Physical Properties and Consumer Evaluation of Cocoa Bean Shell-Functionalized Biscuits Adapted for Diabetic Consumers by the Replacement of Sucrose with Tagatose
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Samples
2.3. Formulation of the Biscuits
2.4. Analytical Procedures
2.4.1. Total Dietary Fiber Determination
2.4.2. Physicochemical Analyses
2.4.3. Structural Analyses
2.5. Sensory Analyses
2.5.1. Consumer Evaluation
2.5.2. Napping® Sensory Analysis
2.6. Statistical Analysis
3. Results and Discussion
3.1. Total Dietary Fiber Content
3.2. Physicochemical Characterization
3.3. Structural Characterization
3.4. Sensory Analyses
3.4.1. Hedonic Consumer Acceptance Evaluation
3.4.2. Napping® Sensory Characterization
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wildman, R.E.; Kelley, M. Nutraceuticals and Functional Foods. In Handbook of Nutraceuticals and Functional Foods, 2nd ed.; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: New York, NY, USA, 2007. [Google Scholar]
- Shahidi, F.; Naczk, M. Phenolics in Food and Nutraceuticals; CRC Press: Boca Raton, FL, USA; Taylor & Francis: Boca Raton, FL, USA, 2006. [Google Scholar]
- Kaczmarczyk, M.M.; Miller, M.J.; Freund, G.G. The health benefits of dietary fiber: Beyond the usual suspects of type 2 diabetes mellitus, cardiovascular disease and colon cancer. Metab.-Clin. Exp. 2012, 61, 1058–1066. [Google Scholar] [CrossRef]
- Lecumberri, E.; Mateos, R.; Izquierdo-Pulido, M.; Rupérez, P.; Goya, L.; Bravo, L. Dietary fibre composition, antioxidant capacity and physico-chemical properties of a fibre-rich product from cocoa (Theobroma cacao L.). Food Chem. 2007, 104, 948–954. [Google Scholar] [CrossRef]
- Nsor-Atindana, J.; Zhong, F.; Mothibe, K.J. In vitro hypoglycemic and cholesterol lowering effects of dietary fiber prepared from cocoa (Theobroma cacao L.) shells. Food Funct. 2012, 3, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific Opinion on Dietary Reference Values for carbohydrates and dietary fibre. EFSA J. 2010, 8, 1462. [Google Scholar]
- Rojo-Poveda, O.; Barbosa-Pereira, L.; Zeppa, G.; Stévigny, C. Cocoa Bean Shell—A By-Product with Nutritional Properties and Biofunctional Potential. Nutrients 2020, 12, 1123. [Google Scholar] [CrossRef] [PubMed]
- Barbosa-Pereira, L.; Rojo-Poveda, O.; Ferrocino, I.; Giordano, M.; Zeppa, G. Assessment of volatile fingerprint by HS-SPME/GC-qMS and E-nose for the classification of cocoa bean shells using chemometrics. Food Res. Int. 2019, 123, 684–696. [Google Scholar] [CrossRef] [PubMed]
- Barbosa-Pereira, L.; Rojo-Poveda, O.; Ferrocino, I.; Giordano, M.; Zeppa, G. Analytical dataset on volatile compounds of cocoa bean shells from different cultivars and geographical origins. Data Brief 2019, 25, 104268. [Google Scholar] [CrossRef] [PubMed]
- Öztürk, E.; Ova, G. Evaluation of Cocoa Bean Hulls as a Fat Replacer On Functional Cake Production. Turk. J. Agric. Food Sci. Technol. 2018, 6, 1043–1050. [Google Scholar] [CrossRef]
- Martínez-Cervera, S.; Salvador, A.; Muguerza, B.; Moulay, L.; Fiszman, S. Cocoa fibre and its application as a fat replacer in chocolate muffins. LWT Food Sci.Technol. 2011, 44, 729–736. [Google Scholar] [CrossRef]
- Collar, C.; Rosell, C.M.; Muguerza, B.; Moulay, L. Breadmaking performance and keeping behavior of cocoa-soluble fiber-enriched wheat breads. Food Sci. Technol. Int. 2009, 15, 79–87. [Google Scholar] [CrossRef]
- Kārkliņa, D.; Gedrovica, I.; Reca, M.; Kronberga, M. Production of biscuits with higher nutritional value. In Proceedings of the Latvian Academy of Sciences. Section B. Natural, Exact, and Applied Sciences; Sciendo: Warsaw, Poland, 2012; pp. 113–116. [Google Scholar]
- Rojo-Poveda, O.; Barbosa-Pereira, L.; Mateus-Reguengo, L.; Bertolino, M.; Stévigny, C.; Zeppa, G. Effects of particle size and extraction methods on cocoa bean shell functional beverage. Nutrients 2019, 11, 867. [Google Scholar] [CrossRef] [PubMed]
- Cantele, C.; Rojo-Poveda, O.; Bertolino, M.; Ghirardello, D.; Cardenia, V.; Barbosa-Pereira, L.; Zeppa, G. In vitro bioaccessibility and functional properties of phenolic pompounds from enriched beverages based on cocoa bean shell. Foods 2020, 9, 715. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Report On Diabetes: World Health Organization; 924156525X; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
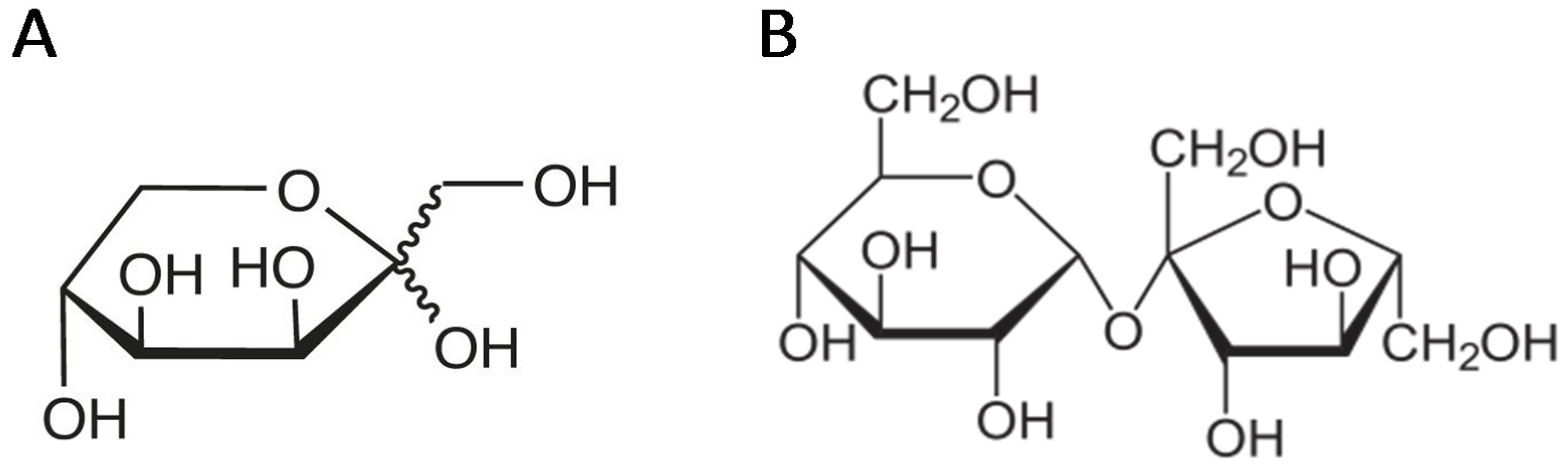
- Roy, S.; Chikkerur, J.; Roy, S.C.; Dhali, A.; Kolte, A.P.; Sridhar, M.; Samanta, A.K. Tagatose as a potential nutraceutical: Production, properties, biological roles, and applications. J. Food Sci. 2018, 83, 2699–2709. [Google Scholar] [CrossRef] [PubMed]
- Oh, D.-K. Tagatose: Properties, applications, and biotechnological processes. Appl. Microbiol. Biotechnol. 2007, 76, 1. [Google Scholar] [CrossRef]
- Levin, G.V. Tagatose, the new GRAS sweetener and health product. J. Med. Food 2002, 5, 23–36. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA); Turck, D.; Bresson, J.L.; Burlingame, B.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; Naska, A. Scientific Opinion on the energy conversion factor of d-tagatose for labelling purposes. EFSA J. 2016, 14, e04630. [Google Scholar]
- Rulis, A. Agency Response Letter GRAS Notice NR GRN 000078. Available online: http://pages.citebite.com/e2f2x0h3y0tjw (accessed on 25 March 2020).
- Taylor, T.; Fasina, O.; Bell, L. Physical properties and consumer liking of cookies prepared by replacing sucrose with tagatose. J. Food Sci. 2008, 73, 145–151. [Google Scholar] [CrossRef]
- Armstrong, L.M.; Luecke, K.J.; Bell, L.N. Consumer evaluation of bakery product flavour as affected by incorporating the prebiotic tagatose. Int. J. Food Sci. Technol. 2009, 44, 815–819. [Google Scholar] [CrossRef]
- Browne, C.A. A Handbook of Sugar Analysis: A Practical and Descriptive Treatise for Use in Research, Technical and Control Laboratories. Available online: https://chestofbooks.com/food/science/Experimental-Cookery/The-Solubility-Of-The-Sugars.html (accessed on 28 April 2020).
- Kawamura, Y. D-Tagatose: Chemical a Technical Assessment; CTA: Chicago, IL, USA, 2004; Available online: http://www.fao.org/3/b-au002e.pdf (accessed on 29 April 2020).
- Lai, H.M.; Lin, T.C. Bakery Products: Science and Technology; Blackwell Publishing: Ames, IA, USA, 2008. [Google Scholar]
- AACC. Approved Methods of Analysis, 11th ed.; Method 10-53.01. Baking Quality of Cookie Flour-Macro Wire-cut Formulation. Approved November 3; AACC International: St. Paul, MN, USA, 1999. [Google Scholar]
- AOAC. Oficial Method 991.43 Total, Soluble and Insoluble Dietary Fibre in Foods; First action 1991 and final action 1994; AOAC Oficial Methods of Analysis: Rockville, MD, USA, 1991. [Google Scholar]
- AACC. Approved Methods of Analysis, 11th ed.; Method 10-50.05. Baking Quality of Cookie Flour. Approved November 3; AACC International: St. Paul, MN, USA, 1999. [Google Scholar]
- Lim, J. Hedonic scaling: A review of methods and theory. Food Qual. Prefer. 2011, 22, 733–747. [Google Scholar] [CrossRef]
- Pagès, J.; Cadoret, M.; Lê, S. The sorted napping: A new holistic approach in sensory evaluation. J. Sens. Stud. 2010, 25, 637–658. [Google Scholar] [CrossRef]
- Perrin, L.; Pagès, J. Construction of a product space from the ultra-flash profiling method: Application to 10 red wines from the Loire Valley. J. Sens. Stud. 2009, 24, 372–395. [Google Scholar] [CrossRef]
- Orden, D.; Tejedor-Romero, M. SensoGraph (Under Registration Process). Available online: https://sensograph.it (accessed on 11 May 2020).
- Orden, D.; Fernández-Fernández, E.; Rodríguez-Nogales, J.M.; Vila-Crespo, J. Testing SensoGraph, a geometric approach for fast sensory evaluation. Food Qual. Prefer. 2019, 72, 1–9. [Google Scholar] [CrossRef]
- Parliament, E. Regulation (EC) No 1924/2006 of the European Parliament and of the Council of 20 December 2006 on nutrition and health claims made on foods. Off. J. Eur. Union 2006, L404, 9–25. [Google Scholar]
- Červenka, L.; Brozková, I.; Vytrasová, J. Effects of the principal ingredients of biscuits upon water activity. J. Food Nutr. Res. 2006, 45, 39–43. [Google Scholar]
- Gallagher, E.; Kenny, S.; Arendt, E.K. Impact of dairy protein powders on biscuit quality. Eur. Food Res. Technol. 2005, 221, 237–243. [Google Scholar] [CrossRef]
- Struck, S.; Jaros, D.; Brennan, C.S.; Rohm, H. Sugar replacement in sweetened bakery goods. Int. J. Food Sci. Technol. 2014, 49, 1963–1976. [Google Scholar] [CrossRef]
- Hill, B.; Roger, T.; Vorhagen, F.W. Comparative analysis of the quantization of color spaces on the basis of the CIELAB color-difference formula. ACM Trans. Graph. (TOG) 1997, 16, 109–154. [Google Scholar] [CrossRef]
- Mamat, H.; Hill, S. Structural and functional properties of major ingredients of biscuit. Int. Food Res. J. 2018, 25, 462–471. [Google Scholar]
- Ktenioudaki, A.; Gallagher, E. Recent advances in the development of high-fibre baked products. Trends Food Sci. Technol. 2012, 28, 4–14. [Google Scholar] [CrossRef]
- Garcia-Serna, E.; Martinez-Saez, N.; Mesias, M.; Morales, F.J.; del Castillo, M.D. Use of coffee silverskin and stevia to improve the formulation of biscuits. Pol. J. Food Nutr. Sci. 2014, 64, 243–251. [Google Scholar] [CrossRef]
- Arimi, J.; Duggan, E.; O’sullivan, M.; Lyng, J.; O’riordan, E. Effect of water activity on the crispiness of a biscuit (Crackerbread): Mechanical and acoustic evaluation. Food Res. Int. 2010, 43, 1650–1655. [Google Scholar] [CrossRef]


| Ingredients | Quantities (g) | ||
|---|---|---|---|
| 0% (Control Biscuits) | 10% CBS Substitution | 20% CBS Substitution | |
| Wheat flour | 225.0 | 202.5 | 180.0 |
| Sucrose or tagatose | 100.0 | 100.0 | 100.0 |
| Butter (>82% fat) | 90.0 | 90.0 | 90.0 |
| CBS powder | 0.0 | 22.5 | 45.0 |
| Baking powder | 7.0 | 7.0 | 7.0 |
| Cocoa powder | 3.0 | 3.0 | 3.0 |
| NaCl | 1.2 | 1.2 | 1.2 |
| Water | 51.0 | 55.0 | 69.0 |
| Biscuit Sample | Sugar | CBS Percentage | Fiber | Moisture | aw | L* | a* | b* | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (g/100 g Dried Product) | (%) | |||||||||||||||||||
| S0 | Sucrose | 0 | 2.48 | ± | 0.23 cA | 1.69 | ± | 0.26 cB | 0.12 | ± | 0.03 cB | 50.78 | ± | 1.93 aA | 11.70 | ± | 0.39 aB | 23.66 | ± | 1.08 aB |
| S10 | 10 | 5.66 | ± | 0.56 bA | 3.21 | ± | 0.14 bB | 0.25 | ± | 0.00 bB | 33.56 | ± | 0.38 bB | 12.04 | ± | 0.29 aB | 17.10 | ± | 0.48 bB | |
| S20 | 20 | 8.70 | ± | 0.34 aA | 4.50 | ± | 1.05 aB | 0.37 | ± | 0.09 aB | 28.54 | ± | 0.67 cA | 9.04 | ± | 2.45 bA | 11.77 | ± | 2.90 cA | |
| Significance | *** | *** | *** | ** | *** | *** | ||||||||||||||
| T0 | Tagatose | 0 | 2.79 | ± | 0.07 cA | 4.76 | ± | 0.11 cA | 0.37 | ± | 0.01 cA | 44.78 | ± | 0.90 aB | 16.04 | ± | 0.34 aA | 30.90 | ± | 0.37 aA |
| T10 | 10 | 5.66 | ± | 0.58 bA | 5.69 | ± | 0.46 bA | 0.43 | ± | 0.02 bA | 35.49 | ± | 1.66 bA | 13.76 | ± | 0.32 bA | 21.63 | ± | 1.20 bA | |
| T20 | 20 | 8.71 | ± | 0.38 aA | 7.36 | ± | 0.51 aA | 0.52 | ± | 0.03 aA | 28.48 | ± | 1.50 cA | 9.54 | ± | 2.27 cA | 13.64 | ± | 3.19 cA | |
| Significance | ** | *** | *** | *** | *** | *** | ||||||||||||||
| Biscuit Sample | Sugar | CBS Percentage | Weight Loss | Width | Thickness | Spread | Hardness | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (%) | (cm) | (cm) | (N) | ||||||||||||||
| S0 | Sucrose | 0 | 15.84 | ± | 1.19 aA | 6.51 | ± | 0.26 aA | 0.86 | ± | 0.05 aB | 75.85 | ± | 6.73 aA | 68.83 | ± | 11.95 aA |
| S10 | 10 | 16.08 | ± | 0.48 aA | 5.93 | ± | 0.10 bA | 0.91 | ± | 0.08 aA | 65.28 | ± | 6.53 bA | 53.92 | ± | 6.46 bB | |
| S20 | 20 | 16.44 | ± | 0.48 aA | 5.81 | ± | 0.12 bA | 0.90 | ± | 0.01 aA | 64.13 | ± | 1.67 bA | 54.75 | ± | 12.21 bB | |
| Significance | ns | *** | ns | *** | ** | ||||||||||||
| T0 | Tagatose | 0 | 15.33 | ± | 1.14 aA | 6.12 | ± | 0.08 aB | 0.92 | ± | 0.07 aA | 66.52 | ± | 4.07 aB | 72.77 | ± | 29.72 bA |
| T10 | 10 | 14.25 | ± | 0.46 aB | 5.88 | ± | 0.07 bA | 0.89 | ± | 0.10 aA | 66.47 | ± | 7.61 aA | 88.96 | ± | 19.39 abA | |
| T20 | 20 | 14.69 | ± | 0.75 aA | 5.81 | ± | 0.12 bA | 0.90 | ± | 0.01 aA | 64.13 | ± | 1.67 aA | 120.34 | ± | 51.14 aA | |
| Significance | ns | *** | ns | ns | * | ||||||||||||
| Biscuit Sample | Sugar | CBS Percentage | Appearance | Odor | Taste | Flavor | Texture | Overall Liking | Purchase Predisposition |
|---|---|---|---|---|---|---|---|---|---|
| S0 | Sucrose | 0 | 2602.0 a | 2890.0 a | 2843.5 a | 2877.5 a | 3057.0 a | 2961.5 a | 2992.5 a |
| S10 | 10 | 2652.0 a | 2394.5 ab | 2737.5 a | 2716.5 a | 2754.0 a | 2773.5 a | 2823.0 a | |
| S20 | 20 | 1692.5 ab | 2175.0 abc | 2451.5 a | 2451.5 a | 2164.0 ab | 2339.0 a | 2365.0 a | |
| T0 | Tagatose | 0 | 1526.0 b | 994.0 d | 986.5 b | 1051.5 b | 1567.0 bc | 1146.5 b | 1009.0 b |
| T10 | 10 | 1781.0 ab | 1502.0 bcd | 1226.0 b | 1225.5 b | 1078.5 c | 1149.5 b | 1199.5 b | |
| T20 | 20 | 921.5 b | 1369.5 cd | 1080.0 b | 1002.5 b | 704.5 c | 955.0 b | 936.0 b | |
| Significance | *** | *** | *** | *** | *** | *** | *** | ||
| Friability | Hardness | Softness | Crunchy | Astringent | Sweet |
|---|---|---|---|---|---|
| Shortbread | Hard on the first try | Damp | Honey | ||
| Dry | Tender | Not sweet | |||
| Granulose | Undercooked | Jam | |||
| Gummy | |||||
| Soluble | |||||
| Sticky | |||||
| Bitter | Salty | Umami | Sour | Butter | Toasted |
| Stale | Stock cube | Fermentation | Hazelnut | ||
| Chocolate | Caramel | Burned | Cooked corn | Milk | Vegetal |
| Unsweetened cocoa powder | Amaretto biscuit | Straw | Condensed milk | Wet grass | |
| Chocolate milk | Puffed rice | ||||
| Sweet cocoa |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rojo-Poveda, O.; Barbosa-Pereira, L.; Orden, D.; Stévigny, C.; Zeppa, G.; Bertolino, M. Physical Properties and Consumer Evaluation of Cocoa Bean Shell-Functionalized Biscuits Adapted for Diabetic Consumers by the Replacement of Sucrose with Tagatose. Foods 2020, 9, 814. https://doi.org/10.3390/foods9060814
Rojo-Poveda O, Barbosa-Pereira L, Orden D, Stévigny C, Zeppa G, Bertolino M. Physical Properties and Consumer Evaluation of Cocoa Bean Shell-Functionalized Biscuits Adapted for Diabetic Consumers by the Replacement of Sucrose with Tagatose. Foods. 2020; 9(6):814. https://doi.org/10.3390/foods9060814
Chicago/Turabian StyleRojo-Poveda, Olga, Letricia Barbosa-Pereira, David Orden, Caroline Stévigny, Giuseppe Zeppa, and Marta Bertolino. 2020. "Physical Properties and Consumer Evaluation of Cocoa Bean Shell-Functionalized Biscuits Adapted for Diabetic Consumers by the Replacement of Sucrose with Tagatose" Foods 9, no. 6: 814. https://doi.org/10.3390/foods9060814
APA StyleRojo-Poveda, O., Barbosa-Pereira, L., Orden, D., Stévigny, C., Zeppa, G., & Bertolino, M. (2020). Physical Properties and Consumer Evaluation of Cocoa Bean Shell-Functionalized Biscuits Adapted for Diabetic Consumers by the Replacement of Sucrose with Tagatose. Foods, 9(6), 814. https://doi.org/10.3390/foods9060814








