Vitamin D3 Loaded Niosomes and Transfersomes Produced by Ethanol Injection Method: Identification of the Critical Preparation Step for Size Control
Abstract
1. Introduction
2. Materials and Methods
2.1. Vesicles Formulation
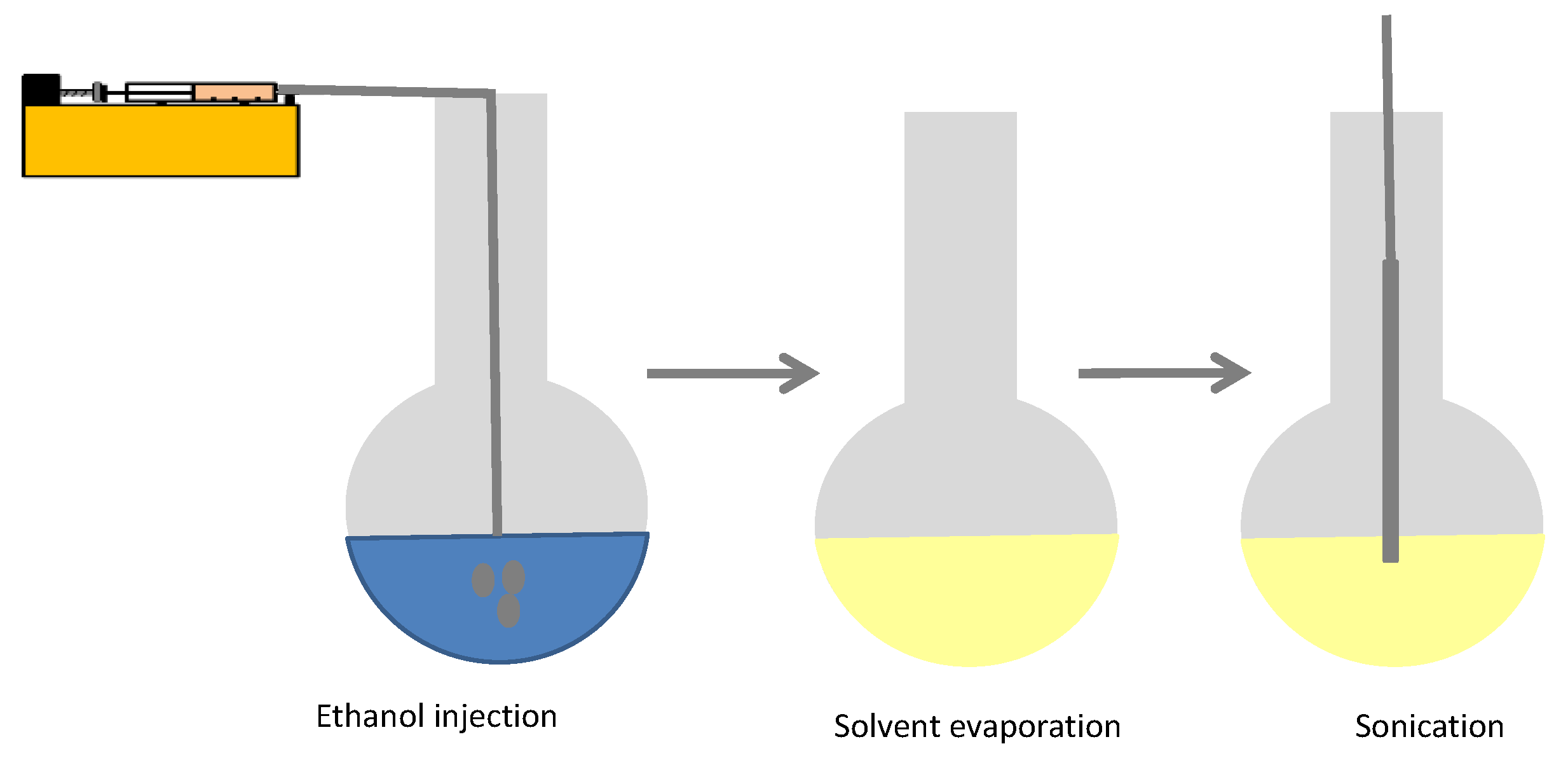
2.2. Preparation Method
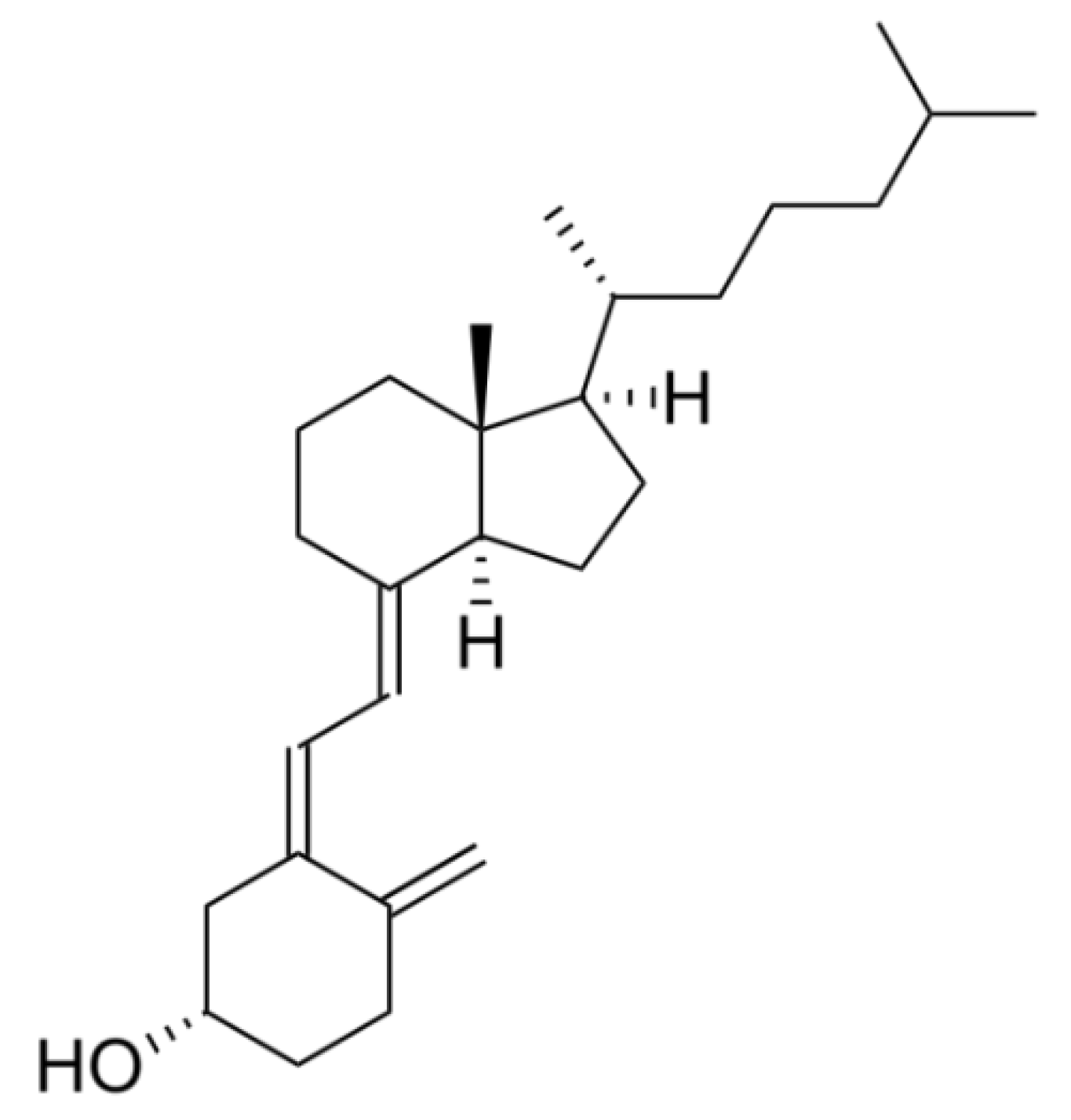
2.3. Vitamine D3 Encapsulation
2.4. Characterization of Nanovesicles
2.5. Statistics Analysis
3. Results
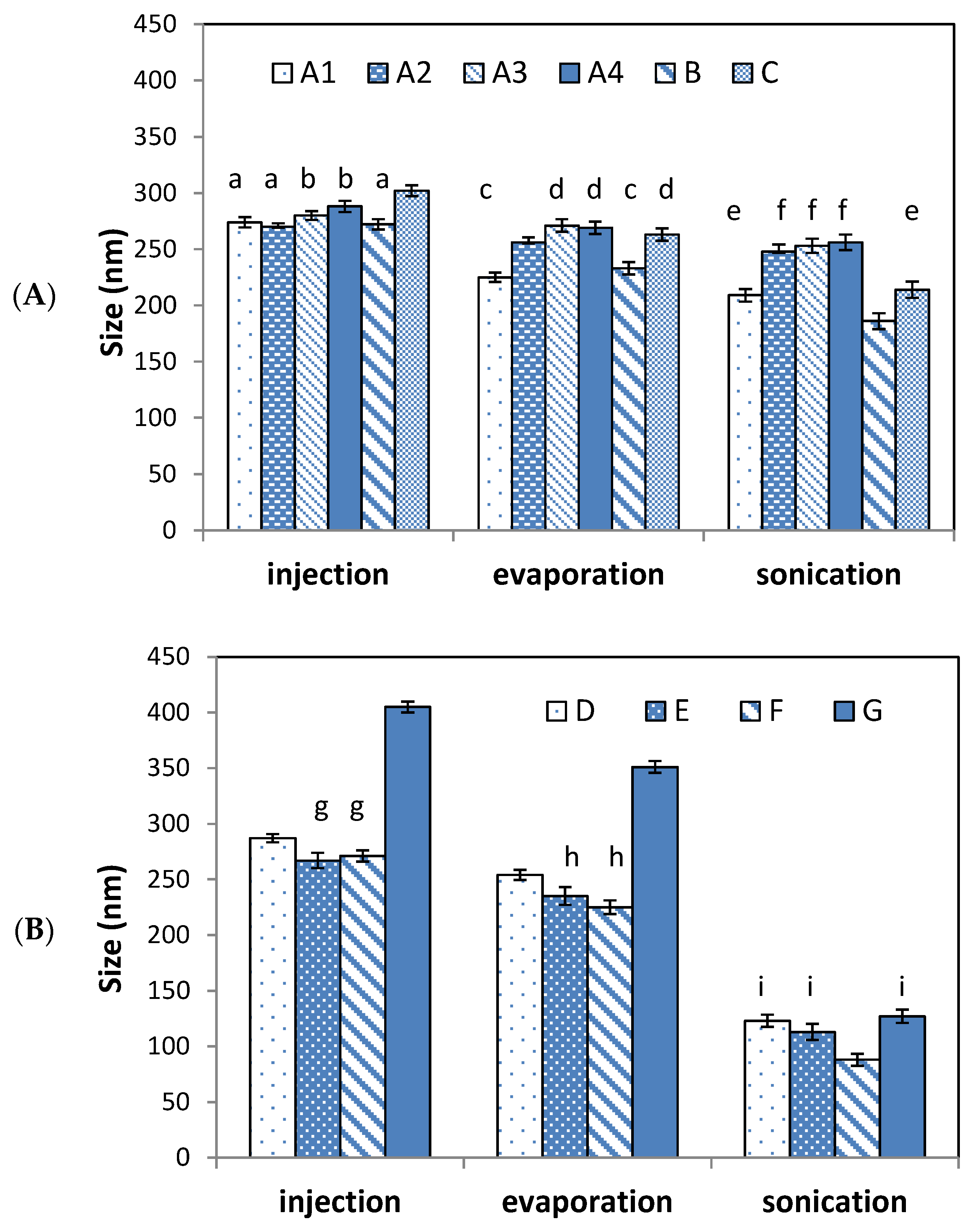
3.1. Effect on Particle Size
3.2. Vesicles as Biocompounds Carriers
3.2.1. Encapsulation Efficiency
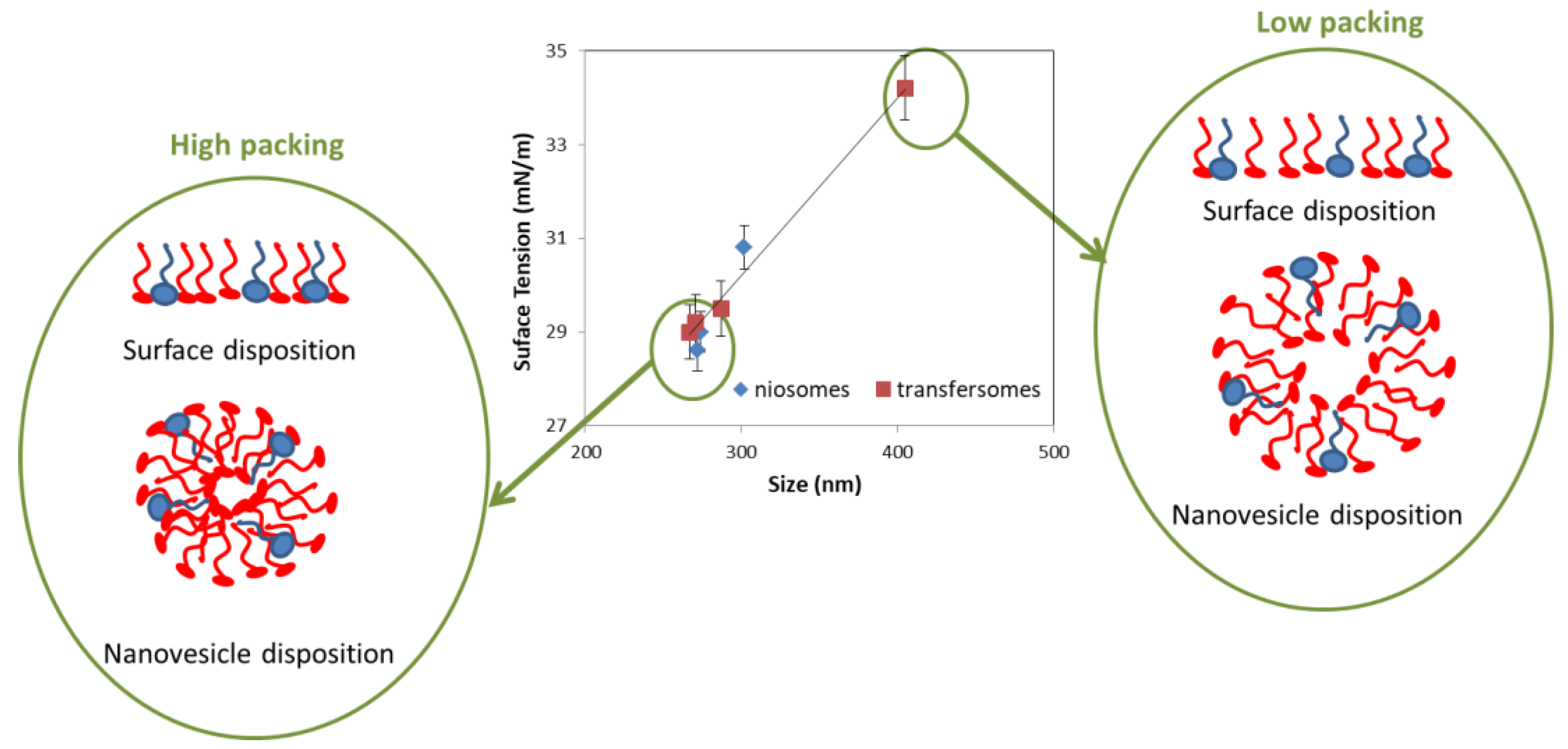
3.2.2. Stability
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Luo, Y.; Teng, Z.; Wang, Q. Development of zein nanoparticles coated with carboxymethyl chitosan for encapsulation and controlled release of vitamin D3. J. Agric. Food Chem. 2012, 60, 836–843. [Google Scholar] [CrossRef]
- Lin, Y.; Wang, Y.H.; Yang, X.Q.; Guo, J.; Wang, J.M. Corn protein hydrolysate as a novel nano-vehicle: Enhanced physicochemical stability and in vitro bioaccessibility of vitamin D3. LWT Food Sci. Techn. 2016, 72, 510–517. [Google Scholar]
- Liu, K.; Kong, X.L.; Li, Q.M.; Zhang, H.L.; Zha, X.Q.; Luo, J.P. Stability and bioavailability of vitamin D3 encapsulated in composite gels of whey protein isolate and lotus root amylopectin. Carbohydr. Polym. 2020, 227, 115337. [Google Scholar] [CrossRef]
- Teng, Z.; Luo, Y.; Wang, Q. Carboxymethyl chitosan—Soy protein complex nanoparticles for the encapsulation and controlled release of vitamin D3. Food Chem. 2013, 141, 524–532. [Google Scholar] [CrossRef]
- Ozturk, B.; Argin, S.; Ozilgen, M.; McClements, D. Formation and Stabilization of Nanoemulsion-Based Vitamin E Delivery Systems using Natural Biopolymers: Whey Protein Isolate and Gum Arabic. Food Chem. 2015, 188, 256–263. [Google Scholar] [CrossRef]
- Ziani, K.; Fang, Y.; McClements, D. Encapsulation of functional lipophilic components in surfactant-based colloidal delivery systems: Vitamin E, vitamin D, and lemon oil. Food Chem. 2012, 134, 1106–1112. [Google Scholar] [CrossRef]
- Mitbumrung, W.; Suphantharika, M.; McClements, D.J.; Winuprasith, T. Encapsulation of Vitamin D3 in Pickering Emulsion Stabilized by NanofibrillatedMangosteen Cellulose: Effect of Environmental Stresses. J. Food Sci. 2019, 84, 3213–3221. [Google Scholar] [CrossRef]
- Jaafari-Maalej, C.; Diab, R.; Andrieu, V.; Elaissari, A.; Fessi, H. Ethanol injectionmethod for hydrophilic and lipophilic drug-loaded liposome preparation. J. Liposome Res. 2010, 20, 228–243. [Google Scholar] [CrossRef]
- Mehanna, M.M.; Motawaa, A.M.; Samaha, M.W. Nanovesicular carrier-mediated Transdermal delivery of Tadalafil: I-Formulation and physicochemical characterization. Drug Dev. Ind. Pharm. 2015, 41, 714–721. [Google Scholar] [CrossRef]
- Pardakhty, A.; Moazeni, E. Nano-niosomes in drug, vaccine and gene delivery: A rapid overview. Nanomed. J. 2013, 1, 1–12. [Google Scholar]
- Marianecci, C.; di Marzio, L.; Rinaldi, F.; Celia, C.; Paolino, D.; Alhaique, F.; Esposito, S.; Carafa, M. Niosomes from 80s to present: The state of the art. Adv. Colloid Interface Sci. 2014, 205, 187–206. [Google Scholar] [CrossRef] [PubMed]
- Rathee, J.; Kanwar, R.; Kaushik, D.; Salunke, D.B.; Mehta, S.K. Niosomes as efficient drug delivery modules for encapsulation of Toll-like receptor 7 agonists and IDO-inhibitor. Appl. Surf. Sci. 2020, 505, a144078. [Google Scholar] [CrossRef]
- Pando, D.; Beltrán, M.; Gerone, I.; Matos, M.; Pazos, C. Resveratrol entrapped niosomes as yogurt additive. Food Chem. 2015, 170, 281–287. [Google Scholar] [CrossRef]
- Lohani, A.; Verma, A. Vesicles: Potential nano carriers for the delivery of skin cosmetics. Cosmet. Laser Ther. 2017, 19, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Mali, N.; Darandale, S.; Vavia, P. Niosomes as vesicular carrier for topical administration of minoxidil: formulation and in vitro assessment. Drug Deliv. Trans. Re. 2013, 3, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Matos, M.; Gutiérrez, G.; Iglesias, O.; Coca, J.; Pazos, C. Enhancing encapsulation efficiency of food-grade double emulsions containing resveratrol or vitamin B12 by membrane emulsification. J. Food Eng. 2015, 166, 212–220. [Google Scholar] [CrossRef]
- Meng, S.; Sun, L.; Wang, L.; Lin, Z.; Liu, Z.; Xi, L.; Wang, Z.; Zheng, Y. Loading of water-insoluble celastrol into niosome hydrogels for improved topical permeation and anti-psoriasis activity. Colloids Surf. B Biointerfaces 2019, 182, 110352. [Google Scholar] [CrossRef]
- Ghadi, Z.; Dinarvand, R.; Asemi, N.; Amiri, F.T.; Ebrahimnejad, P. Preparation, characterization and in vivo evaluation of novel hyaluronan containing niosomes tailored by Box-Behnken design to co-encapsulate curcumin and quercetin. Eur. J. Pharm. Sci. 2019, 130, 234–246. [Google Scholar] [CrossRef]
- Chaikul, P.; Khat-udomkiri, N.; Iangthanarat, K.; Manosroi, J.; Manosroi, A. Characteristics and in vitro anti-skin aging activity of gallic acid loaded in cationic CTAB noisome. Eur. J. Pharm. Sci. 2019, 131, 39–49. [Google Scholar] [CrossRef]
- Pando, D.; Caddeo, C.; Manconi, M.; Fadda, A.M.; Pazos, C. Nanodesign of olein vesicles for the topical delivery of the antioxidant resveratrol. Eur. J. Pharm. Pharmacol. 2013, 65, 1158–1167. [Google Scholar] [CrossRef]
- Hasibi, F.; Nasirpour, A.; Varshosaz, J.; García-Manrique, P.; Gutiérrez, G.; Matos, M. Formulation and Characterization of Taxifolin-Loaded Lipid Nanovesicles (Liposomes, Niosomes, and Transfersomes) for Beverage Fortification. Eur. J. Lipid Sci. Tech. 2020, 122, a1900105. [Google Scholar] [CrossRef]
- Caddeo, C.; Manca, M.L.; Matos, M.; Gutierrez, G.; Díez-Sales, O.; Peris, J.E.; Usach, I.; Fernàndez-Busquets, X.; Fadda, A.M.; Manconi, M. Functional response of novel bioprotective poloxamer-structured vesicles on inflamed skin. Nanomedicine 2017, 13, 1127–1136. [Google Scholar] [CrossRef] [PubMed]
- Vitonyte, J.; Manca, M.L.; Caddeo, C.; Valenti, D.; Peris, J.E.; Usach, I.; Nacher, A.; Matos, M.; Gutiérrez, G.; Orrù, G.; et al. Bifunctional viscous nanovesicles co-loaded with resveratrol and gallic acid for skin protection against microbial and oxidative injuries. Eur. J. P. Biopharm. 2017, 114, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Pan, Z.; Pei, F.; Jin, Z.; Feng, G. In situ growth of hollow Cu2O spheres using anionic vesicles as soft templates. J. Ind. Eng. Chem. 2018, 59, 410–415. [Google Scholar] [CrossRef]
- Umh, H.N.; Kim, Y. Spectroscopic and microscopic studies of vesicle rupture by AgNPs attack to screen the cytotoxicity of nanomaterials. J. Ind. Eng. Chem. 2013, 19, 1944–1948. [Google Scholar] [CrossRef]
- Wang, G.; Zhang, D.; Du, Z.; Li, P. Spontaneous vesicle formation from trisiloxane-tailed gemini surfactant. J. Ind. Eng. Chem. 2014, 20, 1247–1250. [Google Scholar] [CrossRef]
- Moghassemi, S.; Hadjizadeh, A. Nano-niosomes as nanoscale drug delivery systems: An illustrated review. J. Control. Release 2014, 185, 22–36. [Google Scholar] [CrossRef]
- Mahale, N.B.; Thakkar, P.D.; Mali, R.G.; Walunj, D.R.; Chaudhari, S.R. Niosomes: Novel sustained release nonionic stable vesicular systems—An overview. Adv. Colloid Interface Sci. 2012, 183–184, 46–54. [Google Scholar] [CrossRef]
- Grimaldi, N.; Andrade, F.; Segovia, N.; Ferrer-Tasies, L.; Sala, S.; Veciana, J.; Ventosa, N. Lipid-based nanovesicles for nanomedicine. Chem. Soc. Rev. 2016, 45, 6520–6545. [Google Scholar] [CrossRef]
- García-Manrique, P.; Machado, N.D.; Fernández, M.A.; Blanco-López, M.C.; Matos, M.; Gutiérrez, G. Effect of drug molecular weight on niosomes size and encapsulation efficiency. Colloids Surf. B Biointerfaces 2020, 186, 110711. [Google Scholar] [CrossRef]
- Charcosset, C.; Juban, A.; Valour, J.P.; Urbaniak, S.; Fessi, H. Preparation of liposomes at large scale using the ethanol injection method: Effect of scale-up and injection devices. Chem. Eng. Res. Des. 2015, 94, 508–515. [Google Scholar] [CrossRef]
- Machado, N.D.; García-Manrique, P.; Fernández, M.A.; Blanco-López, M.C.; Matos, M.; Gutiérrez, G. Cholesterol free niosome production by microfluidics: comparative with other conventional methods. Chem. Eng. Res. Des. 2020, 162, 162–171. [Google Scholar] [CrossRef]
- De Leo, V.; Ruscignoa, S.; Trapani, A.; di Gioia, S.; Milano, F.; Mandracchia, D.; Comparelli, R.; Castellani, S.; Agostiano, A.; Trapanic, G.L.; et al. Preparation of drug-loaded small unilamellar liposomes and evaluation of their potential for the treatment of chronic respiratory diseases. Int. J. Pharm. 2018, 545, 378–388. [Google Scholar] [CrossRef] [PubMed]
- Jahn, A.; Vreeland, W.N.; DeVoe, D.L.; Locascio, L.E.; Michael, G. Microfluidic directed formation of liposomes of controlled size. Langmuir 2007, 23, 6289–6293. [Google Scholar] [CrossRef] [PubMed]
- Batzri, S.; Korn, E.D. Single bilayer liposomes prepared without sonication. Biochim. Biophys. Acta Biomembr. 1973, 298, 1015–1019. [Google Scholar] [CrossRef]
- García-Manrique, P.; Matos, M.; Gutiérrez, G.; Estupiñán, R.O.; Blanco-López, C.M.; Pazos, C. Using Factorial Experimental Design to Prepare Size-Tuned Nanovesicles. Ind. Eng. Chem. Res. 2016, 55, 9164–9175. [Google Scholar] [CrossRef]
- Pando, D.; Matos, M.; Gutiérrez, G.; Pazos, C. Formulation of resveratrol entrapped niosomes for topical use. Colloids Surf. B 2015, 128, 398–404. [Google Scholar] [CrossRef]
- García-Manrique, P.; Gutiérrez, P.G.; Matos, M.; Cristaldi, A.; Mosayyebi, A.; Carugo, D.; Zhang, X.; Blanco-Lopez, M.C. Continuous flow production of size-controllable niosomes using a thermostatic microreactor. Colloids Surf. B 2019, 182, a110378. [Google Scholar] [CrossRef]
- Gutiérrez, G.; Benito, J.M.; Pazos, C.; Coca, J. Evaporation of aqueous dispersed systems and concentrated emulsions formulated with non-ionic surfactants. Int. J. Heat Mass Tran. 2014, 69, 117–128. [Google Scholar] [CrossRef]
- Roque, L.; Fernández, M.; Benito, J.M.; Escudero, I. Stability and characterization studies of Span 80 niosomes modified with CTAB in the presence of NaCl. Colloids Surf. A Physicochem. Eng. Asp. 2020, 601, a124999. [Google Scholar] [CrossRef]
- Braun, R.; Sachs, J.N. α-Synuclein Reduces Tension and Increases Undulations in Simulations of Small Unilamellar Vesicles. Biophys. J. 2015, 108, 1848–1851. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Guo, J.; Ma, J.; Liu, P.; Zhang, J. Cationic silicon-based gemini surfactants: Effect of hydrophobic chains on surface activity, physic-chemical properties and aggregation behaviors. J. Ind. Eng. Chem 2017, 53, 51–61. [Google Scholar] [CrossRef]
- Barani, M.; Mirzaei, M.; Torkzadeh-Mahani, M.; Lohrasbi-Nejad, A.; Nematollahi, M.H. A new formulation of hydrophobin-coated niosome as a drug carrier to cancer cells. Mat. Sci. Eng. 2020, 113, a110975. [Google Scholar] [CrossRef] [PubMed]
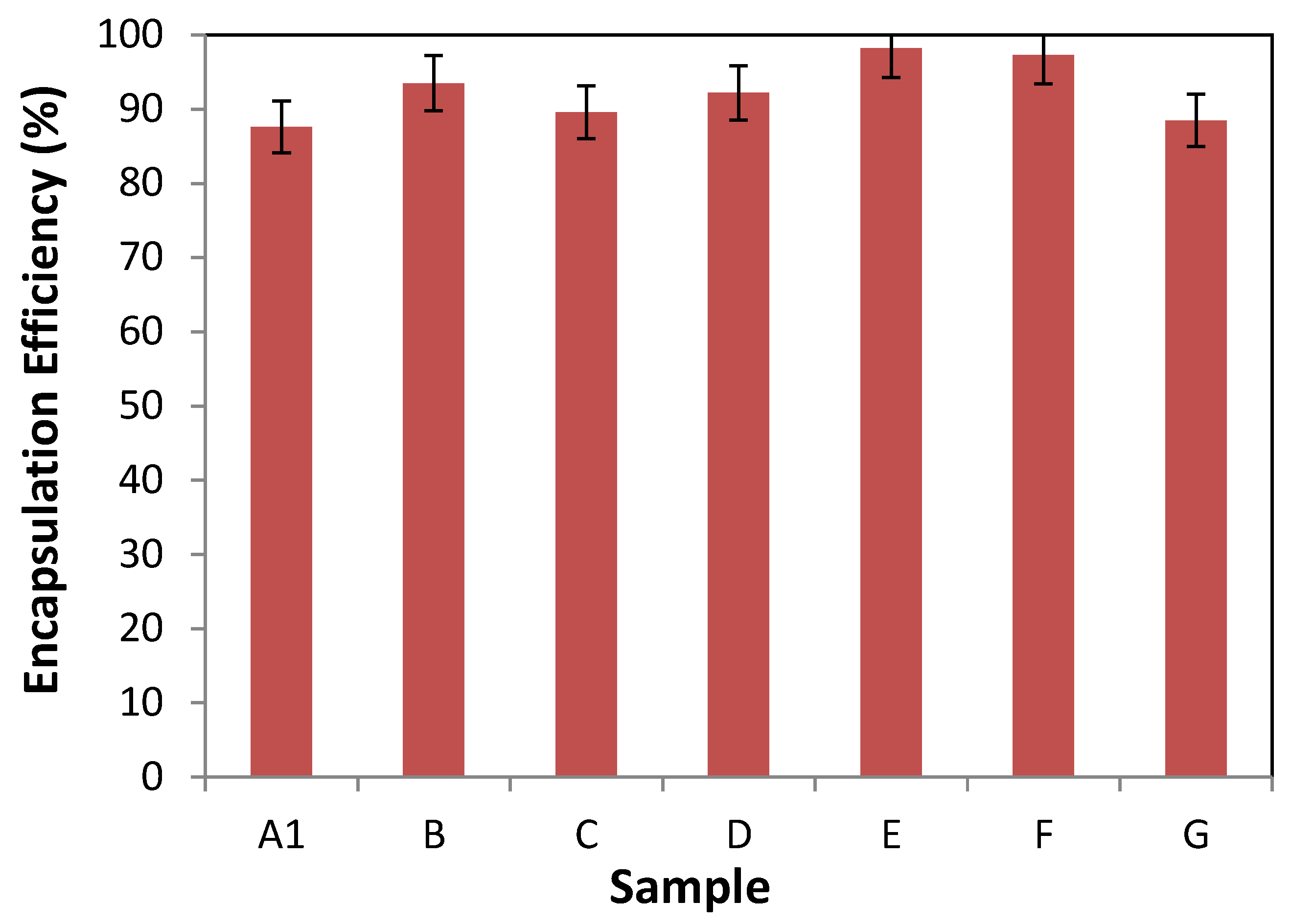
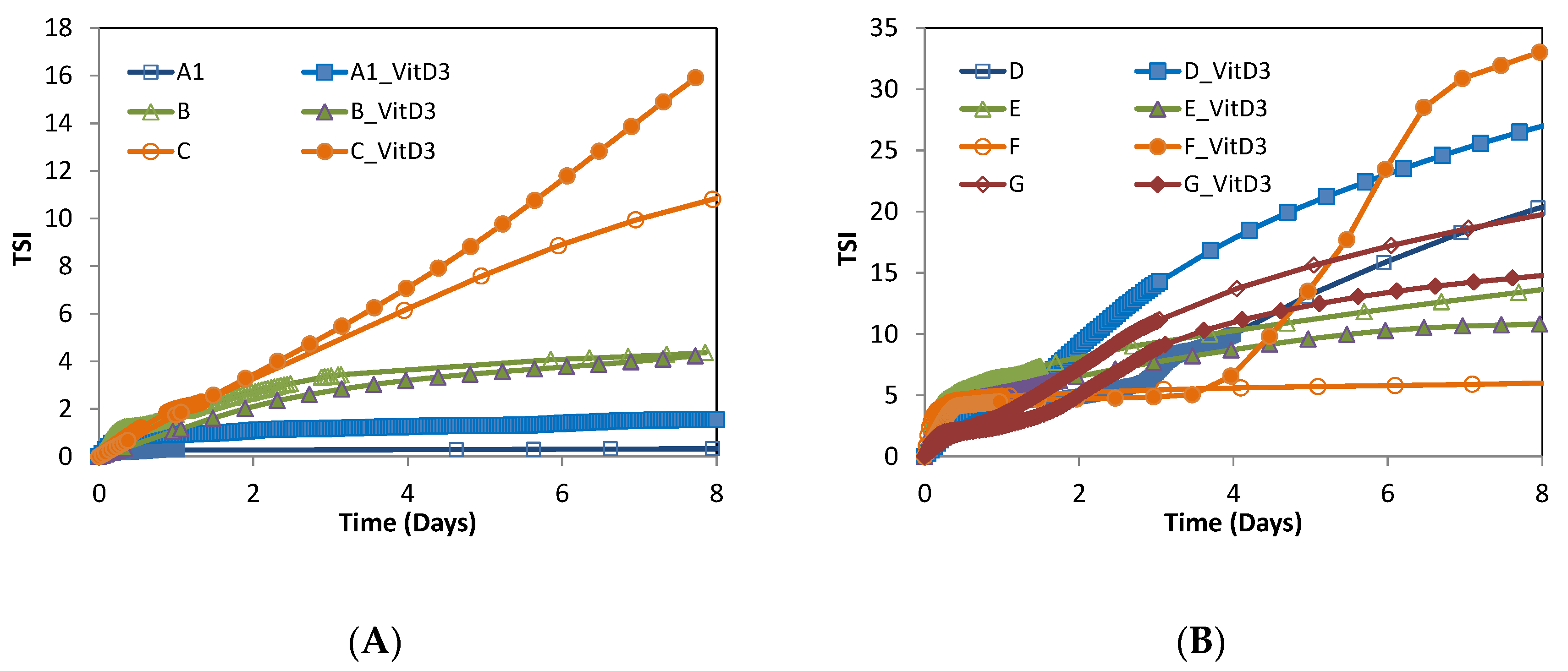
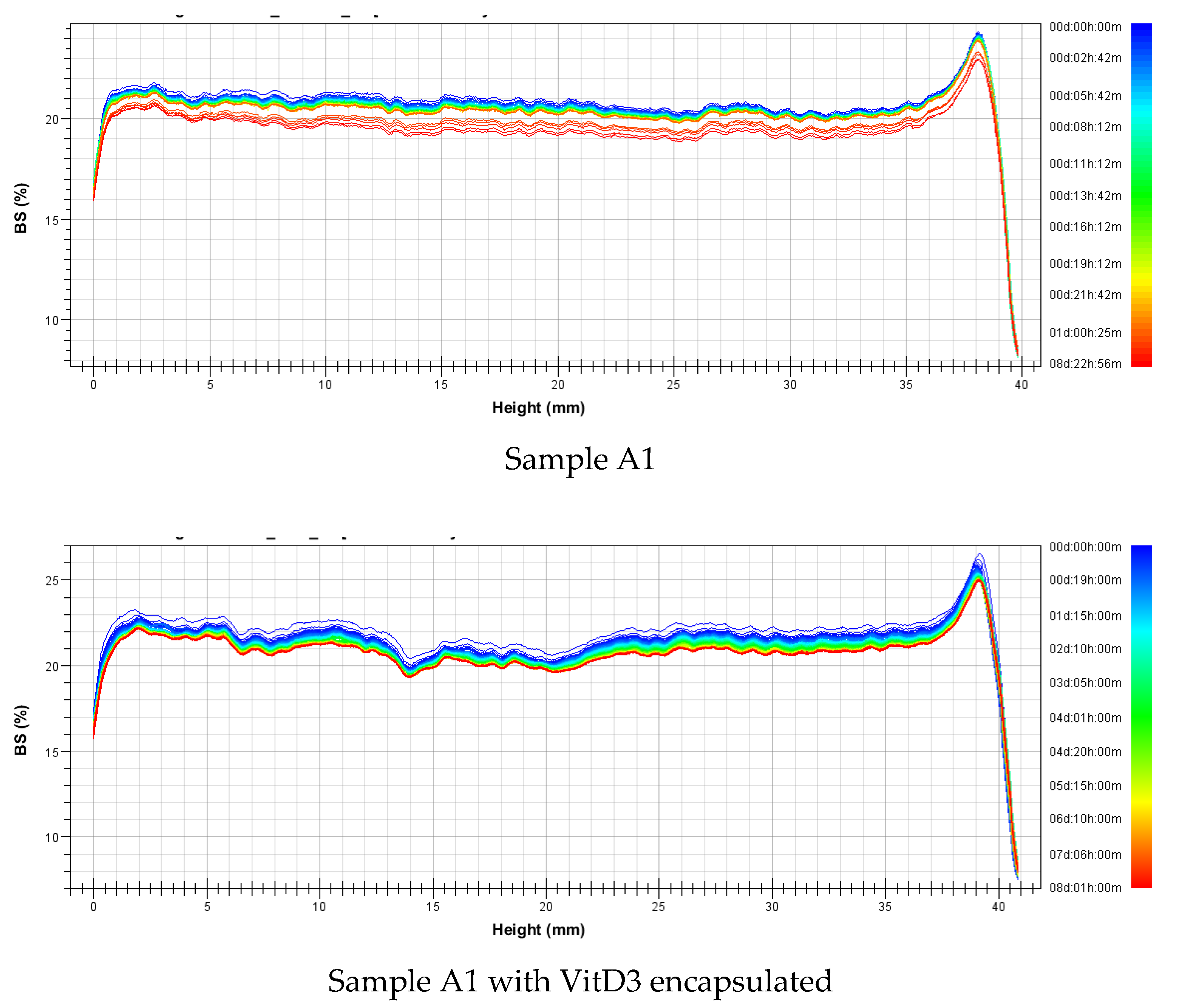
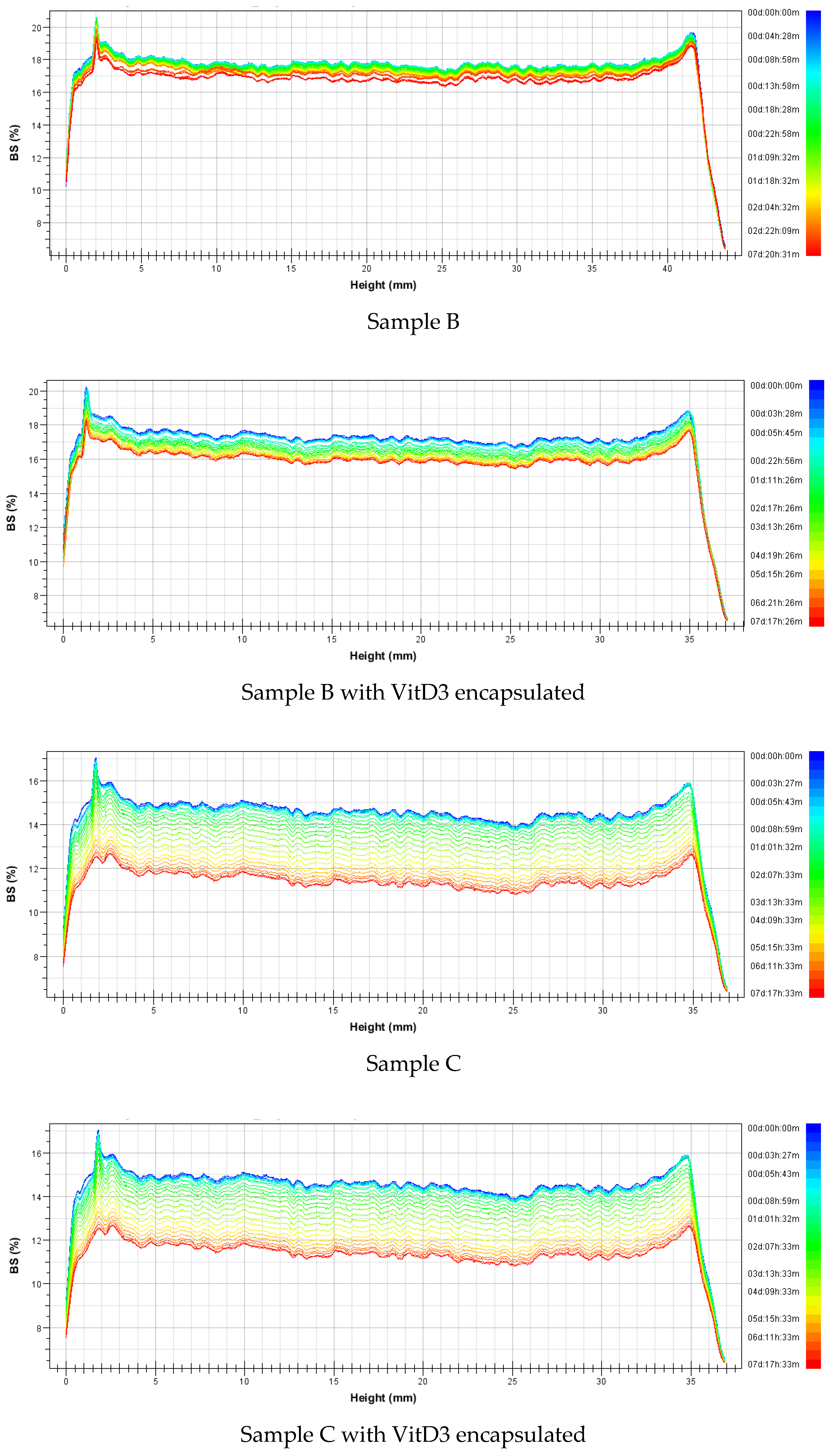
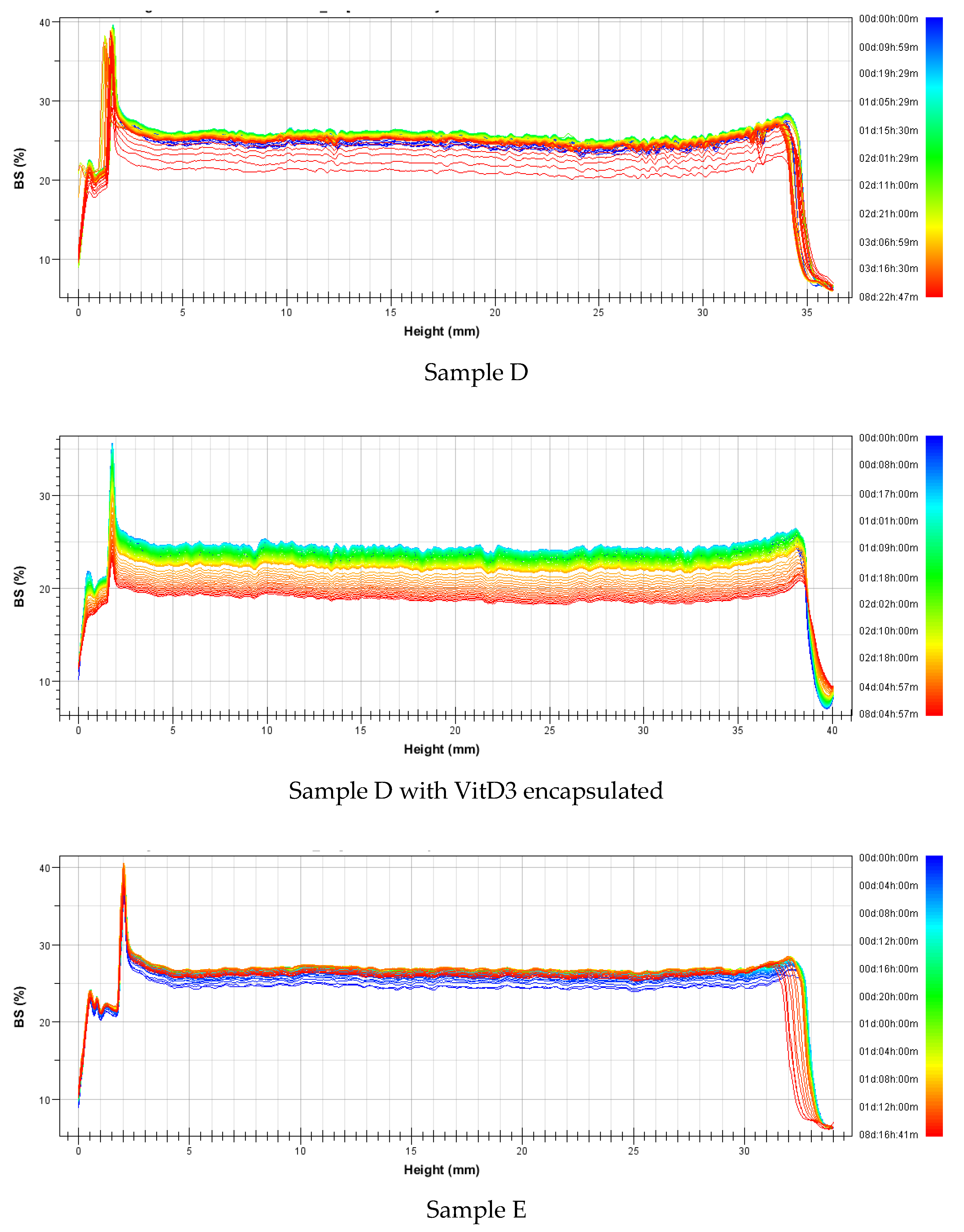
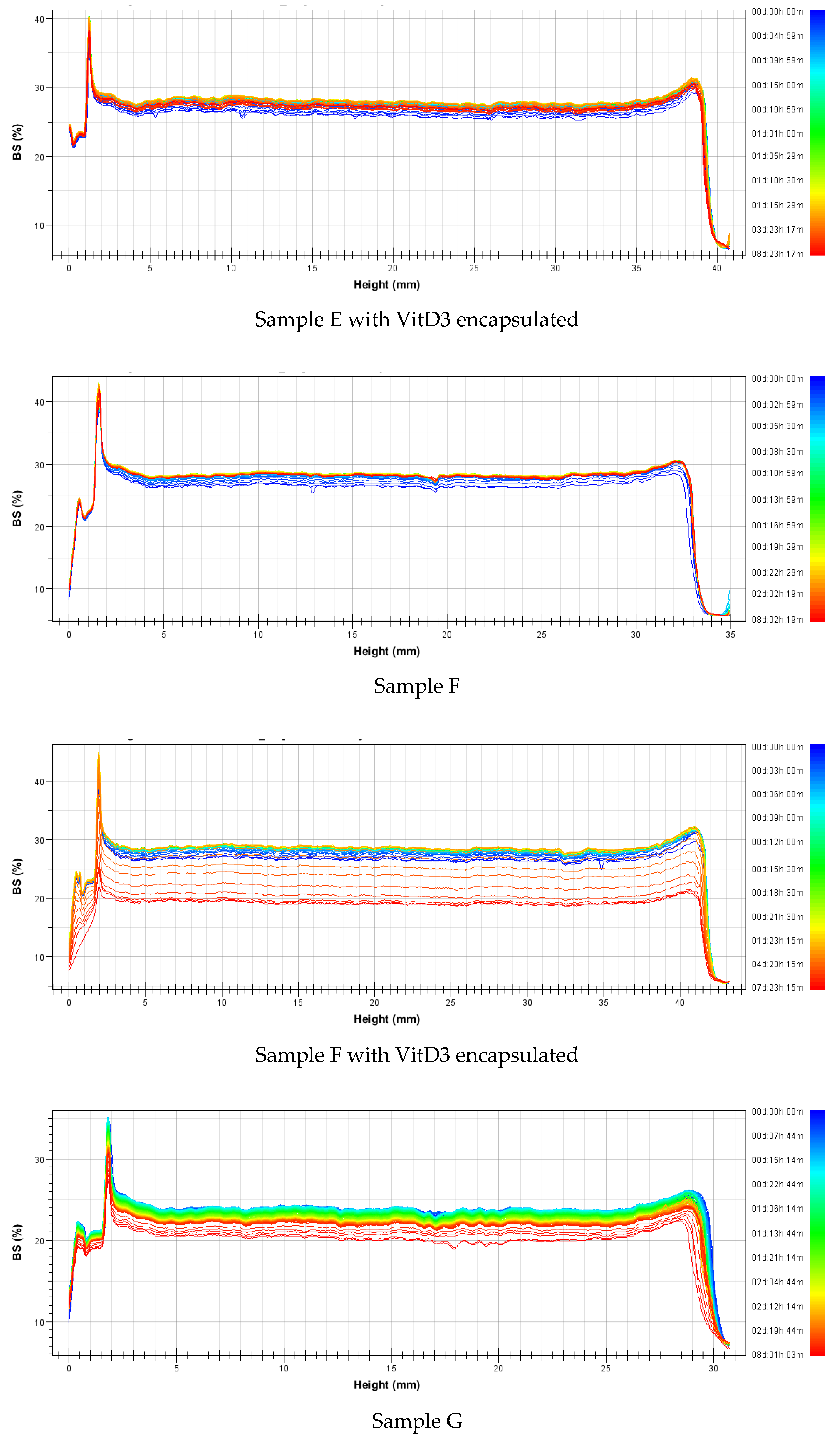
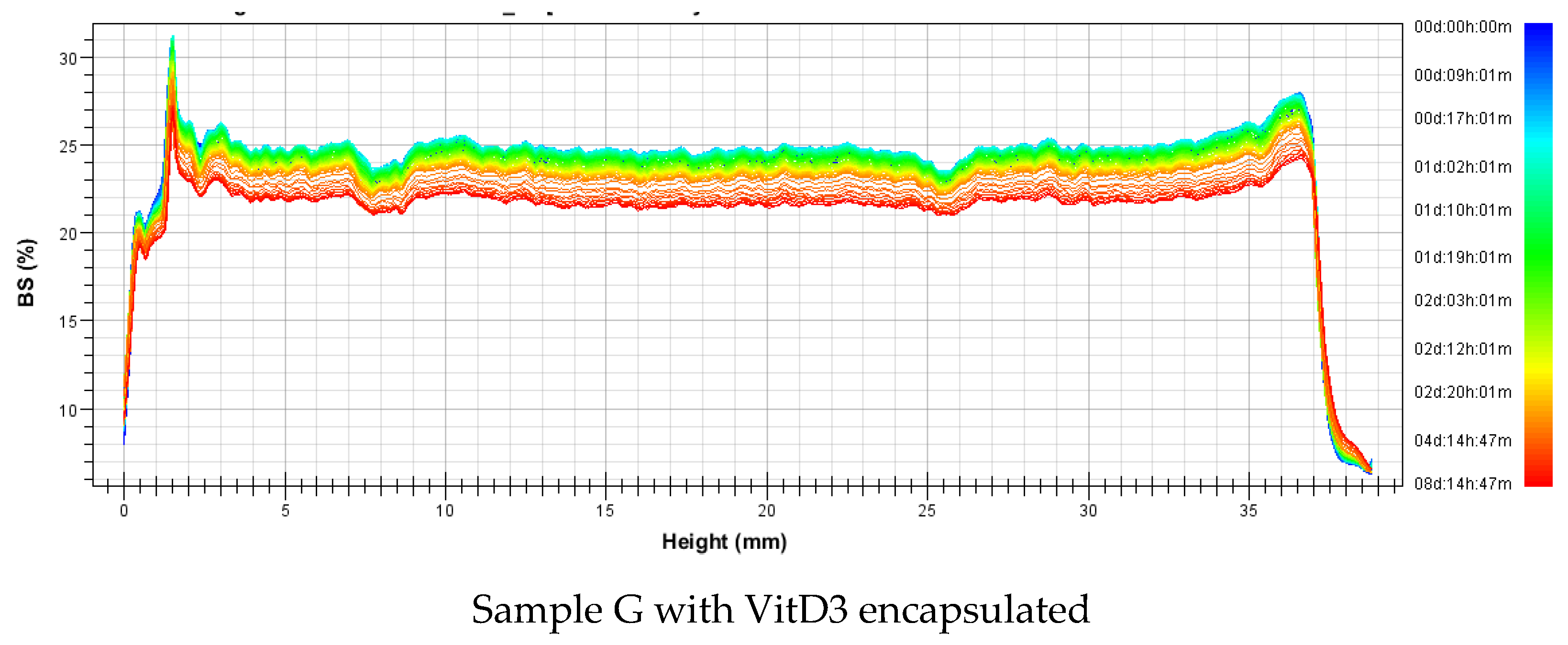
| Nanovesicles | Sample | Mass (%w/w) | Formulation | Average Size (nm) | Polidispersity Index | ||||
|---|---|---|---|---|---|---|---|---|---|
| Span®60 | Tween®20 | Tween®80 | Phospholipid | Cholesterol | |||||
| Niosome | A1 | 2.2 | 0.50 | - | - | - | 0.50 | 209 ± 4 | 0.18 ± 0.05 |
| A2 | 4.3 | 0.50 | - | - | - | 0.50 | 248 ± 5 | 0.17 ± 0.09 | |
| A3 | 6.3 | 0.50 | - | - | - | 0.50 | 253 ± 4 | 0.227 ± 0.09 | |
| A4 | 8.3 | 0.50 | - | - | - | 0.50 | 256 ± 5 | 0.2 ± 0.1 | |
| B | 2.2 | 0.50 | 0.25 | - | - | 0.25 | 186 ± 6 | 0.15 ± 0.01 | |
| C | 2.2 | 0.50 | - | 0.25 | - | 0.25 | 214 ± 5 | 0.161 ± 0.002 | |
| Transfersomes | D | 2.2 | 0.13 | 0.29 | - | 0.29 | 0.29 | 123 ± 6 | 0.202 ± 0.008 |
| E | 2.2 | 0.20 | 0.20 | - | 0.40 | 0.20 | 113 ± 7 | 0.21 ± 0.02 | |
| F | 2.2 | 0.25 | 0.25 | - | 0.50 | - | 88 ± 5 | 0.266 ± 0.001 | |
| G | 2.2 | 0.30 | 0.20 | - | 0.30 | 0.20 | 127 ± 5 | 0.199 ± 0.003 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estupiñan, O.R.; Garcia-Manrique, P.; Blanco-Lopez, M.d.C.; Matos, M.; Gutiérrez, G. Vitamin D3 Loaded Niosomes and Transfersomes Produced by Ethanol Injection Method: Identification of the Critical Preparation Step for Size Control. Foods 2020, 9, 1367. https://doi.org/10.3390/foods9101367
Estupiñan OR, Garcia-Manrique P, Blanco-Lopez MdC, Matos M, Gutiérrez G. Vitamin D3 Loaded Niosomes and Transfersomes Produced by Ethanol Injection Method: Identification of the Critical Preparation Step for Size Control. Foods. 2020; 9(10):1367. https://doi.org/10.3390/foods9101367
Chicago/Turabian StyleEstupiñan, Oscar R., Pablo Garcia-Manrique, Maria del Carmen Blanco-Lopez, Maria Matos, and Gemma Gutiérrez. 2020. "Vitamin D3 Loaded Niosomes and Transfersomes Produced by Ethanol Injection Method: Identification of the Critical Preparation Step for Size Control" Foods 9, no. 10: 1367. https://doi.org/10.3390/foods9101367
APA StyleEstupiñan, O. R., Garcia-Manrique, P., Blanco-Lopez, M. d. C., Matos, M., & Gutiérrez, G. (2020). Vitamin D3 Loaded Niosomes and Transfersomes Produced by Ethanol Injection Method: Identification of the Critical Preparation Step for Size Control. Foods, 9(10), 1367. https://doi.org/10.3390/foods9101367








