Pure Polyphenols and Cranberry Juice High in Anthocyanins Increase Antioxidant Capacity in Animal Organs
Abstract
:1. Introduction
2. Materials and Methods
2.1. PP and CJ
2.2. Animal Studies (Protocols #3-06 and #8-0 Were Approved by the University of Scranton Animal Committee)
2.2.1. Organ Antioxidant Capacity (AC) Analysis
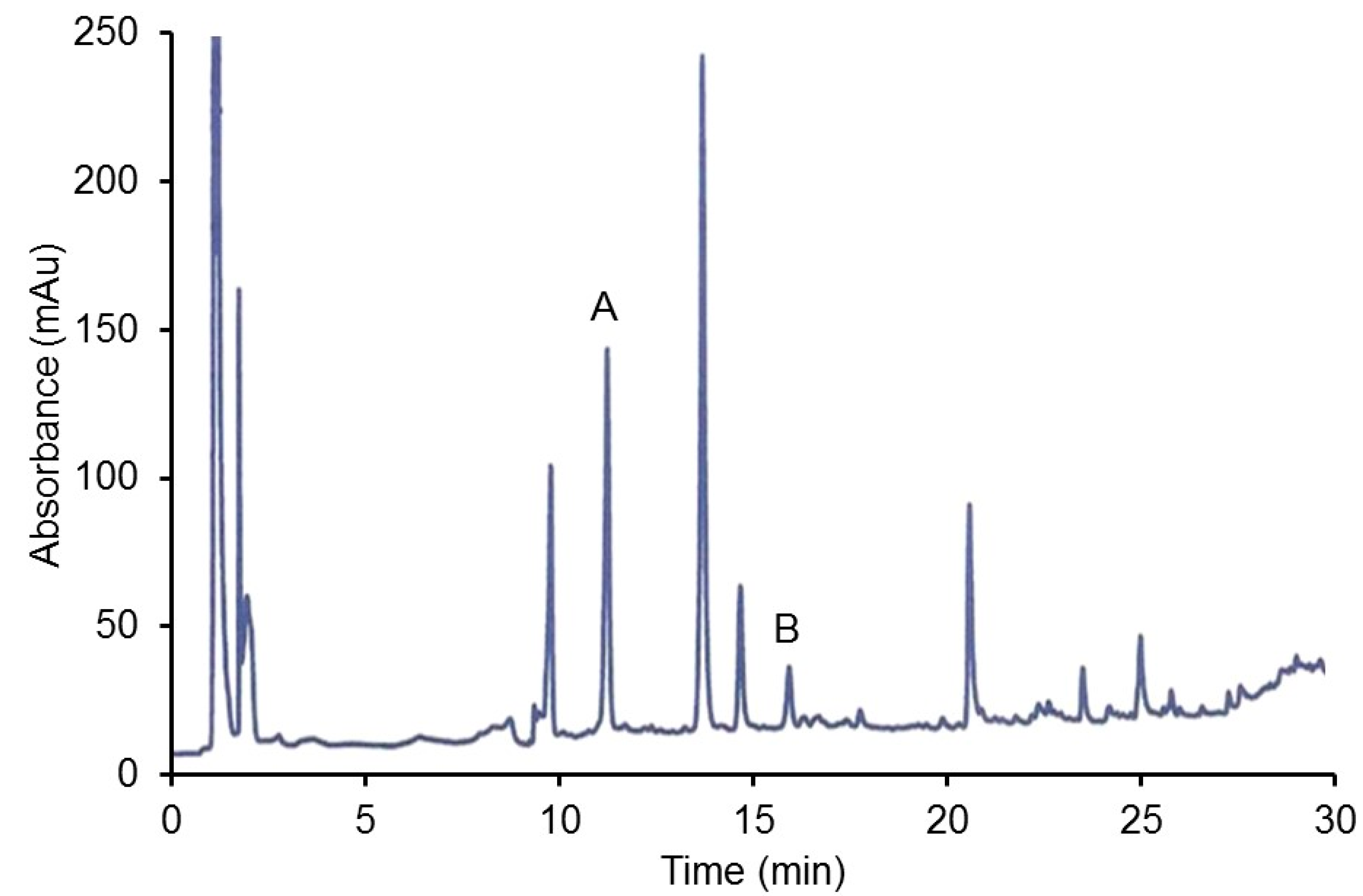
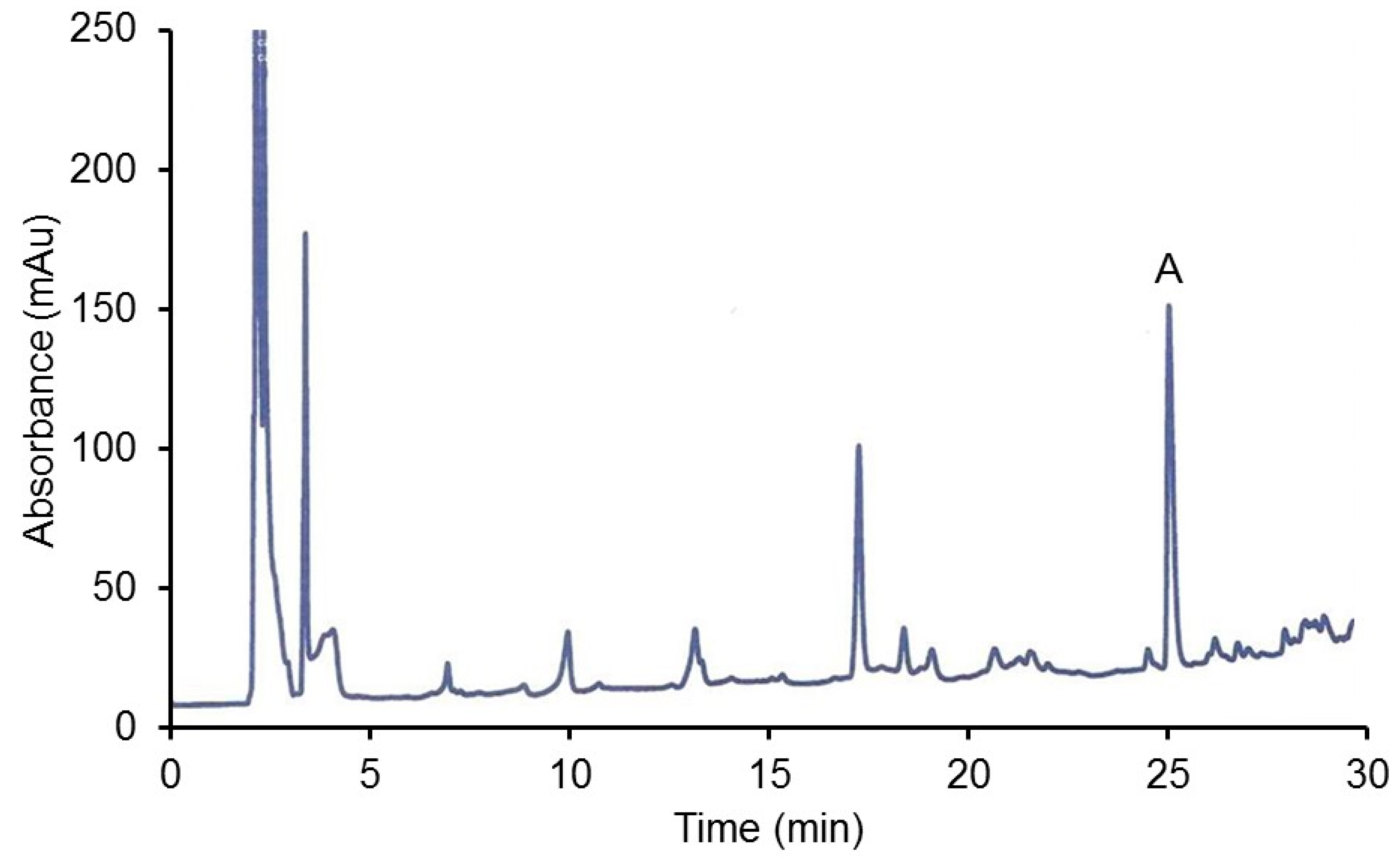
2.2.2. HPLC Analysis of Organ Extracts
2.3. Data Analysis
3. Results
3.1. Rat Study with Pure PP Compounds
3.1.1. Organ AC
3.1.2. HPLC of PP Metabolites
3.2. CJ Study
3.2.1. CJ Analysis
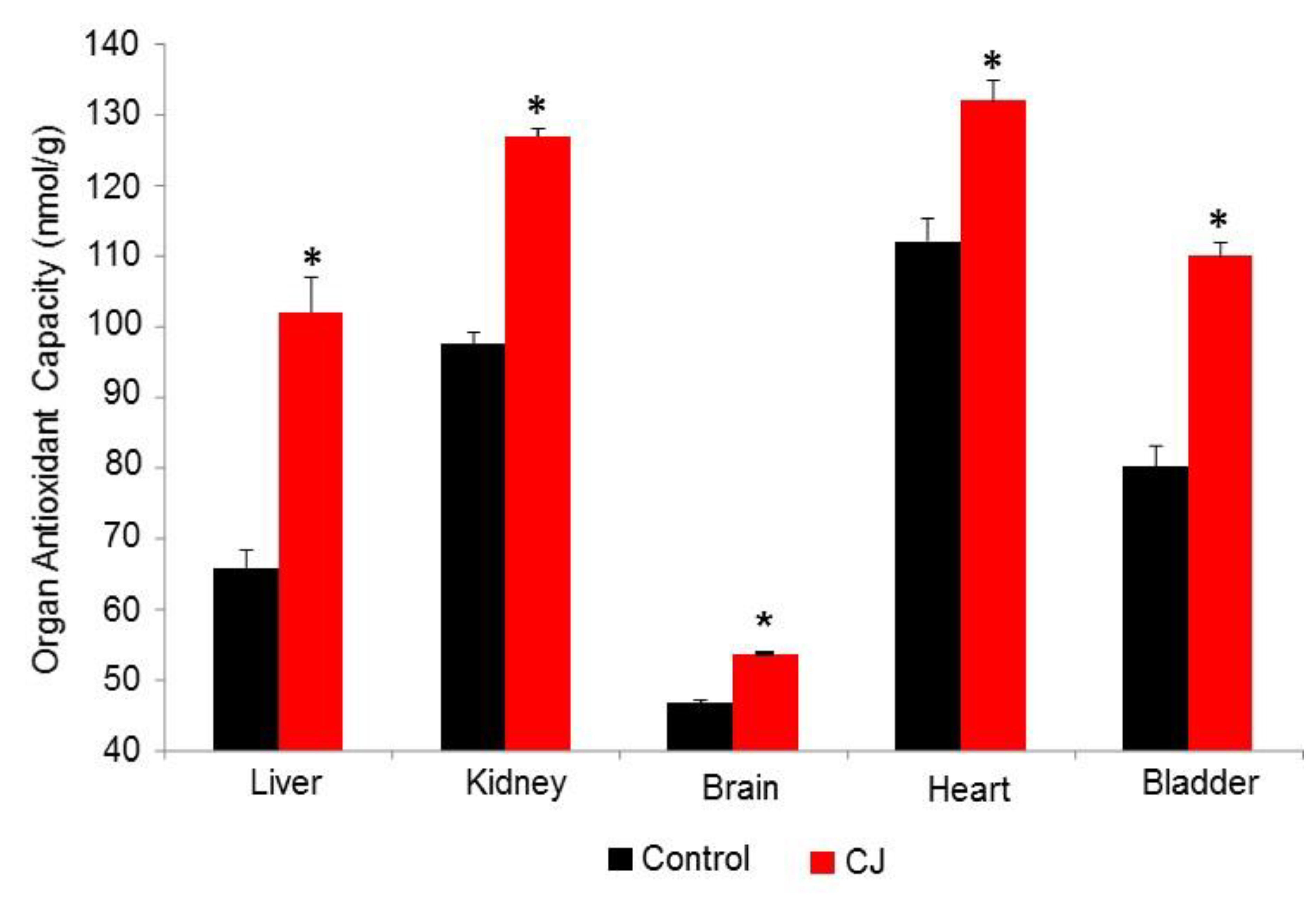
3.2.2. CJ Hamster Organs AC
3.2.3. HPLC Analysis of Hamster CJ Organs
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Redan, B.W.; Buhman, K.K.; Novotny, J.A.; Ferruzzi, M.G. Altered transport and metabolism of phenolic compounds in obesity and diabetes: Implications for functional food development and assessment. Adv. Nutr. 2016, 7, 1090–1104. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.A.; Su, X.; Zubik, L.; Bose, P.J. Phenol antioxidant quantity and quality in foods: Fruits. J. Agric. Food Chem. 2001, 49, 5315–5321. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.A.; Bose, P.; Proch, J.; Al Kharrat, H.; Samman, N.J. Cranberries and cranberry products: Powerful in vitro, ex vivo, and in vivo sources of antioxidants. J. Agric. Food Chem. 2008, 14, 5884–5891. [Google Scholar] [CrossRef]
- Tenore, G.C.; Daglia, M.; Ciampaglia, R.; Novellino, E. Exploring the nutraceutical potential of polyphenols from black, green and white tea infusions–An overview. Curr. Pharm. Biotechnol. 2015, 16, 265–271. [Google Scholar] [CrossRef]
- Scalbert, A.; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr. 2000, 130, 2073S–2085S. [Google Scholar] [CrossRef] [PubMed]
- Sebastian, R.S.; Wilkinson, E.; Goldman, J.D.; Martin, C.L.; Steinfeldt, L.C.; Murayi, T.; Moshfegh, A.J. New database facilitates characterization of flavonoid intake, sources, and positive associations with diet quality among US adults. J. Nutr. 2015, 145, 1239–1248. [Google Scholar] [CrossRef]
- Tresserra-Rimbau, A. Polyphenol intake and mortality risk: A re-analysis of the PREDIMED trial. BMC Med. 2014, 12, 12–77. [Google Scholar] [CrossRef]
- Billingsley, H.E.; Carbone, S. The antioxidant potential of the Mediterranean diet in patients at highcardiovascular risk: An in-depth review of the European PREDIMED trial. Nutr. Diabetes 2018, 8, 13. [Google Scholar] [CrossRef]
- Ivey, K.L.; Jensen, M.K.; Hodgson, J.M.; Eliassen, A.H.; Cassidy, A.; Rimm, E.B. Association of flavonoid-rich foods and flavonoids with risk of all-cause mortality. Br. J. Nutr. 2017, 117, 1470–1477. [Google Scholar] [CrossRef] [Green Version]
- Cassidy, A.; Mukamal, K.J.; Liu, L.; Franz, M.; Eliassen, A.H.; Rimm, E.B. High anthocyanin intake is associated with a reduced risk of myocardial infarction in young and middle-aged women. Circulation 2013, 127, 188–196. [Google Scholar] [CrossRef]
- Vinson, J.A.; Proch, J.; Bose, P. Determination of quantity and quality of polyphenol antioxidants in foods and beverages. Methods Enzymol. 2001, 335, 103–114. [Google Scholar]
- Gabriel, A.A.; Vinson, J.A.; Donnelly, P.E. Folin-Ciocalteau Reagent for Polyphenolic Assay. Int. J. Food Sci. Nutr. Diet. 2014, 147–156. [Google Scholar]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colorants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Koutelidakis, A.E.; Kizis, D.; Argyri, K.; Kyriakou, A.; Komaitis, M.; Kapsokefalou, M. The effect of iron and fat in a diet containing green tea extract (Camellia sinensis) on the antioxidant capacity of some organs and the mRNA expression of specific genes in mice. J. Med. Food. 2014, 11, 1232–1238. [Google Scholar] [CrossRef]
- Katalinic, V.; Modun, D.; Musics, L.; Boban, M. Gender differences in antioxidant capacity of rat tissues determined by 2,2′-azinobis (3-ethylbenzothiazoline 6-sulfonate; ABTS) and ferric reducing antioxidant power (FRAP) assays. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2005, 1, 47–52. [Google Scholar] [CrossRef]
- Bieger, J.; Cermak, R.; Blank, R.; de Boer, V.C.; Hollman, P.C.; Kamphues, J.; Wolffram, S. Tissue distribution of quercetin in pigs after long-term dietary supplementation. J. Nutr. 2008, 8, 1417–1420. [Google Scholar] [CrossRef]
- Xu, F.; Zhang, Y.Z.; Zang, X.Y.; Wang, D.; Shang, M.Y.; Wang, X.; Chui, D.H.; Cai, S.Q.; Liang, J. The profiling and identification of the metabolites of (+)-catechin and study on their distribution in rats by HPLC-DAD-ESI-IT-TOF-MS (n) technique. Biomed. Chromatogr. 2014, 28, 401–411. [Google Scholar]
- Haidari, F.; Keshavarz, S.A.; Rashidi, M.R.; Shahi, M.M. Orange juice and hesperetin supplementation to hyperuricemic rats alter oxidative stress markers and xanthine oxidoreductase activity. J. Clin. Biochem. Nutr. 2009, 45, 285–291. [Google Scholar] [CrossRef]
- Brown, P.N.; Shipley, P.R. Determination of anthocyanins in cranberry fruit and cranberry fruit products by high-performance liquid chromatography with ultraviolet detection: Single-laboratory validation. J. AOAC Int. 2011, 2, 459–466. [Google Scholar]
- Liu, H.; Garrett, T.J.; Tayyari, F.; Gu, L. Profiling the metabolome changes caused by cranberry procyanidins in plasma of female rats using (1) H NMR and UHPLC-Q-Orbitrap-HRMS global metabolomics approaches. Mol. Nutr. Food Res. 2015, 59, 2107–2118. [Google Scholar] [CrossRef]
- Feliciano, R.P.; Mills, C.E.; Istas, G.; Heiss, C.; Rodriguez-Mateos, A. Absorption, metabolism and excretion of cranberry (poly)phenols in humans: A dose response study and assessment of inter-individual variability. Nutrients 2017, 9, 268. [Google Scholar] [CrossRef]
- Felgines, C.; Texier, O.; Garcin, P.; Besson, C.; Lamaison, J.L.; Scalbert, A. Tissue distribution of anthocyanins in rats fed a blackberry anthocyanin-enriched diet. Mol. Nutr. Food Res. 2009, 53, 1098–1103. [Google Scholar] [CrossRef]
- Talavéra, S.; Felgines, C.; Texier, O.; Besson, C.; Gil-Izquierdo, A.; Lamaison, J.L.; Rémésy, C. Anthocyanin metabolism in rats and their distribution to digestive area, kidney, and brain. J. Agric. Food Chem. 2005, 53, 3902–3908. [Google Scholar] [CrossRef]
- Rashid, K.; Wachira, F.N.; Nyabuga, J.N.; Wanyonyi, B.; Murilla, G.; Isaac, A.O. Kenyan purple tea anthocyanins ability to cross the blood brain barrier and reinforce brain antioxidant capacity in mice. Nutr. Neurosci. 2014, 17, 178–185. [Google Scholar] [CrossRef]
- Padayatty, S.J.; Levine, M. Vitamin C. The known and the unknown and Goldilocks. Oral Dis. 2016, 22, 463–493. [Google Scholar] [CrossRef]
- Vinson, J.A.; Bose, P. Comparative bioavailability to humans of ascorbic acid alone or in a citrus extract. Am. J. Clin. Nutr. 1988, 48, 601–604. [Google Scholar] [CrossRef] [Green Version]
- Villarreal, A.; Stoecker, B.J.; Garcia, C.; Garcia, K.; Rios, R.; Gonzales, C.; Mandadi, K.; Faraji, B.; Patil, B.S.; Deyhim, F. Cranberry juice improved antioxidant status without affecting bone quality in orchidectomized male rats. Phytomedicine 2007, 12, 815–820. [Google Scholar] [CrossRef]
- Ruel, G.; Pomerlequ, S.; Couture, P.; Lamarche, B.; Coullard, C. Changes in plasma antioxidant capacity and oxidized low-density lipoprotein levels in men after short-term cranberry juice consumption. Metabolism 2005, 7, 856–861. [Google Scholar] [CrossRef]
- de Almeida Alvarenga, L.; Borges, N.A.; Moreira, L.S.G.; Resende Teixeira, K.T.; Carraro-Eduardo, J.C.; Dai, L.; Stenvinkel, P.; Lindholm, B.; Mafra, D. Cranberries–Potential benefits in patients with chronic kidney disease. Food Funct. 2019, 10, 3103–3112. [Google Scholar] [CrossRef]
- Vidlar, V.; Bouchal, J.; Vrbkova, J.; Kolar, Z.; Kral, M.; Kosina, P.; Vostalova, J. Cranberry Intervention in Patients with Prostate Cancer Prior to Radical Prostatectomy. Clinical, Pathological and Laboratory Findings. Ph.D. Thesis, The University Palacky, Olomouc, Czechia, November 2016. [Google Scholar]
- Weh, K.M.; Clarke, J.; Kresty, L.A. Cranberries and Cancer: An Update of Preclinical Studies Evaluating the Cancer Inhibitory Potential of Cranberry and Cranberry Derived Constituents. Antioxidants 2016, 3, 27. [Google Scholar] [CrossRef]
| Polyphenols | Liver | Kidney | Prostate | Brain | Heart |
|---|---|---|---|---|---|
| Control | 49.3 ± 3.0 | 75.2 ± 8.0 | 224 ± 77 | 18.4 ± 2.8 | 70.0 ± 11.1 |
| Quercetin | 80.1 ± 8.8 ** | 124 ± 9 *** | 255 ± 50 | 13.9 ± 3.9 | 82.3 ± 12.1 |
| Hesperetin | 82.4 ± 6.0 ** | 148 ± 55 ** | 282 ± 8 | 19.5 ± 5.5 | 80.8 ± 12.4 |
| Catechin | 89.2 ± 5.3 ** | 131 ± 6 *** | 193 ± 24 | 27.5 ± 8.0 * | 73.0 ± 6.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bariexca, T.; Ezdebski, J.; Redan, B.W.; Vinson, J. Pure Polyphenols and Cranberry Juice High in Anthocyanins Increase Antioxidant Capacity in Animal Organs. Foods 2019, 8, 340. https://doi.org/10.3390/foods8080340
Bariexca T, Ezdebski J, Redan BW, Vinson J. Pure Polyphenols and Cranberry Juice High in Anthocyanins Increase Antioxidant Capacity in Animal Organs. Foods. 2019; 8(8):340. https://doi.org/10.3390/foods8080340
Chicago/Turabian StyleBariexca, Tracy, Janice Ezdebski, Benjamin W. Redan, and Joe Vinson. 2019. "Pure Polyphenols and Cranberry Juice High in Anthocyanins Increase Antioxidant Capacity in Animal Organs" Foods 8, no. 8: 340. https://doi.org/10.3390/foods8080340
APA StyleBariexca, T., Ezdebski, J., Redan, B. W., & Vinson, J. (2019). Pure Polyphenols and Cranberry Juice High in Anthocyanins Increase Antioxidant Capacity in Animal Organs. Foods, 8(8), 340. https://doi.org/10.3390/foods8080340





