Development and Characterization of the Edible Packaging Films Incorporated with Blueberry Pomace
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Film Preparation
2.3. Color and Texture Analysis
2.4. Ultraviolet-Visible (UV-Vis) Spectroscopy
2.5. Gloss Measurements
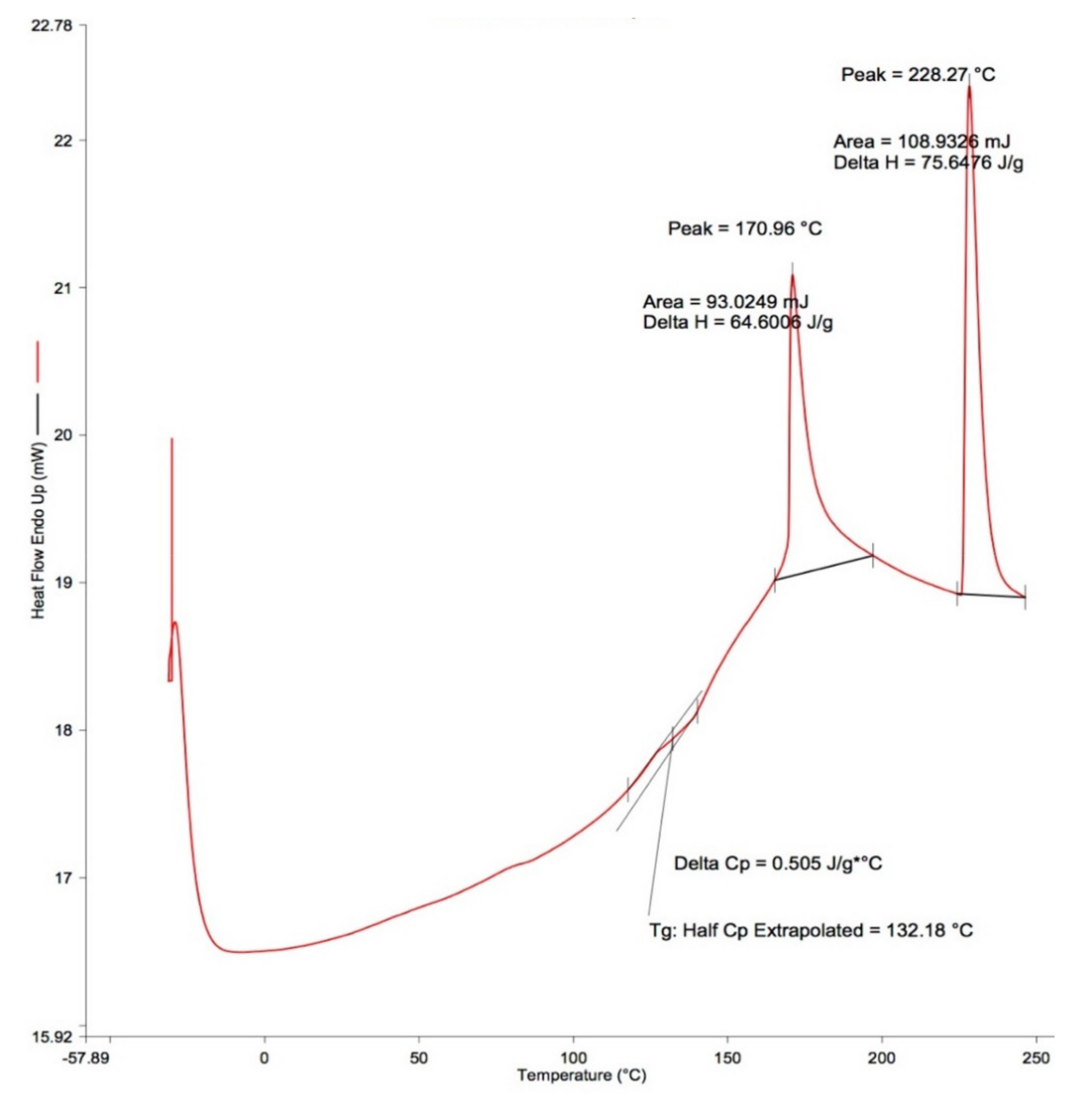
2.6. Differential Scanning Calorimetry (DSC)
2.7. Water Solubility
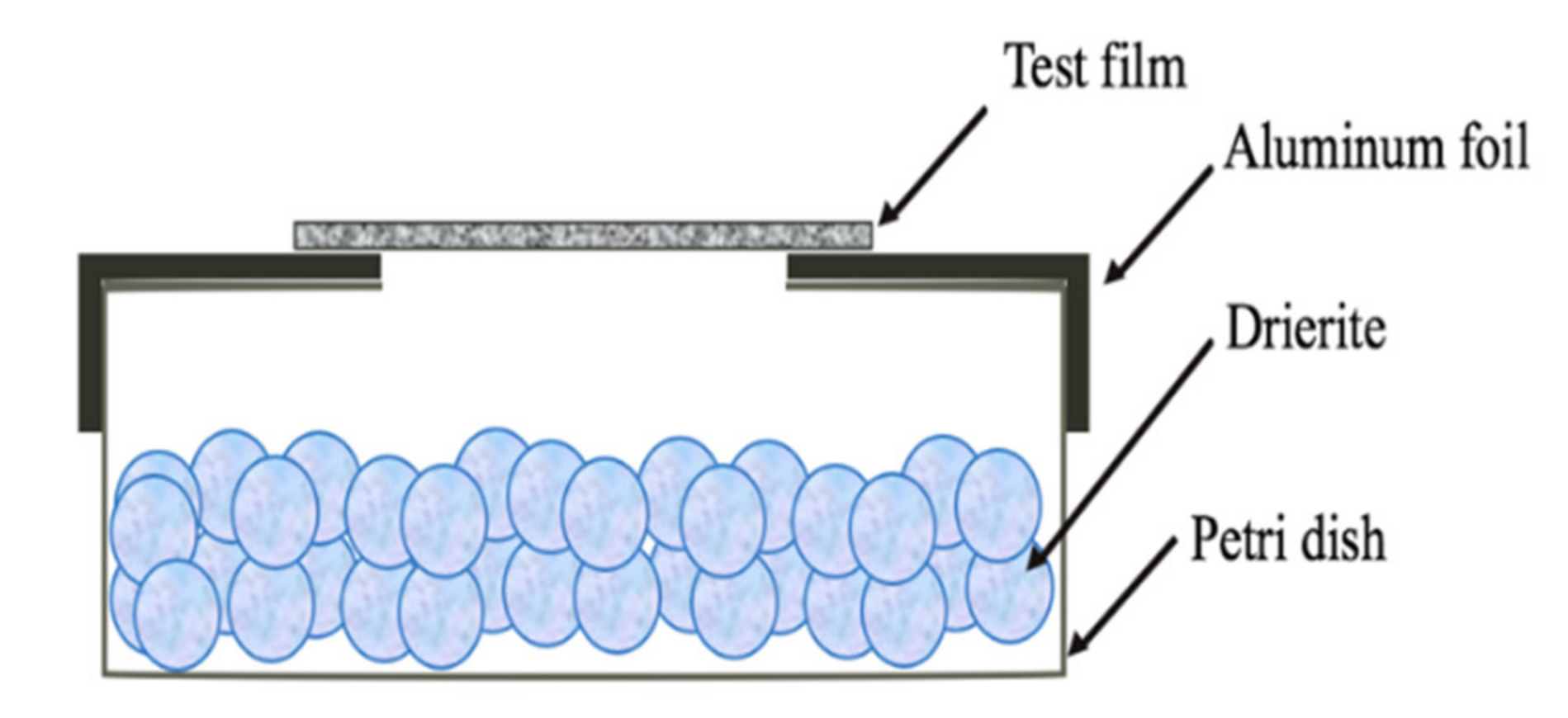
2.8. Water Vapour Transmission Rate (WVTR)
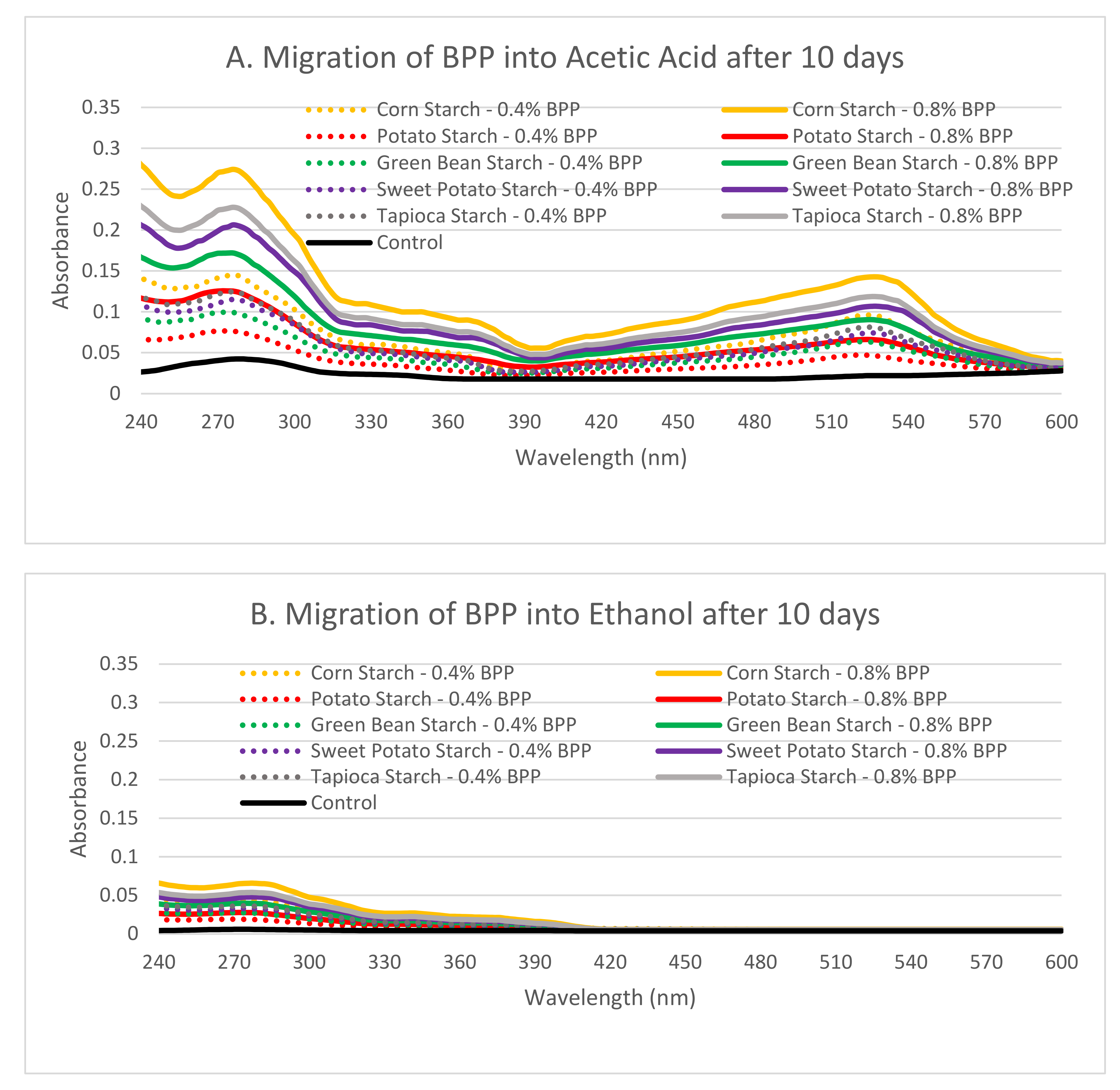
2.9. Migration Analysis
2.10. Statistical Analysis
3. Results
3.1. Optical and Physical Properties
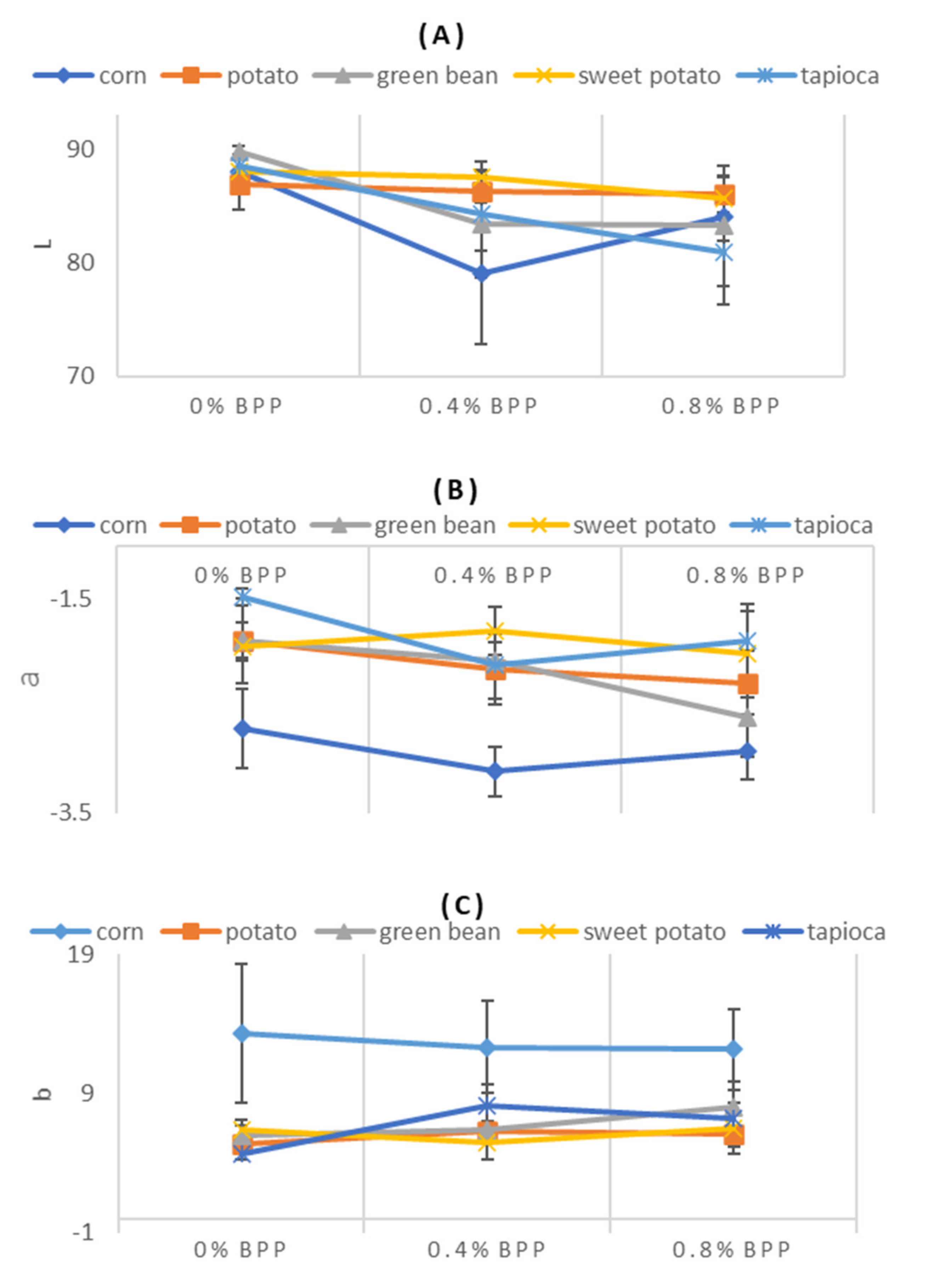
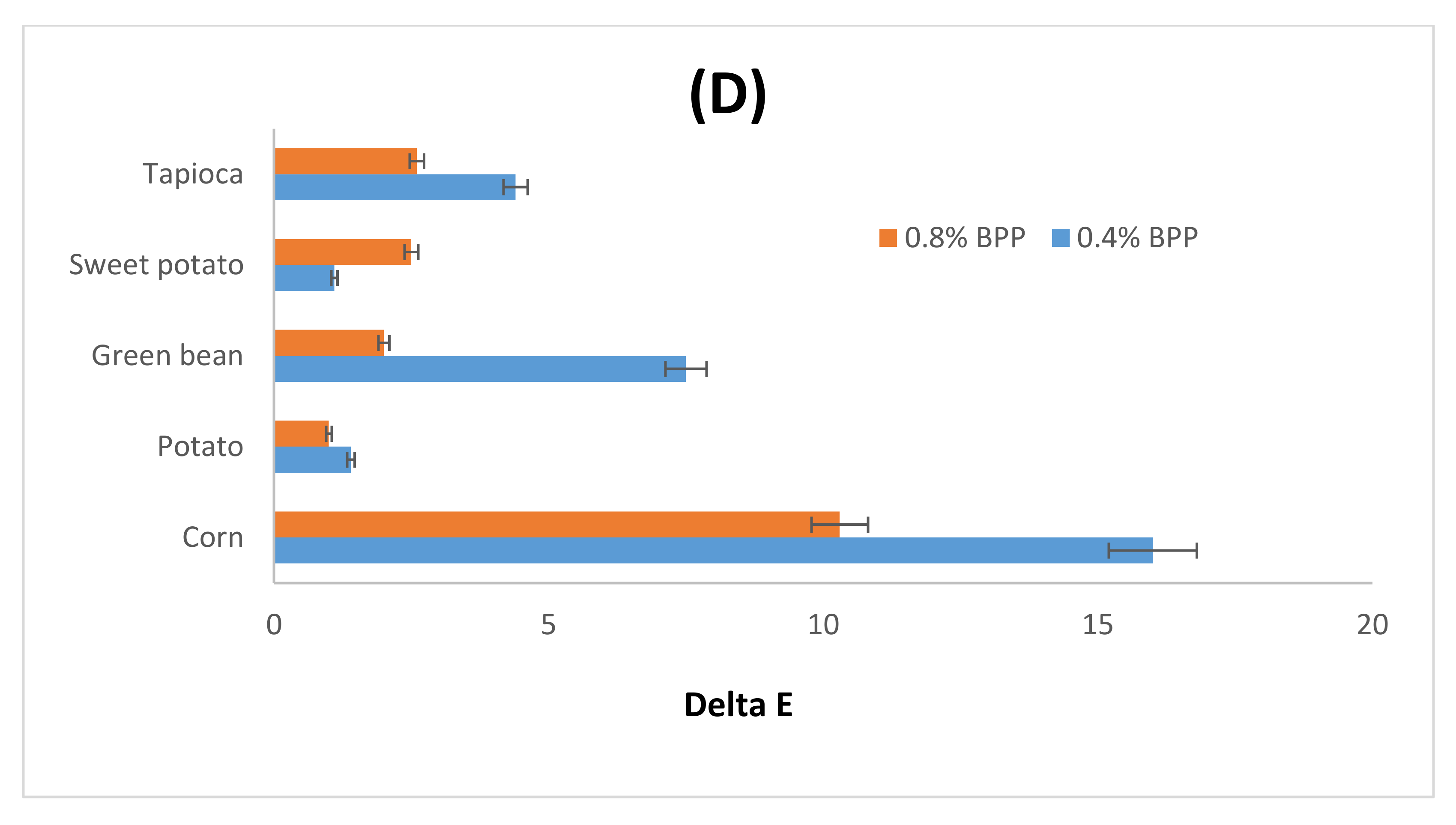
3.1.1. Color
3.1.2. Ultraviolet–Visible (UV-vis) Spectroscopy
3.1.3. Film Thickness and Transparency
3.1.4. Film Gloss
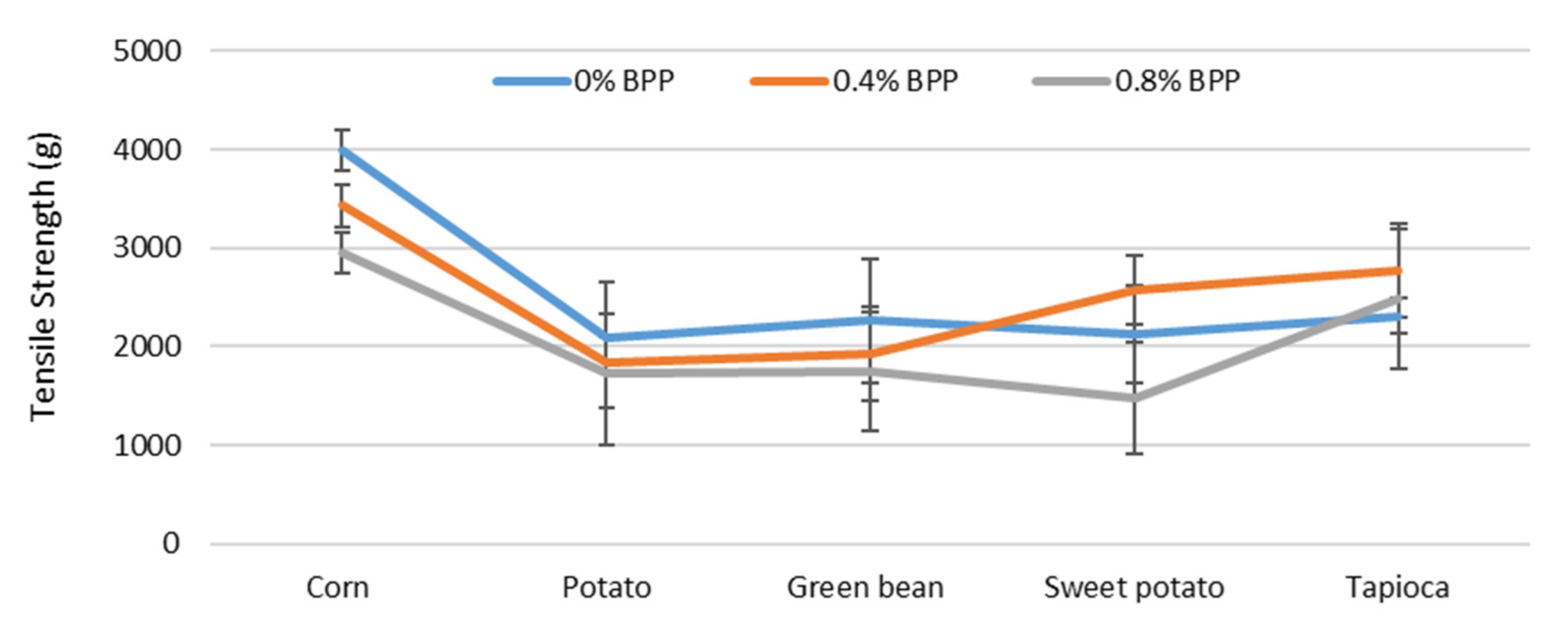
3.1.5. Tensile Strength (TS)
3.2. Physiochemical Properties (WVTR and Solubility)
3.3. Thermal Properties
3.4. Migration Assays
4. Discussion
4.1. Color
4.2. UV-Vis
4.3. Thickness and Transparency
4.4. Gloss
4.5. Tensile Strength (TS)
4.6. Physiochemical (WVTR and Solubility)
4.7. DSC Thermogram
4.8. Migration of Active Components of BPP Starch Films into Aqueous and Fatty Food Simulants
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Luchese, L.L.; Garrido, T.; Spada, J.C.; Tessaro, C.; Caba, K. Development and characterization of cassava starch films incorporated with blueberry pomace. Int. J. Biol. Macromol. 2018, 106, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, M.; Loganathan, S.; Valapa, R.B.; Thomas, S.B.; Varghese, T.O. UV protective polylactic acid/rosin films for sustainable packaging. Int. J. Biol. Macromol. 2017, 99, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Podshivalov, A.; Zakharova, M.; Glazacheva, E.; Uspenskaya, M. Gelatin/potato starch edible biocomposite films: Correlation between morphology and physical properties. Carbohydr. Polym. 2017, 157, 1162–1172. [Google Scholar] [CrossRef] [PubMed]
- Etxabide, A.; Coma, V.; Guerrero, P.; Gardrat, C.; Caba, K. Effect of cross-linking in surface properties and antioxidant activity of gelatin films incorporated with a curcumin derivative. Food Hydrocoll. 2017, 66, 168–175. [Google Scholar] [CrossRef]
- Nawab, A.; Alam, F.; Haq, M.A.; Lutfi, Z.; Hasnain, A. Mango kernel starch-gum composite films: Physical: Mechanical and barrier properties. Int. J. Biol. Macromol. 2017, 98, 869–876. [Google Scholar] [CrossRef]
- Das, D.K.; Dutta, H.; Mahanta, C.L. Development of a rice starch-based coating with antioxidant and microbe-barrier properties and study of its effect on tomatoes stored at room temperature. LWT-Food Sci. Technol. 2013, 50, 272–278. [Google Scholar] [CrossRef]
- Realini, C.E.; Marcos, B. Active and intelligent packaging systems for a modern society. Meat Sci. 2014, 98, 404–419. [Google Scholar] [CrossRef]
- Bonilla, J.; Atarés, L.; Vargas, M.; Chiralt, A. Properties of wheat starch film-forming dispersions and films as affected by chitosan addition. J. Food Eng. 2013, 114, 303–312. [Google Scholar] [CrossRef]
- Basiak, E.; Lenart, A.; Debeaufort, F. Effect of starch type on the physicochemical properties of edible films. Int. J. Biol. Macromol. 2017, 98, 348–356. [Google Scholar] [CrossRef]
- Gutiérrez, T.J.; Tapia, M.S.; Pérez, E.; Famá, L. Structural and mechanical properties of edible films made from native and modified cush-cush yam and cassava starch. Food Hydrocoll. 2015, 45, 211–217. [Google Scholar] [CrossRef]
- Jan, K.N.; Panesar, P.S.; Rana, J.C.; Singh, S. Structural: Thermal and rheological properties of starches isolated from Indian quinoa varieties. Int. J. Biol. Macromol. 2017, 102, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Bunea, A.; Rugină, D.; Sconţa, Z.; Pop, R.M.; Pintea, A.; Socaciu, C.; Tăbăran, F.; Grootaert, C.; Struijs, K.; Vancamp, J. Anthocyanin determination in blueberry extracts from various cultivars and their antiproliferative and apoptotic properties in B16-F10 metastatic murine melanoma cells. Phytochemistry 2013, 95, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Wang, P.; Zhu, Y.; Liu, X.; Hu, X.; Che, F. Blueberry anthocyanins extract inhibits acrylamide-induced diverse toxicity in mice by preventing oxidative stress and cytochrome P450 2E1 activation. J. Funct. Foods 2015, 14, 95–101. [Google Scholar] [CrossRef]
- Choi, I.; Lee, J.Y.; Lacroix, M.; Han, J. Intelligent pH indicator film composed of agar/potato starch and anthocyanin extracts from purple sweet potato. Food Chem. 2017, 218, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Bourtoom, T.; Chinnan, M.S. Preparation and properties of rice starch–chitosan blend biodegradable film. LWT-Food Sci. Technol. 2018, 41, 1633–1641. [Google Scholar] [CrossRef]
- Chakravartula, S.S.N.; Soccio, M.; Lotti, N.; Balestra, F.; Dalla Rosa, M.; Siracusa, V. Characterization of Composite Edible Films Based on Pectin/Alginate/Whey Protein Concentrate. Materials 2019, 12, 2454. [Google Scholar] [CrossRef]
- Hu, Y.; Topolkaraev, V.; Hiltner, A.; Baer, E. Measurement of water vapor transmission rate in highly permeable films. J. Appl. Polymer Sci. 2001, 81, 1624–1633. [Google Scholar] [CrossRef]
- Yap, A.U.J.; Sim, C.P.C.; Loh, W.L.; Teo, J.H. Human-eye versus computerized color matching. Oper. Dent. 1999, 24, 358–363. [Google Scholar]
- Lopez, O.; Garcia, M.A.; Villar, M.A.; Gentili, A.; Rodriguez, M.S.; Albertengo, L. Thermo-compression of biodegradable thermoplastic corn starch films containing chitin and chitosan. LWT-Food Sci. Technol. 2014, 57, 106–115. [Google Scholar] [CrossRef]
- Gaikwad, K.K.; Lee, J.Y.; Lee, Y.S. Development of polyvinyl alcohol and apple pomace bio-composite film with antioxidant properties for active food packaging application. J. Food Sci. Technol. 2016, 53, 1608–1619. [Google Scholar] [CrossRef]
- Fakhouri, F.M.; Martelli, S.M.; Bertan, L.C.; Yamashita, F.; Mei, L.H.I.; Queiroz, F.P.C. Edible films made from blends of manioc starch and gelatin–Influence of different types of plasticizer and different levels of macromolecules on their properties. LWT-Food Sci. Technol. 2012, 49, 149–154. [Google Scholar] [CrossRef]
- Al-Hassan, A.A.; Norziah, M.H. Starch–gelatin edible films: Water vapor permeability and mechanical properties as affected by plasticizers. Food Hydrocol. 2012, 26, 108–117. [Google Scholar] [CrossRef]
- Wang, K.; Wang, W.; Ye, R.; Liu, A.; Xiao, J.; Liu, Y.; Zhao, Y. Mechanical properties and solubility in water of corn starch-collagen composite films: Effect of starch type and concentrations. Food Chem. 2017, 216, 209–216. [Google Scholar] [CrossRef]
- Collado, L.S.; Corke, H. Heat-moisture treatment effects on sweet potato starches differing in amylose content. Food Chem. 1999, 65, 339–346. [Google Scholar] [CrossRef]
- Singh, A.; Pratap-Singh, A.; Ramaswamy, H.S. A Controlled Agitation Process for Improving Quality of Canned Green Beans during Agitation Thermal Processing. J. Food Sci. 2016, 81, E1399–E1411. [Google Scholar] [CrossRef]
- Gutiérrez, T.; Alvarez, V.A. Bionanocomposite films developed from corn starch and natural and modified nano-clays with or without added blueberry extract. Food Hydrocol. 2018, 77, 407–420. [Google Scholar] [CrossRef]
- Kampeerapappun, P.; Aht-ong, D.; Pentrakoon, D.; Srikulkit, K. Preparation of cassava starch/montmorillonite composite film. Carbohydr. Polym. 2007, 67, 155–163. [Google Scholar] [CrossRef]
- Mali, S.; Grossmann, M.V.E.; García, M.A.; Martino, M.N.; Zaritzky, N.E. Barrier, mechanical and optical properties of plasticized yam starch films. Carbohydr. Polym. 2004, 56, 129–135. [Google Scholar] [CrossRef]
- Ban, W.; Song, J.; Argyropoulos, D.S.; Lucia, L.A. The Influence of Biomaterials on the Elastic Properties of Starch-Derived Films: An Optimization Study. Ind. Eng. Chem. Res. 2006, 45, 627–633. [Google Scholar] [CrossRef]
- Trezza, T.A.; Krochta, J.M. The gloss of edible coatings as affected by surfactants, lipids, relative humidity, and time. J. Food Sci. 2000, 65, 658–662. [Google Scholar] [CrossRef]
- Bangyekan, C.; Aht-Ong, D.; Srikulkit, K. Preparation and properties evaluation of chitosan-coated cassava starch films. Carbohydr. Polym. 2006, 63, 61–71. [Google Scholar] [CrossRef]
- Xu, Y.X.; Kim, K.M.; Hanna, M.A.; Nag, D. Chitosan–starch composite film: Preparation and characterization. Ind. Crops Prod. 2005, 21, 185–192. [Google Scholar] [CrossRef]
- Mathew, S.; Abraham, T.E. Characterization of ferulic acid incorporated starch–chitosan blend films. Food Hydrocol. 2008, 22, 826–835. [Google Scholar] [CrossRef]
- Khanal, R.C.; Howard, L.R.; Prior, R.L. Effect of heating on the stability of grape and blueberry pomace procyanidins and total anthocyanins. Food Res. Int. 2010, 43, 1464–1469. [Google Scholar] [CrossRef]
- Teixeira, E.D.M.; Da Róz, A.L.; Carvalho, A.J.F.D.; Curvelo, A.A.D.S. The effect of glycerol/sugar/water and sugar/water mixtures on the plasticization of thermoplastic cassava starch. Carbohydr. Polym. 2007, 69, 619–624. [Google Scholar] [CrossRef]
- Reesha, K.V.; Panda, S.K.; Bindu, J.; Varghese, T.O. Development and characterization of an LDPE/chitosan composite antimicrobial film for chilled fish storage. Int. J. Biol. Macromol. 2015, 79, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.A.; Martino, M.N.; Zaritzky, N.E. Lipid addition to improve barrier properties of edible starch-based films and coatings. J. Food Sci. 2000, 65, 941–944. [Google Scholar] [CrossRef]
- Borah, P.P.; Das, P.; Badwaik, L.S. Ultrasound treated potato peel and sweet lime pomace based biopolymer film development. Ultrason. Sonochem. 2017, 36, 11–19. [Google Scholar] [CrossRef] [PubMed]
- Park, S.I.; Zhao, Y. Incorporation of a high concentration of mineral or vitamin into chitosan-based films. J. Agric. Food Chem. 2004, 52, 1933–1939. [Google Scholar] [CrossRef]
- Li, F.H.; Chen, Y.M.; Li, L.; Bai, X.L.; Li, S. Starch-chitosan blend films prepared by glutaraldehyde cross-linking. Adv. Mater. Res. 2012, 415, 1626–1629. [Google Scholar] [CrossRef]
- Rachtanapun, P.; Tongdeesoontorn, W. Effect of antioxidants on properties of rice flour/cassava starch film blends plasticized with sorbitol. Nat. Sci. 2009, 43, 252–258. [Google Scholar]
- Bizot, H.; Le Bail, P.; Leroux, B.; Davy, J.; Roger, P.; Buleon, A. Calorimetric evaluation of the glass transition in hydrated, linear and branched polyanhydro glucose compounds. Carbohydr. Polym. 1997, 32, 33–50. [Google Scholar] [CrossRef]
- Haque, M.K.; Roos, Y.H. Water sorption and plasticization behaviour of spray-dried lactose/protein mixtures. J. Food Sci. 2004, 69, E384–E391. [Google Scholar] [CrossRef]
- Sin, L.T.; Rahman, W.A.; Rahmat, A.R.; Khan, M.I. Detection of synergistic interactions of polyvinyl alcohol–cassava starch blends through DSC. Carbohydr. Polym. 2010, 79, 224–226. [Google Scholar] [CrossRef]
- Zhu, Z.; Guo, W. Frequency, moisture content, and temperature dependent dielectric properties of potato starch related to drying with radiofrequency/microwave energy. Sci. Rep. 2017, 7, 9311. [Google Scholar] [CrossRef]
- Markakis, P.C.; Francis, F.J. Food colorants: Anthocyanins. Crit. Rev. Food Sci. Nutr. 1989, 28, 273–314. [Google Scholar]
- Buonocore, G.G.; Del Nobile, M.A.; Panizza, A.; Corbo, M.R.; Nicolais, L. A general approach to describe the antimicrobial agent release from highly swellable films intended for food packaging applications. J. Control. Release 2003, 90, 97–107. [Google Scholar] [CrossRef]
| Starch Type * | |||||
|---|---|---|---|---|---|
| BPP (%) | Corn | Potato | Green Bean | Sweet Potato | Tapioca |
| 0 | 58.7 ± 4.4 a,1 | 114.4 ± 4.4 a,2 | 50.3 ± 1.9 a,1 | 60.8 ± 3.4 a,1 | 132.2 ± 1.3 a,2 |
| 0.4 | 86.3 ± 1.4 a,1 | 118.7 ± 8.3 a,1 | 64.3 ± 2.9 a,2 | 48.3 ± 3.8 a,2 | 117.4 ± 4.7 a,1 |
| 0.8 | 164.1 ± 1.7 b,1 | 45.7 ± 18.5 b,2 | 150.9 ± 1.6 b,1 | 72.7 ± 3.8 b,2 | 111.8 ± 2.1 a,2 |
| Thickness (mm) | |||||
|---|---|---|---|---|---|
| BPP (%) | Corn | Potato | Green Bean | Sweet Potato | Tapioca |
| 0 | 0.111 ± 0.01 a,1 | 0.111 ± 0.09 a,1 | 0.090 ± 0.02 a,1 | 0.121 ± 0.04 a,1 | 0.130 ± 0.06 a,1 |
| 0.4 | 0.118 ± 0.02 a,1 | 0.118 ± 0.04 a,1 | 0.128 ± 0.01 a,1 | 0.109 ± 0.09 a,1 | 0.120 ± 0.01 a,1 |
| 0.8 | 0.131 ± 0.01 a,1 | 0.131 ± 0.05 a,1 | 0.123 ± 0.01 a,1 | 0.132 ± 0.03 a,1 | 0.127 ± 0.02 a,1 |
| Transparency | |||||
| 0 | 1.02 ± 0.05 a,1 | 0.53 ± 0.02 a,2 | 0.75 ± 0.10 a,1 | 0.63 ± 0.04 a,2 | 0.47 ± 0.03 a,2 |
| 0.4 | 0.71 ± 0.10 b,1 | 0.63 ± 0.01 a,1 | 0.54 ± 0.02 a,1 | 0.64 ± 0.07 a,1 | 0.59 ± 0.03 a,1 |
| 0.8 | 0.74 ± 0.07 b,1 | 0.48 ± 0.17 a,1 | 0.64 ± 0.04 a,1 | 0.65 ± 0.02 a,1 | 0.63 ± 0.03 a,1 |
| (A) | |||||
|---|---|---|---|---|---|
| BPP (%) | Corn | Potato | Green Bean | Sweet Potato | Tapioca |
| 0 | 42.1 ± 1.6 a,1 | 42.0 ± 4.8 a,1 | 49.0 ± 1.3 a,1 | 42.8 ± 2.5 a,1 | 74.1 ± 2.8 a,2 |
| 0.4 | 44.7 ± 1.8 a,1 | 57.5 ± 3.9 a,2 | 40.5 ± 2.0 a,1 | 42.0 ± 9.9 a,1 | 43.1 ± 1.8 b,1 |
| 0.8 | 36.3 ± 3.9 b,1 | 34.5 ± 2.8 a,1 | 43.4 ± 3.5 a,1 | 43.2 ± 2.0 a,1 | 37.5 ± 1.2 c,1 |
| (B) | |||||
| BPP (%) | Corn | Potato | Green Bean | Sweet Potato | Tapioca |
| 0 | 18.6 ± 1.6 a,1 | 54.0 ± 1.8 a,2 | 66.6 ± 3.7 a,2 | 34.3 ± 9.1 a,3 | 38.5 ± 1.4 a,3 |
| 0.4 | 26.0 ± 6.2 a,1 | 76.9 ± 9.6 b,2 | 42.5 ± 1.6 a,3 | 50.5 ± 1.1 a,3 | 51.8 ± 5.0 a,3 |
| 0.8 | 25.2 ± 0.5 a,1 | 79.5 ± 1.2 b,2 | 53.2 ± 0.7 a,3 | 55.6 ± 7.5 a,3 | 43.6 ± 7.9 a,3 |
| (A) | |||||
|---|---|---|---|---|---|
| WVTR | |||||
| BPP (%) | Corn | Potato | Green Bean | Sweet Potato | Tapioca |
| 0 | 0.00170 a,1 | 0.00055 a,1 | 0.00170 a,1 | 0.00125 a,1 | 1.36000 a,2 |
| 0.4 | 0.00115 a,1 | 0.00065 a,1 | 0.00170 a,1 | 0.00285 a,1 | 0.59649 a,2 |
| 0.8 | 0.00105 a,1 | 0.00070 a,1 | 0.00300 a,1 | 0.00115 a,1 | 2.60870 a,2 |
| (B) | |||||
| BPP | Solubility | ||||
| 0 | 29.2% a,1 | 42.0% a,1 | 37.0% a,1 | 31.6% a.1 | 33.6% a,1 |
| 0.4 | 36.6% a,1 | 37.1% a,1 | 31.5% a,1 | 28.8% a,1 | 32.6% a,1 |
| 0.8 | 24.1% a,1 | 32.5% a,2 | 26.6% b,1 | 32.1% a,2 | 35.3% a,2 |
| BPP (%) | Corn | Potato | Green Bean | Sweet Potato | Tapioca |
|---|---|---|---|---|---|
| The Glass Transition Temperature, Tg (°C) | |||||
| 0 | 129.6 a | 143.1 a | 132.4 a | 124.8 a | 129.6 a |
| 0.4 | 145.7 a | 134.0 a | 141.6 a | 143.9 a | 147.4 a |
| 0.8 | 136.7 a | 143.3 a | 151.5 a | 137.7 a | 140.6 a |
| Enthalpy from the 1st Peak, ΔH1 (J/g) | |||||
| 0 | 30.6 a | 27.4 a | 42.2 a | 29.7 a | 26.3 a |
| 0.4 | 27.7 a | 23.1 a | 32.9 a | 29.1 a | 27.5 a |
| 0.8 | 27.3 a | 26.4 a | 17.6 a | 37.5 a | 31.9 a |
| Enthalpy from the 2nd Peak, ΔH2 (J/g) | |||||
| 0 | 50.9 a | 54.7 a | 73.1 a | 56.6 a | 48.6 a |
| 0.4 | 52.0 a | 48.1 a | 54.5 a | 54.1 a | 43.7 a |
| 0.8 | 42.0 b | 43.3 a | 38.4 b | 61.3 a | 49.1 a |
| Melting Temperatures from the First Peak, Tm1 (°C) | |||||
| 0 | 175.5 a | 175.2 a | 173.1 a | 174.2 a | 173.9 b |
| 0.4 | 175.0 a | 176.7 a | 172.6 a | 175.9 a | 177.1 b |
| 0.8 | 178.2 a | 179.9 a | 180.3 b | 172.6 a | 175.2 b |
| Melting Temperatures from the Second Peak, Tm2 (°C) | |||||
| 0 | 234.7 a | 242.9 a | 204.8 a | 238.6 a | 227.3 a |
| 0.4 | 240.2 a | 236.1 a | 214.3 a | 212.1 a | 237.9 a |
| 0.8 | 235.2 a | 236.2 a | 239.3 a | 226.4 a | 224.9 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, A.; Gu, Y.; Castellarin, S.D.; Kitts, D.D.; Pratap-Singh, A. Development and Characterization of the Edible Packaging Films Incorporated with Blueberry Pomace. Foods 2020, 9, 1599. https://doi.org/10.3390/foods9111599
Singh A, Gu Y, Castellarin SD, Kitts DD, Pratap-Singh A. Development and Characterization of the Edible Packaging Films Incorporated with Blueberry Pomace. Foods. 2020; 9(11):1599. https://doi.org/10.3390/foods9111599
Chicago/Turabian StyleSingh, Anika, Yixin Gu, Simone D. Castellarin, David D. Kitts, and Anubhav Pratap-Singh. 2020. "Development and Characterization of the Edible Packaging Films Incorporated with Blueberry Pomace" Foods 9, no. 11: 1599. https://doi.org/10.3390/foods9111599
APA StyleSingh, A., Gu, Y., Castellarin, S. D., Kitts, D. D., & Pratap-Singh, A. (2020). Development and Characterization of the Edible Packaging Films Incorporated with Blueberry Pomace. Foods, 9(11), 1599. https://doi.org/10.3390/foods9111599






