Active Paper Sheets Including Nanoencapsulated Essential Oils: A Green Packaging Technique to Control Ethylene Production and Maintain Quality in Fresh Horticultural Products—A Case Study on Flat Peaches
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experiment Description
2.3. Ethylene Production and Respiration
2.4. Physicochemical Analyses and Weight Loss
2.5. 1-Aminocyclopropane-1-Carboxylic Acid (ACC)
2.6. Enzymatic Analyses
2.6.1. ACC Oxidase (ACO)
2.6.2. Endo-Polygalacturonase (endo-PG) and Exo-Polygalacturonase (exo-PG)
2.6.3. Polyphenoloxidase (PPO)
2.7. Bioactive Compounds
2.7.1. Phenolic Compounds
2.7.2. Total Antioxidant Capacity
2.8. Statistical Analyses
3. Results
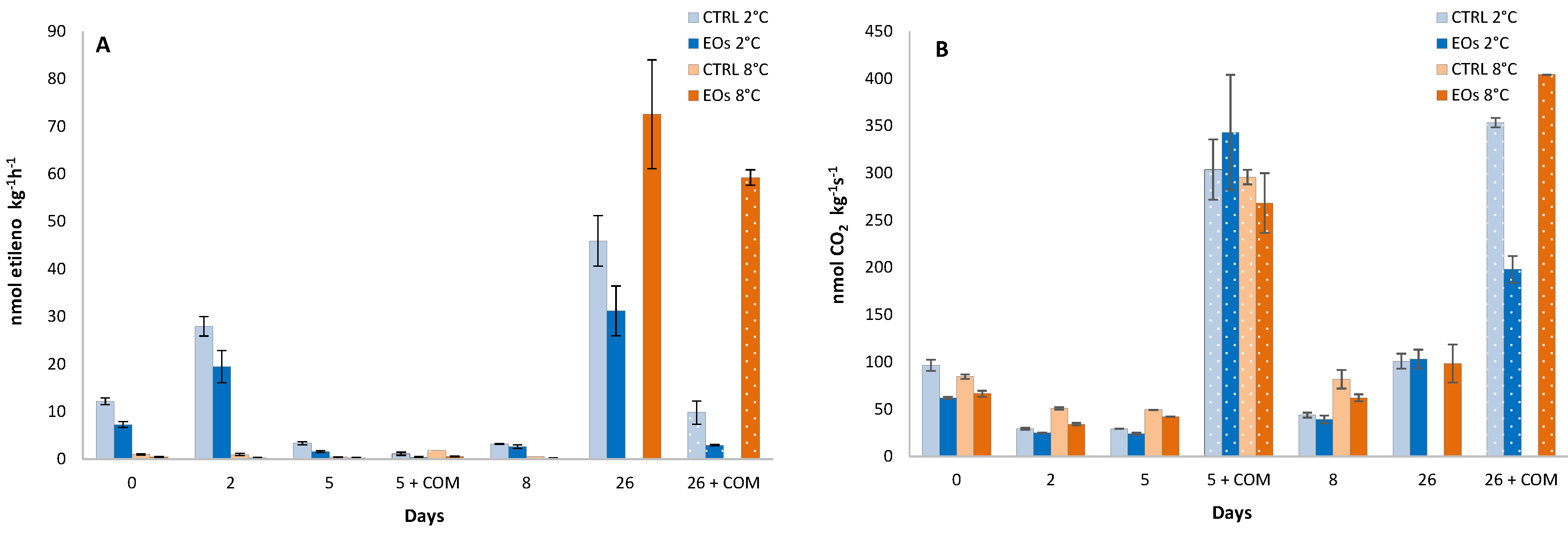
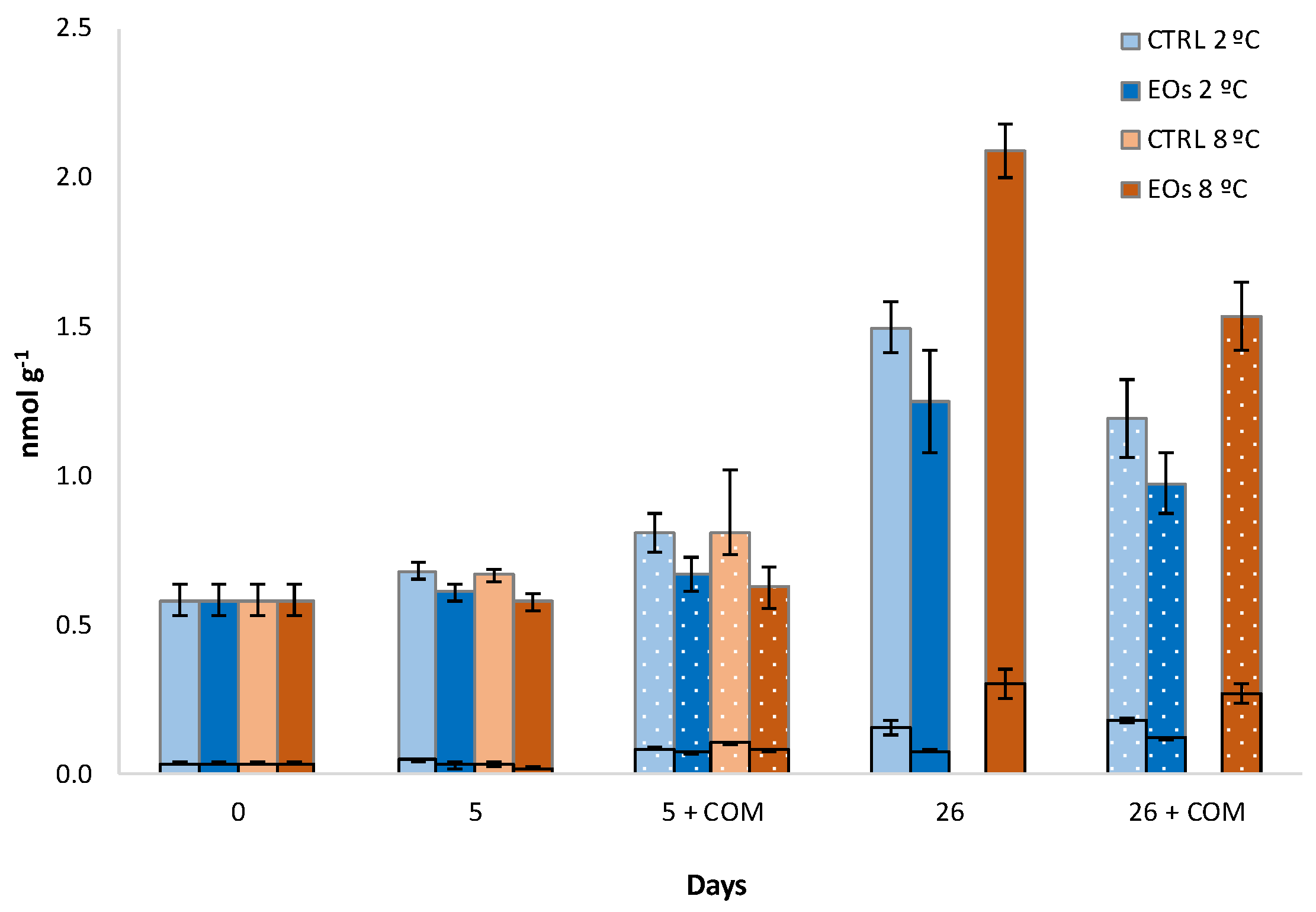
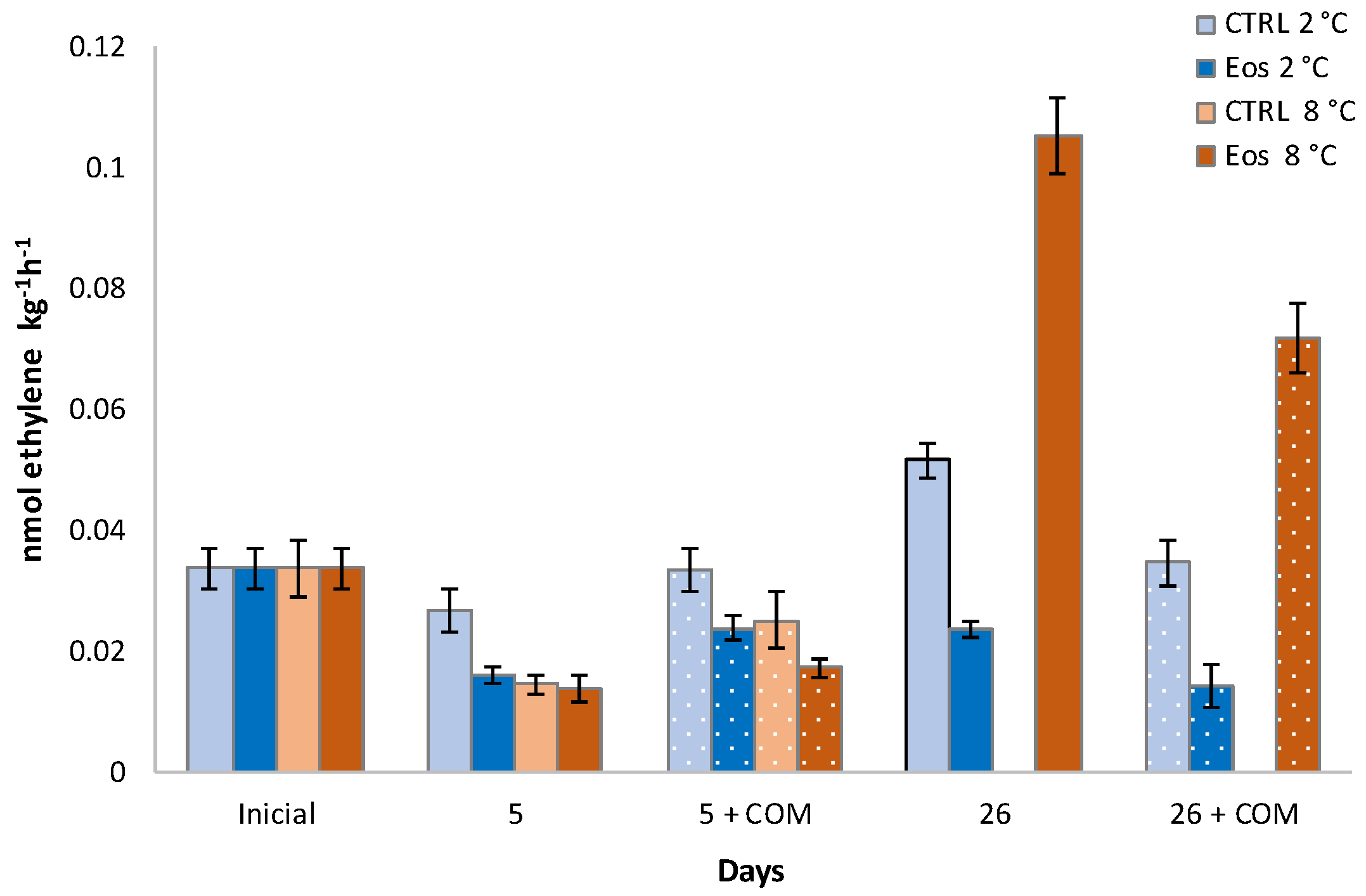
3.1. Ethylene Biosynthesis
3.2. Soluble Solids, pH, and Titratable Acidity
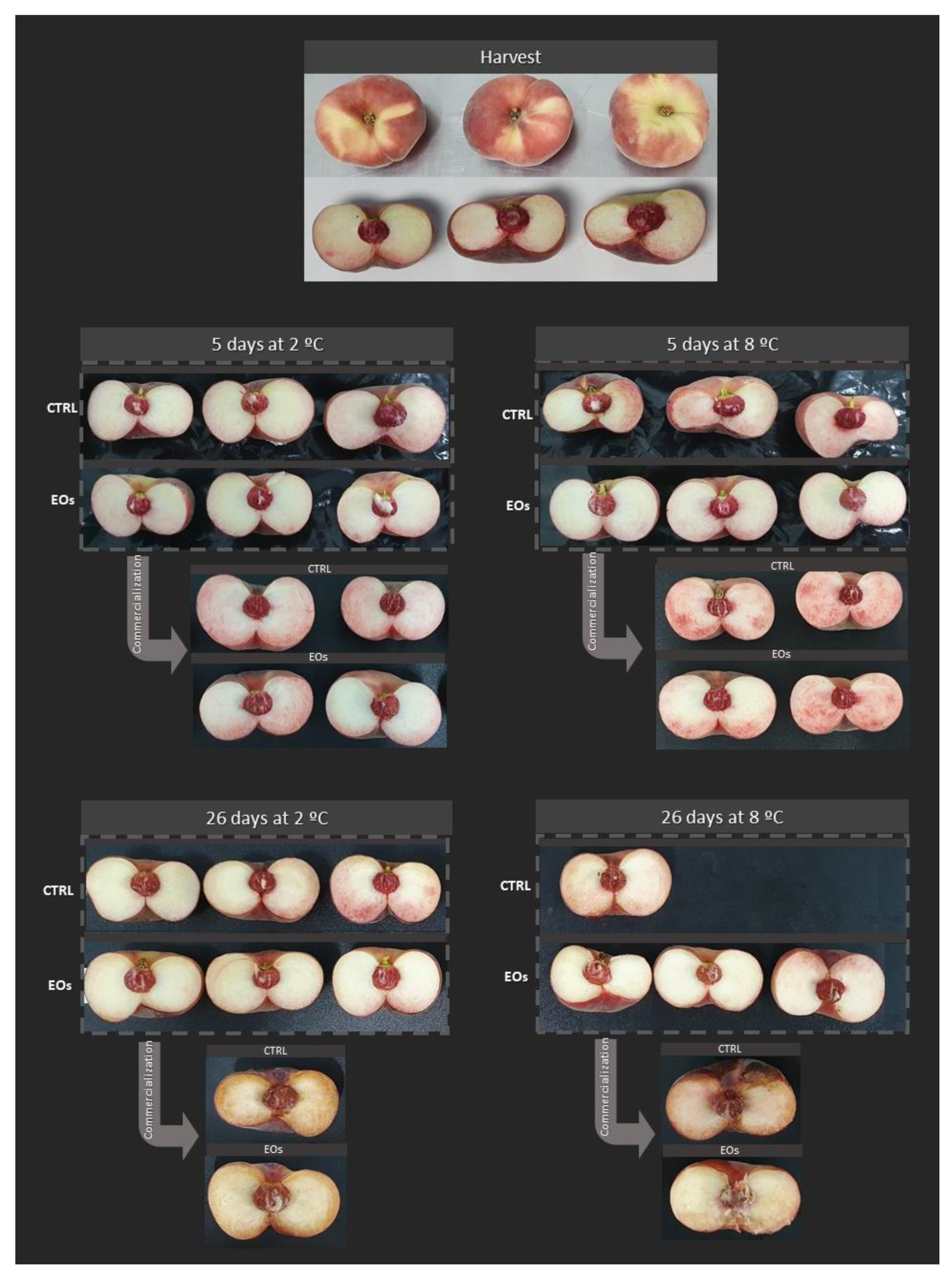
3.3. Color, Appearance, and Decay
3.4. Weight Loss
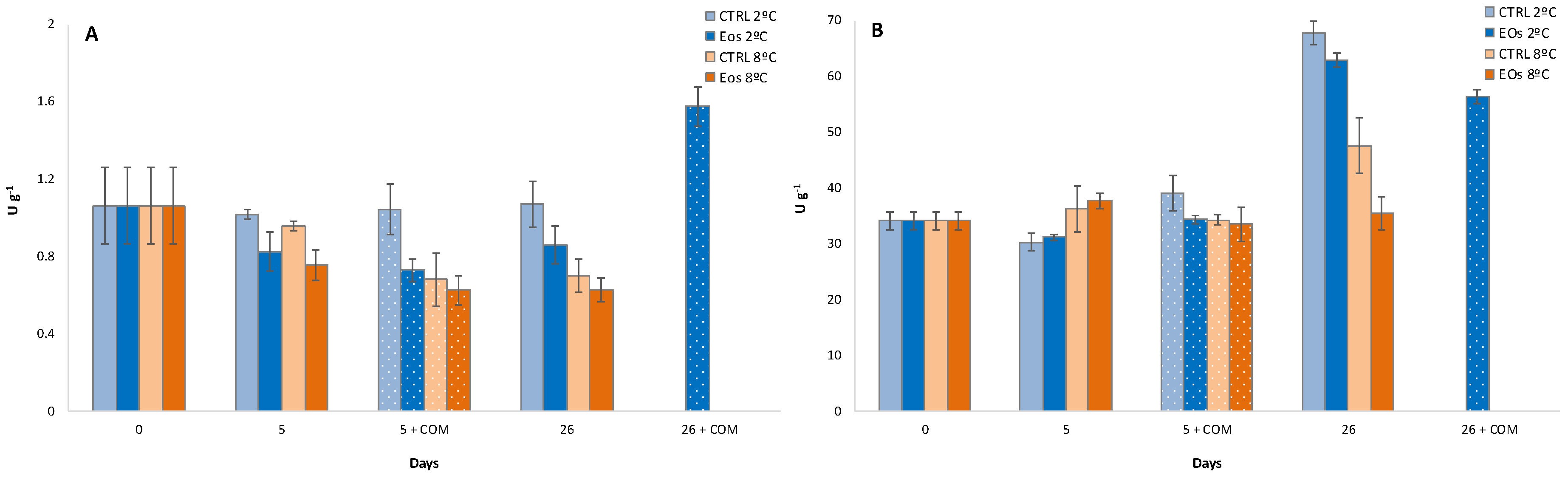
3.5. Firmness and Polygalacturonase Activity
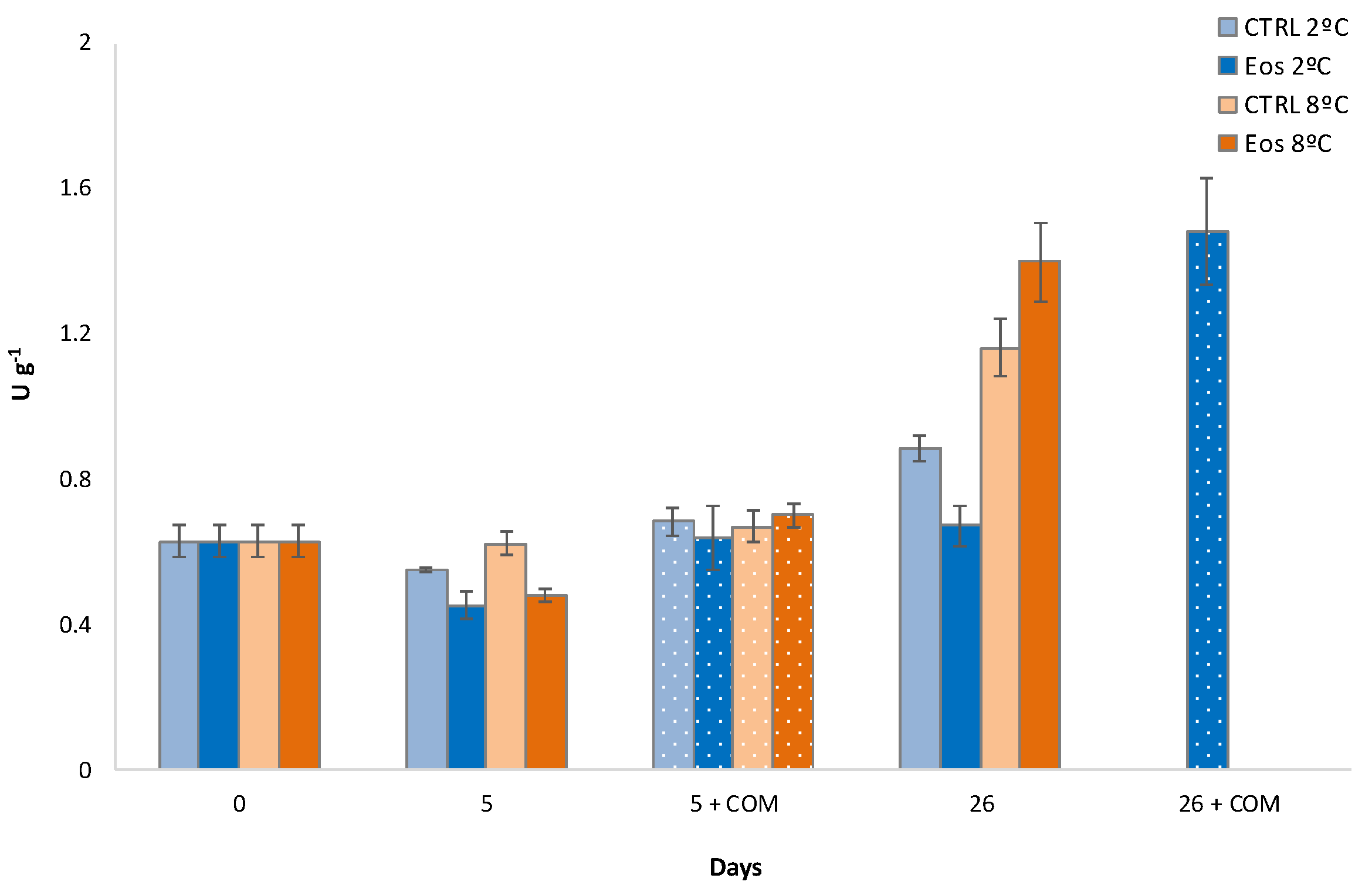
3.6. Polyphenoloxidase Activity and Phenolic Content
3.7. Total Antioxidant Capacity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lurie, S.; Crisosto, C.H. Chilling injury in peach and nectarine. Postharvest Biol. Technol. 2005, 37, 195–208. [Google Scholar] [CrossRef]
- Fernández-Trujillo, J.P.; Cano, A.; Artés, F. Physiological changes in peaches related to chilling injury and ripening. Postharvest Biol. Technol. 1998, 13, 109–119. [Google Scholar] [CrossRef]
- Redondo, D.; Venturini, M.E.; Oria, R.; Arias, E. Inhibitory effect of microwaved thinned nectarine extracts on polyphenol oxidase activity. Food Chem. 2016, 197, 603–610. [Google Scholar] [CrossRef] [PubMed]
- Domingo-Cebrián, R. Proyecto Fin de Carrera Study of Post-Harvest Treatments with 1-methylcyclopropene to Increase the Shelf Life and Quality of Sweet Cab Paraguayos and Platerinas. Bachelor’s Thesis, Universidad de Zaragoza, Zaragoza, Spain, 2014. [Google Scholar]
- Redondo, D.; Gimeno, D.; Calvo, H.; Venturini, M.E.; Oria, R.; Arias, E. Antioxidant Activity and Phenol Content in Different Tissues of Stone Fruits at Thinning and at Commercial Maturity Stages. Waste Biomass Valorization 2020, 1, 3. [Google Scholar] [CrossRef]
- Dorman, H.J.D.; Deans, S.G. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Buendía−Moreno, L.; Soto−Jover, S.; Ros−Chumillas, M.; Antolinos−López, V.; Navarro−Segura, L.; Sánchez−Martínez, M.J.; Martínez−Hernández, G.B.; López−Gómez, A. An innovative active cardboard box for bulk packaging of fresh bell pepper. Postharvest Biol. Technol. 2020, 164, 111171. [Google Scholar] [CrossRef]
- López-Gómez, A.; Boluda-Aguilar, M.; Soto-Jover, S. The use of refrigerated storage, pretreatment with vapors of essential oils, and active flow-packing, improves the shelf life and safety of fresh dill. In Proceedings of the 24th IIR International Congress of Refrigeration, Yokohama, Japan, 16–22 August 2015; pp. 4554–4560. [Google Scholar]
- Zaika, L.L. Spices and herbs: Their antimicrobial activity and its determination. J. Food Saf. 2007, 9, 97–118. [Google Scholar] [CrossRef]
- Rao, J.; Chen, B.; McClements, D.J. Improving the efficacy of essential oils as antimicrobials in foods: Mechanisms of action. Annu. Rev. Food Sci. Technol. 2019, 10, 365–387. [Google Scholar] [CrossRef]
- Chen, X.; Ren, L.; Li, M.; Qian, J.; Fan, J.; Du, B. Effects of clove essential oil and eugenol on quality and browning control of fresh-cut lettuce. Food Chem. 2017, 214, 432–439. [Google Scholar] [CrossRef]
- Rabbany, A.B.M.G.; Mizutani, F. Effect of essential oils on ethylene production and ACC content in apple fruit and peach seed tissues. J. Jpn. Soc. Hortic. Sci. 1996, 65, 7–13. [Google Scholar] [CrossRef]
- Houben, M.; Van de Poel, B. 1-aminocyclopropane-1-carboxylic acid oxidase (ACO): The enzyme that makes the plant hormone ethylene. Front. Plant Sci. 2019, 10, 695. [Google Scholar] [CrossRef] [PubMed]
- Artés-Hernández, F.; Martínez-Hernández, G.B.; Aguayo, E.; Gómez, P.A.; Artés, F. Fresh-cut fruit and vegetables: Emerging eco-friendly techniques for sanitation and preserving safety. In Postharvest Handling; Kahramanoglu, I., Ed.; InTech: London, UK, 2017; pp. 7–45. [Google Scholar]
- López-Gómez, A.; Fernández, P.S.; Palop, A.; Periago, P.M.; Martinez-López, A.; Marin-Iniesta, F.; Barbosa-Cánovas, G.V. Food safety engineering: An emergent perspective. Food Eng. Rev. 2009, 1, 84–104. [Google Scholar] [CrossRef]
- Khaneghah, A.M.; Hashemi, S.M.B.; Limbo, S. Antimicrobial agents and packaging systems in antimicrobial active food packaging: An overview of approaches and interactions. Food Bioprod. Process. 2018, 111, 1–19. [Google Scholar] [CrossRef]
- López-Gómez, A.; Ros-Chumillas, M.; Buendía-Moreno, L.; Navarro-Segura, L.; Martínez-Hernández, G.B. Active cardboard box with smart internal lining based on encapsulated essential oils for enhancing the shelf life of fresh mandarins. Foods 2020, 9, 590. [Google Scholar] [CrossRef]
- Buendía−Moreno, L.; Ros-Chumillas, M.; Navarro-Segura, L.; Sánchez-Martínez, M.J.; Soto-Jover, S.; Antolinos, V.; Martínez-Hernández, G.B.; López-Gómez, A. Effects of an active cardboard box using encapsulated essential oils on the tomato shelf life. Food Bioprocess Technol. 2019, 12, 1548–1558. [Google Scholar] [CrossRef]
- López-Gómez, A.; Ros-Chumillas, M.; Buendía-Moreno, L.; Martínez-Hernández, G.B. Active cardboard packaging with encapsulated essential oils for enhancing the shelf life of fruit and vegetables. Front. Nutr. 2020, 7, 275. [Google Scholar] [CrossRef]
- Kotronia, M.; Kavetsou, E.; Loupassaki, S.; Kikionis, S.; Vouyiouka, S.; Detsi, A. Encapsulation of oregano (Origanum onites L.) essential oil in β-Cyclodextrin (β-CD): Synthesis and characterization of the inclusion complexes. Bioengineering 2017, 4, 74. [Google Scholar] [CrossRef]
- Buendía−Moreno, L.; Sánchez−Martínez, M.J.; Antolinos, V.; Ros−Chumillas, M.; Navarro−Segura, L.; Soto−Jover, S.; Martínez−Hernández, G.B.; López−Gómez, A. Active cardboard box with a coating including essential oils entrapped within cyclodextrins and/or halloysite nanotubes. A case study for fresh tomato storage. Food Control 2020, 107, 106763. [Google Scholar] [CrossRef]
- López-Gómez, A.; Ros-Chumillas, M.; Antolinos, V.; Buendía-Moreno, L.; Navarro-Segura, L.; Sánchez-Martínez, M.J.; Martínez-Hernández, G.B.; Soto-Jover, S. Fresh culinary herbs decontamination with essential oil vapours applied under vacuum conditions. Postharvest Biol. Technol. 2019, 156, 110942. [Google Scholar] [CrossRef]
- Manolikar, M.; Sawant, M. Study of solubility of isoproturon by its complexation with β-cyclodextrin. Chemosphere 2003, 51, 811–816. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Artés-Hernández, F.; Gómez, P.A.; Bretó, J.; Orihuel-Iranzo, B.; Artés, F. Postharvest treatments to control physiological and pathological disorders in lemon fruit. Food Packag. Shelf Life 2017, 14. [Google Scholar] [CrossRef]
- Álvarez-Hernández, M.H.; Martínez-Hernández, G.B.; Avalos-Belmontes, F.; Miranda-Molina, F.D.; Artés-Hernández, F. Postharvest quality retention of apricots by using a novel sepiolite–loaded potassium permanganate ethylene scavenger. Postharvest Biol. Technol. 2020, 160, 111061. [Google Scholar] [CrossRef]
- Carreño, J.; Martínez, A.; Almela, L.; Fernández-López, J.A. Proposal of an index for the objective evaluation of the colour of red table grapes. Food Res. Int. 1995, 28, 373–377. [Google Scholar] [CrossRef]
- Concepcion, M.; Lizada, M.C.; Yang, S.F. A simple and sensitive assay for 1-aminocyclopropane-1-carboxylic acid. Anal. Biochem. 1979, 100, 140–145. [Google Scholar] [CrossRef]
- Bulens, I.; Van de Poel, B.; Hertog, M.L.A.T.M.; De Proft, M.P.; Geeraerd, A.H.; Nicolaï, B.M. Protocol: An updated integrated methodology for analysis of metabolites and enzyme activities of ethylene biosynthesis. Plant Methods 2011, 7, 17. [Google Scholar] [CrossRef]
- Kato, M.; Hyodo, H. Purification and characterization of ACC oxidase and increase in its activity during ripening of pear fruit. J. Jpn. Soc. Hortic. Sci. 1999, 68, 551–557. [Google Scholar] [CrossRef]
- Pressey, R.; Avants, J.K. Difference in polygalacturonase composition of clingstone and freestone peaches. J. Food Sci. 1978, 43, 1415–1417. [Google Scholar] [CrossRef]
- Bach, E.; Schollmeyer, E. An ultraviolet-spectrophotometric method with 2-cyanoacetamide for the determination of the enzymatic degradation of reducing polysaccharides. Anal. Biochem. 1992, 203, 335–339. [Google Scholar] [CrossRef]
- Merck Enzymatic Assay of Polyphenol Oxidase. Available online: https://www.sigmaaldrich.com/content/dam/sigma-aldrich/docs/Sigma/General_Information/polyphenol_oxidase.pdf (accessed on 18 November 2020).
- Antolinos, V.; Sánchez-Martínez, M.J.; Maestre-Valero, J.F.; López-Gómez, A.; Martínez-Hernández, G.B. Effects of Irrigation with Desalinated Seawater and Hydroponic System on Tomato Quality. Water 2020, 12, 518. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Blanco, V.; Blaya-Ros, P.J.; Torres-Sánchez, R.; Domingo, R.; Artés-Hernández, F. Effects of UV–C on bioactive compounds and quality changes during shelf life of sweet cherry grown under conventional or regulated deficit irrigation. Sci. Hortic. (Amst.) 2020, 269. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Gil, M.I.; Cremin, P.; Waterhouse, A.L.; Hess-Pierce, B.; Kader, A.A. HPLC-DAD-ESIMS analysis of phenolic compounds in nectarines, peaches, and plums. J. Agric. Food Chem. 2001, 49, 4748–4760. [Google Scholar] [CrossRef] [PubMed]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Pretel, M.T.; Martinez-Madrid, M.C.; Serrano, M.; Martinez-Reina, G.; Romojaro, F. Physical-Chemical and physiological changes during the ripening of paraguayo (Prunus persica, L.). Acta Hortic. 1998, 463, 399–404. [Google Scholar] [CrossRef]
- Álvarez-Hernández, M.H.; Martínez-Hernández, G.B.; Avalos-Belmontes, F.; Rodríguez-Hernández, A.M.; Castillo-Campohermoso, M.A.; Artés-Hernández, F. An innovative ethylene scrubber made of potassium permanganate loaded on a protonated montmorillonite: A case study on blueberries. Food Bioprocess Technol. 2019, 12, 524–538. [Google Scholar] [CrossRef]
- Barbosa, L.d.N.; Carciofi, B.A.M.; Dannenhauer, C.É.; Monteiro, A.R. Influence of temperature on the respiration rate of minimally processed organic carrots (Daucus Carota L. cv. Brasília). Food Sci. Technol. 2011, 31, 78–85. [Google Scholar] [CrossRef]
- Crisosto, C.H.; Mitchell, F.G.; Ju, Z. Susceptibility to chilling injury of peach, nectarine, and plum cultivars grown in California. Hortscience 1999, 34, 11–16. [Google Scholar] [CrossRef]
- Fan, X.; Argenta, L.; Mattheis, J.P. Interactive effects of 1-MCP and temperature on “Elberta” peach quality. HortScience 2002, 37, 134–138. [Google Scholar] [CrossRef]
- Zapata, P.J.; Castillo, S.; Valero, D.; Guillén, F.; Serrano, M.; Díaz-Mula, H.M. The use of alginate as edible coating alone or in combination with essential oils maintained postharvest quality of tomato. Acta Hortic. 2010, 877, 1529–1534. [Google Scholar] [CrossRef]
- Valverde, J.M.; Guillén, F.; Martínez-Romero, D.; Castillo, S.; Serrano, M.; Valero, D. Improvement of table grapes quality and safety by the combination of modified atmosphere packaging (MAP) and eugenol, menthol, or thymol. J. Agric. Food Chem. 2005, 53, 7458–7464. [Google Scholar] [CrossRef]
- Martínez-Romero, D.; Guillén, F.; Valverde, J.M.; Bailén, G.; Zapata, P.; Serrano, M.; Castillo, S.; Valero, D. Influence of carvacrol on survival of Botrytis cinerea inoculated in table grapes. Int. J. Food Microbiol. 2007, 115, 144–148. [Google Scholar] [CrossRef]
- Hoffman, N.E.; Yang, S.F.; McKeon, T. Identification of 1-(malonylamino)cyclopropane-1-carboxylic acid as a major conjugate of 1-aminocyclopropane-1-carboxylic acid, an ethylene precursor in higher plants. Biochem. Biophys. Res. Commun. 1982, 104, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Brecht, J.; Kader, A. Regulation of ethylene production by ripening nectarine fruit as influenced by ethylene and low temperature. J. Am. Soc. Hortic. Sci. 1984, 109, 869–872. [Google Scholar]
- Yang, S.F.; Hoffman, N.E. Ethylene biosynthesis and its regulation in higher plants. Annu. Rev. Plant Physiol. 1984, 35, 155–189. [Google Scholar] [CrossRef]
- Wu, X.; Yu, M.; Huan, C.; Ma, R.; Yu, Z. Regulation of the protein and gene expressions of ethylene biosynthesis enzymes under different temperature during peach fruit ripening. Acta Physiol. Plant. 2018, 40, 52. [Google Scholar] [CrossRef]
- Teng, Y.; Murtaza, A.; Iqbal, A.; Fu, J.; Ali, S.W.; Iqbal, M.A.; Xu, X.; Pan, S.; Hu, W. Eugenol emulsions affect the browning processes, and microbial and chemical qualities of fresh-cut Chinese water chestnut. Food Biosci. 2020, 38, 100716. [Google Scholar] [CrossRef]
- Esti, M.; Messia, M.C.; Sinesio, F.; Nicotra, A.; Conte, L.; la Notte, E.; Palleschi, G. Quality evaluation of peaches and nectarines by electrochemical and multivariate analyses: Relationships between analytical measurements and sensory attributes. Food Chem. (U. K.) 1997, 60, 659–666. [Google Scholar] [CrossRef]
- Colaric, M.; Veberic, R.; Stampar, F.; Hudina, M. Evaluation of peach and nectarine fruit quality and correlations between sensory and chemical attributes. J. Sci. Food Agric. 2005, 85, 2611–2616. [Google Scholar] [CrossRef]
- Manganaris, G.A.; Vicente, A.R.; Crisosto, C.H.; Labavitch, J.M. Cell wall modifications in chilling-injured plum fruit (Prunus salicina). Postharvest Biol. Technol. 2008, 48, 77–83. [Google Scholar] [CrossRef]
- Ogundiwin, E.A.; Peace, C.P.; Gradziel, T.M.; Dandekar, A.M.; Bliss, F.A.; Crisosto, C.H. Molecular genetic dissection of chilling injury in peach fruit. Acta Hortic. 2007, 633–638. [Google Scholar] [CrossRef]
- Zhu, P.; Xu, L.; Zhang, C.; Toyoda, H.; Gan, S.-S. Ethylene produced by Botrytis cinerea can affect early fungal development and can be used as a marker for infection during storage of grapes. Postharvest Biol. Technol. 2012, 66, 23–29. [Google Scholar] [CrossRef]
- Wadhwa, G.; Kumar, S.; Chhabra, L.; Mahant, S.; Rao, R. Essential oil–cyclodextrin complexes: An updated review. J. Incl. Phenom. Macrocycl. Chem. 2017, 89, 39–58. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; Andrade, M.; Sanches-Silva, A. Application of encapsulated essential oils as antimicrobial agents in food packaging. Curr. Opin. Food Sci. 2017, 14, 78–84. [Google Scholar] [CrossRef]
- Zanetti, M.; Carniel, T.K.; Dalcanton, F.; dos Anjos, R.S.; Riella, H.G.; de Araújo, P.H.H.; de Oliveira, D.; Fiori, M.A. Use of encapsulated natural compounds as antimicrobial additives in food packaging: A brief review. Trends Food Sci. Technol. 2018, 81, 51–60. [Google Scholar] [CrossRef]
- D’hallewin, G.; Arras, G.; Castia, T.; Piga, A. Reducing decay of avana mandarin fruit by the use of uv, heat and thiabendazole treatments. Acta Hortic. 1994, 387–394. [Google Scholar] [CrossRef]
- Thompson, J.; Mitchell, F.G.; Kasmire, R.F. Cooling horticultural commodities. In Postharvest Technology of Horticultural Crops; University of California, Agriculture and Natural Resources: Davis, CA, USA, 2002; pp. 111–130. ISBN 9781879906518. [Google Scholar]
- Zhang, K.; Deng, Y.; Fu, H.; Weng, Q. Effects of Co-60 gamma-irradiation and refrigerated storage on the quality of Shatang mandarin. Food Sci. Hum. Wellness 2014, 3, 9–15. [Google Scholar] [CrossRef]
- Santoro, K.; Maghenzani, M.; Chiabrando, V.; Bosio, P.; Gullino, M.; Spadaro, D.; Giacalone, G. Thyme and Savory Essential Oil Vapor Treatments Control Brown Rot and Improve the Storage Quality of Peaches and Nectarines, but Could Favor Gray Mold. Foods 2018, 7, 7. [Google Scholar] [CrossRef]
- Serrano, M.; Martínez-Romero, D.; Castillo, S.; Guillén, F.; Valero, D. The use of natural antifungal compounds improves the beneficial effect of MAP in sweet cherry storage. Innov. Food Sci. Emerg. Technol. 2005, 6, 115–123. [Google Scholar] [CrossRef]
- Izzo, R.; Scartazza, A.; Masia, A.; Galleschi, L.; Quartacci, M.F.; Navari-Izzo, F. Lipid evolution during development and ripening of peach fruits. Phytochemistry 1995, 39, 1329–1334. [Google Scholar] [CrossRef]
- Maalekuu, K.; Elkind, Y.; Leikin-Frenkel, A.; Lurie, S. The relationship between water loss, lipid content, membrane integrity and LOX activity in ripe pepper fruit after storage. Postharvest Biol. Technol. 2006, 42, 248–255. [Google Scholar] [CrossRef]
- Luza, J.G.; van Gorsel, R.; Polito, V.S.; Kader, A.A. Chilling Injury in Peaches: A Cytochemical and Ultrastructural Cell Wall Study. J. Am. Soc. Hortic. Sci. 2019, 117, 114–118. [Google Scholar] [CrossRef]
- Buendía−Moreno, L.; Soto−Jover, S.; Ros−Chumillas, M.; Antolinos, V.; Navarro−Segura, L.; Sánchez−Martínez, M.J.; Martínez−Hernández, G.B.; López−Gómez, A. Innovative cardboard active packaging with a coating including encapsulated essential oils to extend cherry tomato shelf life. LWT Food Sci. Technol. 2019, 116, 108584. [Google Scholar] [CrossRef]
- Tonutti, P.; Bonghi, C.; Ramina, A. Biochemical and molecular aspects of peach fruit ripening. In Proceedings of the COST-94 Quality Criteria; Hribar, J., Johnson, D.S., Böhling, H., De la Plaza, J., Haffner, K.E., Höhn, E., Somogyi, Z., Eds.; European Commission: Bled, Slovenia, 1994; pp. 101–104. [Google Scholar]
- Gavaric, N.; Mozina, S.S.; Kladar, N.; Bozin, B. Chemical Profile, Antioxidant and Antibacterial Activity of Thyme and Oregano Essential Oils, Thymol and Carvacrol and Their Possible Synergism. J. Essent. Oil Bear. Plants 2015, 18, 1013–1021. [Google Scholar] [CrossRef]
- Cisneros-Zevallos, L. The use of controlled postharvest abiotic stresses as a tool for enhancing the nutraceutical content and adding-value of fresh fruits and vegetables. J. Food Sci. 2003, 68, 1560–1565. [Google Scholar] [CrossRef]
- Redondo, D.; Arias, E.; Oria, R.; Venturini, M.E. Thinned stone fruits are a source of polyphenols and antioxidant compounds. J. Sci. Food Agric. 2017, 97, 902–910. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Rangel, J.C.; Benavides, J.; Heredia, J.B.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. The Folin-Ciocalteu assay revisited: Improvement of its specificity for total phenolic content determination. Anal. Methods 2013, 5, 5990–5999. [Google Scholar] [CrossRef]
| Single Factors and Interactions | |||||||
|---|---|---|---|---|---|---|---|
| Storage Temperature (A) | Package Treatment (B) | Storage Time (C) | A × B | A × C | B × C | A × B × C | |
| Respiration processes | |||||||
| Ethylene production | ns | 2.7 † | 5.7 ‡ | 3.9 † | 8.0 ‡ | ns | ns |
| CO2 production | 7.1 ‡ | 7.1 ‡ | 11.3 ‡ | ns | 15.9 ‡ | 12.1 † | ns |
| ACC | 0.11 ‡ | 0.11 ‡ | 0.14 ‡ | ns | 0.19 ‡ | 0.19 ‡ | 0.27 ‡ |
| MACC | 0.11 ‡ | 0.08 † | 0.14 ‡ | ns | 0.20 ‡ | 0.14 ‡ | 0.28 ‡ |
| ACO | 0.006 ‡ | 0.006 ‡ | 0.008 ‡ | 0.009 ‡ | 0.011 ‡ | 0.011 ‡ | 0.011 † |
| Physicochemical quality | |||||||
| Weight loss | 0.3 ‡ | 0.3 ‡ | 0.3 ‡ | 0.4 ‡ | 0.4 ‡ | 0.4 ‡ | 0.6 ‡ |
| Firmness | 1.2 ‡ | 1.2 ‡ | 1.5 ‡ | ns | 2.1 ‡ | 1.2 * | ns |
| Soluble solids | ns | ns | ns | ns | ns | ns | ns |
| pH | ns | 0.05 † | 0.08 ‡ | ns | 0.09 † | 0.12 ‡ | ns |
| Titratable acidity | ns | ns | 0.4 ‡ | ns | 0.5 † | ns | ns |
| Color index | ns | 0.2 ‡ | 0.3 ‡ | ns | ns | 0.4 ‡ | ns |
| Enzymes | |||||||
| Endo-PG | 0.09 * | 0.09 * | 0.20 ‡ | ns | 0.16 * | ns | ns |
| Exo-PG | 3.0 ‡ | 3.0 ‡ | 3.7 ‡ | ns | 5.3 ‡ | 5.3 ‡ | ns |
| PPO | 0.06 ‡ | ns | 0.08 ‡ | 0.09 ‡ | 0.11 ‡ | 0.08 † | 0.16 ‡ |
| Phenolic contents | |||||||
| Total phenolic content | 7.5 † | ns | 6.8 * | ns | 9.6 * | 9.6 * | ns |
| Sum individual phenolics | 9.1 ‡ | 6.8 † | 11.1 ‡ | 7.1 * | 15.7 ‡ | 15.7 ‡ | 16.6 ‡ |
| Cyanidin-3-glucoside | 2.7 ‡ | 2.7 ‡ | 3.3 ‡ | 2.9 † | 3.5 † | 4.7 ‡ | 6.6 ‡ |
| Cyanidin-3-rutinoside | ns | 0.13 * | 0.30 ‡ | 0.34 ‡ | ns | 0.42 ‡ | 0.59 ‡ |
| Catechin | ns | ns | ns | ns | 11.5 ‡ | ns | ns |
| Neochlorogenic acid | 1.51 ‡ | 0.83 * | 1.85 ‡ | 2.14 ‡ | 2.62 ‡ | 2.62 ‡ | 3.71 ‡ |
| Chlorogenic acid | 1.62 ‡ | 1.62 ‡ | 1.98 ‡ | 2.29 ‡ | 2.80 ‡ | 2.80 ‡ | 3.96 ‡ |
| Total antioxidant capacity | 45.1 * | ns | 55.2 † | ns | 57.6 * | ns | 147.7 ‡ |
| Storage Time | Storage Temperature | Packaging Treatment | SSC | TA | pH | Color Index | Weight Loss | Firmness | |
|---|---|---|---|---|---|---|---|---|---|
| Processing day | 12.0 ± 0.1 | 4.2 ± 0.2 | 4.19 ± 0.03 | 2.1 ± 0.1 | - | 25.9 ± 1.7 | |||
| 5 days | 2 °C | CTRL | 12.2 ± 0.6 | 4.0 ± 0.2 | 4.22 ± 0.02 | 2.7 ± 0.1 | 0.53 ± 0.09 | 14.9 ± 1.2 | |
| 2 °C | EOs | 12.2 ± 0.6 | 4.0 ± 0.2 | 4.27 ± 0.02 | 2.3 ± 0.2 | 0.26 ± 0.05 | 17.4 ± 1.7 | ||
| 8 °C | CTRL | 12.7 ± 0.6 | 3.8 ± 0.3 | 4.29 ± 0.03 | 2.7 ± 0.3 | 0.09 ± 0.13 | 13.4 ± 0.8 | ||
| 8 °C | EOs | 12.1 ± 0.3 | 3.5 ± 0.1 | 4.35 ± 0.02 | 2.0 ± 0.3 | 0.04 ± 0.01 | 16.2 ± 0.4 | ||
| 5 days + COM | 2 °C | CTRL | 12.2 ± 0.1 | 3.8 ± 0.1 | 4.25 ± 0.02 | 2.4 ± 0.1 | 2.65 ± 0.18 | 13.0 ± 1.8 | |
| 2 °C | EOs | 12.2 ± 0.1 | 3.8 ± 0.1 | 4.29 ± 0.01 | 2.5 ± 0.1 | 1.10 ± 0.26 | 13.8 ± 1.8 | ||
| 8 °C | CTRL | 12.5 ± 0.8 | 3.7 ± 0.1 | 4.37 ± 0.04 | 2.2 ± 0.3 | 2.61 ± 0.04 | 10.4 ± 1.3 | ||
| 8 °C | EOs | 12.1 ± 0.5 | 3.1 ± 0.2 | 4.39 ± 0.02 | 2.3 ± 0.1 | 1.04 ± 0.05 | 12.1 ± 0.7 | ||
| 26 days | 2 °C | CTRL | 12.3 ± 0.3 | 1.7 ± 0.1 | 4.96 ± 0.04 | 2.7 ± 0.2 | 3.94 ± 0.14 | 10.8 ± 1.4 | |
| 2 °C | EOs | 12.3 ± 0.2 | 2.0 ± 0.1 | 4.73 ± 0.02 | 2.0 ± 0.1 | 1.28 ± 0.39 | 12.1 ± 0.9 | ||
| 8 °C | CTRL | 12.7 ± 1.0 | 2.1 ± 0.1 | 4.88 ± 0.07 | 2.9 ± 0.1 | 5.52 ± 0.16 | 5.3 ± 1.1 | ||
| 8 °C | EOs | 12.4 ± 0.6 | 2.6 ± 0.2 | 4.66 ± 0.14 | 2.0 ± 0.1 | 1.04 ± 0.12 | 6.8 ± 1.1 | ||
| 26 days + COM | 2 °C | CTRL | 12.6 ± 0.1 | 2.0 ± 0.1 | 5.08 ± 0.01 | 3.1 ± 0.3 | 6.54 ± 0.29 | 6.8 ± 0.3 | |
| 2 °C | EOs | 12.5 ± 0.3 | 2.2 ± 0.2 | 4.88 ± 0.05 | 2.0 ± 0.1 | 1.86 ± 0.08 | 8.3 ± 1.1 | ||
| 8 °C | CTRL | - | - | - | - | - | - | ||
| 8 °C | EOs | 12.7 ± 1.0 | 2.4 ± 0.1 | 4.69 ± 0.02 | 1.9 ± 0.4 | 5.17 ± 0.11 | - | ||
| Storage Time | Temperature | Packaging Treatment | TPC | Cy-3-GLC | Cy-3-RUT | Catechin | 3-CQA | 5-CQA | Sum | TAC |
|---|---|---|---|---|---|---|---|---|---|---|
| Processing day | 126.1 ± 3.8 | 42.7 ± 1.6 | 6.4 ± 0.1 | 174.1 ± 6.9 | 17.4 ± 0.4 | 22.1 ± 0.2 | 262.6 ± 10.2 | 774.2 ± 20.0 | ||
| 5 days | 2 °C | CTRL | 126.9 ± 0.7 | 37.0 ± 1.6 | 5.3 ± 0.3 | 162.5 ± 9.9 | 28.4 ± 1.9 | 23.9 ± 1.3 | 257.2 ± 14.8 | 736.9 ± 43.0 |
| 2 °C | EOs | 118.3 ± 12.4 | 39.8 ± 4.1 | 6.3 ± 0.1 | 167.6 ± 1.9 | 9.8 ± 0.7 | 9.8 ± 1.3 | 233.2 ± 8.0 | 839.2 ± 58.6 | |
| 8 °C | CTRL | 125.9 ± 12.2 | 42.2 ± 1.5 | 6.3 ± 0.2 | 175.5 ± 3.7 | 10.3 ± 1.8 | 20.5 ± 0.5 | 254.8 ± 7.3 | 760.3 ± 48.7 | |
| 8 °C | EOs | 109.6 ± 8.5 | 22.0 ± 0.8 | 5.7 ± 0.1 | 178.3 ± 4.3 | 12.8 ± 0.7 | 9.2 ± 1.6 | 227.9 ± 6.3 | 607.7 ± 62.5 | |
| 5 days + COM | 2 °C | CTRL | 118.1 ± 9.0 | 57.1 ± 3.3 | 6.6 ± 0.1 | 173.9 ± 1.2 | 5.1 ± 0.8 | 4.7 ± 0.5 | 247.4 ± 11.8 | 572.0 ± 41.9 |
| 2 °C | EOs | 116.3 ± 8.2 | 38.6 ± 1.1 | 5.8 ± 0.1 | 179.7 ± 0.8 | 12.7 ± 0.9 | 7.7 ± 0.6 | 244.6 ± 14.2 | 622.7 ± 24.7 | |
| 8 °C | CTRL | 139.4 ± 0.3 | 58.0 ± 3.8 | 5.5 ± 0.1 | 176.1 ± 2.0 | 9.0 ± 1.9 | 13.2 ± 1.8 | 261.8 ± 8.1 | 753.3 ± 77.5 | |
| 8 °C | EOs | 193.3 ± 14.5 | 67.4 ± 3.1 | 7.6 ± 0.4 | 198.0 ± 7.7 | 20.8 ± 1.6 | 20.9 ± 0.4 | 314.8 ± 4.2 | 1287.1 ± 91.0 | |
| 26 days | 2 °C | CTRL | 128.3 ± 8.9 | 49.1 ± 1.3 | 6.8 ± 0.1 | 178.7 ± 7.7 | 8.9 ± 1.9 | 18.6 ± 2.5 | 262.1 ± 10.5 | 746.3 ± 89.3 |
| 2 °C | EOs | 125.2 ± 10.5 | 39.5 ± 3.4 | 6.1 ± 0.3 | 172.3 ± 8.0 | 12.2 ± 1.1 | 18.4 ± 1.9 | 248.5 ± 7.8 | 710.7 ± 54.7 | |
| 8 °C | CTRL | 97.8 ± 5.6 | 40.4 ± 2.1 | 6.7 ± 0.2 | 163.0 ± 1.2 | 5.3 ± 1.1 | 4.0 ± 1.3 | 219.4 ± 13.6 | 593.7 ± 52.2 | |
| 8 °C | EOs | 119.2 ± 11.1 | 39.3 ± 2.3 | 6.0 ± 0.2 | 167.6 ± 9.0 | 11.5 ± 1.8 | 17.1 ± 1.4 | 241.6 ± 10.8 | 760.6 ± 34.1 | |
| 26 days + COM | 2 °C | CTRL | 88.3 ± 15.5 | 28.9 ± 4.1 | 6.2 ± 0.4 | 168.0 ± 9.4 | 2.3 ± 0.4 | 1.5 ± 0.1 | 207.0 ± 14.3 | 450.7 ± 77.7 |
| 2 °C | EOs | 105.9 ± 13.6 | 21.1 ± 2.4 | 5.5 ± 0.1 | 165.9 ± 10.0 | 6.2 ± 0.5 | 4.3 ± 0.6 | 203.0 ± 2.6 | 480.7 ± 16.3 | |
| 8 °C | CTRL | - | - | - | - | - | - | - | - | |
| 8 °C | EOs | 139.1 ± 4.1 | 13.9 ± 0.1 | 5.2 ± 0.1 | 185.6 ± 5.0 | 18.4 ± 0.5 | 16.6 ± 0.4 | 239.7 ± 10.2 | 915.2 ± 22.0 | |
| TAC corelations | 0.86 | 0.47 | 0.17 | 0.50 | 0.37 | 0.41 | 0.69 | - | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Gómez, A.; Navarro-Martínez, A.; Martínez-Hernández, G.B. Active Paper Sheets Including Nanoencapsulated Essential Oils: A Green Packaging Technique to Control Ethylene Production and Maintain Quality in Fresh Horticultural Products—A Case Study on Flat Peaches. Foods 2020, 9, 1904. https://doi.org/10.3390/foods9121904
López-Gómez A, Navarro-Martínez A, Martínez-Hernández GB. Active Paper Sheets Including Nanoencapsulated Essential Oils: A Green Packaging Technique to Control Ethylene Production and Maintain Quality in Fresh Horticultural Products—A Case Study on Flat Peaches. Foods. 2020; 9(12):1904. https://doi.org/10.3390/foods9121904
Chicago/Turabian StyleLópez-Gómez, Antonio, Alejandra Navarro-Martínez, and Ginés Benito Martínez-Hernández. 2020. "Active Paper Sheets Including Nanoencapsulated Essential Oils: A Green Packaging Technique to Control Ethylene Production and Maintain Quality in Fresh Horticultural Products—A Case Study on Flat Peaches" Foods 9, no. 12: 1904. https://doi.org/10.3390/foods9121904
APA StyleLópez-Gómez, A., Navarro-Martínez, A., & Martínez-Hernández, G. B. (2020). Active Paper Sheets Including Nanoencapsulated Essential Oils: A Green Packaging Technique to Control Ethylene Production and Maintain Quality in Fresh Horticultural Products—A Case Study on Flat Peaches. Foods, 9(12), 1904. https://doi.org/10.3390/foods9121904







