1. Introduction
Nowadays, commercial poultry breeding is characterized by specialized fattening (meat-type) and laying lines (egg-type), i.e., meat-type genotypes are not used for egg production and laying genotypes are managed for efficient egg production where carcasses of culled hens are considered a by-product. Contrary to meat-type genotypes, where both sexes are used, in laying hybrids, only hens are used for egg production. Since male offspring of layers do not produce enough meat, they are not used for fattening and are culled on their first day of life, in both organic and conventional farming. This practice has raised ethical concerns in some European Union (EU) countries, including Germany [
1,
2], leading to research into alternatives. One alternative to killing day-old male chicks is the use of dual-purpose breeds: breeds that produce meat (males) and lay eggs (females). As dual-purpose breeds have only become interesting in recent years, they are not able to keep up with specialized meat-type and laying breeds. Dual-purpose hens lay fewer eggs and the males produce less meat even when fattened over a longer period of time. These inefficiencies also mean an increase in production costs associated with feed and housing, resulting in higher product prices [
3]. Yet, dual-purpose breeds could produce an improved meat quality and taste, in addition to meeting consumer animal welfare expectations; therefore, consumers might be willing to pay a higher price for these products [
1,
4,
5].
The movement for dual-purpose breeds does not only stem from the ethical aspects surrounding the culling of day-old chicks. The current specialized lines of production have led to a limited gene pool used in poultry breeding; therefore, the use of dual-purpose breeds, particularly traditional (or local) breeds, is important to the conservation of poultry genetic resources [
6,
7]. Traditional breeds, such as the French Bresse Gauloise (BG) breed, have also been used in Germany as dual-purpose chickens, while local breeds, such as the Vorwerkhuhn (VH) originating from Germany, are mainly kept by hobby breeders. However, their laying performance is rather low. Crossbreeding of such traditional breeds with high-performing commercial laying hens such as White Rock (WR) could be used to produce a dual-purpose genotype with a higher laying performance [
8].
Another problem in the poultry industry is that animal farming requires a high amount of protein-rich feedstuff. The production of these feedstuffs causes a greater environmental impact on the entire poultry farming system than rearing the animals [
9]. Although the requirement for large amounts of protein-rich feedstuffs has attracted criticism in recent years, it will likely remain necessary to meet future demands for human dietary protein [
10]. Soybeans, specifically soybean meal, are widely used as a protein source in poultry diet formulations; however, since the EU’s soybean yield is not sufficient to cover the requirements of its own poultry industry, there is a need to import soybean products from other countries, such as United States of America, Brazil and Argentina [
11,
12]. The large amount of soybean imports (13 million tons by the EU in 2016 [
11] and 3.5 million tons by Germany in 2017 [
13]) contributes to instability in the EU agricultural sector, mainly due to price volatility of soybeans on the global market and production sustainability issues [
12]. Additionally, EU citizens are concerned with genetically modified soy crops and deforestation in the Americas [
12,
14]. Alternatives to soybean products as poultry feed ingredients are regionally grown protein crops, such as beans and peas. These would contribute to a greater independency of local agricultural industries, as they would no longer have to rely on soy imports and their volatile prices, and provide environmental benefits like biological nitrogen fixation, in addition to having the potential to increase poultry production efficiencies [
15].
Faba beans (
Vicia faba L.; FBs) are one of the oldest and most widely cultivated legumes [
16]. They contain approximately 30% protein [
17], which is complemented further by an advantageous amino acid composition rich in lysine, yet variable in methionine and cysteine [
16]. These characteristics make the FB a suitable candidate as poultry feed protein source [
17]. In spite of their high nutritional value, FBs are considered to contain antinutritional factors (i.e., vicin and convicin; together abbreviated as VC) that have challenged their use in poultry diets [
18,
19]. The levels of VC vary depending on the FB cultivar [
16]. It remains unclear whether a modern low-VC cultivar contains a low enough amount of antinutritional factors to allow for the substitution of soybean meal with FB in a poultry diet [
20]. Literature is conflicted regarding the effect of FB as a dietary protein source in poultry diets. Not all findings are conclusive and appear to depend on the antinutritional characteristics of FB as well as bird physiological development [
17]. For example, Laudadio et al. [
21] and Dänner et al. [
20] find that FB can be included in laying hens’ diets without having a significant negative effect on laying performance or egg quality; however, it was found in [
22] that the inclusion of FB in laying hens’ diets can decrease egg weight. In addition, broiler nutrition (apparent metabolizable energy; AMEn) values are found to be adversely affected by antinutritional factors in FB; however, adult cockerels appear to be more resilient towards antinutritional factors [
17].
The main objective of this study was to test the effect of FB as a locally produced soybean meal replacement on meat quality traits, including sensory analysis, of cockerels for two local dual-purpose chicken breeds and one high-performing laying line. The locally grown FBs contain average and reduced VC contents in order to ascertain the limit of antinutritional factors in these genotypes. Furthermore, this system offers the chance to fatten the brothers of laying hens of local breeds in a regional production system and therefore to refrain from culling male chicks. More specifically, the aim of this study is to assess whether traditional breeds can be used as a basis to develop a local alternative poultry production system based on their meat quality and sensory characteristics under different diets. The effect of poultry diets containing different local FB cultivars in these particular genotypes remains the variable under investigation.
2. Materials and Methods
This experiment is in accordance with the European Union directive on the protection of animals used for scientific purposes (Directive 2010/63/EU) and was approved by the Lower Saxony State Office for Consumer Protection and Food Safety (LAVES; ref. 33.9-42502-04-17/2622).
2.1. Animal Management and Sampling
One-day-old male chicks of Bresse Gauloise (BG), Vorwerkhuhn (VH), and White Rock (WR) breeds were reared in indoor pens using a commercial starter at the Friedrich-Loeffler-Institute (FLI; Celle, Germany) for three weeks. The BG and VH chicks were directly hatched at FLI; WR chicks were provided by from Lohmann Tierzucht GmbH (Cuxhaven, Germany). At 21 days, 120 BG, 94 VH, and 120 WR male chicks were transported to the Department of Animal Sciences at the University of Goettingen (Goettingen, Germany), where 40 chicks of each genotype were randomly assigned to one of three feed groups. Decreased hatchability of VH chicks resulted in reduced feed group size (approx. 30 animals per feed group). In total, there were nine different experimental groups (3 breeds × 3 feed groups). The chickens were reared in an indoor-floor system with a solid floor and with fans for ventilation and cooling. The density of each pen was 10 birds/3 m2, with the exception of VH, where seven to eight birds were held per pen; for each feed/breed combination there were four replicates. The temperature was held constant at 20 ± 2 °C, and the photoperiod was 16 h.
Three different diets (
Table 1) were fed across all breeds starting at day 21. The control (C) group was fed soybean-meal-based feed, while the rations of the other two groups were based on FB feed mixture. The difference between the two FB-based diets was the VC content: one diet had a high (0.14%) VC content (VC+); while the VC content of the other diet was low (0.02%) (VC-).
Table 1 outlines the ingredient composition of each experimental diet in percentage of ingredient per kg of feed. All animals were provided feed (pelleted) and water ad libitum.
The animals were reared from September 2017 to December 2017 or January 2018, therefore reaching slaughter ages of 10, 15, and 16 weeks for BG, VH, and WR, respectively. Age differences are due to different growth rates of the breeds reaching the same body weight (approximately 2100 g) at slaughter. At slaughter, the birds were electrically stunned, exsanguinated by neck cut, scalded, eviscerated, weighed, and chilled at 4 °C for 24 h. Twenty-four hours after slaughter (post mortem; p.m.) the carcasses were weighed and manually dissected. Results concerning the animals’ growth performance and carcass parameters have been reported elsewhere [
23].
For each of the nine groups (3 breeds × 3 feeds), approximately 20 animals (BG 20, VH 16, WR 20) were allocated for sensory analysis, and 10 samples per group were used for physicochemical analyses; the rest of the samples were used for analyses not pertaining this study.
2.2. Physicochemical Analysis
The following physicochemical analyses were conducted on 10 samples per feed/breed combination: pH; color; water holding capacity (WHC), measured as storage loss and cooking loss; instrumental tenderness, measured as shear force; and content of flavor-related nucleotides, i.e., inosine-5′-monophosphate (IMP), adenosine-5′-monophosphate (AMP), and inosine. pH and nucleotides were analyzed in the left breast. Remaining parameters were recorded using the right breast samples, which were stored between 24 and 72 h p.m. in modified atmosphere (80% O2/20% CO2) packaging (MAP) using a PP tray with absorbent liners and heat-sealed with an oriented OPET/PP film (<3 cm3/m2/24 h bar oxygen transmission rate; <12 cm3/m2/24 h bar carbon dioxide transmission rate) using a vacuum packaging machine (TS 100, KOMET Maschinenfabrik GmbH, Plochingen, Germany) and stored at 4 °C without illumination. The pH values were determined at three different times (20 min p.m., 24 h p.m., and 72 h p.m.) by inserting a pH-electrode and a thermometer (Portamess 911, Knick Elektronische Messgeräte GmbH & Co. KG, Berlin, Germany) into the cranial part of the left breast. The pH-meter was regularly calibrated between breeds, using standard buffers for pH 4 and pH 7 at room temperature. Color was quantified using CIELAB coordinates (L*a*b* values). Three measurements were taken on non-overlapping areas (free of obvious color defects) using a colorimeter (CR-600d, Konica Minolta, Tokyo, Japan). Color was recorded on the ventral part of the right breast with skin at 24 h p.m. and without skin at 24 and 72 h p.m. The average of the three-color measurements was used in further analysis. The spectrometer was calibrated before every session using a white tile provided by the manufacturer. Storage loss was monitored by weighing the right breast at 24 h p.m. prior to packaging and reweighing at 72 h p.m., where the percent difference in weight was attributed to storage loss. Afterwards, these samples were frozen at −20 °C until cooking loss and shear force analyses were conducted (approx. 8 weeks p.m.). Samples were thawed overnight at 4 °C and freshly vacuum-packaged; then they were immersed in a hot water bath (1092, GFL Gesellschaft für Labortechnik mbH, Burgwedel, Germany) at 80 °C for 50 min until reaching a core temperature of 76 °C, as measured by inserting a thermometer (926, Testo SE & Co. KGaA, Lenzkirch, Germany). After cooling to room temperature, the samples were weighed in order to calculate the cooking loss as a percentage of overall weight loss. The samples were later wrapped in aluminum foil and stored overnight at 4 °C. Prior to conducting shear force analysis, samples were left unwrapped at room temperature for 10 min. Shear force values were measured with a TA.XTplus Texture Analyzer (Stable Micro Systems, Surrey, UK) equipped with a 5 kg load cell and a Meullenet-Owens Razor Shear Blade (MORS-Blade). The conditions for the test were the following: pretest speed 2 mm/s, test speed 10 mm/s, trigger type 10 g. Each breast sample was sheared four times perpendicular to the muscle fiber orientation, with a 1.5 cm distance from each cut. Results of each sample are presented as an average of the four measurements. Shear force is reported as the peak shear force (N) which is necessary to completely shear through the sample.
To determine the content of IMP, AMP, and inosine, samples of raw meat from the left breasts (5 samples per group) and left legs (10 samples per group) were taken at 24 h p.m., frozen with liquid nitrogen, and stored at −72 °C. Six months after slaughter, IMP, AMP, and inosine content was determined using the method of Morzel and Van De Vis [
24] with some modifications. Minced samples (0.200 g) were homogenized (Schuett-homgenplus homogenizer, Schuett-biotec GmbH, Germany) with 1 mL of 5% (
w/v) TCA (aq) for 1 min at 1600 rpm (Pico & Fresco 17/21 centrifuge, ThermoElectron LED GmbH, Osterode, NE, Germany) followed by chilling on ice for 15 min. The liquid extract was centrifuged at 4 °C for 5 min at 12,000 ×
g. The supernatant (200 µL) was diluted 1:4 (
v/v) for the breast samples and 1:2 (
v/v) for the thighs, with 5 % (
w/v) TCA (aq) at pH 7.0. Extracts were kept at −20 °C before being injected into the HPLC system. The system (VWR Hitachi, Chromaster) was equipped with a 5260 pump, a 5260 autosampler (injection volume: 10 µL), and a 5410 UV detector operating at 260 nm. A LiChroCart Licrosphere 100 RP8 (250 × 4.6 mm, 5 μm) column was maintained at 30 °C in a 5310 column oven. The mobile phase consisted of 100 mM KH
2PO
4 (aq), 1.44 mM TBAHS (aq), and 0.5% methanol (aq, pH 7.0). Quantification was performed by an external calibration method. Identification of the analytes was performed by comparison of retention times. All analyses were performed in duplicate.
2.3. Sensory Analysis
The samples for sensory analysis were stored between 24 and 72 h p.m. in MAP packaging under the same conditions as samples for physicochemical analysis (see
Section 2.2). At 72 h p.m., the samples were vacuum-packed in plastic bags and frozen at −20 °C until training or evaluation. All training and evaluation sessions took place in the sensory laboratory at the University of Goettingen, which complies with the international standard ISO 8589. All samples were thawed overnight at 4 °C prior to cooking for training or evaluation. Chicken breast samples were prepared according to the cooking loss procedure (see
Section 2.2). The breasts were cut in 1 cm
2 pieces and served on warm plates (
Figure 1a) marked with a 3-digit code. Leg samples with skin were roasted in a commercial oven for 35 min at 190 °C and 50% air humidity until they reached a minimal core temperature of 76 °C, measured by inserting a thermometer (926, Testo SE & Co. KGaA, Lenzkirch, Germany) into the thigh, and kept warm in a food warmer (Bain Marie, Bartscher, Salzkotten, Germany) until served. Each panelist received one complete leg on a warm plate marked with a 3-digit code (
Figure 1b).
Conventional descriptive analysis was carried out by a trained panel consisting of 10 assessors, who were experienced in descriptive sensory profiling of meat-related products and were trained and selected according to ISO 8586. All assessors provided written informed consent prior to participation.
The panel evaluated different chicken breasts and chicken legs from three breeds (BG, VH, WR) fed with three different diets (C, VC+, VC-), resulting in nine different products per cut (breast, leg). Assessors defined attributes in appearance, odor, taste, flavor, and texture that best described the samples and were trained further in these. To evaluate the breast samples, a total of 21 attributes were collected. Similarly, a total of 20 attributes were collected to describe the leg samples. A list of all attributes along with their definitions and scales for breasts and legs are presented in
Table A1 and
Table A2 (
Appendix A), respectively. Training per cut was directly followed by evaluation of the nine products per cut, i.e., chicken breast training and evaluation was completed prior to starting sensory analysis of leg products.
The trained panelists evaluated the nine chicken breast products in triplicate in four sessions, where each assessor evaluated six samples per session. Leg products were only assessed in duplicate. All samples were evaluated, in a sequential monadic manner, in three different set orders that were allocated to three or four assessors for each session. After the evaluation of each sample, panelists were asked to neutralize their senses by drinking water; additionally, untoasted white bread was available for neutralization. Using EyeQuestion survey software (Version 4.8.7, EyeQuestion, Elst, the Netherlands), each sensory attribute was evaluated on a 10 cm unstructured line with an unmarked scale ranging from 0 (no perception) to 100 (strong perception). The electronically collected data were later used for statistical analysis.
2.4. Statistical Analysis
Due to the different slaughter ages, the statistical analyses of the evaluated physicochemical and sensory data were done separately for each breed; therefore, the feed effect was compared within breed.
Data analysis of physicochemical parameters was performed with SPSS (Version 24, IBM Corporation, New York, NY, USA) statistical software. Mean values were calculated and feed effect was compared within each breed with a one-way ANOVA using Tukey’s multiple comparison statistical test at a 95% confidence level (α = 0.05).
For the statistical analysis of sensory data, the linear mixed model (LMM) procedure from SPSS (Version 26, IBM Corporation, New York, NY, USA) was used for mixed model calculations. All calculations were compared within each breed. In the statistical model for breast samples, “feed” was defined as a fixed effect, while “panelist”, “animal”, “feed*panelist”, and “feed*animal” were defined as random effects. In the statistical model for leg samples, “feed” was defined as a fixed effect, while “panelist” and “feed*panelist” were defined as random effects. Within the model, a least significant difference (LSD) statistical test at a 95% confidence level (α = 0.05) was used.
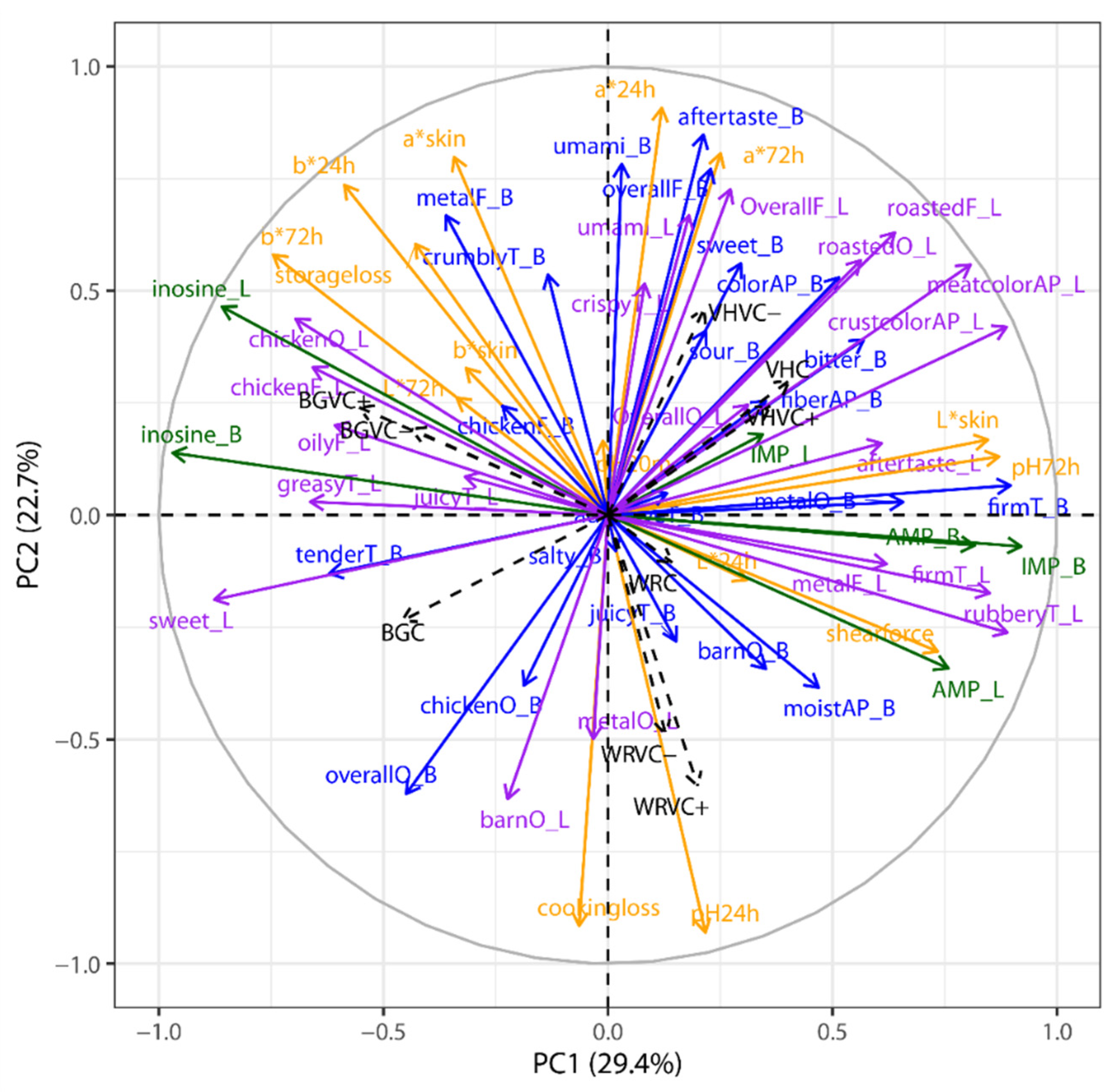
For the sole purpose of visualization, a principle component analysis (PCA) was carried out with treatment group means across all parameters. The PCA was computed using RStudio (version 1.2.5033, R Foundation for Statistical Computing, Vienna, Austria) coupled with the FactoMineR package [
25]. Variables were standardized and number of components was assessed based on a scree-plot.
4. Discussion
Our study illustrates that, as within other more specialized poultry production systems (i.e., meat-type and laying), faba beans present themselves as a feasible dietary protein source. Although total product yield is an important factor to consider with different feedstuff groups, it is not the only defining factor in evaluating the acceptability of FB in poultry meat production. Therefore, our study focused on evaluating the effect different FB diets have on physicochemical as well as organoleptic meat quality of three different breeds; few previous studies focus on such aspects.
pH is one of the most important physicochemical characteristics in meat since it is related to water holding capacity and color. Similar values to BG and VH for pH at 24 h p.m. were obtained by Siekmann et al. [
26] for a dual-purpose hybrid (Lohmann Dual) and by Muth et al. [
27] for BG. Although there are slight differences in pH values between the different breeds (which might be attributed to other factors such as age or genetics), within each breed there has been no effect between feed groups in pH at 20 min and 24 h p.m. This is also reported in [
19,
28], where researchers do not report any significant differences between soy-based and faba-bean-based diets in chicken breast. Nonetheless, we observe that a high content of VC caused a low pH value at 72 h p.m. in BG.
Faba beans were found to influence meat color in broiler chickens by Laudadio et al. [
19] and in guinea fowl broilers by Tufarelli and Laudadio [
28]. This is important given that meat color is assumed to be one of the most important characteristics evaluated at the point of purchase [
29]. An unfamiliar product color can negatively impact consumer expectations [
30]. In our study, FB diets did not affect color uniformly across breeds. In WR, b* values were significantly reduced with FB diets with or without skin and in both 24 and 72 h p.m. chicken breast. No color differences were recorded in other color parameters or in other breeds; therefore, it is likely that the color difference in WR chicken breast skin is due to the refraction of the altered lean meat color (below the partially translucent skin). The lower b* values are contradictory to the findings of Laudadio et al. [
19], who fed a wheat-based diet where the control group dietary protein source was soybean meal and the test group dietary protein source was faba beans. WR chicken breast samples are also lighter (L*) in color when birds are fed a VC- diet; however, FB in general did not increase lightness. Laudadio et al. [
19] observed darker samples with the feeding of faba beans in broiler chicks, whereas Tufarelli and Laudadio [
28] found faba beans to increase lightness in guinea fowl broiler meat. In BG chicken breasts, increased redness values (a*) are observed for both time periods (24 and 72 h p.m.); this corresponds with the findings of [
19] in broiler chicken meat. We do not observe an effect of diet on product color in VH samples, illustrating the need to not overgeneralize the effect of feed on meat color across breeds.
We also observe an effect of FB feed on instrumental tenderness and water holding capacity, where WR chicken breast samples raised on FB diets have increased water holding capacity (reduced storage losses) and are more tender (decreased shear force) as measured by a texture analyzer. Laudadio et al. [
19] also observed a statistically significant increase in water holding capacity (as measured by a filter paper–oven drying method) in their faba-bean-fed broiler chicken breast samples; drip loss also tended to be decreased (as measured with the filter paper method). Tufarelli and Laudadio [
28] also verified faba beans’ effect on water holding capacity in guinea fowl broilers. Unfortunately, to the best of our knowledge, no studies exist investigating instrumental tenderness of poultry meat from chickens fed faba bean diets. However, it would be expected that instrumental tenderness values are reduced (i.e., samples are more tender) with an increased water holding capacity.
AMP, IMP, and inosine all derive from the breakdown of adenosine triphosphate (ATP), which occurs in muscle during the slaughter and the postmortem aging phases [
31]. Among these, IMP plays a predominant role in the formation of meat flavor by contributing to the umami taste [
32]. The data obtained in this study confirm that IMP is the most abundant nucleotide, while AMP shows the lowest concentration values [
33,
34,
35]. Furthermore, across all the three breeds in this study, IMP and inosine contents tend to be higher in chicken breast compared to the legs, as already observed in other studies [
33,
34,
36].
With regard to the effect of the feed for each of the genotypes, from a general point of view it seems that the replacement of soybean meal with FB has no significant effect on the content of the selected nucleotides. In the past, levels of IMP in chicken meat were proved to be related to dietary purine nucleotide, betaine, and soybean isoflavone contents [
37]. Although there is a lack of information about the concentration of these compounds in the three experimental diets, we can assume that the replacement of soybean meal with FB did not lead to significant changes in their levels.
To the best of our knowledge, we are the first to investigate the effect of faba bean diets on organoleptic properties of poultry meat. Furthermore, in profiling three different breeds, our results offer three distinct sensory profiles for Bresse Gauloise (BG), Vorwerkhuhn (VH), and White Rock (WR) poultry meat; to the best of our knowledge, these have also not yet been documented. Instead, research has focused on creating sensory profiles of meat from chickens reared in different production systems (e.g., [
4,
38,
39,
40]) or compared to other indigenous breeds (e.g., [
41,
42]) or to other specific diets (e.g., [
38,
43]).
As research has shown, depending on several factors like breed [
44], feedstuff [
38], and age [
4,
39], chicken breast samples have different attributes in different intensities that best describe their organoleptic properties. However, there are a few general attributes that are present in chicken meat despite the abovementioned factors, such as chicken and metallic aroma, umami taste, and chicken and metallic flavor. Texture is one of the most important sensory qualities associated with consumers’ satisfaction [
45], and attributes such as firmness, tenderness, and crumbliness are usually of interest for meat samples. Horsted et al. [
39] showed that chicken aroma, chicken flavor, and umami taste in breast samples are positively correlated to the product’s overall liking, while a metallic aroma and taste have a negative correlation to the overall liking. A study by Lawlor et al. [
38] showed that for one of five groups of consumers, firmness of the initial bite was correlated to product preference, while for a different group an astringent taste and a crumbly texture were positively correlated to overall liking. On the other hand, for roasted chicken samples, Sow and Grognet [
46] showed that juiciness, oiliness, sweetness, and hardness are attributes correlated to product preference, while chewiness, astringency, and smoothness were negatively correlated to preference.
In general, the effect of the different feed groups led to slight organoleptic changes in BG breast and leg samples. Breast samples of animals fed with FB showed a slight, not statistically significant, improvement in their sensory profile, i.e., higher score in those attributes (often) positively associated with consumer preference, e.g., more intense chicken aroma and more intense umami taste. Similarly, FB diets improved some attributes in BG leg samples, though not significantly, when compared to control, e.g., decrease in barn and metallic aroma. In general, the sensory profiles for Bresse Gauloise, especially reared with FB diets, showed a slight improvement in organoleptic properties.
The effect of FB diets in Vorwerkhuhn (VH) led to several organoleptic changes in breast and leg samples, particularly in aroma, flavor, and texture. In the overall profile of breast samples, the effect of FB was reflected by the improvement of some attributes, particularly barn aroma and tenderness. The VC- content led to the least intense barn aroma of all samples. A similar effect was observed in tenderness, as the VC- diet showed the highest tenderness in breast samples. Interestingly, for both attributes, the opposite effect was noticed with a VC+ content diet, suggesting that a faba-bean-based diet only improves the aroma and tenderness when the VC content is low. Our results also show that FB diets favored the flavor of leg samples by reducing the metallic taste and the overall aftertaste when compared to a soy-based diet. The effect of a low-VC-content diet improved the texture of leg samples, particularly their juiciness. Therefore, there appears to be no consistent faba bean effect for VH, but VC content remains important in determining organoleptic quality.
The White Rock (WR) sensory profile deviated from BG and VH, which was not surprising given that WR is a laying breed, whereas the other two are traditional dual-purpose breeds. In breast samples, the FB diets mostly had a negative effect on texture attributes, particularly for tenderness, which is an important attribute associated with overall liking of chicken. In leg samples, the FB diets also affected aroma and flavor attributes although not statistically significantly. These changes were also observed in the other two breeds, where barn aroma and metallic aroma and taste decreased with the inclusion of faba beans in the diet. Similar to breast samples, FB diets had a negative effect on texture attributes, namely firmness and particularly crispiness in WR. While Lawlor et al. [
38] showed that firmness is a desired attribute in chicken breast meat, the preference for crispiness is unknown. Nonetheless, it could be accepted that (older) WR animals fed with faba beans will not likely produce the most acceptable poultry meat on the market.
Due to the differing slaughter ages, it is difficult to compare the three different breeds since genotype, age, feed, slaughter conditions, and production systems can influence the sensory profile of chicken breast meat. According to [
44], genetic variation only accounts for small differences in taste attributes, whereas age has been shown to increase the intensity of aroma and flavor in meat [
4,
39]. Our results also show a slight increase in overall flavor for breeds reared longer, especially when comparing the overall flavor of the three breeds fed with control (soy-based) feed. Therefore, the numerical difference amongst the breeds of this study should be interpreted with caution.
Finally, it is important to mention that consumers see the concept of dual-purpose breeds as a more animal-friendly practice [
1,
5,
47,
48] for which they would be willing to pay a higher price if meat quality is improved while their expectations on animal welfare are met [
1,
4,
5]. Additionally, studies have shown that consumers are willing to pay more for regional products [
49,
50]. Consequently, consumers should be willing to pay more for this production system of dual-purpose local breeds fed with regional feedstuff, especially when doing so would improve meat quality parameters.
These results are of relevance to the poultry industry, particularly to breeders of particular local breeds used for dual purposes. This production system offers small-scale breeders an opportunity to target niche markets that demand more ethical or sustainable production methods. The improvement of meat quality parameters and the increase in overall flavor of meat from chickens fed with faba beans show a promising future for this production system.