A Revisit to the Formation and Mitigation of 3-Chloropropane-1,2-Diol in Palm Oil Production
Abstract
1. Introduction
2. 3-MCPD in Vegetable Oils
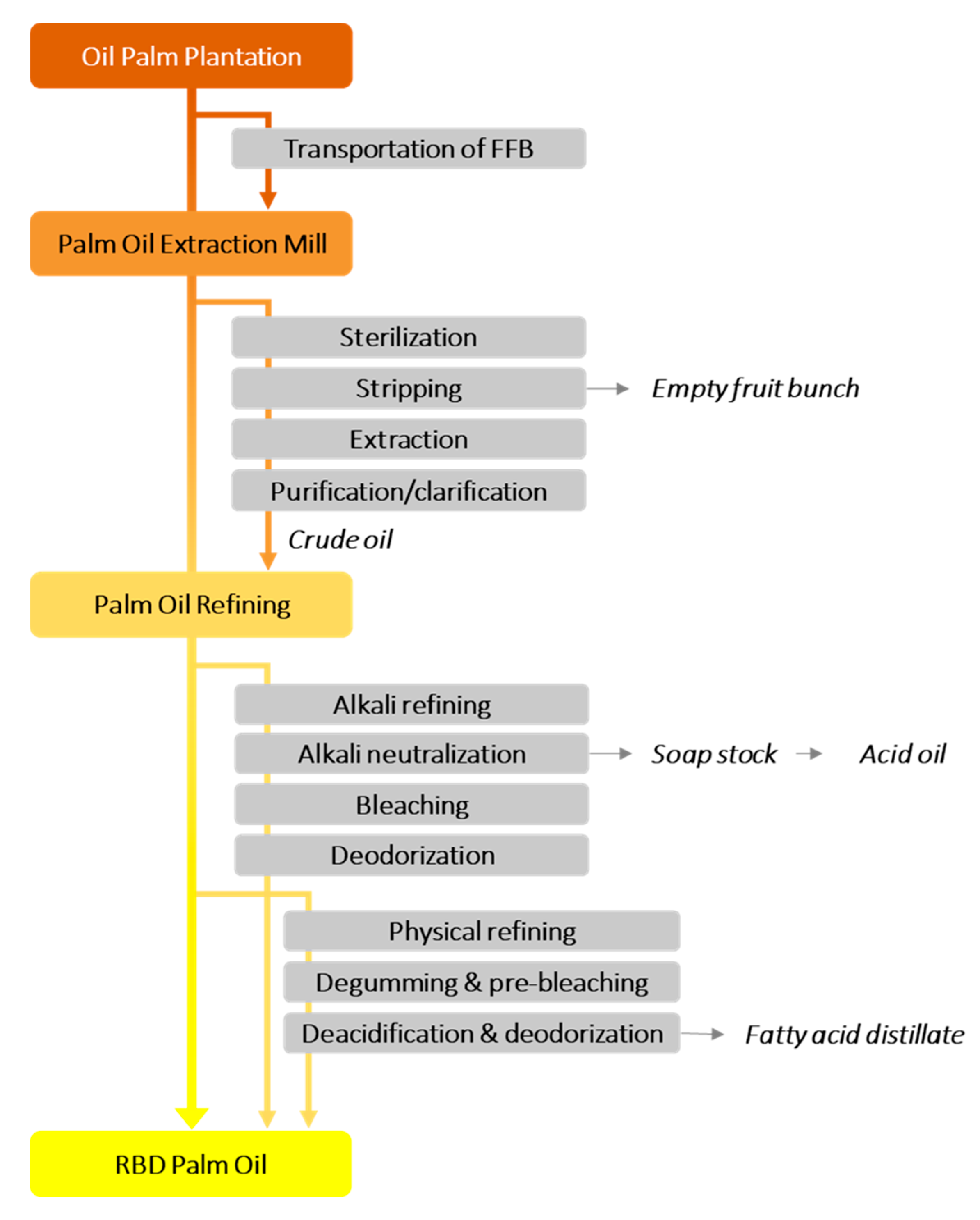
2.1. General Extraction Process
2.2. Occurrence and Formation
3. Trigger Factor in 3-MCPD Formation
3.1. Precursors
3.1.1. Chlorine
3.1.2. Acylglycerols
3.2. Refining Process
3.3. Other Factors
4. Measurement
4.1. Analytical Measurement
4.1.1. Direct Methods
4.1.2. Indirect Methods
5. Mitigation
5.1. Plantation Management
5.2. Extraction and Refining Process Modification
5.3. Post Refining Process
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jedrkiewicz, R.; Głowacz, A.; Gromadzka, J.; Namieśnik, J. Determination of 3-MCPD and 2-MCPD esters in edible oils, fish oils and lipid fractions of margarines available on Polish market. Food Control 2016, 59, 487–492. [Google Scholar] [CrossRef]
- Svejkovska, B.; Novotny, O.; Divinova, V.; Reblova, Z.; Dolezal, M.; Velisek, J. Esters of 3-chloropropane-1,2-diol in foodstuffs. Czech. J. Food Sci. 2004, 22, 190–196. [Google Scholar] [CrossRef]
- Nyman, P.J.; Diachenko, G.W.; Perfetti, G.A. Survey of chloropropanols in soy sauces and related products. Food Addit. Contam. 2003, 20, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Hamlet, C.G.; Sadd, P.A.; Crews, C.; Velíšek, J.; Baxter, D.E. Occurrence of 3-chloro-propane-1, 2-diol (3-MCPD) and related compounds in foods: A review. Food Addit. Contam. 2002, 19, 619–631. [Google Scholar] [CrossRef]
- Velíšek, J.; Davidek, J.; Hajšlová, J.; Kubelka, V.; Janíček, G.; Mánková, B. Chlorohydrins in protein hydrolysates. Z. Für Lebensm. Unters. Und Forsch. 1978, 167, 241–244. [Google Scholar]
- Davidek, J.; Velíšek, J.; Kubelka, V.; Janíček, G.; Šimicová, Z. Glycerol chlorohydrins and their esters as products of the hydrolysis of tripalmitin, tristearin and triolein with hydrochloric acid. Z. Für Lebensm. Unters. Und Forsch. 1980, 171, 14–17. [Google Scholar] [CrossRef]
- Becalski, A.; Feng, S.; Lau, B.P.-Y.; Zhao, T. A pilot survey of 2- and 3-monochloropropanediol and glycidol fatty acid esters in foods on the Canadian market. J. Food Compos. Anal. 2015, 37, 58–66. [Google Scholar] [CrossRef]
- Chung, H.Y.; Chung, S.W.; Chan, B.T.P.; Ho, Y.Y.; Xiao, Y. Dietary exposure of Hong Kong adults to fatty acid esters of 3-monochloropropane-1, 2-diol. Food Addit. Contam. Part A 2013, 30, 1508–1512. [Google Scholar] [CrossRef]
- Custodio-Mendoza, J.A.; Carro, A.M.; Lage-Yusty, M.A.; Herrero, A.; Valente, I.M.; Rodrigues, J.A.; Lorenzo, R.A. Occurrence and exposure of 3-monochloropropanediol diesters in edible oils and oil-based foodstuffs from the Spanish market. Food Chem. 2019, 270, 214–222. [Google Scholar] [CrossRef]
- Divinová, V.; Svejkovská, B.; Novotny, O.; Velisek, J. Survey of 3-chloropropane-1, 2-diol and its precursors in foods in the Czech Republic. Czech. J. Food Sci. 2004, 22, 267. [Google Scholar] [CrossRef]
- Leigh, J.; Macmahon, S. Occurrence of 3-monochloropropanediol esters and glycidyl esters in commercial infant formulas in the United States. Food Addit. Contam. Part A 2017, 34, 356–370. [Google Scholar] [CrossRef] [PubMed]
- Pavesi, A.; Marcolino, A.P.F.C.; Scaranelo, A.C.A.G.R. Contamination of Fried Foods by 3-Monochloropropane-1,2-diol Fatty Acid Esters During Frying. J. Am. Oil. Chem. Soc. 2017, 94, 449–455. [Google Scholar]
- Wöhrlin, F.; Fry, H.; Lahrssen-wiederholt, M.; Preiß-Weigert, A. Occurrence of fatty acid esters of 3-MCPD, 2-MCPD and glycidol in infant formula. Food Addit. Contam. Part A 2015, 32, 1810–1822. [Google Scholar] [CrossRef] [PubMed]
- Zelinková, Z.; Dole, M.; Velí, J. Occurrence of 3-chloropropane-1, 2-diol fatty acid esters in infant and baby foods. Eur. Food Res. Technol. 2009, 228, 571–578. [Google Scholar] [CrossRef]
- Zelinková, Z.; Doležal, M.; Velíšek, J. 3-Chloropropane-1, 2-diol fatty acid esters in potato products. Czech. J. Food Sci. 2009, 27, S421–S424. [Google Scholar] [CrossRef]
- Macarthur, R.; Crews, C.; Davies, A.; Brereton, P.; Hough, P.; Harvey, D. 3-Monochloropropane-1,2-diol (3-MCPD) in soy sauces and similar products available from retail outlets in the UK. Food Addit. Contam. Part A 2000, 17, 903–906. [Google Scholar] [CrossRef]
- Seefelder, W.; Varga, N.; Studer, A.; Williamson, G.; Scanlan, F.P.; Stadler, R.H. Esters of 3-chloro-1,2-propanediol (3-MCPD) in vegetable oils: Significance in the formation of 3-MCPD. Food Addit. Contam. Part A 2008, 25, 391–400. [Google Scholar] [CrossRef]
- Wenzl, T.; Lachenmeier, D.W.; Gökmen, V. Analysis of heat-induced contaminants (acrylamide, chloropropanols and furan) in carbohydrate-rich food. Anal. Bioanal. Chem. 2007, 389, 119. [Google Scholar] [CrossRef]
- Kuhlmann, J. Analysis and occurrence of dichloropropanol fatty acid esters and related process-induced contaminants in edible oils and fats. Eur. J. Lipid Sci. Tech. 2016, 118, 382–395. [Google Scholar] [CrossRef]
- Cho, W.; Han, B.S.; Nam, K.T.; Park, K.; Choi, M.; Kim, S.H.; Jeong, J. Carcinogenicity study of 3-monochloropropane-1, 2-diol in Sprague–Dawley rats. Food Chem. Toxicol. 2008, 46, 3172–3177. [Google Scholar] [CrossRef]
- Jackson, H.; Rooney, F.R.; Fitzpatrick, R.W.; Gibson, K.H. Characterization and antifertility activity in rats of S(+)alpha-chlorohydrin. Chem. Biol. Interact. 1977, 17, 117–120. [Google Scholar] [CrossRef]
- Kwack, S.J.; Kim, S.S.; Choi, Y.W.; Rhee, G.S.; Lee, R.D.; Seok, J.H.; Chae, S.Y.; Won, Y.H.; Lim, K.J.; Choi, K.S.; et al. Mechanism of Antifertility in Male Rats Treated with 3-Monochloro-1,2-Propanediol (3-MCPD). J. Toxicol. Environ. 2004, 67, 2001–2004. [Google Scholar] [CrossRef] [PubMed]
- Schilter, B.; Scholz, G.; Seefelder, W. Fatty acid esters of chloropropanols and related compounds in food: Toxicological aspects. Eur. J. Lipid Sci. Technol. 2011, 113, 309–313. [Google Scholar] [CrossRef]
- Liu, M.; Gao, B.Y.; Qin, F.; Wu, P.P.; Shi, H.M.; Luo, W.; Ma, A.N.; Jiang, Y.R.; Xu, X.B.; Yu, L.L. Acute oral toxicity of 3-MCPD mono- and di-palmitic esters in Swiss mice and their cytotoxicity in NRK-52E rat kidney cells. Food Chem. Toxicol. 2012, 50, 3785–3791. [Google Scholar] [CrossRef]
- Liu, M.; Liu, J.; Wu, Y.; Gao, B.; Wu, P.; Shi, H.; Sun, X.; Huang, H.; Wang, T.T.Y.; Yu, L.L. Preparation of five 3-MCPD fatty acid esters, and the effects of their chemical structures on acute oral toxicity in Swiss mice. J. Sci. 2017, 97, 841–848. [Google Scholar] [CrossRef]
- Abraham, K.; Appel, K.E.; Berger-Preiss, E.; Apel, E.; Gerling, S.; Mielke, H.; Creutzenberg, O.; Lampen, A. Relative oral bioavailability of 3-MCPD from 3-MCPD fatty acid esters in rats. Arch. Toxicol. 2013, 87, 649–659. [Google Scholar] [CrossRef]
- Lee, J.K.; Ryu, M.H.; Byun, J.A. Immunotoxic effect of β-chlorolactic acid on murine splenocyte and peritoneal macrophage function in vitro. Toxicology 2005, 210, 175–187. [Google Scholar] [CrossRef]
- Cavanagh, J.B.; Nolan, C.C.; Seville, M.P. The neurotoxicity of a-chlorohydrin in rats and mice: 1. Evolution of the cellular changes. Neuropathol. Appl. Neurobiol. 1993, 19, 240–252. [Google Scholar] [CrossRef]
- Shah, S.U. Importance of Genotoxicity & S2A guidelines for genotoxicity testing for pharmaceuticals. IOSR J. Pharm. Biol. Sci. 2012, 1, 43–54. [Google Scholar]
- Aasa, J.; Törnqvist, M.; Abramsson-Zetterberg, L. Measurement of micronuclei and internal dose in mice demonstrates that 3-monochloropropane-1, 2-diol (3-MCPD) has no genotoxic potency in vivo. Food Chem. Toxicol. 2017, 109, 414–420. [Google Scholar] [CrossRef]
- Robjohns, S.; Marshall, R.; Fellows, M.; Kowalczyk, G. In vivo genotoxicity studies with 3-monochloropropan-1, 2-diol. Mutagenesis 2003, 18, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zheng, C.; Sun, X.; Ouyang, B.; Ni, P.; Zhang, Y. Identification of 3-chloro-1, 2-propandiol using molecularly imprinted composite solid-phase extraction materials. Anal. Bioanal. Chem. 2014, 406, 6319–6327. [Google Scholar] [CrossRef] [PubMed]
- Baer, I.; De La Calle, B.; Taylor, P. 3-MCPD in food other than soy sauce or hydrolysed vegetable protein (HVP). Anal. Bioanal. Chem. 2010, 396, 443–456. [Google Scholar] [CrossRef] [PubMed]
- Grosse, Y.; Baan, R.; Secretan-Lauby, B.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Islami, F.; Galichet, L.; Straif, K.; et al. Carcinogenicity of chemicals in industrial and consumer products, food contaminants and flavourings, and water chlorination byproducts. Lancet Oncol. 2011, 12, 328–329. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain (CONTAM); Knutsen, H.K.; Alexander, J.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Edler, L.; et al. Update of the risk assessment on 3-monochloropropane diol and its fatty acid esters. EFSA J. 2018, 16, e05083. [Google Scholar]
- May, C.Y.; Nesaretnam, K. Research advancements in palm oil nutrition. Eur. J. Lipid. Sci. Tech. 2014, 116, 1301–1315. [Google Scholar] [CrossRef]
- Kamal-Eldin, A. Minor Components of Fats and Oils. In Bailey’s Industrial Oil and Fat Products; Shahidi, F., Ed.; John Wiley & Sons Inc.: Sussex, UK, 2005; pp. 483–523. ISBN 978-0-4713-8460-1. [Google Scholar]
- Brodnjak-Vončina, D.; Kodba, Z.C.; Novič, M. Multivariate data analysis in classification of vegetable oils characterized by the content of fatty acids. Chemom. Intell. Lab. 2005, 75, 31–43. [Google Scholar] [CrossRef]
- Dahlberg, D.B.; Lee, S.M.; Wenger, S.J.; Vargo, J.A. Classification of vegetable oils by FT-IR. Appl. Spectrosc. 1997, 51, 1118–1124. [Google Scholar] [CrossRef]
- Rusak, D.A.; Brown, L.M.; Martin, S.D. Classification of Vegetable Oils by Principal Component Analysis of FTIR Spectra. J. Chem. Educ. 2003, 80, 541–543. [Google Scholar] [CrossRef]
- Mbesse, Y.G.; Ghalila, H.; Boyomo, M.; Ben, Z. Classification of vegetable oils based on their concentration of saturated fatty acids using laser induced breakdown spectroscopy (LIBS). Food Chem. 2014, 147, 327–331. [Google Scholar] [CrossRef]
- Zamora, R.; Gómez, G.; Hidalgo, F.J. Classification of vegetable oils by high-resolution 13C NMR spectroscopy using chromatographically obtained oil fractions. J. Am. Oil Chem. Soc. 2002, 79, 267–272. [Google Scholar] [CrossRef]
- Lerma-García, M.J.; Ramis-Ramos, G.; Herrero-Martínez, J.M.; Simó-Alfonso, E.F. Classification of vegetable oils according to their botanical origin using sterol profiles established by direct infusion mass spectrometry. Rapid Commun. Mass Sp. 2008, 22, 973–978. [Google Scholar] [CrossRef] [PubMed]
- Troya, F. Classification of vegetable oils according to their botanical origin using n-alkane profiles established by GC–MS. Food Chem. 2015, 167, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Estrada, M.T.; Paciulli, M.; Cerretani, L.; Chiavaro, E. Production of vegetable oils from fruits, oilseeds, and beans: Conventional processing and industry techniques. In Edible Oils; Chemat, S., Ed.; CRC Press: Boca Raton, FL, USA, 2017; pp. 1–34. [Google Scholar]
- Freeman, J.E. Quality Preservation During Harvesting, Conditioning, and Storage of Grains and Oilseeds. Crop. Qual. Storage Util. 1980, 185–226. [Google Scholar]
- Gibon, V. Palm oil and palm kernel oil refining and fractionation technology. In Palm Oil; Lai, O.-M., Tan, C.-P., Akoh, C.C., Eds.; AOCS Press: Champaign, IL, USA, 2012; pp. 329–375. [Google Scholar]
- O’Brien, R.D. Fats and oils processing: Formulating and processing for applications. In Introduction to Fats and Oils Technology; AOCS Press: Champaign, IL, USA, 2000; pp. 90–107. [Google Scholar]
- Franke, K.; Strijowski, U.; Fleck, G.; Pudel, F. LWT—Food Science and Technology Influence of chemical refining process and oil type on bound 3-chloro-1, 2-propanediol contents in palm oil and rapeseed oil. LWT Food Sci. Technol. 2009, 42, 1751–1754. [Google Scholar] [CrossRef]
- Piloto-Rodríguez, R.; Melo, E.A.; Goyos-Pérez, L.; Verhelst, S. Conversion of by-products from the vegetable oil industry into biodiesel and its use in internal combustion engines: A review. Braz. J. Chem. Eng. 2014, 31, 287–301. [Google Scholar] [CrossRef]
- Malaysian Palm Oil Council. Available online: http://mpoc.org.my/processing-flow-chart-2/ (accessed on 30 September 2020).
- Craft, B.D.; Chiodini, A.; Garst, J.; Granvogl, M. Fatty acid esters of monochloropropanediol (MCPD) and glycidol in refined edible oils. Food Addit. Contam. A 2013, 30, 46–51. [Google Scholar] [CrossRef]
- Matthäus, B.; Pudel, F. Mitigation of 3-MCPD and glycidyl esters within the production chain of vegetable oils especially palm oil. Lipid Technol. 2013, 25, 151–155. [Google Scholar] [CrossRef]
- Henson, I.E. A brief history of the oil palm. In Palm Oil; Lai, O.-M., Tan, C.-P., Akoh, C.C., Eds.; AOCS Press: Champaign, IL, USA, 2012; pp. 1–29. [Google Scholar]
- Albert, M.M.E.; Laverdure, D.E.E.; Paul, K. Assessment of the quality of crude palm oil from smallholders in Cameroon. J. Postharvest Res. 2011, 2, 52–58. [Google Scholar]
- Gardner, A.M.; Yurawecs, M.P.; Cunningham, W.C.; Diachenko, G.W.; Mazzola, E.P.; Brumley, W.C. Isolation and Identification of C16 and C18 Fatty Acid Esters of Chloropropanediol in Adulterated Spanish Cooking Oils. Bull. Environ. Contam. Toxicol. 1983, 31, 625–630. [Google Scholar] [CrossRef]
- Arisseto, A.P.; Marcolino, P.F.C.; Vicente, E. Determination of 3-monochloropropane-1,2-diol fatty acid esters in Brazilian vegetable oils and fats by an in-house validated method. Food Addit. Contam. A 2014, 31, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Ermacora, A.; Hrncirik, K. A novel method for simultaneous monitoring of 2-MCPD, 3-MCPD and glycidyl esters in oils and fats. J. Am. Oil Chem. Soc. 2013, 90, 1–8. [Google Scholar] [CrossRef]
- MacMahon, S.; Begley, T.H.; Diachenko, G.W. Occurrence of 3-MCPD and glycidyl esters in edible oils in the United States. Food Addit. Contam. Part A 2013, 30, 2081–2092. [Google Scholar] [CrossRef] [PubMed]
- Weißhaar, R. Determination of total 3-chloropropane-1,2-diol (3-MCPD) in edible oils by cleavage of MCPD esters with sodium methoxide. Eur. J. Lipid Sci. Technol. 2008, 110, 183–186. [Google Scholar] [CrossRef]
- Weißhaar, R.; Perz, R. Fatty acid esters of glycidol in refined fats and oils. Eur. J. Lipid Sci. Technol. 2010, 112, 158–165. [Google Scholar] [CrossRef]
- Zelinkova, Z.; Svejkovska, B.; Velisek, J.; Dolezal, M. Fatty acid esters of 3-chloropropane-1,2-diol in edible oils. Food Addit. Contam. 2006, 23, 1290–1298. [Google Scholar] [CrossRef]
- Hrncirik, K.; van Duijn, G. An initial study on the formation of 3-MCPD esters during oil refining. Eur. J. Lipid Sci. Technol. 2011, 113, 374–379. [Google Scholar] [CrossRef]
- Hung, W.; Peng, G.; Tsai, W.; Chang, M. Identification of 3-MCPD esters to verify the adulteration of extra virgin olive oil. Food Addit. Contam. Part B 2017, 10, 233–239. [Google Scholar] [CrossRef]
- Calta, P.; Velíšek, J.; Doležal, M.; Hasnip, S.; Crews, C.; Réblová, Z. Formation of 3-chloropropane-1,2-diol in systems simulating processed foods. Eur. Food Res. Technol. 2004, 218, 501–506. [Google Scholar] [CrossRef]
- Sim, B.I.; Muhamad, H.; Lai, O.M.; Abas, F.; Yeoh, C.B.; Nehdi, I.A.; Khor, Y.P.; Tan, C.P. New insights on degumming and bleaching process parameters on the formation of 3-monochloropropane-1,2-diol esters and glycidyl esters in refined, bleached, deodorized palm oil. J. OLEO Sci. 2018, 67, 397–406. [Google Scholar] [CrossRef]
- Silva, S.M.; Sampaio, K.A.; Ceriani, R.; Verhé, R.; Stevens, C.; De Greyt, W.; Meirelles, A.J.A. Adsorption of carotenes and phosphorus from palm oil onto acid activated bleaching earth: Equilibrium, kinetics and thermodynamics. J. Food Eng. 2013, 118, 341–349. [Google Scholar] [CrossRef]
- Carter, C.; Finley, W.; Fry, J.; Jackson, D.; Willis, L. Palm oil markets and future supply. Eur. J. Lipid Sci. Technol. 2007, 109, 307–314. [Google Scholar] [CrossRef]
- Corley, R.H.V. How much palm oil do we need? Environ. Sci. Policy 2009, 12, 134–139. [Google Scholar] [CrossRef]
- Von Geibler, J. Market-based governance for sustainability in value chains: Conditions for successful standard setting in the palm oil sector. J. Clean. Prod. 2013, 56, 39–53. [Google Scholar] [CrossRef]
- Gatto, M.; Wollni, M.; Asnawi, R.; Qaim, M. Oil Palm Boom, Contract Farming, and Rural Economic Development: Village-Level Evidence from Indonesia. World Dev. 2017, 95, 127–140. [Google Scholar] [CrossRef]
- Srinivas, A.; Koh, L.P. Oil palm expansion drives avifaunal decline in the Pucallpa region of Peruvian Amazonia. Glob. Ecol. Conserv. 2016, 7, 183–200. [Google Scholar] [CrossRef]
- Economic and Industry Development Division Home Page. Available online: http://bepi.mpob.gov.my/index.php/en/ (accessed on 21 August 2020).
- Razak, R.A.A.; Kuntom, A.; Siew, W.L.; Ibrahim, N.A.; Ramli, M.R.; Hussein, R.; Nesaretnam, K. Detection and monitoring of 3-monochloropropane-1, 2-diol (3-MCPD) esters in cooking oils. Food Control 2012, 25, 355–360. [Google Scholar] [CrossRef]
- Zulkurnain, M.; Lai, O.M.; Tan, S.C.; Abdul Latip, R.; Tan, C.P. Optimization of palm oil physical refining process for reduction of 3-monochloropropane-1, 2-diol (3-MCPD) ester formation. J. Agric. Food Chem. 2013, 61, 3341–3349. [Google Scholar] [CrossRef]
- Weißhaar, R. 3-MCPD-esters in edible fats and oils-a new and worldwide problem. Eur. J. Lipid Sci. Technol. 2008, 110, 671–672. [Google Scholar] [CrossRef]
- Pudel, F.; Benecke, P.; Fehling, P.; Freudenstein, A.; Matthäus, B.; Schwaf, A. On the necessity of edible oil refining and possible sources of 3-MCPD and glycidyl esters. Eur. J. Lipid Sci. Technol. 2011, 113, 368–373. [Google Scholar] [CrossRef]
- Ramli, M.R.; Siew, W.L.; Ibrahim, N.A.; Hussein, R.; Kuntom, A.; Razak, R.A.A.; Nesaretnam, K. Effects of degumming and bleaching on 3-MCPD esters formation during physical refining. J. Am. Oil Chem. Soc. 2011, 88, 1839–1844. [Google Scholar] [CrossRef]
- Matthäus, B.; Pudel, F.; Fehling, P.; Vosmann, K.; Freudenstein, A. Strategies for the reduction of 3-MCPD esters and related compounds in vegetable oils. Eur. J. Lipid Sci. Technol. 2011, 113, 380–386. [Google Scholar] [CrossRef]
- Destaillats, F.; Craft, B.D.; Sandoz, L.; Nagy, K. Formation mechanisms of Monochloropropanediol (MCPD) fatty acid diesters in refined palm (Elaeis guineensis) oil and related fractions. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess 2012, 29, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Nagy, K.; Sandoz, L.; Craft, B.D.; Destaillats, F. Mass-defect filtering of isotope signatures to reveal the source of chlorinated palm oil contaminants. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess 2011, 28, 1492–1500. [Google Scholar] [CrossRef]
- Šmidrkal, J.; Tesařová, M.; Hrádková, I.; Berčíková, M.; Adamčíková, A.; Filip, V. Mechanism of formation of 3-chloropropan-1,2-diol (3-MCPD) esters under conditions of the vegetable oil refining. Food Chem. 2016, 211, 124–129. [Google Scholar] [CrossRef]
- Freudenstein, A.; Weking, J.; Matthäus, B. Influence of precursors on the formation of 3-MCPD and glycidyl esters in a model oil under simulated deodorization conditions. Eur. J. Lipid Sci. Technol. 2013, 115, 286–294. [Google Scholar] [CrossRef]
- Shimizu, M.; Weitkamp, P.; Vosmann, K.; Matthäus, B. Influence of chloride and glycidyl-ester on the generation of 3-MCPD- and glycidyl-esters. Eur. J. Lipid Sci. Technol. 2013, 115, 735–739. [Google Scholar] [CrossRef]
- Tiong, S.H.; Saparin, N.; Teh, H.F.; Ng, T.L.M.; bin Zain, M.M.Z.; Neoh, B.K.; Md Noor, A.; Tan, C.P.; Lai, O.M.; Appleton, D.R. Natural Organochlorines as Precursors of 3-Monochloropropanediol Esters in Vegetable Oils. J. Agric. Food Chem. 2018, 66, 999–1007. [Google Scholar] [CrossRef]
- Svejkovska, B.; Dolezal, M.; Velisek, J. Formation and decomposition of 3-chloropropane-1, 2-diol esters in models simulating processed foods. Czech. J. Food Sci. 2006, 24, 172. [Google Scholar] [CrossRef]
- Ah, T.; Mohd, K.Y.; Nik, M.M.; Joo, G.; Huang, G. Effect of N and K fertilizers on nutrient leaching and groundwater quality under mature oil palm in Sabah during the monsoon period. Am. J. Appl. Sci. 2009, 6, 1788–1799. [Google Scholar]
- Kuntom, A.; Tan, Y.A.; Kamaruddin, N.; Yeoh, C.B. Pesticide application in oil palm plantation. Oil. Palm Bull. 2007, 54, 52–67. [Google Scholar]
- Rutherford, M.; Flood, J.; Sastroutomo, S. Roundtable for sustainable palm oil (RSPO): Research project on integrated weed management strategies for oil palm. Final Rep. 2011, 209, 205. [Google Scholar]
- Hashim, Z.; Muhamad, H.; Subramaniam, V.; May, C.Y. Water footprint: Part 2—FFB production for oil palm planted in Malaysia. J. Oil Palm Res. 2014, 26, 282–291. [Google Scholar]
- Hopkins, B.G.; Horneck, D.A.; Stevens, R.G.; Ellsworth, J.W.; Sullivan, D.M. Managing irrigation water quality for crop production in Pacific Northwest. Or. State Univ. 2007, PNW 597-E, 1–25. [Google Scholar]
- Lee, C.S.; Robinson, J.; Chong, M.F. A review on application of flocculants in wastewater treatment. Process. Saf. Environ. Prot. 2014, 92, 489–508. [Google Scholar] [CrossRef]
- Krisdiarto, A.W.; Sutiarso, L. Study on Oil Palm Fresh Fruit Bunch Bruise in Harvesting and Transportation to Quality. Makara J. Technol. 2016, 20, 67–72. [Google Scholar] [CrossRef]
- Krisdiarto, A.W. Mapping of Bruise of Oil Palm Fresh Fruit Bunch during Loading and Transportation from Field to Mill. Makara J. Technol. 2018, 22, 84–87. [Google Scholar] [CrossRef]
- Elias, M.; Laranjo, M.; Agulheiro-Santos, A.C.; Potes, M.E. The role of salt on food and human health. In Salt in the Earth; Cengiz, M., Karabulut, S., Eds.; InTechOpen: London, UK, 2020; p. 19. [Google Scholar]
- Bognár, E.; Hellner, G.; Radnóti, A.; Somogyi, L.; Kemény, Z. Effect of Different Chlorine Sources on the Formation of 3-Monochloro-1,2-Propanediol and 2-Monochloro-1,3-Propanediol Fatty Acid Esters during Frying. Period. Polytech. Chem. Eng. 2020, 64, 523–529. [Google Scholar] [CrossRef]
- Zhang, X.; Gao, B.; Qin, F.; Shi, H.; Jiang, Y.; Xu, X.; Yu, L. Free radical mediated formation of 3-monochloropropanediol (3-MCPD) fatty acid diesters. J. Agric. Food Chem. 2013, 61, 2548–2555. [Google Scholar] [CrossRef]
- Zhang, Z.; Gao, B.; Zhang, X.; Jiang, Y.; Xu, X.; Yu, L. Formation of 3-monochloro-1,2-propanediol (3-MCPD) Di- and monoesters from tristearoylglycerol (TSG) and the potential catalytic effect of Fe2+ and Fe3+. J. Agric. Food Chem. 2015, 63, 1839–1848. [Google Scholar] [CrossRef]
- Li, C.; Li, L.; Jia, H.; Wang, Y.; Shen, M.; Nie, S.; Xie, M. Formation and reduction of 3-monochloropropane-1,2-diol esters in peanut oil during physical refining. Food Chem. 2016, 199, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, R.; Ito, F.; Shimamura, Y.; Masuda, S. Effect of chloride on the formation of 3-monochloro-1,2-propanediol fatty acid diesters and glycidol fatty acid esters in fish, meats and acylglycerols during heating. Food Addit. Contam. Part A 2019, 36, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Rahn, A.K.K.; Yaylayan, V.A. What do we know about the molecular mechanism of 3-MCPD ester formation? Eur. J. Lipid Sci. Technol. 2011, 113, 323–329. [Google Scholar] [CrossRef]
- Ahmad Tarmizi, A.H.; Abd Razak, R.A.; Abdul Hammid, A.N.; Kuntom, A. Effect of Anti-Clouding Agent on the Fate of 3-Monochloropropane-1, 2-Diol Esters and Glycidyl Esters in Palm Olein during Repeated Frying. Molecules 2019, 24, 2332. [Google Scholar] [CrossRef] [PubMed]
- Wong, Y.H.; Lai, O.M.; Abas, F.; Nyam, K.L.; Nehdi, I.A.; Muhamad, H.; Tan, C.P. Factors impacting the formation of 3-MCPD esters and glycidyl esters during deep fat frying of chicken breast meat. J. Am. Oil Chem. Soc. 2017, 94, 759–765. [Google Scholar] [CrossRef]
- de Oliveira, I.F.; Grimaldi, R.; Gonçalves, L.A.G. Effect of diacylglycerols on crystallization of palm oil (Elaeis guineensis). Eur. J. Lipid Sci. Technol. 2014, 116, 904–909. [Google Scholar] [CrossRef]
- Goh, E.M.; Timms, R.E. Determination of mono-and diglycerides in palm oil, olein and stearin. J. Am. Oil Chem. Soc. 1985, 62, 730–734. [Google Scholar] [CrossRef]
- Kaze, N.; Sato, H.; Yamamoto, H.; Watanabe, Y. Bidirectional conversion between 3-monochloro-1, 2-propanediol and glycidol in course of the procedure of DGF standard methods. J. Am. Oil Chem. Soc. 2011, 88, 1143–1151. [Google Scholar] [CrossRef]
- Tarmizi, A.M.; Mohd Tayeb, D. Nutrient demands of tenera oil palm planted on inland soil of Malaysia. J. Oil Palm Res. 2006, 18, 204. [Google Scholar]
- Basiron, Y.; Weng, C.K. The oil palm and its sustainability. J. Oil Palm Res. 2004, 16, 1–10. [Google Scholar]
- Zieverink, M.M.P.; Berg, I. Oil processing development. In Proceedings of the 8th EuroFed Lipid Congress, Munich, Germany, 21–24 November 2010; pp. 21–24. [Google Scholar]
- Ramli, M.R.; Siew, W.L.; Ibrahim, N.A.; Kuntom, A.; Arni, R.; Razak, A. Other factors to consider in the formation of chloropropandiol fatty esters in oil processes. Food Addit. Contam. Part A 2015, 32, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Japir, A.A.W.; Salimon, J.; Derawi, D.; Bahadi, M.; Al-Shuja’a, S.; Yusop, M.R. Physicochemical characteristics of high free fatty acid crude palm oil. OCL 2017, 24, D506. [Google Scholar] [CrossRef]
- Cherie, D.; Rini, R.; Makky, M. Determination of the optimum harvest window and quality attributes of oil palm fresh fruit bunch using non-destructive shortwave infrared spectroscopy. In AIP Conference Proceedings; AIP: College Park, MD, USA, 2019; Volume 2155. [Google Scholar]
- Pratik, V.; Surekha, D. Refining of palm oil: A review on palm oil refining process, 3-MCPD esters in refined palm oil, and possible reduction tactics for 3-MCPD esters. Int. J. Agric. Eng. 2018, 11, 81–85. [Google Scholar]
- Federal German Institute for Risk Assessment: BfR Method_82_FC-008-01 No Title. Determination of 3-MCPD Fatty Acid Esters in Edible Oils and Solid Fats by GC-MS—An Indirect Determination by Detection of free 3-MCPD released from 3-MCPD-Esters by Acid Hydrolysis and by Derivatization with Phenylboronic Acid; Federal Institute for Risk Assessment: Dahlem, Germany, 2011.
- Federal German Institute for Risk Assessment: BfR Method_82_FC-010-01. Determination of 3-MCPD Fatty Acid Esters in Edible Oils and Solid Fats by GC-MS—An Indirect Determination of free 3-MCPD released from 3-MCPD-Esters by Alkaline Hydrolysis and by Derivatization with Heptafluorobutyric Anhydride; Federal Institute for Risk Assessment: Dahlem, Germany, 2011.
- Federal German Institute for Risk Assessment: BfR Method_82_FC-009-01 No Title. Determination of 3-MCPD Fatty Acid Esters in Edible Oils and Solid Fats by GC-MS—An Indirect Determination by Detection of free 3-MCPD released from 3-MCPD-Esters by Alkaline Hydrolysis and by Derivatization with Phenylboronic Acid; Federal Institute for Risk Assessment: Dahlem, Germany, 2011.
- Deutsche Gesellschalt für Fettewissenschaft (DGF). Deutsche Einheitsmethoden zur Untersuchung von Fetten, Fettprodukten, Tensiden und verwandten Stoffen; Wissenschaftliche Verlagsgesellschaft: Stuttgart, Germany, 1998. [Google Scholar]
- Haines, T.D.; Adlaf, K.J.; Pierceall, R.M.; Lee, I.; Venkitasubramanian, P.; Collison, M.W. Direct determination of MCPD fatty acid esters and glycidyl fatty acid esters in vegetable oils by LC-TOFMS. J. Am. Oil Chem. Soc. 2011, 88, 1–14. [Google Scholar] [CrossRef]
- Xing, X.; Cao, Y. Determination of 3-chloro-1, 2-propanediol in soy sauces by capillary electrophoresis with electrochemical detection. Food Control 2007, 18, 167–172. [Google Scholar] [CrossRef]
- Dininova, V.; Svejkovska, B.; Dolezal, M.; Velisek, J. Determination of free and bound 3-chloropropane-1,2-diol by gas chromatography with mass spectrometric detection using deuterated 3-chloropropane-1,2-diol as internal standard. Czech. J. Food Sci. 2005, 22, 182–189. [Google Scholar] [CrossRef]
- Karasek, L.; Wenzl, T.; Ulberth, F. Determination of 3-MCPD esters in edible oil—methods of analysis and comparability of results. Eur. J. Lipid Sci. Technol. 2011, 113, 1433–1442. [Google Scholar] [CrossRef]
- Brereton, P.; Kelly, J.; Crews, C.; Honour, S.; Wood, R.; Davies, A. Determination of 3-chloro-1,2-propanediol in foods and food ingredients by gas chromatography with mass spectrometric detection: Collaborative study. J. AOAC Int. 2001, 84, 455–465. [Google Scholar] [CrossRef] [PubMed]
- Macmahon, S.; Mazzola, E.; Begley, T.H.; Diachenko, G.W. Analysis of processing contaminants in edible oils. Part 1. Liquid chromatography-tandem mass spectrometry method for the direct detection of 3-monochloropropanediol monoesters and glycidyl esters. J. Agric. Food Chem. 2013, 61, 4737–4747. [Google Scholar] [CrossRef]
- Macmahon, S.; Begley, T.H.; Diachenko, G.W. Analysis of processing contaminants in edible oils. Part 2. Liquid chromatography-tandem mass spectrometry method for the direct detection of 3-monochloropropanediol and 2-monochloropropanediol diesters. J. Agric. Food Chem. 2013, 61, 4748–4757. [Google Scholar] [CrossRef]
- Zhou, H.; Jin, Q.; Wang, X.; Xu, X. Direct measurement of 3-chloropropane-1, 2-diol fatty acid esters in oils and fats by HPLC method. Food Control 2014, 36, 111–118. [Google Scholar] [CrossRef]
- Ashley, J.; Shahbazi, M.; Kant, K.; Aaydha, V. Molecularly imprinted polymers for sample preparation and biosensing in food analysis: Progress and perspectives. Biosens. Bioelectron. 2017, 91, 606–615. [Google Scholar] [CrossRef] [PubMed]
- Fang, M.; Zhou, L.; Zhang, H.; Liu, L.; Gong, Z. A molecularly imprinted polymers/carbon dots-grafted paper sensor for 3-monochloropropane-1,2-diol determination. Food Chem. 2019, 274, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Leung, M.K.; Chiu, B.K.; Lam, M.H. Molecular sensing of 3-chloro-1,2-propanediol by molecular imprinting. Anal. Chim. Acta 2003, 491, 15–25. [Google Scholar] [CrossRef]
- Uzun, L.; Turner, A.P.F. Molecularly-imprinted polymer sensors: Realising their potential. Biosens. Bioelectron. 2016, 76, 131–144. [Google Scholar] [CrossRef]
- Yanez-Sedeno, P.; Campuzano, S.; Pingarron, J.M. Electrochemical sensors based on magnetic molecularly imprinted polymers: A review. Anal. Chim. Acta 2017, 960, 1–17. [Google Scholar] [CrossRef]
- Hrncirik, K.; Zelinkova, Z.; Ermacora, A. Critical factors of indirect determination of 3-chloropropane-1, 2-diol esters. Eur. J. Lipid Sci. Technol. 2011, 113, 361–367. [Google Scholar] [CrossRef]
- Sato, H.; Kaze, N.; Yamamoto, H.; Watanabe, Y. 2-Monochloro-1, 3-propanediol (2-MCPD) dynamics in DGF standard methods and quantification of 2-MCPD. J. Am. Oil Chem. Soc. 2013, 90, 1121–1130. [Google Scholar] [CrossRef]
- Sahibin, A.R.; Shamshuddin, J.; Fauziah, C.I.; Radziah, O.; Razi, I.W.M.; Enio, M.S.K. Impact of Mg rich synthetic gypsum application on the environment and palm oil quality. Sci. Total Environ. 2019, 652, 573–582. [Google Scholar] [CrossRef]
- Shevchenko, A. 3-MCPD and GE Mitigation in Palm Oil Processing. Available online: www.alfalaval.com (accessed on 8 May 2020).
- Shuib, A.R.; Khalid, M.R.; Deraman, M.S. Enhancing field mechanization in oil palm management. Oil Palm Bull. 2010, 61, 1–10. [Google Scholar]
- Abdul-Hamid, A.Q.; Ali, M.H.; Tseng, M.L.; Lan, S.; Kumar, M. Impeding challenges on industry 4.0 in circular economy: Palm oil industry in Malaysia. Comput. Oper. Res. 2020, 123, 105052. [Google Scholar] [CrossRef]
- Kasmin, H.; Lazim, A.M.; Awang, R. Effect of heat treatments on the yield, quality and storage stability of oil extracted from palm fruits. Malays. J. Anal. Sci. 2016, 20, 1373–1381. [Google Scholar]
- Che Mat, C.R.; Yap, K.C.A.; Halim, R.M.; Hadi, N.A.; Mohd Nazim, U.K.H. Effect of double roll crusher complete with preheating on bruising level and free fatty (FFA) acid content of oil palm fruit bunches. Palm Oil Eng. Bull. 2020, 132, 18–23. [Google Scholar]
- Bahadi, M.A.; Japir, A.W.; Salih, N.; Salimon, J. Free fatty acids separation from Malaysian high free fatty acid crude palm oil using molecular distillation. Malays. J. Anal. Sci. 2016, 20, 1042–1051. [Google Scholar]
- Kemeny, Z.; Bhaggan, K.; Bruse, F.; Creanga, A.; Diks, R.; Gambell, L.; Le Bail-Collet, Y.; Ribera, D. MCPDE and GE: An update on mitigation measures. In Encyclopedia of Food Chemistry, 1st ed.; Varelis, P., Melton, L., Shahidi, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 578–587. ISBN 9780128140451. [Google Scholar]
- Oey, S.B.; van der Fels-Klerx, H.J.; Fogliano, V.; van Leeuwen, S.P.J. Mitigation strategies for the reduction of 2- and 3-MCPD esters and glycidyl esters in the vegetable oil processing industry. Compr. Rev. Food Sci. F. 2019, 18, 349–361. [Google Scholar] [CrossRef]
- Silvamany, H.; Md Jahim, J. Enhancement of palm oil extraction using cell wall degrading enzyme formulation. Malays. J. Anal. Sci. 2015, 19, 77–87. [Google Scholar]
- Teixeira, C.B.; Macedo, G.A.; Macedo, J.A.; da Silva, L.H.M.; da Rodrigues, C.A.M. Simultaneous extraction of oil and antioxidant compounds from oil palm fruit (Elaeis guineensis) by an aqueous enzymatic process. Bioresour. Technol. 2013, 123, 575–581. [Google Scholar] [CrossRef]
- Strijowski, U.; Heinz, V.; Franke, K. Removal of 3-MCPD esters and related substances after refining by adsorbent material. Eur. J. Lipid Sci. Technol. 2011, 113, 387–392. [Google Scholar] [CrossRef]
- Gertz, C. Optimising the baking and frying process using oil-improving agents. Eur. J. Lipid Sci. Technol. 2004, 106, 736–745. [Google Scholar] [CrossRef]
- Lin, S.; Akoh, C.C.; Reynolds, A.E. Recovery of used frying oils with adsorbent combinations: Refrying and frequent oil replenishment. Food Res. Int. 2001, 34, 159–166. [Google Scholar] [CrossRef]
- Yates, R.A.; Caldwell, J.D. Regeneration of Oils Used for Deep Frying: A Comparison of Active Filter Aids. J. Am. Oil Chem. Soc. 1993, 70, 507–511. [Google Scholar] [CrossRef]
- Zhang, H.; Jin, P.; Zhang, M.; Cheong, L.-Z.; Hu, P.; Zhao, Y.; Yu, L.; Wang, Y.; Jiang, Y.; Xu, X. Mitigation of 3-monochloro-1,2-propanediol ester formation by radical scavengers. J. Agric. Food Chem. 2016, 64, 5887–5892. [Google Scholar] [CrossRef] [PubMed]
- Bornscheuer, U.T.; Hesseler, M. Enzymatic removal of 3-monochloro-1,2-propanediol (3-MCPD) and its esters from oils. Eur. J. Lipid Sci. Technol. 2010, 112, 552–556. [Google Scholar] [CrossRef]
- Bel-Rhlid, R.; Talmon, J.P.; Fay, L.B.; Juillerat, M.A. Biodegradation of 3-Chloro-1,2-propanediol with Saccharomyces cerevisiae. J. Agric. Food Chem. 2004, 52, 6165–6169. [Google Scholar] [CrossRef]
- Wong, Y.H.; Goh, K.M.; Nyam, K.L.; Nehdi, I.A.; Sbihi, H.M.; Tan, C.P. Effects of natural and synthetic antioxidants on changes in 3-MCPD esters and glycidyl ester in palm olein during deep-fat frying. Food Control 2019, 96, 488–493. [Google Scholar] [CrossRef]
- Hrádková, I.; Merkl, R.; Šmidrkal, J.; Kyselka, J.; Filip, V. Antioxidant effect of mono-and dihydroxyphenols in sunflower oil with different levels of naturally present tocopherols. Eur. J. Lipid Sci. Technol. 2013, 115, 747–755. [Google Scholar] [CrossRef]
- Goh, K.M.; Wong, Y.H.; Abas, F.; Lai, O.M.; Yusoff, M.M.; Tan, T.B.; Tan, C.P. Changes in 3-, 2-Monochloropropandiol and Glycidyl Esters during a Conventional Baking System with Addition of Antioxidants. Foods 2020, 9, 739. [Google Scholar] [CrossRef]


| Samples | Method | 3-MCPD Concentration/Removal | Ref. | |
|---|---|---|---|---|
| Initial | Final | |||
| 3-MCPD and related substances |
| 6.59 ± 0.19 ppm | 4.00 ppm | [144] |
| 6.59 ± 0.19 ppm | 4.50 ppm | [144] | |
| 3-MCPD esters |
| 100% | 82% reduction | [148] |
| 100% | 75% reduction | [148] | |
| 3-MCPD |
| 10.00 mM | 2.9 mM | [149] |
| 10.00 mM | 0.00 mM | [149] | |
| Racemic (R,S) of 3-MCPD |
| 7.3 µmol/L | 68% conversion | [150] |
| 27 mmol/L | 73% conversion | [150] | ||
| Samples | Antioxidant | 3-MCPD, mg/kg | 2-MCPD, mg/kg | GE, mg/kg |
|---|---|---|---|---|
| Butylated hydroxyanisole | 3.439 ± 0.029 a | 2.334 ± 0.021 a | 1.987 ± 0.039 a | |
| Palm Olein | Rosemary | 3.431 ± 0.065 a | 2.051 ± 0.010 b | 1.979 ± 0.029 a |
| Tocopherol | 3.527 ± 0.189 a | 2.143 ± 0.105 b | 1.985 ± 0.047 a | |
| Butylated hydroxyanisole | 2.222 ± 0.028 a | 1.534 ± 0.114 a | 0.665 ± 0.001 a | |
| Soft Stearin | Rosemary | 2.176 ± 0.032 a | 1.489 ± 0.040 a | 0.607 ± 0.032 b |
| Tocopherol | 2.172 ± 0.023 a | 1.435 ± 0.012 a | 0.668 ± 0.010 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arris, F.A.; Thai, V.T.S.; Manan, W.N.; Sajab, M.S. A Revisit to the Formation and Mitigation of 3-Chloropropane-1,2-Diol in Palm Oil Production. Foods 2020, 9, 1769. https://doi.org/10.3390/foods9121769
Arris FA, Thai VTS, Manan WN, Sajab MS. A Revisit to the Formation and Mitigation of 3-Chloropropane-1,2-Diol in Palm Oil Production. Foods. 2020; 9(12):1769. https://doi.org/10.3390/foods9121769
Chicago/Turabian StyleArris, Farrah Aida, Vincent Tiang Soon Thai, Wan Nabilah Manan, and Mohd Shaiful Sajab. 2020. "A Revisit to the Formation and Mitigation of 3-Chloropropane-1,2-Diol in Palm Oil Production" Foods 9, no. 12: 1769. https://doi.org/10.3390/foods9121769
APA StyleArris, F. A., Thai, V. T. S., Manan, W. N., & Sajab, M. S. (2020). A Revisit to the Formation and Mitigation of 3-Chloropropane-1,2-Diol in Palm Oil Production. Foods, 9(12), 1769. https://doi.org/10.3390/foods9121769





