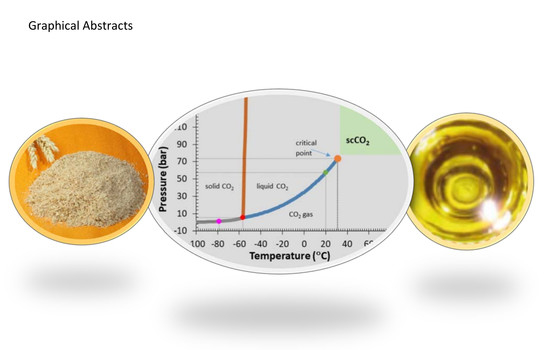
Bioactive Compounds of a Wheat Bran Oily Extract Obtained with Supercritical Carbon Dioxide †
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material
2.2. Supercritical Fluid Extraction (SFE) Equipment and Procedure
2.3. Chemicals
2.4. Analytical Methods
2.4.1. Methods for Quantification of Different Bioactive Compounds
Fatty Acids Content and Profile
Alkylresorcinols Content and Profile
Steryl Ferulates Content and Profile
Tocopherols Content and Profile
Phenolic Compounds Content and Profile
2.4.2. Determination of Some Usual Oil Quality Parameters
Acidity Value (AV)
Peroxide Value (PV)
Hexanal Content
2.4.3. Total Antioxidant Capacity of the Wheat Bran Oily Extract
FRAP Assay
ABTS Assay
DPPH Assay
2.4.4. Evolution of Wheat Bran Oily Extract Composition during Storage
2.5. Statistical Analysis
3. Results and Discussion
3.1. Bioactive Compounds Evaluated in the Obtained Supercritical Wheat Bran Oily Extracts
3.2. Common Quality Parameters Evaluated in the Obtained Supercritical Wheat Bran Oily Extracts
3.3. Evaluation of Antioxidant Capacity of the Obtained Supercritical Wheat Bran Oily Extracts
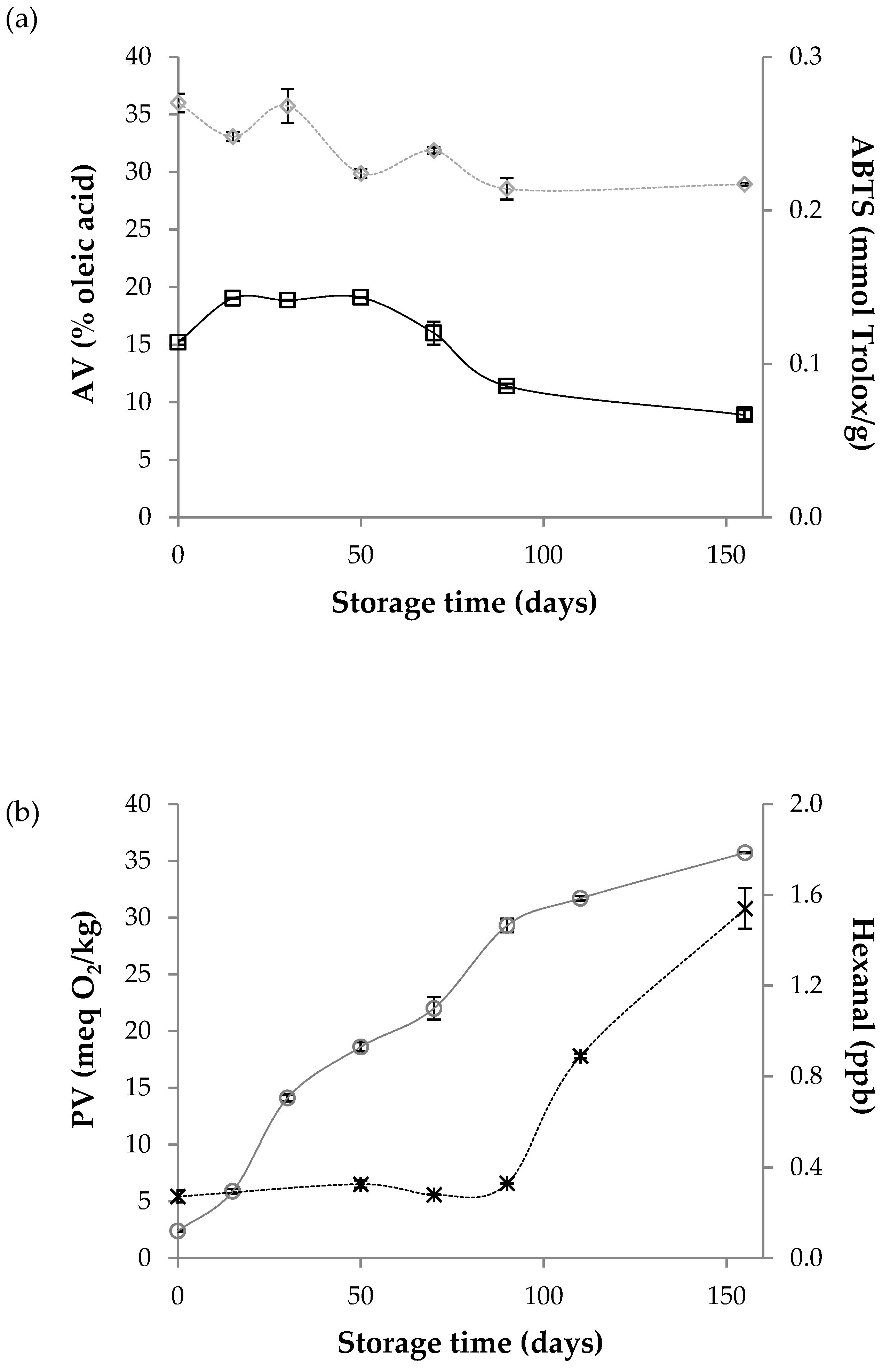
3.4. Evolution of the Obtained Supercritical Wheat Bran Oily Extracts during Storage
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bramley, P.M.; Elmadfa, I.; Kafatos, A.; Kelly, F.J.; Manios, Y.; Roxborough, H.E.; Schuch, W.; Sheehy, P.J.A.; Wagner, K. Vitamin E. J. Sci. Food Agric. 2000, 80, 913–938. [Google Scholar] [CrossRef]
- Rebolleda, S.; Sanz, M.T.; Benito, J.M.; Beltrán, S.; Escudero, I.; González San-José, M.L. Formulation and characterisation of wheat bran oil-in-water nanoemulsions. Food Chem. 2015, 167, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Fardet, A. New hypotheses for the health-protective mechanisms of whole-grain cereals: What is beyond fibre? Nutr. Res. Rev. 2010, 23, 65–134. [Google Scholar] [CrossRef] [PubMed]
- Rebolleda, S.; Beltrán, S.; Sanz, M.T.; González-Sanjosé, M.L.; Solaesa, Á.G. Extraction of alkylresorcinols from wheat bran with supercritical CO2. J. Food Eng. 2013, 119, 814–821. [Google Scholar] [CrossRef]
- Rebolleda, S.; Beltrán, S.; Sanz, M.T.; González-SanJosé, M.L. Supercritical fluid extraction of wheat bran oil: Study of extraction yield and oil quality. Eur. J. Lipid Sci. Technol. 2014, 116, 319–327. [Google Scholar] [CrossRef]
- Zhou, K.; Yu, L. Effects of extraction solvent on wheat bran antioxidant activity estimation. LWT-Food Sci. Technol. 2004, 37, 717–721. [Google Scholar] [CrossRef]
- Dey, E.S.; Mikhailopulo, K. A food grade approach for the isolation of major alkylresorcinols (ARs) from rye bran applying tailored supercritical carbon dioxide (scCO2) extraction combined with HPLC. J. Supercrit. Fluids 2009, 51, 167–173. [Google Scholar] [CrossRef]
- Parikka, K.; Rowland, I.R.; Welch, R.W.; Wähälä, K. In vitro antioxidant activity and antigenotoxicity of 5-n-alkylresorcinols. J. Agric. Food Chem. 2006, 54, 1646–1650. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Shi, L.; Hu, C.; Sang, S. Wheat Bran for Colon Cancer Prevention: The Synergy between Phytochemical Alkylresorcinol C21 and Intestinal Microbial Metabolite Butyrate. J. Agric. Food Chem. 2019, 67, 12761–12769. [Google Scholar] [CrossRef] [PubMed]
- Kruk, J.; Aboul-Enein, B.; Bernstein, J.; Marchlewicz, M. Dietary alkylresorcinols and cancer prevention: A systematic review. Eur. Food Res. Technol. 2017, 243, 1693–1710. [Google Scholar] [CrossRef]
- Tsuzuki, W.; Mogushi, H.; Kawahara, S.; Kotake-Nara, E.; Komba, S.; Kanai, Y.; Yamada, S.; Horigane, A. The content and distribution of steryl ferulates in wheat produced in Japan. Biosci. Biotechnol. Biochem. 2017, 81, 573–580. [Google Scholar] [CrossRef]
- Nyström, L.; Makinen, M.; Lampi, A.M.; Piironen, V. Antioxidant Activity of Steryl Ferulate Extracts from Rye and Wheat Bran. J. Agric. Food Chem. 2005, 53, 2503–2510. [Google Scholar] [CrossRef] [PubMed]
- Luthria, D.L.; Lu, Y.; John, K.M.M. Bioactive phytochemicals in wheat: Extraction, analysis, processing, and functional properties. J. Funct. Foods 2015, 18, 910–925. [Google Scholar] [CrossRef]
- Călinoiu, L.F.; Vodnar, D.C. Whole grains and phenolic acids: A review on bioactivity, functionality, health benefits and bioavailability. Nutrients 2018, 10, 1615. [Google Scholar] [CrossRef]
- Lack, E.; Simándi, B. Supercritical fluid extraction and fractionation from solid materials. In High Pressure Process Technology: Fundamentals and Applications; Bertucco, A., Vetter, G., Eds.; Elsevier: Amsterdam, The Netherlands, 2001; pp. 537–575. [Google Scholar]
- Vaquero, E.M.; Beltrán, S.; Sanz, M.T. Extraction of fat from pigskin with supercritical carbon dioxide. J. Supercrit. Fluids 2006, 37, 142–150. [Google Scholar] [CrossRef]
- AOCS-AOAC. AOAC Official Method 991.39. Fatty Acids in Encapsulated Fish Oils and Fish Oil Methyl and Ethyl Esters; AOAC International: Rockville, MD, USA, 2000. [Google Scholar]
- Hakala, P.; Lampi, A.M.; Ollilaiene, V.; Werner, U.; Murkovik, M.; Wahala, K.; Karkola, S.; Piironen, V. Steryl Phenolic Acid Esters in Cereals and Their Milling. J. Agric. Food Chem. 2002, 50, 5300–5307. [Google Scholar] [CrossRef]
- Rebolleda, S.; Rubio, N.; Beltrán, S.; Sanz, M.T.; González-Sanjosé, M.L. Supercritical fluid extraction of corn germ oil: Study of the influence of process parameters on the extraction yield and oil quality. J. Supercrit. Fluids 2012, 72, 270–277. [Google Scholar] [CrossRef]
- Pérez-Magariño, S.; Ortega-Heras, M.; Cano-Mozo, E.; González-Sanjosé, M.L. The influence of oak wood chips, micro-oxygenation treatment, and grape variety on colour, and anthocyanin and phenolic composition of red wines. J. Food Compos. Anal. 2009, 22, 204–211. [Google Scholar] [CrossRef]
- AOCS. Official Methods and Recommendation Practices of the American Oil Chemists’ Society; AOCS Press: Champaign, IL, USA, 1990. [Google Scholar]
- Rivero-Pérez, M.D.; Muñiz, P.; González-SanJosé, M.L. Antioxidant Profile of Red Wines Evaluated by Total Antioxidant Capacity, Scavenger Activity, and Biomarkers of Oxidative Stress Methodologies. J. Agric. Food Chem. 2007, 55, 5476–5483. [Google Scholar] [CrossRef]
- Vegetable Oils in Food Technology: Composition, Properties and Uses; Gunstone, F.D., Ed.; Blackwell Publishing: Oxford, UK, 2002; ISBN 1841273317. [Google Scholar]
- Kulawinek, M.; Jaromin, A.; Kozubek, A.; Zarnowski, R. Alkylresorcinols in Selected Polish Rye and Wheat Cereals and Whole-Grain Cereal Products. J. Agric. Food Chem. 2008, 56, 7236–7242. [Google Scholar] [CrossRef]
- Athukorala, Y.; Hosseinian, F.S.; Mazza, G. Extraction and fractionation of alkylresorcinols from triticale bran by two-step supercritical carbon dioxide. LWT Food Sci. Technol. 2010, 43, 660–665. [Google Scholar] [CrossRef]
- Moreau, R.A.; Fang, X. Analysis of alkylresorcinols in wheat germ oil and barley germ oil via HPLC and fluorescence detection: Cochromatography with tocols. Cereal Chem. 2016, 93, 293–298. [Google Scholar] [CrossRef]
- Nurmi, T.; Lampi, A.M.; Nyström, L.; Hemery, Y.; Rouau, X.; Piironen, V. Distribution and composition of phytosterols and steryl ferulates in wheat grain and bran fractions. J. Cereal Sci. 2012, 56, 379–388. [Google Scholar] [CrossRef]
- Kumar, G.S.; Krishna, A.G.G. Studies on the nutraceuticals composition of wheat derived oils wheat bran oil and wheat germ oil. J. Food Sci. Technol. 2015, 52, 1145–1151. [Google Scholar] [CrossRef]
- Benito-Román, O.; Varona, S.; Sanz, M.T.; Beltrán, S. Valorization of rice bran: Modified supercritical CO2 extraction of bioactive compounds. J. Ind. Eng. Chem. 2019, 80, 273–282. [Google Scholar] [CrossRef]
- Lerma-García, M.J.; Herrero-Martínez, J.M.; Simó-Alfonso, E.F.; Mendonça, C.R.B.; Ramis-Ramos, G. Composition, industrial processing and applications of rice bran γ-oryzanol. Food Chem. 2009, 115, 389–404. [Google Scholar] [CrossRef]
- Durante, M.; Lenucci, M.S.; Rescio, L.; Mita, G.; Caretto, S. Durum wheat by-products as natural sources of valuable nutrients. Phytochem. Rev. 2012, 11, 255–262. [Google Scholar] [CrossRef]
- Okarter, N.; Liu, C.; Sorrells, M.E.; Hai, R. Phytochemical content and antioxidant activity of six diverse varieties of whole wheat. Food Chem. 2010, 119, 249–257. [Google Scholar] [CrossRef]
- Lampi, A.M.; Kamal-Eldin, A.; Piironen, V. Tocopherols and tocotrienols from oil and cereal grains. In Functional Foods: Biochemical and Processing Aspects; Shi, J., Mazza, G., Le Maguer, M., Eds.; CRC Press: Boca Raton, FL, USA, 2002; Volume 2. [Google Scholar]
- Seppanen, C.M.; Song, Q.; Csallany, A.S. The Antioxidant Functions of Tocopherol and Tocotrienol Homologues in Oils, Fats, and Food Systems. J. Am. Oil Chem. Soc. 2010, 87, 469–481. [Google Scholar] [CrossRef]
- Beltrán, S.; Sanz, M.T.; Santamaría, B.; Murga, R.; Salazar, G. Recovery of antioxidants from grape products by using supercritical fluids and membrane technology. Electron. J. Environ. Agric. Food Chem. 2008, 7, 3270–3278. [Google Scholar]
- Mattila, P.H.; Pihlava, J.M.; Hellström, J.K. Contents of Phenolic Acids, Alkyl- and Alkenylresorcinols, and Avenanthramides in Commercial Grain Products. J. Agric. Food Chem. 2005, 53, 8290–8295. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Rodríguez, N.; Beltrán, S.; Jaime, I.; de Diego, S.M.; Sanz, M.T.; Carballido, J.R. Production of omega-3 polyunsaturated fatty acid concentrates: A review. Innov. Food Sci. Emerg. Technol. 2010, 11, 1–12. [Google Scholar] [CrossRef]
- Khattab, R.Y.; Zeitoun, M.A. LWT—Food Science and Technology Quality evaluation of fl axseed oil obtained by different extraction techniques. LWT Food Sci. Technol. 2013, 53, 338–345. [Google Scholar] [CrossRef]
- Jung, G.; Kang, H.; Chun, B. Characterization of wheat bran oil obtained by supercritical carbon dioxide and hexane extraction. J. Ind. Eng. Chem. 2012, 18, 360–363. [Google Scholar] [CrossRef]
- Matthäus, B.; Guillaume, D.; Gharby, S.; Haddad, A.; Harhar, H.; Charrouf, Z. Effect of processing on the quality of edible argan oil. Food Chem. 2010, 120, 426–432. [Google Scholar] [CrossRef]
- Leffingwell & Associates. Flavor-Base, 10th ed.; Leffingwell & Associates: Canton, GA, USA, 2016. [Google Scholar]
- Saura-Calixto, F.; Goñi, I. Food Chemistry Antioxidant capacity of the Spanish Mediterranean diet. Food Chem. 2006, 94, 442–447. [Google Scholar] [CrossRef]
- Fadda, C.; Del Caro, A.; Sanguinetti, A.M.; Urgeghe, P.P.; Vacca, V.; Arca, P.P.; Piga, A. Changes during storage of quality parameters and in vitro antioxidant activity of extra virgin monovarietal oils obtained with two extraction technologies. Food Chem. 2012, 134, 1542–1548. [Google Scholar] [CrossRef]
- Tawaha, K.; Alali, F.Q.; Gharaibeh, M.; Mohammad, M.; El-Elimat, T. Food Chemistry Antioxidant activity and total phenolic content of selected Jordanian plant species. Food Chem. 2007, 104, 1372–1378. [Google Scholar] [CrossRef]
 Total AR,
Total AR,  C15-AR,
C15-AR,  C17-AR,
C17-AR,  C19-AR,
C19-AR,  C21-AR,
C21-AR,  C23-AR,
C23-AR,  C25-AR.
C25-AR.
 Total AR,
Total AR,  C15-AR,
C15-AR,  C17-AR,
C17-AR,  C19-AR,
C19-AR,  C21-AR,
C21-AR,  C23-AR,
C23-AR,  C25-AR.
C25-AR.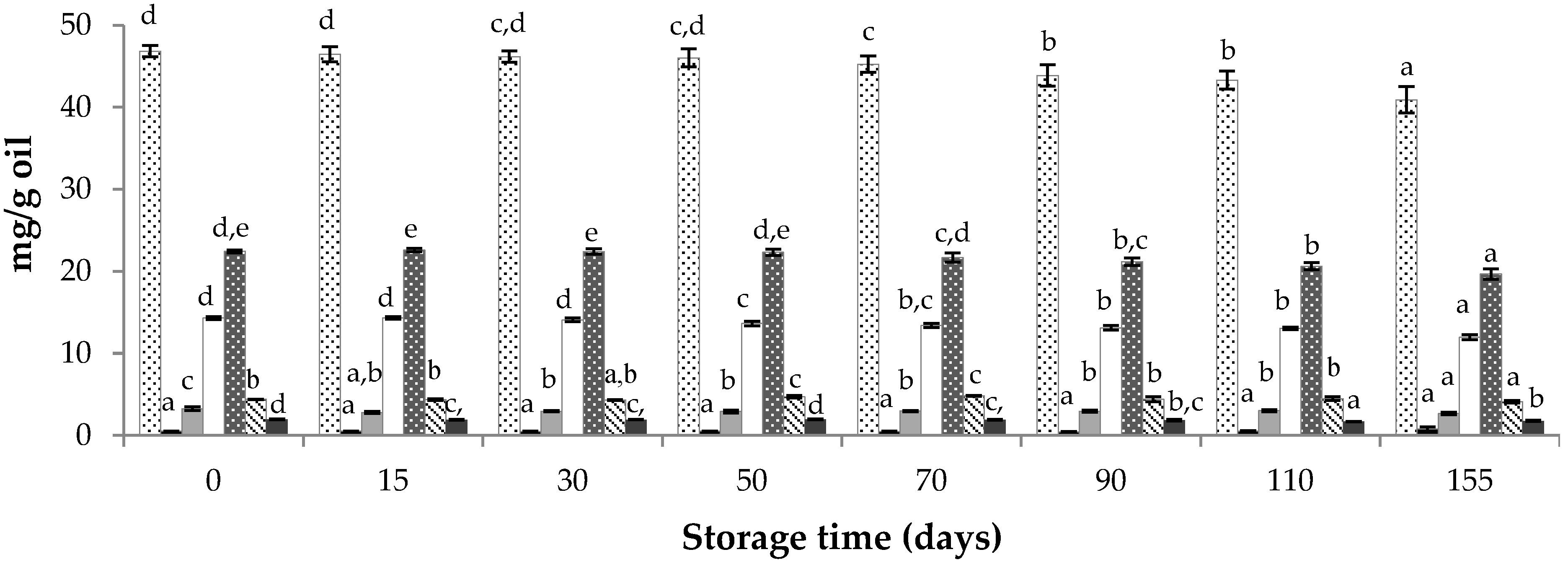
 Total tocopherol,
Total tocopherol,  α-tocopherol,
α-tocopherol,  β-tocopherol,
β-tocopherol,  γ-tocopherol,
γ-tocopherol,  δ-tocopherol.
δ-tocopherol.
 Total tocopherol,
Total tocopherol,  α-tocopherol,
α-tocopherol,  β-tocopherol,
β-tocopherol,  γ-tocopherol,
γ-tocopherol,  δ-tocopherol.
δ-tocopherol.
| Palmitic acid (C16:0) | 118 ± 2 | |
| Stearic acid (C18:0) | 7.9 ± 0.1 | |
| Oleic acid (C18:1) | 114 ± 3 | |
| Linoleic acid (C18:2) | 410 ± 10 | |
| α- linolenic acid (C18:3) | 37.3 ± 0.8 | |
| Other fatty acids | 25 ± 4 | |
| Total fatty acids (mg/g SCWBOE) | 712 ± 20 | |
| C15- alkylresorcinol (R1=C15H31) | 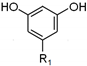 | 0.52 ± 0.01 |
| C17- alkylresorcinol (R1=C17H35) | 3.3 ± 0.2 | |
| C19- alkylresorcinol (R1=C19H39) | 14.3 ± 0.3 | |
| C21- alkylresorcinol (R1=C21H43) | 22.4 ± 0.2 | |
| C23- alkylresorcinol (R1=C23H47) | 4.40 ± 0.03 | |
| C25- alkylresorcinol (R1=C25H51) | 1.97 ± 0.06 | |
| Total alkylresorcinols (mg/g SCWBOE) | 46.9 ± 0.8 | |
| Campesteryl ferulate (R1=H) | 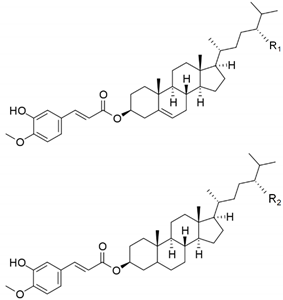 | 2.4 ± 0.1 |
| Sitosteryl ferulate (R1=CH3) + Campestanyl ferulate (R2=H) | 9.9 ± 0.5 | |
| Sitostanyl ferulate (R2=CH3) | 5.9 ± 0.4 | |
| Total steryl ferulates (mg/g SCWBOE) | 18 ± 1 | |
| α- Tocopherol (R1=R2=R3=CH3) |  | 3.84 ± 0.01 |
| β- Tocopherol (R1= R3=CH3; R2=H) | 0.20 ± 0.01 | |
| γ- Tocopherol (R1=H; R2=R3=CH3) | 2.67 ± 0.08 | |
| δ- Tocopherol(R1= R2=H, R3=CH3) | 0.09 ± 0.01 | |
| Total tocopherols (mg/g SCWBOE) | 6.8 ± 0.1 | |
| Vanillin (R1=H; R2=CHO; R3=OCH3) |  | 13.8 ± 0.1 |
| Vanillic acid (R1=H; R2=COOH; R3=OCH3) | 3.5 ± 0.5 | |
| Syringic aldehyde (R1=R3=OCH3; R2=CHO) | 3.4 ± 0.8 | |
| Ferulic acid (R1=H; R2=(CH)2COOH; R3=OCH3) | 1.8 ± 0.3 | |
| Syringic acid (R1=R3=OCH3; R2=COOH) | 1.6 ± 0.4 | |
| p-Hydroxybenzaldehyde (R1=R3=H; R2=CHO) | 0.7 ± 0.1 | |
| Phenolic compounds (mg/kg SCWBOE) | 25 ± 2 |
| Antioxidant Method | Antioxidant Mechanism | Units | Value |
|---|---|---|---|
| DPPH | SET | µmol Trolox/g SCWBOE | 26 ± 2 |
| FRAP | SET | µmol Fe (II)/g SCWBOE | 228 ± 12 |
| ABTS | SET/HAT | µmol Trolox/g SCWBOE | 270 ± 6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rebolleda, S.; González-San José, M.L.; Sanz, M.T.; Beltrán, S.; Solaesa, Á.G. Bioactive Compounds of a Wheat Bran Oily Extract Obtained with Supercritical Carbon Dioxide. Foods 2020, 9, 625. https://doi.org/10.3390/foods9050625
Rebolleda S, González-San José ML, Sanz MT, Beltrán S, Solaesa ÁG. Bioactive Compounds of a Wheat Bran Oily Extract Obtained with Supercritical Carbon Dioxide. Foods. 2020; 9(5):625. https://doi.org/10.3390/foods9050625
Chicago/Turabian StyleRebolleda, Sara, María Luisa González-San José, María Teresa Sanz, Sagrario Beltrán, and Ángela G. Solaesa. 2020. "Bioactive Compounds of a Wheat Bran Oily Extract Obtained with Supercritical Carbon Dioxide" Foods 9, no. 5: 625. https://doi.org/10.3390/foods9050625
APA StyleRebolleda, S., González-San José, M. L., Sanz, M. T., Beltrán, S., & Solaesa, Á. G. (2020). Bioactive Compounds of a Wheat Bran Oily Extract Obtained with Supercritical Carbon Dioxide. Foods, 9(5), 625. https://doi.org/10.3390/foods9050625