Potentials and Safety of Date Palm Fruit against Diabetes: A Critical Review
Abstract
1. Introduction
2. Materials and Methods
3. Physicochemical and Nutritional Composition
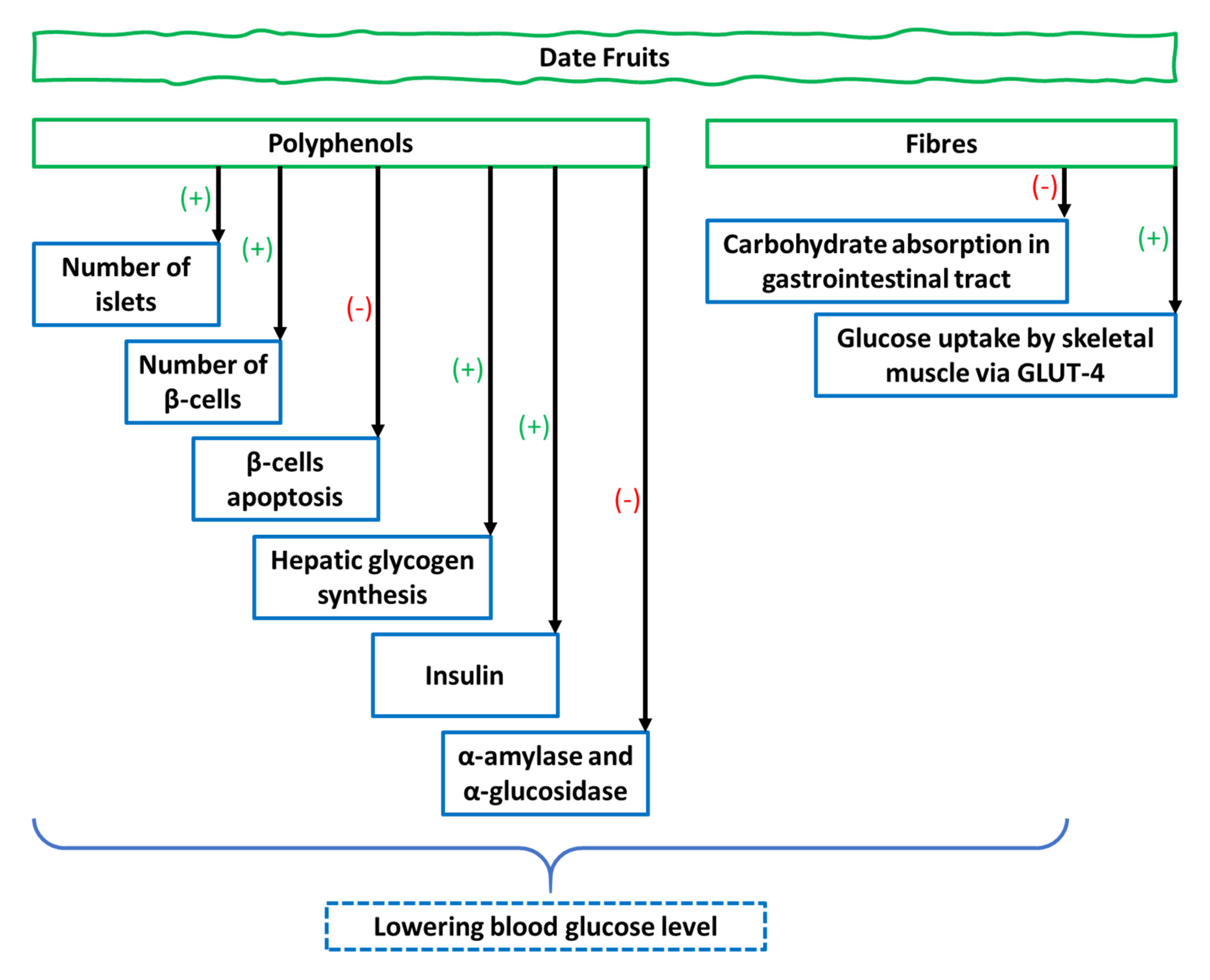
4. Date Fruit Flesh
4.1. Evidence from In Vivo Studies
4.1.1. Antihyperglycemic Activity
4.1.2. Antihyperglycemic Activity
4.1.3. Against Diabetes-Induced Testicular Toxicity
4.1.4. Against Diabetes-Induced Cardiomyopathy
4.1.5. Ameliorating Haematological Parameters
4.1.6. Neuroprotective Activity
4.2. Evidence from Clinical Studies
4.2.1. Antihyperglycemic Activity
4.2.2. Antihyperlipidemic Activity
4.2.3. Ameliorating Liver Function
5. Date Fruit Seed
5.1. Evidence from In Vivo Studies
5.1.1. Antihyperglycemic Activity
5.1.2. Antihyperlipidemic Activity
5.1.3. Against Diabetes-Induced Testicular Toxicity
5.1.4. Ameliorating Liver and Kidney Functions
5.2. Evidence from Clinical Studies
Antihyperglycemic Activity
6. Date Fruit Leaf
6.1. Evidence from In Vivo Studies
6.1.1. Antihyperglycemic Activity
6.1.2. Antihyperlipidemic Activity
6.1.3. Ameliorating Haematological Parameters
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Islam, M.A.; Alam, F.; Solayman, M.; Khalil, M.I.; Kamal, M.A.; Gan, S.H. Dietary phytochemicals: Natural swords combating inflammation and oxidation-mediated degenerative diseases. Oxid. Med. Cell. Longev. 2016, 2016, 1–25. [Google Scholar] [CrossRef]
- Solayman, M.; Ali, Y.; Alam, F.; Islam, M.A.; Alam, N.; Khalil, M.I.; Gan, S.H. Polyphenols: Potential future arsenals in the treatment of diabetes. Curr. Pharm. Des. 2016, 22, 549–565. [Google Scholar] [CrossRef] [PubMed]
- Alam, F.; Islam, M.A.; Mohamed, M.; Ahmad, I.; Kamal, M.A.; Donnelly, R.; Idris, I.; Gan, S.H. Efficacy and safety of pioglitazone monotherapy in type 2 diabetes mellitus: A systematic review and meta-analysis of randomised controlled trials. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Alam, F.; Islam, M.A.; Kamal, M.A.; Gan, S.H. Updates on managing type 2 diabetes mellitus with natural products: Towards antidiabetic drug development. Curr. Med. Chem. 2018, 25, 5395–5431. [Google Scholar] [CrossRef] [PubMed]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The role of polyphenols in human health and food systems: A mini-review. Front. Nutr. 2018, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Souli, I.; Jemni, M.; Rodríguez-Verástegui, L.L.; Chaira, N.; Artés, F.; Ferchichi, A. Phenolic composition profiling of Tunisian 10 varieties of common dates (Phoenix dactylifera L.) at tamar stage using LC-ESI-MS and antioxidant activity. J. Food Biochem. 2018, 42, e12634. [Google Scholar] [CrossRef]
- Hussain, M.I.; Farooq, M.; Syed, Q.A. Nutritional and biological characteristics of the date palm fruit (Phoenix dactylifera L.)–A review. Food Biosci. 2020, 34, 100509. [Google Scholar] [CrossRef]
- Al-Farsi, M.A.; Lee, C.Y. Nutritional and functional properties of dates: A review. Crit. Rev. Food Sci. Nutr. 2008, 48, 877–887. [Google Scholar] [CrossRef]
- Dayang, J.; Reuben, C.; Raji, F. Nutritional, socioeconomic and health benefits of dates. Int. J. Food Nutr. Sci. 2014, 3, 63–73. [Google Scholar]
- El Abed, H.; Chakroun, M.; Fendri, I.; Makni, M.; Bouaziz, M.; Drira, N.; Mejdoub, H.; Khemakhem, B. Extraction optimization and in vitro and in vivo anti-postprandial hyperglycemia effects of inhibitor from Phoenix dactylifera L. parthenocarpic fruit. Biomed. Pharmacother. 2017, 88, 835–843. [Google Scholar] [CrossRef]
- Farag, M.A.; Porzel, A.; Wessjohann, L.A. Unraveling the active hypoglycemic agent trigonelline in Balanites aegyptiaca date fruit using metabolite fingerprinting by NMR. J. Pharm. Biomed. Anal. 2015, 115, 383–387. [Google Scholar] [CrossRef]
- Al-Zuhair, S.; Dowaidar, A.; Kamal, H. Inhibitory effect of dates-extract on α-Amylase and β-glucosidase enzymes relevant to non-insulin dependent diabetes mellitus. J. Biochem. Technol. 2010, 2, 158–160. [Google Scholar]
- Eddine, L.S.; Segni, L.; Noureddine, G.; Redha, O.M.; Sonia, M. Scavenging Effect, Anti-Inflammatory and Diabetes Related Enzyme Inhibition Properties of Leaves Extract from Selected Varieties of Phoenix dactylifera L. Int. J. Pharmacogn. Phytochem. Res. 2014, 6, 66–73. [Google Scholar]
- Chaudhary, S.; Pankaj, A. Dates and Diabetes. J. Soc. Health Diabetes 2018, 6, 109–110. [Google Scholar] [CrossRef][Green Version]
- Alkaabi, J.M.; Al-Dabbagh, B.; Ahmad, S.; Saadi, H.F.; Gariballa, S.; Al Ghazali, M. Glycemic indices of five varieties of dates in healthy and diabetic subjects. Nutr. J. 2011, 10, 1–9. [Google Scholar] [CrossRef]
- Mrabet, A.; Hammadi, H.; Rodríguez-Gutiérrez, G.; Jiménez-Araujo, A.; Sindic, M. Date Palm Fruits as a Potential Source of Functional Dietary Fiber: A Review. Food Sci. Technol. Res. 2019, 25, 1–10. [Google Scholar] [CrossRef]
- Mrabet, A.; Jiménez-Araujo, A.; Guillén-Bejarano, R.; Rodríguez-Arcos, R.; Sindic, M. Date Seeds: A Promising Source of Oil with Functional Properties. Foods 2020, 9, 787. [Google Scholar]
- Alqarni, M.M.; Osman, M.A.; Al-Tamimi, D.S.; Gassem, M.A.; Al-Khalifa, A.S.; Al-Juhaimi, F.; Mohamed Ahmed, I.A. Antioxidant and antihyperlipidemic effects of Ajwa date (Phoenix dactylifera L.) extracts in rats fed a cholesterol-rich diet. J. Food Biochem. 2019, 43, e12933. [Google Scholar] [CrossRef]
- Khan, F.; Khan, T.J.; Kalamegam, G.; Pushparaj, P.N.; Chaudhary, A.; Abuzenadah, A.; Kumosani, T.; Barbour, E.; Al-Qahtani, M. Anti-cancer effects of Ajwa dates (Phoenix dactylifera L.) in diethylnitrosamine induced hepatocellular carcinoma in Wistar rats. BMC Complement. Altern. Med. 2017, 17, 418. [Google Scholar] [CrossRef]
- Maqsood, S.; Adiamo, O.; Ahmad, M.; Mudgil, P. Bioactive compounds from date fruit and seed as potential nutraceutical and functional food ingredients. Food Chem. 2020, 308, 125522. [Google Scholar] [CrossRef]
- Saryono, S. Date Seeds Drinking as Antidiabetic: A Systematic Review. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Purwokerto, Central Java, Indonesia, 14–16 November 2018; p. 012018. [Google Scholar]
- Taha, R.; Ben Maachia, S.; Sindic, M.; Sahli, A.; Namsi, A.; Messaoud, M. Chemical fruit composition of Tunisian date palm ‘Deglet Nour’ collected at maturation from four different oases in Djerid region. J. Food Technol. 2019, 17, 1–10. [Google Scholar]
- Siddeeg, A.; Zeng, X.-A.; Ammar, A.-F.; Han, Z. Sugar profile, volatile compounds, composition and antioxidant activity of Sukkari date palm fruit. J. Food Sci. Technol. 2019, 56, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Nehdi, I.; Omri, S.; Khalil, M.; Al-Resayes, S. Characteristics and chemical composition of date palm (Phoenix canariensis) seeds and seed oil. Ind. Crop. Prod. 2010, 32, 360–365. [Google Scholar] [CrossRef]
- Bentrad, N.; Gaceb-Terrak, R.; Benmalek, Y.; Rahmania, F. Studies on chemical composition and antimicrobial activities of bioactive molecules from date palm (Phoenix dactylifera L.) pollens and seeds. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 242–256. [Google Scholar] [CrossRef]
- Atghaei, M.; Sefidkon, F.; Darini, A.; Sadeghzadeh Hemayati, S.; Abdossi, V. Essential Oil Content and Composition of the Spathe in Some Date Palm (Phoenix dactylifera L.) Varieties in Iran. J. Essent. Oil Bear. Plants 2020, 23, 292–300. [Google Scholar] [CrossRef]
- Hammouda, H.d.; Chérif, J.K.; Trabelsi-Ayadi, M.; Baron, A.; Guyot, S. Detailed polyphenol and tannin composition and its variability in Tunisian dates (Phoenix dactylifera L.) at different maturity stages. J. Agric. Food Chem. 2013, 61, 3252–3263. [Google Scholar] [CrossRef]
- Chaira, N.; Smaali, M.I.; Martinez-Tomé, M.; Mrabet, A.; Murcia, M.A.; Ferchichi, A. Simple phenolic composition, flavonoid contents and antioxidant capacities in water-methanol extracts of Tunisian common date cultivars (Phoenix dactylifera L.). Int. J. Food Sci. Nutr. 2009, 60, 316–329. [Google Scholar] [CrossRef]
- Abbas, F.A.; Ateya, A.-M. Estradiol, esteriol, estrone and novel flavonoids from date palm pollen. Aust. J. Basic Appl. Sci. 2011, 5, 606–614. [Google Scholar]
- Suresh, S.; Guizani, N.; Al-Ruzeiki, M.; Al-Hadhrami, A.; Al-Dohani, H.; Al-Kindi, I.; Rahman, M.S. Thermal characteristics, chemical composition and polyphenol contents of date-pits powder. J. Food Eng. 2013, 119, 668–679. [Google Scholar] [CrossRef]
- Singh, V.; Guizani, N.; Essa, M.; Hakkim, F.; Rahman, M. Comparative analysis of total phenolics, flavonoid content and antioxidant profile of different date varieties (Phoenix dactylifera L.) from Sultanate of Oman. Int. Food Res. J. 2012, 19, 1063. [Google Scholar]
- Eid, N.M.; Al-Awadi, B.; Vauzour, D.; Oruna-Concha, M.J.; Spencer, J.P. Effect of cultivar type and ripening on the polyphenol content of date palm fruit. J. Agric. Food Chem. 2013, 61, 2453–2460. [Google Scholar] [CrossRef]
- El Modafar, C.; El Boustani, E. Cell wall-bound phenolic acid and lignin contents in date palm as related to its resistance to Fusarium oxysporum. Biol. Plant. 2001, 44, 125–130. [Google Scholar] [CrossRef]
- Daoud, A.; Malika, D.; Bakari, S.; Hfaiedh, N.; Mnafgui, K.; Kadri, A.; Gharsallah, N. Assessment of polyphenol composition, antioxidant and antimicrobial properties of various extracts of Date Palm Pollen (DPP) from two Tunisian cultivars. Arab. J. Chem. 2015, 12, 3075–3086. [Google Scholar] [CrossRef]
- Fang, F.; Li, J.-M.; Zhang, P.; Tang, K.; Wang, W.; Pan, Q.-H.; Huang, W.-D. Effects of grape variety, harvest date, fermentation vessel and wine ageing on flavonoid concentration in red wines. Food Res. Int. 2008, 41, 53–60. [Google Scholar] [CrossRef]
- Michael, H.N.; Salib, J.Y.; Eskander, E.F. Bioactivity of diosmetin glycosides isolated from the epicarp of date fruits, Phoenix dactylifera, on the biochemical profile of alloxan diabetic male rats. Phytother. Res. 2013, 27, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Hussein, A.M.; El-Mousalamy, A.M.; Hussein, S.A.; Mahmoud, S.A. Effects of palm dates (Phoenix dactylifera L.) extracts on hepatic dysfunctions in Type 2 diabetic rat model. World J. Pharm. Pharm. Sci. 2015, 4, 62–79. [Google Scholar]
- Al-Malki, A.L.; Barbour, E.K.; Abulnaja, K.O.; Moselhy, S.S. Management of hyperglycaemia by ethyl acetate extract of Balanites aegyptiaca (desert date). Molecules 2015, 20, 14425–14434. [Google Scholar] [CrossRef]
- Al-Alawi, R.A.; Al-Mashiqri, J.H.; Al-Nadabi, J.S.; Al-Shihi, B.I.; Baqi, Y. Date palm tree (Phoenix dactylifera L.): Natural products and therapeutic options. Front. Plant Sci. 2017, 8, 845. [Google Scholar] [CrossRef]
- Regnault-Roger, C.; Hadidane, R.; Biard, J.-F.; Boukef, K. High performance liquid and thin-layer chromatographic determination of phenolic acids in palm (Phoenix dactilifera) products. Food Chem. 1987, 25, 61–71. [Google Scholar] [CrossRef]
- Taleb, H.; Morris, R.K.; Withycombe, C.E.; Maddocks, S.E.; Kanekanian, A.D. Date syrup–derived polyphenols attenuate angiogenic responses and exhibits anti-inflammatory activity mediated by vascular endothelial growth factor and cyclooxygenase-2 expression in endothelial cells. Nutr. Res. 2016, 36, 636–647. [Google Scholar] [CrossRef]
- Ammar, N.M.; Lamia, T.; Abou, E.; Nabil, H.; Lalita, M.; Tom, J. Flavonoid Constituents and Antimicrobial Activity of Date (Phoenix dactylifera L.) Seeds Growing in Egypt. Biology 2009, 3, 1–5. [Google Scholar]
- Sachde, A.G.; Al-Bakir, A.Y.; Abdul-Raheem, J.A. Polyphenol oxidase from Barhee and Zahdi dates. II. Characterization. J. Food Biochem. 1988, 12, 241–252. [Google Scholar] [CrossRef]
- Sirisena, S.; Zabaras, D.; Ng, K.; Ajlouni, S. Characterization of Date (Deglet Nour) Seed Free and Bound Polyphenols by High-Performance Liquid Chromatography-Mass Spectrometry. J. Food Sci. 2017, 82, 333–340. [Google Scholar] [CrossRef]
- Sirisena, S.; Ajlouni, S.; Ng, K. Simulated gastrointestinal digestion and in vitro colonic fermentation of date (Phoenix dactylifera L.) seed polyphenols. Int. J. Food Sci. Technol. 2018, 53, 412–422. [Google Scholar] [CrossRef]
- Saddi, A.A.; Mohamed, A.M.; Shaikh, A.M. Prophylactic mechanisms of Cucumis melo var. flexuosus and Phoenix dactylifera fruit extracts against diabetic cardiomyopathy in streptozotocin induced diabetic rats. Pak. J. Pharm. Sci. 2018, 31, 699–707. [Google Scholar] [PubMed]
- Zaakouk, S.; el-Rasheid, A.; Hesham, G.; Belal, A.; Elfeky, K. Effect of Balanites aegyptiaca (heglig dates) and persea americana (avocado fruit) on some hematological and biochemical parameters in streptozotocin induced diabetic male rats. Al-Azhar Bull. Sci. 2018, 29, 49–59. [Google Scholar]
- Alam, F.; Islam, M.A.; Khalil, M.I.; Gan, S.H. Metabolic control of type 2 diabetes by targeting the GLUT4 glucose transporter: Intervention approaches. Curr. Pharm. Des. 2016, 22, 3034–3049. [Google Scholar] [CrossRef]
- Victor, A.C. Ethanol pulp extract of date palm (Phoenix dactylifera) modulates hematinic indices in diabetic rats. Ann. Food Sci. Technol. 2017, 15, 297–306. [Google Scholar]
- Evans, G.H.; McLaughlin, J.; Yau, A.M. The Effect of Glucose or Fructose Added to a Semi-solid Meal on Gastric Emptying Rate, Appetite, and Blood Biochemistry. Front. Nutr. 2018, 5, 1–7. [Google Scholar] [CrossRef]
- Ahmed, S.; Khan, R.A.; Jamil, S.; Afroz, S. Antidiabetic effects of native date fruit Aseel (Phoenix dactylifera L.) in normal and hyperglycemic rats. Pak. J. Pharm. Sci. 2017, 30, 1797–1802. [Google Scholar]
- Sekhon-Loodu, S.; Rupasinghe, H. Evaluation of antioxidant, antidiabetic and antiobesity potential of selected traditional medicinal plants. Front. Nutr. 2019, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.O.; Ahmad, M.N.; Hamad, H.J.; Hamad, W.J. Effect of Birhi Variety of Date Palm Fruits, (Phoenix dactylifera L.) at the Tamr Stage on Serum Glucose Levels in Streptozotocin-Induced Diabetic Rats. J. Agric. Sci. 2016, 8, 110–118. [Google Scholar]
- Abou Khalil, N.S.; Abou-Elhamd, A.S.; Wasfy, S.I.; El Mileegy, I.M.; Hamed, M.Y.; Ageely, H.M. Antidiabetic and antioxidant impacts of desert date (Balanites aegyptiaca) and parsley (Petroselinum sativum) aqueous extracts: Lessons from experimental rats. J. Diabetes Res. 2016, 2016, 1–10. [Google Scholar] [CrossRef]
- Kim, E.-K.; Kwon, K.-B.; Song, M.-Y.; Seo, S.-W.; Park, S.-J.; Ka, S.-O.; Na, L.; Kim, K.-A.; Ryu, D.-G.; So, H.-S. Genistein protects pancreatic β cells against cytokine-mediated toxicity. Mol. Cell. Endocrinol. 2007, 278, 18–28. [Google Scholar] [CrossRef]
- Bendary, M.; Zawawi, B.M. A study on the possible role of date fruit on the electrophysiology and neuropathic changes in diabetic polyneuropathy. J. Biosci. Appl. Res. 2016, 2, 524–531. [Google Scholar] [CrossRef]
- Olawole, T.D.; Okundigie, M.I.; Rotimi, S.O.; Okwumabua, O.; Afolabi, I.S. Preadministration of fermented sorghum diet provides protection against hyperglycemia-induced oxidative stress and suppressed glucose utilization in alloxan-induced diabetic rats. Front. Nutr. 2018, 5, 1–8. [Google Scholar] [CrossRef]
- Amouoghli, T.B.; Hassanpour, A.; Kohi, V.; Ostovar, A.; Alizadeh, A. The effects of date consumption on serumic levels of glucose, lipids and lipoproteins in diabetic rats. Vet. Clin. Pathol. 2009, 3, 367–375. [Google Scholar]
- Hosseinipour, M.; Goodarzi, N.; Bakhtiari, M. Protective efficiency of Ashrasi date palm hydroalcoholic extract against diabetes-induced testicular toxicity: A biochemical and stereological study. Andrologia 2019, 51, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zangiabadi, N.; Asadi-Shekaari, M.; Sheibani, V.; Jafari, M.; Shabani, M.; Asadi, A.R.; Tajadini, H.; Jarahi, M. Date fruit extract is a neuroprotective agent in diabetic peripheral neuropathy in streptozotocin-induced diabetic rats: A multimodal analysis. Oxid. Med. Cell. Longev. 2011, 2011, 1–9. [Google Scholar] [CrossRef]
- Chung, A.P.; Gurtu, S.; Chakravarthi, S.; Moorthy, M.; Palanisamy, U.D. Geraniin protects high-fat diet-induced oxidative stress in Sprague Dawley Rats. Front. Nutr. 2018, 5, 1–8. [Google Scholar] [CrossRef]
- Ali, Z.; Ma, H.; Wali, A.; Ayim, I.; Rashid, M.T.; Younas, S. A double-blinded, randomized, placebo-controlled study evaluating the impact of dates vinegar consumption on blood biochemical and hematological parameters in patients with type 2 diabetes. Trop. J. Pharm. Res. 2018, 17, 2463–2469. [Google Scholar] [CrossRef]
- Fushimi, T.; Tayama, K.; Fukaya, M.; Kitakoshi, K.; Nakai, N.; Tsukamoto, Y.; Sato, Y. The efficacy of acetic acid for glycogen repletion in rat skeletal muscle after exercise. Int. J. Sports Med. 2002, 23, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Hlebowicz, J.; Darwiche, G.; Björgell, O.; Almér, L.-O. Effect of apple cider vinegar on delayed gastric emptying in patients with type 1 diabetes mellitus: A pilot study. BMC Gastroenterol. 2007, 7, 46. [Google Scholar] [CrossRef] [PubMed]
- Al-Mssallem, M. Consumption of Dates among Saudi Adults and its Association with the Prevalence of Type 2 Diabetes. Asian J. Clin. Nutr. 2018, 10, 58–64. [Google Scholar] [CrossRef]
- Hossain, A.S. Dried Dates Fruit and its Biochemical and Nutrient Content: Uses as Diabetic Food. Asian J. Clin. Nutr. 2015, 7, 90–95. [Google Scholar] [CrossRef]
- Williamson, G.; Carughi, A. Polyphenol content and health benefits of raisins. Nutr. Res. 2010, 30, 511–519. [Google Scholar] [CrossRef]
- Foshati, S.; Nouripour, F.; Akhlaghi, M. Effect of Date and Raisin Snacks on Glucose Response in Type 2 Diabetes. Nutr. Food Sci. Res. 2015, 2, 19–25. [Google Scholar]
- Alkaabi, J.; Al-Dabbagh, B.; Saadi, H.; Gariballa, S.; Yasin, J. Effect of traditional Arabic coffee consumption on the glycemic index of Khalas dates tested in healthy and diabetic subjects. Asia Pac. J. Clin. Nutr. 2013, 22, 565–572. [Google Scholar]
- Forghani, B.; Kasaeian, N.; Tala, M.M.; Zare, M.; Haghighi, S.; Amini, A. Effect of dates (khorma) on 2hour postprandial (2hpp) blood glucose level in type 2 diabetic patients reterred to isfahan endocrine and metabolism research center. J. Shahid Sadoughi Univ. Med. Sci. Health Serv. 2003, 10, 52–55. [Google Scholar]
- Avogaro, A.; Toffolo, G.; Valerio, A.; Cobelli, C. Epinephrine exerts opposite effects on peripheral glucose disposal and glucose-stimulated insulin secretion: A stable label intravenous glucose tolerance test minimal model study. Diabetes 1996, 45, 1373–1378. [Google Scholar] [CrossRef]
- Ahmed, M.; Al-Othaimeen, A.; De Vol, E.; Bold, A. Comparative responses of plasma glucose, insulin and C-peptide following ingestion of isocaloric glucose, a modified urban Saudi breakfast and dates in normal Saudi persons. Ann. Saudi Med. 1991, 11, 414–417. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aldughpassi, A.; Wolever, T.M. Effect of coffee and tea on the glycaemic index of foods: No effect on mean but reduced variability. Br. J. Nutr. 2009, 101, 1282–1285. [Google Scholar] [CrossRef] [PubMed]
- Razaghi-azar, M.; Noori, N.; Afsharian, K. Effect of date on blood sugar in patients with type 1 diabetes mellitus. Iran. J. Diabetes Metab. 2005, 4, 27–34. [Google Scholar]
- AlGeffari, M.A.; Almogbel, E.S.; Alhomaidan, H.T.; El-Mergawi, R.; Barrimah, I.A. Glycemic indices, glycemic load and glycemic response for seventeen varieties of dates grown in Saudi Arabia. Ann. Saudi Med. 2016, 36, 397–403. [Google Scholar] [CrossRef]
- Abiola, T.; Dibie, D.; Akinwale, O.; Shomuyiwa, O. Assessment of the Antidiabetic Potential of the Ethanolic Extract of Date Palm (Phoenix Dactylifera) Seed in Alloxan-Induced Diabetic Rats. J. Diabetes Metab. 2018, 9, 1–9. [Google Scholar]
- Bouaziz, F.; Ben Abdeddayem, A.; Koubaa, M.; Ellouz Ghorbel, R.; Ellouz Chaabouni, S. Date Seeds as a Natural Source of Dietary Fibers to Improve Texture and Sensory Properties of Wheat Bread. Foods 2020, 9, 737. [Google Scholar] [CrossRef]
- Turki, M.; Barbosa-Pereira, L.; Bertolino, M.; Essaidi, I.; Ghirardello, D.; Torri, L.; Bouzouita, N.; Zeppa, G. Physico-Chemical Characterization of Tunisian Canary Palm (Phoenix canariensis Hort. Ex Chabaud) Dates and Evaluation of Their Addition in Biscuits. Foods 2020, 9, 695. [Google Scholar] [CrossRef]
- Ayatollahi, S.A.; Sharifi-Rad, M.; Roointan, A.; Baghalpour, N.; Salehi, B.; Shinwari, Z.K.; Khalil, A.T.; Sharifi-Rad, J. Antidiabetic Activity of Date Seed Methanolic Extracts in Alloxan-Induced Diabetic Rats. Pak. Vet. J. 2019, 39, 583–587. [Google Scholar] [CrossRef]
- Thouri, A.; Chahdoura, H.; El Arem, A.; Hichri, A.O.; Hassin, R.B.; Achour, L. Effect of solvents extraction on phytochemical components and biological activities of Tunisian date seeds (var. Korkobbi and Arechti). Bmc Complement. Altern. Med. 2017, 17, 1–10. [Google Scholar] [CrossRef]
- Hasan, M.; Mohieldein, A. In vivo evaluation of anti diabetic, hypolipidemic, antioxidative activities of Saudi date seed extract on streptozotocin induced diabetic rats. J. Clin. Diagn. Res. 2016, 10, 1–12. [Google Scholar] [CrossRef]
- Abdelaziz, D.H.; Ali, S.A.; Mostafa, M.M. Phoenix dactylifera seeds ameliorate early diabetic complications in streptozotocin-induced diabetic rats. Pharm. Biol. 2015, 53, 792–799. [Google Scholar] [CrossRef]
- Khalil, M.M.; Abou-Raya, M.; Ghoniem, G.A.; Hussain, T. Effect of feeding at different levels of wheat bran and date seeds powder on diabetic rats. J. Food Dairy Sci. 2015, 6, 435–449. [Google Scholar] [CrossRef]
- Olagunju, J.; Jobi, A.; Oyedapo, O. An investigation into the biochemical basis of the observed hyperglycaemia in rats treated with ethanol root extract of Plumbago zeylanica. Phytother. Res. 1999, 13, 346–348. [Google Scholar] [CrossRef]
- El Fouhil, A.F.; Ahmed, A.M.; Darwish, H.H. Hypoglycemic effect of an extract from date seeds on diabetic rats. Saudi Med. J. 2010, 31, 747–751. [Google Scholar]
- El Fouhil, A.F.; Ahmed, A.M.; Atteya, M.; Mohamed, R.A.; Moustafa, A.S.; Al-Roalle, A.H.; Darwish, H.H. Hypoglycemic effects of date seed extract. Possible mechanism of action, and potential therapeutic implications. Saudi Med. J. 2013, 34, 1125–1132. [Google Scholar] [PubMed]
- Halaby, M.S.; Farag, M.H.; Gerges, A.H. Potential effect of date pits fortified bread on diabetic rats. Int. J. Nutr. Food Sci. 2014, 3, 49–59. [Google Scholar] [CrossRef]
- Ardekani, M.R.S.; Khanavi, M.; Hajimahmoodi, M.; Jahangiri, M.; Hadjiakhoondi, A. Comparison of antioxidant activity and total phenol contents of some date seed varieties from Iran. Iran. J. Pharm. Res. 2010, 9, 141. [Google Scholar]
- Amany, M.; Shaker, M.; Abeer, A. Antioxidant activities of date pits in a model meat system. Int. Food Res. J. 2012, 19, 223–227. [Google Scholar]
- Abdallaha, I.Z.; Khattab, H.A.; Ragheb, E.M.; Yousef, F.M.; Alkreathy, H.M. Date Pits Alleviate Reproductive Disorders in Male Diabetic Rats. Glob. J. Pharmacol. 2015, 9, 208–221. [Google Scholar]
- El Fouhil, A.F.; Ahmed, A.M.; Darwish, H.H.; Atteya, M.; Al-Roalle, A.H. An extract from date seeds having a hypoglycemic effect. Is It Safe to Use? Saudi Med. J. 2011, 32, 791–796. [Google Scholar]
- Gharib, M.A.A.-K.; Bakrand, E.-S.H.; Baz, S.M. Hypoglycemic Efficacy of Date Kernels Coffee on Diabetic and Nephrodiabetic Patients. Life Sci. J. 2016, 13, 10–18. [Google Scholar]
- Esfandiari, E.; Dorali, A.; Ghanadian, M.; Rashidi, B.; Mostafavi, F.S. Protective and therapeutic effects of Phoenix dactylifera leaf extract on pancreatic β-cells and diabetic parameters in streptozotocin-induced diabetic rats. Comp. Clin. Path. 2020. [Google Scholar] [CrossRef]
- Mohamed, S.; Abuzaid, O.I.; El-Ashmawy, I.M. Effect of aqueous extract of tops of date palm leaves on blood glucose of diabetic rats. Pak. J. Pharm. Sci. 2017, 30, 2031–2037. [Google Scholar]
- Nuha, A.D.M.A.-M.; Al-Obaid, A.M. Hypoglycemic Action of Synergistic Interaction of Phenolic Compounds Isolated from Iraqi Phoenix dactylifera (Breim) Leaflets in Alloxan–Induced Diabetic Rabbits. J. Nat. Sci. Res. 2016, 6, 22–31. [Google Scholar]
- Chakroun, M.; Khemakhem, B.; Mabrouk, H.B.; El Abed, H.; Makni, M.; Bouaziz, M.; Drira, N.; Marrakchi, N.; Mejdoub, H. Evaluation of anti-diabetic and anti-tumoral activities of bioactive compounds from Phoenix dactylifera L’s leaf: In vitro and in vivo approach. Biomed. Pharmacother. 2016, 84, 415–422. [Google Scholar] [CrossRef]
- Shirdel, Z.; Mirbadalzadeh, R. Improvement of hyperglycemia in diabetic rats by Ethanolic extract of red date leaves. Hor. Sig. 2011, 31, 1957–1959. [Google Scholar]
- Mard, S.A.; Jalalvand, K.; Jafarinejad, M.; Balochi, H.; Naseri, M.K.G. Evaluation of the antidiabetic and antilipaemic activities of the hydroalcoholic extract of Phoenix dactylifera palm leaves and its fractions in alloxan-induced diabetic rats. Malays. J. Med. Sci. 2010, 17, 4–13. [Google Scholar]
- Abuelgassim, A.O. Effect of flax seeds and date palm leaves extracts on serum concentrations of glucose and lipids in alloxan diabetic rats. Pak. J. Biol. Sci. 2010, 13, 1141–1145. [Google Scholar]
- Venkateswaran, S.; Pari, L. Effect of Coccinia indica on blood glucose, insulin and key hepatic enzymes in experimental diabetes. Pharm. Biol. 2002, 40, 165–170. [Google Scholar] [CrossRef]
- Jurgoński, A.; Juśkiewicz, J.; Zduńczyk, Z. Ingestion of black chokeberry fruit extract leads to intestinal and systemic changes in a rat model of prediabetes and hyperlipidemia. Plant Foods Hum. Nutr. 2008, 63, 176–182. [Google Scholar] [CrossRef]
| No. | Types of Date | Polyphenols | Country | References |
|---|---|---|---|---|
| 1 | Deglet Nour and Ftimi (Phoenix dactylifera L.) |
| Tunisia | [27] |
| 2 | Date palm (Phoenix dactylifera L.) |
| Tunisia | [28] |
| 3 | Date palm pollen (Phoenix dactylifera L.) |
| Egypt | [29] |
| 4 | Date palm (Phoenix dactylifera L.) |
| Oman | [30] |
| 5 | Fardh, Khasab, and Khalas date fruits (Phoenix dactylifera L.) |
| Oman | [31] |
| 6 | Date palm Ajwa, Barni (Phoenix dactylifera L.) |
| United Kingdom | [32] |
| 7 | Date palm (Phoenix dactylifera L.) |
| Morocco | [33] |
| 8 | Date palm (Phoenix dactylifera L.) |
| Tunisia | [34] |
| 9 | Harvest date |
| China | [35] |
| 10 | Date palm (Phoenix dactylifera L.) |
| Egypt | [36] |
| 11 | Palm Date (Phoenix dactylifera L.) |
| Egypt | [37] |
| 12 | Desert date (Balanites aegyptiaca) |
| Saudi Arabia | [38] |
| 13 | Desert date (Balanites aegyptiaca) |
| Egypt | [11] |
| 14 | Date palm (Phoenix dactylifera L.) |
| Oman | [39] |
| 15 | Date Palm (Phoenix dactylifera L.) |
| Tunisia | [40] |
| 16 | Date syrup |
| United Kingdom | [41] |
| 17 | Date palm (Phoenix dactylifera L.) |
| Egypt | [42] |
| 18 | Barhee and Zahdi dates (Phoenix dactylifera L.) |
| Iraq | [43] |
| 19 | Deglet Nour (Phoenix dactylifera L.) |
| Australia | [44] |
| 20 | Date palm (Phoenix dactylifera L.) |
| Australia | [45] |
| Study ID [References] | Country | Study Design | Number of Participants (Female) | Date Consumption | Study Duration | Outcomes |
|---|---|---|---|---|---|---|
| Al-Mssallem 2018 [65] | Saudi Arabia | Cross-sectional | 2177 (1133) | 100 g/day | Weekly and monthly consumption of dates was recorded | Consumption of dates has no association with the prevalence of T2DM |
| Ali 2018 [62] | Pakistan | Double-blinded randomised-controlled trial | 60 (29) | 20 mL/day | 10 weeks | Date vinegar improved blood concentrations of HbA1c and FBS (p < 0.05) in patients with T2DM |
| Foshati 2015 [68] | Iran | Non-randomised crossover clinical trial | 15 (10) | 24.2 g | 3 days | Consumption of dates did not increase blood glucose |
| Alkaabi 2013 [69] | United Arab Emirates | Case-control | 10 (5) | 50 g | 5 days | Dates exhibited as a low-GI fruit for patients with T2DM when consumed with and without Arabic coffee |
| Alkaabi 2011 [15] | United Arab Emirates | Non-randomised clinical trial | 10 | 50 g | 3 days | Diabetic individuals do not result in significant postprandial glucose excursions due to the date consumption |
| Forghani 2003 [70] | Iran | Non-randomised clinical trial | 16 | NR | 2 days | Glucose level decreases substantially following replacing the bread content in a diabetic diet with dates |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mia, M.A.-T.; Mosaib, M.G.; Khalil, M.I.; Islam, M.A.; Gan, S.H. Potentials and Safety of Date Palm Fruit against Diabetes: A Critical Review. Foods 2020, 9, 1557. https://doi.org/10.3390/foods9111557
Mia MA-T, Mosaib MG, Khalil MI, Islam MA, Gan SH. Potentials and Safety of Date Palm Fruit against Diabetes: A Critical Review. Foods. 2020; 9(11):1557. https://doi.org/10.3390/foods9111557
Chicago/Turabian StyleMia, Md Al-Tareq, Md Golam Mosaib, Md Ibrahim Khalil, Md Asiful Islam, and Siew Hua Gan. 2020. "Potentials and Safety of Date Palm Fruit against Diabetes: A Critical Review" Foods 9, no. 11: 1557. https://doi.org/10.3390/foods9111557
APA StyleMia, M. A.-T., Mosaib, M. G., Khalil, M. I., Islam, M. A., & Gan, S. H. (2020). Potentials and Safety of Date Palm Fruit against Diabetes: A Critical Review. Foods, 9(11), 1557. https://doi.org/10.3390/foods9111557








