A Review of Registered Clinical Trials on Dietary (Poly)Phenols: Past Efforts and Possible Future Directions
Abstract
1. Introduction
2. Materials and Methods
2.1. Database Search Strategy
- ClinicalTrials.gov: polyphenols OR flavonoids OR flavanols OR anthocyanidins OR anthocyanins OR isoflavones OR flavones OR flavonols OR flavanones OR flavanonols OR nonflavonoids OR phenolic acids OR stilbenes OR lignans.
- ISRCTN registry: (“polyphenols”) OR (“flavonoids”) OR (“flavanols”) OR (“anthocyanidins”) OR (“anthocyanins”) OR (“isoflavones”) OR (“flavones”) OR (“flavonols”) OR (“flavanones”) OR (“flavanonols”) OR (“nonflavonoids”) OR (“phenolic acids”) OR (“stilbenes”) OR (“lignans”).
2.2. Study Selection
2.3. Data Extraction and Analysis
3. Results
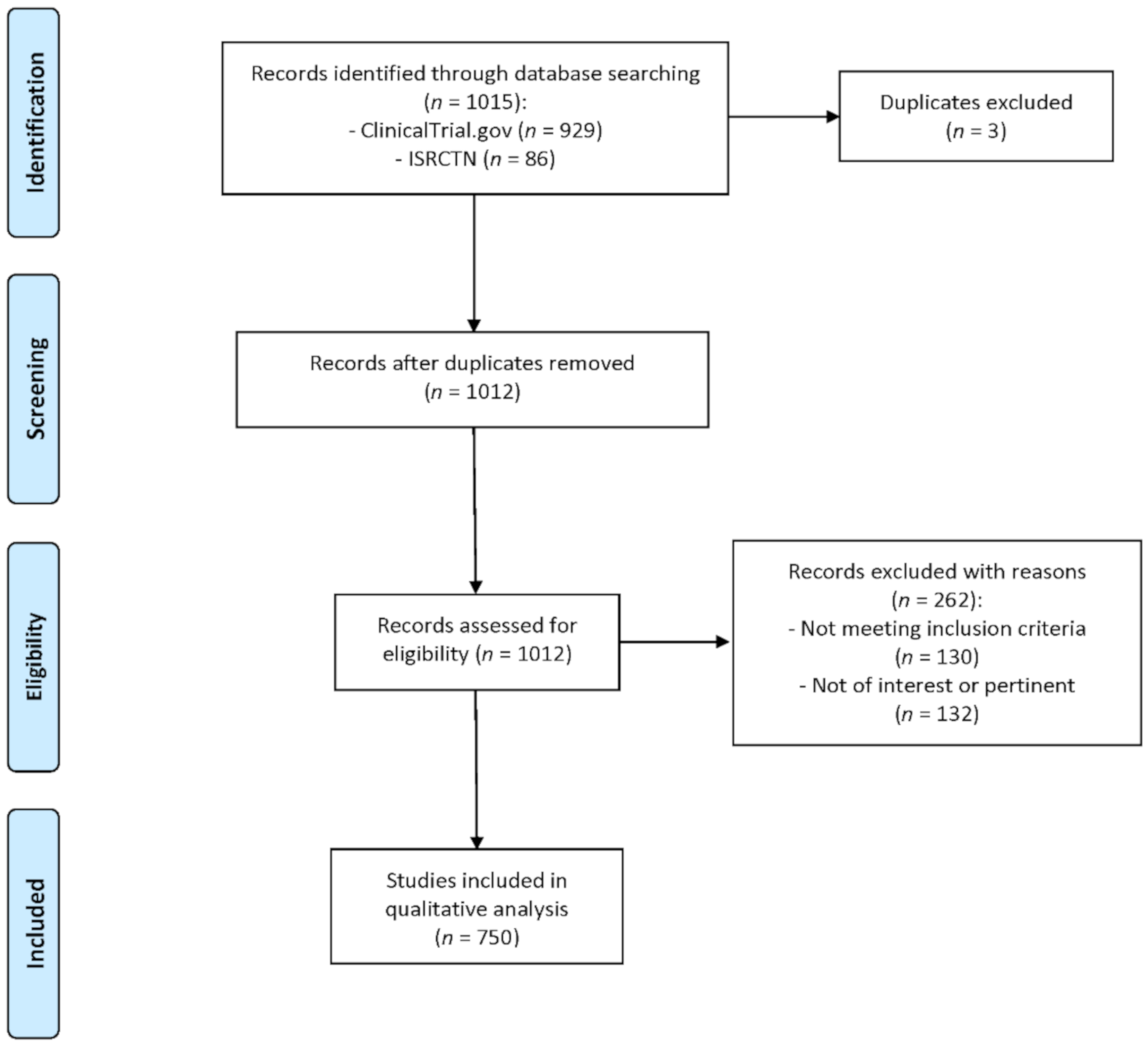
3.1. Study Selection
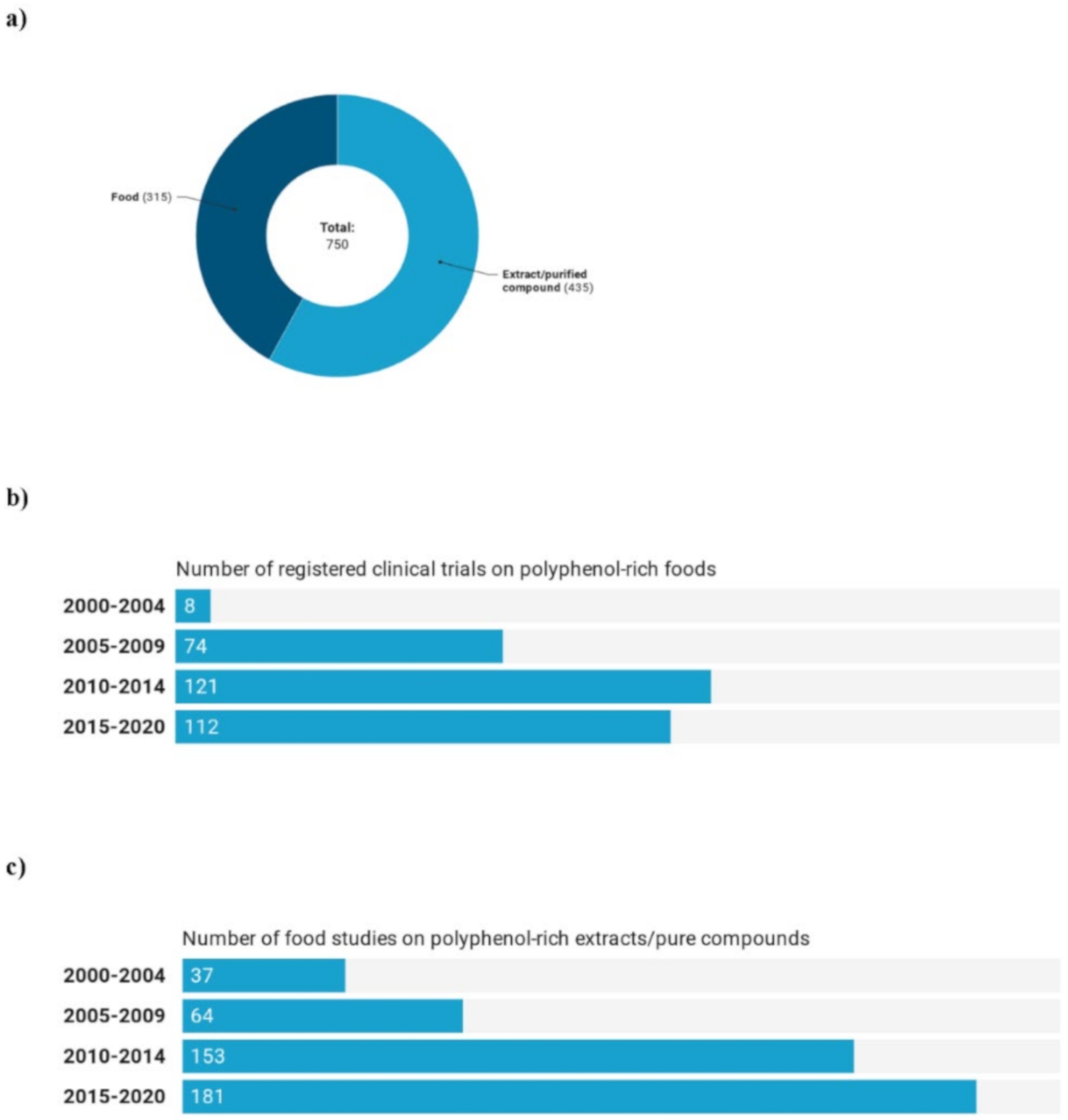
3.2. Trials on Polyphenol-Rich Foods and Extracts
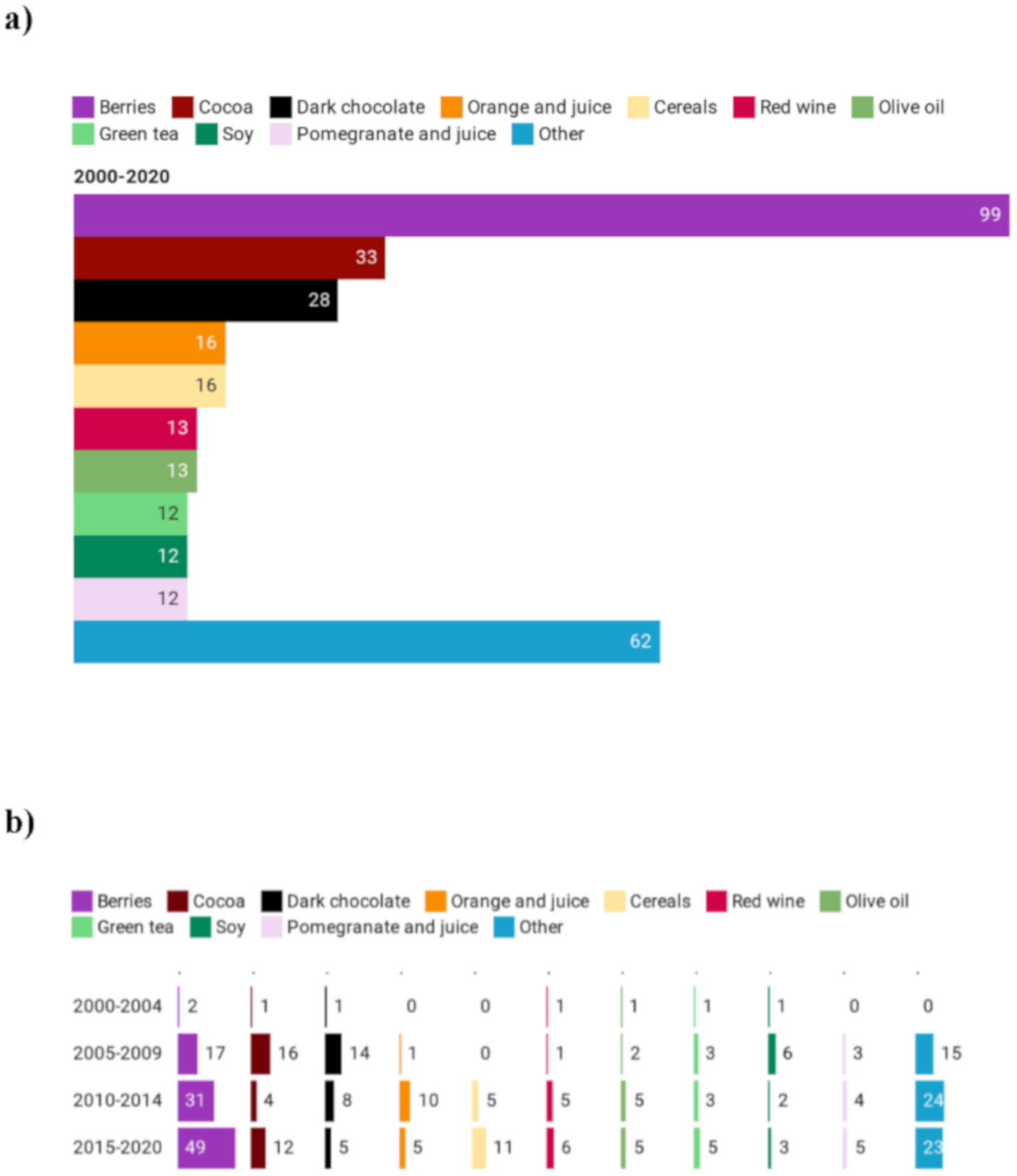
3.2.1. Types of Polyphenol-Rich Foods
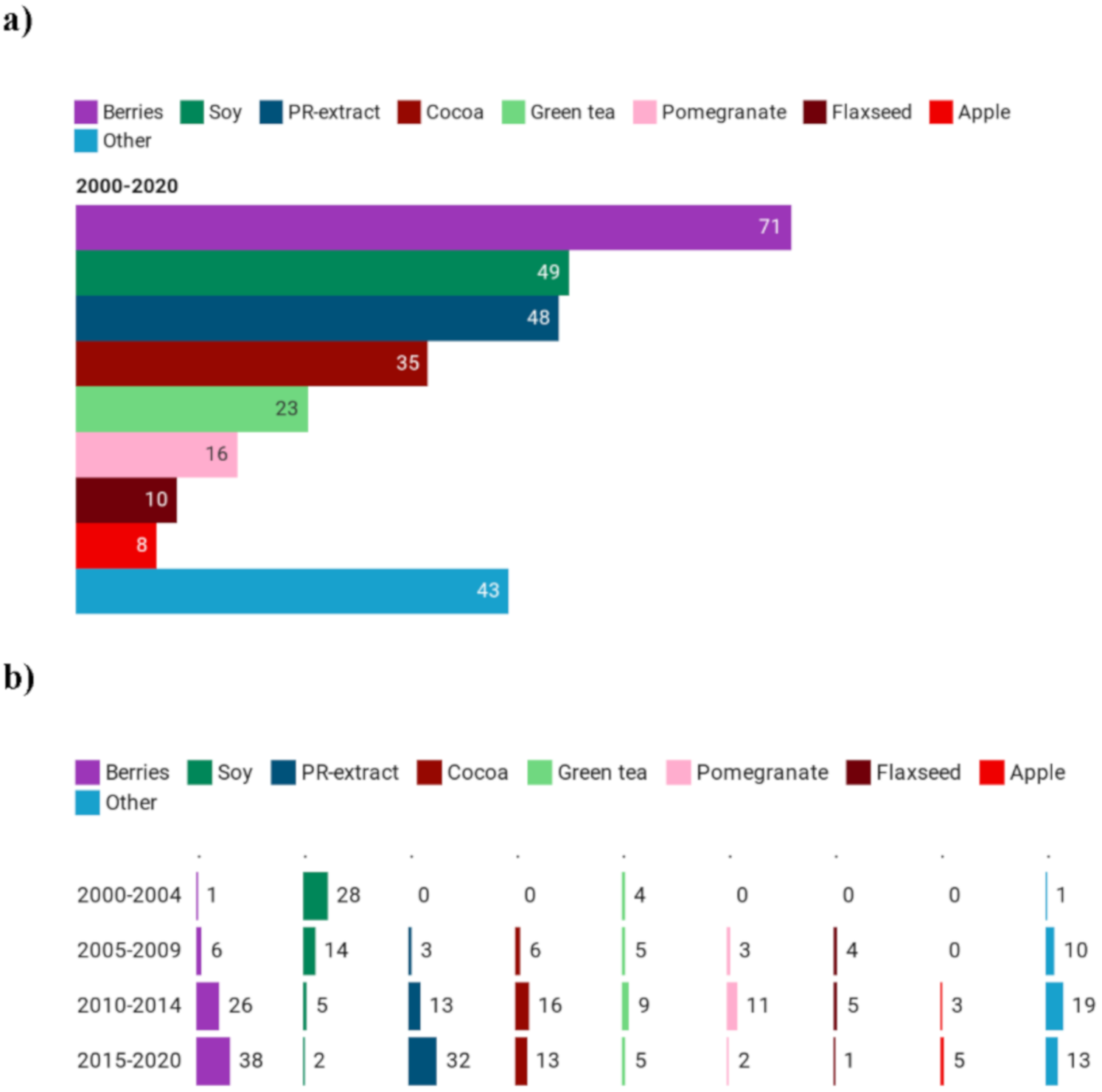
3.2.2. Types of Polyphenol-Rich Extracts
3.2.3. Types of Pure (Poly)Phenolic Compounds
3.3. Characteristics of Subjects
3.4. Main Goals of the Registered Trials
3.5. Other Characteristics of the Registered Trials
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/ (accessed on 30 April 2020).
- Adebooye, O.C.; Alashi, A.M.; Aluko, R.E. A brief review on emerging trends in global polyphenol research. J. Food Biochem. 2018, 42, e12519. [Google Scholar] [CrossRef]
- Abbas, M.; Saeed, F.; Anjum, F.M.; Afzaal, M.; Tufail, T.; Bashir, M.S.; Ishtiaq, A.; Hussain, S.; Suleria, H.A.R. Natural polyphenols: An overview. Int. J. Food Prop. 2017, 20, 1689–1699. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010, 64, S112–S120. [Google Scholar] [CrossRef] [PubMed]
- D’Archivio, M.; Filesi, C.; Di Benedetto, R.; Gargiulo, R.; Giovannini, C.; Masella, R. Polyphenols, dietary sources and bioavailability. Ann. Ist. Super. Sanita 2007, 43, 348–361. [Google Scholar] [PubMed]
- Fraga, C.G.; Oteiza, P. Bioactives and their impact on human health. Mol. Asp. Med. 2018, 61, 1. [Google Scholar] [CrossRef]
- Jin, K. New perspectives on healthy aging. Prog. Neurobiol. 2017, 157, 1. [Google Scholar] [CrossRef]
- Fraga, C.G.; Croft, K.D.; Kennedy, D.O.; Tomás-Barberán, F.A. The effects of polyphenols and other bioactives on human health. Food Funct. 2019, 10, 514–528. [Google Scholar] [CrossRef]
- Giacco, R.; Costabile, G.; Fatati, G.; Frittitta, L.; Maiorino, M.I.; Marelli, G.; Parillo, M.; Pistis, D.; Tubili, C.; Vetrani, C.; et al. Effects of polyphenols on cardio-metabolic risk factors and risk of type 2 diabetes. A joint position statement of the Diabetes and Nutrition Study Group of the Italian Society of Diabetology (SID), the Italian Association of Dietetics and Clinical Nutrition (ADI) and the Italian Association of Medical Diabetologists (AMD). Nutr. Metab. Cardiovasc. Dis. 2020, 30, 355–367. [Google Scholar]
- Gibney, E.R.; Milenkovic, D.; Combet, E.; Ruskovska, T.; Greyling, A.; González-Sarrías, A.; de Roos, B.; Tomás-Barberán, F.; Morand, C.; Rodriguez-Mateos, A. Factors influencing the cardiometabolic response to (poly)phenols and phytosterols: A review of the COST Action POSITIVe activities. Eur. J. Nutr. 2019, 58, 37–47. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Selma, M.V.; Espín, J.C. Interactions of gut microbiota with dietary polyphenols and consequences to human health. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Shortt, C.; Hasselwander, O.; Meynier, A.; Nauta, A.; Fernández, E.N.; Putz, P.; Rowland, I.; Swann, J.; Türk, J.; Vermeiren, J.; et al. Systematic review of the effects of the intestinal microbiota on selected nutrients and non-nutrients. Eur. J. Nutr. 2018, 57, 25–49. [Google Scholar] [CrossRef]
- Godos, J.; Vitale, M.; Micek, A.; Ray, S.; Martini, D.; Del Rio, D.; Riccardi, G.; Galvano, F.; Grosso, G. Dietary Polyphenol Intake, Blood Pressure, and Hypertension: A Systematic Review and Meta-Analysis of Observational Studies. Antioxidants 2019, 8, 152. [Google Scholar] [CrossRef]
- Pham, N.M.; Do, V.V.; Lee, A.H. Polyphenol-rich foods and risk of gestational diabetes: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 2019, 73, 647–656. [Google Scholar] [CrossRef]
- Rienks, J.; Barbaresko, J.; Nöthlings, U. Association of Polyphenol Biomarkers with Cardiovascular Disease and Mortality Risk: A Systematic Review and Meta-Analysis of Observational Studies. Nutrients 2017, 9, 415. [Google Scholar]
- Renaud, J.; Martinoli, M. Considerations for the Use of Polyphenols as Therapies in Neurodegenerative Diseases. Int. J. Mol. Sci. 2019, 20, 1883. [Google Scholar] [CrossRef] [PubMed]
- Ruskovska, T.; Maksimova, V.; Milenkovic, D. Polyphenols in human nutrition: From the in vitro antioxidant capacity to the beneficial effects on cardiometabolic health and related inter-individual variability—An overview and perspective. Br. J. Nutr. 2020, 123, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Del Bo’, C.; Bernardi, S.; Marino, M.; Porrini, M.; Tucci, M.; Guglielmetti, S.; Cherubini, A.; Carrieri, B.; Kirkup, B.; Kroon, P.; et al. Systematic Review on Polyphenol Intake and Health Outcomes: Is there Sufficient Evidence to Define a Health-Promoting Polyphenol-Rich Dietary Pattern? Nutrients 2019, 11, 1355. [Google Scholar]
- ClinicalTrials Registry. Available online: https://clinicaltrials.gov (accessed on 30 April 2020).
- ISRCTN Registry. Available online: https://www.isrctn.com (accessed on 30 April 2020).
- Saaiq, M.; Ashraf, B. Modifying “Pico” Question into “Picos” Model for More Robust and Reproducible Presentation of the Methodology Employed in A Scientific Study. World J. Plast. Surg. 2017, 6, 390–392. [Google Scholar] [PubMed]
- Methley, A.M.; Campbell, S.; Chew-Graham, C.; McNally, R.; Cheraghi-Sohi, S. PICO, PICOS and SPIDER: A comparison study of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Serv. Res. 2014, 14, 579. [Google Scholar] [CrossRef]
- Guru Murthy, G.S.; Szabo, A.; Michaelis, L.; Carlson, K.-S.; Runaas, L.; Abedin, S.; Atallah, E. Improving Outcomes of Acute Promyelocytic Leukemia in the Current Era: Analysis of the SEER Database. J. Natl. Compr. Cancer Netw. 2020, 18, 169–175. [Google Scholar]
- Parikh, S.H.; Satwani, P.; Ahn, K.W.; Sahr, N.A.; Fretham, C.; Abraham, A.A.; Agrawal, V.; Auletta, J.J.; Abdel-Azim, H.; Copelan, E.; et al. Survival Trends in Infants Undergoing Allogeneic Hematopoietic Cell Transplant. JAMA Pediatr. 2019, 173, e190081. [Google Scholar] [CrossRef]
- Chen, Y.; Chomsky-Higgins, K.; Nwaogu, I.; Gosnell, J.E.; Seib, C.; Shen, W.T.; Suh, I.; Duh, Q.-Y. Trends in Adrenal Surgery—The Changing Nature of Tumors and Patients. J. Surg. Res. 2019, 236, 129–133. [Google Scholar] [CrossRef]
- Blanch, G.P.; Gómez-Jiménez, M.C.; Ruiz del Castillo, M.L. Enrichment of Olive Fruits in Antioxidant Content by Pre-Harvest Salicylic Acid Treatment. Foods 2020, 9, 1513. [Google Scholar] [CrossRef]
- Martini, D.; Taddei, F.; Nicoletti, I.; Ciccoritti, R.; Corradini, D.; D’Egidio, M.G. Effects of Genotype and Environment on Phenolic Acids Content and Total Antioxidant Capacity in Durum Wheat. Cereal Chem. J. 2014, 91, 310–317. [Google Scholar] [CrossRef]
- Struiving, S.; Hacke, A.C.M.; Simionatto, E.L.; Scharf, D.R.; Klimaczewski, C.V.; Besten, M.A.; Heiden, G.; Boligon, A.A.; Rocha, J.B.T.; Vellosa, J.C.R.; et al. Effects of Gender and Geographical Origin on the Chemical Composition and Antiradical Activity of Baccharis myriocephala and Baccharis trimera. Foods 2020, 9, 1433. [Google Scholar] [CrossRef] [PubMed]
- Alashi, A.M.; Taiwo, K.A.; Oyedele, D.J.; Adebooye, O.C.; Aluko, R.E. Polyphenol composition and antioxidant properties of vegetable leaf-fortified bread. J. Food Biochem. 2019, 43, e12625. [Google Scholar] [CrossRef] [PubMed]
- Antonic, B.; Jancikova, S.; Dordevic, D.; Tremlova, B. Apple pomace as food fortification ingredient: A systematic review and meta-analysis. J. Food Sci. 2020, 85, 2977–2985. [Google Scholar] [CrossRef]
- McDougall, G.J. Phenolic-enriched foods: Sources and processing for enhanced health benefits. Proc. Nutr. Soc. 2017, 76, 163–171. [Google Scholar] [CrossRef]
- Martini, D.; Ciccoritti, R.; Nicoletti, I.; Nocente, F.; Corradini, D.; D’Egidio, M.G.; Taddei, F. From seed to cooked pasta: Influence of traditional and non-conventional transformation processes on total antioxidant capacity and phenolic acid content. Int. J. Food Sci. Nutr. 2018, 69, 24–32. [Google Scholar] [CrossRef]
- Pycia, K.; Ivanišová, E. Physicochemical and Antioxidant Properties of Wheat Bread Enriched with Hazelnuts and Walnuts. Foods 2020, 9, 1081. [Google Scholar] [CrossRef] [PubMed]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef]
- Koch, W. Dietary Polyphenols—Important Non-Nutrients in the Prevention of Chronic Noncommunicable Diseases. A Systematic Review. Nutrients 2019, 11, 1039. [Google Scholar] [CrossRef]
- Rasouli, H.; Farzaei, M.H.; Khodarahmi, R. Polyphenols and their benefits: A review. Int. J. Food Prop. 2017, 1–42. [Google Scholar] [CrossRef]
- Manach, C.; Milenkovic, D.; Van de Wiele, T.; Rodriguez-Mateos, A.; de Roos, B.; Garcia-Conesa, M.T.; Landberg, R.; Gibney, E.R.; Heinonen, M.; Tomás-Barberán, F.; et al. Addressing the inter-individual variation in response to consumption of plant food bioactives: Towards a better understanding of their role in healthy aging and cardiometabolic risk reduction. Mol. Nutr. Food Res. 2017, 61, 1600557. [Google Scholar] [CrossRef]
- Bento-Silva, A.; Koistinen, V.M.; Mena, P.; Bronze, M.R.; Hanhineva, K.; Sahlstrøm, S.; Kitrytė, V.; Moco, S.; Aura, A.-M. Factors affecting intake, metabolism and health benefits of phenolic acids: Do we understand individual variability? Eur. J. Nutr. 2020, 59, 1275–1293. [Google Scholar] [CrossRef]
- Gross, G.; Jacobs, D.M.; Peters, S.; Possemiers, S.; van Duynhoven, J.; Vaughan, E.E.; van de Wiele, T. In Vitro Bioconversion of Polyphenols from Black Tea and Red Wine/Grape Juice by Human Intestinal Microbiota Displays Strong Interindividual Variability. J. Agric. Food Chem. 2010, 58, 10236–10246. [Google Scholar] [CrossRef] [PubMed]
- Martini, D.; Chiavaroli, L.; González-Sarrías, A.; Bresciani, L.; Palma-Duran, S.A.; Dall’Asta, M.; Deligiannidou, G.-E.; Massaro, M.; Scoditti, E.; Combet, E.; et al. Impact of Foods and Dietary Supplements Containing Hydroxycinnamic Acids on Cardiometabolic Biomarkers: A Systematic Review to Explore Inter-Individual Variability. Nutrients 2019, 11, 1805. [Google Scholar] [CrossRef]
- García-Conesa, M.-T.; Chambers, K.; Combet, E.; Pinto, P.; Garcia-Aloy, M.; Andrés-Lacueva, C.; de Pascual-Teresa, S.; Mena, P.; Konic Ristic, A.; Hollands, W.; et al. Meta-Analysis of the Effects of Foods and Derived Products Containing Ellagitannins and Anthocyanins on Cardiometabolic Biomarkers: Analysis of Factors Influencing Variability of the Individual Responses. Int. J. Mol. Sci. 2018, 19, 694. [Google Scholar]
- González-Sarrías, A.; García-Villalba, R.; Romo-Vaquero, M.; Alasalvar, C.; Örem, A.; Zafrilla, P.; Tomás-Barberán, F.A.; Selma, M.V.; Espín, J.C. Clustering according to urolithin metabotype explains the interindividual variability in the improvement of cardiovascular risk biomarkers in overweight-obese individuals consuming pomegranate: A randomized clinical trial. Mol. Nutr. Food Res. 2017, 61, 1600830. [Google Scholar]
- Lavefve, L.; Howard, L.R.; Carbonero, F. Berry polyphenols metabolism and impact on human gut microbiota and health. Food Funct. 2020, 11, 45–65. [Google Scholar] [CrossRef] [PubMed]
- Catalkaya, G.; Venema, K.; Lucini, L.; Rocchetti, G.; Delmas, D.; Daglia, M.; De Filippis, A.; Xiao, H.; Quiles, J.L.; Xiao, J.; et al. Interaction of dietary polyphenols and gut microbiota: Microbial metabolism of polyphenols, influence on the gut microbiota, and implications on host health. Food Front. 2020, 1, 109–133. [Google Scholar] [CrossRef]
- Mena, P.; Bresciani, L.; Brindani, N.; Ludwig, I.A.; Pereira-Caro, G.; Angelino, D.; Llorach, R.; Calani, L.; Brighenti, F.; Clifford, M.N.; et al. Phenyl-γ-valerolactones and phenylvaleric acids, the main colonic metabolites of flavan-3-ols: Synthesis, analysis, bioavailability, and bioactivity. Nat. Prod. Rep. 2019, 36, 714–752. [Google Scholar] [CrossRef]
- Corrêa, T.A.F.; Rogero, M.M.; Hassimotto, N.M.A.; Lajolo, F.M. The Two-Way Polyphenols-Microbiota Interactions and Their Effects on Obesity and Related Metabolic Diseases. Front. Nutr. 2019, 6, 188. [Google Scholar]
- European Food Safety Authority (EFSA) Panel on Dietetic Products, Nutrition and Allergies (NDA). General scientific guidance for stakeholders on health claim applications. EFSA J. 2016, 14, 4367. [Google Scholar]
- Gakidou, E.; Afshin, A.; Abajobir, A.A.; Abate, K.H.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abdulle, A.M.; Abera, S.F.; Aboyans, V.; et al. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1345–1422. [Google Scholar] [CrossRef]
- Petersen, K.S.; Flock, M.R.; Richter, C.K.; Mukherjea, R.; Slavin, J.L.; Kris-Etherton, P.M. Healthy Dietary Patterns for Preventing Cardiometabolic Disease: The Role of Plant-Based Foods and Animal Products. Curr. Dev. Nutr. 2017, 1, cdn.117.001289. [Google Scholar] [CrossRef]
- Angelino, D.; Godos, J.; Ghelfi, F.; Tieri, M.; Titta, L.; Lafranconi, A.; Marventano, S.; Alonzo, E.; Gambera, A.; Sciacca, S.; et al. Fruit and vegetable consumption and health outcomes: An umbrella review of observational studies. Int. J. Food Sci. Nutr. 2019, 70, 652–667. [Google Scholar] [CrossRef]
- Oak, M.-H.; Auger, C.; Belcastro, E.; Park, S.-H.; Lee, H.-H.; Schini-Kerth, V.B. Potential mechanisms underlying cardiovascular protection by polyphenols: Role of the endothelium. Free Radic. Biol. Med. 2018, 122, 161–170. [Google Scholar] [CrossRef]
- Schini-Kerth, V.B.; Auger, C.; Kim, J.-H.; Étienne-Selloum, N.; Chataigneau, T. Nutritional improvement of the endothelial control of vascular tone by polyphenols: Role of NO and EDHF. Pflüg. Arch. Eur. J. Physiol. 2010, 459, 853–862. [Google Scholar] [CrossRef]
- Foito, A.; McDougall, G.J.; Stewart, D. Evidence for Health Benefits of Berries. Annu. Plant Rev. Online 2018, 1, 105–148. [Google Scholar]
- Martini, D.; Marino, M.; Angelino, D.; Del Bo’, C.; Del Rio, D.; Riso, P.; Porrini, M. Role of berries in vascular function: A systematic review of human intervention studies. Nutr. Rev. 2019, 78, 189–206. [Google Scholar] [CrossRef]
- Del Bo’, C.; Martini, D.; Porrini, M.; Klimis-Zacas, D.; Riso, P. Berries and oxidative stress markers: An overview of human intervention studies. Food Funct. 2015, 6, 2890–2917. [Google Scholar] [CrossRef]
- Joseph, S.V.; Edirisinghe, I.; Burton-Freeman, B.M. Berries: Anti-inflammatory Effects in Humans. J. Agric. Food Chem. 2014, 62, 3886–3903. [Google Scholar] [CrossRef]
- Fahmy, H.; Hegazi, N.; El-Shamy, S.; Farag, M.A. Pomegranate juice as a functional food: A comprehensive review of its polyphenols, therapeutic merits, and recent patents. Food Funct. 2020, 11, 5768–5781. [Google Scholar] [CrossRef]
- Tenore, G.C.; Caruso, D.; Buonomo, G.; D’Urso, E.; D’Avino, M.; Campiglia, P.; Marinelli, L.; Novellino, E. Annurca (Malus pumila Miller cv. Annurca) apple as a functional food for the contribution to a healthy balance of plasma cholesterol levels: Results of a randomized clinical trial. J. Sci. Food Agric. 2017, 97, 2107–2115. [Google Scholar] [CrossRef]
- Hosseini, B.; Saedisomeolia, A.; Wood, L.G.; Yaseri, M.; Tavasoli, S. Effects of pomegranate extract supplementation on inflammation in overweight and obese individuals: A randomized controlled clinical trial. Complement. Ther. Clin. Pract. 2016, 22, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Fuster-Muñoz, E.; Roche, E.; Funes, L.; Martínez-Peinado, P.; Sempere, J.M.; Vicente-Salar, N. Effects of pomegranate juice in circulating parameters, cytokines, and oxidative stress markers in endurance-based athletes: A randomized controlled trial. Nutrition 2016, 32, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.-Y.; Chen, C.-Y.; Chen, G.-C.; Chen, W.-G.; Pan, A.; Pan, C.-W.; Zhang, Y.-H.; Qin, L.-Q.; Chen, L.-H. Effect of Flaxseed Intervention on Inflammatory Marker C-Reactive Protein: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2016, 8, 136. [Google Scholar] [CrossRef]
- Shoji, T.; Masumoto, S.; Moriichi, N.; Ohtake, Y.; Kanda, T. Administration of Apple Polyphenol Supplements for Skin Conditions in Healthy Women: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients 2020, 12, 1071. [Google Scholar] [CrossRef] [PubMed]
- Califf, R.M. Characteristics of Clinical Trials Registered in ClinicalTrials.gov, 2007–2010. JAMA 2012, 307, 1838. [Google Scholar] [CrossRef]
- Roumiantseva, D.; Carini, S.; Sim, I.; Wagner, T.H. Sponsorship and design characteristics of trials registered in ClinicalTrials.gov. Contemp. Clin. Trials 2013, 34, 348–355. [Google Scholar] [CrossRef]
- Turner, B.; Rajeshuni, N.; Tran, E.M.; Ludwig, C.A.; Tauqeer, Z.; Weeks, B.; Kinde, B.; Pershing, S. Characteristics of Ophthalmology Trials Registered in ClinicalTrials.gov, 2007–2018. Am. J. Ophthalmol. 2020, 211, 132–141. [Google Scholar] [CrossRef]
- Allareddy, V.; Rampa, S.; Masoud, M.I.; Lee, M.K.; Nalliah, R.; Allareddy, V. Overview of registered studies in orthodontics: Evaluation of the ClinicalTrials.gov registry. Am. J. Orthod. Dentofac. Orthop. 2014, 146, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Goswami, N.D.; Pfeiffer, C.D.; Horton, J.R.; Chiswell, K.; Tasneem, A.; Tsalik, E.L. The State of Infectious Diseases Clinical Trials: A Systematic Review of ClinicalTrials.gov. PLoS ONE 2013, 8, e77086. [Google Scholar] [CrossRef] [PubMed]
- Bourgeois, F.T. Outcome Reporting Among Drug Trials Registered in ClinicalTrials.gov. Ann. Intern. Med. 2010, 153, 158. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Tresserra-Rimbau, A.; Medina-Remón, A.; Pérez-Jiménez, J.; Martínez-González, M.A.A.; Covas, M.I.I.; Corella, D.; Salas-Salvadó, J.; Gómez-Gracia, E.; Lapetra, J.; Arós, F.; et al. Dietary intake and major food sources of polyphenols in a Spanish population at high cardiovascular risk: The PREDIMED study. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 953–959. [Google Scholar] [CrossRef]




| PICOS Item | Inclusion Criteria |
|---|---|
| Population | Healthy or diseased children, adults and/or older adults |
| Intervention | Food, extract or pure polyphenols tested alone. No other bioactive compound or drug |
| Comparison | Control group without polyphenols |
| Outcome | Any effect on human health and bioavailability |
| Study design | No restriction on study design |
| Foods (n = 315) | Extracts or Pure Compounds (n = 435) | |
|---|---|---|
| Goal | ||
| Health effect | 197 | 323 |
| Bioavailability | 33 | 22 |
| Both | 85 | 90 |
| Duration | ||
| Acute | 91 | 92 |
| Chronic | 194 | 316 |
| Both | 30 | 27 |
| Subjects | ||
| Healthy | 174 | 196 |
| Subject with risk factors | 84 | 165 |
| Patients | 57 | 74 |
| Primary outcome | ||
| Lipid profile and blood pressure | 74 | 65 |
| Vascular and endothelial function | 66 | 54 |
| Glucose and insulin parameters | 40 | 52 |
| Blood polyphenols’ concentration | 46 | 29 |
| Urinary polyphenols’ concentration | 36 | 26 |
| Cognitive function | 28 | 31 |
| Oxidative stress | 28 | 27 |
| Inflammation | 30 | 22 |
| Gut Microbiota | 18 | 8 |
| Safety and tolerability | 4 | 19 |
| Other | 98 | 122 |
| Location | ||
| USA | 94 | 136 |
| UK | 76 | 72 |
| Spain | 23 | 35 |
| Canada | 21 | 22 |
| Italy | 17 | 20 |
| Germany | 13 | 21 |
| China | 4 | 24 |
| Netherlands | 4 | 18 |
| France | 6 | 14 |
| Switzerland | 10 | 5 |
| Other | 47 | 68 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marino, M.; Del Bo’, C.; Martini, D.; Porrini, M.; Riso, P. A Review of Registered Clinical Trials on Dietary (Poly)Phenols: Past Efforts and Possible Future Directions. Foods 2020, 9, 1606. https://doi.org/10.3390/foods9111606
Marino M, Del Bo’ C, Martini D, Porrini M, Riso P. A Review of Registered Clinical Trials on Dietary (Poly)Phenols: Past Efforts and Possible Future Directions. Foods. 2020; 9(11):1606. https://doi.org/10.3390/foods9111606
Chicago/Turabian StyleMarino, Mirko, Cristian Del Bo’, Daniela Martini, Marisa Porrini, and Patrizia Riso. 2020. "A Review of Registered Clinical Trials on Dietary (Poly)Phenols: Past Efforts and Possible Future Directions" Foods 9, no. 11: 1606. https://doi.org/10.3390/foods9111606
APA StyleMarino, M., Del Bo’, C., Martini, D., Porrini, M., & Riso, P. (2020). A Review of Registered Clinical Trials on Dietary (Poly)Phenols: Past Efforts and Possible Future Directions. Foods, 9(11), 1606. https://doi.org/10.3390/foods9111606









