Grape (Vitis vinifera L.) Seed Oil: A Functional Food from the Winemaking Industry
Abstract
:1. Introduction: An Overview of Wine to the Past from the Present
2. Grapes, Seeds, and Seed Extracts: Natural Sources of Nutrients and Bioactive Compounds from Winemaking Industry
3. Grape Seed Oil: A Novel Functional Food
3.1. Grape Seed Oil: Extraction Processes
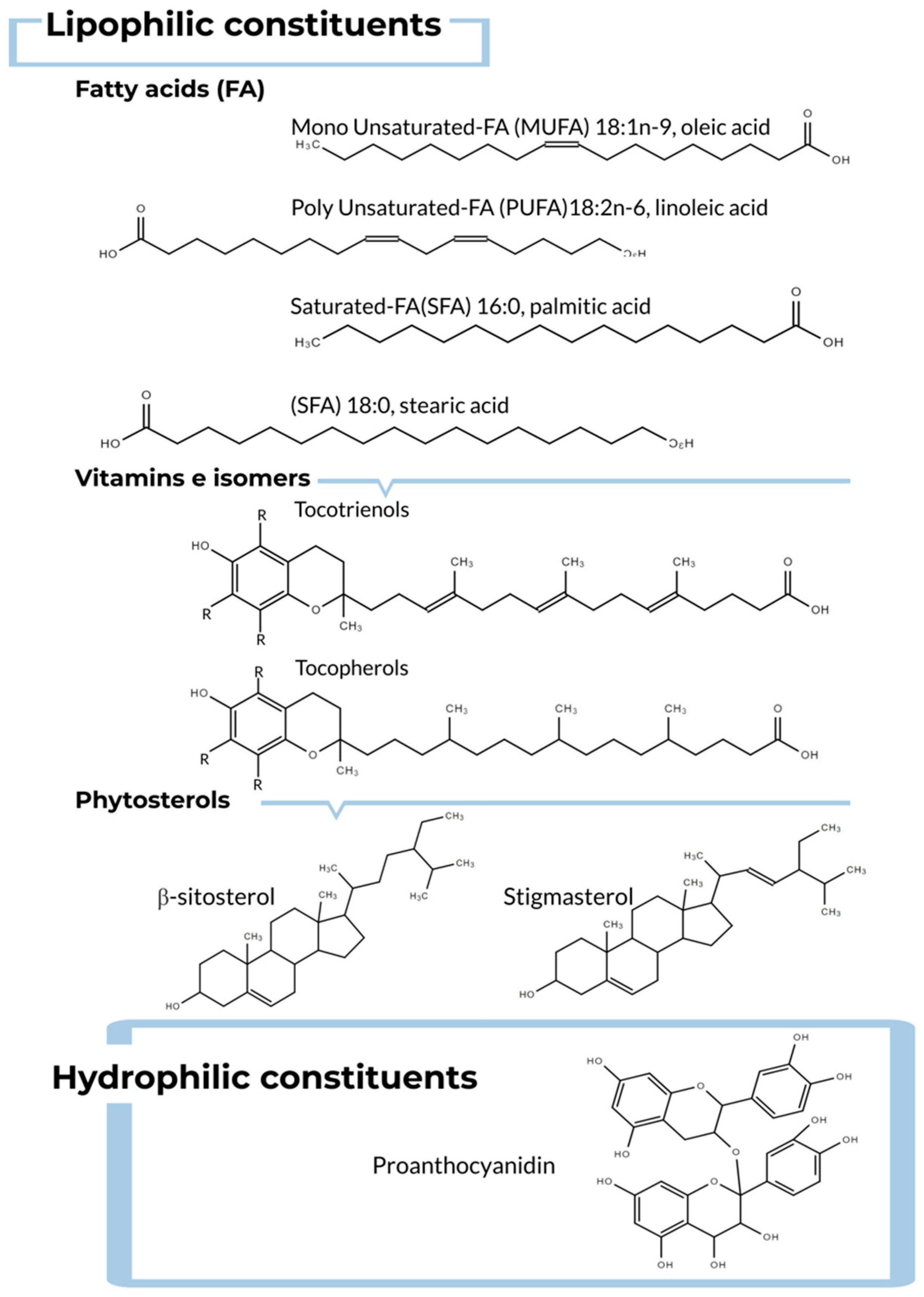
3.2. Grape Seed Oil: Chemical Composition
3.3. Grape Seed Oil: Nutraceutical Activities
3.3.1. Antioxidant Activity
3.3.2. Anti-Inflammatory Activity
3.3.3. Antimicrobial Activity
3.3.4. Antitumoral Activities
3.3.5. Protective Activities
3.3.6. Dietary Activities
3.4. Grape Seed Oil: Other Applications of Interest
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Haseeb, S.; Alexander, B.; Santi, R.L.; Liprandi, A.S.; Baranchuk, A. What’s in wine? A clinician’s perspective. Trends Cardiovasc. Med. 2019, 29, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Sotiropoulou, E.I.; Varelas, V.; Liouni, M.; Nerantzis, E.T. Grape Seed Oil: From a Winery Waste to a Value Added Cosmetic Product-a Review. 2015. Available online: https://www.researchgate.net/publication/312578959 (accessed on 20 April 2020).
- Baranchuk, A.; Alexander, B.; Haseeb, S. Drinking red wine is good for you—Or maybe not. The Washington Post. 2017. Available online: https://www.washingtonpost.com/national/health-science/drinking-red-wine-is-good-for-you--or-maybe-not/2017/12/01/49f55e7a-cbd3-11e7-aa96-54417592cf72_story.html (accessed on 1 May 2018).
- Shield, K.D.; Gmel, G.; Gmel, G.; Mäkelä, P.; Probst, C.; Room, R.; Rehm, J. Life-time risk of mortality due to different levels of alcohol consumption in seven European countries: Implications for low-risk drinking guidelines. Addiction 2017, 112, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- OIV: Organisation Internationale de la vigne et du vin. Global Economic Vitiviniculture Data. 2017. Available online: http://www.oiv.int/public/medias/5681/en-communiqu-depresse-octobre-017.pdf (accessed on 10 January 2019).
- Stockley, C.; Teissedre, P.L.; Boban, M.; Lorenzo, C.D.; Restani, P. Bioavailability of wine-derived phenolic compounds in humans: A review. Food Funct. 2012, 3, 995–1007. [Google Scholar] [CrossRef] [PubMed]
- Haseeb, S.; Alexander, B.; Baranchuk, A. Wine and cardiovascular health: A comprehensive review. Circulation 2017, 136, 1434–1448. [Google Scholar] [CrossRef]
- Champ, C.E.; Kundu-Champ, A. Maximizing Polyphenol Content to Uncork the Relationship Between Wine and Cancer. Front. Nutr. 2019, 6, 44. [Google Scholar] [CrossRef] [Green Version]
- Gülçin, İ. Antioxidant properties of resveratrol: A structure–activity insight. Inn. Food Sci. Emerg. Technol. 2010, 11, 210–218. [Google Scholar] [CrossRef]
- Malaguarnera, L. Influence of resveratrol on the immune response. Nutrients 2019, 11, 946. [Google Scholar] [CrossRef] [Green Version]
- Tabrizi, R.; Tamtaji, O.R.; Lankarani, K.B.; Mirhosseini, N.; Akbari, M.; Dadgostar, E.; Peymanic, P.; Asemi, Z. The effects of resveratrol supplementation on biomarkers of inflammation and oxidative stress among patients with metabolic syndrome and related disorders: A systematic review and meta-analysis of randomized controlled trials. Food Funct. 2018, 9, 6116–6128. [Google Scholar] [CrossRef]
- Hoseini, A.; Namazi, G.; Farrokhian, A.; Reiner, Ž.; Aghadavod, E.; Bahmani, F.; Asemi, Z. The effects of resveratrol on metabolic status in patients with type 2 diabetes mellitus and coronary heart disease. Food Funct. 2019, 10, 6042–6051. [Google Scholar] [CrossRef]
- Breuss, J.M.; Atanasov, A.G.; Uhrin, P. Resveratrol and its effects on the vascular system. Int. J. Mol. Sci. 2019, 20, 1523. [Google Scholar] [CrossRef] [Green Version]
- Springer, M.; Moco, S. Resveratrol and Its Human Metabolites—Effects on Metabolic Health and Obesity. Nutrients 2019, 11, 143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, C.K.; Luo, J.Y.; Lau, C.W.; Chen, Z.Y.; Tian, X.Y.; Huang, Y. Pharmacological basis and new insights of resveratrol action in the cardiovascular system. Br. J. Pharmacol. 2020, 177, 1258–1277. [Google Scholar] [CrossRef]
- Huang, X.T.; Li, X.; Xie, M.L.; Huang, Z.; Huang, Y.X.; Wu, G.X.; Peng, Z.R.; Sun, Y.N.; Ming, Q.L.; Liu, Y.X.; et al. Resveratrol: Review on its discovery, anti-leukemia effects and pharmacokinetics. Chem. Biol. Interact. 2019, 306, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Vervandier-Fasseur, D.; Latruffe, N. The Potential Use of Resveratrol for Cancer Prevention. Molecules 2019, 24, 4506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galiniak, S.; Aebisher, D.; Bartusik-Aebisher, D. Health benefits of resveratrol administration. Acta Biochim. Pol. 2019, 66, 13–21. [Google Scholar] [CrossRef] [Green Version]
- Meng, X.; Zhou, J.; Zhao, C.N.; Gan, R.Y.; Li, H.B. Health Benefits and Molecular Mechanisms of Resveratrol: A Narrative Review. Foods 2020, 9, 340. [Google Scholar] [CrossRef] [Green Version]
- Shaito, A.; Posadino, A.M.; Younes, N.; Hasan, H.; Halabi, S.; Alhababi, D.; Al-Mohannadi, A.; Abdel-Rahman, W.M.; Eid, A.H.; Nasrallah, G.K.; et al. Potential adverse effects of resveratrol: A literature review. Int. J. Mol. Sci. 2020, 21, 2084. [Google Scholar] [CrossRef] [Green Version]
- Vestergaard, M.; Ingmer, H. Antibacterial and antifungal properties of resveratrol. Int. J. Antimicrob. Agents 2019, 53, 716–723. [Google Scholar] [CrossRef]
- Lin, S.C.; Ho, C.T.; Chuo, W.H.; Li, S.; Wang, T.T.; Lin, C.C. Effective inhibition of MERS-CoV infection by resveratrol. BMC Infect. Dis. 2017, 17, 144. [Google Scholar] [CrossRef] [Green Version]
- Annunziata, G.; Maisto, M.; Schisano, C.; Ciampaglia, R.; Narciso, V.; Tenore, G.C.; Novellino, E. Resveratrol as a novel anti-herpes simplex virus nutraceutical agent: An overview. Viruses 2018, 10, 473. [Google Scholar] [CrossRef] [Green Version]
- Mohd, A.; Zainal, N.; Tan, K.K.; AbuBakar, S. Resveratrol affects Zika virus replication in vitro. Sci. Rep. 2019, 9, 14336. [Google Scholar] [CrossRef] [PubMed]
- Paradelo, R.; Moldes, A.B.; Barral, M.T. Utilization of a factorial design to study the composting of hydrolyzed grape marc and vinification lees. J. Agric. Food Chem. 2010, 58, 3085–3092. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Ahmedna, M. Functional components of grape pomace: Their composition, biological properties and potential applications. Int. J. Food Sci. Technol. 2013, 48, 221–237. [Google Scholar] [CrossRef]
- Pérez-Serradilla, J.A.; Castro, M.D.L.D. Microwave-assisted extraction of phenolic compounds from wine lees and spray-drying of the extract. Food Chem. 2011, 124, 1652–1659. [Google Scholar] [CrossRef]
- Jara-Palacios, M.J. Wine Lees as a Source of Antioxidant Compounds. Antioxidants 2019, 8, 45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nassiri-Asl, M.; Hosseinzadeh, H. Review of the Pharmacological Effects of Vitis vinifera (Grape) and its Bioactive Constituents: An Update. Phytother. Res. 2016, 30, 1392–1403. [Google Scholar] [CrossRef] [PubMed]
- Pasini, F.; Chinnici, F.; Caboni, M.F.; Verardo, V. Recovery of Oligomeric Proanthocyanidins and Other Phenolic Compounds with Established Bioactivity from Grape Seed By-Products. Molecules 2019, 24, 677. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, L.; Zhang, Y.; Lu, J. Phenolic contents and compositions in skins of red wine grape cultivars among various genetic backgrounds and originations. Int. J. Mol. Sci. 2012, 13, 3492–3510. [Google Scholar] [CrossRef] [Green Version]
- Mattos, G.N.; Tonon, R.V.; Furtado, A.A.; Cabral, L.M. Grape by-product extracts against microbial proliferation and lipid oxidation: A review. J. Sci. Food Agric. 2017, 97, 1055–1064. [Google Scholar] [CrossRef]
- Teixeira, A.; Eiras-Dias, J.; Castellarin, S.D.; Gerós, H. Berry phenolics of grapevine under challenging environments. Int. J. Mol. Sci. 2013, 14, 18711–18739. [Google Scholar] [CrossRef] [Green Version]
- Tang, G.Y.; Zhao, C.N.; Liu, Q.; Feng, X.L.; Xu, X.Y.; Cao, S.Y.; Meng, X.; Li, S.; Gan, R.Y.; Li, H.B. Potential of Grape Wastes as a Natural Source of Bioactive Compounds. Molecules 2018, 23, 2598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shinagawa, F.B.; De Santana, F.C.; Torres, L.R.O.; Mancini-Filho, J. Grape seed oil: A potential functional food? Food Sci. Technol. 2015, 35, 399–406. [Google Scholar] [CrossRef] [Green Version]
- Devesa-Rey, R.; Vecino, X.; Varela-Alende, J.L.; Barral, M.T.; Cruz, J.M.; Moldes, A.B. Valorization of winery waste vs. the costs of not recycling. Waste Manag. 2011, 31, 2327–2335. [Google Scholar] [CrossRef]
- Unterkofler, J.; Muhlack, R.A.; Jeffery, D.W. Processes and purposes of extraction of grape components during winemaking: Current state and perspectives. Appl. Microbiol. Biotechnol. 2020, 104, 4737–4755. [Google Scholar] [CrossRef] [PubMed]
- Ky, I.; Lorrain, B.; Kolbas, N.; Crozier, A.; Teissedre, P.L. Wine by-products: Phenolic characterization and antioxidant activity evaluation of grapes and grape pomaces from six different French grape varieties. Molecules 2014, 19, 482–506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rousserie, P.; Rabot, A.; Geny-Denis, L. From Flavanols Biosynthesis to Wine Tannins: What Place for Grape Seeds? J. Agric. Food Chem. 2019, 675, 1325–1343. [Google Scholar] [CrossRef] [PubMed]
- Guaita, M.; Bosso, A. Polyphenolic Characterization of Grape Skins and Seeds of Four Italian Red Cultivars at Harvest and after Fermentative Maceration. Foods 2019, 8, 395. [Google Scholar] [CrossRef] [Green Version]
- Bordiga, M.; Meudec, E.; Williams, P.; Montella, R.; Travaglia, F.; Arlorio, M.; Coisson, J.D.; Doco, T. The impact of distillation process on the chemical composition and potential prebiotic activity of different oligosaccharidic fractions extracted from grape seeds. Food Chem. 2019, 285, 423–430. [Google Scholar] [CrossRef]
- Antoniolli, A.; Fontana, A.R.; Piccoli, P.; Bottini, R. Characterization of polyphenols and evaluation of antioxidant capacity in grape pomace of the cv. Malbec. Food Chem. 2015, 178, 172–178. [Google Scholar] [CrossRef]
- Lucarini, M.; Durazzo, A.; Romani, A.; Campo, M.; Lombardi-Boccia, G.; Cecchini, F. Bio-based compounds from grape seeds: A biorefinery approach. Molecules 2018, 23, 1888. [Google Scholar] [CrossRef] [Green Version]
- Luo, L.; Bai, R.; Zhao, Y.; Li, J.; Wei, Z.; Wang, F.; Sun, B. Protective Effect of Grape Seed Procyanidins against H2O2-Induced Oxidative Stress in PC-12 Neuroblastoma Cells: Structure-Activity Relationships. J. Food Sci. 2018, 83, 2622–2628. [Google Scholar] [CrossRef]
- Mossalayi, M.D.; Rambert, J.; Renouf, E.; Micouleau, M.; M’erillon, J.M. Grape polyphenols and propolis mixture inhibits inflammatory mediator release from human leukocytes and reduces clinical scores in experimental arthritis. Phytomedicine 2014, 21, 290–297. [Google Scholar] [CrossRef] [PubMed]
- Kemperman, R.A.; Gross, G.; Mondot, S.; Possemiers, S.; Marzorati, M.; Wiele, T.V.; Vaughan, E.E. Impact of polyphenols from black tea and red wine/grape juice on a gut model microbiome. Food Res. Int. 2013, 53, 659–669. [Google Scholar] [CrossRef]
- Saito, M.; Hosoyama, H.; Ariga, T.; Kataoka, S.; Yamaji, N. Antiulcer activity of grape seed extract and procyanidins. J. Agric. Food Chem. 1998, 46, 1460–1464. [Google Scholar] [CrossRef]
- Sun, T.; Chen, Q.Y.; Wu, L.J.; Yao, X.M.; Sun, X.J. Antitumor and antimetastatic activities of grape skin polyphenols in a murine model of breast cancer. Food Chem. Toxicol. 2012, 50, 3462–3467. [Google Scholar] [CrossRef] [PubMed]
- Jara-Palacios, M.J.; Hernanz, D.; Cifuentes-Gomez, T.; Escudero-Gilete, M.L.; Heredia, F.J.; Spencer, J.P.E. Assessment of white grape pomace from winemaking as source of bioactive compounds and its antiproliferative activity. Food Chem. 2015, 183, 78–82. [Google Scholar] [CrossRef] [Green Version]
- Zhang, R.; Yu, Q.; Lu, W.; Shen, J.; Zhou, D.; Wang, Y.; Gao, S.; Wang, Z. Grape seed procyanidin B2 promotes the autophagy and apoptosis in colorectal cancer cells via regulating PI3K/Akt signaling pathway. Onco Targets Ther. 2019, 12, 4109–4118. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Pérez, C.; García-Villanova, B.; Guerra-Hernández, E.; Verardo, V. Grape Seeds Proanthocyanidins: An Overview of In Vivo Bioactivity in Animal Models. Nutrients 2019, 11, 2435. [Google Scholar] [CrossRef] [Green Version]
- Ma, Z.F.; Zhang, H. Phytochemical Constituents, Health Benefits, and Industrial Applications of Grape Seeds: A Mini-Review. Antioxidants 2017, 6, 71. [Google Scholar] [CrossRef] [Green Version]
- Butkhup, L.; Chowtivannakul, S.; Gaensakoo, R.; Prathepha, P.; Samappito, S. Study of the phenolic composition of Shiraz red grape cultivar (Vitis vinifera L.) cultivated in north-eastern Thailand and its antioxidant and antimicrobial activity. S. Afr. J. Enol. Vitic. 2010, 31, 89–98. [Google Scholar] [CrossRef] [Green Version]
- Adamez, J.D.; Samino, E.G.; Sánchez, E.V.; Gónzalez-Gómez, D. In vitro estimation of the antibacterial activity and antioxidant capacity of aqueous extracts from grape-seeds (Vitis vinifera L.). Food Control 2012, 24, 136–141. [Google Scholar] [CrossRef]
- Mirkarimi, M.; Amin-Marashi, S.; Bargrizan, M.; Abtahi, A.; Imani, F.A.A. The Antimicrobial Activity of Grape Seed Extract against Two Important Oral Pathogens. Zahedan J. Res. Med. Sci. 2013, 15, 43–46. [Google Scholar]
- Leone, A.; Longo, C.; Gerardi, C.; Trosko, J.E. Pro-Apoptotic Effect of Grape Seed Extract on MCF-7 Involves Transient Increase of Gap Junction Intercellular Communication and Cx43 Up-Regulation: A Mechanism of Chemoprevention. Int. J. Mol. Sci. 2019, 20, 3244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, A.; D’Silva, M.; Dholakia, K.; Levenson, A.S. In Vitro Anticancer Properties of Table Grape Powder Extract (GPE) in Prostate Cancer. Nutrients 2018, 10, 1804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ravindranathan, P.; Pasham, D.; Goel, A. Oligomeric proanthocyanidins (OPCs) from grape seed extract suppress the activity of ABC transporters in overcoming chemoresistance in colorectal cancer cells. Carcinogenesis 2019, 40, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.T.; Lu, Q.; Xue, B.; Neis, P.; Zamora, F.; Lundmark, L.; Qualls, C.; Massie, L. A Pilot Study of a Grape Seed Procyanidin Extract for Lung Cancer Chemoprevention. Cancer Prev. Res. 2019, 12, 557–566. [Google Scholar] [CrossRef]
- Targhi, R.G.; Banaei, A.; Saba, V. Radioprotective effect of grape seed extract against gamma irradiation in mouse bone marrow cells. J. Cancer Res. Ther. 2019, 15, 512–516. [Google Scholar]
- Eldaim, M.A.A.; Tousson, E.; Sayed, I.E.T.E.; El-Aleim, A.E.A.H.A.; Elsharkawy, H.N. Grape seeds proanthocyanidin extract ameliorates Ehrlich solid tumor induced renal tissue and DNA damage in mice. Biomed. Pharmacother. 2019, 115, 108908. [Google Scholar] [CrossRef]
- Du, Y.; Guo, H.; Lou, H. Grape seed polyphenols protect cardiac cells from apoptosis via induction of endogenous antioxidant enzymes. J. Agric. Food Chem. 2007, 55, 1695–1701. [Google Scholar] [CrossRef]
- Hao, J.P.; Shi, H.; Zhang, J.; Zhang, C.M.; Feng, Y.M.; Qie, L.Y.; Dong, M.; Ji, X. Role of GSPE in improving early cerebral vascular damage by inhibition of Profilin-1 expression in a ouabain-induced hypertension model. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 6999–7012. [Google Scholar]
- Vinson, J.A.; Mandarano, M.A.; Shuta, D.L.; Bagchi, M.; Bagchi, D. Beneficial effects of a novel IH636 grape seed proanthocyanidin extract and a niacin-bound chromium in a hamster atherosclerosis model. Mol. Cell Biochem. 2002, 240, 99–103. [Google Scholar] [CrossRef]
- Bijak, M.; Sut, A.; Kosiorek, A.; Saluk-Bijak, J.; Golanski, J. Dual Anticoagulant/Antiplatelet Activity of Polyphenolic Grape Seeds Extract. Nutrients 2019, 11, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tu, X.; Wang, M.; Liu, Y.; Zhao, W.; Ren, X.; Li, Y.; Liu, H.; Gu, Z.; Jia, H.; Liu, J.; et al. Pretreatment of Grape Seed Proanthocyanidin Extract Exerts Neuroprotective Effect in Murine Model of Neonatal Hypoxic-ischemic Brain Injury by Its Antiapoptotic Property. Cell Mol. Neurobiol. 2019, 39, 953–961. [Google Scholar] [CrossRef] [PubMed]
- Kadri, S.; El-Ayed, M.; Cosette, P.; Jouenne, T.; Elkhaoui, S.; Zekri, S.; Limam, F.; Aouani, E.; Mokni, M. Neuroprotective effect of grape seed extract on brain ischemia: A proteomic approach. Metab. Brain Dis. 2019, 34, 889–907. [Google Scholar] [CrossRef]
- Wang, Y.J.; Thomas, P.; Zhong, J.H.; Bi, F.F.; Kosaraju, S.; Pollard, A.; Zhou, X.F. Consumption of grape seed extract prevents amyloid-β deposition and attenuates inflammation in brain of an Alzheimer’s disease mouse. Neurotox Res. 2009, 15, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Youssef, S.B.; Brisson, G.; Doucet-Beaupré, H.; Castonguay, A.M.; Gora, C.; Amri, M.; Lévesque, M. Neuroprotective benefits of grape seed and skin extract in a mouse model of Parkinson’s disease. Nutr. Neurosci. 2019, 25, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Pons, Z.; Margalef, M.; Bravo, F.; Arola-Arnal, A.; Muguerza, B. Chronic administration of grape-seed polyphenols attenuates the development of hypertension and improves other cardiometabolic risk factors associated with the metabolic syndrome in cafeteria diet-fed rats. Br. J. Nutr. 2017, 117, 200–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rameshrad, M.; Razavi, B.M.; Imenshahidi, M.; Hosseinzadeh, H. Vitis vinifera (grape) seed extract and resveratrol alleviate bisphenol-A-induced metabolic syndrome: Biochemical and molecular evidences. Phytother. Res. 2019, 33, 832–844. [Google Scholar] [CrossRef]
- Griffin, L.E.; Witrick, K.A.; Klotz, C.; Dorenkott, M.R.; Goodrich, K.M.; Fundaro, G.; McMillan, R.P.; Hulver, M.W.; Ponder, M.A.; Neilson, A.P. Alterations to metabolically active bacteria in the mucosa of the small intestine predict anti-obesity and anti-diabetic activities of grape seed extract in mice. Food Funct. 2017, 8, 3510–3522. [Google Scholar] [CrossRef]
- Gurger, M.; Yilmaz, E.; Yilmaz, S.; Once, G.K.; Konuk, M.; Kaya, E.; Say, Y.; Artas, G.; Artas, H. Grape seed extract supplement increases bone callus formation and mechanical strength: An animal study. J. Orthop. Surg. Res. 2019, 14, 206. [Google Scholar] [CrossRef]
- Soleymani, S.; Iranpanah, A.; Najafi, F.; Belwal, T.; Ramola, S.; Abbasabadi, Z.; Momtaz, S.; Farzaei, M.H. Implications of grape extract and its nanoformulated bioactive agent resveratrol against skin disorders. Arch. Dermatol. Res. 2019, 311, 577–588. [Google Scholar] [CrossRef] [PubMed]
- Izadpanah, A.; Soorgi, S.; Geraminejad, N.; Hosseini, M. Effect of grape seed extract ointment on cesarean section wound healing: A double-blind, randomized, controlled clinical trial. Complement. Ther. Clin. 2019, 35, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Decean, H.P.; Brie, I.C.; Tatomir, C.B.; Perde-Schrepler, M.; Fischer-Fodor, E.; Virag, P. Targeting MAPK (p38, ERK, JNK) and inflammatory CK (GDF-15, GM-CSF) in UVB-Activated Human Skin Cells with Vitis vinifera Seed Extract. J. Environ. Pathol. Toxicol. Oncol. 2018, 37, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Brenes, A.; Viveros, A.; Goñi, I.; Centeno, C.; Saura-Calixto, F.; Arija, I. Effect of grape seed extract on growth performance, protein and polyphenol digestibilities, and antioxidant activity in chickens. Span. J. Agric. Res. 2010, 8, 326–335. [Google Scholar] [CrossRef] [Green Version]
- Martineau, A.S.; Leray, V.; Lepoudere, A.; Blanchard, G.; Bensalem, J.; Gaudout, D.; Ouduerram, K.; Nguyen, P. A mixed grape and blueberry extract is safe for dogs to consume. BMC Vet. Res. 2016, 12, 162. [Google Scholar] [CrossRef] [Green Version]
- Grases, F.; Prieto, R.M.; Fernández-Cabot, R.A.; Costa-Bauzá, A.; Sánchez, A.M.; Prodanov, M. Effect of consuming a grape seed supplement with abundant phenolic compounds on the oxidative status of healthy human volunteers. Nutr. J. 2015, 14, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García-Lomillo, J.; Gónzalez-San-José, M.L.; Del-Pino-García, R.; Rivero-Pérez, M.D.; Muniz-Rodríguez, P. Antioxidant and antimicrobial properties of wine byproducts and their potential uses in the food industry. J. Agric. Food Chem. 2014, 62, 12595–12602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Özvural, E.B.; Vural, H. Which is the best grape seed additive for frankfurters: Extract, oil or flour? J. Sci. Food Agric. 2014, 94, 792–797. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, L.; Casal, S.; Cruz, R.; Pereira, J.A.; Ramalhosa, E. Seed oils of ten traditional Portuguese grape varieties with interesting chemical and antioxidant properties. Food Res. Int. 2013, 50, 161–166. [Google Scholar] [CrossRef]
- Lachman, J.; Hejtmánková, A.; Táborský, J.; Kotíková, Z.; Pivec, V.; Střalková, R.; Vollmannová, A.; Bojňanská, T.; Dědina, M. Evaluation of oil content and fatty acid composition in the seed of grapevine varieties. LWT Food Sci. Technol. 2015, 63, 620–625. [Google Scholar] [CrossRef]
- Agostini, F.; Bertussi, R.A.; Agostini, G.; Atti dos Santos, A.C.; Rossato, M.; Vanderlinde, R. Supercritical Extraction from Vinification Residues: Fatty Acids, α-Tocopherol, and Phenolic Compounds in the Oil Seeds from Different Varieties of Grape. Sci. World J. 2012, 2012, 790486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baydar, N.G.; Akkurt, M. Oil content and oil quality properties of some grape seeds. Turk. J. Agric. For. 2001, 25, 163–168. [Google Scholar]
- Al Juhaimi, F.; Gecgel, Ü.; Gülcü, M.; Hamaur, M.; Özcan, M.M. Bioactive properties, fatty acid composition and mineral contents of grape seed and oils. S. Afr. J. Enol. Vitic. 2017, 38, 103–108. [Google Scholar] [CrossRef] [Green Version]
- Özcan, M.M.; Al Juhaimi, F.; Gülcü, M.; Ulsu, N.; Gecgel, Ü. Determination of bioactive compounds and mineral contents of seedless parts and seeds of grapes. S. Afr. J. Enol. Vitic. 2017, 38, 212–220. [Google Scholar] [CrossRef] [Green Version]
- Elagamey, A.A.; Abdel-Wahab, M.A.; Shimaa, M.M.E.; Abdel-Mogib, M. Comparative study of morphological characteristics and chemical constituents of seeds of some grape table varieties. J. Am. Sci. 2013, 9, 447–454. [Google Scholar]
- Tangolar, S.G.; Özogul, Y.; Tangolar, S.; Torun, A. Evaluation of fatty acid profiles and mineral content of grape seed oil of some grape genotypes. Int. J. Food Sci. Nutr. 2009, 60, 32–39. [Google Scholar] [CrossRef]
- Bada, J.C.; León-Camacho, M.; Copovi, P.; Alonso, L. Characterization of grape seed oil from wines with protected denomination of origin (PDO) from Spain. Grasas Aceites 2015, 66, e085. [Google Scholar]
- Sabir, A.; Unver, A.; Kara, Z. The fatty acid and tocopherol constituents of the seed oil extracted from 21 grape varieties (Vitis spp.). J. Sci. Food Agric. 2012, 92, 1982–1987. [Google Scholar] [CrossRef]
- Crews, C.; Hough, P.; Godward, J.; Brereton, P.; Lees, M.; Guiet, S.; Winkelmann, W. Quantitation of the main constituents of some authentic grape-seed oils of different origin. J. Agric. Food Chem. 2006, 54, 6261–6265. [Google Scholar] [CrossRef]
- Dimić, I.; Teslić, N.; Putnik, P.; Bursać Kovačević, D.; Zeković, Z.; Šojić, B.; Mrkonjić, Ž.; Čolović, D.; Montesano, D.; Pavlić, B. Innovative and Conventional Valorizations of Grape Seeds from Winery By-Products as Sustainable Source of Lipophilic Antioxidants. Antioxidants 2020, 9, 568. [Google Scholar] [CrossRef]
- Tobar, P.; Moure, A.; Soto, C.; Chamy, R.; Zúñiga, M.E. Winery solid residue revalorization into oil and antioxidant with nutraceutical properties by an enzyme assisted process. Water Sci. Technol. 2005, 51, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Karaman, S.; Karasu, S.; Tornuk, F.; Toker, O.S.; Geçgel, Ü.; Sagdic, O.; Ozcan, N.; Gül, O. Recovery Potential of Cold Press By-products Obtained from the Edible Oil Industry: Physicochemical, Bioactive, and Antimicrobial Properties. J. Agric. Food Chem. 2015, 63, 2305–2313. [Google Scholar] [CrossRef] [PubMed]
- Venkitasamy, C.; Teh, H.E.; Atungulu, G.G.; McHugh, T.H.; Pan, Z. Optimization of mechanical extraction conditions for producing grape seed oil. Trans. ASABE 2014, 57, 1699–1705. [Google Scholar]
- Teh, H.E.; Yokoyama, W.H.; German, J.B.; McHugh, T.H.; Pan, Z. Hypocholesterolemic Effects of Expeller-Pressed and Solvent-Extracted Fruit Seed Oils and Defatted Pomegranate Seed Meals. J. Agric. Food Chem. 2019, 22, 6150–6159. [Google Scholar] [CrossRef] [PubMed]
- Jayawardena, B.; Smith, R.M. Superheated water extraction of essential oils from Cinnamomum zeylanicum (L.). Phytochem. Anal. 2010, 21, 470–472. [Google Scholar] [CrossRef]
- Çam, M.; Yüksel, E.; Alaşalvar, H.; Basygit, B.; Sen, H.; Yilmaztekin, M.; Ahhmed, A.; Sagdic, O. Simultaneous extraction of phenolics and essential oil from peppermint by pressurized hot water extraction. J. Food Sci. Technol. 2019, 56, 200–207. [Google Scholar] [CrossRef]
- Bravi, M.; Spinoglio, F.; Verdone, N.; Adami, M.; Aliboni, A.; D’Andrea, A.; De Santis, A.; Ferri, D. Improving the extraction of α-tocopherol-enriched oil from grape seeds by supercritical CO2. Optimisation of the extraction conditions. J. Food Eng. 2007, 78, 488–493. [Google Scholar] [CrossRef]
- Duba, K.S.; Fiori, L. Supercritical CO2 extraction of grape seed oil: Effect of process parameters on the extraction kinetics. J Supercrit. Fluids 2015, 98, 33–43. [Google Scholar] [CrossRef]
- Villanueva-Bermejo, D.; Calvo, M.V.; Castro-Gómez, P.; Fornari, T.; Fontecha, J. Production of omega 3-rich oils from underutilized chia seeds. Comparison between supercritical fluid and pressurized liquid extraction methods. Food Res. Int. 2019, 115, 400–407. [Google Scholar] [CrossRef]
- Ballesteros-Vivas, D.; Álvarez-Rivera, G.; Ibánez, E.; Parada-Alfonso, F.; Cifuentes, A. Integrated strategy for the extraction and profiling of bioactive metabolites from Passiflora mollissima seeds combining pressurized-liquid extraction and gas/liquid chromatography–high resolution mass spectrometry. J. Chromatogr. A 2019, 1595, 144–157. [Google Scholar] [CrossRef]
- Da Porto, C.; Porretto, E.; Decorti, D. Comparison of ultrasound-assisted extraction with conventional extraction methods of oil and polyphenols from grape (Vitis vinifera L.) seeds. Ultrason. Sonochem 2013, 20, 1076–1080. [Google Scholar] [CrossRef] [PubMed]
- Carrera, C.; Ruiz-Rodríguez, A.; Palma, M.l.; Barroso, C.G. Ultrasound assisted extraction of phenolic compounds from grapes. Anal. Chim. Acta 2012, 732, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Böger, B.R.; Salviato, A.; Valezi, D.F.; Di Mauro, E.; Georgetti, S.R.; Kurozawa, L.E. Optimization of ultrasound-assisted extraction of grape-seed oil to enhance process yield and minimize free radical formation. J. Sci. Food Agric. 2018, 98, 5019–5026. [Google Scholar] [CrossRef] [PubMed]
- Castejón, N.; Luna, P.; Señoráns, F.J. Alternative oil extraction methods from Echium plantagineum L. seeds using advanced techniques and green solvents. Food Chem. 2018, 244, 75–82. [Google Scholar] [CrossRef]
- Krishnaswamy, K.; Orsat, V.; Gariépy, Y.; Thangavel, K. Optimization of microwave-assisted extraction of phenolic antioxidants from grape seeds (Vitis vinifera). Food Bioprocess Technol. 2012, 6, 441–455. [Google Scholar] [CrossRef]
- Garavaglia, J.; Markoski, M.M.; Oliveira, A.; Marcadenti, A. Grape Seed Oil Compounds: Biological and Chemical Actions for Health. Nutr. Metab. Insights 2016, 9, 59–64. [Google Scholar] [CrossRef] [Green Version]
- Lutterodt, H.; Slavin, M.; Whent, M.; Turner, E.; Yu, L. Fatty acid composition, oxidative stability, antioxidant and antiproliferative properties of selected cold-pressed grape seed oils and flours. Food Chem. 2011, 128, 391–399. [Google Scholar] [CrossRef]
- Navas, P.B. Chemical composition of the virgin oil obtained by mechanical pressing form several grape seed varieties (Vitis vinifera L.) with emphasis on minor constituents. Arch. Latinoam. Nutr. 2009, 59, 214–219. [Google Scholar]
- Kochhar, S.P.; Henry, C.J. Oxidative stability and shelf-life evaluation of selected culinary oils. Int. J. Food Sci. Nutr. 2009, 60, 289–296. [Google Scholar] [CrossRef]
- Kim, D.J.; Jeon, G.; Sung, J.; Oh, S.K.; Hong, H.C.; Lee, J. Effect of grape seed oil supplementation on plasma lipid profile in rats. Food Sci. Biotechnol. 2010, 19, 249–252. [Google Scholar] [CrossRef]
- Orsavova, J.; Misurcova, L.; Ambrozova, J.V.; Vicha, R.; Mlcek, J. Fatty acids composition of vegetable oils and its contribution to dietary energy intake and dependence of cardiovascular mortality on dietary intake of fatty acids. Int. J. Mol. Sci. 2015, 16, 12871–12890. [Google Scholar] [CrossRef]
- Mahanna, M.; Millán-Linares, M.C.; Grao-Cruces, E.; Claro, C.; Toscano, R.; Rodríguez-Martín, N.M.; Naranjo, M.C.; Montserrat-de la Paz, S. Resveratrol-enriched grape seed oil (Vitis vinifera L.) protects from white fat dysfunction in obese mice. J. Funct. Foods 2019, 62, 103546. [Google Scholar] [CrossRef]
- Montserrat de la Paz, S.; Marín-Aguilar, F.; García-Giménez, M.D.; Fernández-Arche, M.A. Hemp (Cannabis sativa) seed oil: Analytical and phytochemical characterization of the unsaponifiable fraction. J. Agric. Food Chem. 2014, 62, 1105–1110. [Google Scholar] [CrossRef] [PubMed]
- López, S.; Bermúdez, B.; Montserrat-de la Paz, S.; Jaramillo, S.; Varela, L.M.; Ortega-Gómez, A.; Abia, R.; Muriana, F.J.G. Membrane composition and dynamics: A target of bioactive virgin olive oil constituents. Biochim. Biophys. Acta 2014, 1838, 1638–1656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montserrat-de la Paz, S.; Fernández-Arche, M.A.; Ángel-Martín, M.; García-Giménez, M.D. Phytochemical characterization of potential nutraceutical ingredients from Evening Primrose oil (Oenothera biennis L.). Phytochem. Lett. 2014, 8, 158–162. [Google Scholar] [CrossRef]
- Górnaś, P.; Rudzińska, M.; Grygier, A.; Lācis, G. Diversity of oil yield, fatty acids, tocopherols, tocotrienols, and sterols in the seeds of 19 interspecific grapes crosses. J. Sci. Food Agric. 2019, 99, 2078–2087. [Google Scholar] [CrossRef]
- Assumpção, C.F.; Nunes, I.L.; Mendonça, T.A.; Bortolin, R.C.; Jablonski, A.; Flôres, S.H.; de Oliveira Rios, A. Bioactive Compounds and Stability of Organic and Conventional Vitis labrusca Grape Seed Oils. J. Am. Oil Chem. Soc. 2016, 93, 115–124. [Google Scholar] [CrossRef]
- Freitas, L.D.S.; Jacques, R.A.; Richter, M.F.; Silva, A.L.D.; Caramão, E.B. Pressurized liquid extraction of vitamin E from Brazilian grape seed oil. J. Chromatogr. A 2008, 1200, 80–83. [Google Scholar] [CrossRef]
- Madawala, S.R.P.; Kochhar, S.P.; Dutta, P.C. Lipid components and oxidative status of selected specialty oils. Grasas Aceites 2012, 63, 143–151. [Google Scholar] [CrossRef] [Green Version]
- Maier, T.; Schieber, A.; Kammerer, D.R.; Carle, R. Residues of grape (Vitis vinifera L.) seed oil production as a valuable source of phenolic antioxidants. Food Chem. 2009, 112, 551–559. [Google Scholar] [CrossRef]
- Rombaut, N.; Savoire, R.; Thomasset, B.; Castello, J.; Hecke, E.V.; Lanoisellé, J.L. Optimization of oil yield and oil total phenolic content during grapeseed cold screw pressing. Ind. Crops Prod. 2015, 63, 26–33. [Google Scholar] [CrossRef]
- Kaseb, F.; Biregani, A.N. Effects of Olive Oil and Grape Seed Oil on Lipid Profile and Blood Pressure in Patients with Hyperlipidemia: A Randomized Clinical Trial. Food Nutr. Sci. 2016, 7, 682. [Google Scholar] [CrossRef] [Green Version]
- Ebrahimi-Mameghani, M.; Irandoost, P.; Pourmoradian, S. The Effects of Grape Seed Oil on the Cardiovascular Risk Factors in Overweight and Obese Women: A Double-Blind Randomized Clinical Trial. Curr. Top. Nutraceutical Res. 2020, 18, 221–226. [Google Scholar]
- Khurana, S.; Venkataraman, K.; Hollingsworth, A.; Piche, M.; Tai, T.C. Polyphenols: Benefits to the Cardiovascular System in Health and in Aging. Nutrients 2013, 5, 3779–3827. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Lee, J. Antioxidant and antiproliferative properties of a tocotrienol-rich fraction from grape seeds. Food Chem. 2009, 11, 1386–1390. [Google Scholar] [CrossRef]
- Santangelo, C.; Varì, R.; Scazzocchio, B.; Benedetto, R.D.; Filesi, C.; Masella, R. Polyphenols, intracellular signalling and inflammation. Ann. Ist. Super. Sanita 2007, 43, 394–405. [Google Scholar]
- Zhao, L.; Yagiz, Y.; Xu, C.; Lu, J.; Chung, S.; Marshall, M.R. Muscadine grape seed oil as a novel source of tocotrienols to reduce adipogenesis and adipocyte inflammation. Food Funct. 2015, 6, 2293–2302. [Google Scholar] [CrossRef]
- Irandoost, P.; Ebrahimi-Mameghani, M.; Pirouzpanah, S. Does grape seed oil improve inflammation and insulin resistance in overweight or obese women? Int. J. Food Sci. Nutr. 2013, 64, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Lai, X.; Kang, X.; Zeng, L.; Li, J.; Yang, Y.; Liu, D. The protective effects and genetic pathways of thorn grape seeds oil against high glucose-induced apoptosis in pancreatic β-cells. BMC Complement. Altern. Med. 2014, 14, 10. [Google Scholar] [CrossRef] [Green Version]
- Shinagawa, F.B.; Santana, F.C.; Mancini-Filho, J. Effect of cold pressed grape seed oil on rats biochemical markers and inflammatory profile. Rev. Nutr. 2015, 28, 65–76. [Google Scholar] [CrossRef] [Green Version]
- Vilahur, G.; Ben-Aicha, S.; Diaz, E.; Badimon, L.; Padro, T. Phytosterols and Inflammation. Curr. Med. Chem. 2018, 25, 1. [Google Scholar] [CrossRef] [PubMed]
- Kolar, M.J.; Konduri, S.; Chang, T.; Wang, H.; McNerlin, C.; Ohlsson, L.; Härröd, M.; Siegel, D.; Saghatelian, A. Linoleic acid esters of hydroxy linoleic acids are anti-inflammatory lipids found in plants and mammals. J. Biol. Chem. 2019, 294, 10698–10707. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.F.M.; Moawed, F.S.M.; Mohamed, M.A. Protective mechanism of grape seed oil on carbon tetrachloride-induced brain damage in γ-irradiated rats. J. Photochem. Photobiol. B 2015, 153, 317–323. [Google Scholar] [CrossRef]
- Ismail, A.F.M.; Salem, A.A.M.; Eassawy, M.M.T. Hepatoprotective effect of grape seed oil against carbon tetrachloride induced oxidative stress in liver of γ-irradiated rat. J. Photochem. Photobiol. B 2016, 160, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Millán-Linares, M.C.; Bermúdez, B.; Martín, M.E.; Muñoz, E.; Abia, R.; Millán, F.; Muriana, F.J.G.; Montserrat-de la Paz, S. Unsaponifiable fraction isolated from grape (Vitis vinifera L.) seed oil attenuates oxidative and inflammatory responses in human primary monocytes. Food Funct. 2018, 9, 2517–2523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rotava, R.; Zanella, I.; Silva, L.P.D.; Manfron, M.P.; Ceron, C.S.; Alves, S.H.; Karkow, A.K.; Santos, J.P.A. Antibacterial, antioxidant and tanning activity of grape by product. Cienc. Rural 2009, 39, 941–944. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Zhang, Y.J.; Xu, X.R.; Chen, Y.M.; Li, H.B. Resources and biological activities of natural polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef]
- Huang, S.; Yang, N.; Liu, Y.; Gao, J.; Huang, T.; Hu, L.; Zhao, J.; Li, Y.; Li, C.; Zhang, X. Grape seed proanthocyanidins inhibit colon cancer-induced angiogenesis through suppressing the expression of VEGF and Ang1. Int. J. Mol. Med. 2012, 30, 1410–1416. [Google Scholar] [CrossRef] [Green Version]
- Cheah, K.Y.; Howarth, G.S.; Bindon, K.A.; Kennedy, J.A.; Bastian, S.E. Low molecular weight procyanidins from grape seeds enhance the impact of 5-Fluorouracil chemotherapy on Caco-2 human colon cancer cells. PLoS ONE 2014, 9, e98921. [Google Scholar] [CrossRef]
- Hutchinson, S.A.; Lianto, P.; Moore, J.B.; Hughes, T.A.; Thorne, J.L. Phytosterols Inhibit Side-Chain Oxysterol Mediated Activation of LXR in Breast Cancer Cells. Int. J. Mol. Sci. 2019, 20, 3241. [Google Scholar] [CrossRef] [Green Version]
- Sánchez-Crisóstomo, I.; Fernández-Martínez, E.; Cariño-Cortés, R.; Betanzos-Cabrera, G.; Bobadilla-Lugo, R.A. Phytosterols and Triterpenoids for Prevention and Treatment of Metabolic-related Liver Diseases and Hepatocellular Carcinoma. Curr. Pharm. Biotechnol. 2019, 20, 197–214. [Google Scholar] [CrossRef] [PubMed]
- Fakih, O.; Sanver, D.; Kane, D.; Thorne, J.L. Exploring the biophysical properties of phytosterols in the plasma membrane for novel cancer prevention strategies. Biochimie 2018, 153, 150–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lacatusu, I.; Badea, N.; Badea, G.; Oprea, O.; Mihaila, M.A.; Kaya, D.A.; Stan, R.; Meghea, A. Lipid nanocarriers based on natural oils with high activity against oxygen free radicals and tumor cell proliferation. Mater Sci. Eng. C 2015, 56, 88–94. [Google Scholar] [CrossRef]
- Asadi, F.; Shahriari, A.; Chahardah-Cheric, M. Effect of long-term optional ingestion of canola oil, grape seed oil, corn oil and yogurt butter on serum, muscle and liver cholesterol status in rats. Food Chem. Toxicol. 2010, 48, 2454–2457. [Google Scholar] [CrossRef] [PubMed]
- Scognamiglio, M.; Costa, D.; Sorriento, A.; Napoli, C. Current Drugs and Nutraceuticals for the Treatment of Patients with Dyslipidemias. Curr. Pharm. Des. 2019, 25, 85–95. [Google Scholar] [CrossRef]
- Berahmand, F.; Anoush, G.; Hosseini, M.J.; Anoush, M. Grape Seed Oil as a Natural Therapy in Male Rats with Alzheimer’s Diseases. Adv. Pharm. Bull. 2020, 10, 430–436. [Google Scholar] [CrossRef]
- Bialek, A.; Czerwonka, M.; Bialek, M.; Lepionka, T.; Kaszperuk, K.; Banaszkiewicz, T.; Tokarz, A. Influence of Pomegranate Seed Oil and Grape Seed Oil on Cholesterol Content and Fatty Acids Profile in Livers of Chickens. Acta Pol. Pharm. 2017, 74, 624–632. [Google Scholar]
- Wall-Medrano, A.; de la Rosa, L.A.; Vázquez-Flores, A.A.; Mercado-Mercado, G.; González-Arellanes, R.; López-Díaz, J.A.; González-Córdova, A.F.; González-Aguilar, G.A.; Vallejo-Cordoba, B.; Molina-Corral, F.J. Lipidomic and Antioxidant Response to Grape Seed, Corn and Coconut Oils in Healthy Wistar Rats. Nutrients 2017, 9, 82. [Google Scholar] [CrossRef] [Green Version]
- Sharifi, M.; Bashtani, M.; Naserian, A.A.; Farhangfar, H.; Emami, A. The effect of grapeseed oil on performance, rumen fermentation, antioxidant status and subcutaneous adipose fatty acid profile in lambs. J. Anim. Physiol. Anim. Nutr. 2018, 102, 157–165. [Google Scholar] [CrossRef] [Green Version]
- Fernández-Iglesias, A.; Quesada, H.; Díaz, S.; Pajuelo, D.; Bladé, C.; Arola, L.; Salvadó, M.J.; Mulero, M. Combination of grape seed proanthocyanidin extract and docosahexaenoic acid-rich oil increases the hepatic detoxification by GST mediated GSH conjugation in a lipidic postprandial state. Food Chem. 2014, 165, 14–20. [Google Scholar] [CrossRef]
- Calder, P.C. Fatty acids and inflammation: The cutting edge between food and pharma. Eur. J. Pharmacol. 2011, 668, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Raphael, W.; Sordillo, L.M. Dietary polyunsaturated fatty acids and inflammation: The role of phospholipid biosynthesis. Int. J. Mol. Sci. 2013, 14, 21167–21188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matthäus, B. Virgin grape seed oil: Is it really a nutritional highlight? Eur. J. Lipid Sci. Technol. 2008, 110, 645–650. [Google Scholar] [CrossRef]
- Hurtado de Catalfo, G.E.; Alaniz, M.J.D.; Marra, C.A. Influence of commercial dietary oils on lipid composition and testosterone production in interstitial cells isolated from rat testis. Lipids 2009, 44, 345–357. [Google Scholar] [CrossRef]
- Hurtado de Catalfo, G.E.; Alaniz, M.J.D.; Marra, C.A. Dietary lipids modify redox homeostasis and steroidogenic status in rat testis. Nutrition 2006, 24, 717–726. [Google Scholar] [CrossRef]
- Kim, T.K.; Yong, H.I.; Jung, S.; Kim, Y.B.; Choi, Y.S. Effects of replacing pork fat with grape seed oil and gelatine/alginate for meat emulsions. Meat Sci. 2020, 163, 108079. [Google Scholar] [CrossRef]
- Choi, Y.S.; Choi, J.H.; Han, D.J.; Kim, H.Y.; Lee, M.A.; Kim, H.W.; Lee, J.W.; Chung, H.J.; Kim, C.J. Optimization of replacing pork back fat with grape seed oil and rice bran fiber for reduced-fat meat emulsion systems. Meat Sci. 2010, 84, 212–218. [Google Scholar] [CrossRef]
- Lee, H.J.; Jung, E.H.; Lee, S.H.; Kim, J.H.; Lee, J.J.; Choi, Y.I. Effect of Replacing Pork Fat with Vegetable Oils on Quality Properties of Emulsion-type Pork Sausages. Korean J. Food Sci. Anim. Resour. 2015, 35, 130–136. [Google Scholar] [CrossRef] [Green Version]
- Gorna’s, P.; Rudzińska, M. Seeds recovered from industry by-products of nine fruit species with a high potential utility as a source of unconventional oil for biodiesel and cosmetic and pharmaceutical sectors. Ind. Crops Prod. 2016, 83, 329–338. [Google Scholar] [CrossRef]
- Katiyar, S.K. Grape seed proanthocyanidins and cancer prevention: Inhibition of oxidative stress and protection of immune system. Mol. Nutr. Food Res. 2008, 52, 871–876. [Google Scholar]
- Sharif, A.; Akhtar, N.; Khan, M.S.; Menaa, A.; Menaa, B.; Khan, B.A.; Menaa, F. Formulation and evaluation on human skin of a water-in-oil emulsion containing Muscat hamburg black grape seed extract. Int. J. Cosmet. Sci. 2015, 37, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Shivananda Nayak, B.; Dan Ramdath, D.; Marshall, J.R.; Isitor, G.; Xue, S.; Shi, J. Wound-healing Properties of the Oils of Vitis vinifera and Vaccinium macrocarpon. Phytother. Res. 2011, 25, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.H.; Lee, S.Y.; Jeong, D.W.; Choi, E.J.; Kim, Y.J.; Lee, J.G.; Yi, Y.H.; Cha, H.S. Effect of pumpkin seed oil on hair growth in men with androgenetic alopecia: A randomized, double-blind, placebo-controlled trial. Evid. Based Complement. Alternat. Med. 2014, 2014, 549721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernández, C.M.; Ramos, M.J.; Pérez, A.; Rodríguez, J.F. Production of biodiesel from winery waste: Extraction, refining and transesterification of grape seed oil. Bioresour. Technol. 2010, 101, 7019–7024. [Google Scholar] [CrossRef]
- Hariram, V.; Bose, A.; Seralathan, S. Dataset on optimized biodiesel production from seeds of Vitis vinifera using ANN, RSM and ANFIS. Data Brief 2019, 25, 104298. [Google Scholar] [CrossRef]

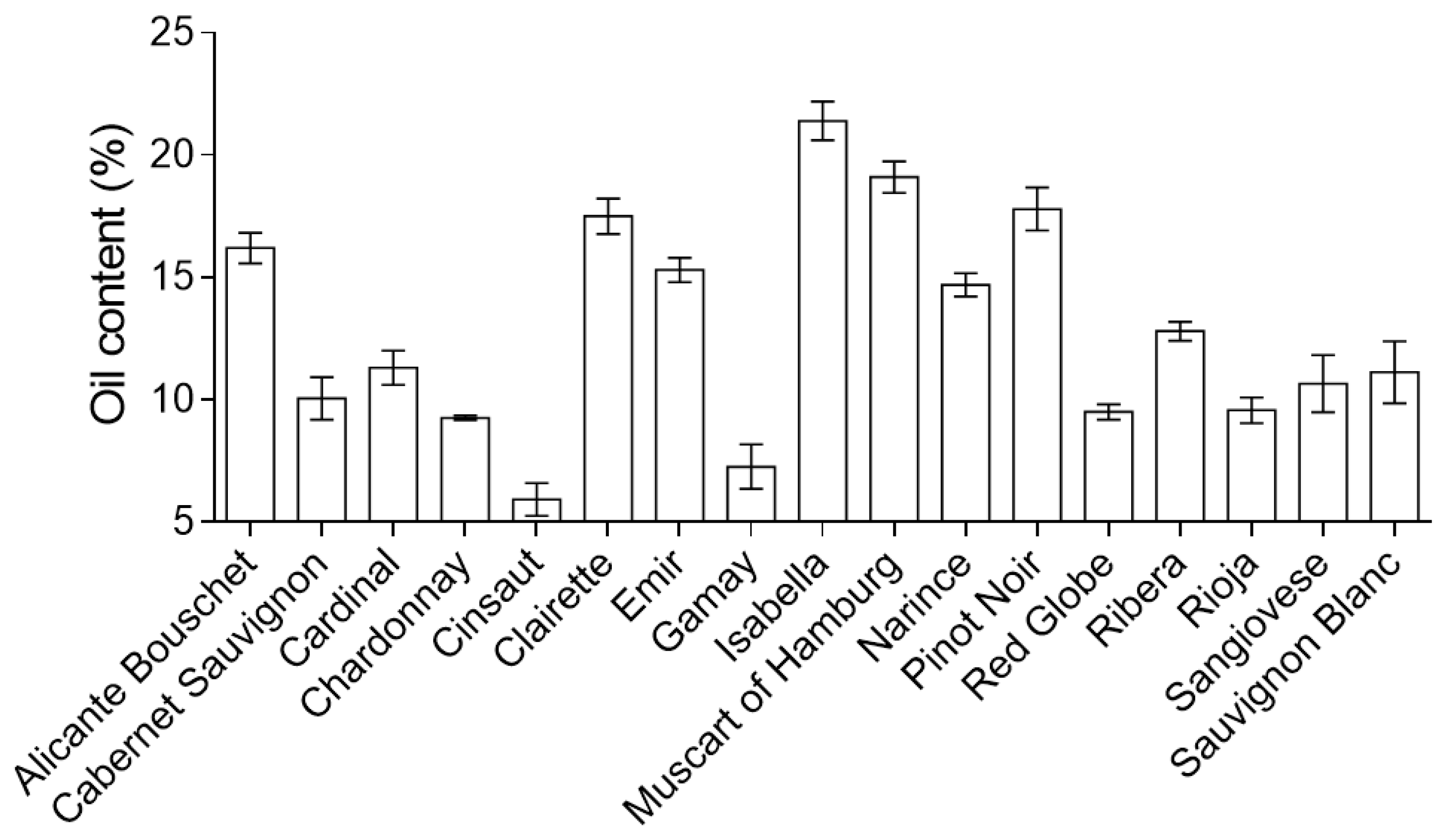
| Country | 2014 | 2015 | 2016 | 2017 | 2018 |
|---|---|---|---|---|---|
| Italy | 44,200 | 50,000 | 50,900 | 42,500 | 54,800 |
| France | 46,500 | 47,000 | 45,200 | 36,400 | 49,100 |
| Spain | 39,500 | 37,700 | 39,700 | 32,500 | 44,400 |
| United States | 23,100 | 21,700 | 23,700 | 23,300 | 23,900 |
| Argentina | 15,200 | 13,400 | 9400 | 11,800 | 14,500 |
| Chile | 9900 | 12,900 | 10,100 | 9500 | 12,900 |
| Australia | 11,900 | 11,900 | 13,100 | 13,700 | 12,900 |
| Germany | 9200 | 8800 | 9000 | 7500 | 9800 |
| South Africa | 11,500 | 11,200 | 10,500 | 10,800 | 9500 |
| China | 11,600 | 11,500 | 11,400 | 11,600 | 9300 |
| Russia | 4800 | 5600 | 5200 | 6300 | 6500 |
| Portugal | 6200 | 7000 | 6000 | 6700 | 6100 |
| Romania | 3700 | 3600 | 3300 | 4300 | 5100 |
| Hungary | 2400 | 2600 | 2500 | 3200 | 3600 |
| Brazil | 2600 | 2700 | 1300 | 3600 | 3100 |
| Rest of the world | 27,100 | 29,800 | 29,900 | 26,100 | 26,800 |
| World | 270,000 | 277,000 | 273,000 | 249,800 | 292,300 |
| FAs | SAF | GRP | SIL | HMP | SFL | WHG | PMS | SES | RB | ALM | RPS | PNT | OL | COC | EPO |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C6:0 | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | 0.52 | nd |
| C8:0 | nd | 0.01 | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | 7.6 | nd |
| C10:0 | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | 0.01 | nd | nd | 5.5 | nd |
| C12:0 | nd | 0.01 | 0.01 | nd | 0.02 | 0.07 | nd | nd | nd | 0.09 | nd | nd | nd | 47.7 | nd |
| C14:0 | 0.10 | 0.05 | 0.09 | 0.07 | 0.09 | nd | 0.17 | nd | 0.39 | 0.07 | nd | 0.04 | nd | 19.9 | nd |
| C15:0 | nd | 0.01 | 0.02 | nd | nd | 0.04 | nd | nd | nd | nd | 0.02 | nd | nd | nd | nd |
| C16:0 | 6.7 | 6.7 | 7.9 | 5.6 | 6.2 | 17.4 | 13.1 | 9.7 | 20.0 | 6.8 | 4.6 | 7.5 | 7.5–20 | nd | 6.3 |
| C17:0 | 0.04 | 0.06 | 0.06 | 0.05 | 0.02 | 0.03 | 0.13 | nd | nd | 0.05 | 0.04 | 0.07 | nd | nd | nd |
| C18:0 | 2.4 | 3.8 | 4.5 | 2.68 | 2.8 | 0.7 | 5.7 | 6.5 | 2.1 | 2.3 | 1.7 | 2.1 | 0.5–5 | 2.7 | 1.9 |
| C20:0 | nd | 0.16 | 2.6 | 2.5 | 0.21 | nd | 0.47 | 0.63 | nd | 0.09 | nd | 1.01 | 0.43 | nd | 0.3 |
| C22:0 | nd | nd | nd | 0.4 | nd | nd | nd | 0.14 | nd | nd | nd | nd | 0.15 | nd | 0.1 |
| C16:1 (n-7) | 0.08 | 0.2 | 0.05 | 0.31 | 0.12 | 0.21 | 0.12 | 0.11 | 0.19 | 0.53 | 0.21 | 0.07 | 0.3–3.5 | nd | nd |
| C17:1 (n-7) | nd | nd | 0.03 | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd |
| C18:1 cis (n-9) | 11.5 | 14.8 | 20.4 | 11.9 | 28.0 | 12.7 | 24.9 | 41.5 | 42.7 | 67.2 | 63.3 | 71.1 | 55–83 | 6.2 | 6.9 |
| C18:1 trans (n-9) | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | 0.14 | nd | nd | nd | nd |
| C20:1(n-9) | nd | 0.40 | 0.15 | 1.44 | 0.18 | 7.91 | 1.08 | 0.32 | 1.11 | 0.16 | 9.1 | nd | 0.30 | nd | 0.6 |
| C18:2 cis (n-6) | 79.0 | 74.2 | 63.3 | 55.1 | 62.2 | 59.7 | 54.2 | 40.9 | 33.1 | 22.8 | 19.6 | 18.2 | 3.5–21 | 1.6 | 73.9 |
| C18:3 (n-3) | 0.15 | 0.11 | 0.88 | 16.7 | 0.16 | 1.2 | 0.12 | 0.21 | 0.45 | nd | 1.2 | nd | <1 | nd | nd |
| C18:3 (n-6) | nd | nd | nd | 3.4 | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd | 9.2 |
| SFAs | 9.3 | 10.6 | 15.1 | 11.2 | 9.4 | 18.2 | 19.6 | 16.9 | 22.5 | 9.3 | 6.3 | 10.7 | 8–26 | 92.1 | nd |
| MUFAs | 11.6 | 14.9 | 20.7 | 13.3 | 28.3 | 20.9 | 26.1 | 42.0 | 44.0 | 67.9 | 72.8 | 71.1 | 53–87 | 6.2 | nd |
| PUFAs | 79.1 | 74.3 | 64.2 | 75.5 | 62.4 | 61.0 | 54.3 | 41.2 | 33.6 | 22.8 | 20.9 | 18.2 | 3–22 | 1.6 | nd |
| n-3 PUFAs | 0.2 | 0.2 | 0.9 | 0.4 | 0.2 | 1.2 | 0.1 | 0.2 | 0.5 | 0.0 | 1.2 | 0.0 | 1.6 | 0.0 | nd |
| n-6 PUFAs | 79.0 | 74.7 | 63.3 | 62.4 | 62.2 | 59.7 | 54.2 | 40.9 | 33.1 | 22.8 | 19.6 | 18.2 | 16.4 | 1.6 | nd |
| Study 1 | Study 2 | Study 3 | |||
|---|---|---|---|---|---|
| France | Italy | Spain | |||
| Tocopherols | |||||
| α-T | 47–56 | 86–244 | 18–229 | 14–160 | tr–75 |
| β-T | 38–48 | nd | nd–109 | nd–133 | nd–127 |
| γ-T | 17–29 | 3–28 | nd–61 | nd–119 | nd–168 |
| Δ-T | nd–3 | tr–1 | nd–47 | nd | nd–69 |
| Tocotrienols | |||||
| α-T3 | 216–278 | 69–319 | nd–163 | nd–352 | nd–60 |
| β-T3 | - | 4–18 | nd–67 | nd–22 | nd–125 |
| γ-T3 | 482–556 | 499–1575 | nd–500 | nd–785 | nd–399 |
| Δ-T3 | 13–17 | 6–18 | nd | nd | nd–82 |
| Phytosterols | Content (mg/kg Oil) |
|---|---|
| Cholesterol | nd–0.10 |
| Cholestanol | nd |
| Brassicasterol | 0.6–0.9 |
| 2,4 methylenecholesterol | nd–0.18 |
| Campesterol | 0.1–9.3 |
| Campestenol | - |
| Stigmasterol | 10.2–10.8 |
| α-7 campesterol | 0.16–0.27 |
| α-52,3 stigmastadienol | - |
| Clerosterol | 0.90–0.94 |
| β-sitosterol | 66.6–67.4 |
| Sitostanol | 3.92–4.70 |
| α-5 avenasterol | 1.98–2.09 |
| α-52,4 stigmastadienol | 0.41–0.47 |
| α-7 estigmastenol | 1.99–2.30 |
| α-7 avenasterol | 0.98–1.10 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martin, M.E.; Grao-Cruces, E.; Millan-Linares, M.C.; Montserrat-de la Paz, S. Grape (Vitis vinifera L.) Seed Oil: A Functional Food from the Winemaking Industry. Foods 2020, 9, 1360. https://doi.org/10.3390/foods9101360
Martin ME, Grao-Cruces E, Millan-Linares MC, Montserrat-de la Paz S. Grape (Vitis vinifera L.) Seed Oil: A Functional Food from the Winemaking Industry. Foods. 2020; 9(10):1360. https://doi.org/10.3390/foods9101360
Chicago/Turabian StyleMartin, Maria E, Elena Grao-Cruces, Maria C Millan-Linares, and Sergio Montserrat-de la Paz. 2020. "Grape (Vitis vinifera L.) Seed Oil: A Functional Food from the Winemaking Industry" Foods 9, no. 10: 1360. https://doi.org/10.3390/foods9101360
APA StyleMartin, M. E., Grao-Cruces, E., Millan-Linares, M. C., & Montserrat-de la Paz, S. (2020). Grape (Vitis vinifera L.) Seed Oil: A Functional Food from the Winemaking Industry. Foods, 9(10), 1360. https://doi.org/10.3390/foods9101360







