A Randomized, Double-Blind, Placebo Controlled Trial to Determine the Effectiveness a Polyphenolic Extract (Hibiscus sabdariffa and Lippia citriodora) in the Reduction of Body Fat Mass in Healthy Subjects
Abstract
1. Introduction
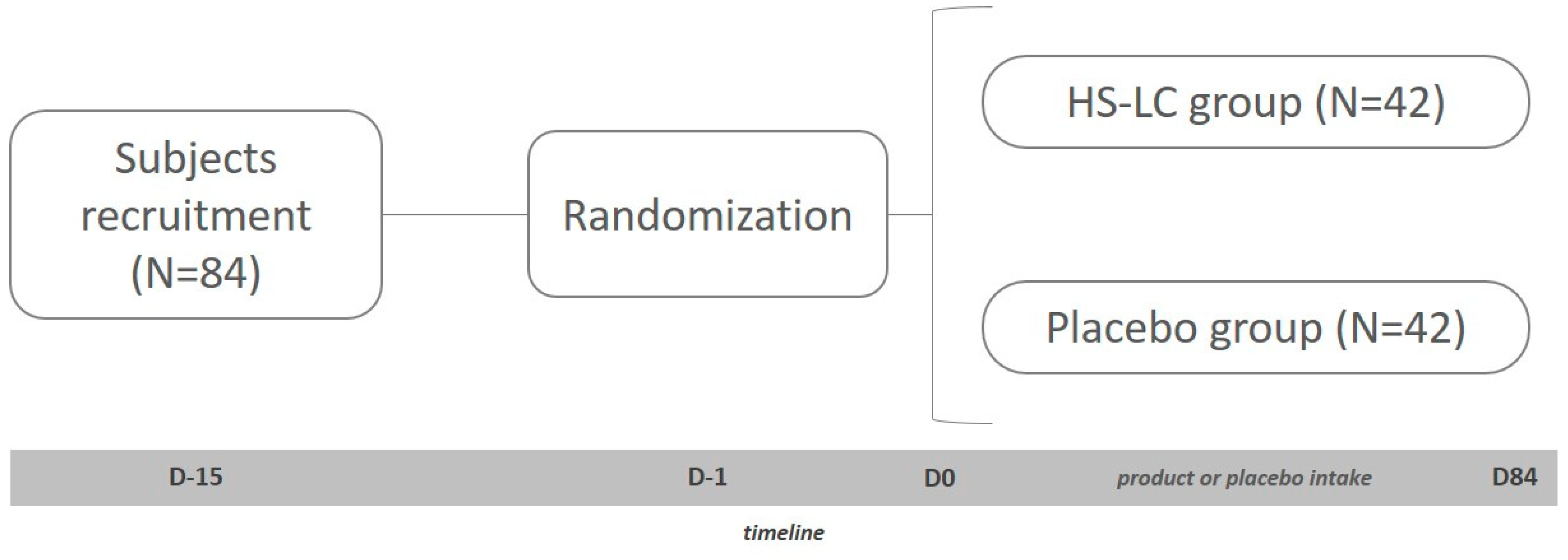
2. Material and Methods
3. Statistical Determinations
4. Results and Discussion
4.1. Blood Parameters
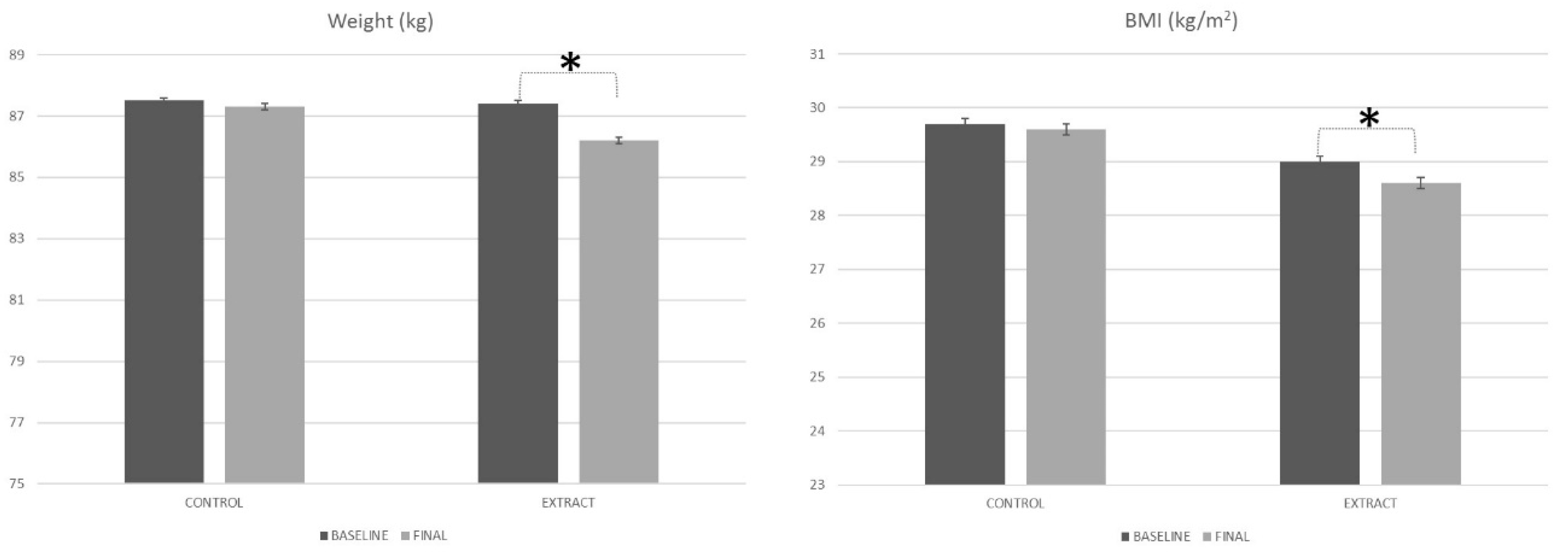
4.2. Weight and BMI
4.3. Body Composition
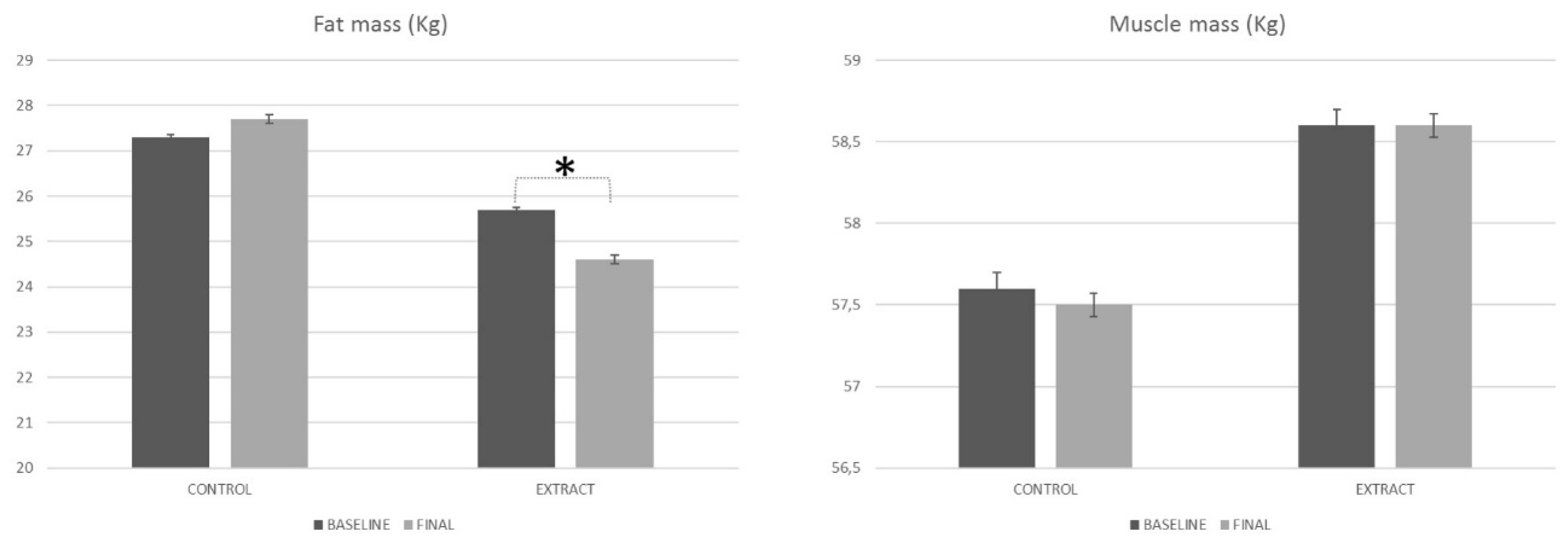
4.3.1. Bioimpedance
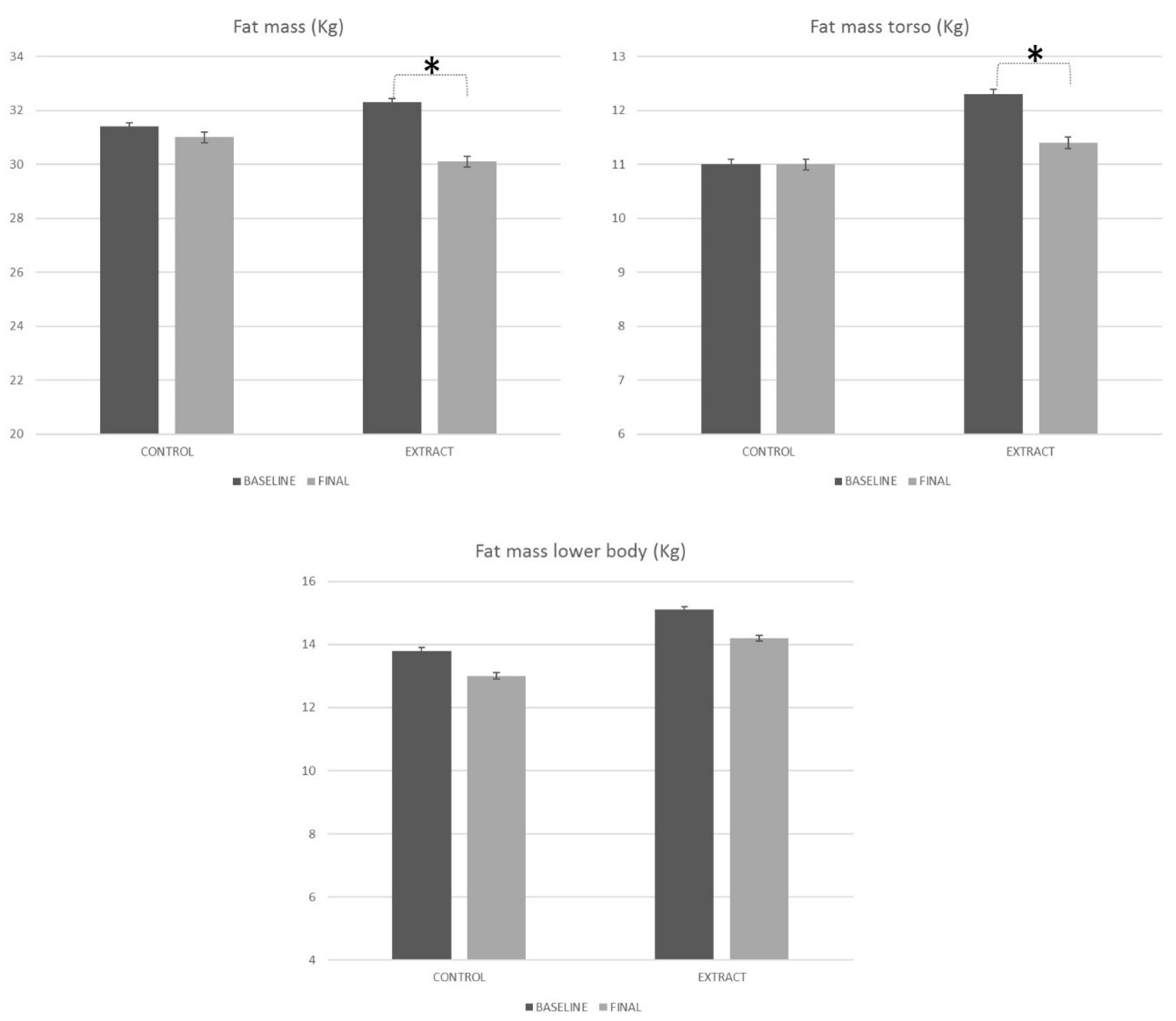
4.3.2. Densitometry
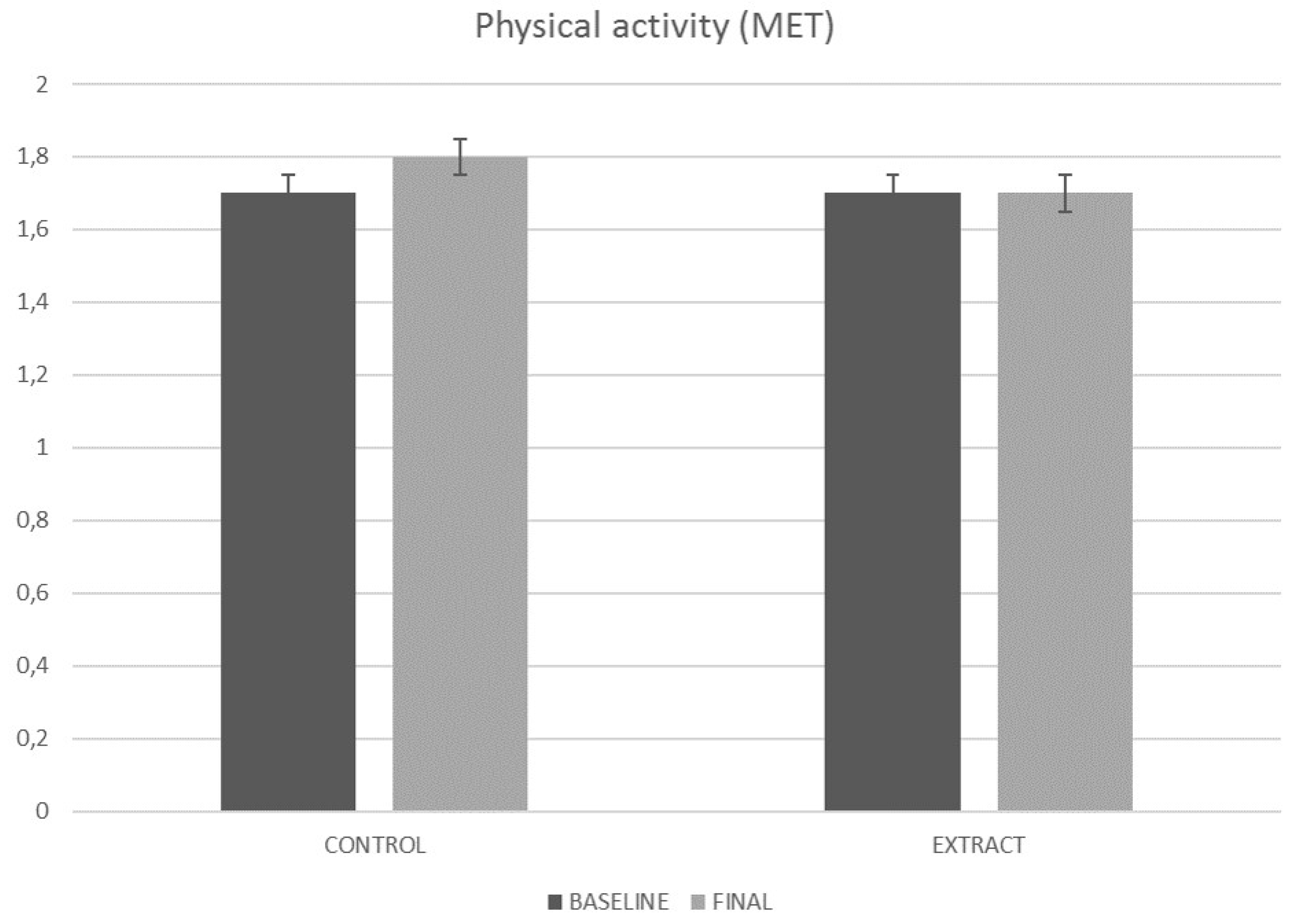
4.4. Physical Activity
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Díaz, M. Presente y futuro del tratamiento farmacológico de la obesidad. Present Future Pharmacol. Treat. Obes. 2005, 73, 137–144. [Google Scholar]
- Blancas-Flores, G.; Almanza-Pérez, J.C.; López-Roa, R.I.; Alarcón-Aguilar, F.J.; García-Macedo, R.; Cruz, M. La obesidad como un proceso inflamatorio. Boletín médico del Hospital Infantil de México 2010, 67, 88–97. [Google Scholar]
- Grundy, S.M. Metabolic Syndrome; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Alvídrez-Morales, A.; González-Martínez, B.E.; Jiménez-Salas, Z. Tendencias en la producción de alimentos: Alimentos funcionales. Available online: https://www.medigraphic.com/pdfs/revsalpubnut/spn-2002/spn023g.pdf (accessed on 30 November 2019).
- Anchia, I.A. Alimentos y Nutrición en La Práctica Sanitaria; Ediciones Díaz de Santos: Madrid, Spain, 2003. [Google Scholar]
- Serban, C.; Sahebkar, A.; Ursoniu, S.; Andrica, F.; Banach, M. Effect of sour tea (Hibiscus sabdariffa L.) on arterial hypertension: A systematic review and meta-analysis of randomized controlled trials. J. Hypertens. 2015, 33, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Marhuenda, J.; Medina, S.; Martínez-Hernández, P.; Arina, S.; Zafrilla, P.; Mulero, J.; Oger, C.; Galano, J.-M.; Durand, T.; Ferreres, F.; et al. Melatonin and hydroxytyrosol protect against oxidative stress related to the central nervous system after the ingestion of three types of wine by healthy volunteers. Food Funct. 2017, 8, 64–74. [Google Scholar] [CrossRef]
- Marhuenda, J.; Medina, S.; Martínez-Hernández, P.; Arina, S.; Zafrilla, P.; Mulero, J.; Oger, C.; Galano, J.-M.; Durand, T.; Solana, A. Effect of the dietary intake of melatonin-and hydroxytyrosol-rich wines by healthy female volunteers on the systemic lipidomic-related oxylipins. Food Funct. 2017, 8, 3745–3757. [Google Scholar] [CrossRef]
- Zaki, M.E.; Hala, T.; El-Gammal, M.; Kamal, S. Indicators of the metabolic syndrome in obese adolescents. Arch. Med. Sci. AMS 2015, 11, 92. [Google Scholar] [CrossRef]
- Min, S.Y.; Yang, H.; Seo, S.G.; Shin, S.H.; Chung, M.Y.; Kim, J.; Lee, S.J.; Lee, H.J.; Lee, K.W. Cocoa polyphenols suppress adipogenesis in vitro and obesity in vivo by targeting insulin receptor. Int. J. Obes. 2013, 37, 584. [Google Scholar] [CrossRef]
- Shimoda, H.; Tanaka, J.; Kikuchi, M.; Fukuda, T.; Ito, H.; Hatano, T.; Yoshida, T. Effect of polyphenol-rich extract from walnut on diet-induced hypertriglyceridemia in mice via enhancement of fatty acid oxidation in the liver. J. Agric. Food Chem. 2009, 57, 1786–1792. [Google Scholar] [CrossRef]
- Matsui, N.; Ito, R.; Nishimura, E.; Yoshikawa, M.; Kato, M.; Kamei, M.; Shibata, H.; Matsumoto, I.; Abe, K.; Hashizume, S. Ingested cocoa can prevent high-fat diet-induced obesity by regulating the expression of genes for fatty acid metabolism. Nutrient 2005, 21, 594–601. [Google Scholar] [CrossRef]
- Stohs, S.J.; Badmaev, V. A review of natural stimulant and non-stimulant thermogenic agents. Phytother. Res. 2016, 30, 732–740. [Google Scholar] [CrossRef]
- Gu, Y.; Hurst, W.J.; Stuart, D.A.; Lambert, J.D. Inhibition of key digestive enzymes by cocoa extracts and procyanidins. J. Agric. Food Chem. 2011, 59, 5305–5311. [Google Scholar] [CrossRef] [PubMed]
- Boix-Castejón, M.; Herranz-López, M.; Gago, A.P.; Olivares-Vicente, M.; Caturla, N.; Roche, E.; Micol, V. Hibiscus and lemon verbena polyphenols modulate appetite-related biomarkers in overweight subjects: A randomized controlled trial. Food Funct. 2018, 9, 3173–3184. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Yang, W.K.; Kim, H.; Min, B.; Caturla, N.; Jones, J.; Park, Y.-C.; Lee, Y.-C.; Kim, S.H. Metabolaid® combination of lemon verbena and hibiscus flower extract prevents high-fat diet-induced obesity through AMP-activated protein kinase activation. Nutrient 2018, 10, 1204. [Google Scholar] [CrossRef] [PubMed]
- Ahmad-Qasem, M.H.; Caánovas, J.; Barrajón-Catalán, E.; Carreres, J.E.; Micol, V.; García-Pérez, J.V. Influence of olive leaf processing on the bioaccessibility of bioactive polyphenols. J. Agric. Food Chem. 2014, 62, 6190–6198. [Google Scholar] [CrossRef]
- Herranz-López, M.; Fernández-Arroyo, S.; Pérez-Sanchez, A.; Barrajón-Catalán, E.; Beltrán-Debón, R.; Menéndez, J.A.; Alonso-Villaverde, C.; Segura-Carretero, A.; Joven, J.; Micol, V. Synergism of plant-derived polyphenols in adipogenesis: Perspectives and implications. Phytomed 2012, 19, 253–261. [Google Scholar]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett Jr, D.R.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. Compendium of physical activities: A second update of codes and MET values. Med. Sci. Sports Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef]
- Beltrán-Debón, R.; Alonso-Villaverde, C.; Aragones, G.; Rodriguez-Medina, I.; Rull, A.; Micol, V.; Segura-Carretero, A.; Fernandez-Gutierrez, A.; Camps, J.; Joven, J. The aqueous extract of Hibiscus sabdariffa calices modulates the production of monocyte chemoattractant protein-1 in humans. Phytomed 2010, 17, 186–191. [Google Scholar] [CrossRef]
- Joven, J.; March, I.; Espinel, E.; Fernández-Arroyo, S.; Rodríguez-Gallego, E.; Aragonès, G.; Beltran-Debon, R.; Alonso-Villaverde, C.; Rios, L. Hibiscus sabdariffa extract lowers blood pressure and improves endothelial function. Mol. Nutr. Food Res. 2014, 58, 1374–1378. [Google Scholar] [CrossRef]
- Joven, J.; Espinel, E.; Rull, A.; Aragonès, G.; Rodríguez-Gallego, E.; Camps, J.; Micol, V.; Herranz-Lopez, M.; Menendez, J.A.; Borras, I.; et al. Plant-derived polyphenols regulate expression of miRNA paralogs miR-103/107 and miR-122 and prevent diet-induced fatty liver disease in hyperlipidemic mice. Biochim. et Biophys. Acta (BBA)-Gen. Subj. 2012, 1820, 894–899. [Google Scholar] [CrossRef]
- Olivares-Vicente, M.; Barrajon-Catalan, E.; Herranz-Lopez, M.; Segura-Carretero, A.; Joven, J.; Encinar, J.A.; Micol, V. Plant-derived polyphenols in human health: Biological activity, metabolites and putative molecular targets. Curr. Drug Metab. 2018, 19, 351–369. [Google Scholar] [CrossRef]
- Herranz-López, M.; Barrajón-Catalán, E.; Segura-Carretero, A.; Menéndez, J.A.; Joven, J.; Micol, V. Lemon verbena (Lippia citriodora) polyphenols alleviate obesity-related disturbances in hypertrophic adipocytes through AMPK-dependent mechanisms. Phytomed 2015, 22, 605–614. [Google Scholar]
- Shirwany, N.A.; Zou, M.H. AMPK: A cellular metabolic and redox sensor: A mini review. Front. Biosci. 2014, 19, 447. [Google Scholar] [CrossRef] [PubMed]
- Nuttall, F.Q. Body mass index: Obesity, BMI, and health: A critical review. Nutr. Today 2015, 50, 117. [Google Scholar] [CrossRef] [PubMed]
- Barbosa-Silva, M.C.G.; Barros, A.J.; Post, C.L.; Waitzberg, D.L.; Heymsfield, S.B. Can bioelectrical impedance analysis identify malnutrition in preoperative nutrition assessment? Nutrient 2003, 19, 422–426. [Google Scholar] [CrossRef]
- Ellis, K.J.; Bell, S.J.; Chertow, G.M.; Chumlea, W.C.; Knox, T.A.; Kotler, D.P.; Lukaski, H.C.; Schoeller, D.A. Bioelectrical impedance methods in clinical research: A follow-up to the NIH Technology Assessment Conference. Nutrient 1999, 15, 874–880. [Google Scholar] [CrossRef]
- Alvero-Cruz, J.R.; Gómez, L.C.; Ronconi, M.; Vázquez, R.F.; i Manzañido, J.P. La bioimpedancia eléctrica como método de estimación de la composición corporal: Normas prácticas de utilización. Rev. Andal. Med. Deporte 2011, 4, 167–174. [Google Scholar]
- Blake, G.M.; Fogelman, I. The clinical role of dual energy X-ray absorptiometry. Mag. Andal. Med. Sport. 2009, 71, 406–414. [Google Scholar] [CrossRef]
- Meyer, N.L.; Sundgot-Borgen, J.; Lohman, T.G.; Ackland, T.R.; Stewart, A.D.; Maughan, R.J.; Smith, S.; Müller, W. Body composition for health and performance: A survey of body composition assessment practice carried out by the Ad Hoc Research Working Group on Body Composition, Health and Performance under the auspices of the IOC Medical Commission. Br J Sports Med. 2013, 47, 1044–1053. [Google Scholar] [CrossRef]
- Ackland, T.R.; Lohman, T.G.; Sundgot-Borgen, J.; Maughan, R.J.; Meyer, N.L.; Stewart, A.D.; & Müller, W. Current status of body composition assessment in sport. Sports Med. 2012, 42, 227–249. [Google Scholar] [CrossRef]
- Huang, T.T.K.; Johnson, M.S.; Figueroa-Colon, R.; Dwyer, J.H.; Goran, M.I. Growth of visceral fat, subcutaneous abdominal fat, and total body fat in children. Obes. Res. 2001, 9, 283–289. [Google Scholar] [CrossRef]
- Gutiérrez, S.A.G.; Orozco, G.E.M.; Rodríguez, E.M.; Vázquez, J.D.J.S.; Camacho, R.B. La grasa visceral y su importancia en obesidad. J. Endocrinol. Nutr. 2002, 10, 121–127. [Google Scholar]
- Ross, T.T.; Overton, J.D.; Houmard, K.F.; Kinsey, S.T. β-GPA treatment leads to elevated basal metabolic rate and enhanced hypoxic exercise tolerance in mice. Physiol. Rep. 2017, 5, e13192. [Google Scholar] [CrossRef] [PubMed]
- Thomson, D.M.; Porter, B.B.; Tall, J.H.; Kim, H.J.; Barrow, J.R.; Winder, W.W. Skeletal muscle and heart LKB1 deficiency causes decreased voluntary running and reduced muscle mitochondrial marker enzyme expression in mice. A. J. Physiol.-Endocrinol. Metab. 2007, 292, E196–E202. [Google Scholar] [CrossRef] [PubMed]
- Chaube, B.; Malvi, P.; Singh, S.V.; Mohammad, N.; Viollet, B.; Bhat, M.K. AMPK maintains energy homeostasis and survival in cancer cells via regulating p38/PGC-1α-mediated mitochondrial biogenesis. Cell Death Discov. 2015, 1, 15063. [Google Scholar] [CrossRef] [PubMed]
- Dal-Pan, A.; Blanc, S.; Aujard, F. Resveratrol suppresses body mass gain in a seasonal non-human primate model of obesity. BMC Physiol. 2010, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Weltman, A.; Levine, S.; Seip, R.L.; Tran, Z.V. Accurate assessment of body composition in obese females. Am. J. Clin. Nutr. 1988, 48, 1179–1183. [Google Scholar] [CrossRef]
- Sanz, J.M.M.; Otegui, A.U.; Mielgo-Ayuso, J. Necesidades energéticas, hídricas y nutricionales en el deporte. Eur. J. Hum. Mov. 2013, 30, 37–52. [Google Scholar]
| Baseline | Final | ||
|---|---|---|---|
| Total Cholesterol | Control | 228.2 ± 6.2 | 232.8 ± 9.5 |
| EXTRACT | 234.9 ± 6 | 223.2 ± 9 | |
| HDL | Control | 56.5 ± 2.5 | 58.9 ± 2.4 |
| Extract | 58.5 ± 2.1 | 60.6 ± 2.2 | |
| LDL | Control | 144.1 ± 6.3 | 144.17.6 |
| Extract | 134.1 ± 7.2 | 133.8 ± 7.5 | |
| Triglycerides | Control | 138.8 ± 12.1 | 134.1 ± 612.4 |
| Extract | 136 ± 612.4 | 133.1 ± 12.4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marhuenda, J.; Perez, S.; Victoria-Montesinos, D.; Abellán, M.S.; Caturla, N.; Jones, J.; López-Román, J. A Randomized, Double-Blind, Placebo Controlled Trial to Determine the Effectiveness a Polyphenolic Extract (Hibiscus sabdariffa and Lippia citriodora) in the Reduction of Body Fat Mass in Healthy Subjects. Foods 2020, 9, 55. https://doi.org/10.3390/foods9010055
Marhuenda J, Perez S, Victoria-Montesinos D, Abellán MS, Caturla N, Jones J, López-Román J. A Randomized, Double-Blind, Placebo Controlled Trial to Determine the Effectiveness a Polyphenolic Extract (Hibiscus sabdariffa and Lippia citriodora) in the Reduction of Body Fat Mass in Healthy Subjects. Foods. 2020; 9(1):55. https://doi.org/10.3390/foods9010055
Chicago/Turabian StyleMarhuenda, Javier, Silvia Perez, Desirée Victoria-Montesinos, María Salud Abellán, Nuria Caturla, Jonathan Jones, and Javier López-Román. 2020. "A Randomized, Double-Blind, Placebo Controlled Trial to Determine the Effectiveness a Polyphenolic Extract (Hibiscus sabdariffa and Lippia citriodora) in the Reduction of Body Fat Mass in Healthy Subjects" Foods 9, no. 1: 55. https://doi.org/10.3390/foods9010055
APA StyleMarhuenda, J., Perez, S., Victoria-Montesinos, D., Abellán, M. S., Caturla, N., Jones, J., & López-Román, J. (2020). A Randomized, Double-Blind, Placebo Controlled Trial to Determine the Effectiveness a Polyphenolic Extract (Hibiscus sabdariffa and Lippia citriodora) in the Reduction of Body Fat Mass in Healthy Subjects. Foods, 9(1), 55. https://doi.org/10.3390/foods9010055







