Comparison of High Hydrostatic Pressure, Ultrasound, and Heat Treatments on the Quality of Strawberry–Apple–Lemon Juice Blend
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples
2.2. Chemicals
2.3. Preparation of Juice Blend
2.4. HHP, US, and HT Treatments of the Juice Blend
2.5. Storage Study
2.6. Microbial Analysis
2.7. pH and TSS Analysis
2.8. Turbidity Analysis
2.9. Instrumental Color Assessment
2.10. Ascorbic Acid Analysis
2.11. Total Phenol Analysis
2.12. Total Anthocyanin Analysis
2.13. Determination of Antioxidant Capacity
2.14. Sensory Analyses
2.15. Statistical Analysis
3. Results and Discussion
3.1. Microbial Counts after Storage in Juice Blends Treated by HHP, US, and HT
3.2. pH, Total Soluble Solids (TSS), and Turbidity after Storage in Juice Blends Treated by HHP, US, and HT
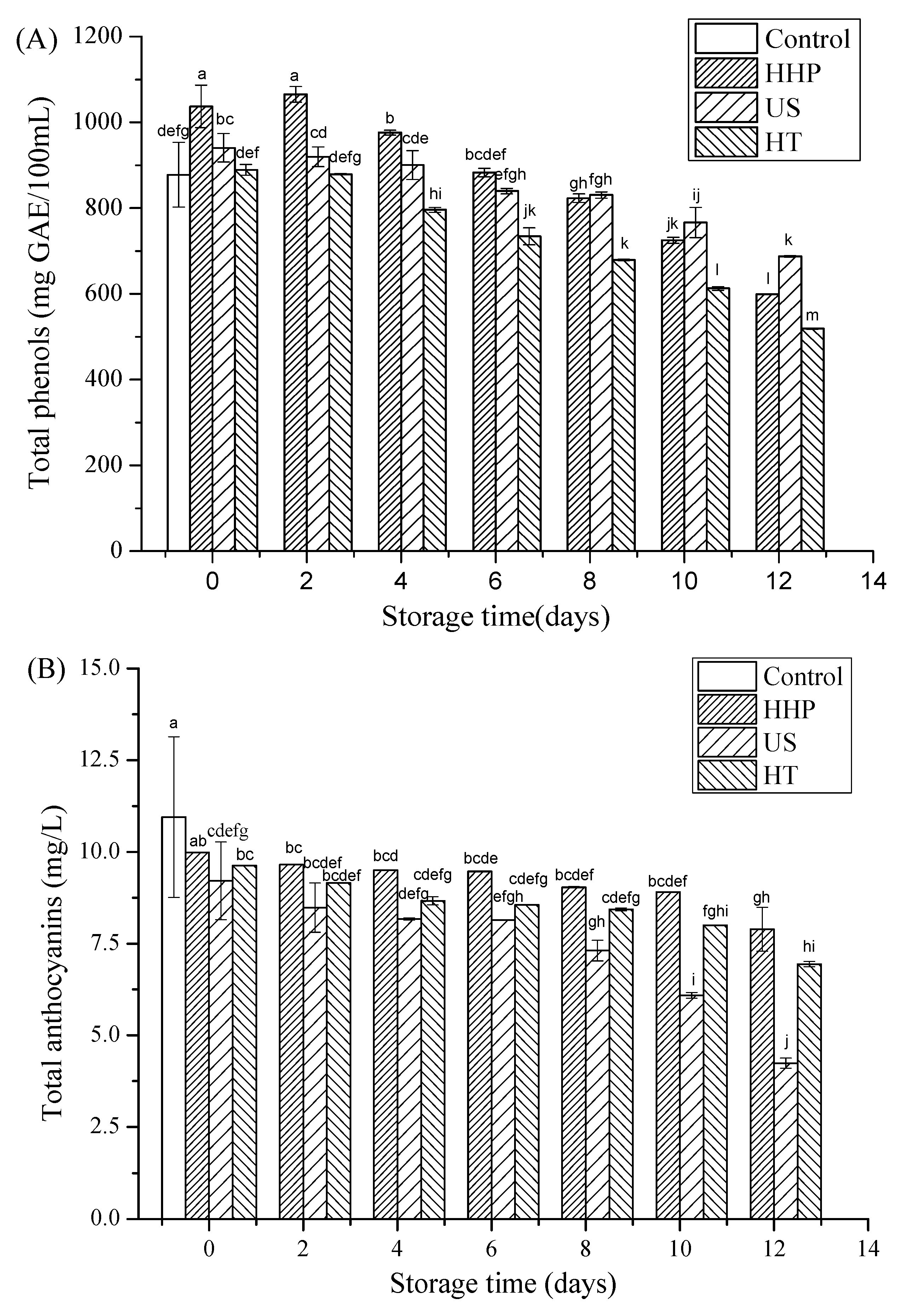
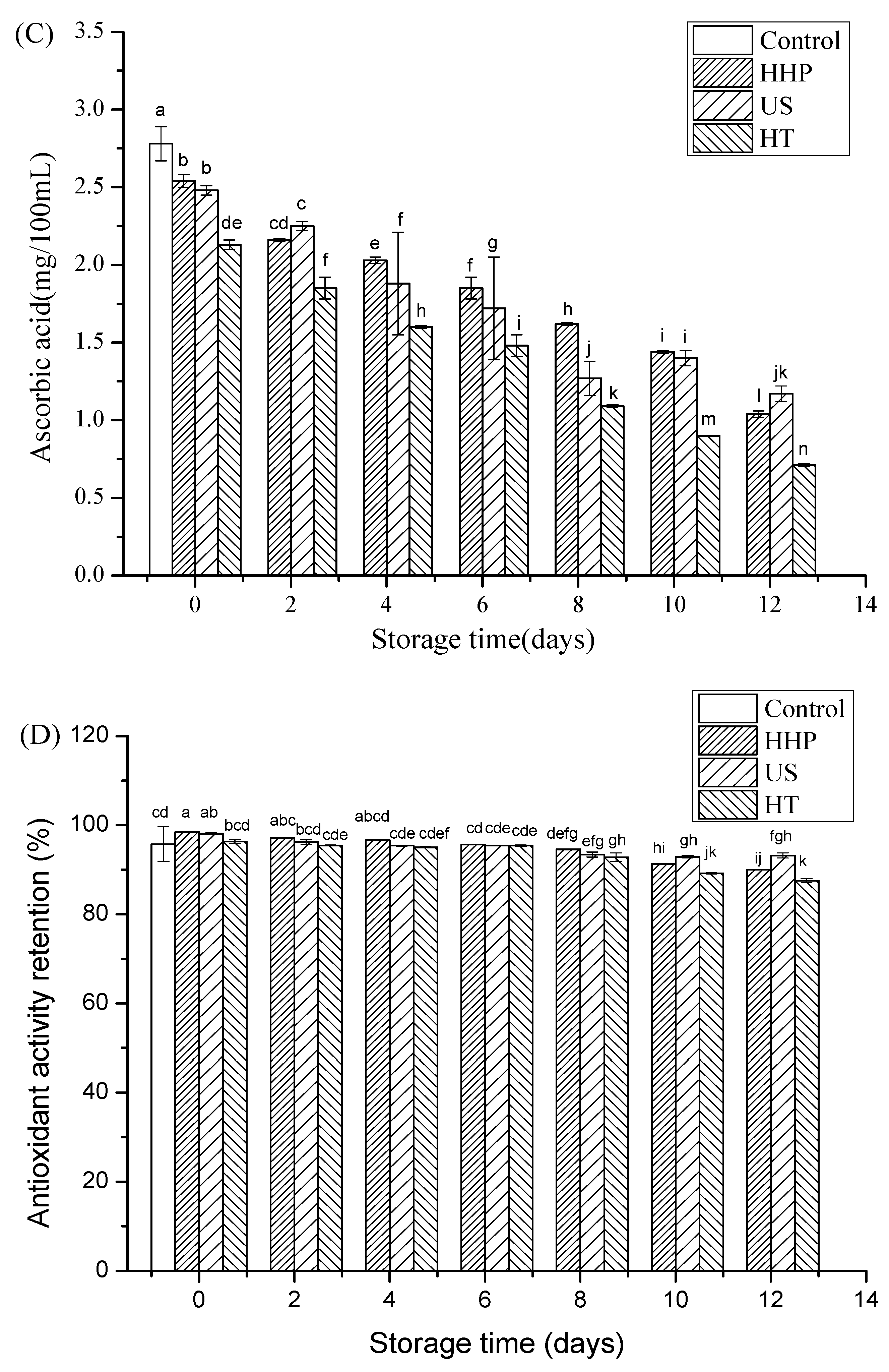
3.3. Total Phenols, Total Anthocyanins, and Ascorbic Acid after Storage in Juice Blends Treated by HHP, US, and HT
3.4. Antioxidant Capacity after Storage of Juice Blends Treated by HHP, US, and HT
3.5. Color Changes after Storage in Juice Blends Treated by HHP, US, and HT
3.6. Sensory Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rathod, A.S.; Shakya, B.; Ade, K.D. Studies on effect of thermal processing on preparation of bael fruit RTS blended effect of thermal processing on preparation of bael fruit RTS blended with aonla. Int. J. Res. Eng. Adv. Technol. 2014, 2, 1–5. [Google Scholar]
- Rojo, M.C.; Cristiani, M.; Szerman, N.; Gonzalez, M.L.; Lerena, M.C.; Mercado, L.A.; Combina, M. Reduction of Zygosaccharomyces rouxii population in concentrated grape juices by thermal pasteurization and hydrostatic high pressure processing. Food Bioprocess Technol. 2019, 12, 781–788. [Google Scholar] [CrossRef]
- Adekunte, A.; Brijesh kumar, T.; Cullen, P.J.; Scannell, A.; O’Donnell, C. Effect of sonication on colour, ascorbic acid and yeast inactivation in tomato juice. Food Chem. 2010, 122, 500–507. [Google Scholar] [CrossRef]
- Gómez-López, V.; Orsolani, L.; Martínez-Yépez, A.; Tapia, M. Microbiological and sensory quality of sonicated calcium-added orange juice. LWT-Food Sci. Technol. 2010, 43, 808–813. [Google Scholar] [CrossRef]
- Santhirasegaram, V.; Razali, Z.; George, D.; Somasundram, C. Effects of thermal and non-thermal processing on phenolic compounds, antioxidant activity and sensory attributes of chokanan Mango (Mangifera indica L.) Juice. Food Bioprocess Technol. 2015, 8, 2256–2267. [Google Scholar] [CrossRef]
- Aadil, R.; Zeng, X.A.; Han, Z.; Sun, D.W. Effects of ultrasound treatments on quality of grapefruit juice. Food Chem. 2013, 141, 3201–3206. [Google Scholar] [CrossRef]
- San Martin-Gonzalez, M.F.; Barbosa-Cánovas, G.; Swanson, B. Food processing by high hydrostatic pressure. Crit. Rev. Food Sci. Nutr. 2002, 42, 627–645. [Google Scholar] [CrossRef]
- Ferrari, G.; Maresca, P.; Ciccarone, R. The effects of high hydrostatic pressure on the polyphenols and anthocyanins in red fruit products. Procedia Food Sci. 2011, 1, 847–853. [Google Scholar] [CrossRef]
- Barba, F.; Jäger, H.; Meneses, N.; Esteve, M.J.; Frigola, A.; Knorr, D. Evaluation of quality changes of blueberry juice during refrigerated storage after high-pressure and pulsed electric fields processing. Innov. Food Sci. Emerg. Technol. 2012, 14, 18–24. [Google Scholar] [CrossRef]
- Juarez-Enriquez, E.; Salmeron, I.; Gutierrez-Mendez, N.; Ramaswamy, H.; Ortega-Rivas, E. Shelf life studies on apple juice pasteurised by ultrahigh hydrostatic pressure. LWT-Food Sci. Technol. 2014, 62, 915–919. [Google Scholar] [CrossRef]
- Varela-Santos, E.; Ochoa, A.; Tabilo, G.; Reyes, J.; Pérez-Won, M.; Briones-Labarca, V.; Morales-Castro, J. Effect of high hydrostatic pressure (HHP) processing on physicochemical properties, bioactive compounds and shelf-life of pomegranate juice. Innov. Food Sci. Emerg. Technol. 2011, 13, 12–22. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, Y.; Lin, T.; Liao, X. Quality assurance in pepper and orange juice blend treated by high pressure processing and high temperature short time treatment. Innov. Food Sci. Emerg. Technol. 2015, 31, 28–36. [Google Scholar] [CrossRef]
- Fernandez, A.; Butz, P.; Bognàr, A.; Tauscher, B. Antioxidative capacity, nutrient content and sensory quality of orange juice and an orange-lemon-carrot juice product after high pressure treatment and storage in different packaging. Eur. Food Res. Technol. 2001, 13, 290–296. [Google Scholar] [CrossRef]
- Piyasena, P.; Ohareb, E.; McKellar, R.C. Inactivation of microbes using ultrasound: A review. Int. J. Food Microbiol. 2013, 87, 207–216. [Google Scholar] [CrossRef]
- O’Donnell, C.; Brijesh kumar, T.; Bourke, P.; Cullen, P.J. Effect of ultrasonic processing on food enzymes of industrial importance. Trends Food Sci. Technol. 2010, 21, 358–367. [Google Scholar] [CrossRef]
- Nadeem, M.; Ubaid, N.; Qureshi, T.; Munir, M.; Mehmood, A. Effect of ultrasound and chemical treatment on total phenol, flavonoids and antioxidant properties on carrot-grape juice blend during storage. Ultrason. Sonochem. 2018, 45, 1–6. [Google Scholar] [CrossRef]
- Gao, J.; Rupasinghe, H.V. Nutritional, physicochemical and microbial quality of ultrasound-treated apple-carrot juice blends. Food Nutr. Sci. 2012, 3, 212. [Google Scholar] [CrossRef]
- Beirão-da-Costa, S.; Steiner, A.; Correia, L.; Empis, J.; Moldão-Martins, M. Effects of maturity stage and mild heat treatments on quality of minimally processed kiwifruit. J. Food Eng. 2006, 76, 616–625. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, F.; Cao, X.; Chen, F.; Hu, X.; Liao, X. Comparison of high hydrostatic pressure and high temperature short time processing on quality of purple sweet potato nectar. Innov. Food Sci. Emerg. Technol. 2012, 16, 326–334. [Google Scholar] [CrossRef]
- Daoudi, L.; Quevedo, J.; Trujillo, A.J.; Capdevila, F.; Bartra, E.; Mínguez, S.; Guamis, B. Effects of high-pressure treatment on the sensory quality of white grape juice. Int. J. High Pressure Res. 2002, 22, 705–709. [Google Scholar] [CrossRef]
- Engmann, F.; Ma, Y.; Tchabo, W.; Ma, H.; Zhang, H. Optimization of ultrasonic and high hydrostatic pressure conditions on quality parameters of mulberry (Morus Moraceae) juice using response surface methodology. J. Food Qual. 2014, 37, 297–308. [Google Scholar] [CrossRef]
- Feng, P.; Weagant, S.D.; Grant, M.A.; Burkhardt, W.; Shellfish, M.; Water, B. BAM: Enumeration of Escherichia coli and the coliform bacteria. Bacteriol. Anal. Manual. 2002, 213, 290–296. [Google Scholar]
- Abid, M.; Jabbar, D.S.; Wu, T.; Hashim, M.; Hu, B.; Shicheng, L.; Zhang, X. Effect of ultrasound on different quality parameters of apple juice. Ultrason. Sonochem. 2013, 20, 1185–1187. [Google Scholar] [CrossRef] [PubMed]
- Kubo, M.; Augusto, P.; Cristianini, M. Effect of high pressure homogenization (HPH) on the physical stability of tomato juice. Food Res. Int. 2013, 51, 170–179. [Google Scholar] [CrossRef]
- Chen, D.; Pan, S.; Chen, J.; Pang, X.; Guo, X.; Gao, L.; Liao, X.; Wu, J. Comparing the effects of high hydrostatic pressure and ultrahigh temperature on quality and shelf life of cloudy ginger juice. Food Bioprocess Technol. 2016, 9, 1779–1793. [Google Scholar] [CrossRef]
- Cassani, L.; Tomadoni, B.; Ponce, A.G.; Agüero, M.; Moreira, M. Combined use of ultrasound and vanillin to improve quality parameters and safety of strawberry juice enriched with prebiotic fibers. Food Bioprocess Technol. 2017, 10, 1454–1465. [Google Scholar] [CrossRef]
- Cao, X.; Zhang, Y.; Zhang, F.; Wang, Y.; Yi, J.; Liao, X. Effects of high hydrostatic pressure on enzymes, phenolic compounds, anthocyanins, polymeric color and color of strawberry pulps. J. Sci. Food Agric. 2011, 91, 877–885. [Google Scholar] [CrossRef]
- Zou, H.; Lin, T.; Bi, X.; Zhao, L.; Wang, Y.; Liao, X. Comparison of high hydrostatic pressure, high-pressure carbon dioxide and high-temperature short-time processing on quality of mulberry juice. Food Bioprocess Technol. 2015, 9, 217–231. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Kaya, Z.; Yildiz, S.; Ünlütürk, S. Effect of UV-C irradiation and heat treatment on the shelf life stability of a lemon-melon juice blend: Multivariate statistical approach. Innov. Food Sci. Emerg. Technol. 2015, 29, 230–239. [Google Scholar] [CrossRef]
- Lavinas, F.; Miguel, M.; Lopes, M.; Valente Mesquita, V. Effect of high hydrostatic pressure on cashew apple (Anacardium occidentale L.) juice preservation. J. Food Sci. 2008, 73, M273–M277. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Flores, H.E.; Garnica-Romo, M.G.; Bermudez-Aguirre, D.; Pokhrel, P.; Barbosa-Cánovas, G.V. Physico-chemical parameters, bioactive compounds and microbial quality of thermo-sonicated carrot juice during storage. Food Chem. 2015, 172, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Del Caro, A.; Piga, A.; Vacca, V.; Agabbio, M. Changes of flavonoids, vitamin C and antioxidant capacity in minimally processed citrus segments and juices during storage. Food Chem. 2004, 84, 99–105. [Google Scholar] [CrossRef]
- Kang, D.; Dougherty, R.; Swanson, B. Controlling Alicyclobacillus acidoterrestris in fruit juices by high pressure and high temperature. Nutr. Reprod. Food Sci. Human Nutr. 2003, 34, 311–316. [Google Scholar]
- Brijesh kumar, T.; Muthukumarappan, K.; O’Donnell, C.; Cullen, P.J. Effects of sonication on the kinetics of orange juice quality parameters. J. Agric. Food Chem. 2008, 56, 2423–2428. [Google Scholar]
- Cao, X.; Bi, X.; Huang, W.; Wu, J.; Hu, X.; Liao, X. Changes of quality of high hydrostatic pressure processed cloudy and clear strawberry juices during storage. Innov. Food Sci. Emerg. Technol. 2012, 16, 181–190. [Google Scholar] [CrossRef]
- Tiwari, B.K.; O’Donnell, C.P.; Muthukumarappan, K.; Cullen, P.J. Effect of sonication on orange juice quality parameters during storage. Int. J. Food Sci. Technol. 2009, 44, 586–595. [Google Scholar] [CrossRef]
- Huang, W.; Bi, X.; Zhang, X.; Liao, X.; Hu, X.; Wu, J. Comparative study of enzymes, phenolics, carotenoids and color of apricot nectars treated by high hydrostatic pressure and high temperature short time. Innov. Food Sci. Emerg. Technol. 2013, 18, 74–82. [Google Scholar] [CrossRef]
- Kim, H.K.; Leem, K.H.; Lee, S.; Kim, B.Y.; Hahm, Y.; Cho, H.Y.; Lee, J. Effect of high hydrostatic pressure on immunomodulatory activity of cloudy apple juice. Food Sci. Biotechnol. 2012, 21, 175–181. [Google Scholar] [CrossRef]
- Sadilova, E.; Carle, R.; Stintzing, F. Thermal degradation of anthocyanin and impact on color and in vitro antioxidant capacity. Mol. Nutr. Food Res. 2007, 51, 1461–1471. [Google Scholar] [CrossRef]
- Dubrovi, I.; Herceg, Z.; Režek Jambrak, A.; Badanjak Sabolović, M.; Verica, D.U. Effect of high intensity ultrasound and pasteurization on anthocyanin content in strawberry juice. Food Technol. Biotechnol. 2013, 49, 196–204. [Google Scholar]
- Adams, J. Thermal degradation of anthocyanins with particular reference to the 3-glycosides of cyanidin. I. In acidified aqueous solution at 100 °C. J. Sci. Food Agric. 1973, 24, 747–762. [Google Scholar] [CrossRef]
- Patras, A.; Brunton, N.; Brijesh kumar, T.; Butler, F. Stability and degradation kinetics of bioactive compounds and colour in strawberry jam during storage. Food Bioprocess Technol. 2011, 4, 1245–1252. [Google Scholar] [CrossRef]
- Castañeda, A.; de Lourdes Pacheco-Hernandez, M.; Páez-Hernández, M.E.; Rodriguez, J.A.; Galán-Vidal, C.A. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859–871. [Google Scholar]
- Sánchez-Moreno, C.; Plaza, L.; Elez-Martinez, P.; De Ancos, B.; Martin-Belloso, O.; Cano, M.P. Impact of high pressure and pulsed electric fields on bioactive compounds and antioxidant activity of orange juice in comparison with traditional thermal processing. J. Agric. Food Chem. 2005, 53, 4403–4409. [Google Scholar] [CrossRef]
- Korn, M.; Primo, P.M.; de Sousa, C.S. Influence of ultrasonic waves on phosphate determination by the molybdenum blue method. Microchem. J. 2002, 73, 273–277. [Google Scholar] [CrossRef]
- Patras, A.; Brunton, N.; Pieve, S.; Butler, F. Impact of high pressure processing on total antioxidant activity, phenolic, ascorbic acid, anthocyanin content and color of strawberry and blackberry purées. Innov. Food Sci. Emerg. Technol. 2009, 10, 308–313. [Google Scholar] [CrossRef]
- Dede, S.; Alpas, H.; Bayindirli, A. High hydrostatic pressure treatment and storage of carrot and tomato juice: Antioxidant activity and microbial safety. J. Sci. Food Agric. 2007, 87, 773–782. [Google Scholar] [CrossRef]
- Zenker, M.; Heinz, V.; Knorr, D. Application of ultrasound-assisted thermal processing for preservation and quality retention of liquid foods. J. Food Prot. 2003, 66, 1642–1649. [Google Scholar] [CrossRef] [PubMed]
- Davey, M.W.; Montagu, M.V.; Inze, D.; Sanmartin, M.; Kanellis, A.; Smirnoff, N.; Benzie, I.J.J.; Strain, J.J.; Favell, D.; Fletcher, J. Plant L-ascorbic acid: Chemistry, function, metabolism, bioavailability and effects of processing. J. Sci. Food Agric. 2000, 80, 825–860. [Google Scholar] [CrossRef]
- Siddiqui, M.W.; Chakraborty, I.; Ayala-Zavala, J.; Dhua, R. Advances in minimal processing of fruits and vegetables: A review. J. Sci. Ind. Res. 2011, 70, 823–834. [Google Scholar]
- Bhat, R.; Kamaruddin, N.; Min-Tze, L.; Karim, A. Sonication improves kasturi lime (Citrus microcarpa) juice quality. Ultrason. Sonochem. 2011, 18, 1295–1300. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, F.L.; Escribano-Bailón, M.T.; Alonso, J.J.P.; Rivas-Gonzalo, J.C.; Santos-Buelga, C. Anthocyanin pigments in strawberry. LWT-Food Sci. Technol. 2007, 40, 374–382. [Google Scholar] [CrossRef]
- Gorinstein, S.; Zachwieja, Z.; Katrich, E.; Pawelzik, E.; Haruenkit, R.; Trakhtenberg, S.; Martin-Belloso, O. Comparison of the contents of the main antioxidant compounds and the antioxidant activity of white grapefruit and his new hybrid. LWT-Food Sci. Technol. 2004, 37, 337–343. [Google Scholar] [CrossRef]
- Costa, M.; Fonteles, T.; De Jesus, A.L.; Almeida, F.; Miranda, M.; Fernandes, F.; Rodrigues, S. High-intensity ultrasound processing of pineapple juice. Food Bioprocess Technol. 2011, 6, 997–1006. [Google Scholar] [CrossRef]
- Tomadoni, B.; Cassani, L.; Viacava, G.; Moreira, M.; Ponce, A.G. Effect of ultrasound and storage time on quality attributes of strawberry juice. J. Food Process Eng. 2017, 40, e12533. [Google Scholar] [CrossRef]
- Sadilova, E.; Stintzing, F.; Kammerer, D.; Carle, R. Matrix dependent impact of sugar and ascorbic acid addition on color and anthocyanin stability of black carrot, elderberry and strawberry single strength and from concentrate juices upon thermal treatment. Food Res. Int. 2009, 42, 1023–1033. [Google Scholar] [CrossRef]
- Barbosa-Cánovas, G.; Rodríguez, J. Update on nonthermal food processing technologies: Pulsed electric field, high hydrostatic pressure, irradiation and ultrasound. Food Aust. 2002, 54, 513–520. [Google Scholar]
- Mason, T.J.; Paniwnyk, L.; Lorimer, J. The uses of ultrasound in food technology. Ultrason. Sonochem. 1996, 3, S253–S260. [Google Scholar] [CrossRef]
- Brijesh kumar, T.; O’Donnell, C.; Patras, A.; Cullen, P.J. Anthocyanin and ascorbic acid degradation in sonicated strawberry juice. J. Agric. Food Chem. 2008, 56, 10071–10077. [Google Scholar]
- Sentandreu, E.; Carbonell, L.; Carbonell, J.; Izquierdo, L. Effects of heat treatment conditions on fresh taste and on pectinmethylesterase activity of chilled mandarin and orange juices. Food Sci. Technol. Int. 2005, 11, 217–222. [Google Scholar] [CrossRef]
- Parish, M. Orange juice quality after treatment by thermal pasteurization or isostatic high pressure. LWT-Food Sci. Technol. 1998, 31, 439–442. [Google Scholar] [CrossRef]
- Igual, M.; Contreras, C.; Camacho, M.; Martínez-Navarrete, N. Effect of thermal treatment and storage conditions on the physical and sensory properties of grapefruit juice. Food Bioprocess Technol. 2014, 7, 191–203. [Google Scholar] [CrossRef]
- Brijesh kumar, T.; O’Donnell, C.; Muthukumarappan, K.; Cullen, P.J. Ascorbic acid degradation kinetics of sonicated orange juice during storage and comparison with thermally pasteurised juice. LWT-Food Sci. Technol. 2009, 42, 700–704. [Google Scholar]
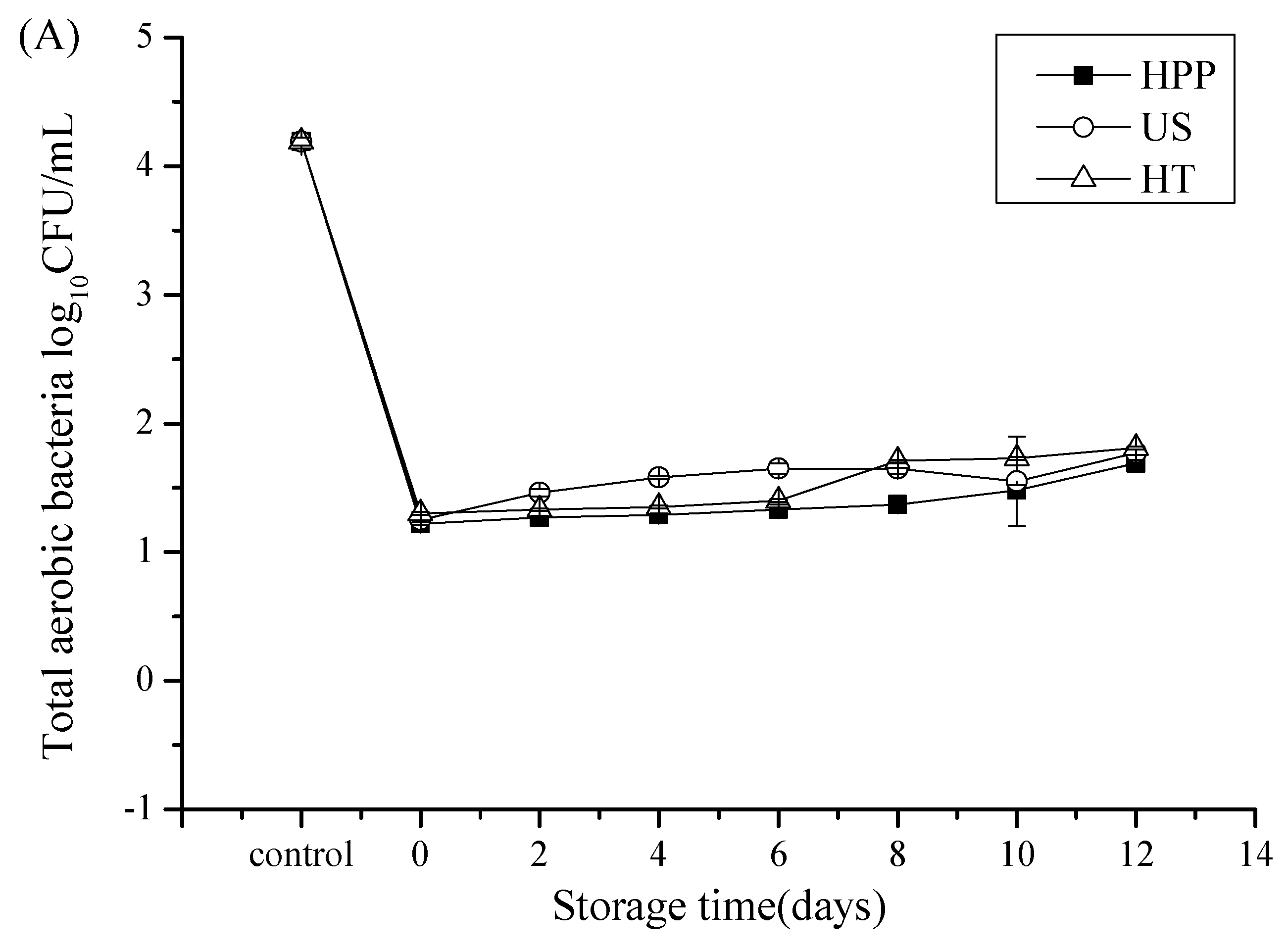
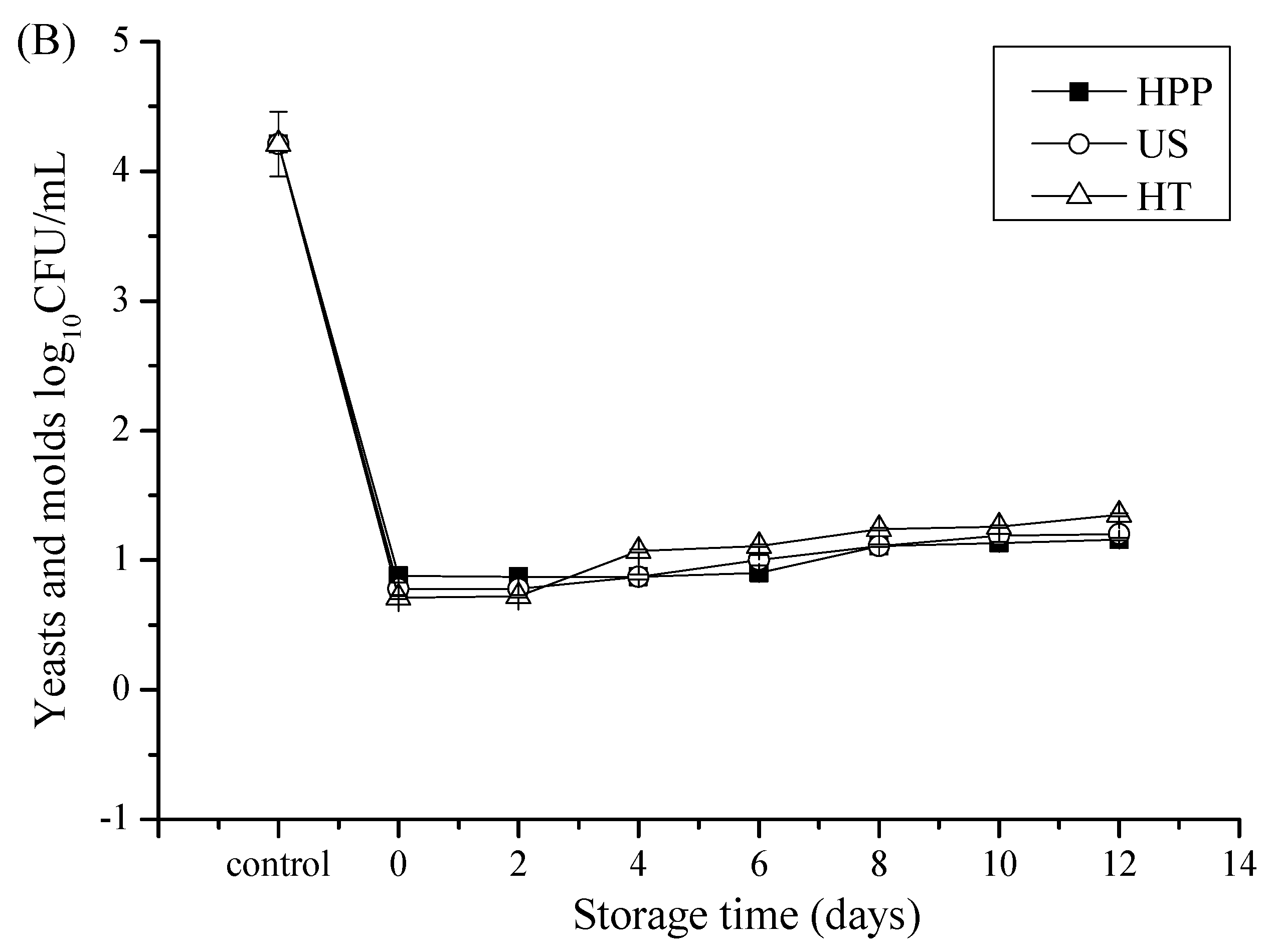
| Storage Time | Treatments | |||
|---|---|---|---|---|
| Control | HHP | US | HT | |
| 0 | 120 | <3 | <3 | <3 |
| 2 | ND | <3 | <3 | <3 |
| 4 | ND | <3 | <3 | <3 |
| 6 | ND | <3 | <3 | <3 |
| 8 | ND | <3 | <3 | <3 |
| 10 | ND | <3 | <3 | <3 |
| 12 | ND | <3 | <3 | <3 |
| Process | Storage (Days) | pH | TSS (°Brix) | Turbidity (A660) | Color | |||
|---|---|---|---|---|---|---|---|---|
| L* | a* | b* | ΔE* | |||||
| Control | 0 | 3.38 ± 0.01 bc | 7.80 ± 0 a | 0.19 ± 0.12 bcd | 21.59 ± 0.92 a | 7.29 ± 0.38 bc | 4.3 ± 0.20 abc | 0 |
| HHP (500 MPa/15 min/15 °C) | 0 | 3.37 ± 0.01 cde | 7.80 ± 0 a | 0.11 ± 0 cdefg | 20.02 ± 0 bc | 7.57 ± 0 a | 4.43 ± 0.01 a | 1.60 p |
| 2 | 3.38 ± 0.01 bcd | 7.77 ± 0.06 a | 0.05 ± 0.06 efg | 19.88 ± 0.02 bcd | 7.50 ± 0 ab | 4.4 ± 0.01 ab | 1.73 o | |
| 4 | 3.38 ± 0.01 bcd | 7.77 ± 0.06 a | 0.09 ± 0 defg | 19.74 ± 0.18 cde | 7.4 ± 0.04 abc | 4.4 ± 0 ab | 1.86 m | |
| 6 | 3.38 ± 0.01 bcd | 7.80 ± 0 a | 0.08 ± 0 efg | 19.40 ± 0.07 cdef | 7.25 ± 0.01 bcd | 4.37 ± 0 ab | 2.02 l | |
| 8 | 3.37 ± 0.01 cde | 7.77 ± 0.06 a | 0.07 ± 0 efg | 19.13 ± 0.10 defg | 7.19 ± 0.01 bc | 4.37 ± 0.04 abc | 2.46 h | |
| 10 | 3.37 ± 0.01 cde | 7.80 ± 0 a | 0.05 ± 0 fg | 18.91 ± 0.06 fgh | 7.03 ± 0.02 de | 4.21 ± 0 abcd | 2.68 f | |
| 12 | 3.37 ± 0.01 cde | 7.80 ± 0 a | 0.03 ± 0 g | 18.45 ± 0.16 ghi | 6.91 ± 0.02 efg | 4.11 ± 0.01 abcd | 3.17 c | |
| US (376 W/10 min/35 °C) | 0 | 3.43 ± 0 a | 7.80 ± 0 a | 0.56 ± 0.03 a | 21.59 ± 0.92 a | 6.91 ± 0.04 efg | 4.19 ± 0a bcd | 0.40 u |
| 2 | 3.35 ± 0 ef | 7.80 ± 0 a | 0.23 ± 0.09 b | 21.91 ± 0.71 a | 6.60 ± 0.06 hi | 4.08 ± 0.05 bcde | 0.79 t | |
| 4 | 3.34 ± 0.0 g | 7.80 ± 0 a | 0.16 ± 0.10 bcdef | 22.34 ± 0.10 a | 6.76 ± 0.03 fgh | 4.11 ± 0.18 abcd | 0.94 s | |
| 6 | 3.32 ± 0.02 e | 7.80 ± 0 a | 0.11 ± 0.07 cdefg | 22.04 ± 0.53 a | 6.30 ± 0.21 jk | 3.96 ± 0.38 def | 1.14 r | |
| 8 | 3.28 ± 0.03 h | 7.80 ± 0 a | 0.06 ± 0.05 efg | 20.62 ± 0.31 a | 6.31 ± 0.16 jk | 4.11 ± 0.18 abcd | 1.39 h | |
| 10 | 3.26 ± 0.01 hi | 7.80 ± 0 a | 0.03 ± 0.03 g | 20.16 ± 0.05 bc | 6.22 ± 0.05 k | 3.78 ± 0.31 ef | 2.15 k | |
| 12 | 3.25 ± 0 i | 7.80 ± 0 a | 0.02 ± 0 g | 20.01 ± 0.01 bc | 5.94 ± 0.14 l | 3.74 ± 0.01 f | 2.21 j | |
| HT (86 °C/1 min) | 0 | 3.39 ± 0.01 b | 7.80 ± 0 a | 0.05 ± 0 fg | 19.39 ± 0.02 cdef | 7.42 ± 0.03 abc | 4.40 ± 0.1 ab | 2.20 j |
| 2 | 3.39 ± 0.01 b | 7.80 ± 0 a | 0.07 ± 0 efg | 19.18 ± 0.04 defg | 7.21 ± 0.02 bc | 4.36 ± 0.01 ab | 2.41 i | |
| 4 | 3.38 ± 0.01 bc | 7.77 ± 0.06 a | 0.08 ± 0 efg | 19.01 ± 0.01 efgh | 7.01 ± 0.01 de | 4.31 ± 0.01 abc | 2.50 g | |
| 6 | 3.37 ± 0 cde | 7.80 ± 0 a | 0.09 ± 0 cdefg | 18.76 ± 0 fghi | 6.94 ± 0.06 ef | 4.29 ± 0.01 abcd | 2.84 e | |
| 8 | 3.37 ± 0.01 cde | 7.77 ± 0.06 a | 0.10 ± 0 efg | 18.51 ± 0.01 ghi | 6.87 ± 0.04 ef | 4.24 ± 0.01 abcd | 3.11 d | |
| 10 | 3.37 ± 0 cde | 7.80 ± 0 a | 0.17 ± 0 bcde | 18.01 ± 0.01 i | 6.67 ± 0.03 ghi | 4.10 ± 0.01 abcd | 3.64 a | |
| 12 | 3.36 ± 0.01 def | 7.80 ± 0 a | 0.20 ± 0 bc | 18.22 ± 0.02 hi | 6.51 ± 0.01 ij | 4.0 ± 0.02c def | 3.54 b | |
| Antioxidant Capacity | |||
|---|---|---|---|
| HHP | US | HT | |
| Total phenols | 0.971 ** | 0.854 * | 0.933 ** |
| Total anthocyanins | 0.943 ** | 0.831 * | 0.917 ** |
| Ascorbic acid | 0.968 ** | 0.963 ** | 0.922 ** |
| Storage Time | Process | Sensory Attributes | ||||
|---|---|---|---|---|---|---|
| Taste | Aroma | Color | Consistency | Total Score | ||
| 0 | Untreated | 33.25 ± 2.25 a | 24.38 ± 2.00 a | 11.63 ± 1.77 a | 12.25 ± 1.75 a | 81.50 ± 4.87 a |
| 0 | HHP | 33.25 ± 2.25 a | 23.50 ± 2.20 ab | 11.00 ± 1.6 a | 12.25 ± 1.75 a | 80.00 ± 5.37 a |
| 0 | US | 31.38 ± 2.82 ab | 22.63 ± 2.33 ab | 11.63 ± 1.92 a | 12.00 ± 1.51 a | 77.25 ± 3.37 ab |
| 0 | HT | 23.75 ± 3.78 c | 21.63 ± 3.16 ab | 10.38 ± 1.51 a | 11.63 ± 1.77 a | 67.13 ± 4.49 c |
| 10 | Untreated | ND | ND | ND | ND | ND |
| 10 | HHP | 31.38 ± 3.78 ab | 23.13 ± 2.36 ab | 10.38 ± 1.41 a | 12.00 ± 1.51 a | 76.88 ± 3.44 ab |
| 10 | US | 30.13 ± 3.76 b | 22.9 ± 3.00 ab | 11.00 ± 0.93 a | 11.63 ± 1.77 a | 73.50 ± 5.04 b |
| 10 | HT | 20.25 ± 0.71 d | 21.38 ± 2.13 b | 8.38 ± 0.52 b | 9.50 ± 0.53 b | 61.88 ± 3.00 d |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, X.; Zhou, Z.; Wang, X.; Bi, X.; Ma, Y.; Xing, Y. Comparison of High Hydrostatic Pressure, Ultrasound, and Heat Treatments on the Quality of Strawberry–Apple–Lemon Juice Blend. Foods 2020, 9, 218. https://doi.org/10.3390/foods9020218
Feng X, Zhou Z, Wang X, Bi X, Ma Y, Xing Y. Comparison of High Hydrostatic Pressure, Ultrasound, and Heat Treatments on the Quality of Strawberry–Apple–Lemon Juice Blend. Foods. 2020; 9(2):218. https://doi.org/10.3390/foods9020218
Chicago/Turabian StyleFeng, Xiaoping, Zhongyu Zhou, Xiaoqiong Wang, Xiufang Bi, Yuan Ma, and Yage Xing. 2020. "Comparison of High Hydrostatic Pressure, Ultrasound, and Heat Treatments on the Quality of Strawberry–Apple–Lemon Juice Blend" Foods 9, no. 2: 218. https://doi.org/10.3390/foods9020218
APA StyleFeng, X., Zhou, Z., Wang, X., Bi, X., Ma, Y., & Xing, Y. (2020). Comparison of High Hydrostatic Pressure, Ultrasound, and Heat Treatments on the Quality of Strawberry–Apple–Lemon Juice Blend. Foods, 9(2), 218. https://doi.org/10.3390/foods9020218