Seasonal Variation of Health-Promoting Bioactives in Broccoli and Methyl-Jasmonate Pre-Harvest Treatments to Enhance Their Contents
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Growing Conditions
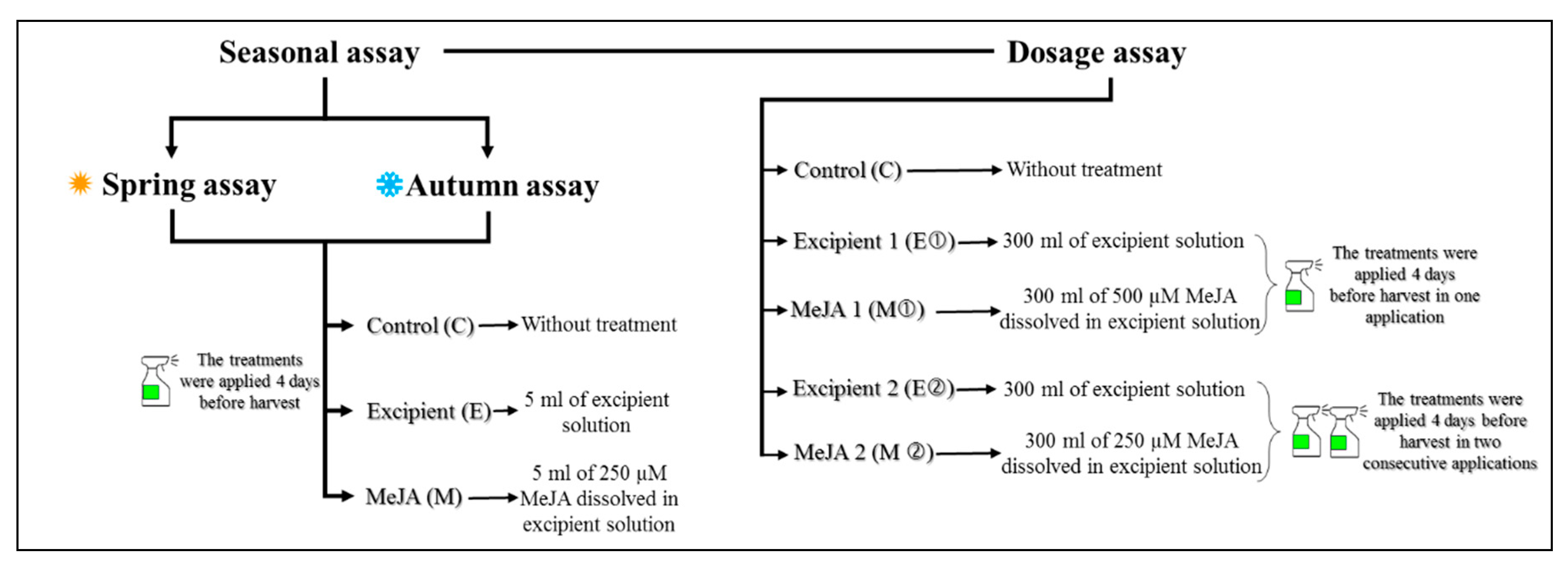
2.2. Experimental Design
2.2.1. Seasonal Assays
2.2.2. Dose of Application Assay
2.3. Color Parameters
2.4. Chlorophyll Determination
2.5. Total Phenolics and Total Flavonoids Analysis
2.6. Oxigen Radical Absorbance Capacity (ORAC)
2.7. Analysis of Carotenoids by High Performance Liquid Chromatography with Diode Array Detection (HPLC-DAD)
2.8. Analysis of Glucosinolates and Phenolic Compounds by HPLC-DAD
2.9. Data Analysis
3. Results and Discussion
3.1. Seasonal Variation of the Quality Parameters
3.1.1. Color Parameters and Chlorophyll Content
3.1.2. Total Phenolic Compounds (TPCs), Total Flavonoids (TF) and Oxygen Radical Absorbance Capacity (ORAC)
3.2. Effect of Elicitor Application Dosage on the Quality Parameters
3.2.1. Color Parameters and Chlorophyll Content
3.2.2. TPC, TF and ORAC
3.3. Changes in the Contents of Carotenoids
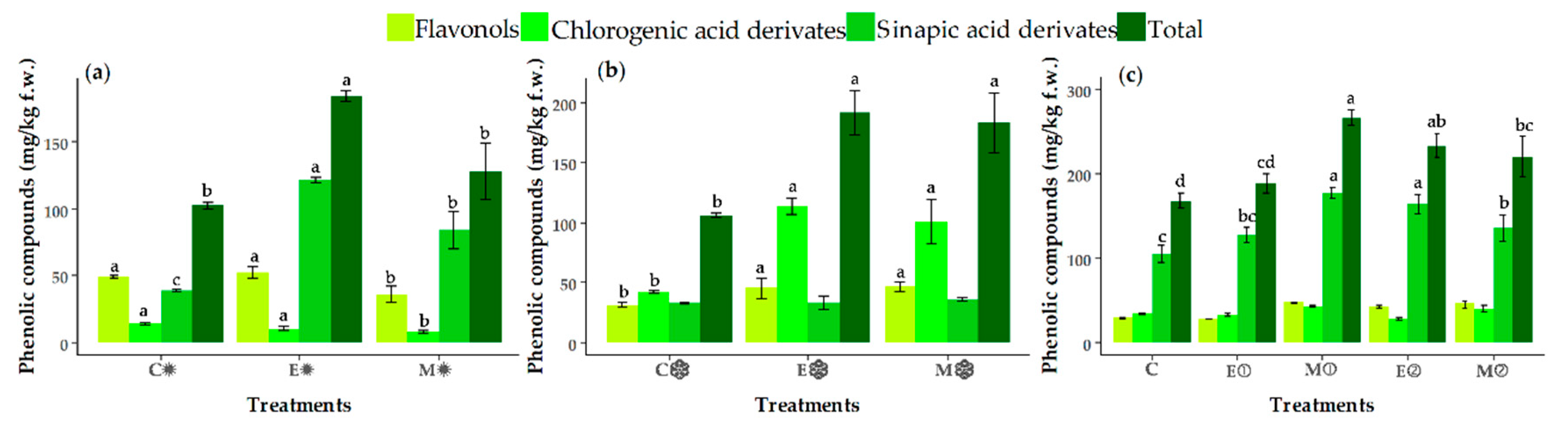
3.4. Changes in the Contents of Individual Phenolic Compounds
3.5. Changes in the Glucosinolate Content
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fahey, J. Brassica: Characteristics and Properties. Encycl. Food Health 2016, 469–477. [Google Scholar] [CrossRef]
- Food and Agriculture Organization Corporate Statistical Database (FAOSTAT). New Food Balances. Available online: http://www.fao.org/faostat/en/#data/FBS (accessed on 2 December 2019).
- Ministry of Agriculture, Fisheries and Food. Available online: https://www.mapa.gob.es/es/estadistica/temas/publicaciones/anuario-de-estadistica/2019/default.aspx?parte=3&capitulo=07&grupo=6&seccion=32 (accessed on 18 September 2020).
- Shi, J.; Gao, L.; Zuo, J.; Wang, Q.; Wang, Q.; Fan, L. Exogenous sodium nitroprusside treatment of broccoli florets extends shelf life, enhances antioxidant enzyme activity, and inhibits chlorophyll-degradation. Postharvest Boil. Technol. 2016, 116, 98–104. [Google Scholar] [CrossRef]
- Branca, F. Cauliflower and Broccoli. In Handbook of Plant Breeding; Springer Science and Business Media LLC: Berlin, Germany, 2007; Volume 1, pp. 151–186. [Google Scholar]
- Ciancaleoni, S.; Onofri, A.; Torricelli, R.; Negri, V. Broccoli yield response to environmental factors in sustainable agriculture. Eur. J. Agron. 2016, 72, 1–9. [Google Scholar] [CrossRef]
- Mijts, B.N.; Lee, P.C.; Schmidt-Dannert, C. Engineering Carotenoid Biosynthetic Pathways. Enzyme Eng. Evolut. Gen. Methods 2004, 388, 315–329. [Google Scholar] [CrossRef]
- Schonhof, I.; Kläring, H.-P.; Krumbein, A.; Clausen, W.; Schreiner, M. Effect of temperature increase under low radiation conditions on phytochemicals and ascorbic acid in greenhouse grown broccoli. Agric. Ecosyst. Environ. 2007, 119, 103–111. [Google Scholar] [CrossRef]
- Moreira-Rodríguez, M.; Nair, V.; Benavides, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. UVA, UVB Light, and Methyl Jasmonate, Alone or Combined, Redirect the Biosynthesis of Glucosinolates, Phenolics, Carotenoids, and Chlorophylls in Broccoli Sprouts. Int. J. Mol. Sci. 2017, 18, 2330. [Google Scholar] [CrossRef]
- Neugart, S.; Baldermann, S.; Hanschen, F.S.; Klopsch, R.; Wiesner-Reinhold, M.; Schreiner, M. The intrinsic quality of brassicaceous vegetables: How secondary plant metabolites are affected by genetic, environmental, and agronomic factors. Sci. Hortic. 2018, 233, 460–478. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, L.; Ser, S.L.; Cumming, J.; Ku, K.-M. Comparative Phytonutrient Analysis of Broccoli By-Products: The Potentials for Broccoli By-Product Utilization. Molecules 2018, 23, 900. [Google Scholar] [CrossRef]
- Dos Reis, L.C.R.; De Oliveira, V.R.; Hagenn, M.; Jablonski, A.; Flôres, S.H.; Rios, A.D.O. Carotenoids, flavonoids, chlorophylls, phenolic compounds and antioxidant activity in fresh and cooked broccoli (Brassica oleracea var. Avenger) and cauliflower (Brassica oleracea var. Alphina F1). LWT 2015, 63, 177–183. [Google Scholar] [CrossRef]
- Minatel, I.O.; Borges, C.V.; Ferreira, M.I.; Gomez, H.A.; Chen, C.Y.; Lima, G.P. Phenolic Compounds: Functional Properties, Impact of Processing and Bioavailability. Phenolic Compd. Biol. Act. 2017, 1–24. [Google Scholar] [CrossRef]
- Cartea, M.E.; Francisco, M.; Soengas, P.; Velasco, P. Phenolic Compounds in Brassica Vegetables. Molecules 2010, 16, 251–280. [Google Scholar] [CrossRef] [PubMed]
- Vallejo, F.; Tomás-Barberán, F.A.; García-Viguera, C. Phenolic compound contents in edible parts of broccoli inflorescences after domestic cooking. J. Sci. Food Agric. 2003, 83, 1511–1516. [Google Scholar] [CrossRef]
- Moreno, D.A.; Carvajal, M.; Lopez-Berenguer, C.; García-Viguera, C. Chemical and biological characterisation of nutraceutical compounds of broccoli. J. Pharm. Biomed. Anal. 2006, 41, 1508–1522. [Google Scholar] [CrossRef]
- Jeffery, E.; E Stewart, K. Upregulation of Quinone Reductase by Glucosinolate Hydrolysis Products from Dietary Broccoli. Enzyme Eng. Evolut. Gen. Methods 2004, 382, 457–469. [Google Scholar] [CrossRef]
- Chiu, Y.-C.; Matak, K.; Ku, K.-M. Methyl jasmonate treated broccoli: Impact on the production of glucosinolates and consumer preferences. Food Chem. 2019, 299, 125099. [Google Scholar] [CrossRef]
- War, A.R.; Paulraj, M.G.; Ahmad, T.; Buhroo, A.A.; Hussain, B.; Ignacimuthu, S.; Sharma, H.C. Mechanisms of plant defense against insect herbivores. Plant Signal. Behav. 2012, 7, 1306–1320. [Google Scholar] [CrossRef]
- Raiola, A.; Errico, A.; Petruk, G.; Monti, D.M.; Barone, A.; Rigano, M.M. Bioactive Compounds in Brassicaceae Vegetables with a Role in the Prevention of Chronic Diseases. Molecules 2017, 23, 15. [Google Scholar] [CrossRef]
- Latté, K.P.; Appel, K.-E.; Lampen, A. Health benefits and possible risks of broccoli—An overview. Food Chem. Toxicol. 2011, 49, 3287–3309. [Google Scholar] [CrossRef]
- Charron, C.S.; Saxton, A.M.; Sams, C.E. Relationship of climate and genotype to seasonal variation in the glucosinolate-myrosinase system. I. Glucosinolate content in ten cultivars ofBrassica oleracea grown in fall and spring seasons. J. Sci. Food Agric. 2005, 85, 671–681. [Google Scholar] [CrossRef]
- Baenas, N.; García-Viguera, C.; Moreno, D.A. Elicitation: A Tool for Enriching the Bioactive Composition of Foods. Molecules 2014, 19, 13541–13563. [Google Scholar] [CrossRef] [PubMed]
- Asensio, E.; Sanvicente, I.; Mallor, C.; Menal-Puey, S. Spanish traditional tomato. Effects of genotype, location and agronomic conditions on the nutritional quality and evaluation of consumer preferences. Food Chem. 2019, 270, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Yuan, G.; Wang, Q. Effect of sucrose and mannitol on the accumulation of health-promoting compounds and the activity of metabolic enzymes in broccoli sprouts. Sci. Hortic. 2011, 128, 159–165. [Google Scholar] [CrossRef]
- Baenas, N.; García-Viguera, C.; Moreno, D.A. Biotic Elicitors Effectively Increase the Glucosinolates Content in Brassicaceae Sprouts. J. Agric. Food Chem. 2014, 62, 1881–1889. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Chen, X.; Jin, P.; Wang, X.; Wang, J.; Zheng, Y. Effect of ethanol treatment on quality and antioxidant activity in postharvest broccoli florets. Eur. Food Res. Technol. 2012, 235, 793–800. [Google Scholar] [CrossRef]
- Duarte-Sierra, A.; Nadeau, F.; Angers, P.; Michaud, M.; Arul, J. UV-C hormesis in broccoli florets: Preservation, phyto-compounds and gene expression. Postharvest Boil. Technol. 2019, 157, 110965. [Google Scholar] [CrossRef]
- Ku, K.M.; Juvik, J. Environmental Stress and Methyl Jasmonate-mediated Changes in Flavonoid Concentrations and Antioxidant Activity in Broccoli Florets and Kale Leaf Tissues. HortScience 2013, 48, 996–1002. [Google Scholar] [CrossRef]
- Parsons, T.R.; Strickland, J.D.H. Discussion of spectrophotometric determination of Marine-plant Pigments, with Revised Equations far Ascertaining Chlorophylls and Carotenoids. J. Mar. Res. 1963, 21, 155–163. [Google Scholar]
- Periago, M.J.; García-Alonso, J.; Jacob, K.; Olivares, A.B.; Bernal, M.J.; Iniesta, M.D.; Martínez, C.; Ros, G. Bioactive compounds, folates and antioxidant properties of tomatoes (Lycopersicum esculentum) during vine ripening. Int. J. Food Sci. Nutr. 2009, 60, 694–708. [Google Scholar] [CrossRef]
- Wheeler, T.L. Organization Security Metrics: Can Organizations Protect Themselves? Inf. Secur. J. A Glob. Perspect. 2008, 17, 228–242. [Google Scholar] [CrossRef]
- Dewanto, V.; Wu, X.; Adom, K.K.; Liu, R.H. Thermal Processing Enhances the Nutritional Value of Tomatoes by Increasing Total Antioxidant Activity. J. Agric. Food Chem. 2002, 50, 3010–3014. [Google Scholar] [CrossRef] [PubMed]
- Prior, R.L.; Hoang, H.; Gu, L.; Wu, X.; Bacchiocca, M.; Howard, L.; Hampsch-Woodill, M.; Huang, D.; Ou, A.B.; Jacob, R. Assays for Hydrophilic and Lipophilic Antioxidant Capacity (oxygen radical absorbance capacity (ORACFL)) of Plasma and Other Biological and Food Samples. J. Agric. Food Chem. 2003, 51, 3273–3279. [Google Scholar] [CrossRef] [PubMed]
- González-Barrio, R.; Periago, M.J.; Luna-Recio, C.; Garcia-Alonso, F.J.; Navarro-González, I.; Javier, G.-A.F. Chemical composition of the edible flowers, pansy (Viola wittrockiana) and snapdragon (Antirrhinum majus) as new sources of bioactive compounds. Food Chem. 2018, 252, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Bravo, S.; García-Alonso, J.; Martín-Pozuelo, G.; Gómez, V.; Santaella, M.; Navarro-González, I.; Periago, M.J. The influence of post-harvest UV-C hormesis on lycopene, β-carotene, and phenolic content and antioxidant activity of breaker tomatoes. Food Res. Int. 2012, 49, 296–302. [Google Scholar] [CrossRef]
- Böhm, V. Use of column temperature to optimize carotenoid isomer separation by C30 high performance liquid chromatography. J. Sep. Sci. 2001, 24, 955–959. [Google Scholar] [CrossRef]
- Francisco, M.; Moreno, D.A.; Cartea, M.E.; Ferreres, F.; García-Viguera, C.; Velasco, P. Simultaneous identification of glucosinolates and phenolic compounds in a representative collection of vegetable Brassica rapa. J. Chromatogr. A 2009, 1216, 6611–6619. [Google Scholar] [CrossRef]
- Baenas, N.; Moreno, D.A.; García-Viguera, C. Selecting Sprouts of Brassicaceae for Optimum Phytochemical Composition. J. Agric. Food Chem. 2012, 60, 11409–11420. [Google Scholar] [CrossRef]
- Wei, K.; Wang, L.; Zhou, J.; He, W.; Zeng, J.; Jiang, Y.; Cheng, H. Catechin contents in tea (Camellia sinensis) as affected by cultivar and environment and their relation to chlorophyll contents. Food Chem. 2011, 125, 44–48. [Google Scholar] [CrossRef]
- Fang, H.; Luo, F.; Li, P.; Zhou, Q.; Zhou, X.; Wei, B.; Cheng, S.; Zhou, H.; Ji, S.-J. Potential of jasmonic acid (JA) in accelerating postharvest yellowing of broccoli by promoting its chlorophyll degradation. Food Chem. 2020, 309, 125737. [Google Scholar] [CrossRef]
- Tan, X.-L.; Fan, Z.-Q.; Shan, W.; Yin, X.-R.; Kuang, J.-F.; Lu, W.-J.; Chen, J.-Y. Association of BrERF72 with methyl jasmonate-induced leaf senescence of Chinese flowering cabbage through activating JA biosynthesis-related genes. Hortic. Res. 2018, 5, 22. [Google Scholar] [CrossRef]
- Fan, X.; Mattheis, J.P.; Fellman, J.K. A role for jasmonates in climacteric fruit ripening. Planta 1998, 204, 444–449. [Google Scholar] [CrossRef]
- El Atki, Y.; Aouam, I.; El Kamari, F.; Taroq, A.; Lyoussi, B.; Taleb, M.; Abdellaoui, A. Total phenolic and flavonoid contents and antioxidant activities of extracts from Teucrium polium growing wild in Morocco. Mater. Today Proc. 2019, 13, 777–783. [Google Scholar] [CrossRef]
- Vallejo, F.; Tomás-Barberán, F.A.; García-Viguera, C. Effect of climatic and sulphur fertilisation conditions, on phenolic compounds and vitamin C, in the inflorescences of eight broccoli cultivars. Eur. Food Res. Technol. 2003, 216, 395–401. [Google Scholar] [CrossRef]
- Shiva, R.B.; Jung-Ho, K. Seasonal variation in phytochemicals and antioxidant activities in different tissues of various Broccoli cultivars. Afr. J. Biotechnol. 2014, 13, 604–615. [Google Scholar] [CrossRef]
- Cohen, S.D.; Tarara, J.M.; A Kennedy, J. Assessing the impact of temperature on grape phenolic metabolism. Anal. Chim. Acta 2008, 621, 57–67. [Google Scholar] [CrossRef]
- Guidi, L.; Brunetti, C.; Fini, A.; Agati, G.; Ferrini, F.; Gori, A.; Tattini, M. UV radiation promotes flavonoid biosynthesis, while negatively affecting the biosynthesis and the de-epoxidation of xanthophylls: Consequence for photoprotection? Environ. Exp. Bot. 2016, 127, 14–25. [Google Scholar] [CrossRef]
- Natella, F.; Maldini, M.; Nardini, M.; Azzini, E.; Foddai, M.S.; Giusti, A.M.; Baima, S.; Morelli, G.; Scaccini, C. Improvement of the nutraceutical quality of broccoli sprouts by elicitation. Food Chem. 2016, 201, 101–109. [Google Scholar] [CrossRef]
- Carvacho, H.B.; Pérez, C.; Zúñiga, G.E.; Mahn, A. Effect of methyl jasmonate, sodium selenate and chitosan as exogenous elicitors on the phenolic compounds profile of broccoli sprouts. J. Sci. Food Agric. 2014, 94, 2555–2561. [Google Scholar] [CrossRef]
- Strati, I.F.; Sinanoglou, V.J.; Kora, L.; Miniadis-Meimaroglou, S.; Oreopoulou, V. Carotenoids from Foods of Plant, Animal and Marine Origin: An Efficient HPLC-DAD Separation Method. Foods 2012, 1, 52–65. [Google Scholar] [CrossRef]
- Becerra-Moreno, A.; Alanís-Garza, P.A.; Mora-Nieves, J.L.; Mora-Mora, J.P.; Jacobo-Velázquez, D.A. Kale: An excellent source of vitamin C, pro-vitamin A, lutein and glucosinolates. CyTA J. Food 2013, 12, 298–303. [Google Scholar] [CrossRef]
- Villarreal-García, D.; Alanís-Garza, P.A.; Cuéllar-Villarreal, M.D.R.; Redondo-Gil, M.; Mora-Nieves, J.L.; Jacobo-Velázquez, D.A. Effects of different defrosting methods on the stability of bioactive compounds and consumer acceptability of frozen broccoli. CyTA J. Food 2014, 13, 312–320. [Google Scholar] [CrossRef]
- Samuolienė, G.; Brazaitytė, A.; Viršilė, A.; Miliauskienė, J.; Vaštakaitė-Kairienė, V.; Duchovskis, P. Nutrient Levels in Brassicaceae Microgreens Increase Under Tailored Light-Emitting Diode Spectra. Front. Plant Sci. 2019, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Thoma, F.; Somborn-Schulz, A.; Schlehuber, D.; Keuter, V.; Deerberg, G. Effects of Light on Secondary Metabolites in Selected Leafy Greens: A Review. Front. Plant Sci. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Wierstra, I. Differential Effects of Methyl Jasmonate on the Expression of the Early Light-Inducible Proteins and Other Light-Regulated Genes in Barley. Plant Physiol. 2000, 124, 833–844. [Google Scholar] [CrossRef] [PubMed]
- Collini, E. Carotenoids in Photosynthesis: The Revenge of the “Accessory” Pigments. Chem 2019, 5, 494–495. [Google Scholar] [CrossRef]
- Kirilovsky, D.; Kerfeld, C.A. The Orange Carotenoid Protein: A blue-green light photoactive protein. Photochem. Photobiol. Sci. 2013, 12, 1135. [Google Scholar] [CrossRef] [PubMed]
- Neugart, S.; Fiol, M.; Schreiner, M.; Rohn, S.; Zrenner, R.; Kroh, L.W.; Krumbein, A. Interaction of Moderate UV-B Exposure and Temperature on the Formation of Structurally Different Flavonol Glycosides and Hydroxycinnamic Acid Derivatives in Kale (Brassica oleracea var. sabellica). J. Agric. Food Chem. 2014, 62, 4054–4062. [Google Scholar] [CrossRef]
- Rechner, O.; Neugart, S.; Schreiner, M.; Wu, S.; Poehling, H.-M. Different Narrow-Band Light Ranges Alter Plant Secondary Metabolism and Plant Defense Response to Aphids. J. Chem. Ecol. 2016, 42, 989–1003. [Google Scholar] [CrossRef]
- Pérez-Balibrea, S.; Moreno, D.A.; García-Viguera, C. Improving the phytochemical composition of broccoli sprouts by elicitation. Food Chem. 2011, 129, 35–44. [Google Scholar] [CrossRef]
- Ku, K.-M.; Jeffery, E.; Juvik, J. Optimization of methyl jasmonate application to broccoli florets to enhance health-promoting phytochemical content. J. Sci. Food Agric. 2014, 94, 2090–2096. [Google Scholar] [CrossRef]
- Champolivier, L.; Merrien, A. Effects of water stress applied at different growth stages to Brassica napus L. var. oleifera on yield, yield components and seed quality. Eur. J. Agron. 1996, 5, 153–160. [Google Scholar] [CrossRef]
- Ku, K.-M.; Jeffery, E.; Juvik, J.A. Influence of Seasonal Variation and Methyl Jasmonate Mediated Induction of Glucosinolate Biosynthesis on Quinone Reductase Activity in Broccoli Florets. J. Agric. Food Chem. 2013, 61, 9623–9631. [Google Scholar] [CrossRef] [PubMed]
- Mølmann, J.A.; Steindal, A.L.; Bengtsson, G.B.; Seljåsen, R.; Lea, P.; Skaret, J.; Johansen, T.J. Effects of temperature and photoperiod on sensory quality and contents of glucosinolates, flavonols and vitamin C in broccoli florets. Food Chem. 2015, 172, 47–55. [Google Scholar] [CrossRef] [PubMed]

| Season | Treatment | L* | a* | b* | C* | h* | Total Chlorophylls | TPCs | TF | ORAC |
|---|---|---|---|---|---|---|---|---|---|---|
| Spring | C✹ | 43 ± 2 a | −8 ± 2 | 13 ± 4 | 15 ± 4 | 122 ± 7 | 149 ± 17 | 1081 ± 147 | 207 ± 37 | 31 ± 9 ab |
| E✹ | 42 ± 2 ab | −7 ± 2 | 14 ± 4 | 15. ± 4 | 116 ± 7 | 152 ± 38 | 932 ± 29 | 187 ± 24 | 20 ± 8 b | |
| M✹ | 41 ± 2 b | −5 ± 4 | 15 ± 3 | 16 ± 3 | 108 ± 12 | 128 ± 27 | 1102 ± 136 | 180 ± 13 | 44 ± 0 a | |
| Autumn | C❄ | 38 ± 2 b | −10 ± 1 | 10 ± 2 | 14 ± 2 b | 135 ± 3 | 264 ± 25 a | 828 ± 86 | 74 ± 7 b | 18 ± 4 ab |
| E❄ | 41 ± 2 a | −11 ± 2 | 14 ± 4 | 18 ± 4 a | 129 ± 4 | 147 ± 32 c | 912 ± 113 | 92 ± 15 a | 22 ± 2 a | |
| M❄ | 37 ± 1 b | −10 ± 1 | 12 ± 2 | 16 ± 2 ab | 132 ± 3 | 202 ± 29 b | 870 ± 108 | 26 ± 8 c | 14 ± 6 b | |
| ANOVA | SEASON | *** | *** | * | ns | *** | *** | *** | *** | *** |
| TREATMENT | *** | ns | ns | ns | ns | *** | ns | *** | ** | |
| T × S | ** | * | ns | ns | * | *** | ** | *** | *** |
| Sample | L* | a* | b* | C* | h* | Total Chlorophylls | TPCs | TF | ORAC |
|---|---|---|---|---|---|---|---|---|---|
| C | 20 ± 14 | −4 ± 3 | 5 ± 2 | 7 ± 3 | 121 ± 14 | 362 ± 32 a | 2578 ± 157 a | 409 ± 44 | 66 ± 7 b |
| E① | 17 ± 12 | −2 ± 3 | 6 ± 3 | 6 ± 4 | 102 ± 20 | 276 ± 27 b | 2531 ± 4 ab | 405 ± 48 | 75 ± 5 ab |
| M① | 16 ± 6 | −4 ± 0 | 9 ± 0 | 10 ± 0 | 114 ± 2 | 205 ± 28 b | 2038 ± 179 ab | 444 ± 55 | 79 ± 6 a |
| E② | 18 ± 11 | −2 ± 2 | 8 ± 4 | 8 ± 4 | 103 ± 11 | 202 ± 21 b | 2342 ± 295 ab | 477 ±53 | 69 ± 6 b |
| M② | 27 ± 23 | −6 ± 7 | 15 ± 9 | 16 ± 11 | 107 ± 11 | 243 ± 30 b | 1994 ± 261 b | 446 ± 52 | 83 ± 3 a |
| TREATMENT | ns | ns | ns | ns | ns | ns | ** | ns | *** |
| DOSAGE | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| T × D | ns | ns | ns | ns | ns | ** | ns | ns | * |
| ANOVA | B-Carotene | Lutein | Neoxanthin | Violaxanthin | Total Carotenoids | Flavonols | Chlorogenic Acid Derivatives | Sinapic Acid Derivatives | Total Phenolics | GIB | GRA | HGB | GBS | MGB | NGB | Aliphatics | Indoles | Total GLS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SEASON | *** | ns | *** | *** | *** | *** | *** | *** | ** | ** | *** | *** | *** | ** | *** | *** | *** | *** |
| TREATMENT | *** | ns | ** | ns | *** | *** | * | *** | *** | ns | * | *** | ** | ns | * | ns | * | ns |
| T × S | *** | ns | *** | * | *** | *** | ** | *** | * | ns | ** | *** | ns | * | *** | ns | ** | ns |
| TREATMENT | *** | *** | * | ** | *** | *** | *** | *** | *** | ns | *** | − | * | *** | *** | *** | *** | *** |
| DOSAGE | ** | * | ns | ns | ** | *** | ** | * | * | ns | *** | − | ** | *** | *** | ns | *** | *** |
| T × D | * | ns | ns | ns | * | *** | Ns | *** | *** | ns | *** | - | *** | *** | *** | ** | *** | *** |
| Assay | Sample | Glucosinolates | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| GIB | GRA | HGB | GBS | MGB | NGB | Total Aliphatic | Total Indole | Total | ||
| Spring | C✹ | 198 ± 9 a | 48 ± 20 b | 3 ± 1 b | 23 ± 1 | 19 ± 1 | 20 ± 0 b | 245 ± 16 | 65 ± 2 | 310 ± 15 |
| E✹ | 127 ± 8 b | 124 ± 35 a | 10 ± 1 a | 23 ± 2 | 25 ± 1 | 12 ± 0 b | 251 ± 27 | 70 ± 4 | 322 ± 23 | |
| M✹ | 150 ± 34 ab | 106 ± 9 ab | 4 ± 1 b | 16 ± 5 | 19 ± 5 | 57 ± 15 a | 255 ± 43 | 96 ± 26 | 351 ± 69 | |
| Autumn | C❄ | 254 ± 63 | 300 ± 9 a | 2 ± 1 | 119 ± 12 ab | 39 ± 10 | 53 ± 13 b | 554 ± 64 | 212 ± 16 a | 766 ± 77 |
| E❄ | 227 ± 3 | 300 ± 18 a | 1 ± 1 | 132 ± 1 a | 27 ± 2 | 71 ± 1 a | 527 ± 20 | 232 ± 2 a | 759 ± 20 | |
| M❄ | 219 ± 12 | 258 ± 19 b | 1 ± 0 | 107 ± 7 b | 27 ± 2 | 48 ± 5 b | 477 ± 32 | 183 ± 12 b | 661 ± 43 | |
| Dosage Application | C | 153 ± 8 | 216 ± 16 a | tr | 8 ± 1 c | trb | 0 ± 1 c | 369 ± 24 a | 8 ± 2 c | 377 ± 25 b |
| E① | 169 ± 6 | 137 ± 3 b | tr | 14 ± 0 b | 1 ± 0 b | 19 ± 1 b | 306 ± 9 b | 34 ± 2 b | 340 ± 11 b | |
| M① | 153 ± 50 | 82 ± 13 d | tr | tr d | trb | 25 ± 8 b | 236 ± 40 c | 25 ± 8 b | 261 ± 32 c | |
| E② | 171 ± 7 | 103 ± 4 cd | tr | 1 ± 2 d | 1 ± 1 b | 6 ± 3 c | 273 ± 11 bc | 84 c | 281 ± 14 c | |
| M② | 186 ± 8 | 106 ± 5 c | tr | 21 ± 4 a | 37 ± 2 a | 175 ± 6 a | 291 ± 13 bc | 233 ± 12 a | 524 ± 25 a | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nuñez-Gómez, V.; Baenas, N.; Navarro-González, I.; García-Alonso, J.; Moreno, D.A.; González-Barrio, R.; Periago-Castón, M.J. Seasonal Variation of Health-Promoting Bioactives in Broccoli and Methyl-Jasmonate Pre-Harvest Treatments to Enhance Their Contents. Foods 2020, 9, 1371. https://doi.org/10.3390/foods9101371
Nuñez-Gómez V, Baenas N, Navarro-González I, García-Alonso J, Moreno DA, González-Barrio R, Periago-Castón MJ. Seasonal Variation of Health-Promoting Bioactives in Broccoli and Methyl-Jasmonate Pre-Harvest Treatments to Enhance Their Contents. Foods. 2020; 9(10):1371. https://doi.org/10.3390/foods9101371
Chicago/Turabian StyleNuñez-Gómez, Vanesa, Nieves Baenas, Inma Navarro-González, Javier García-Alonso, Diego A. Moreno, Rocío González-Barrio, and Mª Jesús Periago-Castón. 2020. "Seasonal Variation of Health-Promoting Bioactives in Broccoli and Methyl-Jasmonate Pre-Harvest Treatments to Enhance Their Contents" Foods 9, no. 10: 1371. https://doi.org/10.3390/foods9101371
APA StyleNuñez-Gómez, V., Baenas, N., Navarro-González, I., García-Alonso, J., Moreno, D. A., González-Barrio, R., & Periago-Castón, M. J. (2020). Seasonal Variation of Health-Promoting Bioactives in Broccoli and Methyl-Jasmonate Pre-Harvest Treatments to Enhance Their Contents. Foods, 9(10), 1371. https://doi.org/10.3390/foods9101371








