CrossRef]
- Wang, F.; Yang, L.; Zhang, B.; Liu, G.; Wang, C.; Zhang, Y.; Wang, T. Neurobehavioral performance of PAH exposure in male coal miners in Shanxi, China: A cross-sectional study. Int. Arch. Occup. Environ. Health 2020, 93, 707–714. [Google Scholar] [CrossRef]
- Amor, S.; Puentes, F.; Baker, D.; Van Der Valk, P. Inflammation in neurodegenerative diseases. Immunology 2010, 129, 154–169. [Google Scholar] [CrossRef]
- Przedborski, S.; Vila, M.; Jackson-Lewis, V. Series Introduction: Neurodegeneration: What is it and where are we? J. Clin. Investig. 2003, 111, 3–10. [Google Scholar] [CrossRef]
- Chin-Chan, M.; Navarro-Yepes, J.; Quintanilla-Vega, B. Environmental pollutants as risk factors for neurodegenerative disorders: Alzheimer and Parkinson diseases. Front. Cell. Neurosci. 2015, 9, 124. [Google Scholar] [CrossRef]
- Genc, S.; Zadeoglulari, Z.; Fuss, S.H.; Genc, K. The Adverse Effects of Air Pollution on the Nervous System. J. Toxicol. 2012, 2012, 782462. [Google Scholar] [CrossRef]
- Valand, R.; Magnusson, P.; Dziendzikowska, K.; Gajewska, M.; Wilczak, J.; Oczkowski, M.; Kamola, D.; Krolikowski, T.; Kruszewski, M.; Lankoff, A.; et al. Gene expression changes in rat brain regions after 7- and 28 days inhalation exposure to exhaust emissions from 1st and 2nd generation biodiesel fuels—The FuelHealth project. Inhal. Toxicol. 2018, 30, 299–312. [Google Scholar] [CrossRef]
- Haghani, A.; Johnson, R.; Safi, N.; Zhang, H.; Thorwald, M.; Mousavi, A.; Woodward, N.C.; Shirmohammadi, F.; Coussa, V.; Wise, J.P.; et al. Toxicity of urban air pollution particulate matter in developing and adult mouse brain: Comparison of total and filter-eluted nanoparticles. Environ. Int. 2020, 136, 105510. [Google Scholar] [CrossRef]
- Srinivasan, M.; Lahiri, D.K. Significance of NF-κB as a pivotal therapeutic target in the neurodegenerative pathologies of Alzheimer’s disease and multiple sclerosis. Expert. Opin. Ther. Targets 2015, 19, 471–487. [Google Scholar] [CrossRef]
- Miquel, S.; Champ, C.; Day, J.; Aarts, E.; Bahr, B.A.; Bakker, M.; Banati, D.; Calabrese, V.; Cederholm, T.; Cryan, J.; et al. Poor cognitive ageing: Vulnerabilities, mechanisms and the impact of nutritional interventions. Ageing Res. Rev. 2018, 42, 40–55. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, V.; Giordano, J.; Crupi, R.; Di Paola, R.; Ruggieri, M.; Bianchini, R.; Ontario, M.L.; Cuzzocrea, S.; Calabrese, E.J. Hormesis, cellular stress response and neuroinflammation in schizophrenia: Early onset versus late onset state. J. Neurosci. Res. 2017, 95, 1182–1193. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Mahapatra, K.K.; Mishra, S.R.; Mallick, S.; Negi, V.D.; Sarangi, I.; Patil, S.; Patra, S.K.; Bhutia, S.K. Bacopa monnieri inhibits apoptosis and senescence through mitophagy in human astrocytes. Food Chem. Toxicol. 2020, 141, 111367. [Google Scholar] [CrossRef]



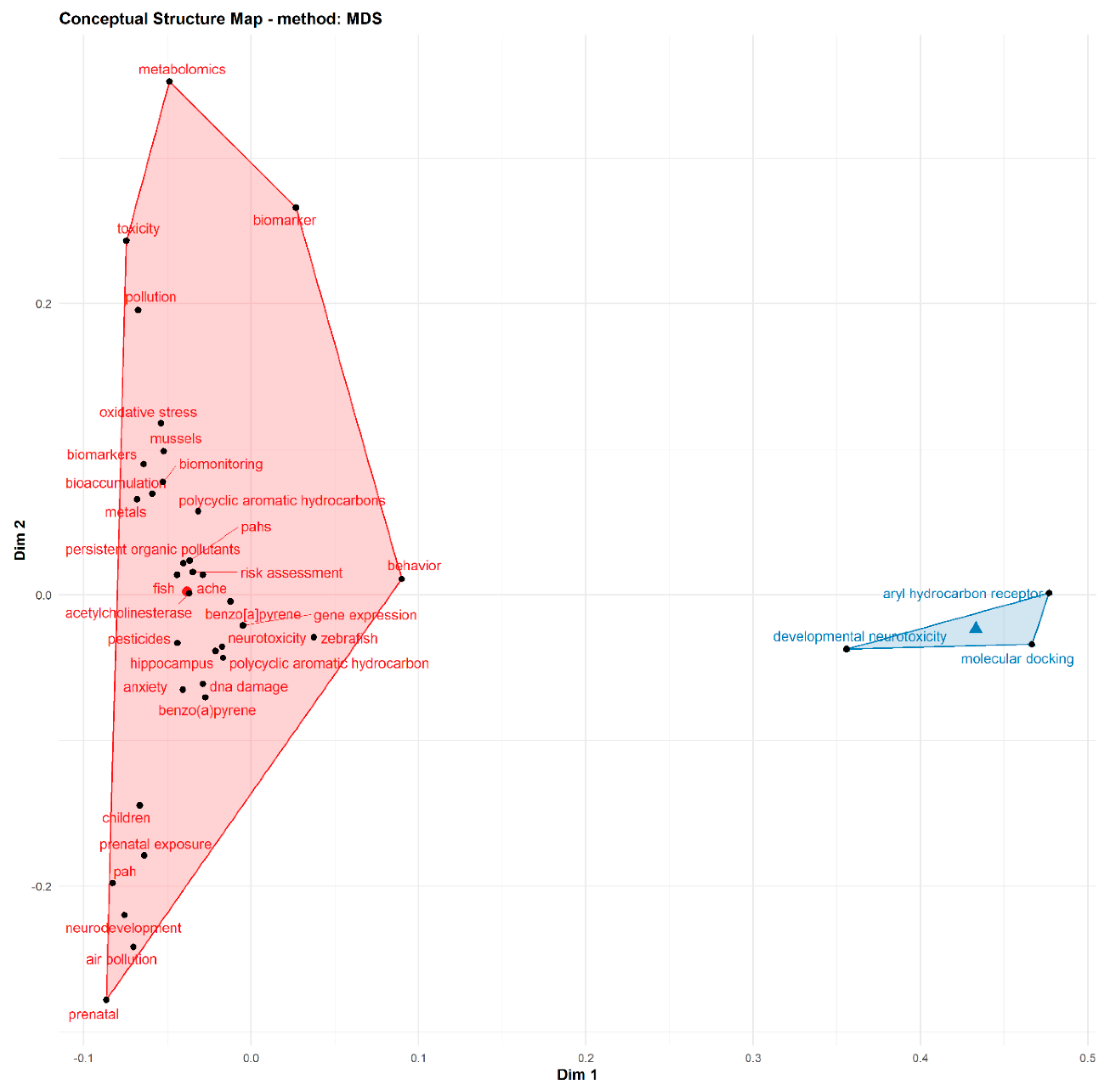
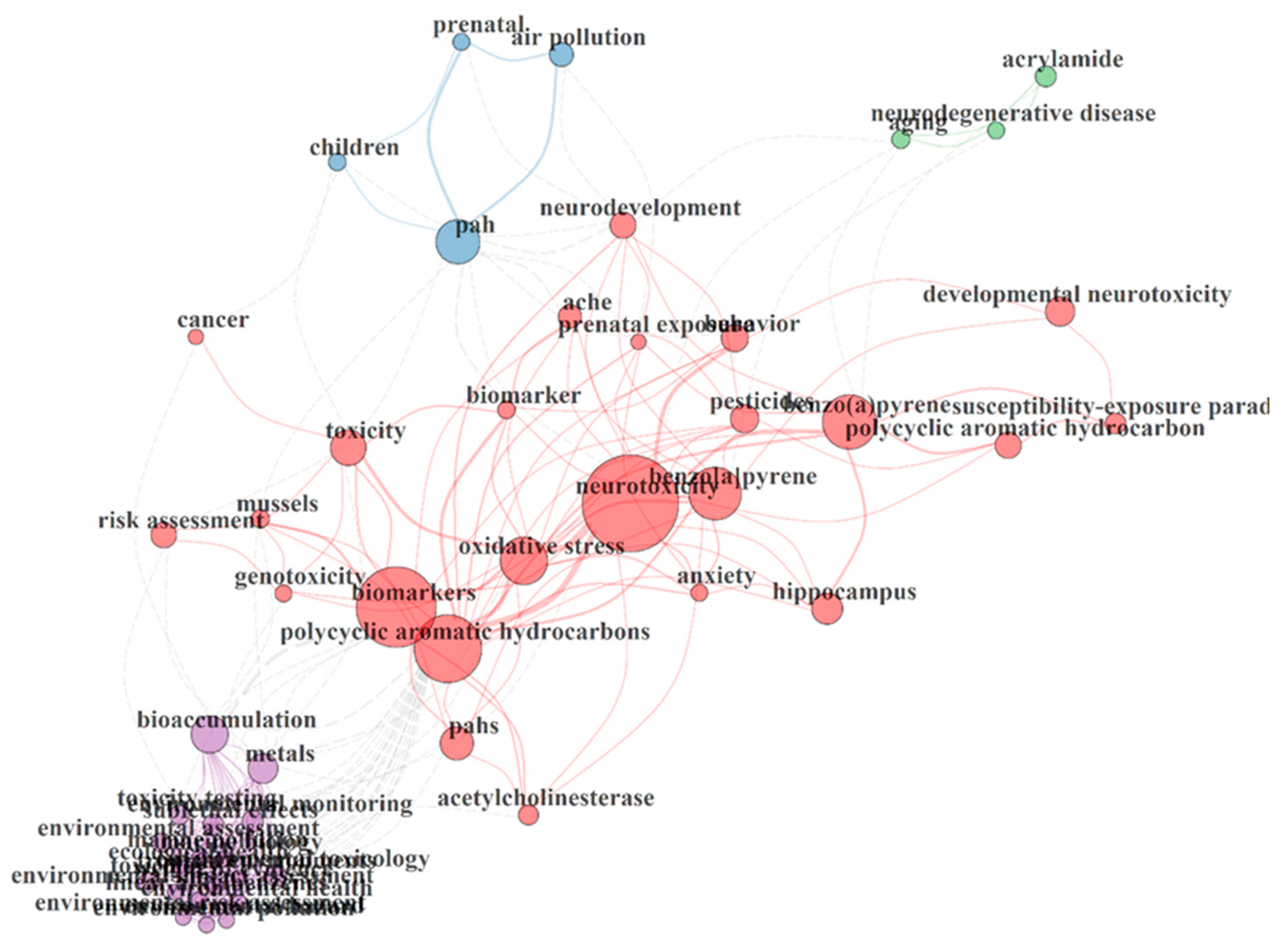
| Variable | Count/Rate | Authors | Articles |
|---|---|---|---|
| Documents | 258 | Perera F. | 14 |
| Sources (journals, books, etc.) | 129 | Ramesh A. | 10 |
| Keyword plus (ID) | 2970 | Raugh V. | 10 |
| Author’s keywords (DE) | 851 | Tang D. | 10 |
| Period | 1979–2020 | Hood D. | 9 |
| Average citations per document | 29.41 | Schroeder H. | 8 |
| Authors | 1192 | Das S. | 7 |
| Author’s appearances | 1592 | Budzinski H. | 6 |
| Authors of single-authored documents | 12 | Herbstman J. | 6 |
| Authors of multi-authored documents | 1180 | Rychen G. | 6 |
| Single authored documents | 13 | Wang S. | 6 |
| Documents per author | 0.216 | ||
| Authors per document | 4.62 | ||
| Co-authors per document | 6.17 | ||
| Collaboration index | 4.82 | ||
| Documents types | |||
| Article | 246 | ||
| Article, book chapter | 4 | ||
| Article, early access | 1 | ||
| Article, proceedings paper | 7 |
| Productivity Based on Articles Published | Productivity Based on Citations | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Rank | Country | Articles | Freq | SCP | MCP | MCP Ratio | Rank | Country | Total Citations | Average Total Citations |
| 1 | USA | 72 | 0.30769 | 64 | 8 | 0.1111 | 1 | USA | 2145 | 29.7 |
| 2 | China | 23 | 0.09829 | 18 | 5 | 0.2174 | 2 | Portugal | 699 | 63.5 |
| 3 | France | 18 | 0.07692 | 12 | 6 | 0.3333 | 3 | Canada | 643 | 71.4 |
| 4 | Italy | 14 | 0.05983 | 9 | 5 | 0.3571 | 4 | France | 597 | 33.2 |
| 5 | India | 12 | 0.05128 | 11 | 1 | 0.0833 | 5 | Italy | 453 | 32.4 |
| 6 | Portugal | 11 | 0.04701 | 5 | 6 | 0.5455 | 6 | China | 295 | 12.8 |
| 7 | Brazil | 10 | 0.04274 | 9 | 1 | 0.1 | 7 | Spain | 269 | 26.9 |
| 7 | Spain | 10 | 0.04274 | 9 | 1 | 0.1 | 8 | Sweden | 264 | 88 |
| 8 | Canada | 9 | 0.03846 | 8 | 1 | 0.1111 | 9 | United Kingdom | 228 | 45.6 |
| 9 | Germany | 7 | 0.02991 | 7 | 0 | 0 | 10 | Australia | 175 | 87.5 |
| 9 | Norway | 7 | 0.02991 | 5 | 2 | 0.2857 | Germany | 163 | 23.3 | |
| 10 | United Kingdom | 5 | 0.02137 | 5 | 0 | 0 | 10 | India | 163 | 13.6 |
| 11 | Finland | 3 | 0.01282 | 1 | 2 | 0.6667 | 11 | Poland | 118 | 39.3 |
| 11 | Korea | 3 | 0.01282 | 2 | 1 | 0.3333 | 12 | Belgium | 108 | 54 |
| 11 | Poland | 3 | 0.01282 | 2 | 1 | 0.3333 | 13 | Finland | 87 | 29 |
| 11 | Sweden | 3 | 0.01282 | 3 | 0 | 0 | 14 | Norway | 86 | 12.3 |
| 11 | Tunisia | 3 | 0.01282 | 0 | 3 | 1 | 15 | Brazil | 75 | 7.5 |
| 16 | Japan | 65 | 32.5 | |||||||
| 19 | Tunisia | 64 | 21.3 | |||||||
| 20 | Korea | 60 | 20 | |||||||
| Sources | Articles |
|---|---|
| Science Of The Total Environment | 16 |
| Neurotoxicology | 13 |
| Toxicological Sciences | 11 |
| Environmental Research | 9 |
| Environmental Science and Pollution Research | 9 |
| Aquatic Toxicology | 8 |
| Marine Pollution Bulletin | 7 |
| Environment International | 6 |
| Plos One | 6 |
| Toxicology Letters | 6 |
| Authors | Study Duration/Design/Model of Experiment | Sample Size | PAHs Investigated | Dosage/Route of Administration | Main Outcome Indicator | Neuropathological Mechanism |
|---|---|---|---|---|---|---|
| Das and Patri [49] | male Wistar pups (adolescent rats)/30 days | 54 | benzo[a]pyrene | 0.2 µg/kg BW/Intracisternal | 1, 2, 3, 4 |
|
| Liang, Tang, Duan, Cheng, Luo, Cao and Tu [50] | adult male rats/90 days | 96 | benzo[a]pyrene | 2 mg/kg BW/Intragastric | 1, 5, 6, 7 |
|
| Lin, Wu, Hu, Pai, Chen and Wang [48] | wild type and transgenic zebrafish (embryos)/20–22 h | benzo[a]pyrene | 10 and 20 µM | 1, 5, 8 |
| |
| Patel et al. [52] | male Wistar pups (5 day old)/30 days | 18 | benzo[a]pyrene | 0.2 µg/kg BW Intracisternal | 1, 2, 4, 7, 9, 10, 11, 12, 13 |
|
| Saunders, Das, Ramesh, Shockley and Mukherjee [39] | rats/2–96 h | 50 | benzo[a]pyrene | 25–200 mg/kg BW | 1, 5, 9, 13 | - Redox imbalance; - altered behavioural response. |
| Mohanty, Das and Patri [47] | adult zebrafish/7 days | benzo[a]pyrene | 1–4 nM | 1, 9, 10, 11, 15, 16 |
| |
| Gao et al. [53] | zebrafish/230 days | benzo[a]pyrene | 0.05–53.93 nmol/L | 15, 17, 18, 19 |
| |
| Mehri, Barangi, Zamiri and Karimi [46] | male Razi mice/28 days | 30 | benzo[a]pyrene | 75 mg/kg | 1, 12, 20, 21, 22, 23 |
|
| Xia et al. [54] | rats/13 weeks | benzo[a]pyrene | 2.5 and 6.25 mg/kg BW | 1, 5, 6, 24, 25,2 6, 27, 2829 |
| |
| Slotkin, Skavicus, Ko, Levin and Seidler [44] | sprague dawley rats/ | benzo[a]pyrene | 30 mg/kg/day | 26, 29 |
| |
| Gauthier et al. [55] | amphipod—Hyalella azteca | phenanthrene | 195 µg L−1 | 24, 30 |
| |
| Saunders et al. [56] | F-344 rats | fluoranthene | 100–400 mg/kg |
| ||
| He et al. [57] | rock fish (Sebastiscus marmoratus) | pyrene | 0.5 and 50 nmol/L | 24, 26, 31, 32 |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olasehinde, T.A.; Olaniran, A.O. Neurotoxicity of Polycyclic Aromatic Hydrocarbons: A Systematic Mapping and Review of Neuropathological Mechanisms. Toxics 2022, 10, 417. https://doi.org/10.3390/toxics10080417
Olasehinde TA, Olaniran AO. Neurotoxicity of Polycyclic Aromatic Hydrocarbons: A Systematic Mapping and Review of Neuropathological Mechanisms. Toxics. 2022; 10(8):417. https://doi.org/10.3390/toxics10080417
Chicago/Turabian StyleOlasehinde, Tosin A., and Ademola O. Olaniran. 2022. "Neurotoxicity of Polycyclic Aromatic Hydrocarbons: A Systematic Mapping and Review of Neuropathological Mechanisms" Toxics 10, no. 8: 417. https://doi.org/10.3390/toxics10080417
APA StyleOlasehinde, T. A., & Olaniran, A. O. (2022). Neurotoxicity of Polycyclic Aromatic Hydrocarbons: A Systematic Mapping and Review of Neuropathological Mechanisms. Toxics, 10(8), 417. https://doi.org/10.3390/toxics10080417






