Antioxidant and Antimicrobial Properties of Semi-Processed Frozen Prickly Pear Juice as Affected by Cultivar and Harvest Time
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Physical and Chemical Analyses
2.3. Bioactive Compounds and Antioxidant Activity
2.3.1. Total Polyphenols and Flavonoids
2.3.2. Total Betalains
2.3.3. DPPH Assay
2.4. Color Analysis
2.5. In Vitro Evaluation of Prickly Pear Juice Antimicrobial Activity
2.6. Statistical Analysis
3. Results and Discussion
3.1. Evaluation of Physical and Chemical Properties
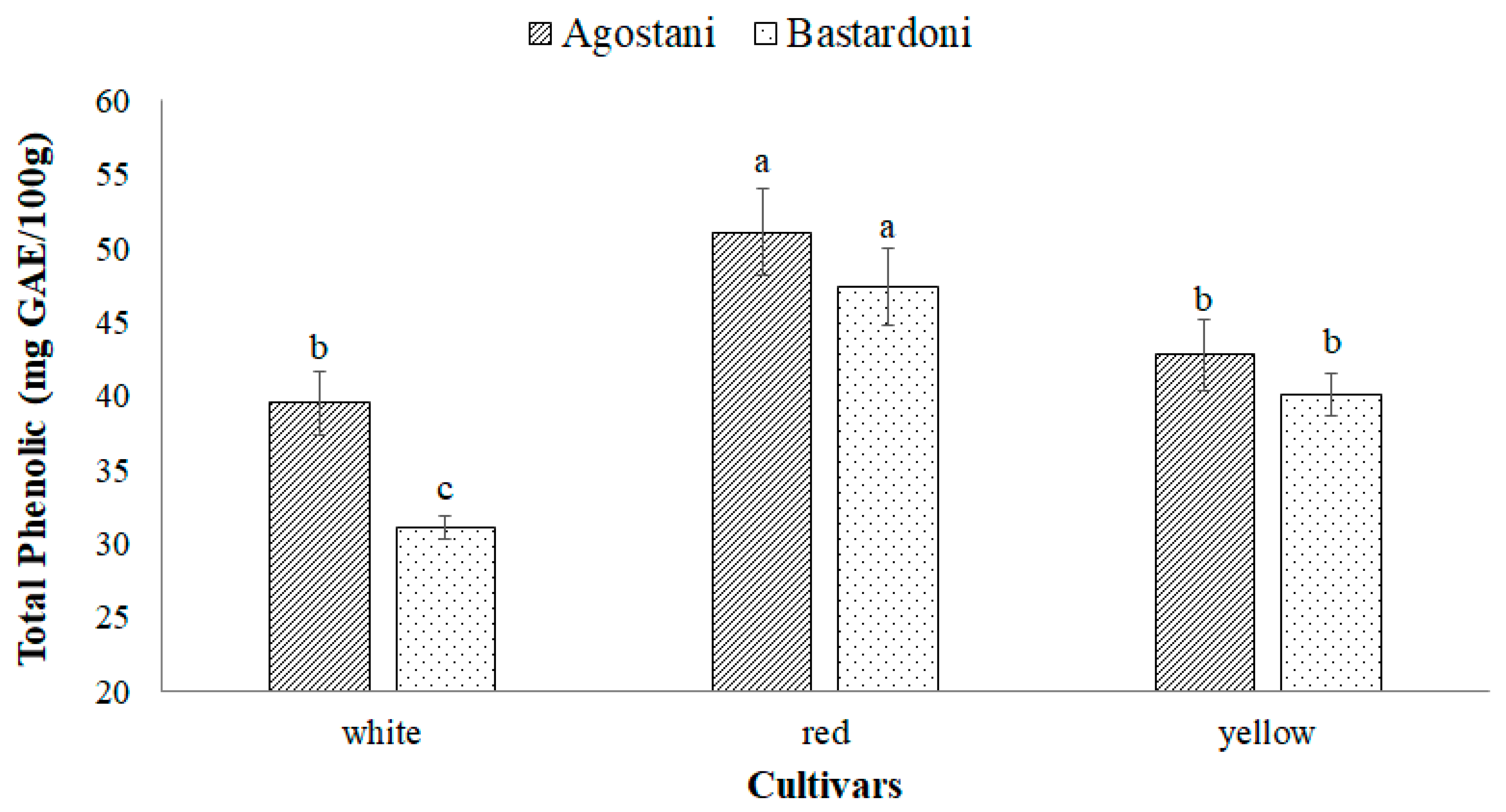
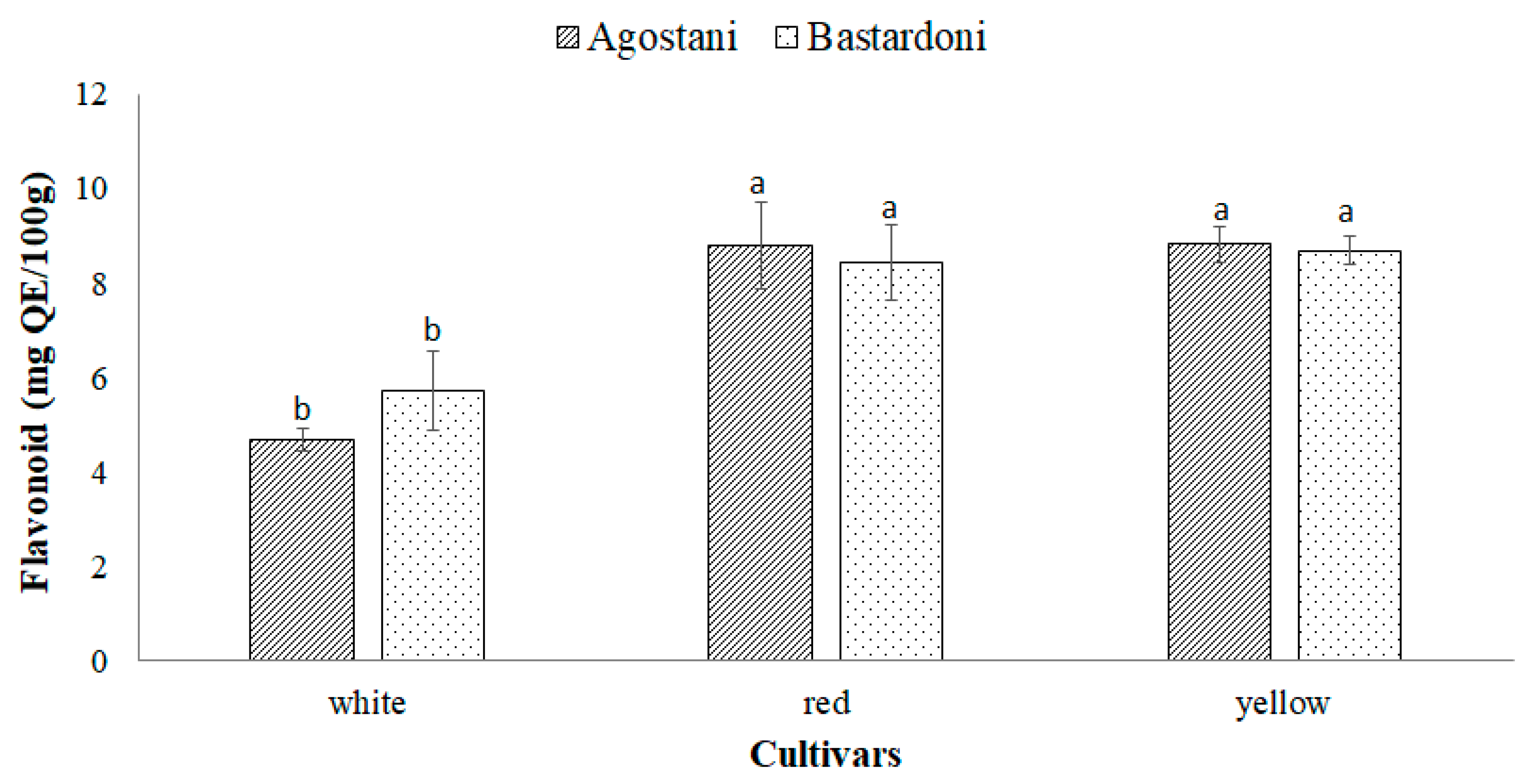
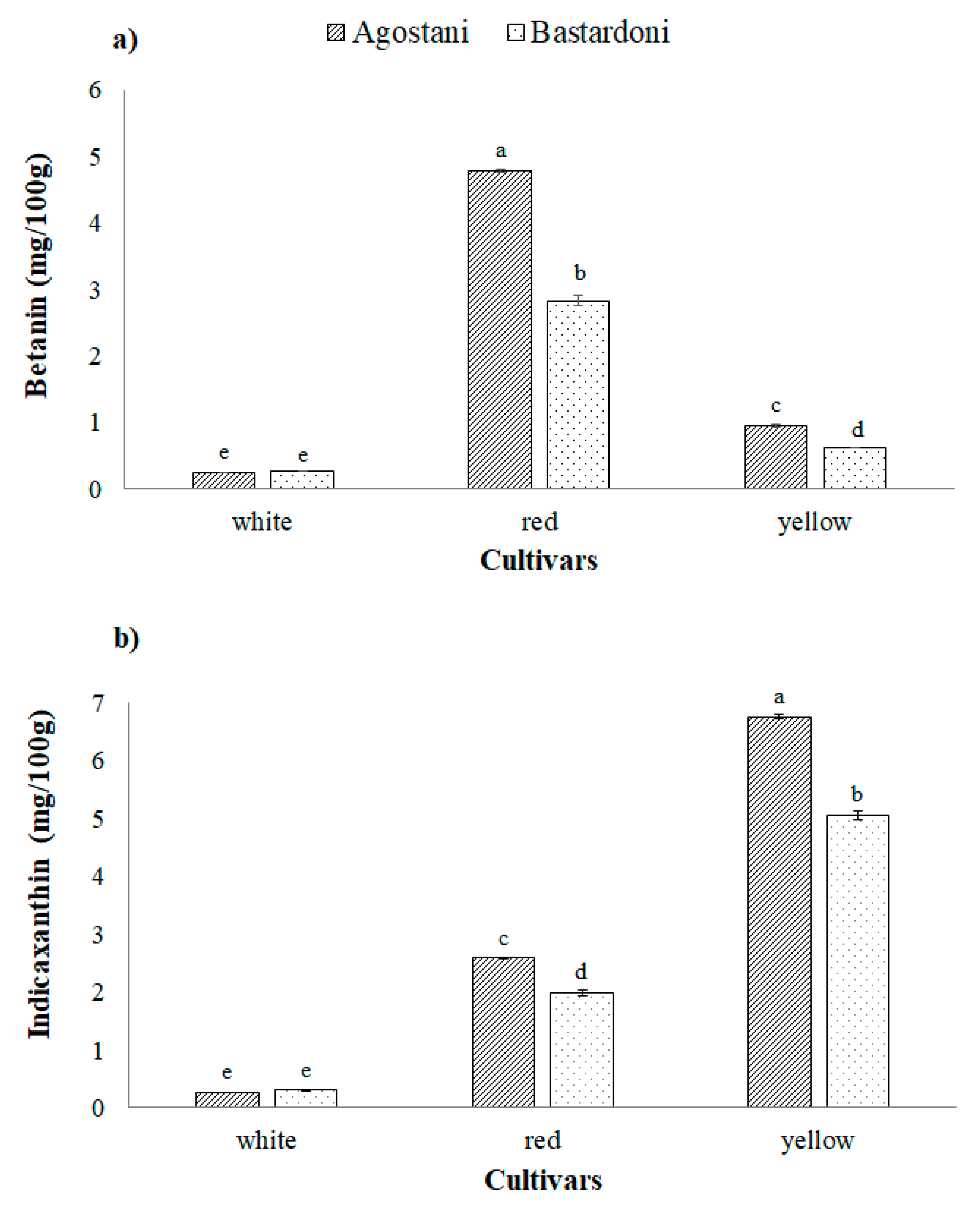
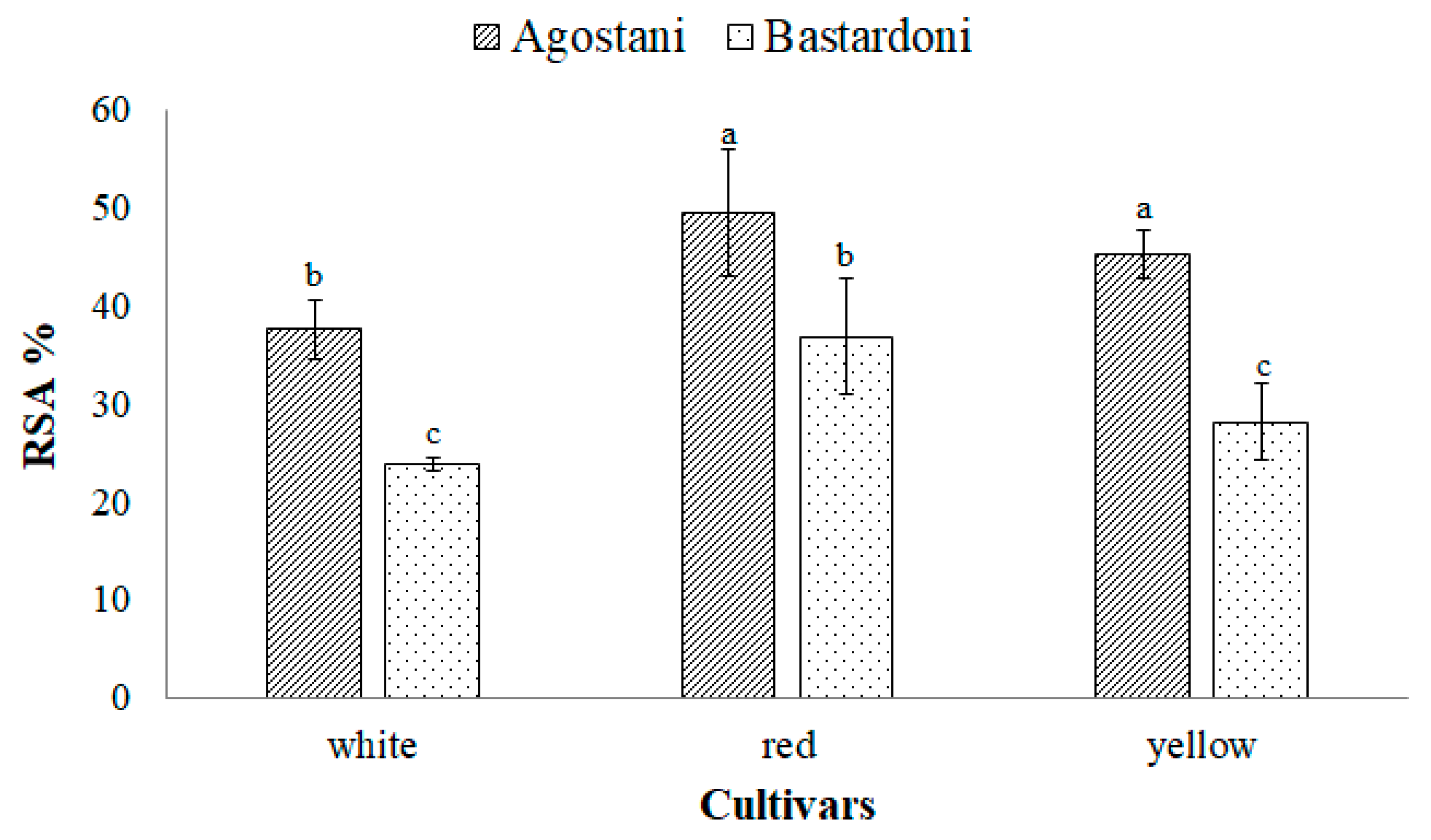
3.2. Bioactive Compounds
3.3. Color Evaluation
3.4. Antimicrobial Activity of Ppjs Against Target Bacteria
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sawaya, W.N.; Khatchadourian, H.A.; Safi, W.M.; Al-Hammad, H.M. Chemical characterization of prickly pear pulp, Opuntia ficus-indica, and the manufacturing of prickly pear jam. J. Food Tech. 1983, 18, 183–193. [Google Scholar] [CrossRef]
- Gouws, C.A.; Georgousopoulou, E.N.; Mellor, D.D.; McKune, A.; Naumovski, N. Effects of the consumption of prickly pear cacti (Opuntia spp.) and its products on blood glucose levels and insulin: A systematic review. Medicina 2019, 55, 138. [Google Scholar] [CrossRef]
- Fernández-López, J.A.; Almela, L.; Obón, J.M.; Castellar, R. Determination of antioxidant constituents in cactus pear fruits. Plant. Foods Hum. Nutr. 2010, 65, 253–259. [Google Scholar] [CrossRef]
- Madrigal-Santillán, E.; García-Melo, F.; Morales-González, J.A.; Vázquez-Alvarado, P.; Muñoz-Juárez, S.; Zuñiga-Pérez, C.; Sumaya-Martínez, M.T.; Madrigal-Bujaidar, E.; Hernández-Ceruelos, A. Antioxidant and anticlastogenic capacity of prickly pear juice. Nutrients 2013, 5, 4145–4158. [Google Scholar] [CrossRef]
- Mena, P.; Tassotti, M.; Andreu, L.; Nuncio-Jáuregui, N.; Legua, P.; Del Rio, D.; Hernández, F. Phytochemical characterization of different prickly pear (Opuntia ficus-indica (L.) Mill.) cultivars and botanical parts: UHPLC-ESI-MSn metabolomics profiles and their chemometric analysis. Food Res. Int. 2018, 108, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hameed, E.S.; Nagaty, M.A.; Salman, M.S.; Bazaid, S.A. Phytochemicals, nutritionals and antioxidant properties of two prickly pear cactus cultivars (Opuntia ficus indica Mill.) growing in Taif, KSA. Food Chem. 2014, 160, 31–38. [Google Scholar] [CrossRef]
- Cova, A.M.; Crascì, L.; Panico, A.; Catalfo, A.; De Guidi, G. Antioxidant capability and phytochemicals content of Sicilian prickly fruits. Int. J. Food Sci. Nutr. 2015, 66, 881–886. [Google Scholar] [CrossRef]
- Ali, H.S.M.; Al-Khalifa, A.S.; Brückner, H. Taurine is absent from amino components in fruits of Opuntia ficus-indica. SpringerPlus 2014, 3, 663. [Google Scholar] [CrossRef] [PubMed]
- Rocchetti, G.; Pellizzoni, M.; Montesano, D.; Lucini, L. Italian Opuntia ficus-indica cladodes as rich source of bioactive compounds with health-promoting properties. Foods 2018, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, P.; Abedimanesh, S.; Mesbah-Namin, S.A.; Ostadrahimi, A. Betalains, the nature-inspired pigments, in health and diseases. Crit. Rev. Food Sci. Nutr. 2018, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Livrea, M.A.; Tesoriere, L. Antioxidative effects of cactus pear [Opuntia ficus-indica (L.) Mill.] fruits from Sicily and bioavailability of betalain components in healthy humans. Acta Horticult. 2009, 811, 197–204. [Google Scholar] [CrossRef]
- Castellar, R.; Obón, J.M.; Alacid, M.; Fernández-López, J.A. Color properties and stability of betacyanins from Opuntia fruits. J. Agric. Food Chem. 2003, 51, 2772–2776. [Google Scholar] [CrossRef]
- Triebold, H.O. Quantitative Analysis with Applications to Agricultural and Food Products, 2nd ed.; D. van Nostrand Company, Inc.: New York, NY, USA, 2000; pp. 204–221. [Google Scholar]
- AOAC. Official Methods of Analysis, 16th ed.; AOAC International: Washington, DC, USA, 1998. [Google Scholar]
- Vazquez-Roncero, A.; Janer del Valle, C.; Janer del Valle, M.L. Determinación de los polifenoles totales del aceite de oliva. Grasas Aceites 1973, 24, 350–355. [Google Scholar]
- Lin, J.Y.; Tang, C.Y. Determination of total phenolic and flavonoid contents in selected fruits and vegetables, as well as their stimulatory effect on mouse splenocyte proliferation. Food Chem. 2007, 101, 140–147. [Google Scholar] [CrossRef]
- Cai, Y.; Sun, M.; Corke, H. Colourant properties and stability of Amaranthus betacyanin pigments. J. Agric. Food Chem. 1998, 46, 4491–4495. [Google Scholar] [CrossRef]
- Pulipati, S.; Srinivasa babu, P.; Lakshmi, N.M. Quantitative Determination of Tannin Content and Evaluation of Antibacterial Activity of Amaranthus tricolor (L). Int. J. Biol. Pharm. Res. 2014, 5, 623–626. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free-radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Sant’Anna, V.; Gurak, P.; Marczak, L.; Tessaro, I. Tracking bioactive compounds with colour changes in foods-A review. Dye. Pigment. 2013, 98, 601–608. [Google Scholar] [CrossRef]
- Gurrieri, S.; Miceli, L.; Lanza, C.M.; Tomaselli, F.; Bonomo, R.P.; Rizzarelli, E. Chemical characterization of sicilian prickly pear (Opuntia ficus indica) and perspectives for the storage of its juice. J. Agric. Food Chem. 2000, 48, 5424–5431. [Google Scholar] [CrossRef]
- Dehbi, F.; Hasib, A.; Ouatmane, A.; Elbatal, H.; Jaouad, A. Physicochemical Characteristics of Moroccan Prickly Pear Juice (Opuntia ficus indica L.). IJETAE 2014, 4, 300–306. [Google Scholar]
- El-Gharras, H.; Hasib, A.; Jaouad, A.; El-Bouadili, A. Chemical and physical characterization of three cultivars of moroccan yellow prickly pears (Opuntia ficus-indica) at three stages of maturity -Caracterización química y física de tres variedades de higos chumbos amarillas de marruecos (Opuntia ficus-indica) en tres etapas de madurez. CYTA J. Food 2006, 5, 93–99. [Google Scholar] [CrossRef]
- Díaz Medina, E.M.; Rodríguez Rodríguez, E.M.; Díaz Romero, C. Chemical characterization of Opuntia dillenii and Opuntia ficus-indica fruits. Food Chem. 2007, 103, 38–45. [Google Scholar] [CrossRef]
- Sáenz, C.; Sepúlveda, E. Cactus-pear juices. J. Prof. Assoc. Cactus Dev. 2001, 4, 3–10. [Google Scholar]
- Silos-Espino, H.; Fabian-Morales, L.; Osuna-Castro, J.A.; Valverde, E.; Guevara-Lara, F.; Paredes-López, O. Chemical and biochemical changes in prickly pears with different ripening behavior. Nahrung/Food 2003, 47, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Stintzing, F.C.; Herbach, K.M.; Mosshammer, M.R.; Carle, R.; Yi, W.; Sellappan, S.; Felker, P. Color, betalain pattern, and antioxidant properties of cactus pear (Opuntia spp.) clones. J. Agric. Food Chem. 2005, 53, 442–451. [Google Scholar] [CrossRef] [PubMed]
- Chavez Santoscoy, R.A.; Gutierrez Uribe, J.A.; Serna Saldivar, S.O. Phenolic composition, antioxidant capacity and in vitro cancer cell cytotoxicity of nine prickly pear (Opuntia spp.) juices. Plant. Foods Hum. Nutr. 2009, 64, 146–152. [Google Scholar] [CrossRef]
- Butera, D.; Tesoriere, L.; Di Gaudio, F.; Bongiorno, A.; Allegra, M.; Pintaudi, A.M.; Kohen, R.; Livrea, M.A. Antioxidant Activities of Sicilian Prickly Pear (Opuntia ficus indica) Fruit Extracts and Reducing Properties of Its Betalains: Betanin and Indicaxanthin. J. Agric. Food Chem. 2002, 50, 6895–6901. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Sun, M.; Corke, H. Antioxidant activity of betalins from plants of the Amaranthaceae. J. Agric. Food Chem. 2003, 51, 2288–2294. [Google Scholar] [CrossRef]
- Alimi, H.; Hfaeidh, N.; Bouoni, Z.; Sakly, M.; Rhouma, K.B. Ameliorative effect of Opuntia ficus indica juice on ethanol-induced oxidative stress in rat erythrocytes. Exp. Toxicol. Pathol. 2013, 65, 391–396. [Google Scholar] [CrossRef]
- Albano, C.; Negro, C.; Tommasi, N.; Gerardi, C.; Mita, G.; Miceli, A.; Blando, F. Betalains, phenols and antioxidant capacity in cactus pear [Opuntia ficus-indica (L.) Mill.] fruits from Apulia (South Italy) genotypes. Antioxidants 2015, 4, 269–280. [Google Scholar] [CrossRef]
- Cano, M.P.; Gómez-Maqueo, A.; García-Cayuela, T.; Welti-Chanes, J. Characterization of carotenoid profile of Spanish Sanguinos and Verdal prickly pear (Opuntia ficus-indica, spp.) tissues. Food Chem. 2017, 237, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Yahia, E.M.; Mondragon-Jacob, C. Nutritional components and antioxidant capacity of ten cultivars and lines of cactus pear fruit (Opuntia spp.). Food Res. Int. 2011, 44, 2311–2318. [Google Scholar] [CrossRef]
- Francis, F.J.; Clydesdale, F.M. Food Colorimetry: Theory and Applications; Avi Publishing Co Inc.: Westport, CT, USA, 1975. [Google Scholar] [CrossRef]
- Melgar, B.; Inês, M.; Ciric, A.; Sokovic, M.; García-Castello, E.M.; Rodríguez-López, A.D.; Ferreira, I. By-product recovery of Opuntia spp. peels: Betalainic and phenolic profiles and bioactive properties. Ind. Crop. Prod. 2017, 107, 353–359. [Google Scholar] [CrossRef]
- Ramírez-Moreno, E.; Cariño-Cortés, R.; Cruz-Cansino, N.S.; Delgado-Olivares, L.; Ariza-Ortega, J.A.; Montañez-Izquierdo, V.Y.; Hernández-Herrero, M.M.; Filardo-Kerstupp, T. Antioxidant and antimicrobial properties of cactus pear (Opuntia) seed oils. J. Food Qual. 2017, 1–8. [Google Scholar] [CrossRef]
- Blando, F.; Russo, R.; Negro, C.; De Bellis, L.; Frassinetti, S. Antimicrobial and antibiofilm activity against Staphylococcus aureus of Opuntia ficus-indica (L.) Mill. cladode polyphenolic extracts. Antioxidants 2019, 8, 117. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.F.; El-Mohamedy, R.S.R. Eco-friendly and protective natural dye from red prickly pear (Opuntia Lasiacantha Pfeiffer) plant. J. Saudi Chem. Soc. 2011, 15, 257–261. [Google Scholar] [CrossRef]
- Sanchez, E.; Garcia, S.; Heredia, N. Extracts of edible and medicinal plants damage membranes of Vibrio cholerae. Appl. Environ. Microbiol. 2010, 76, 6888–6894. [Google Scholar] [CrossRef] [PubMed]
- Rabhi, A.; Falleh, H.; Limam, F.; Ksouri, R.; Abdelly, C.; Raies, A. Upshot of the ripening time on biological activities, phenol content and fatty acid composition of Tunisian Opuntia ficus indica fruit. Afr. J. Biotechnol. 2013, 40, 5875–5885. [Google Scholar] [CrossRef]
- El-Mostafa, K.; El Kharrassi, Y.; Badreddine, A.; Andreoletti, P.; Vamecq, J.; El Kebbaj, M.S.; Latruffe, N.; Lizard, G.; Nasser, B.; Cherkaoui-Malki, M. Nopal cactus (Opuntia ficus indica) as a source of bioactive compounds for nutrition, health and disease. Molecules 2014, 19, 14879–14901. [Google Scholar] [CrossRef]
- Palmeri, R.; Parafati, L.; Restuccia, C.; Fallico, B. Application of prickly pear fruit extract to improve domestic shelf life, quality and microbial safety of sliced beef. Food Chem. Toxicol. 2018, 118, 355–360. [Google Scholar] [CrossRef]
- Parafati, L.; Palmeri, R.; Trippa, D.; Restuccia, C.; Fallico, B. Quality Maintenance of Beef Burger Patties by Direct Addiction or Encapsulation of a Prickly Pear Fruit Extract. Front. Microbiol. 2019, 10, 1760. [Google Scholar] [CrossRef] [PubMed]
- Rana, S.M.; Billah, M.M.; Hossain, M.S.; Saifuddin, A.K.M.; Azizul Islam, S.K.M.; Banik, S.; Naim, Z.; Raju, G.S. Susceptibility of microorganism to selected medicinal plants in Bangladesh. Asian Pac. J. Trop. Med. 2014, 4, 911–917. [Google Scholar] [CrossRef]
- Koohsari, H.; Ghaemi, E.A.; Sadegh Sheshpoli, M.; Jahedi, M.; Zahiri, M. The investigation of antibacterial activity of selected native plants from North of Iran. J. Med. Life 2015, 8, 38–42. [Google Scholar] [PubMed]
- Scavo, A.; Pandino, G.; Restuccia, C.; Parafati, L.; Cirvilleri, G.; Mauromicale, G. Antimicrobial activity of cultivated cardoon (Cynara cardunculus L. Var. Altilis DC.) leaf extracts against bacterial species of agricultural and food interest. Ind. Crop. Prod. 2019, 129, 206–211. [Google Scholar] [CrossRef]
| pH | Citric Acid (%) | °Brix | Reducing Sugar (%) | Dry Matter (%) | Protein (%) | ||
|---|---|---|---|---|---|---|---|
| Agostani | white | 6.30 ± 0.03 d | 0.019 ± 0.000 b | 13.00 ± 0.00 a | 12.73 ± 1.05 a | 13.99 ± 0.23 ab | 1.24 ± 0.01 b |
| red | 6.43 ± 0.03 b | 0.026 ± 0.000 a | 13.25 ± 0.75 a | 12.79 ± 2.28 a | 12.85 ± 0.12 ab | 1.22 ± 0.05 b | |
| yellow | 6.18 ± 0.05 e | 0.026 ± 0.000 a | 11.75 ± 0.25 b | 11.05 ± 1.69 a | 12.48 ± 1.36 b | 1.23 ± 0.24 b | |
| Bastardoni | white | 6.50 ± 0.03 a | 0.026 ± 0.000 a | 12.70 ± 0.75 a | 11.80 ± 1.21 a | 14.10 ± 1.97 a | 1.65 ± 0.40 a |
| red | 6.40 ± 0.03 c | 0.022 ± 0.003 ab | 13.50 ± 0.50 a | 12.60 ± 1.11 a | 13.36 ± 0.53 ab | 1.74 ± 0.14 a | |
| yellow | 6.40 ± 0.02 b | 0.0026 ± 0.006a | 13.00 ± 0.00 a | 11.50 ± 1.12 a | 14.05 ± 0.93 a | 1.65 ± 0.18 a |
| Samples | Color Parameters | |||||||
|---|---|---|---|---|---|---|---|---|
| L* | a* | b* | C | h | ΔE | Δh | ||
| White | Agostani | 58.52 ± 0.53 a | −8.43 ± 0.26 e | 55.95 ± 0.75 a | 56.58 ± 0.76 a | 98.57 ± 0.22 b | 11.13 ± 0.98 a | −2.42 ± 0.54 b |
| Bastardoni | 48.60 ± 0.69 b | −9.94 ± 0.42 f | 51.19 ± 0.81 b | 52.15 ± 0.81 c | 100.99 ± 0.48 a | |||
| Red | Agostani | 10.53 ± 0.42 e | 25.72 ± 0.56 c | 10.36 ± 0.25 d | 27.73 ± 0.58 e | 21.94 ± 0.44 e | 2.21 ± 1.06 c | −0.02 ± 0.04 a |
| Bastardoni | 9.53 ± 0.68 f | 24.16 ± 0.64 d | 10.22 ± 0.95 d | 26.25 ± 0.40 f | 22.94 ± 2.35 e | |||
| Yellow | Agostani | 25.74 ± 0.37 c | 39.87 ± 0.61 a | 34.97 ± 0.34 c | 53.04 ± 0.53 b | 41.25 ± 0.49 d | 7.53 ± 0.78 b | −0.10 ± 0.01 a |
| Bastardoni | 24.59 ± 0.97 d | 32.57 ± 0.67 b | 35.04 ± 0.86 c | 47.84 ± 0.74 d | 47.09 ± 0.97 c | |||
| Agostani | Bastardoni | |||||
|---|---|---|---|---|---|---|
| White | Red | Yellow | White | Red | Yellow | |
| E. coli | - | 0.60 ± 0.00 a | 0.60 ± 0.00 a | - | 0.50 ± 0.00 b | 0.60 ± 0.00 a |
| S. enterica | 1.00 ± 0.00 a | 0.97 ± 0.06 a | 0.97 ± 0.06 a | 1.00 ± 0.00 a | 1.00 ± 0.00 a | 0.97 ± 0.06 a |
| Ps. fluorescens | 0.23 ± 0.03 a | 0.10 ± 0.00 c | - | 0.27 ± 0.03 a | 0.17 ± 0.03 b | 0.27 ± 0.03 a |
| L. innocua | - | - | - | - | - | - |
| L. gray | - | - | - | - | - | - |
| St. aureus | - | - | - | - | - | - |
| B. subtilis | 0.77 ± 0.06 a | 0.27 ± 0.06 c | 0.20 ± 0.00 c | 0.27 ± 0.06 c | 0.47 ± 0.06 b | 0.50 ± 0.00 b |
| B. cereus | - | - | - | - | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palmeri, R.; Parafati, L.; Arena, E.; Grassenio, E.; Restuccia, C.; Fallico, B. Antioxidant and Antimicrobial Properties of Semi-Processed Frozen Prickly Pear Juice as Affected by Cultivar and Harvest Time. Foods 2020, 9, 235. https://doi.org/10.3390/foods9020235
Palmeri R, Parafati L, Arena E, Grassenio E, Restuccia C, Fallico B. Antioxidant and Antimicrobial Properties of Semi-Processed Frozen Prickly Pear Juice as Affected by Cultivar and Harvest Time. Foods. 2020; 9(2):235. https://doi.org/10.3390/foods9020235
Chicago/Turabian StylePalmeri, Rosa, Lucia Parafati, Elena Arena, Emanuela Grassenio, Cristina Restuccia, and Biagio Fallico. 2020. "Antioxidant and Antimicrobial Properties of Semi-Processed Frozen Prickly Pear Juice as Affected by Cultivar and Harvest Time" Foods 9, no. 2: 235. https://doi.org/10.3390/foods9020235
APA StylePalmeri, R., Parafati, L., Arena, E., Grassenio, E., Restuccia, C., & Fallico, B. (2020). Antioxidant and Antimicrobial Properties of Semi-Processed Frozen Prickly Pear Juice as Affected by Cultivar and Harvest Time. Foods, 9(2), 235. https://doi.org/10.3390/foods9020235






