Adsorption Characteristics of Modified Bamboo Charcoal on Cu(II) and Cd(II) in Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Main Experimental Apparatus and Materials
2.2. Preparation Method of Bamboo Charcoal
3. Results
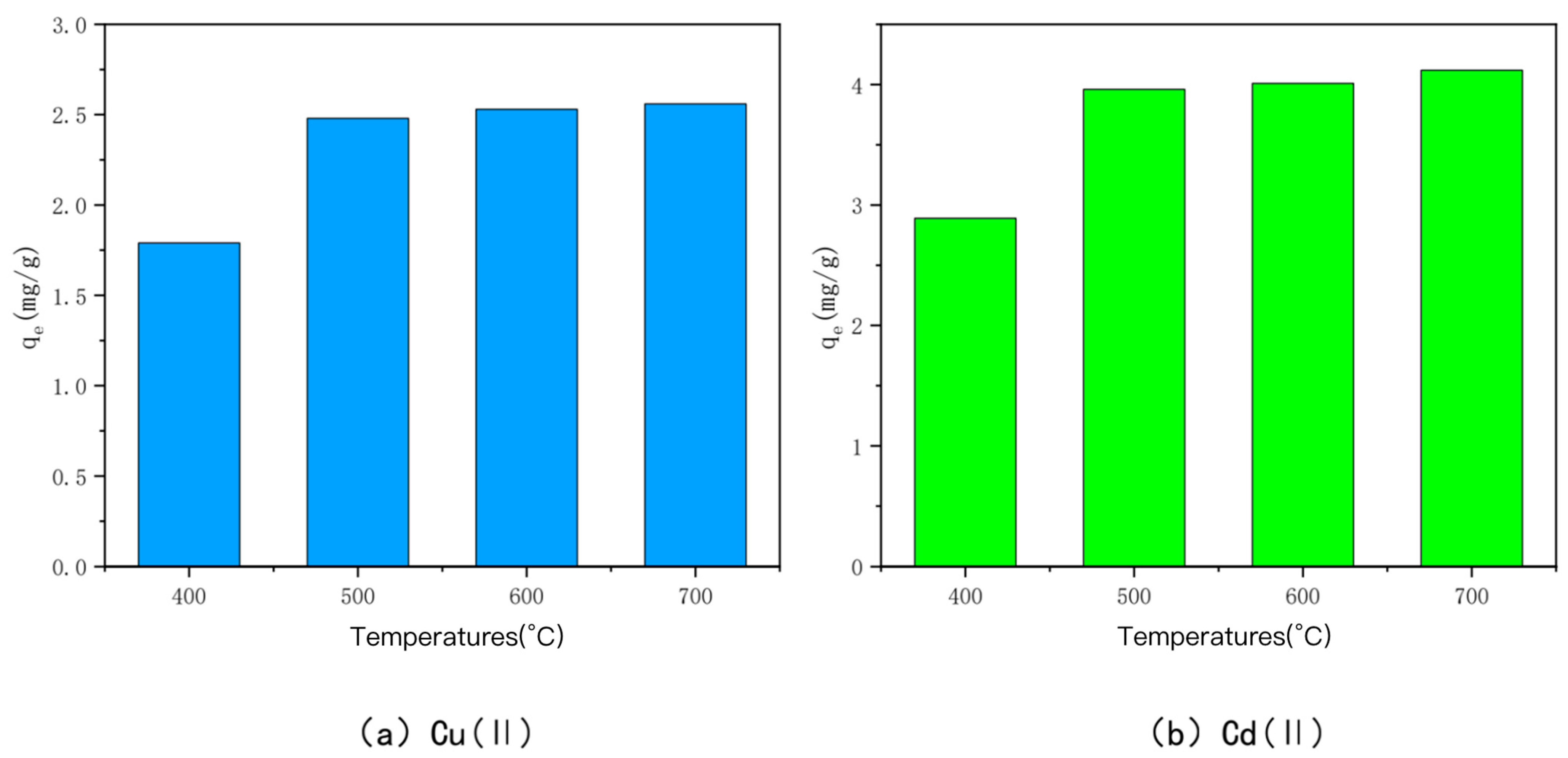
3.1. Effect of Charring Temperature
3.2. Effects of Different Modification Methods
- (1)
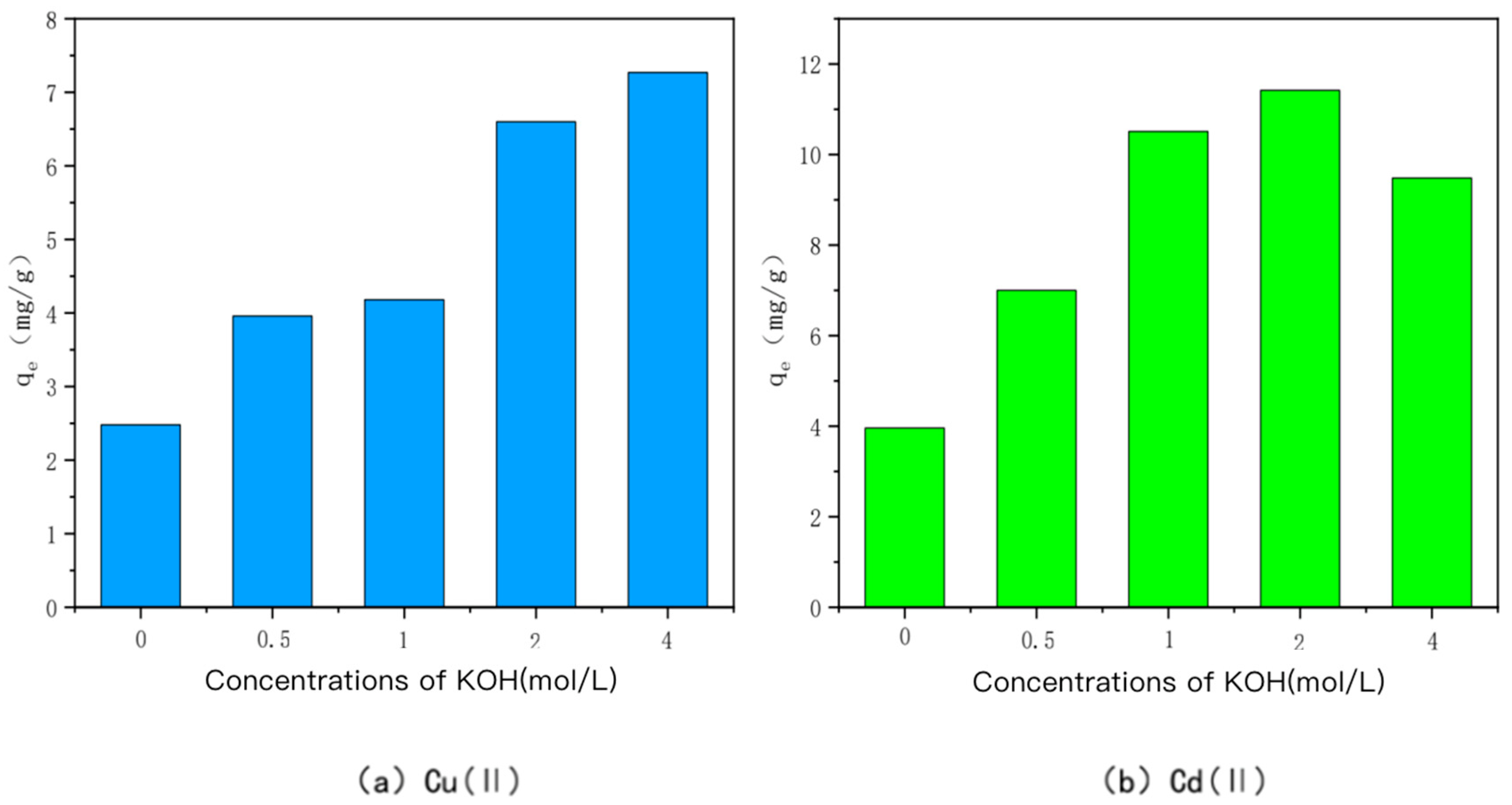
- KOH-modified bamboo charcoal
- (2)
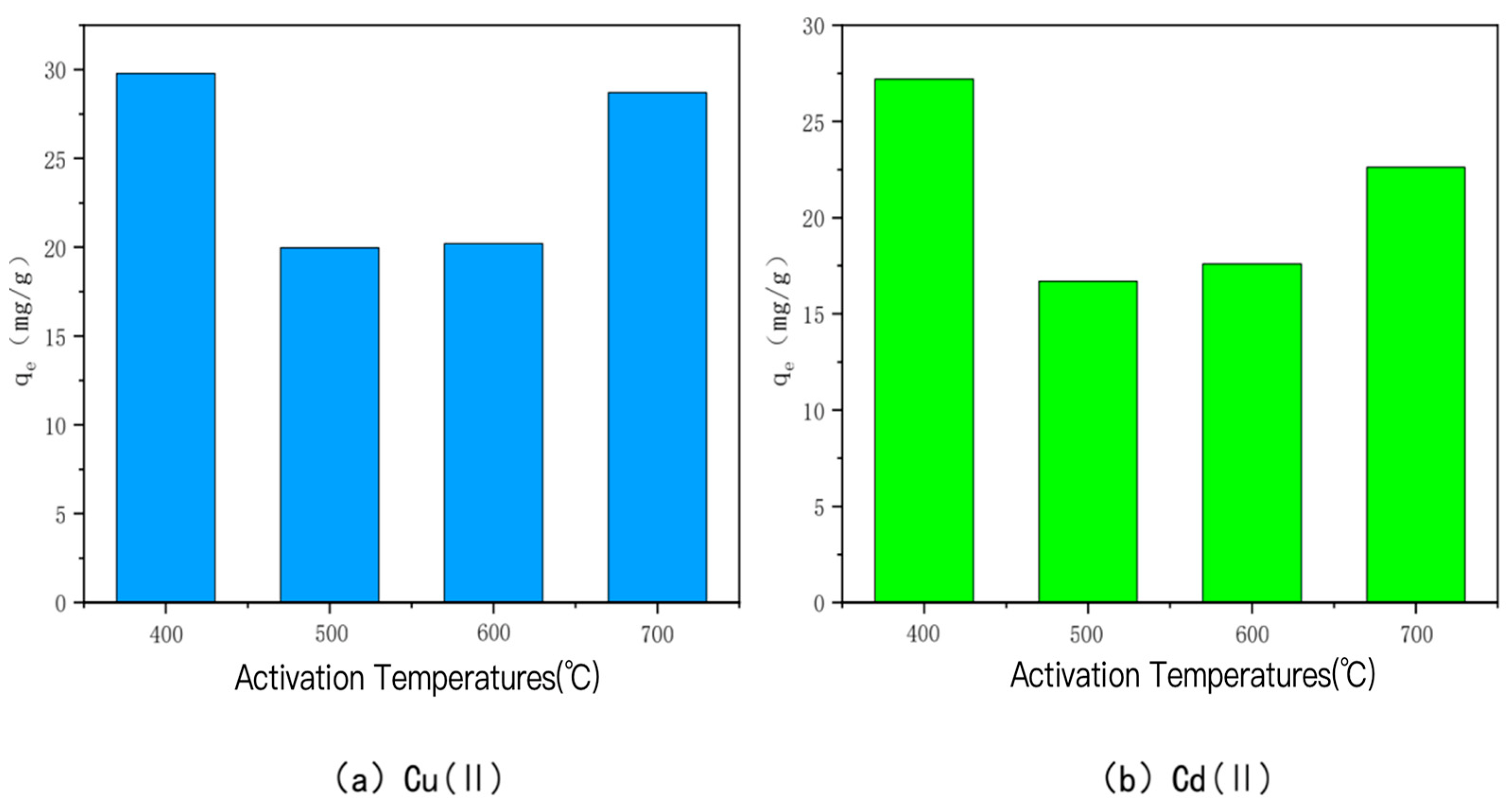
- KOH-activated bamboo charcoal
- ①
- Charcoal-to-alkali ratio
- ②
- Temperature
- (3)
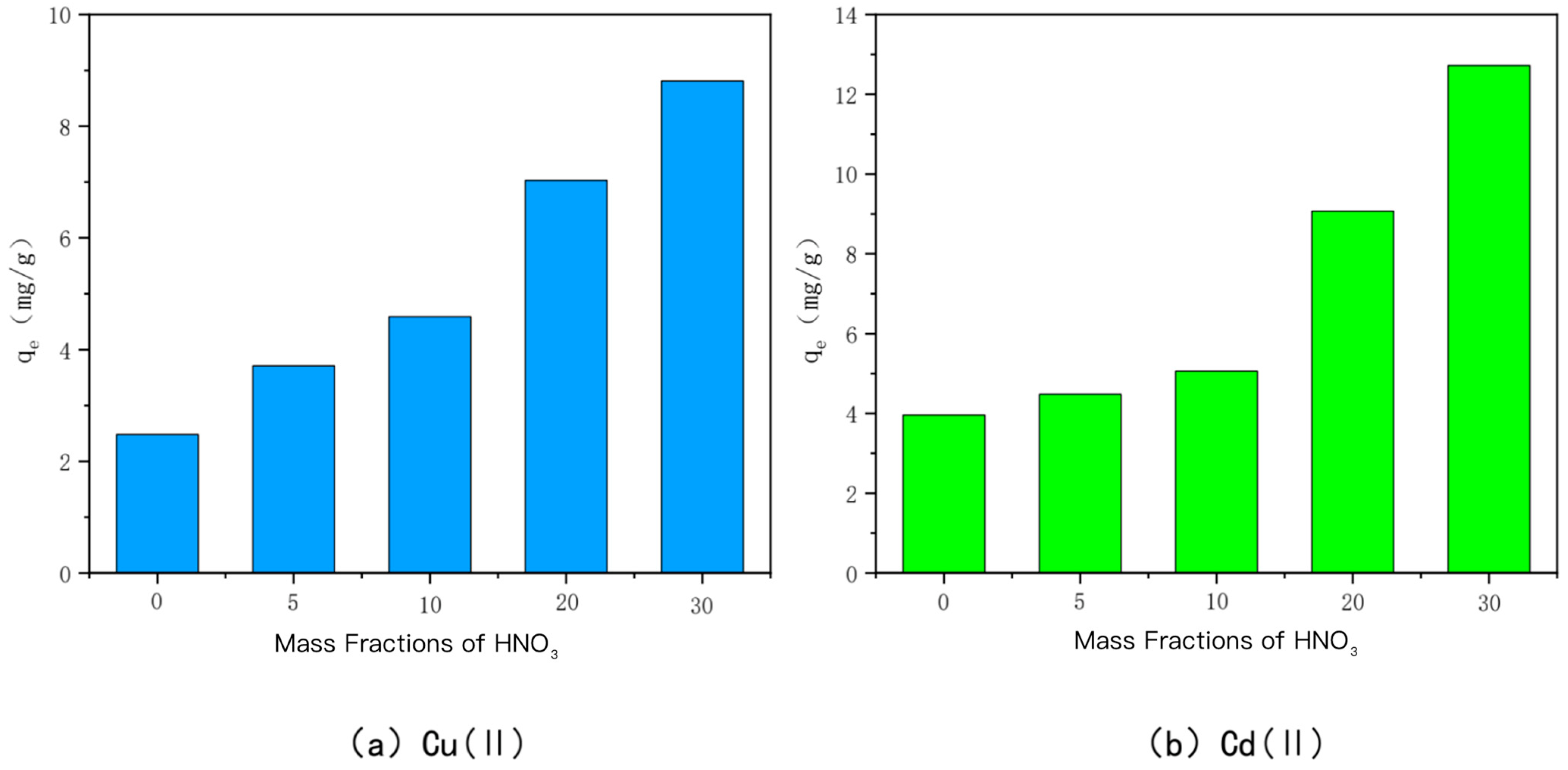
- HNO3-modified bamboo charcoal
3.3. Characterization of Modified Bamboo Charcoal
3.3.1. Morphological Analysis
3.3.2. Measurement of Specific Surface Area
3.3.3. Surface Functional Group Determination
3.3.4. Element Distribution
3.4. Effect of Solution Concentration
3.5. Effect of Solution pH
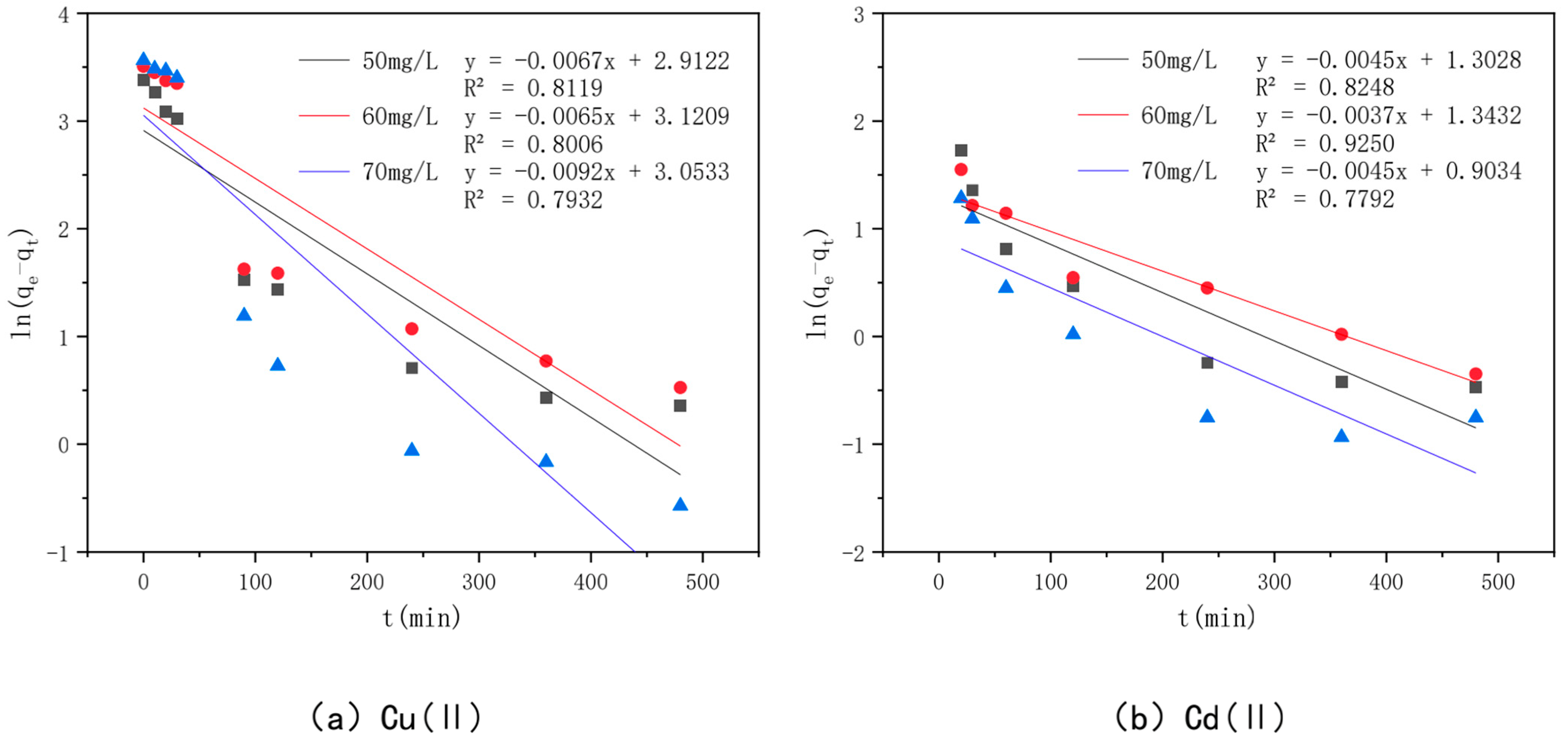
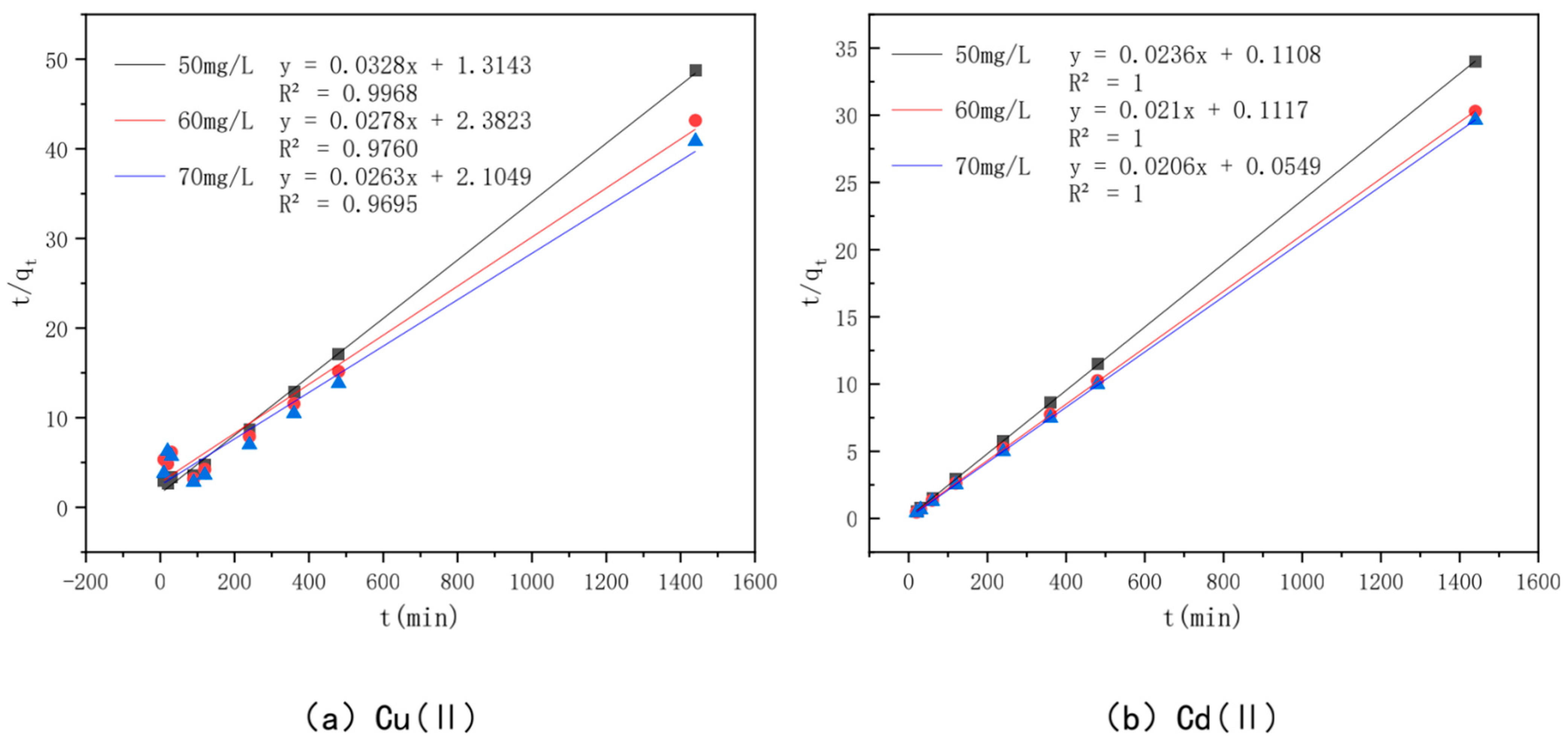
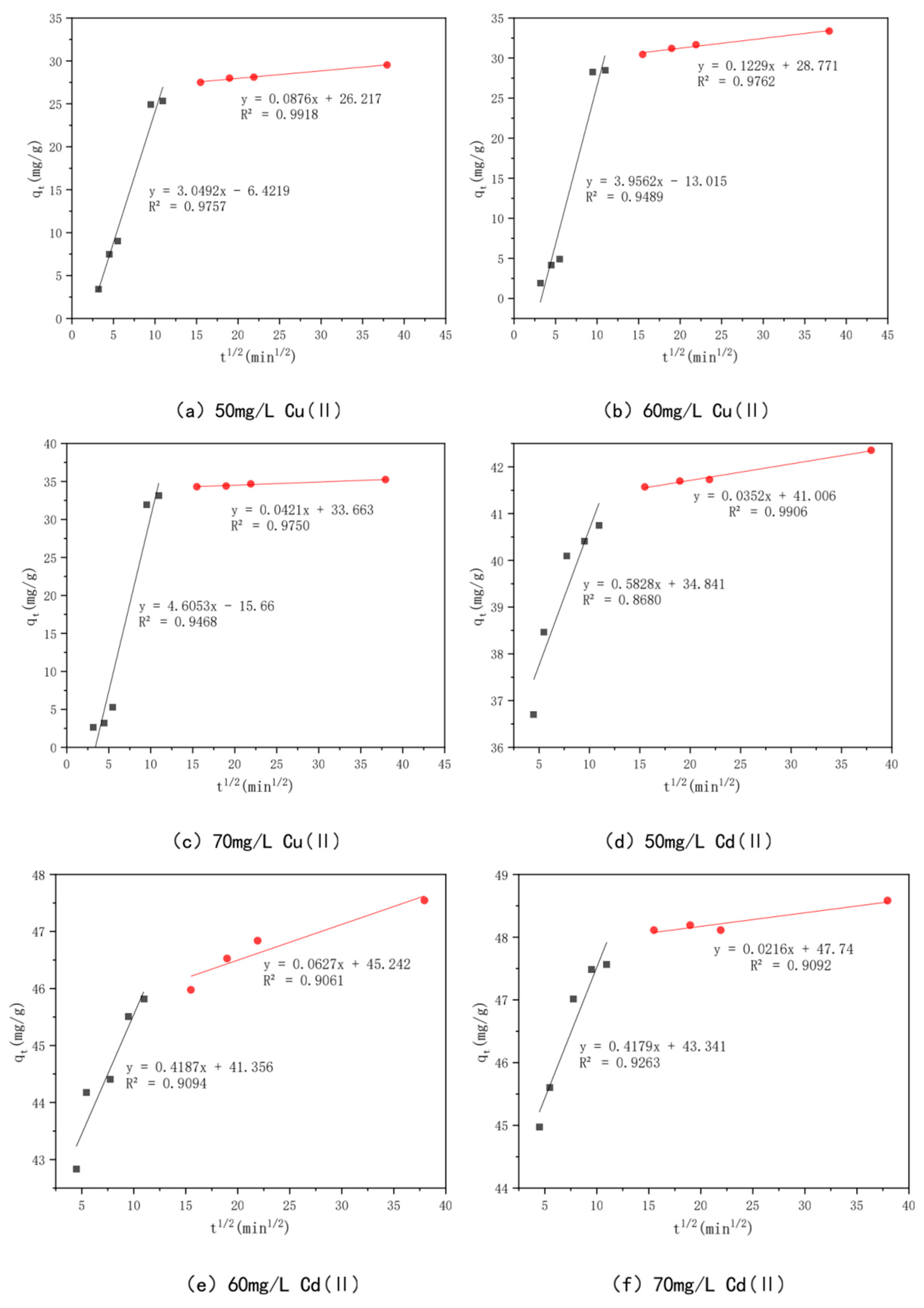
3.6. Effect of Adsorption Time and Adsorption Kinetics
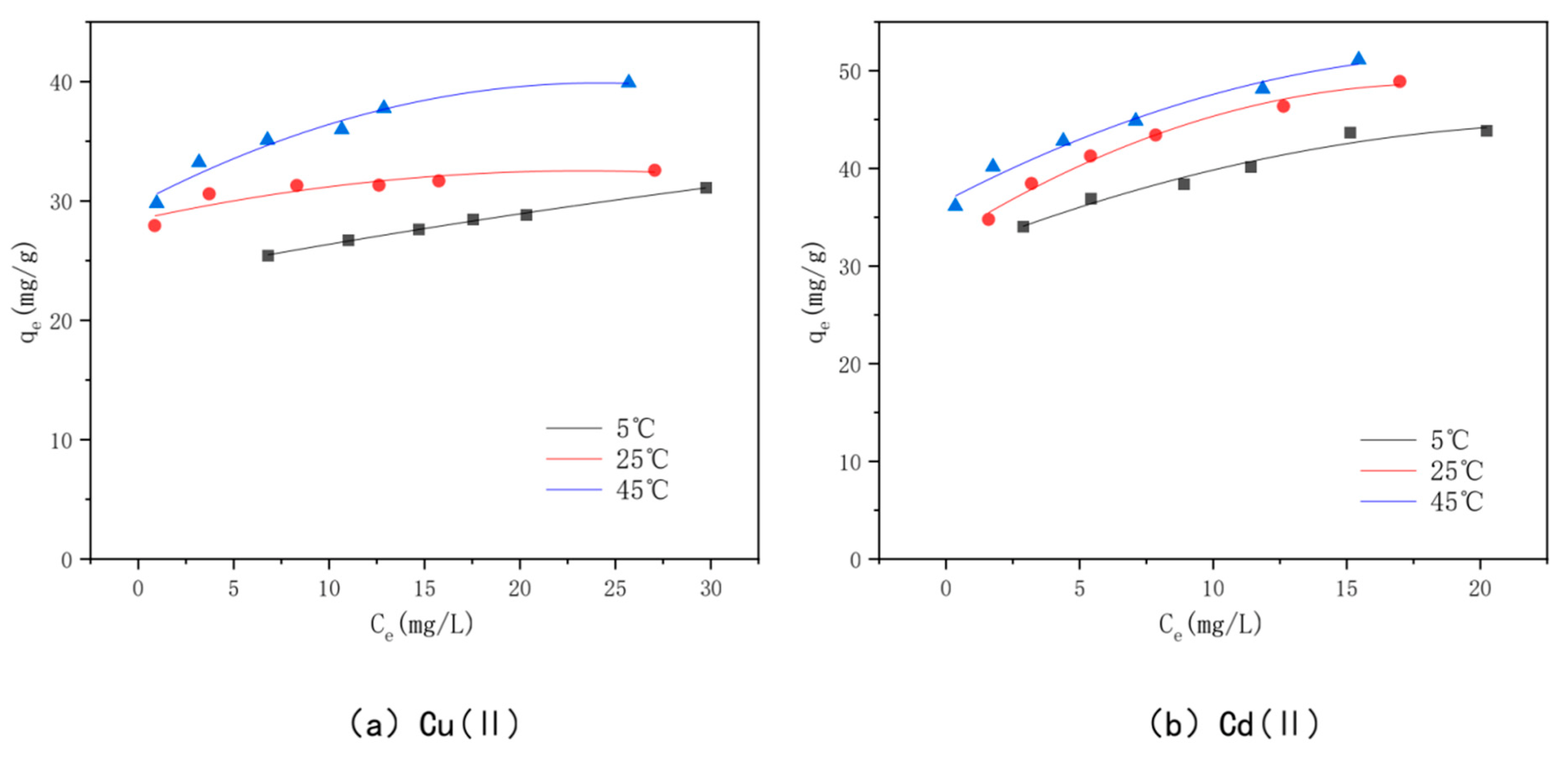
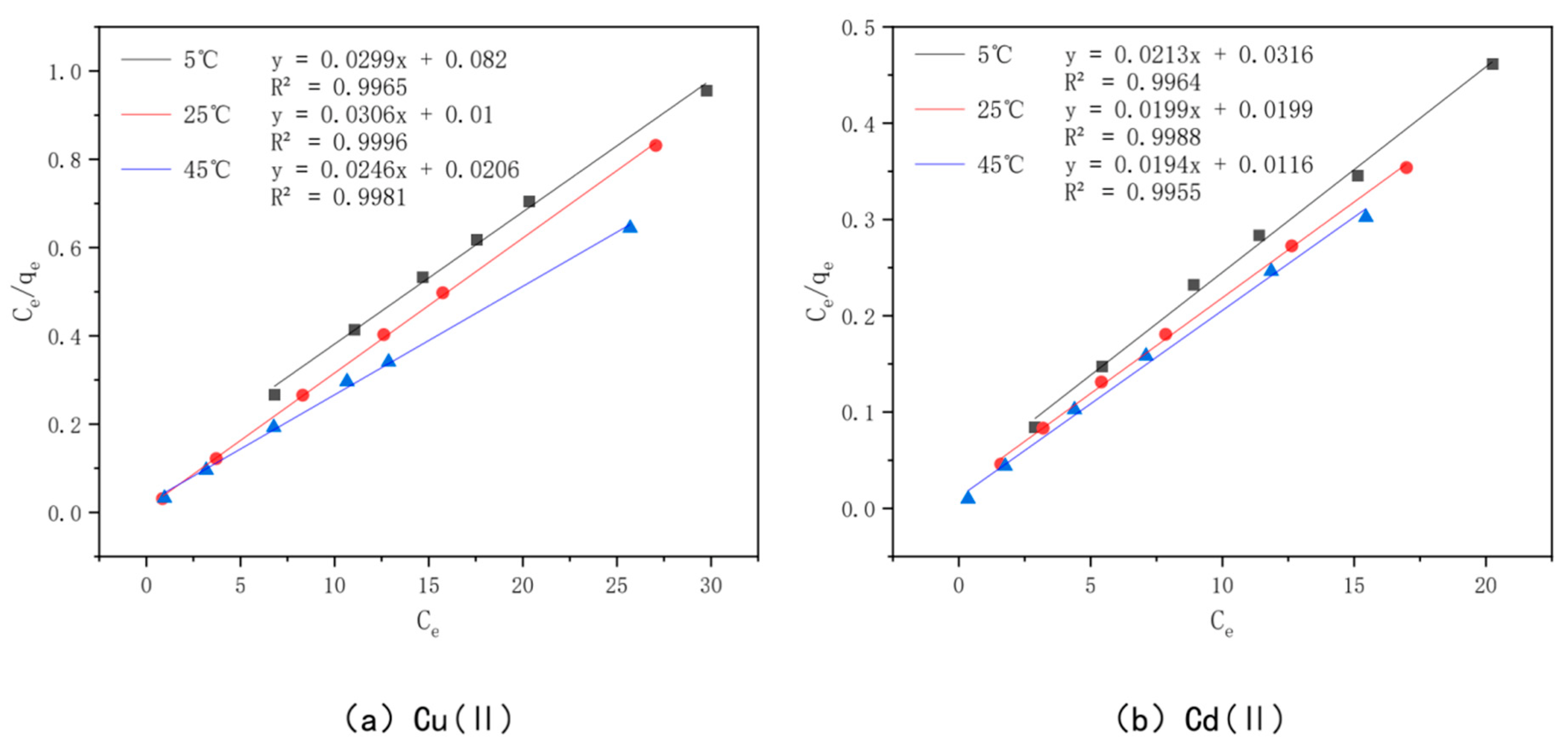
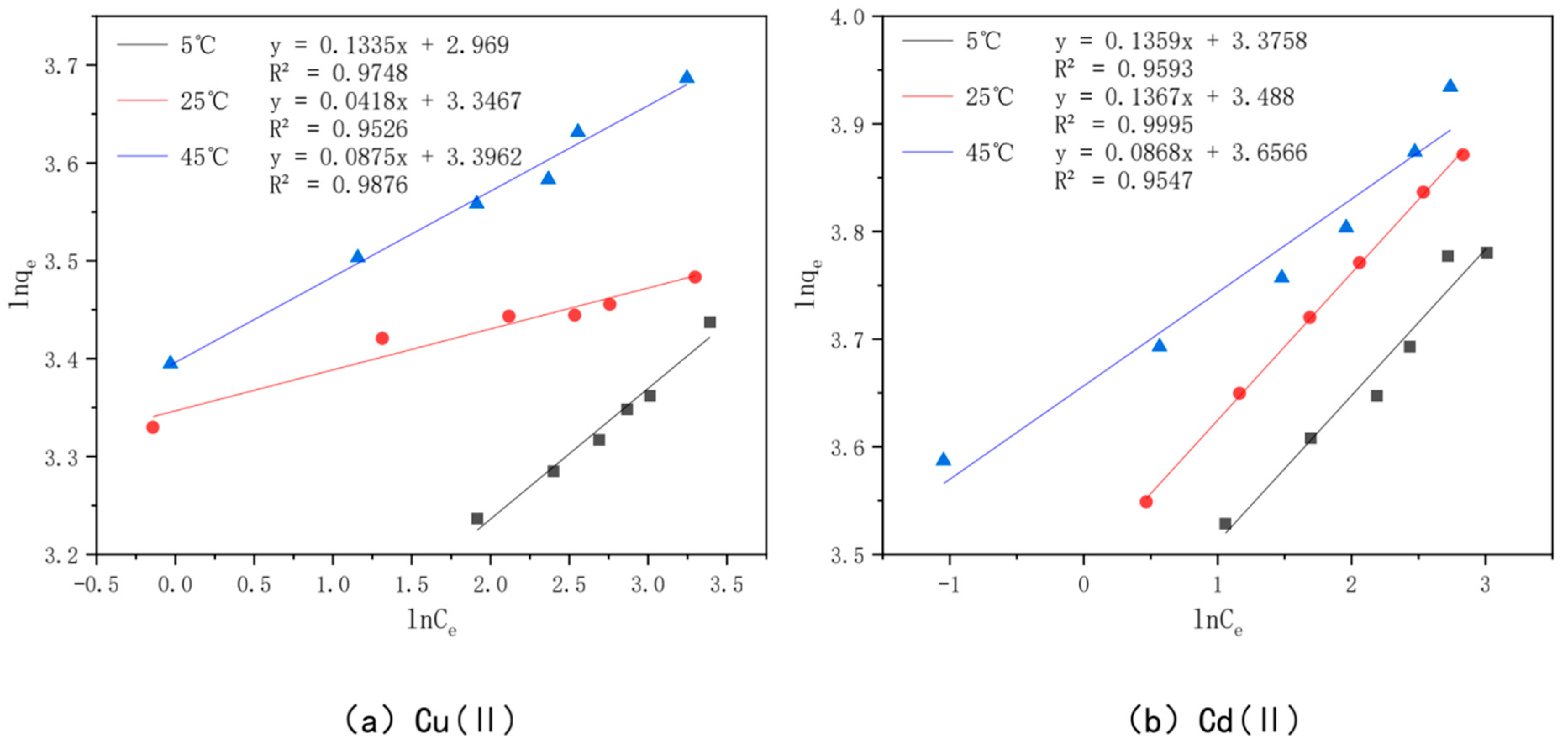
3.7. Adsorption Isotherms
3.8. Adsorption Thermodynamics
4. Conclusions
- (1)
- The adsorption of heavy metals Cu(II) and Cd(II) by KAM500-400-3 is affected by solution concentration, adsorption time, pH, temperature, and other factors. When the initial concentration of heavy metals Cu(II) and Cd(II) increases, the adsorption amount also increases, and the relatively higher pH (pH = 6) and temperature are more favorable to the adsorption of the optimal modified effect carbon for heavy metals Cu(II) and Cd(II);
- (2)
- The results of the adsorption kinetic fitting showed that both the proposed primary and secondary kinetics could describe the process of Cu(II) and Cd(II) adsorption by KAM500-400-3 well, but the latter was a better fit. This indicates that chemisorption and physical adsorption of Cu(II) and Cd(II) by KAM500-400-3 occur simultaneously, with chemisorption dominating. This chemisorption involves electron covalency or electron migration between the adsorbed material and the adsorbent, and physical adsorption such as precipitation and ion exchange may also exist.
- (1)
- The biomass material selected for this experiment was Moso bamboo. In future studies, we can try to select a variety of biomass materials for carbonization and modification to investigate their adsorption capacity for Cu(II) and Cd(II) in water as well as their immobilization effect on Cu(II) and Cd(II) contaminated sediment, and to determine the optimal biomass material;
- (2)
- The modification methods adopted in this experiment are mainly acid–base surface modification and activation modification. In future studies, we can also try redox modification by adding metal salts to biochar or adsorbent compound modification, or combine several modification methods to explore a better modification method for biochar [87];
- (3)
- In this experiment, we only investigated the adsorption of Cu(II) and Cd(II) in the water body; we could consider the competition of heavy metals adsorption in the water body with mixed metals in a subsequent study;
- (4)
- This experiment did not consider the regeneration and reuse of modified bamboo charcoal adsorbed with Cu(II) and Cd(II). In subsequent experiments, we may consider regenerating the contaminated modified bamboo charcoal by acid regeneration and thermal regeneration, investigate the number of times it can be regenerated, and compare the effectiveness of each method in regenerating the modified bamboo charcoal.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wang, J. Research progress of source and treatment methods of heavy metals in water. Guangdong Chem. Ind. 2014, 41, 87–88. [Google Scholar]
- Chen, G. Electrochemical technologies in wastewater treatment. Sep. Purif. Technol. 2004, 38, 11–41. [Google Scholar]
- Yi, Y.; Yang, Z.; Zhang, S. Ecological risk assessment of heavy metals in sediment and human health risk assessment of heavy metals in fishes in the middle and lower reaches of the Yangtze River basin. Environ. Pollut. 2011, 159, 2575–2585. [Google Scholar] [CrossRef]
- Fang, J.; Xiong, Y.; Wu, F.; Wang, S.; Yang, H.; Xie, W.; Xie, Y. Composition and source identification of biomarkers in surface sediments from typical freshwater lakes in China. Environ. Pollut. Control 2017, 39, 822–828. [Google Scholar]
- Wang, T. The Effects of Additives on Adsorption of Heavy Metals onto Surficial Sediments. Master’s Thesis, Jilin University, Nanjing, China, 2008. [Google Scholar]
- Islam, M.S.; Ahmed, M.K.; Raknuzzaman, M.; Habibullah-Al-Mamun, M.; Islam, M.K. Heavy metal pollution in surface water and sediment: A preliminary assessment of an urban river in a developing country. Ecol. Indic. 2015, 48, 282–291. [Google Scholar] [CrossRef]
- Chang, T.C.; Yen, J.H. On-site mercury-contaminated soils remediation by using thermal desorption technology. J. Hazard. Mater. 2006, 128, 208–217. [Google Scholar] [CrossRef]
- Wang, F.; Bao, K.; Huang, C.; Zhao, X.; Han, W.; Yin, Z. Adsorption and pH values determine the distribution of cadmium in terrestrial and marine soils in the Nansha area, Pearl River Delta. Int. J. Environ. Res. Public Health 2022, 19, 793. [Google Scholar] [CrossRef]
- Diao, Z.; Shi, T.; Wang, S.; Huang, X.; Zhang, T.; Tang, Y.; Zhang, X.; Qiu, R. Silane-based coatings on the pyrite for remediation of acid mine drainage. Water Res. 2013, 47, 4391–4402. [Google Scholar] [CrossRef]
- Tan, X.; Liu, Y.; Zeng, G.; Wang, X.; Hu, X.; Gu, Y.; Yang, Z. Application of biochar for the removal of pollutants from aqueous solutions. Chemosphere 2015, 125, 70–85. [Google Scholar] [CrossRef]
- Inyang, M.I.; Gao, B.; Yao, Y.; Xue, Y.; Zimmerman, A.; Mosa, A.; Pullammanappallil, P.; Ok, Y.S.; Cao, X. A review of biochar as a low-cost adsorbent for aqueous heavy metal removal. Crit. Rev. Environ. Sci. Technol. 2016, 46, 406–433. [Google Scholar] [CrossRef]
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L. Effects of biochar and greenwaste compost amendments on mobility, bioavailability and toxicity of inorganic and organic contaminants in a multi-element polluted soil. Environ. Pollut. 2010, 158, 2282–2287. [Google Scholar] [CrossRef] [PubMed]
- Higashikawa, F.S.; Conz, R.F.; Colzato, M. Effects of feedstock type and slow pyrolysis temperature in the production of biochars on the removal of cadmium and nickel from water. J. Clean. Prod. 2016, 137, 965–972. [Google Scholar] [CrossRef]
- Qian, L.; Zhang, W.; Yan, J.; Han, L.; Gao, W.; Liu, R.; Chen, M. Effective removal of heavy metal by biochar colloids under different pyrolysis temperatures. Bioresour. Technol. 2016, 206, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Zimmerman, A.R.; Harris, W. Surface chemistry variations among a series of laboratory-produced biochars. Geoderma 2011, 163, 247–255. [Google Scholar] [CrossRef]
- Zhuang, C.; Yuan, L. Adsorption characteristics of modified biochar on Cr. J. Fudan 2021, 60, 779–788. [Google Scholar]
- Tong, X. Removal of Cu(II) from Aqueous Solutions and Its Fixation in Red Soil by Biochars from Crop Straws. Master’s Thesis, Nanjing Agricultural University, Nanjing, China, 2011. [Google Scholar]
- Dong, X.; Ma, L.; Zhu, Y.J.; Li, Y.C.; Gu, B.H. Mechanistic investigation of mercury sorption by Brazilian pepper biochars of different pyrolytic temperatures based on X-ray photoelectron spectroscopy and flow calorimetry. J. Environ. Sci. Technol. 2013, 47, 12156–12164. [Google Scholar] [CrossRef]
- Li, H.; Dong, X.; da Silva, E.B.; de Oliveira, L.M.; Chen, Y.; Ma, L.Q. Mechanisms of metal sorption by biochars: Biochar characteristics and modifications. Chemosphere 2017, 178, 466–478. [Google Scholar]
- Li, A.Y.; Deng, H.; Jiang, Y.H.; Ye, C.H.; Yu, B.G.; Zhou, X.L.; Ma, A.Y. Superefficient removal of heavy metals from wastewater by Mg-loaded biochars: Adsorption characteristics and removal mechanisms. Langmuir 2020, 36, 9160–9174. [Google Scholar] [CrossRef]
- Alothman, Z.A.; Yilmaz, E.; Habila, M.; Soylak, M. Solid phase extraction of metal ions in environmental samples on 1-(2-pyridylazo)-2-naphthol impregnated activated carbon cloth. Ecotoxicol. Environ. Saf. 2015, 112, 74–79. [Google Scholar]
- Kadirvelu, K.; Faur-Brasquet, C.; Le Cloirec, P. Removal of Cu(II), Pb(II), and Ni(II) by adsorption onto activated carbon cloths. Langmuir 2000, 16, 8404–8409. [Google Scholar] [CrossRef]
- Wang, F.; Shih, K.M.; Li, X.Y. The partition behavior of perfluorooctanesulfonate (PFOS) and perfluorooctanesulfonamide (FOSA) on microplastics. Chemosphere 2015, 119, 841–847. [Google Scholar] [PubMed]
- Chen, J.Y.; Zhu, D.Q.; Sun, C. Effect of heavy metals on the sorption of hydrophobic organic compounds to wood charcoal. Environ. Sci. Technol. 2007, 41, 2536–2541. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Cao, X.; Zhao, L. Comparison of rice husk- and dairy manure-derived biochars for simultaneously removing heavy metals from aqueous solutions: Role of mineral components in biochars. Chemosphere 2013, 92, 955–961. [Google Scholar] [CrossRef] [PubMed]
- Zama, E.F.; Zhu, Y.G.; Reid, B.J.; Sun, G.X. The role of biochar properties in influencing the sorption and desorption of Pb(II), Cd(II) and As(III) in aqueous solution. J. Clean. Prod. 2017, 148, 127–136. [Google Scholar] [CrossRef]
- Mohan, D.; Sarswat, A.; Ok, Y.S.; Pittman, C.U. Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent—A critical review. Bioresour. Technol. 2014, 160, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Singh, E.; Kumar, A.; Mishra, R.; You, S.; Singh, L.; Kumar, S.; Kumar, R. Pyrolysis of waste biomass and plastics for production of biochar and its use for removal of heavy metals from aqueous solution. Bioresour. Technol. 2021, 320, 124278. [Google Scholar] [CrossRef]
- Park, J.H.; Ok, Y.S.; Kim, S.H.; Cho, J.S.; Heo, J.S.; Delaune, R.D.; Seo, D.C. Competitive adsorption of heavy metals onto sesame straw biochar in aqueous solutions. Chemosphere 2016, 142, 77–83. [Google Scholar] [CrossRef]
- Prochaska, J.O.; DiClemente, C.C.; Norcross, J.C. In search of how people change: Applications to addictive behaviors. Am. Psychol. 1992, 47, 1102–1114. [Google Scholar] [CrossRef]
- Su, H.; Fang, Z.; Tsang, P.E.; Fang, J.; Zhao, D. Stabilisation of nanoscale zero-valent iron with biochar for enhanced transport and in-situ remediation of hexavalent chromium in soil. Environ. Pollut. 2016, 214, 94–100. [Google Scholar] [CrossRef]
- Kaczala, F.; Marques, M.; Hogland, W. Lead and vanadium removal from a real industrial wastewater by gravitational settling/sedimentation and sorption onto Pinus sylvestris sawdust. Bioresour. Technol. 2009, 100, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Velkova, Z.; Kirova, G.; Stoytcheva, M.; Kostadinova, S.; Todorova, K.; Gochev, V. Immobilized microbial biosorbents for heavy metals removal. Eng. Life Sci. 2018, 18, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.P.; Zhang, Z.Y.; Liu, X.Y.; Che, S.; Shi, N.W.; Chen, Y.M.; Yan, M.Y. Adsorption behavior and mechanism of Lead (Pb2+) by sulfate polysaccharide from Enteromorpha prolifera. Int. J. Biol. Macromol. Adv. 2022, 207, 760–770. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Kuila, A. Bioremediation of heavy metals by microbial process. Environ. Technol. Innov. 2019, 14, 100369. [Google Scholar]
- Patel, P.R.; Shaikh, S.S.; Sayyed, R.Z. Dynamism of PGPR in bioremediation and plant growth promotion in heavy metal contaminated soil. Indian J. Exp. Biol. 2016, 54, 286–290. [Google Scholar] [PubMed]
- Sharma, S.; Tiwari, S.; Hasan, A.; Saxena, V.; Pandey, L.M. Recent advances in conventional and contemporary methods for remediation of heavy metal-contaminated soils. 3 Biotech 2018, 8, 216–233. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhou, K.; Qin, W.; Tian, C.; Qi, M.; Yan, X.; Han, W. A review on heavy metals contamination in soil: Effects, sources, and remediation techniques. Soil Sediment Contam. Int. J. 2019, 28, 380–394. [Google Scholar] [CrossRef]
- Song, J. Heavy Metal Pollution Characteristics of Shandong Peninsula River Sediment and Landfill Disposal Risk Assessment. Ph.D. Thesis, Yantai Institute of Coastal Zone, Chinese Academy of Sciences, Yantai, China, 2019. [Google Scholar]
- Wei, Z.; Shurong, C.; Ping, H. Heavy metal pollution of sediment and its potential ecological risk evaluation before and after dredging in Puyang River Basin. J. Zhejiang A F Univ. 2016, 33, 33–41. [Google Scholar]
- Shimin, Z.; Rongzhan, L.; Xuesong, G. Research progress of biochar preparation and application of sludge in sewage treatment plant. Chem. Prog. 2016, 35, 363–368. [Google Scholar]
- Hao, L.; Zhaohui, L.; Haiyan, Z. planted modified rice straw for the adsorption of Cd2+ in solution. Chin. Environ. Sci. 2018, 38, 596–607. [Google Scholar]
- Patra, J.M.; Panda, S.S.; Dhal, N.K. Biochar as a low-cost adsorbent for heavy metal removal: A review. Int. J. Res. Biosci. 2017, 6, 1–7. [Google Scholar]
- Wang, Y.; Gu, K.; Wang, H.S.; Shi, B.; Tang, C. Remediation of heavy-metal-contaminated soils by biochar: A review. J. Environ. Geotech. 2022, 9, 135–148. [Google Scholar] [CrossRef]
- Yi, Y.; Wang, X.; Zhang, Y.; Ma, J.; Ning, P. Adsorption properties and mechanism of Cr(VI) by Fe2(SO4)3 modified biochar derived from Egeria najas. J. Colloids Surf. a-Physicochem. Eng. Asp. 2022, 645, 128938. [Google Scholar] [CrossRef]
- Jing, D.; Yangsheng, L. The adsorption characteristics of Pb2+ and Cd2+ produced by the biochar generated from the pyrolysis of the four raw materials were studied. J. Peking Univ. 2013, 49, 1075–1082. [Google Scholar]
- Kumar, A.; Bhattacharya, T. Biochar: A sustainable solution. Environ. Dev. Sustain. 2021, 23, 6642–6680. [Google Scholar] [CrossRef]
- Dass, K.; Ghoshg, K.; Avasthe, R. Conversion of crop, weed and tree biomass into biochar for heavy metal removal and wastewater treatment. Biomass Convers. Biorefin. 2021, 1–14. [Google Scholar] [CrossRef]
- Jia, Y.; Shi, S.; Liu, J.; Su, S.; Liang, Q.; Zeng, X.B.; Li, T. Study of the effect of pyrolysis temperature on the Cd2+ adsorption characteristics of biochar. Appl. Sci. 2018, 8, 1019. [Google Scholar] [CrossRef]
- Xiao, Y.; Xue, Y.; Gao, F.; Mosa, A. Sorption of heavy metal ions onto crayfish shell biochar: Effect of pyrolysis temperature, pH and ionic strength. J. Taiwan Inst. Chem. Eng. 2017, 80, 114–121. [Google Scholar] [CrossRef]
- Ahmed, M.; Zhou, J.; Ngo, H.; Guo, W.; Chen, M. Progress in the preparation and application of modified biochar for improved contaminant removal from water and wastewater. J. Bioresour. Technol. 2016, 214, 836–851. [Google Scholar]
- Jie, Z.; Yuhong, H.; Xiaoming, Z. the influence of isoacid-base modification on Cr (W) performance. Environ. Eng. 2020, 38, 28–34. [Google Scholar]
- Peng, H.; Gao, P.; Chu, G.; Pan, B.; Peng, J.; Xing, B. Enhanced adsorption of Cu (II) and Cd (II) by phosphoric acid-modified biochars. Environ. Pollut. 2017, 229, 846–853. [Google Scholar]
- Herath, A.; Layne, C.A.; Perez, F.; Hassan, E.; Pittman, C.; Mlsna, T. KOH-activated high surface area Douglas Fir biochar for adsorbing aqueous Cr (VI), Pb (II) and Cd (II). Chemosphere 2021, 269, 128409. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.; Wang, Y.; Tian, X.; Jiang, Z.; Deng, F.; Tao, Y.; Jiang, Q.; Wang, L.; Zhang, Y. KOH-activated porous biochar with high specific surface area for adsorptive removal of chromium (VI) and naphthalene from water: Affecting factors, mechanisms and reusability exploration. J. Hazard. Mater. 2021, 401, 123292. [Google Scholar] [CrossRef]
- Monser, L.; Adhoum, N. Modified activated carbon for the removal of copper, zinc, chromium and cyanide from wastewater. Sep. Purif. Technol. 2002, 26, 137–146. [Google Scholar] [CrossRef]
- Yang, L.; Li, B.; Wang, C. passivation effect of modified biochar materials on original and exogenous cadmium contaminated soil in rice fields. Environ. Sci. 2016, 37, 3562–3574. [Google Scholar]
- Xing, B.; Chen, L.; Zhang, C.; Huang, G.; Ma, M. Study on the activation mechanism of lignite-based activated carbon prepared by KOH activation method. J. China Univ. Min. Technol. 2014, 43, 1038–1045. [Google Scholar]
- Li, W.; Zhang, C.; University, S. Research progress on the remediation of soil heavy metal pollution by biochar. Biol. Chem. Eng. 2019, 39, 5266–5274. [Google Scholar]
- Bian, R.; Chen, D.; Liu, X. Biochar soil amendment as a solution to prevent Cd-tainted rice from China: Results from a cross-site field experiment. Ecol. Eng. 2013, 58, 378–383. [Google Scholar] [CrossRef]
- Lu, X.; Wu, J.; Zheng, Y. Passivation of cadmium in soil by walnut biochar. Environ. Eng. 2020, 38, 196–202. [Google Scholar]
- Tang, J.; Zhu, W.; Kookana, R.; Katayama, A. Characteristics of biochar and its application in remediation of contaminated soil. J. Biosci. Bioeng. 2013, 116, 653659. [Google Scholar] [CrossRef]
- Que, W.; Zhou, Y.; Liu, Y.; Wen, J.; Tan, X.; Liu, S.; Jiang, L. Appraising the effect of in-situ remediation of heavy metal contaminated sediment by biochar and activated carbon on Cu immobilization and microbial community. Ecol. Eng. 2019, 127, 519–526. [Google Scholar] [CrossRef]
- Zhang, C.; Shan, B.; Zhu, Y.; Wen, T. Remediation effectiveness of Phyllostachys pubescens biochar in reducing the bioavailability and bioaccumulation of metals in sediments. Environ. Pollut. 2018, 242, 1768–1776. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.; Huang, D.; Liu, Y.; Zeng, G.; Chen, S.; Wang, R.; Xu, P.; Cheng, M.; Zhang, C.; Xue, W. Biochar facilitated the phytoremediation of cadmium contaminated sediments: Metal behavior, plant toxicity, and microbial activity. Sci. Total Environ. 2019, 666, 1126–1133. [Google Scholar] [CrossRef] [PubMed]
- Zou, Q.; An, W.; Wu, C.; Li, W.; Fu, A.; Xiao, R.; Chen, H.; Xue, S. Red mud-modified biochar reduces soil arsenic availability and changes bacterial composition. Environ. Chem. Lett. 2018, 16, 615–622. [Google Scholar] [CrossRef]
- Liu, S.; Liu, Y.; Tan, X.; Zeng, G.; Zhou, Y.; Liu, S.; Yin, Z.; Jiang, L.; Li, M.; Wen, J. The effect of several activated biochars on Cd immobilization and microbial community composition during in-situ remediation of heavy metal contaminated sediment. Chemosphere 2018, 208, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Wang, P.; Chen, J.; Wang, S.; Yang, G. Effect of modified biochar material for in-situ restoration of contaminated sediment. J. Environ. Eng. Technol. 2020, 10, 661–670. [Google Scholar]
- Sun, T.; Li, K.; Fu, Y.; Ma, W.; Xie, X.; Sun, Y. Effect of modified biochar on passivation repair effect and soil environmental quality of weakly alkaline Cd contaminated soil. J. Environ. Sci. 2020, 40, 2571–2580. [Google Scholar]
- Ayar, A.; Gursal, S.; Gurten, A.A.; Gezici, O. On the removal of some phenolic compounds from aqueous solutions by using asporopollenin-based ligand-exchange fixed bed one Isotherm analysis. Desalination 2008, 219, 160–170. [Google Scholar] [CrossRef]
- Yan, T.; Li, Q.; Li, H.; Zhang, F. Dynamic adsorption and regeneration of Cr (VI) in water. New Chem. Mater. 2021, 49, 179–184. [Google Scholar]
- Bohart, G.S.; Adams, E.Q. Some aspects of the behavior of charcoal with respect to chlorine. J. Am. Chem. Soc. 1920, 42, 523–544. [Google Scholar] [CrossRef]
- Mistar, E.M.; Hasmita, I.; Alfatah, T.; Muslim, A.; Supardan, M. Adsorption of mercury (II) using activated carbon produced from Bambusa vulgaris var.striata in a fixed-bed column. Sains Malays. 2019, 48, 719–725. [Google Scholar] [CrossRef]
- Yuan, C.; Sun, Y.; Yang, Y.; Song, X.; Yu, J. Exploration and model fitting of D301 resin. J. Process Eng. 2020, 20, 655–666. [Google Scholar]
- Peng, K.; Kong, H.; Zhang, D.; Wu, D. Pyrolysis and reduction and harmless treatment of Cr (Qiao) contaminated soil. Environ. Pollut. Prev. 2009, 31, 31–35. [Google Scholar]
- Jin, H.; Capareda, S.; Chang, Z.; Gao, J.; Xu, Y.; Zhang, J. Biochar pyrolytically produced from municipal solid wastes for aqueous As (V) removal: Adsorption property and its improvement with KOH activation. Bioresour. Technol. 2014, 169, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.; Cheng, X.; Li, S.; Yang, Y. Preparation of cotton stalk-based activated carbon and its adsorption to phenol-containing wastewater by KOH activation method. Biomass Chem. Eng. 2019, 53, 13–18. [Google Scholar]
- Zhao, Y.; Gao, J.; Hao, X.; Chen, Y. Preparation and characterization of walnut-based activated carbon based on KOH activation method. J. Saf. Environ. 2016, 16, 262–266. [Google Scholar]
- Garcia, T.; Murillo, R.; Cazorla-Amoros, D.; Mastral, A.; Linares-Solano, A. Role of the activated carbon surface chemistry in the adsorption of phenanthrene. Carbon 2004, 42, 1683–1689. [Google Scholar] [CrossRef]
- Yi, P.; Wu, G.; Duan, W.; Wu, M.; Pan, B. Progress in the modification and aging and environmental effects of biochar. Mater. Guide 2020, 34, 43–49. [Google Scholar]
- Ertugay, N.; Bayhan, Y.K. Biosorption of Cr (VI) from aqueous solutions by biomass of Agaricus bisporus. J. Hazard. Mater. 2008, 154, 432–439. [Google Scholar] [CrossRef]
- Chen, Y.; Lyu, J.; Wang, Y.; Chen, T.; Tian, Y.; Bai, P.; Guo, X. Synthesis, characterization, adsorption, and isotopic separation studies of pyrocatechol-modified MCM-41 for efficient boron removal. Ind. Eng. Chem. Res. 2019, 58, 3282–3292. [Google Scholar] [CrossRef]
- Zhang, M.; Gao, B.; Varnoosfaderani, S.; Hebard, A.; Yao, Y.; Inyang, M. Preparation and characterization of a novel magnetic biochar for arsenic removal. Bioresour. Technol. 2013, 130, 457–462. [Google Scholar] [CrossRef]
- Chen, S.; Yue, Q.; Gao, B.; Li, Q.; Xu, X.; Fu, K. Adsorption of hexavalent chromium from aqueous solution by modified corn stalk: A fixed-bed column study. Bioresour. Technol. 2012, 113, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Malkoc, E.; Nuhoglu, Y.; Dundar, M. Adsorption of chromium(VI) on pomace one an olive oil industry waste: Batch and column studies. J. Hazard. Mater. 2006, 138, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Shen, Y.; Zhang, P.; Zhao, Q.; An, L. Adsorption of Pb and Cd in water by a steel slag fixation bed. Environ. Eng. 2016, 34, 25–30. [Google Scholar]
- Lu, X.; Zhong, L.; Meng, F. Study on the dynamic adsorption characteristics of Cr (VI) in simulated wastewater from peanut shell. Water Resour. Prot. 2013, 29, 87–90. [Google Scholar]


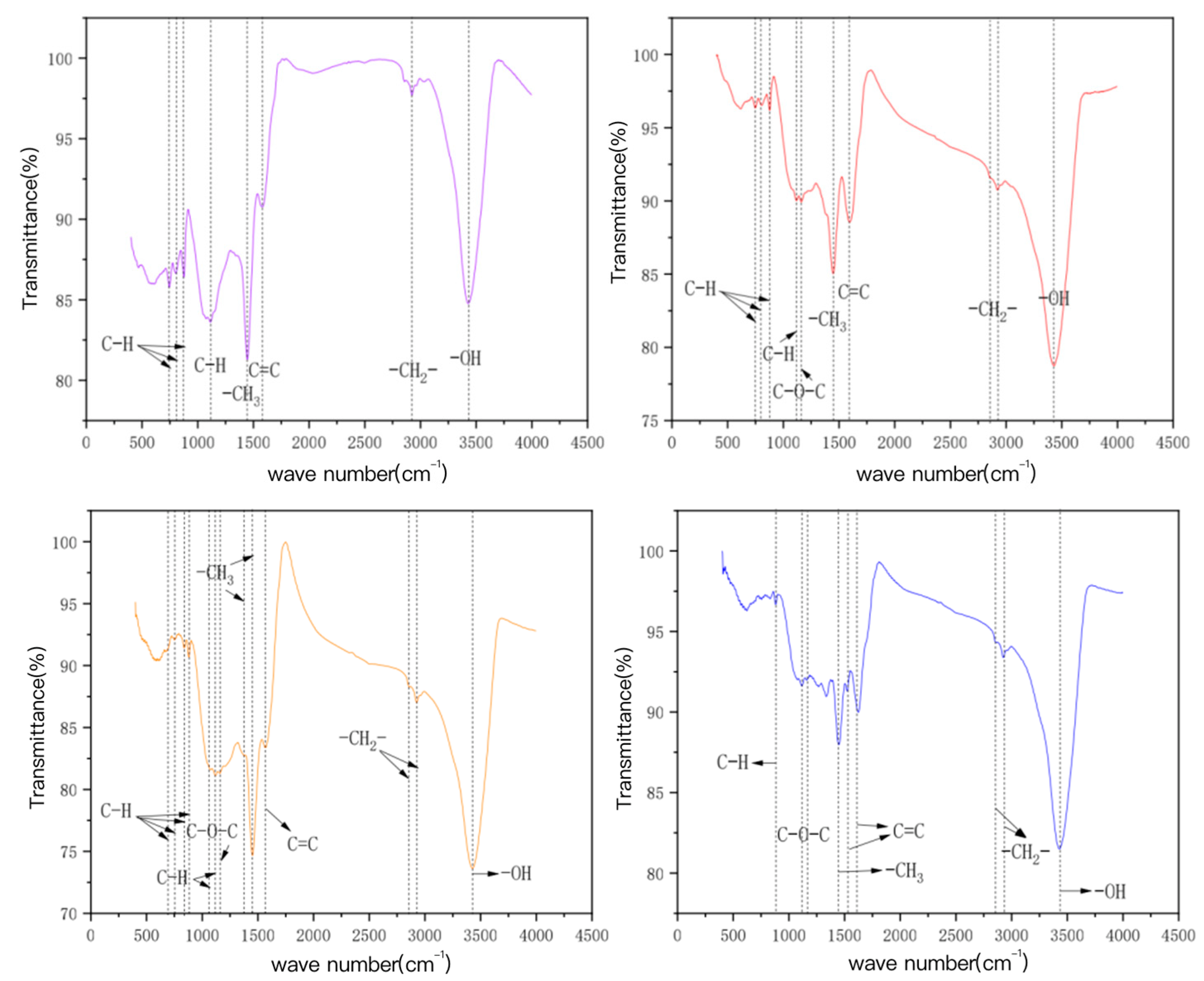
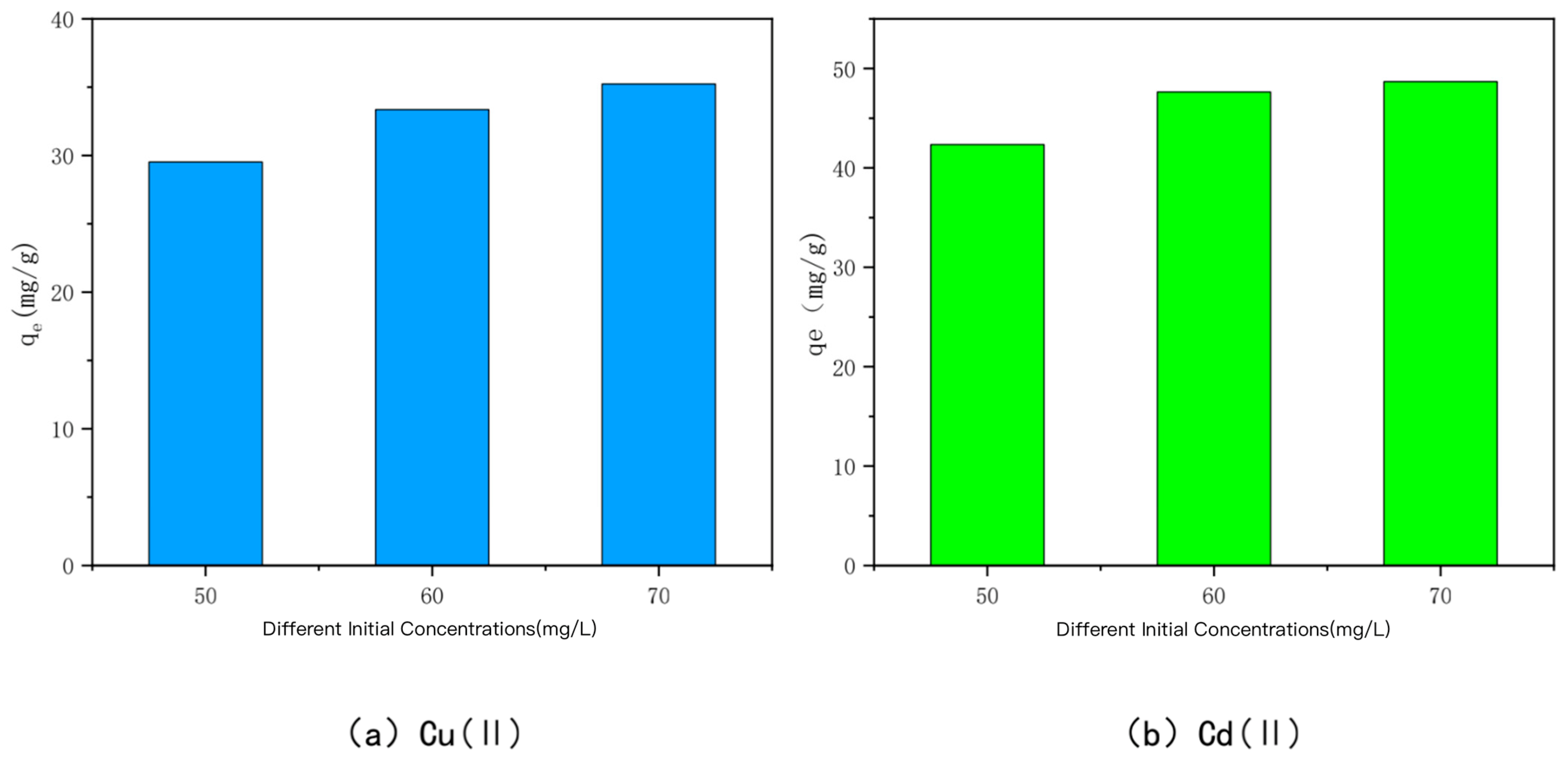
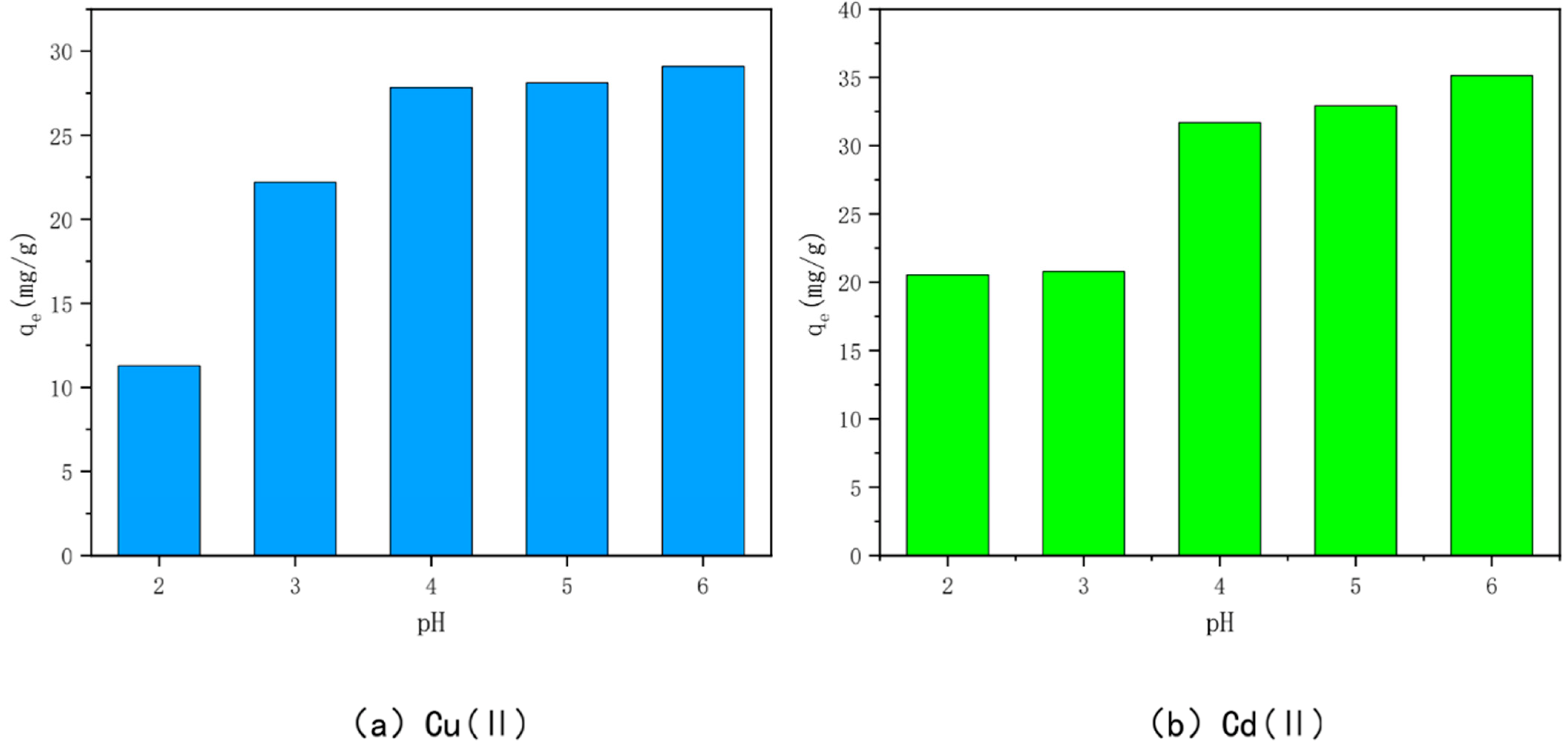
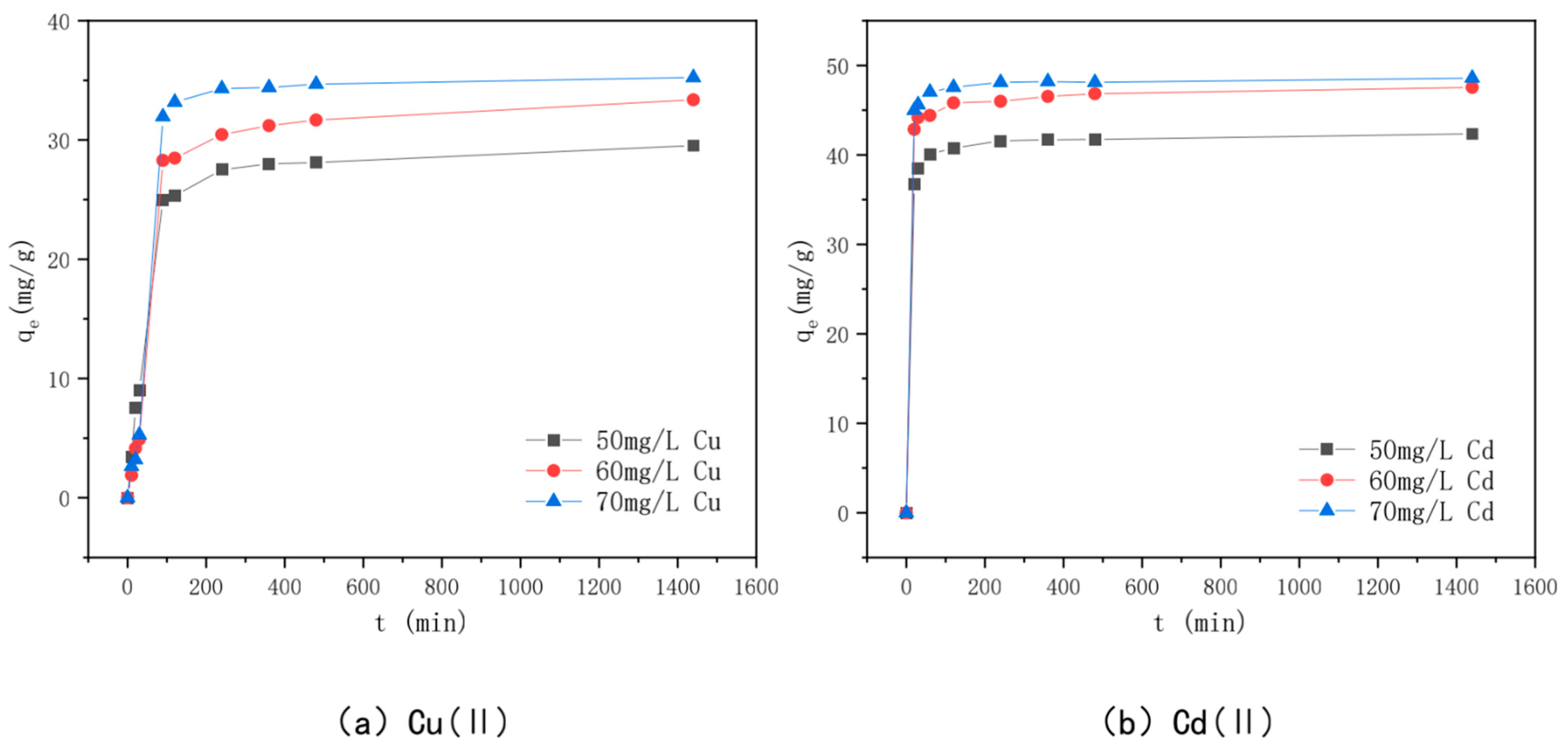
| Mechanism | Principle | Main Influencing Factors | References |
|---|---|---|---|
| Surfaceabsorption | The surface of biochar is rich in acidic groups such as carboxyl groups and phenolic hydroxyl groups, which can form specific metal complexes with heavy metal ions in water/soil and form active adsorption sites, etc. |
| [10,11,12,13,14] |
| ElectrostaticAdherence | Formation of ionic bonds (formed when atoms gain or lose electrons) between anions and cations by electrostatic interaction (chemical bonding). |
| [15,16,17] |
| Ion Exchange | The charged cations and protons on the surface of biochar exchange with dissolved heavy metal ions in an exchange reaction. |
| [18,19,20,21,22] |
| ChemicalPrecipitation | Anions react with heavy metal ions to form a water-insoluble precipitate. |
| [23,24,25,26] |
| Number | Experimental Instruments | Specifications | Use |
|---|---|---|---|
| 1 | Electronic balance | AUY220 | Weight |
| 2 | Tube furnace | SG-GL1400 | Carbonized sample |
| 3 | Constant temperature drying oven | DZ47-60 | Dry sample |
| 4 | pH meter | S2211T | Measuring pH of heavy metal solution |
| 5 | Atomic absorption spectrophotometer | TAS-990SUPERAFG | Measure the concentration of heavy metal solution |
| 6 | Constant temperature culture oscillator | THZ-82 | Oscillation |
| 7 | Pulverizer | LD-500A | Crushed sample |
| 8 | Vacuum pump | 2XZ-4 | Pump the tubular furnace into a vacuum state |
| 9 | Environmental scanning electron microscope—energy spectrometer | QUANTA200 | Obtain sample surface morphology and micro area elements |
| 10 | Specific surface area analyzer | ASAP200 | Aperture structure of analytical sample |
| 11 | Fourier transform infrared spectrometer | VERTEX80v | Analysis of sample surface |
| 12 | Constant-temperature water bath (HH-2) | HH-2 | Heated sample |
| Number | Chemical Reaction | Vender | Fineness | Use |
|---|---|---|---|---|
| 1 | CuSO4·5H2O | Nanjing Chemical Reagents Co., Ltd. | AR | Compound Cu(II) solution |
| 2 | CdSO4·8/3H2O | MacClean | AR | Compound Cd(II) solution |
| 3 | NaOH | Xilong Science Co., Ltd. | AR | Adjust the solution pH |
| 4 | HCl | Nanjing Chemical Reagents Co., Ltd. | AR | Adjust the solution pH |
| 5 | HNO3 | Nanjing Chemical Reagents Co., Ltd. | AR | Acid modification of bamboo charcoal |
| 6 | KOH | Sinopharm Group Chemical Reagent Co., Ltd. | AR | Bamboo charcoal was modified to alkali |
| 7 | SiO3 | Sinopharm Group Chemical Reagent Co., Ltd. | AR | Fixed charcoal bed |
| 8 | HCl | Nanjing Chemical Reagents Co., Ltd. | AR | Nitric acid-hydrochloric acid mixture is configured |
| 9 | CH3COOH | Nanjing Chemical Reagents Co., Ltd. | AR | Extracted acid extracts from the heavy metals Cu(II) and Cd(II) |
| 10 | NH2OH·HCI | Sinopharm Group Chemical Reagent Co., Ltd. | AR | Extracting acid-recoverable heavy metals Cu(II) and Cd(II) |
| 11 | H2O2 | Nanjing Chemical Reagents Co., Ltd. | AR | Extracting acid oxidation state heavy metals Cu(II) and Cd(II) |
| 12 | NH4COOH | Sinopharm Group Chemical Reagent Co., Ltd. | AR | A TCLP toxic leaching extract was prepared |
| Sample Name | BET Specific Area (m2/g) | BJH Total Hole Capacity (cm3/g) | Mean Pore Size (nm) |
|---|---|---|---|
| BC500 | 4.2422 | 0.000693 | 20.604 |
| KBC500-2 | 0.1112 | — | — |
| KAM500-400-3 | 6.1656 | 0.003165 | 106.317 |
| HBC500-30 | 2.8851 | 0.000442 | 20.056 |
| (a) BC500 | ||||||
|---|---|---|---|---|---|---|
| The BC500 Was Not Adsorbed | BC500 Adsorption of Cu(II) | BC500 Adsorption of Cd(II) | ||||
| Element | Wt % | At % | Wt % | At % | Wt % | At % |
| C K | 74.21 | 79.31 | 75.3 | 80.38 | 80.41 | 85.28 |
| O K | 25.79 | 20.69 | 24.42 | 19.57 | 18.23 | 14.57 |
| Cu K | — | — | 0.27 | 0.05 | — | — |
| Cd L | — | — | — | — | 1.36 | 0.15 |
| Totals | 100 | 100 | 100 | 100 | 100 | 100 |
| (b) KBC500-2 | ||||||
| The KBC500-2 Was Not Adsorbed | KBC500-2 Adsorption of Cu(II) | KBC500-2 Adsorption of Cd(II) | ||||
| Element | Wt % | At % | Wt % | At % | Wt % | At % |
| C K | 69.29 | 75.05 | 73.08 | 79.11 | 70.03 | 76.54 |
| O K | 30.71 | 24.94 | 25.3 | 20.56 | 28.36 | 23.27 |
| Cu K | — | — | 1.62 | 0.33 | — | — |
| Cd L | — | — | — | — | 1.61 | 0.19 |
| Totals | 100 | 100 | 100 | 100 | 100 | 100 |
| (c) KAM500-400-3 | ||||||
| KAM500-400-3 Was Not Adsorbed | KAM500-400-3 Adsorption of Cu(II) | KAM500-400-3 Adsorption of Cd(II) | ||||
| Element | Wt % | At % | Wt % | At % | Wt % | At % |
| C K | 57.65 | 64.74 | 56.2 | 66.34 | 51.53 | 62.43 |
| O K | 42.35 | 35.26 | 36.02 | 31.92 | 40.05 | 36.48 |
| Cu K | — | — | 7.77 | 1.73 | — | — |
| Cd L | — | — | — | — | 8.42 | 1.09 |
| Totals | 100 | 100 | 100 | 100 | 100 | 100 |
| (d) HBC500-30 | ||||||
| KAM500-30 Was Not Adsorbed | KAM500-30 Adsorption of Cu(II) | KAM500-30 Adsorption of Cd(II) | ||||
| Element | Wt % | At % | Wt % | At % | Wt % | At % |
| C K | 54.72 | 61.59 | 55.05 | 62.15 | 55.32 | 63.02 |
| O K | 45.28 | 38.41 | 44.54 | 37.76 | 42.98 | 36.77 |
| Cu K | — | — | 0.41 | 0.09 | — | — |
| Cd L | — | — | — | — | 1.7 | 0.21 |
| Totals | 100 | 100 | 100 | 100 | 100 | 100 |
| Heavy Metal | Dynamic Model | Constant | 50 mg/L | 60 mg/L | 70 mg/L |
|---|---|---|---|---|---|
| Cu(II) | Level 1 power | k1 (g/mg·h) | 0.0067 | 0.0065 | 0.0092 |
| Learn the model | qe (mg/g) | 18.4 | 22.67 | 21.185 | |
| R2 | 0.8119 | 0.8006 | 0.7932 | ||
| Planned secondary power | k2 (g/mg·h) | 0.0008 | 0.0003 | 0.0003 | |
| Learn the model | qe (mg/g) | 30.49 | 35.97 | 38.02 | |
| h0 | 0.7609 | 0.4198 | 0.4751 | ||
| R2 | 0.9968 | 0.976 | 0.9695 | ||
| Intraparticle diffusion | kp1 (mg/g min1/2) | 3.0492 | 3.9562 | 4.6053 | |
| model | Ci | −6.422 | −13.015 | −15.66 | |
| R12 | 0.9757 | 0.9489 | 0.9468 | ||
| kp2(mg/g min1/2) | 0.0876 | 0.1229 | 0.0421 | ||
| C2 | 26.217 | 28.711 | 33.663 | ||
| R2 | 0.9918 | 0.9762 | 0.975 | ||
| Cd(II) | Level 1 power | k1 (g/mg·h) | 0.0045 | 0.0037 | 0.0045 |
| Learn the model | qe (mg/g) | 3.68 | 3.83 | 2.47 | |
| R22 | 0.8248 | 0.925 | 0.7792 | ||
| Planned secondary power | k2 | 0.005 | 0.0039 | 0.0077 | |
| Learn the model | qe (mg/g) | 42.37 | 47.62 | 48.54 | |
| h0 | 9.0353 | 8.9526 | 18.215 |
| Heavy Metal | Temperature (°C) | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|---|
| qe | Ki | R2 | Kf | n | R2 | ||
| (mg/g) | (L/mg) | (mg1−1/n·L1/n·g−1) | |||||
| Cu(II) | 5 | 33.44 | 0.3646 | 0.9965 | 19.4724 | 7.4906 | 0.9748 |
| 25 | 32.68 | 3.06 | 0.9996 | 28.4088 | 23.9234 | 0.9526 | |
| 45 | 40.65 | 1.1942 | 0.9981 | 29.8505 | 11.4286 | 0.9876 | |
| Cd(II) | 5 | 46.95 | 0.6741 | 0.9964 | 29.2477 | 7.3584 | 0.9593 |
| 25 | 50.25 | 1 | 0.9988 | 32.7204 | 7.3153 | 0.9995 | |
| 45 | 51.55 | 1.6724 | 0.9955 | 38.7294 | 11.5207 | 0.9547 | |
| Heavy Metal | Temperature (K) | K d | ΔG0 (kJ/mol) | ΔH0 (kJ/mol) | ΔS0 (kJ/mol) | R2 (kJ/mol) |
|---|---|---|---|---|---|---|
| Cu(II) | 278 | 33.44 | −9.7 | 7.19 | 60.64 | 0.9582 |
| 298 | 32.68 | −10.74 | ||||
| 318 | 40.65 | −12.14 | ||||
| Cd(II) | 278 | 46.95 | −12.7 | 7.82 | 73.93 | 0.9884 |
| 298 | 50.25 | −14.27 | ||||
| 318 | 51.55 | −15.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Li, H.; Lin, S. Adsorption Characteristics of Modified Bamboo Charcoal on Cu(II) and Cd(II) in Water. Toxics 2022, 10, 787. https://doi.org/10.3390/toxics10120787
Wang Y, Li H, Lin S. Adsorption Characteristics of Modified Bamboo Charcoal on Cu(II) and Cd(II) in Water. Toxics. 2022; 10(12):787. https://doi.org/10.3390/toxics10120787
Chicago/Turabian StyleWang, Yizhuo, He Li, and Shaohua Lin. 2022. "Adsorption Characteristics of Modified Bamboo Charcoal on Cu(II) and Cd(II) in Water" Toxics 10, no. 12: 787. https://doi.org/10.3390/toxics10120787
APA StyleWang, Y., Li, H., & Lin, S. (2022). Adsorption Characteristics of Modified Bamboo Charcoal on Cu(II) and Cd(II) in Water. Toxics, 10(12), 787. https://doi.org/10.3390/toxics10120787






