Chemical Characteristics and Source-Specific Health Risks of the Volatile Organic Compounds in Urban Nanjing, China
Abstract
1. Introduction
2. Experimental Methods
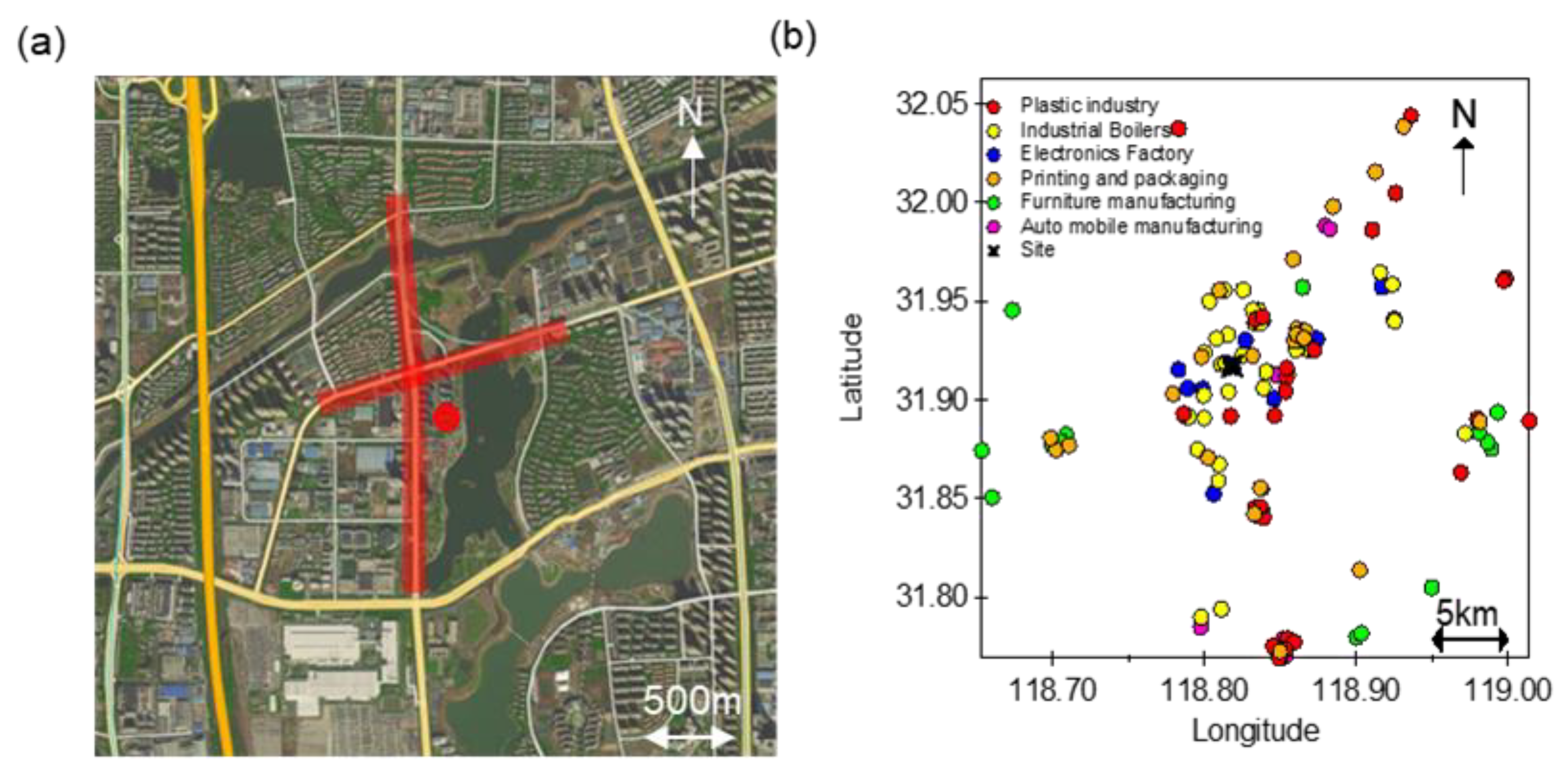
2.1. Sampling Site, Instrumentation, and Chemical Analysis
2.2. Data Analysis
2.2.1. Source Apportionment
2.2.2. Calculation of Ozone Formation Potential (OFP) and Secondary Organic Aerosol Formation Potential (SOAFP)
2.2.3. Health Risk Assessment
3. Results and Discussion
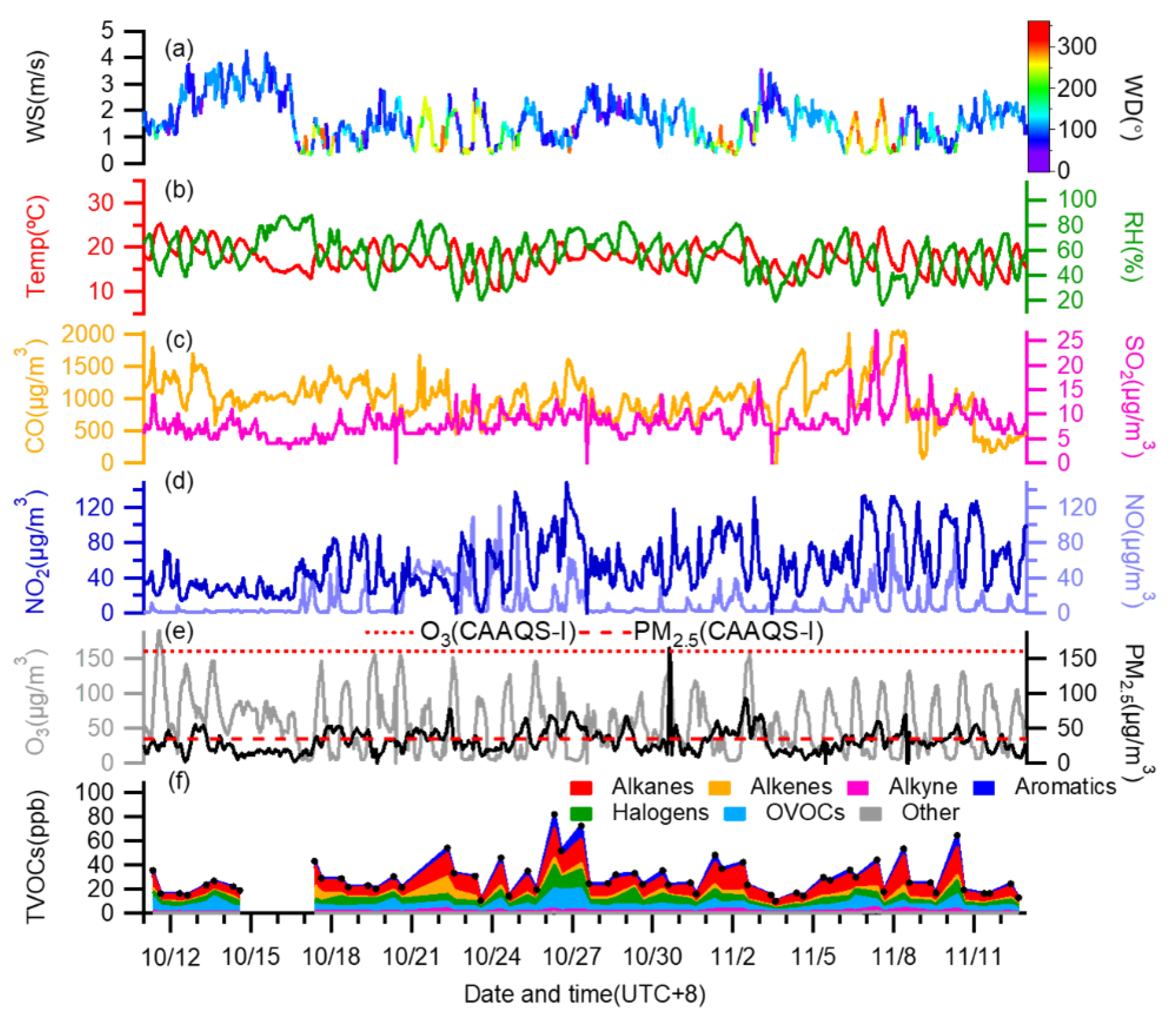
3.1. Overview of Air Pollutants and Meteorological Conditions
3.2. Chemical Characteristics of VOCs
3.2.1. Mass Concentration and Composition
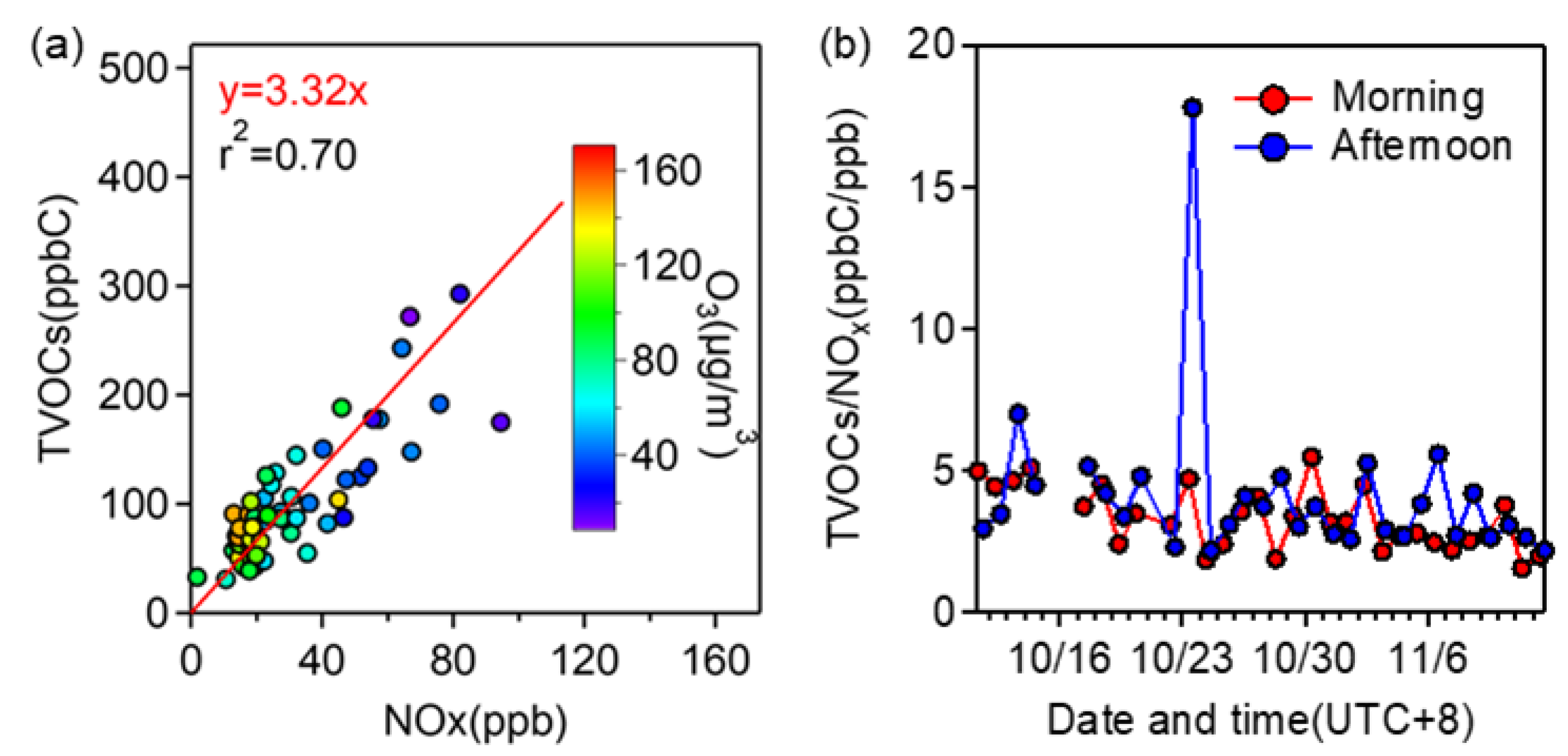
3.2.2. Relationship with NOx
3.3. Contributions of VOCs to Ozone and SOA Formations
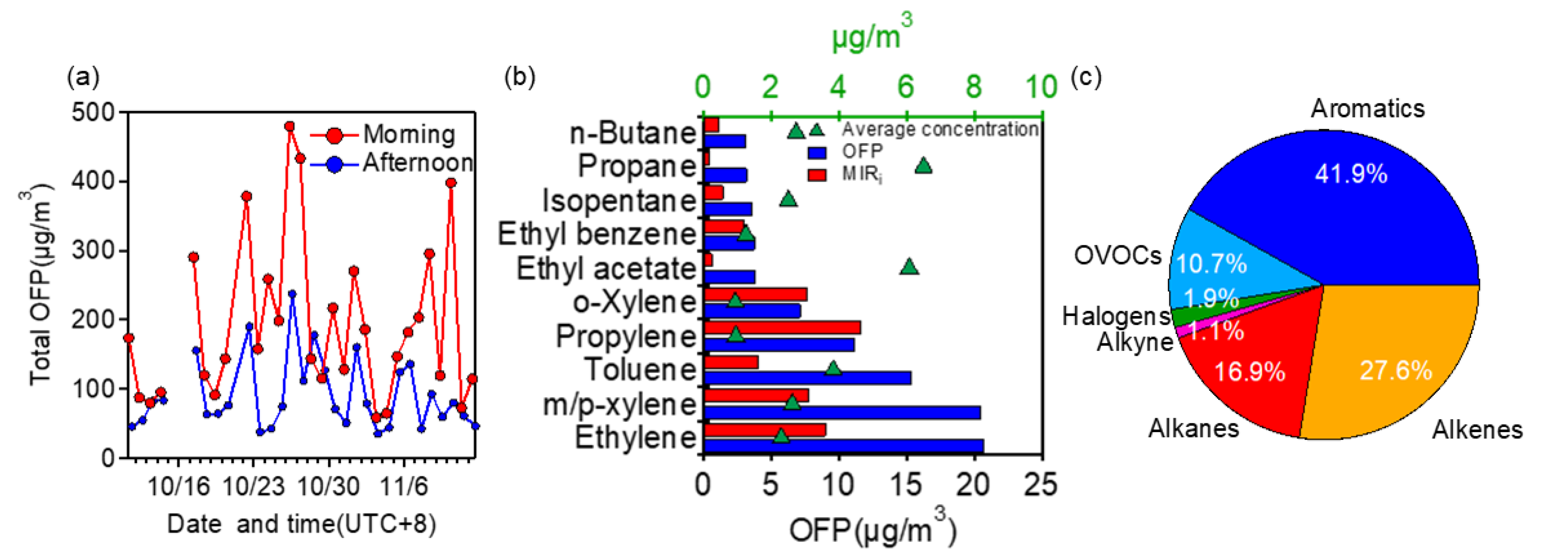
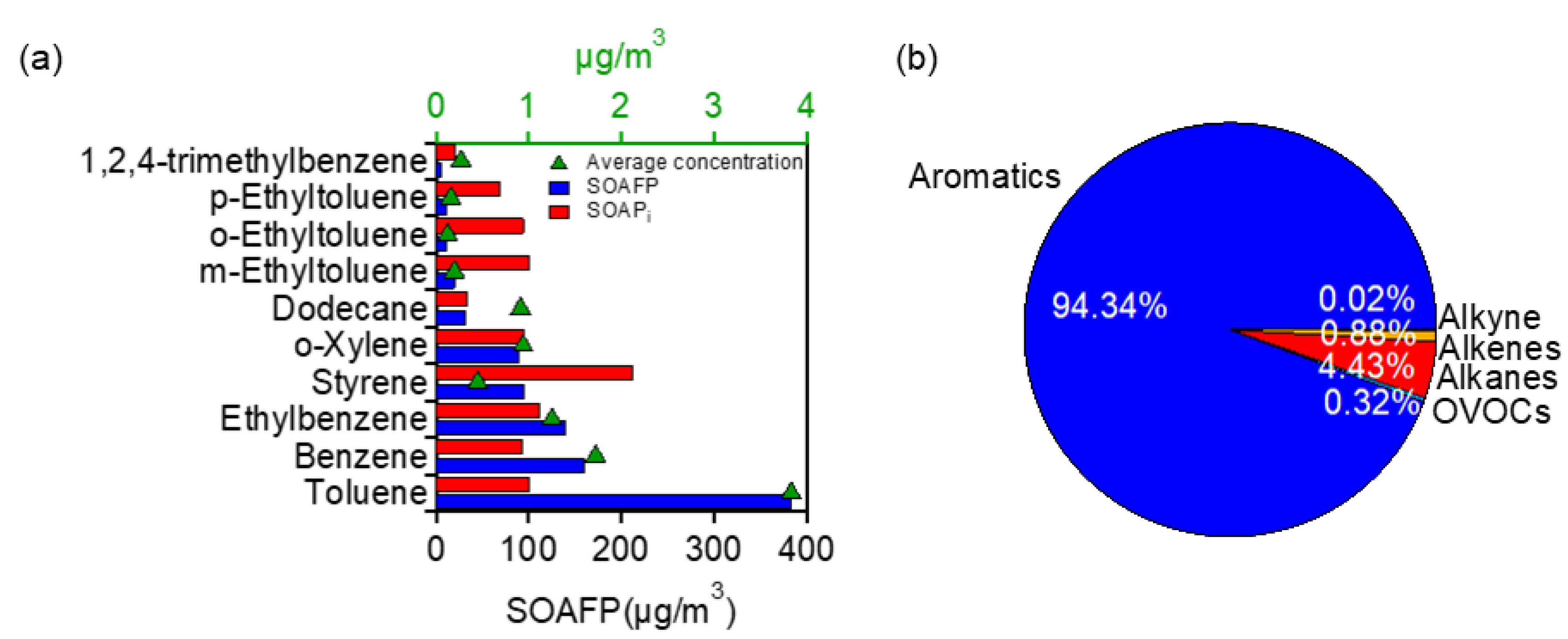
3.3.1. Contributions to OFP
3.3.2. Contributions to SOAFP
3.4. Sources of VOCs
3.4.1. Diagnostic Ratios
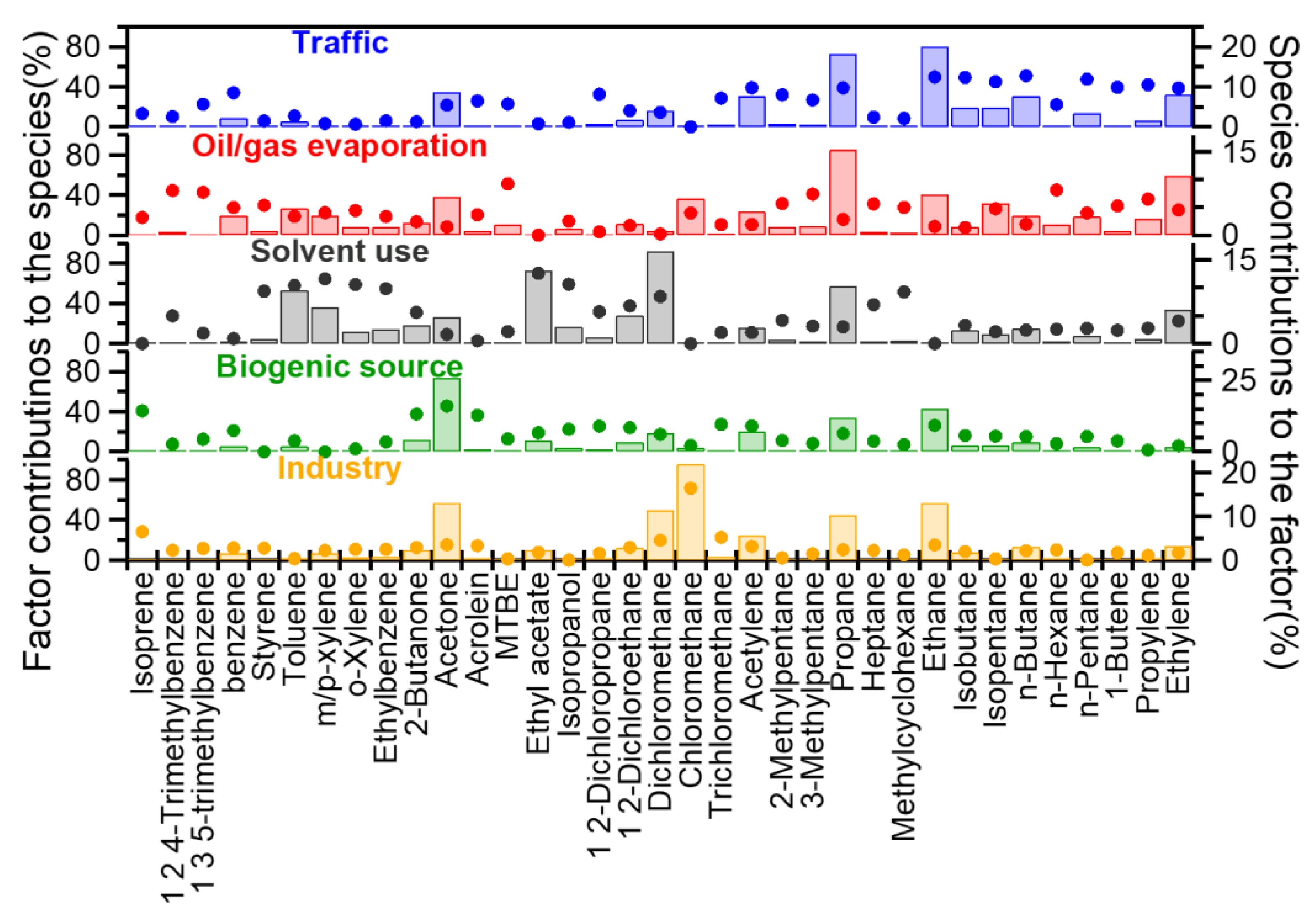
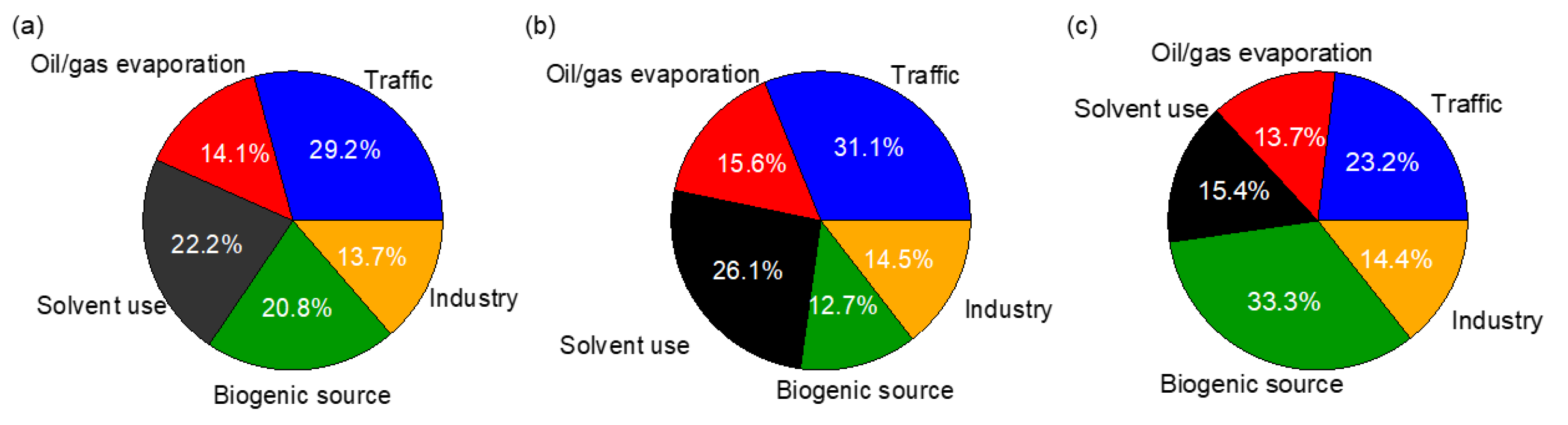
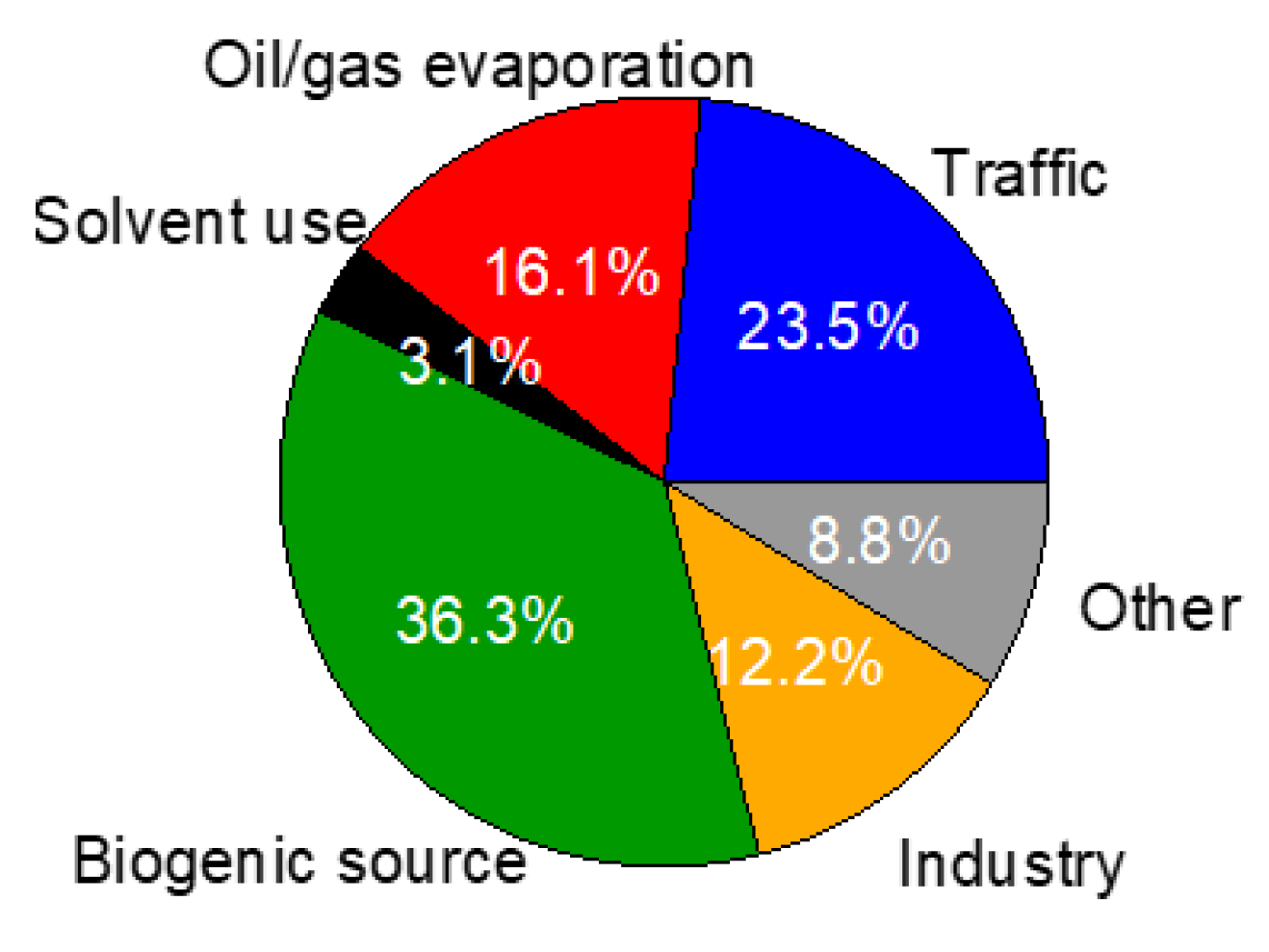
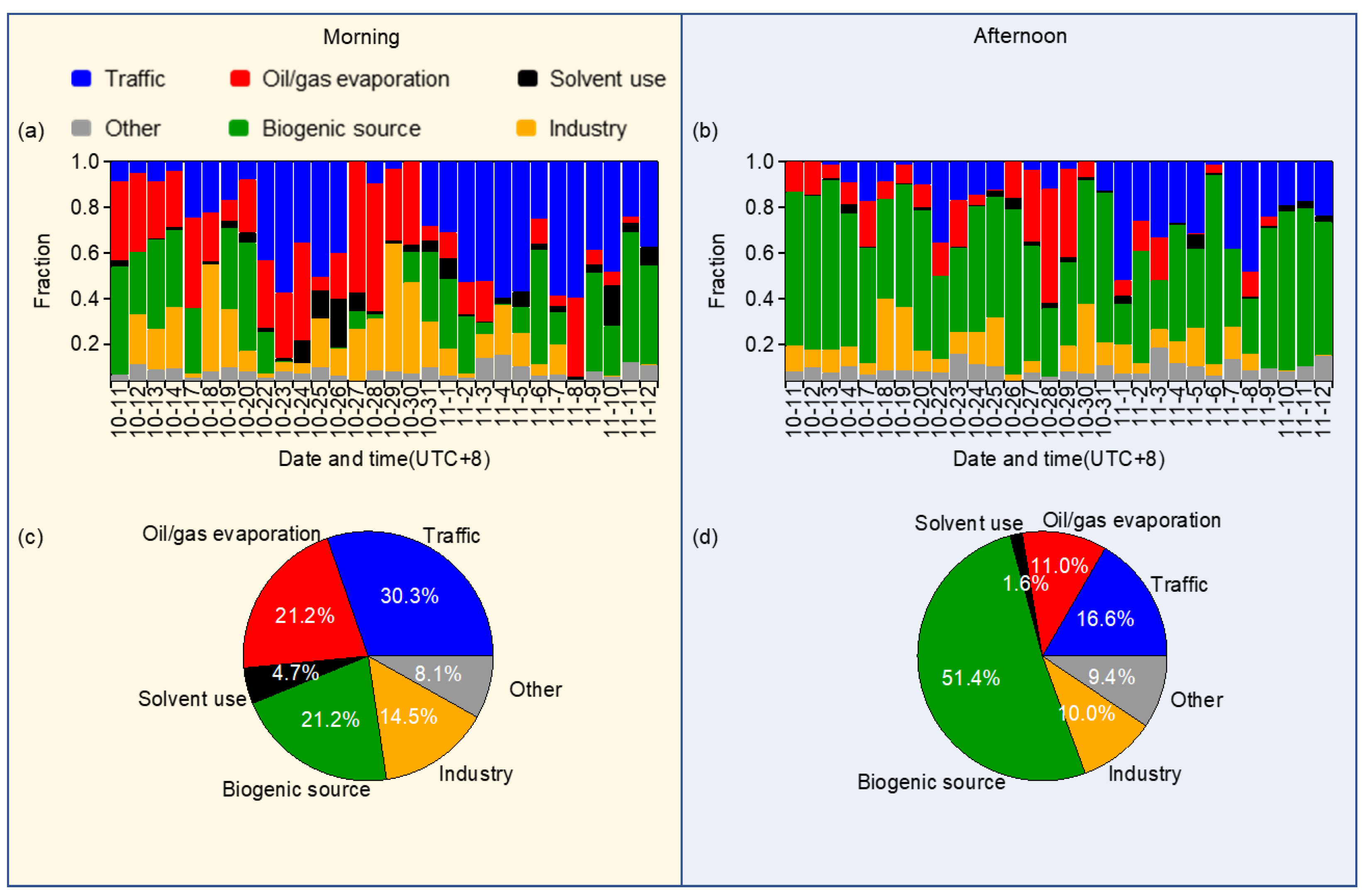
3.4.2. Source Apportionment
3.5. Health Risks of VOCs
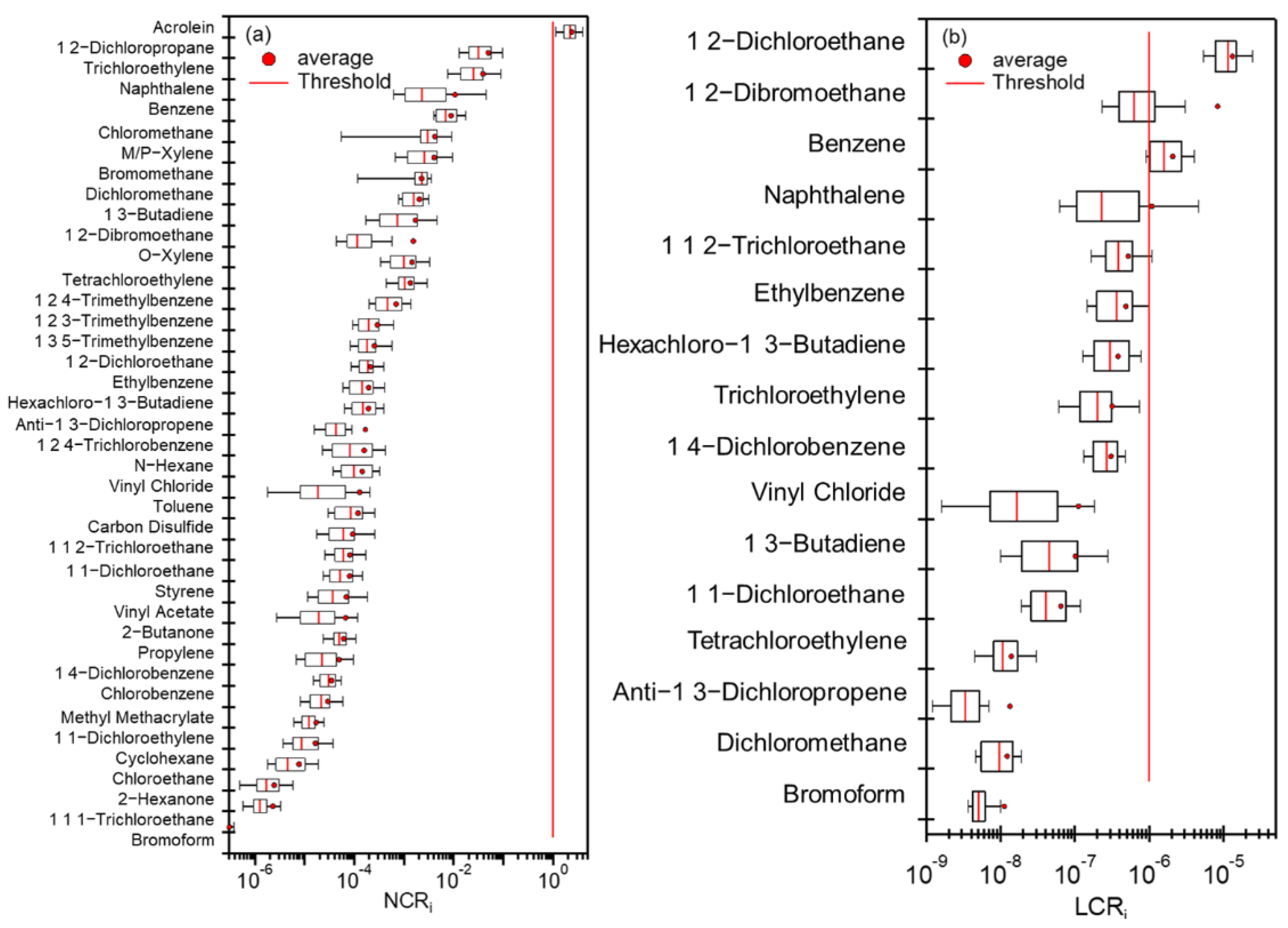
3.5.1. Calculations of LCR and NCR
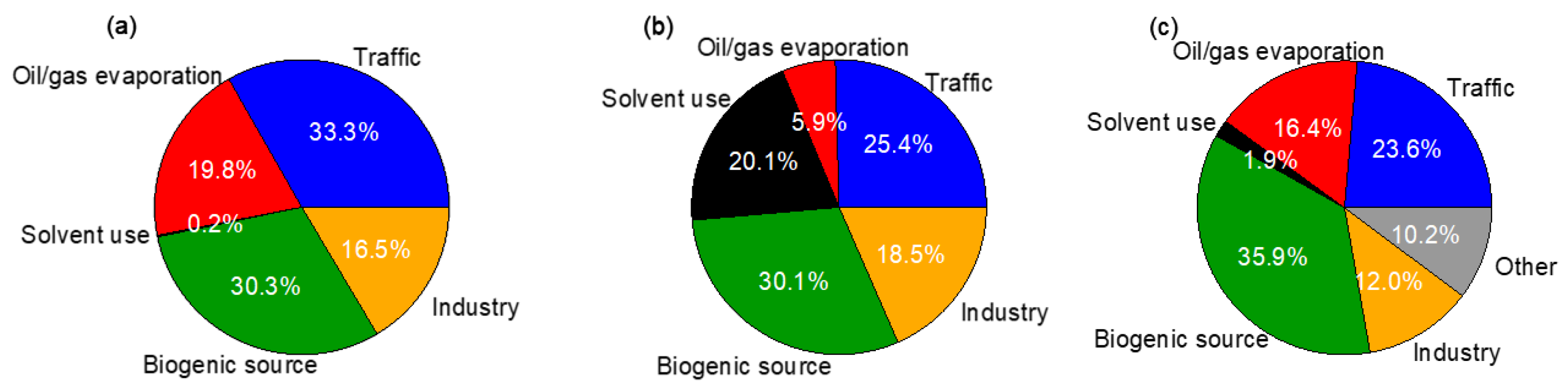
3.5.2. Source-Specific Health Risks
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Haagen-Smit, A.J.; Fox, M.M. Photochemical Ozone Formation with Hydrocarbons and Automobile Exhaust. Air Repair 1954, 4, 105–136. [Google Scholar] [CrossRef]
- Wu, R.; Li, J.; Hao, Y.; Li, Y.; Zeng, L.; Xie, S. Evolution process and sources of ambient volatile organic compounds during a severe haze event in Beijing, China. Sci. Total Environ. 2016, 560, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Shang, X.; Chen, H.; Xie, G.; Fu, Y.; Wu, D.; Sun, W.; Liu, P.; Zhang, C.; Mu, Y.; et al. Significant impact of coal combustion on VOCs emissions in winter in a North China rural site. Sci. Total Environ. 2020, 720, 137617. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, R.; Fu, H.; Zhou, D.; Chen, J. Observation and analysis of atmospheric volatile organic compounds in a typical petrochemical area in Yangtze River Delta, China. J. Environ. Sci. 2018, 71, 233–248. [Google Scholar] [CrossRef]
- Luo, H.; Li, G.; Chen, J.; Lin, Q.; Ma, S.; Wang, Y.; An, T. Spatial and temporal distribution characteristics and ozone formation potentials of volatile organic compounds from three typical functional areas in China. Environ. Res. 2020, 183, 109141. [Google Scholar] [CrossRef]
- Wang, W.; Xiong, T.; Zhang, W.; Luo, B.; Wang, D.; Jiang, X.; Rao, Z.; Jiang, Y.; Liu, Y.; Cheng, H.; et al. Observation and analysis of VOCs in nine prefecture-level cities of Sichuan Province, China. Environ. Monit. Assess. 2020, 192, 511. [Google Scholar] [CrossRef]
- Atkinson, R. Atmospheric chemistry of VOCs and NOx. Atmos. Environ. 2000, 34, 2063–2101. [Google Scholar] [CrossRef]
- Zhang, J.J.; Wei, Y.; Fang, Z. Ozone Pollution: A Major Health Hazard Worldwide. Front. Immunol. 2019, 10, 2518. [Google Scholar] [CrossRef]
- Kašpar, V.; Zapletal, M.; Samec, P.; Komárek, J.; Bílek, J.; Juráň, S. Unmanned aerial systems for modelling air pollution removal by urban greenery. Urban For. Urban Green. 2022, 78, 127757. [Google Scholar] [CrossRef]
- Ismail, I.M.; Basahi, J.M.; Hassan, I.A. Gas exchange and chlorophyll fluorescence of pea (Pisum sativum L.) plants in response to ambient ozone at a rural site in Egypt. Sci. Total Environ. 2014, 497–498, 585–593. [Google Scholar] [CrossRef]
- Nuvolone, D.; Petri, D.; Voller, F. The effects of ozone on human health. Environ. Sci. Pollut. Res. Int. 2018, 25, 8074–8088. [Google Scholar] [CrossRef]
- Koman, P.D.; Mancuso, P. Ozone Exposure, Cardiopulmonary Health, and Obesity: A Substantive Review. Chem. Res. Toxicol. 2017, 30, 1384–1395. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, H.; Jing, S.; Gao, Y.; Peng, Y.; Lou, S.; Cheng, T.; Tao, S.; Li, L.; Li, Y.; et al. Characteristics and sources of volatile organic compounds (VOCs) in Shanghai during summer: Implications of regional transport. Atmos. Environ. 2019, 215, 116902. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, X.; Li, H.; Cheng, N.; Zhang, Y.; Zhang, K.; Li, L. Variations in Levels and Sources of Atmospheric VOCs during the Continuous Haze and Non-Haze Episodes in the Urban Area of Beijing: A Case Study in Spring of 2019. Atmosphere 2021, 12, 171. [Google Scholar] [CrossRef]
- Meng, Y.; Song, J.; Zeng, L.; Zhang, Y.; Zhao, Y.; Liu, X.; Guo, H.; Zhong, L.; Ou, Y.; Zhou, Y.; et al. Ambient volatile organic compounds at a receptor site in the Pearl River Delta region: Variations, source apportionment and effects on ozone formation. J. Environ. Sci. 2022, 111, 104–117. [Google Scholar] [CrossRef]
- Xiong, C.; Wang, N.; Zhou, L.; Yang, F.; Qiu, Y.; Chen, J.; Han, L.; Li, J. Component characteristics and source apportionment of volatile organic compounds during summer and winter in downtown Chengdu, southwest China. Atmos. Environ. 2021, 258, 118485. [Google Scholar] [CrossRef]
- Wang, S.; Liu, G.; Zhang, H.; Yi, M.; Liu, Y.; Hong, X.; Bao, X. Insight into the environmental monitoring and source apportionment of volatile organic compounds (VOCs) in various functional areas. Air Qual. Atmos. Health 2021, 15, 1121–1131. [Google Scholar] [CrossRef]
- Jing, S.A.; Gao, Y.Q.; Shen, J.D.; Wang, Q.; Peng, Y.R.; Li, Y.J.; Wang, H.L. Characteristics and Reactivity of Ambient VOCs in Urban Hangzhou, China. Huan Jing Ke Xue 2020, 41, 5306–5315. [Google Scholar]
- Mozaffar, A.; Zhang, Y.-L.; Fan, M.; Cao, F.; Lin, Y.-C. Characteristics of summertime ambient VOCs and their contributions to O3 and SOA formation in a suburban area of Nanjing, China. Atmos. Res. 2020, 240, 104923. [Google Scholar] [CrossRef]
- Li, B.; Ho, S.S.H.; Li, X.; Guo, L.; Feng, R.; Fang, X. Pioneering observation of atmospheric volatile organic compounds in Hangzhou in eastern China and implications for upcoming 2022 Asian Games. J. Environ. Sci. 2023, 124, 723–734. [Google Scholar] [CrossRef]
- Shusterman, D. The effects of air pollutants and irritants on the upper airway. Proc. Am. Thorac. Soc. 2011, 8, 101–105. [Google Scholar] [CrossRef]
- Doty, R.L.; Cometto-Muniz, J.E.; Jalowayski, A.A.; Dalton, P.; Kendal-Reed, M.; Hodgson, M. Assessment of upper respiratory tract and ocular irritative effects of volatile chemicals in humans. Crit. Rev. Toxicol. 2004, 34, 85–142. [Google Scholar] [CrossRef]
- Wu, D.; Ding, X.; Li, Q.; Sun, J.F.; Huang, C.; Yao, L.; Wang, X.M.; Ye, X.N.; Chen, Y.J.; He, H.; et al. Pollutants emitted from typical Chinese vessels: Potential contributions to ozone and secondary organic aerosols. J. Clean. Prod. 2019, 238, 117862. [Google Scholar] [CrossRef]
- Tomatis, M.; Moreira, M.T.; Xu, H.H.; Deng, W.; He, J.; Parvez, A.M. Removal of VOCs from waste gases using various thermal oxidizers: A comparative study based on life cycle assessment and cost analysis in China. J. Clean. Prod. 2019, 233, 808–818. [Google Scholar] [CrossRef]
- El-Maghrabey, M.H.; El-Shaheny, R.; El Hamd, M.A.; Al-Khateeb, L.A.; Kishikawa, N.; Kuroda, N. Aldehydes’ Sources, Toxicity, Environmental Analysis, and Control in Food. In Organic Pollutants: Toxicity and Solutions; Vasanthy, M., Sivasankar, V., Sunitha, T.G., Eds.; Springer International Publishing: Cham, Switzerland, 2022; pp. 117–151. [Google Scholar]
- Cassani, C.; Martin-Rapun, R.; Arceo, E.; Bravo, F.; Melchiorre, P. Synthesis of 9-amino(9-deoxy)epi cinchona alkaloids, general chiral organocatalysts for the stereoselective functionalization of carbonyl compounds. Nat. Protoc 2013, 8, 325–344. [Google Scholar] [CrossRef]
- Nabizadeh, R.; Sorooshian, A.; Baghani, A.N.; Ashournejad, Q. On the nature of airborne aldehydes in a middle eastern megacity: Tehran, Iran. Sustain. Cities Soc. 2020, 53, 101895. [Google Scholar] [CrossRef]
- Adhikari, N.; Joshi, D.R. An Overview on Common Organic Solvents and Their Toxicity. J. Pharm. Res. Int. 2019, 28, 1–18. [Google Scholar]
- Todd, G.D.; Melia, J. Toxicological Profile for 2-Butanone: Draft for Public Comment; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2019. [Google Scholar]
- Lee, D.; Wexler, A.S. Atmospheric amines—Part III: Photochemistry and toxicity. Atmos. Environ. 2013, 71, 95–103. [Google Scholar] [CrossRef]
- Ge, X.; Wexler, A.S.; Clegg, S.L. Atmospheric amines—Part I. A review. Atmos. Environ. 2011, 45, 524–546. [Google Scholar] [CrossRef]
- Jia, H.; Gao, S.; Duan, Y.; Fu, Q.; Che, X.; Xu, H.; Wang, Z.; Cheng, J. Investigation of health risk assessment and odor pollution of volatile organic compounds from industrial activities in the Yangtze River Delta region, China. Ecotoxicol. Environ. Saf. 2021, 208, 111474. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.Y.; Ko, H.J.; Kim, H.T.; Kim, Y.S.; Roh, Y.M.; Lee, C.M.; Kim, H.S.; Kim, C.N. Sulfuric odorous compounds emitted from pig-feeding operations. Atmos. Environ. 2007, 41, 4811–4818. [Google Scholar] [CrossRef]
- Huang, H.; Miller, G.Y.; Ellis, M.; Funk, T.; Zhang, Y.; Hollis, G.; Heber, A.J. Odor management in swine finishing operations: Cost effectiveness. J. Food Agric. Environ. 2004, 22, 130–135. [Google Scholar]
- Conti, C.; Guarino, M.; Bacenetti, J. Measurements techniques and models to assess odor annoyance: A review. Environ. Int. 2020, 134, 105261. [Google Scholar] [CrossRef] [PubMed]
- Sahu, L.K.; Pal, D.; Yadav, R.; Munkhtur, J. Aromatic VOCs at Major Road Junctions of a Metropolis in India: Measurements Using TD-GC-FID and PTR-TOF-MS Instruments. Aerosol. Air Qual. Res. 2016, 16, 2405–2420. [Google Scholar] [CrossRef]
- Dumanoglu, Y.; Kara, M.; Altiok, H.; Odabasi, M.; Elbir, T.; Bayram, A. Spatial and seasonal variation and source apportionment of volatile organic compounds (VOCs) in a heavily industrialized region. Atmos. Environ. 2014, 98, 168–178. [Google Scholar] [CrossRef]
- Huang, L.; Qian, H.; Deng, S.; Guo, J.; Li, Y.; Zhao, W.; Yue, Y. Urban residential indoor volatile organic compounds in summer, Beijing: Profile, concentration and source characterization. Atmos. Environ. 2018, 188, 1–11. [Google Scholar] [CrossRef]
- Huang, X.; Zhang, Y.; Yang, W.; Huang, Z.; Wang, Y.; Zhang, Z.; He, Q.; Lü, S.; Huang, Z.; Bi, X.; et al. Effect of traffic restriction on reducing ambient volatile organic compounds (VOCs): Observation-based evaluation during a traffic restriction drill in Guangzhou, China. Atmos. Environ. 2017, 161, 61–70. [Google Scholar] [CrossRef]
- Sarkar, C.; Sinha, V.; Sinha, B.; Panday, A.K.; Rupakheti, M.; Lawrence, M.G. Source apportionment of NMVOCs in the Kathmandu Valley during the SusKat-ABC international field campaign using positive matrix factorization. Atmos. Chem. Phys. 2017, 17, 8129–8156. [Google Scholar] [CrossRef]
- He, Z.; Wang, X.; Ling, Z.; Zhao, J.; Guo, H.; Shao, M.; Wang, Z. Contributions of different anthropogenic volatile organic compound sources to ozone formation at a receptor site in the Pearl River Delta region and its policy implications. Atmos. Chem. Phys. 2019, 19, 8801–8816. [Google Scholar] [CrossRef]
- Mozaffar, A.; Zhang, Y.-L.; Lin, Y.-C.; Xie, F.; Fan, M.-Y.; Cao, F. Measurement report: High contributions of halocarbon and aromatic compounds to atmospheric volatile organic compounds in an industrial area. Atmos. Chem. Phys. 2021, 21, 18087–18099. [Google Scholar] [CrossRef]
- Zhao, Q.; Bi, J.; Liu, Q.; Ling, Z.; Shen, G.; Chen, F.; Qiao, Y.; Li, C.; Ma, Z. Sources of volatile organic compounds and policy implications for regional ozone pollution control in an urban location of Nanjing, East China. Atmos. Chem. Phys. 2020, 20, 3905–3919. [Google Scholar] [CrossRef]
- Atkinson, R.; Arey, J. Atmospheric degradation of volatile organic compounds. Chem. Rev. 2003, 103, 4605–4638. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, C.; Sinha, V.; Kumar, V.; Rupakheti, M.; Panday, A.; Mahata, K.S.; Rupakheti, D.; Kathayat, B.; Lawrence, M.G. Overview of VOC emissions and chemistry from PTR-TOF-MS measurements during the SusKat-ABC campaign: High acetaldehyde, isoprene and isocyanic acid in wintertime air of the Kathmandu Valley. Atmos. Chem. Phys. 2016, 16, 3979–4003. [Google Scholar] [CrossRef]
- Kleinman, L. Ozone process insights from field experiments-part II: Observation-based analysis for ozone production. Atmos. Environ. 2000, 34, 2023–2033. [Google Scholar] [CrossRef]
- Cardelino, C.A.; Chameides, W.L. An observation-based model for analyzing ozone precursor relationships in the urban atmosphere. J. Air Waste Manag. Assoc. 1995, 45, 161–180. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Holloway, T.; Oki, T.; Streets, D.G.; Richter, A. Multi-−scale model analysis of boundary layer ozone over East Asia. Atmos. Chem. Phys. 2009, 9, 3277–3301. [Google Scholar] [CrossRef]
- Carter, W.P.L. Development of Ozone Reactivity Scales for Volatile Organic Compounds. J. Air Waste Manag. Assoc. 2012, 44, 881–899. [Google Scholar] [CrossRef]
- Carter, W.P.L. Development of the SAPRC-07 chemical mechanism. Atmos. Environ. 2010, 44, 5324–5335. [Google Scholar] [CrossRef]
- Chen, P.; Zhao, X.; Wang, O.; Shao, M.; Xiao, X.; Wang, S.; Wang, Q.g. Characteristics of VOCs and their Potentials for O3 and SOA Formation in a Medium-sized City in Eastern China. Aerosol. Air Qual. Res. 2022, 22, 210239. [Google Scholar] [CrossRef]
- Cui, L.; Wu, D.; Wang, S.; Xu, Q.; Hu, R.; Hao, J. Measurement report: Ambient volatile organic compound (VOC) pollution in urban Beijing: Characteristics, sources, and implications for pollution control. Atmos. Chem. Phys. 2022, 22, 11931–11944. [Google Scholar] [CrossRef]
- Zhang, X.; Xue, Z.; Li, H.; Yan, L.; Yang, Y.; Wang, Y.; Duan, J.; Li, L.; Chai, F.; Cheng, M.; et al. Ambient volatile organic compounds pollution in China. J. Environ. Sci. 2017, 55, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Hui, L.; Liu, X.; Tan, Q.; Feng, M.; An, J.; Qu, Y.; Zhang, Y.; Cheng, N. VOC characteristics, sources and contributions to SOA formation during haze events in Wuhan, Central China. Sci. Total Environ. 2019, 650, 2624–2639. [Google Scholar] [CrossRef] [PubMed]
- Derwent, R.G.; Jenkin, M.E.; Utembe, S.R.; Shallcross, D.E.; Murrells, T.P.; Passant, N.R. Secondary organic aerosol formation from a large number of reactive man-made organic compounds. Sci. Total Environ. 2010, 408, 3374–3381. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wu, F.; Hu, B.; Tang, G.; Zhang, J.; Wang, Y. VOC characteristics, emissions and contributions to SOA formation during hazy episodes. Atmos. Environ. 2016, 141, 560–570. [Google Scholar] [CrossRef]
- Yu, S.; Su, F.; Yin, S.; Wang, S.; Xu, R.; He, B.; Fan, X.; Yuan, M.; Zhang, R. Characterization of ambient volatile organic compounds, source apportionment, and the ozone–NOx–VOC sensitivities in a heavily polluted megacity of central China: Effect of sporting events and. Atmos. Chem. Phys. 2021, 21, 15239–15257. [Google Scholar] [CrossRef]
- Xie, G.; Chen, H.; Zhang, F.; Shang, X.; Zhan, B.; Zeng, L.; Mu, Y.; Mellouki, A.; Tang, X.; Chen, J. Compositions, sources, and potential health risks of volatile organic compounds in the heavily polluted rural North China Plain during the heating season. Sci. Total Environ. 2021, 789, 147956. [Google Scholar] [CrossRef]
- Zhang, H.; Ji, Y.; Wu, Z.; Peng, L.; Bao, J.; Peng, Z.; Li, H. Atmospheric volatile halogenated hydrocarbons in air pollution episodes in an urban area of Beijing: Characterization, health risk assessment and sources apportionment. Sci. Total Environ. 2021, 806, 150283. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, R.; Yu, W. The Effects of PM2.5 Concentrations and Relative Humidity on Atmospheric Visibility in Beijing. J. Geophys. Res. Atmos. 2019, 124, 2235–2259. [Google Scholar] [CrossRef]
- Miao, Y.; Li, J.; Miao, S.; Che, H.; Wang, Y.; Zhang, X.; Zhu, R.; Liu, S. Interaction Between Planetary Boundary Layer and PM2.5 Pollution in Megacities in China: A Review. Curr. Pollut. Rep. 2019, 5, 261–271. [Google Scholar] [CrossRef]
- Zhang, Y.; Mu, Y.; Liu, J.; Mellouki, A. Levels, sources and health risks of carbonyls and BTEX in the ambient air of Beijing, China. J. Environ. Sci. 2012, 24, 124–130. [Google Scholar] [CrossRef]
- Fan, M.Y.; Zhang, Y.L.; Lin, Y.C.; Li, L.; Xie, F.; Hu, J.; Mozaffar, A.; Cao, F. Source apportionments of atmospheric volatile organic compounds in Nanjing, China during high ozone pollution season. Chemosphere 2021, 263, 128025. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.Y.; Lin, Y.C.; Zhang, Y.L. Characteristics and Source Apportionment of Atmospheric VOCs in the Nanjing Industrial Area in Autumn. Huan Jing Ke Xue 2020, 41, 2565–2576. [Google Scholar] [PubMed]
- Wang, T.; Xue, L.; Brimblecombe, P.; Lam, Y.F.; Li, L.; Zhang, L. Ozone pollution in China: A review of concentrations, meteorological influences, chemical precursors, and effects. Sci. Total Environ. 2017, 575, 1582–1596. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Ho, S.S.H.; Gong, S.; Ni, J.; Li, H.; Han, L.; Yang, Y.; Qi, Y.; Zhao, D. Characterization of VOCs and their related atmospheric processes in a central Chinese city during severe ozone pollution periods. Atmos. Chem. Phys. 2019, 19, 617–638. [Google Scholar] [CrossRef]
- Wang, M.; Lu, S.H.; Shao, M.; Zeng, L.M.; Zheng, J.; Xie, F.J.; Lin, H.T.; Hu, K.; Lu, X.D. Impact of COVID-19 lockdown on ambient levels and sources of volatile organic compounds (VOCs) in Nanjing, China. Sci. Total Environ. 2021, 757, 143823. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yan, Y.; Duan, X.; Chai, J.; Li, R.; Xu, Y.; Li, Z.; Peng, L. Sources and Seasonal Variance of Ambient Volatile Organic Compounds in the Typical Industrial City of Changzhi, Northern China. Atmosphere 2022, 13, 393. [Google Scholar] [CrossRef]
- Sheng, T.; Chen, X.J.; Gao, S.; Liu, Q.Z.; Li, X.F.; Fu, Q.Y. Pollution Characteristics and Health Risk Assessment of VOCs in Areas Surrounding a Petrochemical Park in Shanghai. Huan Jing Ke Xue 2018, 39, 4901–4908. [Google Scholar]
- Han, C.; Liu, R.; Luo, H.; Li, G.; Ma, S.; Chen, J.; An, T. Pollution profiles of volatile organic compounds from different urban functional areas in Guangzhou China based on GC/MS and PTR-TOF-MS: Atmospheric environmental implications. Atmos. Environ. 2019, 214, 116843. [Google Scholar] [CrossRef]
- Ying, H.; Yuan, H.; Ye, Z.; Li, S.; Liang, J. Spatial and temporal distribution characteristics of volatile organic compounds in the atmosphere and ozone generation potential in Chengdu. Acta Sci. Circumstantiae 2015, 35, 386–393. [Google Scholar]
- Qin, J.; Wang, X.; Yang, Y.; Qin, Y.; Shi, S.; Xu, P.; Chen, R.; Zhou, X.; Tan, J.; Wang, X. Source apportionment of VOCs in a typical medium-sized city in North China Plain and implications on control policy. J. Environ. Sci. (China) 2021, 107, 26–37. [Google Scholar] [CrossRef]
- Tan, Q.; Liu, H.; Xie, S.; Zhou, L.; Song, T.; Shi, G.; Jiang, W.; Yang, F.; Wei, F. Temporal and spatial distribution characteristics and source origins of volatile organic compounds in a megacity of Sichuan Basin, China. Environ. Res. 2020, 185, 109478. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-C.; Wang, J.-L.; Liu, S.-C.; Candice Lung, S.-C. Assessment of vehicular and non-vehicular contributions to hydrocarbons using exclusive vehicular indicators. Atmos. Environ. 2006, 40, 6349–6361. [Google Scholar] [CrossRef]
- Goldan, P.D.; Parrish, D.D.; Kuster, W.C.; Trainer, M.; McKeen, S.A.; Holloway, J.; Jobson, B.T.; Sueper, D.T.; Fehsenfeld, F.C. Airborne measurements of isoprene, CO, and anthropogenic hydrocarbons and their implications. J. Geophys. Res. Atmos. 2000, 105, 9091–9105. [Google Scholar] [CrossRef]
- Zheng, H.; Kong, S.; Xing, X.; Mao, Y.; Hu, T.; Ding, Y.; Li, G.; Liu, D.; Li, S.; Qi, S. Monitoring of volatile organic compounds (VOCs) from an oil and gas station in northwest China for 1 year. Atmos. Chem. Phys. 2018, 18, 4567–4595. [Google Scholar] [CrossRef]
- Yan, Y.; Peng, L.; Li, R.; Li, Y.; Li, L.; Bai, H. Concentration, ozone formation potential and source analysis of volatile organic compounds (VOCs) in a thermal power station centralized area: A study in Shuozhou, China. Environ. Pollut. 2017, 223, 295–304. [Google Scholar] [CrossRef]
- Kumar, A.; Singh, D.; Kumar, K.; Singh, B.B.; Jain, V.K. Distribution of VOCs in urban and rural atmospheres of subtropical India: Temporal variation, source attribution, ratios, OFP and risk assessment. Sci. Total Environ. 2018, 613–614, 492–501. [Google Scholar] [CrossRef]
- Rappenglück, B.; Fabian, P.; Kalabokas, P.; Viras, L.G.; Ziomas, I.C. Quasi-continuous measurements of non-methane hydrocarbons (NMHC) in the Greater Athens area during medcaphot-trace. Atmos. Environ. 1998, 32, 2103–2121. [Google Scholar] [CrossRef]
- Schauer, J.J.; Kleeman, M.J.; Cass, G.R.; Simoneit, B.R. Measurement of emissions from air pollution sources. 5. C1-C32 organic compounds from gasoline-powered motor vehicles. Environ. Sci. Technol. 2002, 36, 1169–1180. [Google Scholar] [CrossRef]
- Seila, R.L.; Main, H.H.; Arriaga, J.L.; Martínez, V.G.; Ramadan, A.B. Atmospheric volatile organic compound measurements during the 1996 Paso del Norte Ozone Study. Sci. Total Environ. 2001, 276, 153–169. [Google Scholar] [CrossRef]
- Liu, Y.; Shao, M.; Lu, S.; Chang, C.-C.; Wang, J.-L.; Fu, L. Source apportionment of ambient volatile organic compounds in the Pearl River Delta, China: Part II. Atmos. Environ. 2008, 42, 6261–6274. [Google Scholar] [CrossRef]
- Liu, B.; Liang, D.; Yang, J.; Dai, Q.; Bi, X.; Feng, Y.; Yuan, J.; Xiao, Z.; Zhang, Y.; Xu, H. Characterization and source apportionment of volatile organic compounds based on 1-year of observational data in Tianjin, China. Environ. Pollut. 2016, 218, 757–769. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, X.; Zhang, Z.; Lü, S.; Shao, M.; Lee, F.S.C.; Yu, J. Species profiles and normalized reactivity of volatile organic compounds from gasoline evaporation in China. Atmos. Environ. 2013, 79, 110–118. [Google Scholar] [CrossRef]
- Deichao, J. Analysis of Urban Atmospheric Volatile Organic Compounds (VOCs) Pollution Characteristics and Sources. Master’s Thesis, Shandong Jianzhu University, Shandong, China, 2015. [Google Scholar]
- Tian, L.; Wei, w.; Cheng, S.; Li, G.; Wang, G. VOCs composition spectrum and ozone generation potential in typical organic solvent-using industries. J. Saf. Environ. 2017, 17, 314–320. [Google Scholar]
- Alghamdi, M.A.; Khoder, M.; Abdelmaksoud, A.S.; Harrison, R.M.; Hussein, T.; Lihavainen, H.; Al-Jeelani, H.; Goknil, M.H.; Shabbaj, I.I.; Almehmadi, F.M.; et al. Seasonal and diurnal variations of BTEX and their potential for ozone formation in the urban background atmosphere of the coastal city Jeddah, Saudi Arabia. Air Qual., Atmos. Health 2014, 7, 467–480. [Google Scholar] [CrossRef]
- Hui, L.; Liu, X.; Tan, Q.; Feng, M.; An, J.; Qu, Y.; Zhang, Y.; Jiang, M. Characteristics, source apportionment and contribution of VOCs to ozone formation in Wuhan, Central China. Atmos. Environ. 2018, 192, 55–71. [Google Scholar] [CrossRef]
- An, J.L.; Wang, Y.S.; Wu, F.K.; Zhu, B. Characterizations of volatile organic compounds during high ozone episodes in Beijing, China. Environ. Monit. Assess. 2012, 184, 1879–1889. [Google Scholar]
- Chen, W.H.; Chen, Z.B.; Yuan, C.S.; Hung, C.H.; Ning, S.K. Investigating the differences between receptor and dispersion modeling for concentration prediction and health risk assessment of volatile organic compounds from petrochemical industrial complexes. J. Environ. Manag. 2016, 166, 440–449. [Google Scholar] [CrossRef]
- Liang, X.M.; Chen, L.G.; Sun, X.B.; Zhao, W.; Lu, Q.; Sun, J.R.; Chen, P.L.; Ye, D.Q. Raw Materials and End Treatment-based Emission Factors for Volatile Organic Compounds (VOCs) from Typical Solvent Use Sources. Huan Jing Ke Xue 2019, 40, 4382–4394. [Google Scholar]
- Hewitt, C.N.; Langford, B.; Possell, M.; Karl, T.; Owen, S.M. Quantification of VOC emission rates from the biosphere. TrAC Trends Anal. Chem. 2011, 30, 937–944. [Google Scholar] [CrossRef]
- Yao, D.; Tang, G.; Wang, Y.; Yang, Y.; Wang, L.; Chen, T.; He, H.; Wang, Y. Significant contribution of spring northwest transport to volatile organic compounds in Beijing. J. Environ. Sci. 2021, 104, 169–181. [Google Scholar] [CrossRef]
- Aucott, M.L.; McCulloch, A.; Graedel, T.E.; Kleiman, G.; Midgley, P.; Li, Y.-F. Anthropogenic emissions of trichloromethane (chloroform, CHCl3) and chlorodifluoromethane (HCFC-22): Reactive Chlorine Emissions Inventory. J. Geophys. Res. Atmos. 1999, 104, 8405–8415. [Google Scholar] [CrossRef]
- Zheng, H.; Kong, S.F.; Chen, N.; Niu, Z.Z.; Zhang, Y.; Jiang, S.N.; Yan, Y.Y.; Qi, S.H. Source apportionment of volatile organic compounds: Implications to reactivity, ozone formation, and secondary organic aerosol potential. Atmos. Res. 2021, 249, 105344. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, C.; Yan, W.; Wu, N.; Bo, Y.; Zhang, Q.; He, K. Characteristics and sources of non-methane VOCs and their roles in SOA formation during autumn in a central Chinese city. Sci. Total Environ. 2021, 782, 146802. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Ji, D.; Sun, J.; Wang, Y.; Yao, D.; Zhao, S.; Yu, X.; Zeng, L.; Zhang, R.; Zhang, H.; et al. Ambient volatile organic compounds in a suburban site between Beijing and Tianjin: Concentration levels, source apportionment and health risk assessment. Sci. Total Environ. 2019, 695, 133889. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Li, Q.; Wei, D.; Gao, L.; Tan, L.; Su, G.; Liu, G.; Liu, W.; Li, C.; Wang, Q. Emission characteristics of 99 NMVOCs in different seasonal days and the relationship with air quality parameters in Beijing, China. Ecotoxicol. Environ. Saf. 2019, 169, 797–806. [Google Scholar] [CrossRef]
- Liu, Y.; Li, L.; An, J.Y.; Huang, L.; Yan, R.S.; Huang, C.; Wang, H.L.; Wang, Q.; Wang, M.; Zhang, W. Estimation of biogenic VOC emissions and its impact on ozone formation over the Yangtze River Delta region, China. Atmos. Environ. 2018, 186, 113–128. [Google Scholar] [CrossRef]
- Liu, Y.; Song, M.; Liu, X.; Zhang, Y.; Hui, L.; Kong, L.; Zhang, Y.; Zhang, C.; Qu, Y.; An, J.; et al. Characterization and sources of volatile organic compounds (VOCs) and their related changes during ozone pollution days in 2016 in Beijing, China. Environ Pollut 2020, 257, 113599. [Google Scholar] [CrossRef]
- Luo, D.T.; Zhang, Q.M.; Liu, Z.; You, X.Y.; Huang, J.; Song, Y.F.; Zhang, J.Q. Characteristics and Source Apportionment of Volatile Organic Compounds in August in the Chang-Zhu-Tan Urban Area. Huan Jing Ke Xue 2022, 43, 3463–3472. [Google Scholar] [CrossRef]
- Li, J.; Zhai, C.; Yu, J.; Liu, R.; Li, Y.; Zeng, L.; Xie, S. Spatiotemporal variations of ambient volatile organic compounds and their sources in Chongqing, a mountainous megacity in China. Sci. Total Environ. 2018, 627, 1442–1452. [Google Scholar] [CrossRef]
- Zhou, Z.; Xiao, L.H.; Fei, L.L.; Yu, W.; Lin, M.; Huang, J.J.; Zhang, Z.S.; Tao, J. Characteristics and Source Apportionment of Volatile Organic Compounds (VOCs) in a Typical Industrial Area in Dongguan During Periods of Ozone and Non-ozone Pollution in Summer. Huan Jing Ke Xue 2022, 43, 4497–4505. [Google Scholar] [CrossRef]
- Chen, C.; Wang, L.; Zhang, Y.; Zheng, S.; Tang, L. Spatial and Temporal Distribution Characteristics and Source Apportionment of VOCs in Lianyungang City in 2018. Atmosphere 2021, 12, 1598. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Yue, H.; Cui, S.; Zhang, Y.; Li, H.; Wang, J.; Ge, X. Chemical Characteristics and Source-Specific Health Risks of the Volatile Organic Compounds in Urban Nanjing, China. Toxics 2022, 10, 722. https://doi.org/10.3390/toxics10120722
Wang J, Yue H, Cui S, Zhang Y, Li H, Wang J, Ge X. Chemical Characteristics and Source-Specific Health Risks of the Volatile Organic Compounds in Urban Nanjing, China. Toxics. 2022; 10(12):722. https://doi.org/10.3390/toxics10120722
Chicago/Turabian StyleWang, Jingyun, Hao Yue, Shijie Cui, Yunjiang Zhang, Haiwei Li, Junfeng Wang, and Xinlei Ge. 2022. "Chemical Characteristics and Source-Specific Health Risks of the Volatile Organic Compounds in Urban Nanjing, China" Toxics 10, no. 12: 722. https://doi.org/10.3390/toxics10120722
APA StyleWang, J., Yue, H., Cui, S., Zhang, Y., Li, H., Wang, J., & Ge, X. (2022). Chemical Characteristics and Source-Specific Health Risks of the Volatile Organic Compounds in Urban Nanjing, China. Toxics, 10(12), 722. https://doi.org/10.3390/toxics10120722





