Lime (Citrus aurantifolia (Christm.) Swingle) Essential Oils: Volatile Compounds, Antioxidant Capacity, and Hypolipidemic Effect
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation and Reagents
2.2. Essential Oils Preparation and Analysis
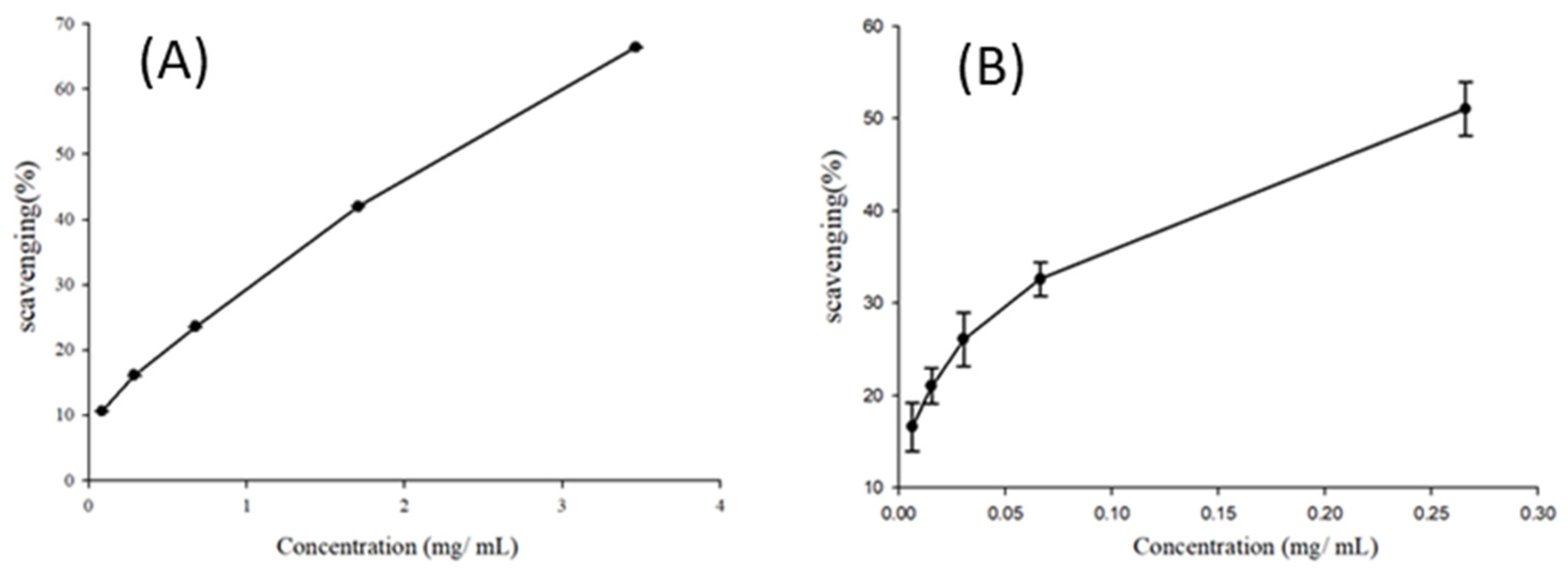
2.3. Antioxidant Capacity Assay
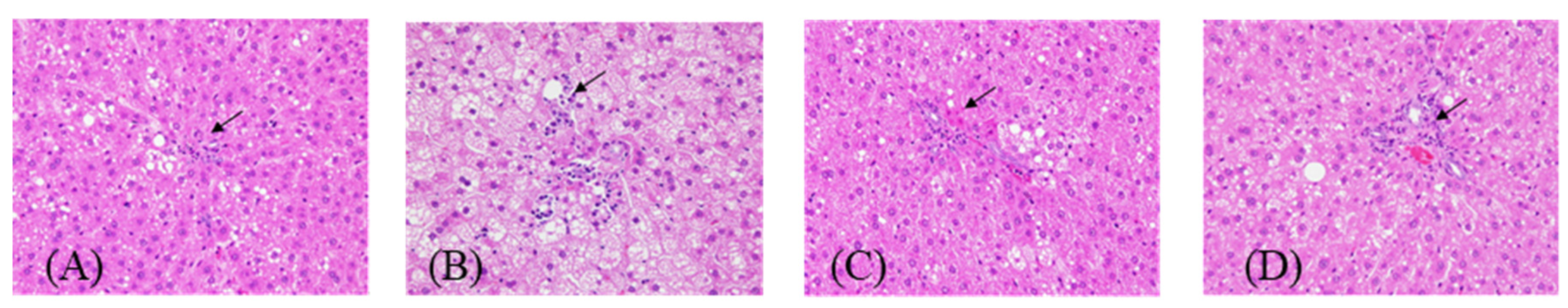
2.4. Animals and Treatments
2.5. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| EO | essential oil |
| LEO | lime essential oil |
| TG | triglyceride |
| TC | total cholesterol |
| LDL | low-density lipoprotein cholesterol |
| VLDL | very low-density lipoprotein cholesterol |
| HDL | high-density lipoprotein cholesterol |
| CVD | cardiovascular disease |
| DPPH | 1,1-diphenyl-2-picrylhydrazyl |
| ABTS | 2,2′-azinobis(3-ethylbenzothiazolin-6-sulfonic acid) |
| AI | atherogenic index |
| HTR | HDL-C/TC ratio |
| ACAT | cholesterol acyltransferase |
| ALT | aspartate aminotransferase |
| AST | alanine aminotransferase |
| GRAS | generally recognized as safe |
References
- Lv, X.; Zhao, S.; Ning, Z.; Zeng, H.; Shu, Y.; Tao, O.; Xiao, C.; Lu, C.; Liu, Y. Citrus fruits as a treasure trove of active natural metabolites that potentially provide benefits for human health. Chem. Cent. J. 2015, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Kelebek, H.; Selli, S. Determination of volatile, phenolic, organic acid and sugar components in a Turkish cv. Dortyol (Citrus sinensis L. Osbeck) orange juice. J. Sci. Food Agr. 2011, 91, 1855–1862. [Google Scholar] [CrossRef] [PubMed]
- Rafiq, S.; Kaul, R.; Sofi, S.A.; Bashir, N.; Nazir, F.; Nayik, G.A. Citrus peel as a source of functional ingredient: A review. J. Saudi Soc. Agric. Sci. 2018, 17, 351–358. [Google Scholar] [CrossRef]
- Pak, C.Y. Medical management of urinary stone disease. Nephron Clin. Pract. 2004, 98, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Dosoky, N.; Setzer, W. Biological activities and safety of Citrus spp. essential oils. Int. J. Mol. Sci. 2018, 19, 1966. [Google Scholar] [CrossRef] [PubMed]
- Jantan, I.; Ahmad, A.S.; Ahmad, A.R.; Ali, N.A.M.; Ayop, N. Chemical composition of some citrus oils from Malaysia. J. Essent. Oil Res. 1996, 8, 627–632. [Google Scholar] [CrossRef]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Alam, M.A.; Subhan, N.; Rahman, M.M.; Uddin, S.J.; Reza, H.M.; Sarker, S.D. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv. Nutr. 2014, 5, 404–417. [Google Scholar] [CrossRef]
- Fattore, M.; Montesano, D.; Pagano, E.; Teta, R.; Borrelli, F.; Mangoni, A.; Albrizio, S. Carotenoid and flavonoid profile and antioxidant activity in “Pomodorino Vesuviano” tomatoes. J. Food Compost. Anal. 2016, 53, 61–68. [Google Scholar] [CrossRef]
- González-Molina, E.; Domínguez-Perles, R.; Moreno, D.A.; García-Viguera, C. Natural bioactive compounds of Citrus limon for food and health. J. Pharm. Biomed. Anal. 2010, 51, 327–345. [Google Scholar] [CrossRef]
- Montesano, D.; Fallarino, F.; Cossignani, L.; Bosi, A.; Simonetti, M.S.; Puccetti, P.; Damiani, P. Innovative extraction procedure for obtaining high pure lycopene from tomato. Eur. Food Res. Technol. 2008, 226, 327. [Google Scholar] [CrossRef]
- Papadaki, A.; Johnson, L.; Toumpakari, Z.; England, C.; Rai, M.; Toms, S.; Penfold, C.; Zazpe, I.; Martínez-González, M.A.; Feder, G. Validation of the English version of the 14-item Mediterranean diet adherence screener of the PREDIMED study, in people at high cardiovascular risk in the UK. Nutrients 2018, 10, 138. [Google Scholar] [CrossRef] [PubMed]
- Keys, A. Coronary heart disease in seven countries. Nutrition 1997, 13, 249–253. [Google Scholar] [CrossRef]
- Lee, L.C.; Wei, L.; Huang, W.C.; Hsu, Y.J.; Chen, Y.M.; Huang, C.C. Hypolipidemic effect of tomato juice in hamsters in high cholesterol diet-induced hyperlipidemia. Nutrients 2015, 7, 10525–10537. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.H.; Yang, K.M.; Huang, T.C.; Wu, M.L. Traditional small-size citrus from Taiwan: Essential oils, bioactive compounds and antioxidant capacity. Medicines 2017, 4, 28. [Google Scholar] [CrossRef] [PubMed]
- Delgado, A.M.; Issaoui, M.; Chammem, N. Analysis of main and healthy phenolic compounds in foods. J AOAC Int. 2019, 102, 1356–1364. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.Y.; Peng, C.C.; Liang, Y.J.; Yeh, W.T.; Wang, H.E.; Yu, T.H.; Peng, R.Y. Alpinia zerumbet potentially elevates high-density lipoprotein cholesterol level in hamsters. J. Agr. Food Chem. 2008, 56, 4435–4443. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.H.; Huang, T.C. Volatile and nonvolatile constituents and antioxidant capacity of oleoresins in three Taiwan citrus varieties as determined by supercritical fluid extraction. Molecules 2016, 21, 1735. [Google Scholar] [CrossRef]
- Mehl, F.; Marti, G.; Boccard, J.; Debrus, B.; Merle, P.; Delort, E.; Wolfender, J.L. Differentiation of lemon essential oil based on volatile and non-volatile fractions with various analytical techniques: A metabolomic approach. Food Chem. 2014, 143, 325–335. [Google Scholar] [CrossRef]
- Sadgrove, N.; Jones, G. A contemporary introduction to essential oils: Chemistry, bioactivity and prospects for Australian agriculture. Agriculture 2015, 5, 48–102. [Google Scholar] [CrossRef]
- Tangpao, T.; Chung, H.H.; Sommano, S. Aromatic profiles of essential oils from five commonly used Thai basils. Foods 2018, 7, 175. [Google Scholar] [CrossRef] [PubMed]
- Md Othman, S.N.A.; Hassan, M.A.; Nahar, L.; Basar, N.; Jamil, S.; Sarker, S.D. Essential oils from the Malaysian citrus (Rutaceae) medicinal plants. Medicines 2016, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Torres-Alvarez, C.; Núñez González, A.; Rodríguez, J.; Castillo, S.; Leos-Rivas, C.; Báez-González, J.G. Chemical composition, antimicrobial, and antioxidant activities of orange essential oil and its concentrated oils. CyTa J. Food 2017, 15, 129–135. [Google Scholar] [CrossRef]
- Zhu, Z.; Li, S.; He, J.; Thirumdas, R.; Montesano, D.; Barba, F.J. Enzyme-assisted extraction of polyphenol from edible lotus (Nelumbo nucifera) rhizome knot: Ultra-filtration performance and HPLC-MS2 profile. Food Res. Int. 2018, 111, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Schaich, K.M.; Tian, X.; Xie, J. Hurdles and pitfalls in measuring antioxidant efficacy: A critical evaluation of ABTS, DPPH, and ORAC assays. J. Funct. Foods 2015, 14, 111–125. [Google Scholar] [CrossRef]
- Yang, C.; Chen, H.; Chen, H.; Zhong, B.; Luo, X.; Chun, J. Antioxidant and anticancer activities of essential oil from Gannan navel orange peel. Molecules 2017, 22, 1391. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Bok, S.H.; Jeon, S.M.; Park, Y.B.; Lee, S.J.; Jeong, T.S.; Choi, M.S. Effect of rutin and tannic acid supplements on cholesterol metabolism in rats1. Nutr. Res. 2002, 22, 283–295. [Google Scholar] [CrossRef]
- Shinnick, F.L.; Ink, S.L.; Marlett, J.A. Dose response to a dietary oat bran fraction in cholesterol-fed rats. J. Nutr. 1990, 120, 561–568. [Google Scholar] [CrossRef]
- Cheng, M.C.; Ker, Y.B.; Yu, T.H.; Lin, L.Y.; Peng, R.Y.; Peng, C.H. Chemical synthesis of 9 (Z)-octadecenamide and its hypolipidemic effect: A bioactive agent found in the essential oil of mountain celery seeds. J. Agr. Food Chem. 2010, 58, 1502–1508. [Google Scholar] [CrossRef]
- Peng, C.H.; Ker, Y.B.; Weng, C.F.; Peng, C.C.; Huang, C.N.; Lin, L.Y.; Peng, R.Y. Insulin secretagogue bioactivity of finger citron fruit (Citrus medica L. var. sarcodactylis Hort, Rutaceae). J. Agr. Food Chem. 2009, 57, 8812–8819. [Google Scholar] [CrossRef]
- Aldosari, S.; Awad, M.; Harrington, E.O.; Sellke, F.W.; Abid, M.R. Subcellular reactive oxygen species (ROS) in cardiovascular pathophysiology. Antioxidants 2018, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, T.A.; Miller, M.; Schaefer, E.J. Hypertriglyceridemia and cardiovascular risk reduction. Clin. Ther. 2007, 29, 763–777. [Google Scholar] [CrossRef] [PubMed]
- Misharina, T.A.; Samusenko, A.L. Antioxidant properties of essential oils from lemon, grapefruit, coriander, clove, and their mixtures. Appl. Biochem. Micro. 2008, 44, 438–442. [Google Scholar] [CrossRef]
- Ruberto, G.; Baratta, M.T. Antioxidant activity of selected essential oil components in two lipid model systems. Food Chem. 2000, 69, 167–174. [Google Scholar] [CrossRef]
- De Andrade, T.U.; Brasil, G.A.; Endringer, D.C.; Da Nóbrega, F.R.; De Sousa, D.P. Cardiovascular activity of the chemical constituents of essential oils. Molecules 2017, 22, 1539. [Google Scholar] [CrossRef] [PubMed]
- Tundis, R.; Loizzo, M.R.; Bonesi, M.; Menichini, F.; Mastellone, V.; Colica, C.; Menichini, F. Comparative study on the antioxidant capacity and cholinesterase inhibitory activity of Citrus aurantifolia Swingle, C. aurantium L., and C. bergamia Risso and Poit. peel essential oils. J. Food Sci. 2012, 77, 40–46. [Google Scholar] [CrossRef]
| Groups | CN | HF | LEO-N | LEO-H |
|---|---|---|---|---|
| Diet Content (%) | ||||
| Casein | 20 | 20 | 20 | 20 |
| d,l-methionine | 0.3 | 0.3 | 0.3 | 0.3 |
| Corn starch | 15 | 5 | 5 | 5 |
| Sucrose | 50 | 50 | 50 | 50 |
| Cellulose | 5 | 5 | 5 | 5 |
| Ain 76 Mineral | 3.5 | 3.5 | 3.5 | 3.5 |
| Ain 76 Vitamin | 1 | 1 | 1 | 1 |
| Choline bitartrate | 0.2 | 0.2 | 0.2 | 0.2 |
| Corn oil | 5 | 5 | 5 | 5 |
| Lard | - | 9.8 | 9.8 | 9.8 |
| Cholesterol | - | 0.2 | 0.2 | 0.2 |
| Total | 100 | 100 | 100 | 100 |
| Lime essential Oil | - | - | 0.744 | 2.232 |
| Compounds * | RIs # | Composition (%) |
|---|---|---|
| Monoterpenes | ||
| α-thujene | 925 | 1.01 ± 0.15 |
| α-pinene | 941 | 3.12 ± 0.26 |
| camphene | 950 | 0.14 ± 0.01 |
| sabinene | 967 | 2.12 ± 0.17 |
| β-pinene | 971 | 12.57 ± 0.8 |
| β-myrcene | 983 | 1.92 ± 0.03 |
| α-phellandrene | 996 | 0.13 ± 0.01 |
| α-terpinene | 1009 | 0.37 ± 0.02 |
| o-cymene | 1013 | 1.30 ± 0.21 |
| limonene | 1030 | 42.35 ± 1.89 |
| β-ocimene | 1040 | 0.28 ± 0.07 |
| γ-terpinene | 1051 | 15.44 ± 1.89 |
| α-terpinolene | 1094 | 1.12 ± 0.06 |
| Terpene alcohols | ||
| linalool | 1087 | 0.64 ± 0.03 |
| fenchol | 1107 | 0.04 ± 0.00 |
| 1-terpinenol | 1125 | 0.04 ± 0.00 |
| borneol | 1156 | 0.06 ± 0.04 |
| 4-terpineol | 1164 | 0.40 ± 0.57 |
| α-terpineol | 1174 | 1.58 ± 0.11 |
| nerol | 1203 | 0.85 ± 0.15 |
| geraniol | 1311 | 0.58 ± 0.07 |
| α-bisabolol | 1615 | 0.01 ± 0.01 |
| β-santalol | 1642 | 0.08 ± 0.06 |
| ledol | 1656 | 0.08 ± 0.06 |
| α-bisabolol | 1669 | 0.18 ± 0.02 |
| Terpene aldehydes | ||
| β-citronellal | 1131 | 0.05 ± 0.00 |
| α-citral | 1197 | 1.98 ± 0.27 |
| β-citral | 1233 | 1.76 ± 0.25 |
| Terpene ketones | ||
| camphor | 1118 | 0.01 ± 0.01 |
| Terpene esters | ||
| citronellyl acetate | 1328 | 0.05 ± 0.03 |
| neryl acetate | 1343 | 2.20 ± 0.23 |
| trans-geranyl acetate | 1357 | 0.60 ± 0.10 |
| Sesquiterpenes | ||
| δ-elemene | 1331 | 0.23 ± 0.01 |
| α-farnesene | 1373 | 0.41 ± 0.02 |
| β-elemene | 1382 | 0.31 ± 0.02 |
| trans-α-bergamotene | 1408 | 0.12 ± 0.01 |
| γ-elemene | 1427 | 0.05 ± 0.01 |
| β-caryophyllene | 1430 | 0.86 ± 0.21 |
| trans-α-bergamotene | 1432 | 1.44 ± 0.09 |
| trans-β-farnesene | 1442 | 0.16 ± 0.02 |
| α-humulene | 1448 | 0.11 ± 0.03 |
| β-santalene | 1454 | 0.07 ± 0.00 |
| γ-curcumene | 1470 | 0.05 ± 0.01 |
| germacrene-D | 1473 | 0.23 ± 0.02 |
| cis-α-bisabolene | 1489 | 0.18 ± 0.01 |
| β-selinene | 1492 | 0.02 ± 0.02 |
| β-bisabolene | 1499 | 2.00 ± 0.13 |
| cis-γ-bisabolene | 1505 | 0.06 ± 0.00 |
| trans-γ-bisabolene | 1520 | 0.02 ± 0.00 |
| trans-α-bisabolene | 1529 | 0.07 ± 0.00 |
| germacrene-B | 1554 | 0.10 ± 0.02 |
| α-santalene | 1598 | 0.02 ± 0.02 |
| Oxygen-containing aliphatics | ||
| p-menth-2-en-1-ol | 1108 | 0.05 ± 0.00 |
| camphene hydrate | 1136 | 0.01 ± 0.00 |
| isopulegone | 1159 | 0.02 ± 0.03 |
| decanal | 1181 | 0.17 ± 0.01 |
| bornyl acetate | 1269 | 0.02 ± 0.00 |
| undecanal | 1284 | 0.03 ± 0.01 |
| tetradecanal | 1388 | 0.12 ± 0.02 |
| dodecanal | 1395 | 0.01 ± 0.01 |
| Groups | CN | HF | LEO-N | LEO-H |
|---|---|---|---|---|
| Serum | ||||
| AST (U/L) | 87.0 ± 8.0 a | 104.7 ± 8.8 a | 75.6 ± 8.7 ab | 54.3 ± 6.0 b |
| ALT (U/L) | 32.1 ± 2.8 b | 51.3 ± 5.0 a | 32.5± 5.8 b | 28.0 ± 4.7 b |
| TC (mg/dL) | 40.2 ± 6.3 b | 49.9 ± 4.9 a | 36.6 ± 3.7 b | 35.5 ± 6.3 b |
| TG (mg/dL) | 59.6 ± 12.8 b | 72.6 ± 14.4 a | 50.8 ± 12.5 b | 43.8 ± 6.9 c |
| LDL (mg/dL) | 32.8 ± 3.4 a | 39.2 ± 1.4 b | 31.5 ± 2.9 a | 34.8 ± 3.9 a |
| HDL (mg/dL) | 10.7 ± 1.5 a | 9.8 ± 0.9 a | 10.4 ± 1.5 a | 11.1 ± 2.6 a |
| HTR (%) | 26.7 ± 0.5 a | 19.63 ± 0.5 b | 28.3 ± 1.2 a | 30.9 ± 1.8 a |
| AI | 2.7 ± 0.1 b | 4.1 ± 0.1 a | 2.5 ± 0.2 b | 2.2 ± 0.2 b |
| Liver | ||||
| TC (mg/dL) | 58.4 ± 1.2 b | 72.5 ± 2.8 a | 56.3 ± 2.6 b | 53.8 ± 3.1 b |
| TG (mg/dL) | 57.0 ± 2.3 b | 77.3 ± 1.5 a | 59.4 ± 1.8 b | 55.9 ± 2.2 b |
| Feces | ||||
| Neutral sterols (mg/g) | 4.0 ± 0.4 b | 7.6 ± 1.5 b | 8.8 ± 0.4 b | 10.0 ± 0.4 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, L.-Y.; Chuang, C.-H.; Chen, H.-C.; Yang, K.-M. Lime (Citrus aurantifolia (Christm.) Swingle) Essential Oils: Volatile Compounds, Antioxidant Capacity, and Hypolipidemic Effect. Foods 2019, 8, 398. https://doi.org/10.3390/foods8090398
Lin L-Y, Chuang C-H, Chen H-C, Yang K-M. Lime (Citrus aurantifolia (Christm.) Swingle) Essential Oils: Volatile Compounds, Antioxidant Capacity, and Hypolipidemic Effect. Foods. 2019; 8(9):398. https://doi.org/10.3390/foods8090398
Chicago/Turabian StyleLin, Li-Yun, Cheng-Hung Chuang, Hsin-Chun Chen, and Kai-Min Yang. 2019. "Lime (Citrus aurantifolia (Christm.) Swingle) Essential Oils: Volatile Compounds, Antioxidant Capacity, and Hypolipidemic Effect" Foods 8, no. 9: 398. https://doi.org/10.3390/foods8090398
APA StyleLin, L.-Y., Chuang, C.-H., Chen, H.-C., & Yang, K.-M. (2019). Lime (Citrus aurantifolia (Christm.) Swingle) Essential Oils: Volatile Compounds, Antioxidant Capacity, and Hypolipidemic Effect. Foods, 8(9), 398. https://doi.org/10.3390/foods8090398