Evolution of the Aroma of Treixadura Wines during Bottle Aging
Abstract
1. Introduction
2. Materials and Methods
2.1. Wine Samples
2.2. Determination of Volatile Compounds
2.3. Aromatic Index
2.4. Sensory Evaluation
2.5. Statistical Analysis
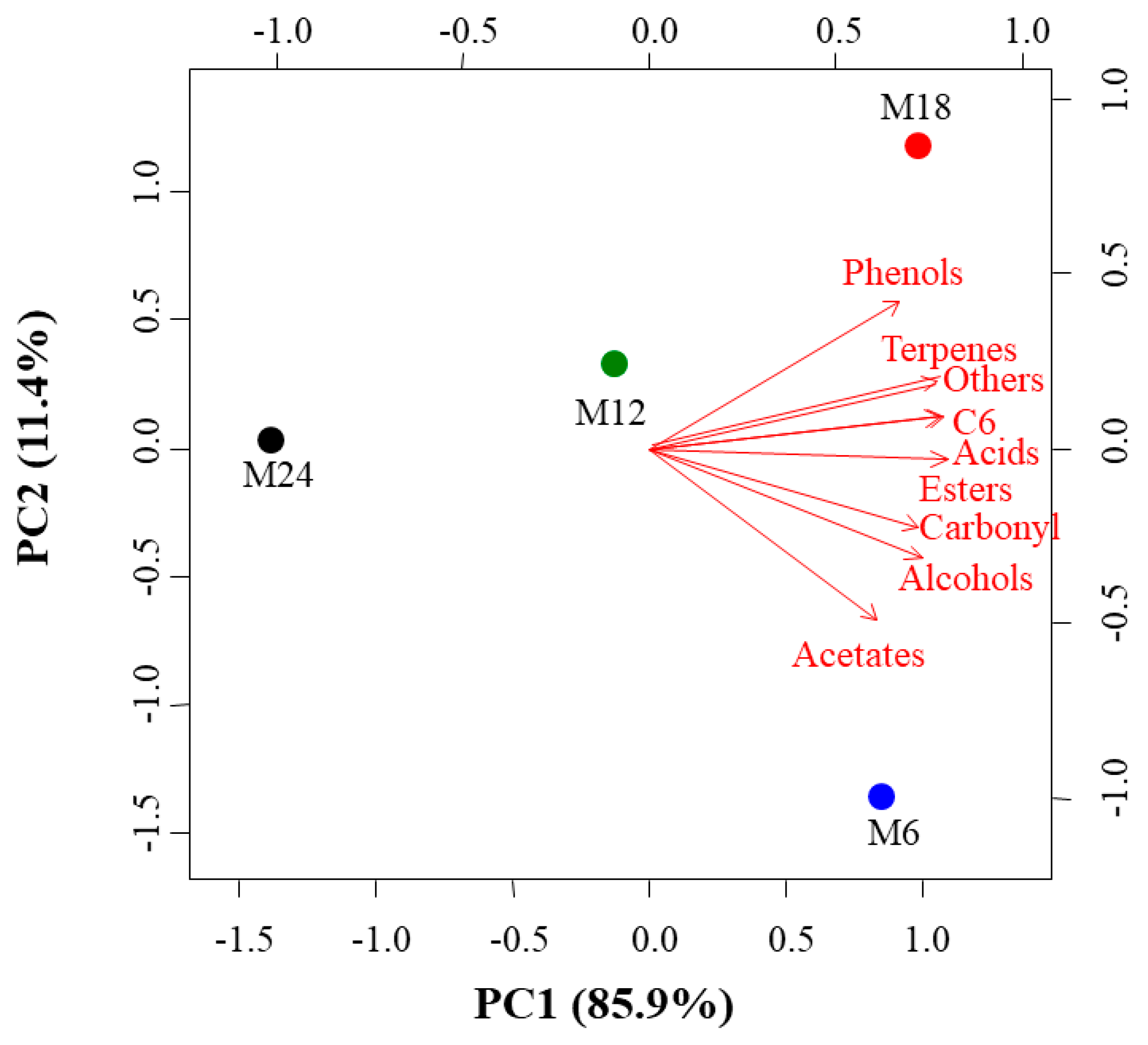
3. Results
3.1. Evolution of the Concentrations of Volatile Compounds Over Storage Time in the Bottle
3.2. Effect of Bottle Storage Time on Odor Activity Values
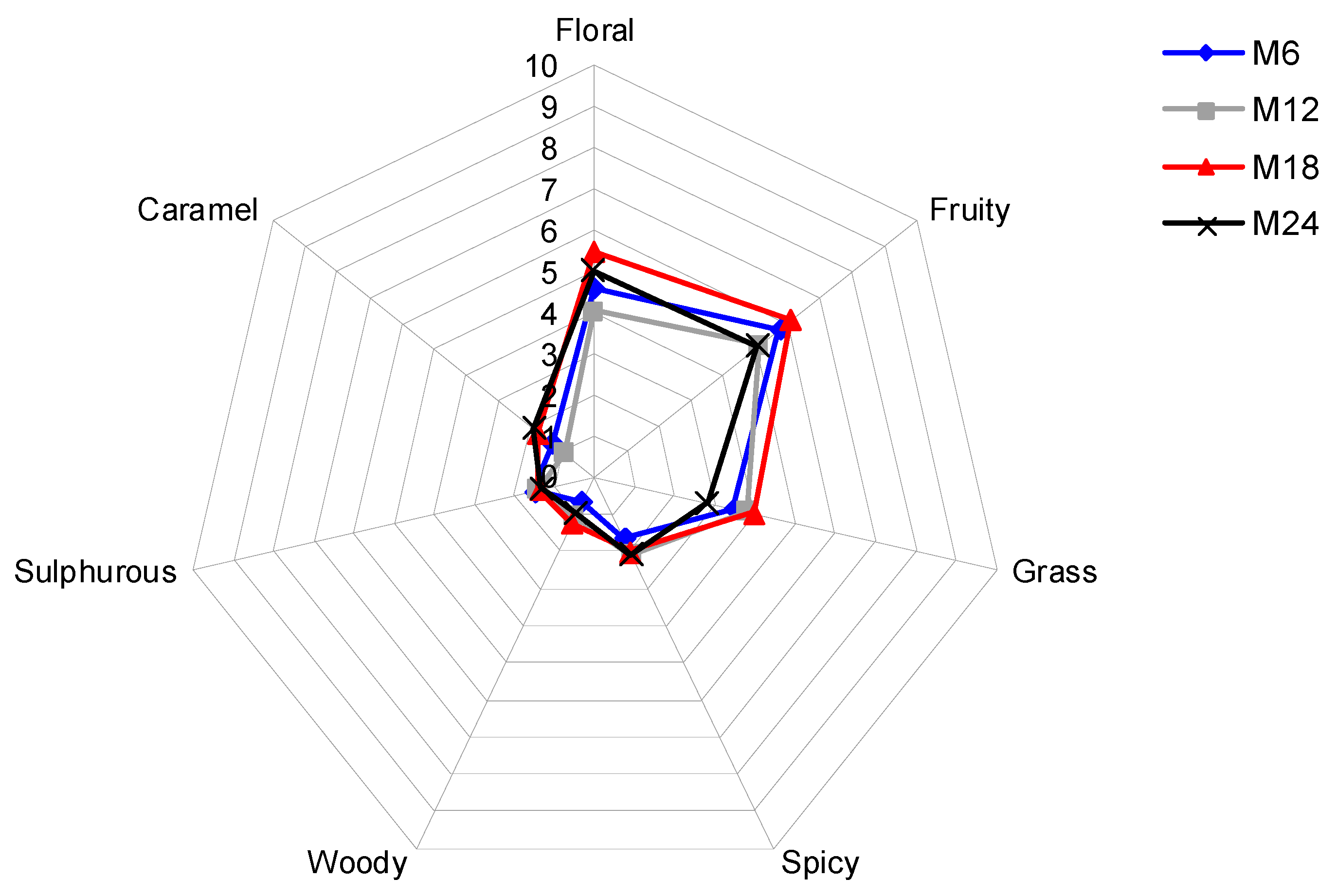
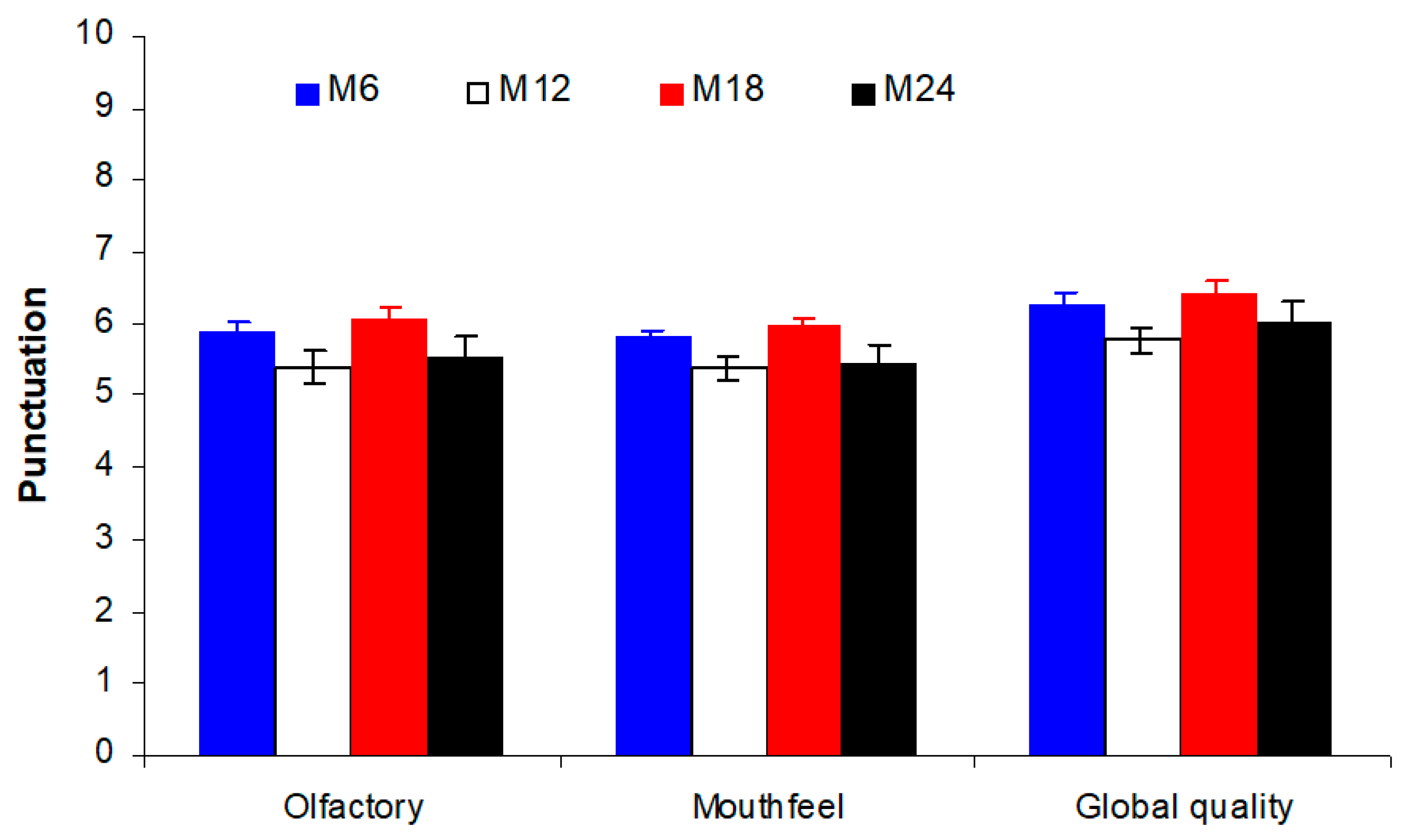
3.3. Evolution of the Sensory Profile of Treixadura Wines over Bottle Storage
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Robinson, A.L.; Boss, P.K.; Solomon, P.S.; Trengove, R.D.; Heymann, H.; Ebeler, S.E. Origins of grape and wine aroma. Part 1. Chemical components and viticultural impacts. Am. J. Enol. Vitic. 2014, 65, 1–24. [Google Scholar] [CrossRef]
- Ebeler, S.E.; Thorngate, J.H. Wine chemistry and flavor: Looking into the crystal glass. J. Agric. Food Chem. 2009, 57, 8098–8108. [Google Scholar] [CrossRef] [PubMed]
- González-Barreiro, C.; Rial-Otero, R.; Cancho-Grande, B.; Simal-Gándara, J. Wine aroma compounds in grapes: A critical review. Crit. Rev. Food Sci. Nutr. 2015, 55, 202–218. [Google Scholar] [CrossRef] [PubMed]
- Swiegers, J.H.; Bartwosky, E.J.; Henschke, P.A.; Pretorius, I.S. Yeast and bacterial modulation of wine aroma and flavour. Aust. J. Grape Wine Res. 2005, 11, 139–173. [Google Scholar] [CrossRef]
- Garde-Cerdán, T.; Ancín-Azpilicueta, C. Review of quality factors on wine ageing in oak barrels. Trends Food Sci. Technol. 2006, 17, 438–447. [Google Scholar] [CrossRef]
- Garde-Cerdán, T.; Lorenzo, C.; Carot, J.M.; Esteve, M.D.; Climent, M.D.; Salinas, M.R. Effects of composition, storage time, geographic origin and oak type on the accumulation of some volatile oak compounds and ethylphenols in wines. Food Chem. 2010, 122, 1076–1082. [Google Scholar] [CrossRef]
- Ling, M.Q.; Xie, H.; Hua, Y.B.; Cai, J.; Li, S.Y.; Lan, Y.B.; Li, R.N.; Duan, C.Q.; Shi, Y. Flavor profile evolution of bottle aged rosé and white wines sealed with different closures. Molecules 2019, 24, 836. [Google Scholar] [CrossRef]
- Maza, M.A.; Martínez, J.M.; Cebrián, G.; Sánchez-Gimeno, A.C.; Camargo, A.; Álvarez, I.; Raso, J. Evolution of polyphenolic compounds and sensory properties of wines obtained from Grenache grapes treated by pulsed electric fields during aging in bottles and oak barrels. Foods 2020, 9, 542. [Google Scholar] [CrossRef]
- Francis, I.L.; Newton, J.L. Determining wine aroma from compositional data. Aust. J. Grape Wine Res. 2005, 11, 14–126. [Google Scholar] [CrossRef]
- Kwiatkowski, M.J.; Skouroumounis, G.K.; Lattey, K.A.; Waters, E.J. The impact of closures, including screw cap with three different headspace volumes, on the composition, colour and sensory properties of a Cabernet Sauvignon wine during two years’ storage. Aust. J. Grape Wine Res. 2007, 13, 81–94. [Google Scholar] [CrossRef]
- Liu, D.; Xing, R.-R.; Li, Z.; Yang, D.-M.; Pan, Q.-H. Evolution of volatile compounds, aroma attributes, and sensory perception in bottle-aged red wines and their correlation. Eur. Food Res. Technol. 2016, 242, 1937–1948. [Google Scholar] [CrossRef]
- Smith, P.A.; McRae, J.M.; Bindon, K.A. Impact of winemaking practices on the concentration and composition of tannins in red wine. Aust. J. Grape Wine Res. 2015, 21, 601–614. [Google Scholar] [CrossRef]
- Silva, M.A.; Julien, M.; Jourdes, M.; Teissedre, P.L. Impact of closures on wine post-bottling development: A review. Eur. Food Res. Technol. 2011, 233, 905–914. [Google Scholar] [CrossRef]
- Ugliano, M. Oxygen contribution to wine aroma evolution during bottle aging. J. Agric. Food Chem. 2013, 61, 6125–6136. [Google Scholar] [CrossRef] [PubMed]
- Styger, G.; Prior, B.; Bauer, F.F. Wine flavor and aroma. J. Ind. Microbiol. Biotechnol. 2011, 38, 1145–1159. [Google Scholar] [CrossRef] [PubMed]
- Belda, I.; Ruiz, J.; Esteban-Fernández, A.; Navascués, E.; Marquina, D.; Santos, A.; Moreno-Arribas, M.V. Microbial contribution to wine aroma and its intended use for wine quality improvement. Molecules 2017, 22, 189. [Google Scholar] [CrossRef]
- Falqué, E.; Fernández, E.; Dubourdieu, D. Volatile components of Loureira, Dona Branca, and Treixadura wines. J. Agric. Food Chem. 2002, 50, 538–543. [Google Scholar] [CrossRef]
- Cortés, S.; Blanco, P. Yeast strain effect on the concentration of major volatile compounds and sensory profile of wines from Vitis vinifera var. Treixadura. World J. Microbiol. Biotechnol. 2011, 27, 925–932. [Google Scholar] [CrossRef]
- Mirás-Avalos, J.M.; Bouzas-Cid, Y.; Trigo-Córdoba, E.; Orriols, I.; Falqué, E. Amino acid profiles to differentiate white wines from three autochtonous Galician varieties. Foods 2020, 9, 114. [Google Scholar] [CrossRef]
- Vilanova, M.; Escudero, A.; Graña, M.; Cacho, J. Volatile composition and sensory properties of North West Spain white wines. Food Res. Int. 2013, 54, 562–568. [Google Scholar] [CrossRef]
- Versini, G.; Orriols, I.; Dalla Serra, A. Aroma components of Galician Albariño, Loureira and Godello wines. Vitis 1994, 33, 165–170. [Google Scholar]
- Falqué, E.; Fernández, E.; Dubourdieu, D. Differentiation of white wines by their aromatic index. Talanta 2001, 54, 271–281. [Google Scholar] [CrossRef]
- Genisheva, Z.; Oliveira, J.M. Monoterpenic characterization of white cultivars from Vinhos Verdes Appellation of Origin (North Portugal). J. Inst. Brew. 2009, 115, 308–317. [Google Scholar] [CrossRef]
- Moreira, N.; Guedes-de-Pinho, P.; Santos, C.; Vasconcelos, I. Volatile sulphur compounds composition of monovarietal white wines. Food Chem. 2010, 123, 1198–1203. [Google Scholar] [CrossRef]
- Blanco, P.; Mirás-Avalos, J.M.; Suárez, V.; Orriols, I. Inoculation of Treixadura musts with Saccharomyces cerevisiae strains: Fermentative performance and influence on the wine characteristics. Food Sci. Technol. Int. 2012, 19, 177–186. [Google Scholar] [CrossRef]
- Bouzas-Cid, Y.; Falqué, E.; Orriols, I.; Mirás-Avalos, J.M. Effects of irrigation over three years on the amino acid composition of Treixadura (Vitis vinifera L.) musts and wines, and on the aromatic composition and sensory profiles of its wines. Food Chem. 2018, 240, 707–716. [Google Scholar] [CrossRef]
- Bertrand, A.; Ribéreau-Gayon, P. Determination of volatile components of wine by gas-phase chromatography. Ann. Falsif. Expert. Chim. 1970, 63, 148–156. [Google Scholar]
- Armada, L.; Fernández, E.; Falqué, E. Influence of several enzymatic treatments on aromatic composition of white wines. LWT Food Sci. Technol. 2010, 43, 1517–1525. [Google Scholar] [CrossRef]
- Guth, H. Quantitation and sensory studies of character impact odorants of different white wine varieties. J. Agric. Food Chem. 1997, 45, 3027–3032. [Google Scholar] [CrossRef]
- Ferreira, V. Volatile aroma compounds and wine sensory attributes. In Managing Wine Quality: Viticulture and Wine Quality; Reynolds, A.G., Ed.; Woodhead Publishing Limited: Cambridge, UK, 2010; pp. 3–28. [Google Scholar]
- Ribéreau-Gayon, P.; Dubourdieu, D.; Doneche, B.; Lonvaud, A. Handbook of Enology, Volume 1: The Microbiology of Wine and Vinifications; Dunond: Paris, France, 1998. [Google Scholar]
- Simpson, R.F. Some important aroma composition of components of white wine. Food Technol. 1979, 31, 516–522. [Google Scholar]
- Peinado, R.A.; Moreno, J.; Bueno, J.E.; Moreno, J.A.; Mauricio, J.C. Comparative study of aromatic compounds in two young white wines subjected to pre-fermentative cryomaceration. Food Chem. 2004, 84, 585–590. [Google Scholar] [CrossRef]
- Meilgaard, M. Flavor chemistry of beer. Part II: Flavor and threshold of 239 aroma volatiles. MBBA Tech. Quart. 1975, 12, 151–168. [Google Scholar]
- Zoecklein, B.W.; Fugelsang, K.C.; Gump, B.H.; Nury, F.S. Wine Analysis and Production; Springer Science: New York, NY, USA, 1994. [Google Scholar]
- López, R.; Ferreira, V.; Cacho, J.F. Quantitative determination of the odorants of young red wines from different grape varieties. An assessment of their sensory role. In Enology 99, Proceedings of the 6th International Symposium on Enology, Bordeaux, France, 10–12 June 1999; Lonvaud-Funel, A., Ed.; Tec & Doc Éditions: Paris, France, 1999; pp. 148–151. [Google Scholar]
- Ferreira, V.; López, R.; Cacho, J.F. Quantitative determination of the odorants of young red wines from different grape varieties. J. Sci. Food Agric. 2000, 80, 1659–1667. [Google Scholar] [CrossRef]
- Étievant, P.X. Wine. In Volatile Compounds in Food and Beverages; Maarse, H., Ed.; TNO-CIVO Food Analysis Institute: Zeist, The Netherlands, 1991; pp. 483–546. [Google Scholar]
- Salo, P. Determining the odor thresholds for some compounds in alcoholic beverages. J. Food Sci. 1970, 35, 95–99. [Google Scholar] [CrossRef]
- Boidron, J.; Chatonnet, P.; Pons, M. Influence du bois sur certaines substances odorantes des vines. Connaiss. Vigne Vin 1988, 22, 275–294. [Google Scholar]
- R Core Team. R: A Language for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Karbowiak, T.; Gougeon, R.D.; Alinc, J.-B.; Brachais, L.; Debeaufort, F.; Voilley, A.; Chassagne, D. Wine oxidation and the role of cork. Crit. Rev. Food Sci. Nutr. 2009, 50, 20–52. [Google Scholar] [CrossRef]
- Tarko, T.; Duda-Chodak, A.; Sroka, P.; Siuta, M. The impact of oxygen at various stages of vinification on the chemical composition and the antioxidant and sensory properties of white and red wines. Int. J. Food Sci. 2020, 2020, 7902974. [Google Scholar] [CrossRef]
- Lee, D.H.; Kang, B.S.; Park, H.J. Effect of oxygen on volatile and sensory characteristics of Cabernet Sauvignon during secondary shelf life. J. Agric. Food Chem. 2011, 59, 11657–11666. [Google Scholar] [CrossRef]
- Campo, E.; Ferreira, V.; Escudero, A.; Cacho, J. Prediction of the wine sensory properties related to grape variety from dynamic-headspace gas chromatography-olfactometry data. J. Agric. Food Chem. 2005, 53, 5682–5690. [Google Scholar] [CrossRef]
- Iland, P.; Gago, P. Discovering Australian Wine: A Taster’s Guide. An Introduction to Australian Wine, the Vineyards, the Wineries, How to Taste the Wine, Australian Wine Styles, Wine and Food; Patrick Iland Wine Promotions: Campbelltown, Australia, 2014. [Google Scholar]
- Blackman, J.W.; Hopfer, H.; Saliba, A.J.; Schmidtke, L.M.; Barril, C.; Scollary, G.R. Sensory characterization of Hunter Valley Semillon aged in bottle. Flavour Fragr. J. 2014, 29, 340–349. [Google Scholar] [CrossRef]
- Rocha, S.M.; Coutinho, P.; Delgadillo, I.; Dias Cardoso, A.; Coimbra, M.A. Effect of enzymatic aroma release on the volatile compounds of white wines presenting different aroma potentials. J. Sci. Food Agric. 2005, 85, 199–205. [Google Scholar] [CrossRef]
- Oliveira, C.M.; Ferreira, A.C.S.; de Freitas, V.; Silva, A.M.S. Oxidation mechanisms occurring in wines. Food Res. Int. 2011, 44, 1115–1126. [Google Scholar] [CrossRef]
- Robinson, A.L.; Ebeler, S.E.; Heymann, H.; Boss, P.K.; Solomon, P.S.; Trengove, R.D. Interactions between wine volatile compounds and grape and wine matrix components influence aroma compound headspace partitioning. J. Agric. Food Chem. 2009, 57, 10313–10322. [Google Scholar] [CrossRef] [PubMed]
- Sáenz-Navajas, M.P.; Campo, E.; Culleré, L.; Fernández-Zurbano, P.; Valentin, D.; Ferreira, V. Effects of the nonvolatile matriz on the aroma perception of wine. J. Agric. Food Chem. 2010, 58, 5574–5585. [Google Scholar] [CrossRef]
| Family | Compound | Odor Threshold (mg L−1) | Matrix | Descriptor | Reference |
|---|---|---|---|---|---|
| Terpenes | linalool | 0.050 | Wine | Rose | [31] |
| α-terpineol | 0.400 | Wine | Flowers, linden | ||
| C6 Compounds | 1-hexanol | 4 | Ethanol (11%) | Herbaceous | [32] |
| cis-3-hexen-1-ol | 1 | Ethanol (10%) | Green, bitter | [33] | |
| trans-3-hexen-1-ol | 13 | Beer | [34] | ||
| Higher Alcohols | 1-propanol | 30 | Not specified | Ripe fruit | [35] |
| 1-butanol | 11 | Not specified | Medicine | ||
| isobutanol | 75 | Ethanol (10%) | Clove | [33] | |
| isoamyl alcohol | 40 | Ethanol (10%) | Fusel | [36] | |
| 2-phenylethanol | 14 | Ethanol (10%) | Rose, honey | [37] | |
| Alcohols | methanol | 2000 | Not specified | Alcohol | [35] |
| benzyl alcohol | 900 | Beer | Blackberry | [34] | |
| Carbonyl Compounds | benzaldehyde | 2 | Ethanol (10%) | Almond | [33] |
| furfural | 150 | Beer | Toasted | [34] | |
| acetoine | 150 | Ethanol (12%) | - | [38] | |
| Ethyl Esters | ethyl butyrate | 0.4 | Ethanol (10%) | Blueberry | [33] |
| ethyl hexanoate | 0.08 | Green apple | |||
| ethyl octanoate | 0.58 | Sweet, flower | |||
| ethyl decanoate | 0.5 | Brandy, grape | |||
| ethyl lactate | 150 | Butter | |||
| diethyl succinate | 1.2 | Melon | |||
| Acetates of Higher Alcohols | isoamyl acetate | 0.16 | Ethanol (10%) | Banana | [33] |
| hexyl acetate | 0.67 | Pear, apple, cherry | |||
| 2-phenylethyl acetate | 1.8 | Rose, flower | |||
| Volatile Fatty Acids | butyric acid | 4 | Ethanol (9.5%) | Butter, cheese | [39] |
| isobutyric acid | 2.3 | Ethanol (11%) | - | [37] | |
| isovaleric acid | 0.03 | - | |||
| hexanoic acid | 3 | Beer | Cheese, fatty | [34] | |
| octanoic acid | 10 | Fatty, rancid | |||
| decanoic acid | 6 | ||||
| Volatile Phenols | 4-vinyl-guaiacol | 0.440 | Ethanol (12%) | Paint, watercolor | [40] |
| 4-vinyl-phenol | 0.375 | Pharmacy, clove |
| Family | Compound 1 | M6 | M12 | M18 | M24 | P-Value 1 |
|---|---|---|---|---|---|---|
| Terpenes | linalool * | 29.6 ± 2.4 a | 29.7 ± 1.0 a | 32.6 ± 1.6 a | 18.4 ± 0.7 b | 0.003 |
| α-terpineol * | 21.9 ± 5.2 | 22.0 ± 6.8 | 34.9 ± 5.4 | 16.7 ± 2.2 | 0.597 | |
| C6 Compounds | 1-hexanol | 1.3 ± 0.1 a | 1.1 ± 0.1 ab | 1.4 ± 0.1 a | 0.7 ± 0.0 b | 0.011 |
| cis-3-hexen-1-ol | 0.22 ± 0.05 | 0.18 ± 0.04 | 0.19 ± 0.04 | 0.11 ± 0.02 | 0.736 | |
| trans-3-hexen-1-ol | 0.20 ± 0.08 | 0.26 ± 0.11 | 0.28 ± 0.12 | 0.11 ± 0.04 | 0.861 | |
| Alcohols | methanol | 63.3 ± 3.5 | 58.4 ± 2.3 | 63.4 ± 2.6 | 63.1 ± 2.9 | 0.710 |
| 1-propanol | 15.1 ± 1.3 | 12.8 ± 1.0 | 13.3 ± 1.4 | 16.1 ± 1.4 | 0.494 | |
| isobutanol | 20.2 ± 1.2 | 19.2 ± 1.1 | 22.3 ± 1.6 | 17.9 ± 1.2 | 0.297 | |
| 1-butanol | 1.3 ± 0.1 | 1.3 ± 0.1 | 1.3 ± 0.1 | 0.9 ± 0.1 | 0.134 | |
| isoamyl alcohol | 164.3 ± 6.9 | 158.0 ± 7.2 | 159.5 ± 8.0 | 152.3 ± 6.4 | 0.824 | |
| benzyl alcohol | 0.08 ± 0.01 | 0.29 ± 0.08 | 0.21 ± 0.02 | 0.09 ± 0.01 | 0.129 | |
| 2-phenylethanol | 8.5 ± 0.5 a | 7.8 ± 0.3 a | 9.1 ± 0.5 a | 3.7 ± 0.3 a | <0.001 | |
| 3-methyl-1-pentanol | 0.07 ± 0.01 ab | 0.07 ± 0.00 ab | 0.07 ± 0.00 a | 0.04 ± 0.00 b | 0.031 | |
| 3-ethoxy-1-propanol # | 38.3 ± 7.9 | 38.9 ± 7.0 | 35.1 ± 7.2 | 19.9 ± 3.9 | 0.456 | |
| 1,2-propanodiol # | 8.6 ± 0.7 | 8.0 ± 0.3 | 7.4 ± 0.7 | 4.8 ± 0.2 | 0.221 | |
| 1,3-butanediol # | 355.8 ± 35.1 a | 258.9 ± 15.1 ab | 247.1 ± 19.8 ab | 132.8 ± 15.8 b | <0.001 | |
| 2,3-butanediol # | 78.9 ± 7.2 a | 66.0 ± 5.9 a | 59.5 ± 4.1 ab | 32.9 ± 3.7 b | 0.003 | |
| Ethyl Esters | ethyl butyrate | 0.44 ± 0.04 | 0.39 ± 0.03 | 0.44 ± 0.03 | 0.31 ± 0.02 | 0.167 |
| ethyl hexanoate | 0.65 ± 0.03 a | 0.54 ± 0.05 a | 0.63 ± 0.02 a | 0.35 ± 0.02 b | <0.001 | |
| ethyl octanoate | 1.41 ± 0.13 a | 1.48 ± 0.06 a | 1.21 ± 0.05 a | 0.63 ± 0.02 b | <0.001 | |
| ethyl decanoate | 0.58 ± 0.02 a | 0.63 ± 0.04 a | 0.63 ± 0.02 a | 0.30 ± 0.01 b | <0.001 | |
| ethyl-3-hydroxybutyrate | 0.14 ± 0.01 a | 0.14 ± 0.01 a | 0.16 ± 0.01 a | 0.08 ± 0.01 b | 0.006 | |
| ethyl-4-hydroxybutyrate # | 92.7 ± 13.6 a | 62.7 ± 6.3 ab | 56.4 ± 6.6 ab | 22.3 ± 2.2 b | 0.003 | |
| ethyl lactate | 13.8 ± 1.7 | 9.7 ± 0.5 | 15.1 ± 2.1 | 8.4 ± 1.3 | 0.170 | |
| monoethyl succinate # | 50.3 ± 3.0 b | 54.2 ± 2.5 b | 79.3 ± 3.9 a | 27.7 ± 1.4 c | <0.001 | |
| diethyl succinate | 0.78 ± 0.06 b | 1.16 ± 0.11 b | 1.80 ± 0.11 a | 0.85 ± 0.08 b | <0.001 | |
| Volatile Fatty Acids | isobutyric acid | 1.01 ± 0.08 a | 0.93 ± 0.07 a | 0.93 ± 0.06 a | 0.56 ± 0.03 b | 0.002 |
| butyric acid | 2.88 ± 0.13 a | 2.75 ± 0.11 a | 2.90 ± 0.13 a | 1.62 ± 0.14 b | <0.001 | |
| isovaleric acid | 0.75 ± 0.04 a | 0.73 ± 0.02 a | 0.74 ± 0.03 a | 0.40 ± 0.02 b | <0.001 | |
| hexanoic acid | 4.7 ± 0.1 a | 4.2 ± 0.1 a | 4.7 ± 0.1 a | 2.2 ± 0.1 b | <0.001 | |
| octanoic acid | 6.4 ± 0.2 a | 6.1 ± 0.2 a | 6.9 ± 0.1 a | 3.2 ± 0.1 b | <0.001 | |
| decanoic acid | 2.1 ± 0.1 a | 2.0 ± 0.1 a | 2.1 ± 0.0 a | 0.9 ± 0.0 b | <0.001 | |
| lauric acid | 0.16 ± 0.02 | 0.16 ± 0.02 | 0.15 ± 0.02 | 0.18 ± 0.07 | 0.987 | |
| trans-2-hexenoic acid # | 16.6 ± 1.9 | 16.7 ± 2.2 | 19.0 ± 1.9 | 11.2 ± 0.9 | 0.262 | |
| Acetates of Higher Alcohols | isoamyl acetate | 2.1 ± 0.2 a | 1.4 ± 0.1 a | 1.0 ± 0.1 a | 0.5 ± 0.1 b | <0.001 |
| hexyl acetate | 0.14 ± 0.02 | 0.17 ± 0.05 | 0.15 ± 0.04 | 0.08 ± 0.02 | 0.738 | |
| 2-phenylethyl acetate | 0.10 ± 0.01 a | 0.09 ± 0.01 a | 0.07 ± 0.01 ab | 0.02 ± 0.00 b | 0.003 | |
| Carbonyl Compounds | furfural | 0.03 ± 0.00 c | 0.04 ± 0.00 bc | 0.07 ± 0.01 a | 0.05 ± 0.00 b | <0.001 |
| benzaldehyde | 0.02 ± 0.00 | 0.03 ± 0.00 | 0.03 ± 0.0 | 0.01 ± 0.00 | 0.126 | |
| acetoine | 3.1 ± 0.5 | 2.1 ± 0.2 | 2.6 ± 0.4 | 1.8 ± 0.3 | 0.427 | |
| Volatile Phenols | 4-vinyl-phenol | 3.8 ± 0.3 b | 5.5 ± 0.4 ab | 5.7 ± 0.4 a | 1.9 ± 0.1 c | <0.001 |
| 4-vinyl-guaiacol | 1.4 ± 0.1 b | 1.6 ± 0.1 ab | 2.0 ± 0.1 a | 0.5 ± 0.0 c | <0.001 | |
| Others | γ-butyrolactone | 1.8 ± 0.2 ab | 1.4 ± 0.1 ab | 1.9 ± 0.2 a | 1.0 ± 0.1 b | 0.016 |
| methionol # | 34.2 ± 2.4 a | 34.1 ± 2.0 a | 37.0 ± 2.1 a | 16.4 ± 1.4 b | <0.001 |
| Family | Compound | M6 | M12 | M18 | M24 | p-Value |
|---|---|---|---|---|---|---|
| Terpenes | linalool 1 | 0.6 a | 0.6 a | 0.7 a | 0.4 b | 0.006 |
| α-terpineol | 0.0 | 0.0 | 0.1 | 0.0 | 0.301 | |
| C6 Compounds | 1-hexanol | 0.3 ab | 0.3 ab | 0.4 a | 0.2 b | 0.008 |
| cis-3-hexen-1-ol | 0.2 | 0.2 | 0.2 | 0.2 | 0.994 | |
| trans-3-hexen-1-ol | 0.0 | 0.0 | 0.0 | 0.0 | 0.819 | |
| Alcohols | methanol | 0.0 | 0.0 | 0.0 | 0.0 | 0.999 |
| 1-propanol | 0.5 | 0.4 | 0.5 | 0.5 | 0.572 | |
| isobutanol | 0.3 | 0.3 | 0.3 | 0.2 | 0.332 | |
| 1-butanol | 0.1 | 0.1 | 0.1 | 0.1 | 0.240 | |
| isoamyl alcohol | 4.1 | 4.0 | 4.0 | 3.8 | 0.781 | |
| benzyl alcohol | 0.0 | 0.0 | 0.0 | 0.0 | 0.999 | |
| 2-phenylethanol | 0.6 a | 0.5 a | 0.6 a | 0.3 b | <0.001 | |
| Ethyl Esters | ethyl butyrate | 1.1 | 1.0 | 1.1 | 0.8 | 0.306 |
| ethyl hexanoate | 8.2 a | 6.7 ab | 7.8 a | 4.8 b | 0.003 | |
| ethyl octanoate | 2.5 a | 2.5 a | 2.1 a | 1.2 b | <0.001 | |
| ethyl decanoate | 1.2 a | 1.3 a | 1.3 a | 0.7 b | 0.001 | |
| ethyl lactate | 0.1 a | 0.1 a | 0.1 a | 0.0 b | 0.003 | |
| diethyl succinate | 0.7 b | 1.0 a | 1.5 a | 0.7 b | <0.001 | |
| Volatile Fatty Acids | isobutyric acid | 0.5 a | 0.4 a | 0.4 a | 0.2 b | 0.002 |
| butyric acid | 0.7 a | 0.7 a | 0.7 a | 0.4 b | <0.001 | |
| isovaleric acid | 25.1 a | 24.4 a | 24.7 a | 13.3 b | <0.001 | |
| hexanoic acid | 1.6 a | 1.4 a | 1.6 a | 0.7 b | <0.001 | |
| octanoic acid | 0.6 a | 0.6 a | 0.7 a | 0.3 b | <0.001 | |
| decanoic acid | 0.4 | 1.1 | 1.1 | 0.1 | 0.471 | |
| Acetates of Higher Alcohols | isoamyl acetate | 13.3 a | 8.7 ab | 6.1 bc | 2.8 c | <0.001 |
| hexyl acetate | 0.2 | 0.2 | 0.2 | 0.1 | 0.877 | |
| 2-phenylethyl acetate | 0.1 | 0.1 | 0.0 | 0.0 | 0.059 | |
| Carbonyl Compounds | furfural | 0.0 | 0.0 | 0.0 | 0.0 | 0.999 |
| benzaldehyde | 0.0 | 0.0 | 0.0 | 0.0 | 0.999 | |
| acetoine | 0.0 | 0.0 | 0.0 | 0.0 | 0.451 | |
| Volatile Phenols | 4-vinyl-phenol | 10.2 b | 14.7 ab | 15.2 a | 5.2 c | <0.001 |
| 4-vinyl-guaiacol | 3.2 b | 3.6 b | 4.6 a | 1.1 c | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vázquez-Pateiro, I.; Arias-González, U.; Mirás-Avalos, J.M.; Falqué, E. Evolution of the Aroma of Treixadura Wines during Bottle Aging. Foods 2020, 9, 1419. https://doi.org/10.3390/foods9101419
Vázquez-Pateiro I, Arias-González U, Mirás-Avalos JM, Falqué E. Evolution of the Aroma of Treixadura Wines during Bottle Aging. Foods. 2020; 9(10):1419. https://doi.org/10.3390/foods9101419
Chicago/Turabian StyleVázquez-Pateiro, Iván, Uxía Arias-González, José Manuel Mirás-Avalos, and Elena Falqué. 2020. "Evolution of the Aroma of Treixadura Wines during Bottle Aging" Foods 9, no. 10: 1419. https://doi.org/10.3390/foods9101419
APA StyleVázquez-Pateiro, I., Arias-González, U., Mirás-Avalos, J. M., & Falqué, E. (2020). Evolution of the Aroma of Treixadura Wines during Bottle Aging. Foods, 9(10), 1419. https://doi.org/10.3390/foods9101419







